Abstract
Pituitary tumors are rare in chinchillas. This report describes the clinical, gross, histologic, and immunohistochemical characteristics of pituitary tumors in 4 chinchillas. The affected chinchillas were females between 4 and 18 years of age. Clinically, neurologic signs were most commonly reported and included depression, obtundation, seizure, head-pressing, ataxia, and possible blindness. Computed tomography scanning of 2 chinchillas revealed solitary intracranial extra-axial masses in the region of the pituitary gland. Two pituitary tumors were confined to the pars distalis; the other 2 invaded the brain. Based on their microscopic appearances and lack of distant metastases, all 4 tumors were diagnosed as pituitary adenomas. Immunohistochemically, all pituitary adenomas were weakly to strongly positive for growth hormone, most consistent with the diagnosis of somatotropic pituitary adenomas. To the authors’ knowledge, this is the first detailed report of the clinical, pathologic, and immunohistochemical features of pituitary tumors in chinchillas.
Keywords
Compared with other rodents, there is a low incidence of reported neoplasia in chinchillas.2,3,7,9–11,18–20 Although there is speculation that the paucity of reports is indicative of low occurrence of cancer in chinchillas, there has been a recent increase in the number of case reports in the literature, which suggests neoplasia may be more common than previously suspected.6,10,11 Of the reported neoplasia in chinchillas, pituitary tumors are thought to be rare. To the authors’ knowledge, there is a single report of a pituitary tumor in a chinchilla. 10 The case was reported as a pituitary adenoma in a castrated 10-year-old male chinchilla; however, additional details including clinical, pathologic, and hormonal features were not provided. 10 This study aims to add to the existing literature and characterize a series of pituitary tumors in chinchillas based on their clinical, gross, histologic, and immunohistochemical characteristics.
Between 2013 and 2021, 4 pituitary neoplasms were identified in chinchillas from 3 diagnostic pathology services (a private diagnostic laboratory in Washington, USA [cases 1 and 2]; University of Wisconsin–Madison, Madison, Wisconsin, USA [case 3]; and City University of Hong Kong Veterinary Diagnostic Laboratory, Hong Kong SAR, China [case 4]). The 4 affected chinchillas in this study were females between 4 and 18 years of age. Three chinchillas presented with neurologic signs including seizure-like activity (case 1); depression and obtundation (case 2); and head-pressing, ataxia, and possible blindness (case 3). The chinchilla in case 4 initially collapsed and was comatose; however, seizure activity and circling were observed later in the hospital. In case 4, the presenting clinical signs were attributed to hypoglycemia (1.3 mmol/L; reference range = 6.1–10.7).
Computed tomography (CT) scanning of 2 chinchillas (cases 3 and 4) revealed solitary, intracranial, extra-axial masses in the region of the pituitary gland. In case 3, there was evidence of obstructive hydrocephalus. One chinchilla was found dead (case 2), 1 chinchilla died naturally after CT scanning (case 3), and 2 chinchillas were humanely euthanized (cases 1 and 4).
On gross examination of the brain in case 4, the pituitary gland was enlarged, measured 8 × 5 × 7 mm, and compressed the overlying brain. In case 3, it was difficult to identify the origin of the neoplasm grossly. The mass extended from the mid-diencephalon to the metencephalon and compressed and obscured several structures in the brain, including the hypothalamus, thalamus, hippocampus, caudal colliculus, and pons, as well as the third ventricle and mesencephalic aqueduct. The mass measured 8 × 20 × 14 mm, was poorly demarcated, gray to tan, and friable (Fig. 1a). Gross descriptions of the neoplasms from cases 1 and 2 were unavailable.
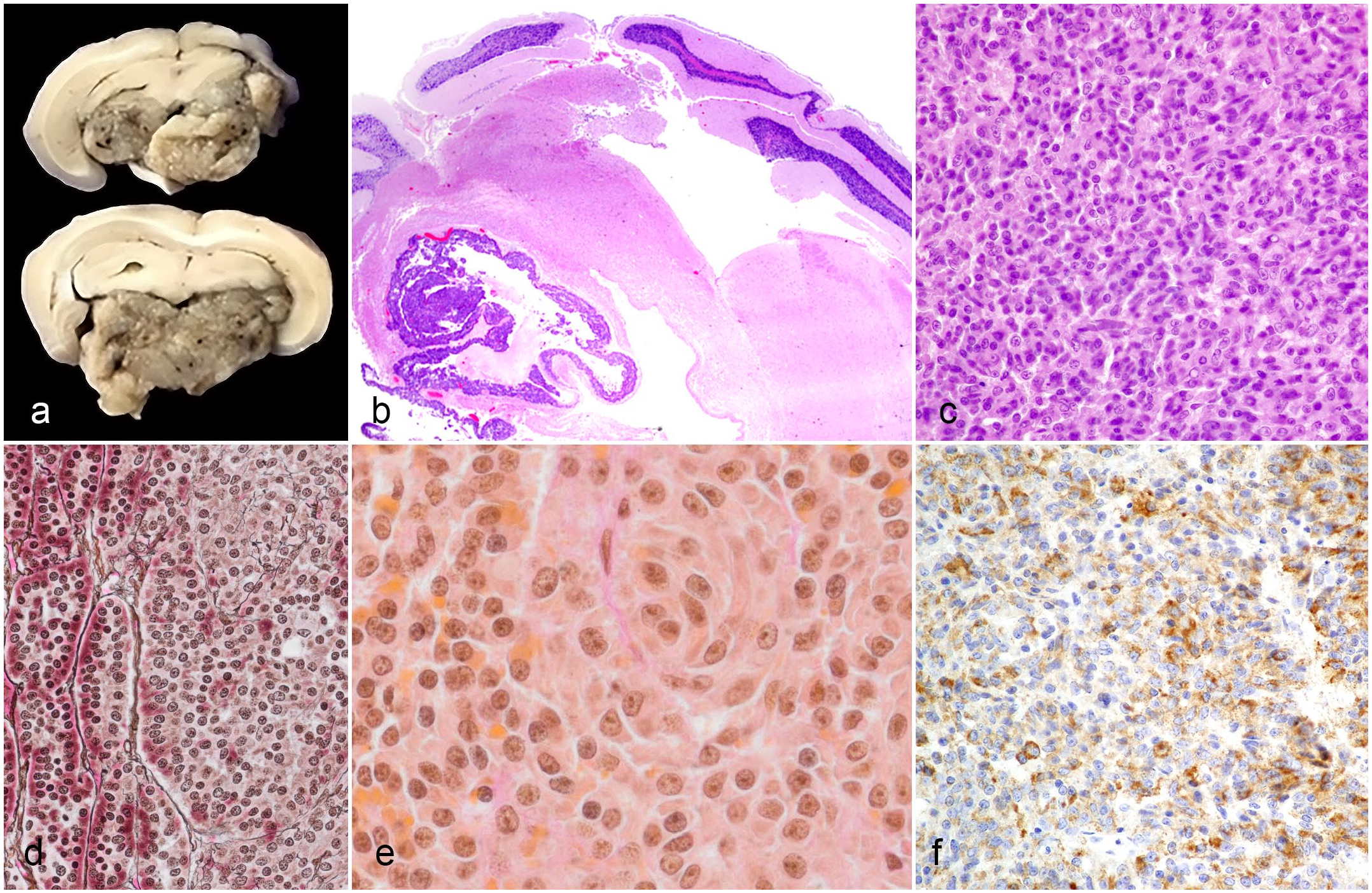
Gross, histopathologic, and immunohistochemical features of pituitary adenomas in the pituitary glands of chinchillas. (a) Transverse sections of formalin-fixed brain and pituitary gland. The pituitary gland and surrounding structures in the brain are compressed and obscured by a poorly demarcated, gray to tan, friable neoplasm. Case 3. (b) The pituitary adenoma infiltrates the leptomeninges and brain. Case 3, hematoxylin and eosin (HE). (c) Granulation is not seen in most neoplastic cells. Case 2, HE. (d) The pituitary adenoma exhibits gradual loss and disorganization of reticulin fibers. Case 3, Gomori’s reticulin. (e) Some neoplastic cells exhibit sparse to absent orange-G-positive staining that is typical of growth hormone cells. Case 3, Periodic acid-Schiff/orange G. (f) Neoplastic cells exhibit labeling for growth hormone. Case 2, immunohistochemistry for growth hormone.
Specimens were fixed in 10% neutral-buffered formalin, routinely processed and paraffin-embedded, sectioned at a thickness of 5 μm, and stained with hematoxylin and eosin. Sections from case 3 were also stained with Gomori’s reticulin and periodic acid-Schiff (PAS)/orange G stains.
Immunohistochemistry was performed on sections of all tumors for adrenocorticotropic hormone (ACTH), follicle-stimulating hormone (FSH), growth hormone (GH), luteinizing hormone (LH), melanocyte-stimulating hormone (MSH), and thyroid-stimulating hormone (TSH) following previously published protocols. 8 Protocols and antibodies used in this study have been previously successfully demonstrated to label various cell types in a variety of mammalian and avian species, including other rodents, and there was labeling for each antibody within normal pituitary tissue from a chinchilla that followed the expected pattern.4,5,8,12,17 Immunohistochemistry for prolactin was omitted from this study because the authors were unable to validate an antibody in chinchillas.
Histologically, the neoplasms ranged from multiple microadenomas (case 1) to a large solitary mass (macroadenoma) that effaced the pituitary gland (cases 2, 3, and 4). In cases 2 and 3, the macroadenomas infiltrated the leptomeninges and contiguous brain (Fig. 1b) in the absence of metastases. Morphologic features of the adenomas varied. In the case with multiple microadenomas (case 1), there were 3 small, expansile nodules that minimally compressed the surrounding tissue. In the macroadenomas (cases 2, 3, and 4), the neoplasms ranged from poorly demarcated to well-demarcated nodules and were unencapsulated. In both microadenomas and macroadenomas, neoplastic cells were arranged in palisades, trabeculae, and nests supported by a scant to fine fibrovascular stroma. Neoplastic cells were cuboidal to polygonal to elongate with variably distinct cell borders and a small to moderate amount of eosinophilic cytoplasm. Nuclei were round to oval with finely clumped to condensed chromatin and contained a variably distinct magenta nucleolus. Variably throughout the neoplasms in 3 cases (cases 1, 3, and 4), approximately 20% to 60% of cells contained intracytoplasmic hypereosinophilic granules, suggestive of acidophils, that were not evident in case 2 (Fig. 1c). Anisocytosis and anisokaryosis were mild to moderate, and mitoses were rare with less than 1 per 10 high-power fields (2.37 mm2). In addition, in the pituitary gland of case 4, there were multiple cystic spaces. In case 1, the chinchilla had a concurrent thymoma.
In case 3, the reticulin stain revealed progressive loss of normal architecture with disruption of the reticulin framework within the neoplasm (Fig. 1d). The PAS/orange G stain demonstrated positive staining of acidophils throughout the neoplasm, as well as sparse to absent staining in the cytoplasm of some atypical cells (Fig. 1e).
Immunohistochemically, cases 1, 3 and 4 had focal to multifocal, weak to moderate labeling for growth hormone. Case 2 had multifocal to diffuse, moderate to strong labeling for growth hormone (Fig. 1f). All cases were negative for ACTH, FSH, LH, MSH, and TSH. All antibodies labeled the appropriate cell types within normal pituitary tissue.
This case series documents the clinical, gross, histologic, and immunohistochemical characteristics of pituitary tumors in 4 chinchillas. Based on the expansile mass effect with loss of the normal reticulin fiber pattern (highlighted with reticulin stain in case 3), absence of metastases, and presence of growth hormone within both densely granulated and sparsely granulated neoplastic cells as assessed by immunohistochemical labeling, all 4 tumors were diagnosed as somatotroph pituitary adenomas.
Although somatotroph pituitary adenomas have been associated with a variety of conditions secondary to secretion of excess growth hormone including diabetes mellitus and acromegaly, particularly in cats, 15 there was no definitive evidence of these conditions in the chinchillas of this study, suggesting the tumors may have been nonfunctional. In these chinchillas, the clinical signs were neurologic and likely related to a mass effect resulting from increased intracranial pressure and/or displaced soft tissues of the brain. One chinchilla, case 4, initially presented for collapse and coma, which was later attributed to hypoglycemia; however, there was no definitive evidence of downregulation of insulin receptors and/or insulin resistance secondary to excess secretion of growth hormone.
For pituitary tumors, differentiating between nodular hyperplasia, adenoma, and carcinoma can be challenging, as these entities often present a continuous spectrum of proliferative lesions with carcinomas thought to arise from invasive, clinically aggressive adenomas.15,16 Although 2 chinchilla pituitary tumors were locally invasive and of remarkable size relative to the size of the chinchilla brain, current veterinary literature defines pituitary carcinomas solely by the presence of cerebrospinal and/or systemic metastasis; thus, both tumors were diagnosed as macroadenomas. 15 Recently, there was a major nomenclature change in the human literature from the classification of these tumors as “adenomas” to “pituitary neuroendocrine tumors” (PitNETs).1,13 This change attempts to reconcile the fact that pituitary adenomas can behave aggressively, such as infiltrating surrounding structures, and negatively impact the lives of patients.1,13
In the human literature, some subtypes of pituitary tumors have been designated as “high-risk PitNETs” due to aggressive clinical behavior and include sparsely granulated somatotroph tumors. 13 The distinction between densely and sparsely granulated somatotroph tumors is determined by the density of GH-containing secretory granules and the low-molecular-weight cytokeratin expression patterns, which is determined through immunohistochemical labeling for cytokeratin CAM5.2 or CK18, and/or evaluation of ultrastructural features via electron microscopy. 14 While neither labeling for cytokeratins nor electron microscopy was performed in these cases, 1 case (case 2) did not contain granules on H&E; however, this case also lacked recognizable fibrous bodies which are characteristic of low-molecular-weight cytokeratin. 14 The extent of invasion combined with the microscopic appearance of these tumors may suggest that chinchillas are predisposed to developing a particularly aggressive subtype of pituitary tumor.
There are several limitations of this study that may provide direction for future investigation of pituitary tumors in chinchillas. The authors did not feel that prevalence could be accurately reported due to inconsistencies in criteria for which pituitary glands were histologically evaluated at these facilities. At some of these facilities, histologic examination of the pituitary gland was only performed based on presenting clinical and/or gross neurologic abnormalities. Hyperplasia and microadenomas that were not clinically or grossly evident would therefore have been under-represented in this article. The prevalence would be better reported in a study in which there is consistent gross and histologic examination of pituitary glands of chinchillas.
In addition, in this study, prolactin secretion was not evaluated because the authors were unable to validate an antibody in chinchillas. In humans, mixed somatotroph-lactotroph PitNET/adenomas have been described. These neoplasms are composed of 2 distinct populations of tumor cells—those that secrete growth hormone and those that secrete prolactin. 1 Although the majority of both densely granulated and sparsely granulated neoplastic cells in these 4 chinchillas labeled for growth hormone with immunohistochemistry, in the absence of a validated prolactin antibody, a mixed tumor cannot be entirely ruled out.
Despite its limitations, this study provides insight into the clinical, pathologic, and immunohistochemical features of pituitary tumors in chinchillas. All chinchillas in this case series were female, which may suggest female predisposition; however, the single report documented prior to this study was male. 10 All cases were fatal, indicating a poor prognosis associated with pituitary adenomas in chinchillas. Pituitary tumors should be considered as a differential diagnosis for chinchillas presenting with neurological signs, and a combination of histology and immunohistochemistry is necessary for a definitive diagnosis of such pituitary neoplasms.
Footnotes
Acknowledgements
We thank Dr. Shahriar Salamat for his investigative assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
