Abstract
The development of lesions after infection with Mycobacterium avium subsp paratuberculosis (MAP) was examined in an experimental infection model. Goat kids were orally inoculated 10 times with 10 mg bacterial wet mass of MAP (total dose 2.6 × 108 colony-forming units). Six to 7 inoculated goats and 3 controls were autopsied 3, 6, 9, and 12 months postinoculation (mpi), lesions were documented, and samples were collected for histology, immunohistochemistry (IHC), and bacterial culture. Twenty-five of the 26 inoculated goats did not develop clinical signs. Macroscopic lesions were detected in 3 of the 7 inoculated goats as soon as 3 mpi. Jejunal Peyer’s patches (JPPs) were thickened and had ulcerated surfaces and circumscribed serositis. Characteristic granulomatous infiltrates were seen in all goats in gut-associated lymphoid tissues (GALTs), especially JPPs and lymphoid tissue at the ileocecal valve and in intestinal lymph nodes. Granulomatous intestinal infiltrates not associated with GALT were seen beginning at 6 mpi and with increasing frequency thereafter. Interindividual differences in lesions were most pronounced at 12 mpi, varying from mild focal paucibacillary to severe diffuse multibacillary patterns. Bacterial culture of MAP confirmed the IHC findings but was more sensitive and revealed widespread dissemination at 3 and 12 mpi. Granulomatous arteritis was found in intestinal submucosa of several goats. This may contribute to the spreading of MAP to the intestinal wall and possibly systemically. The different lesions observed during the clinically inapparent period of paratuberculosis are most likely indicators for the later progression of infection and development of clinical disease.
Keywords
Mycobacterium avium subsp paratuberculosis (MAP) is the cause of paratuberculosis, a chronic emaciating disease with characteristic lesions of granulomatous enteritis, lymphangitis, and lymphadenitis of regional lymph nodes. 5 Paratuberculosis is an important infectious disease of domestic and wild ruminants worldwide and causes severe economic losses. 28 A special feature of the disease is that infection of newborn and young animals is followed by a clinically inapparent period that may last for years before the disease becomes manifest. During this inapparent period, intermittent shedding of MAP occurs. 5,44 Thus, these apparently healthy animals are a source of infection and reinfection of herd mates and contaminate premises and pastures. They may also introduce MAP into paratuberculosis-free herds. Detection of infected cattle during the inapparent phase is unreliable using the currently available antemortem tests, since no tests with high specificity as well as high sensitivity are available. 27
Field studies in cattle, sheep, and goat herds with a history of paratuberculosis revealed a wide variation in severity and distribution of lesions (focal to multifocal to diffuse), inflammatory cell infiltrates (lymphocytes, multinucleated giant cells, epithelioid macrophages), and mycobacteria within lesions (paucibacillary, multibacillary) of animals both in the clinically inapparent and apparent phase of paratuberculosis. 7,14,30 Since the duration of infection is unknown in field infections, it remains unclear if these findings represent different stages of the disease or different host reactions and if there is a continuous progression or waxing and waning of lesions. A better understanding of host-pathogen interactions during the clinically inapparent interval should also help to improve and innovate diagnostic approaches.
The sequence of events in paratuberculosis can be studied only under experimental conditions. Goats were chosen for the experimental infection, because they are a natural host for MAP and develop paratuberculosis under natural conditions, and they have a shorter clinically inapparent interval than do cattle. 37,38 Experimental infections of goats revealed granulomatous intestinal lesions predominantly in the jejunal and ileal Peyer’s patches as early as 2 and 4 months postinoculation (mpi). 25,36 In one of the studies, no progression of lesions was found between 6 and 9 mpi and no lesions at 12 mpi. 36 In the other study, lesions were comparable at 2, 3, 4, 5, and 9 mpi and more severe at 6 and 7 mpi. 25 The changes during the first year after inoculation are, however, difficult to interpret, since in both studies, only 1 animal was examined per time. At 24 mpi, diffuse granulomatous paucibacillary infiltrates in jejunal Peyer’s patches and the patch at the ileocecal valve were most prevalent, which was interpreted as a general progression of lesions with age. 41
The objective of this investigation was to characterize the development of lesions and tissue distribution of MAP during the clinically inapparent interval of paratuberculosis. Groups of 6 to 7 goat kids were examined at each sequential time point to account for differences in individual disease progression, which are highly likely since immune reactions of the host have been suggested to have a major influence on the type of lesions. 31,32
Materials and Methods
Animals
Thirty-eight Thuringian Forest goats (34 males and 4 females) from a herd free of paratuberculosis were used. They were transferred at the age of 7 to 16 days to the animal facility at the Friedrich-Loeffler-Institut (FLI) in Jena. Fecal samples were collected from each kid upon arrival and cultivated for MAP. MAP was not detected in any sample. Goat kids were divided in groups of 6 to 7 animals with an even distribution based on body weight and sire. Infected and control animals were confined in separate air-conditioned rooms on deep straw bedding. They had free access to water and hay and received in addition milk replacer (Denkamilk capritop; Denkavit, Warendorf, Germany) until 10 weeks of life (12 kg body weight [BW]) and thereafter pellets. Male kids were castrated when they were 6 to 8 weeks old. All animals were treated against coccidia at 2 and 3 months of life (Baycox, 20 mg/kg BW, orally; Bayer, Leverkusen, Germany) and against endo- and ectoparasites at 4, 8, and 10 months of life (Dectomax, 3 mg/kg BW, intramuscularly [IM]; Pfizer, Berlin, Germany). They received vitamin B supplementation (B1-Hevert, 200 mg/animal, IM; Hevert, Nussbaum, Germany) at 4, 8, and 10 months of life. The animal experiment was approved by the animal ethical committee of Thuringia and the Thuringia Regional Office for Food Safety and Consumer Protection (Registration No. 04-001/11).
Study Design
Twenty-six goats (animal Nos. 1–26) kept in 4 groups were inoculated with MAP, and 12 goats (animal Nos. 27–38) served as sham-inoculated controls. Goats received 10 times every 2 to 3 days 10 mg bacterial wet mass (bwm) of MAP mixed into the milk replacer beginning when they were 11 to 20 days old. Sham-inoculated controls received pure milk replacer. MAP used for inoculation was a field isolate (JII-1961) from the ileocecal lymph node of a cow with paratuberculosis, characterized as nonpigmented type II strain. 3 Bacteria, which underwent altogether 5 laboratory passages before inoculation, were propagated in Middlebrook 7H9 bouillon (Becton Dickinson, Heidelberg, Germany) containing glycerine (Merck, Darmstadt, Germany), OADC (Becton Dickinson), and mycobactin J (Allied Monitor, Fayette, MO). After centrifugation, the bacterial pellet was recovered and bacterial wet mass determined. Acid-fast bacilli (AFB) were confirmed by Ziehl-Neelsen staining. MAP was confirmed by IS900 polymerase chain reaction (PCR) using primers according to Englund et al, 12 and freedom from contaminating bacteria was proven by inoculation on blood agar plates. The bwm was adjusted to 10 mg per dose using phosphate-buffered saline (PBS). The total inoculation dose per animal amounted to 2.6 × 108 colony-forming units (cfu).
After inoculation, goats were daily examined for clinical signs, and body weight was recorded every 4 weeks. Fecal samples were tested for MAP every 4 weeks, and blood and serum samples were collected for further immunological and serological testings (data not shown). At 3, 6, 9, and 12 months after the last application of inoculum (mpi), 6 to 7 goats inoculated with MAP and 3 sham-inoculated controls were necropsied. The time of necropsy was predetermined for each goat at the beginning of the experiment. At necropsy, macroscopic lesions were documented and samples collected for histology, immunohistochemistry, and cultivation of MAP.
Necropsy and Collection of Samples
To preserve the integrity of small intestinal mucosa, goats were sedated with Rompun 2% (0.25 mg/kg BW, IM; Bayer) and anesthesized with Ketamin 10% (2.5 mg/kg BW, intravenously [IV]; Intervet, Unterschleißheim, Germany) and Faustan (0.5 mg/kg BW, IV; AWD, Radebeul, Germany). The abdominal cavity was opened and segments of about 5 cm were ligated and filled with fixative at sites to be collected. Then, goats were euthanized with Release (20 ml, IV; WDT, Garbsen, Germany), and the intestine was cut at the duodenum and rectum, collected in toto, detached from the mesentery, and placed full length on a table. The following sites were collected: duodenum, 4 sites of the jejunum (2 m apart at 3 mpi, 2.5 m apart at 6 mpi, and 3 m apart at 9 and 12 mpi), at least 1 JPP from the proximal and distal jejunum, ileal Peyer’s patch (IPP) 90 cm proximal to the ileocecal valve (IPP proximal) and in the terminal ileum (IPP distal), cecum, organized lymphoid tissue in the colon next to the ileocecal valve (ICVPP), at the end of the proximal colon (PCPP) and in the rectum, central flexure of the colon, and descending colon. The remaining intestine was opened, and consistency, amount, and color of the ingesta were recorded. Then the ingesta was rinsed off and the mucosa examined. The intestinal lymph nodes were examined and samples collected from proximal and distal jejunal lymph nodes (J-LNs), ileocolic lymph nodes (ICV-LNs), and colonic lymph nodes (Co-LNs). Then a complete necropsy was performed and representative tissue samples were collected from tonsils, thymus, lung, heart, aorta, liver, hepatic lymph node (LN), spleen, pancreas, kidney, adrenal, rumen, abomasum, bone marrow, retropharyngeal LN, superficial cervical LN, and superficial inguinal LN.
Histology and Immunohistochemistry
Tissue samples were fixed in 4% neutral buffered formalin. Intestinal samples were opened at the mesenteric attachment and pinned flat on Styrofoam before immersion. Tissues were embedded in paraffin and lesions examined in hematoxylin and eosin (HE)–stained paraffin sections. Severity of lesions was graded from 1 to 3, with 1 (mild) representing small circumscribed lymphocytic or granulomatous infiltrates, no change of tissue architecture; 2 (moderate) representing granulomatous infiltrates with altered tissue architecture; and 3 (marked) indicating massive granulomatous infiltrates with partially or completely disrupted tissue architecture. Distribution of lesions was graded: focal (f), up to 3 distinct granulomatous infiltrates per section; multifocal (m), more than 3 granulomatous distinct infiltrates per section; and diffuse (d), infiltrates throughout the section.
Paraffin sections of all intestinal sites, intestinal lymph nodes, liver, tonsil, hepatic LN, superficial inguinal LN, and tissues with granulomatous lesions were examined for MAP by the indirect immunoperoxidase method. Sections were pretreated with trypsin (0.1%, 37°C, 20 minutes) for antigen retrieval. Polyclonal rabbit anti-MAP serum (Dako, Glostrup, Denmark) diluted 1:4000 in Dulbecco’s PBS (pH 7.4) containing 12.5% bovine serum albumin was used as primary antibody and peroxidase-conjugated goat anti–rabbit IgG as secondary antibody (Dianova, Hamburg, Germany). 3-Amino-9-ethyl carbazole (AEC; Sigma-Aldrich, Taufkirchen, Germany) was used as chromogen, because the red precipitate was distinct from brown-black pigments frequently encountered in tissues of the digestive tract. Sections were counterstained with hematoxylin for 90 seconds. As positive control, a slide from an experimentally inoculated goat in which mycobacteria had been detected by Ziehl Neelsen staining and from which MAP had been isolated by culture was included. As negative control, a consecutive section of the positive control was incubated with a polyclonal antiserum directed against nonrelated bacteria (Brachyspira hyodysenteriae) instead of the polyclonal rabbit anti-MAP serum. The number of mycobacteria per section was graded as none (<2 labeled bacteria per section), single (†), few (‡), and many (#), using a scoring system adapted from previous studies (Table 1). 6,10 Positive labeling was also seen in necrotic centers of granulomas (Table 1).
Scoring of Mycobacteria Detected by Immunohistochemistry in Tissue Sections.

To further characterize the granulomatous arteritis, serial paraffin sections were stained with Elastica–van-Gieson and Masson-Goldner-Trichrome stain. In addition, endothelial cells and subendothelial matrix of blood vessels were demonstrated by immunohistochemistry (IHC) for factor VIII (FVIII)–related antigen using a polyclonal rabbit anti-FVIII serum (Dako, Glostrup, Denmark) as primary antibody, diaminobenzidine as chromogen, and hematoxylin as counterstain.
Microbiological Examination
Fat and outer connective tissue were removed from LN and other tissue. Intestine was opened and ingesta removed. From each sampling site, 1 g of sample was collected from different locations, minced with scissors, and transferred into a plastic bag containing 7 ml 0.9% hexadecylpyridinium chloride (HPC) in distilled water. The samples were homogenized in a stomacher for 6 minutes, transferred to a 50-ml tube, and agitated on a shaker at 200 rpm for 10 minutes at room temperature. Afterward, they were incubated in an upright position for 24 hours at room temperature (RT) in the dark. After centrifugation at 1880 × g for 20 minutes at RT, supernatants were discarded and the pellets resuspended with 1 ml of sterile PBS (pH 7.2). Then, 200 μl of the pellet was seeded on each of 4 slopes of Herrold’s Egg Yolk Medium with Mycobactin J and amphotericin, nalidixic acid, and vancomycin (ANV, HEYM; Becton Dickinson). The cultures were incubated up to 6 months at 37°C and checked every 2 weeks for contamination and colonies. MAP was confirmed by IS900 PCR as described above. As soon as colonies became visible, colony counts were estimated semi-quantitatively by a score (CS) from 1 to 5, with 1 = 1–10, 2 = 11–20, 3 = 21–50, 4 = 50–100 individual colonies per slope, and 5 = bacterial lawn, and the week of appearance (WA) was recorded. The mean score of the 4 corresponding slopes was determined. For normalization of data, a growth index (GI) was calculated according to the following formula: GI = CS × 100/WA. Bacterial load was graded from 0 to ++++ with 0, no growth; +, GI >0, <25 (low); ++, GI ≥25, <50 (moderate); +++, GI ≥50, <75 (high); and ++++, GI ≥75 (very high).
Results
At the respective time of necropsy, 25 of the 26 goats inoculated with MAP were clinically healthy and in very good nutritional condition. One goat necropsied 12 mpi had undulating fever over several weeks and developed cachexia. At 3, 6, 9, and 12 mpi, lesions predominated at sites with gut-associated lymphoid tissue (GALT) throughout the digestive tract. JPPs and ICVPPs were most severely affected, whereas lesions were usually less severe and more seldom seen in other sites of GALT, especially in the IPP.
All control animals remained in good health during the observation period. One focal granulomatous infiltrate with eosinophils was seen in the PCPP of 1 goat at 6 mpi and was interpreted as a reaction to intestinal nematodes that had been detected in a few controls and MAP-inoculated goats at 3 and 6 mpi. MAP was neither detected by IHC nor cultured from any of the control animals during the entire observation period.
Three Months Postinoculation (7 Goats)
Intestinal Tract
In 3 goats, gross lesions were seen in JPPs of the proximal and mid-jejunum, which had diffuse or nodular thickening (Table 2). A circumscribed mild fibrous serositis characterized by firmly attached multiple delicate, filamentous strands of connective tissue was observed at the serosal aspect of altered JPPs. Severely affected JPPs had also often an ulcerated surface (Fig. 1). One of the 3 goats had ulcerations at the ICVPP and PCPP; another had multiple 2-mm nodules in the IPP.
Lesions and MAP in Gut-Associated Lymphoid Tissue of Goats Inoculated With MAP at 3, 6, 9, and 12 mpi: Gross Lesions, Severity,a and Distributionb of Microscopic Lesions, Detection of Mycobacteria by IHC,c and Bacterial Load.d
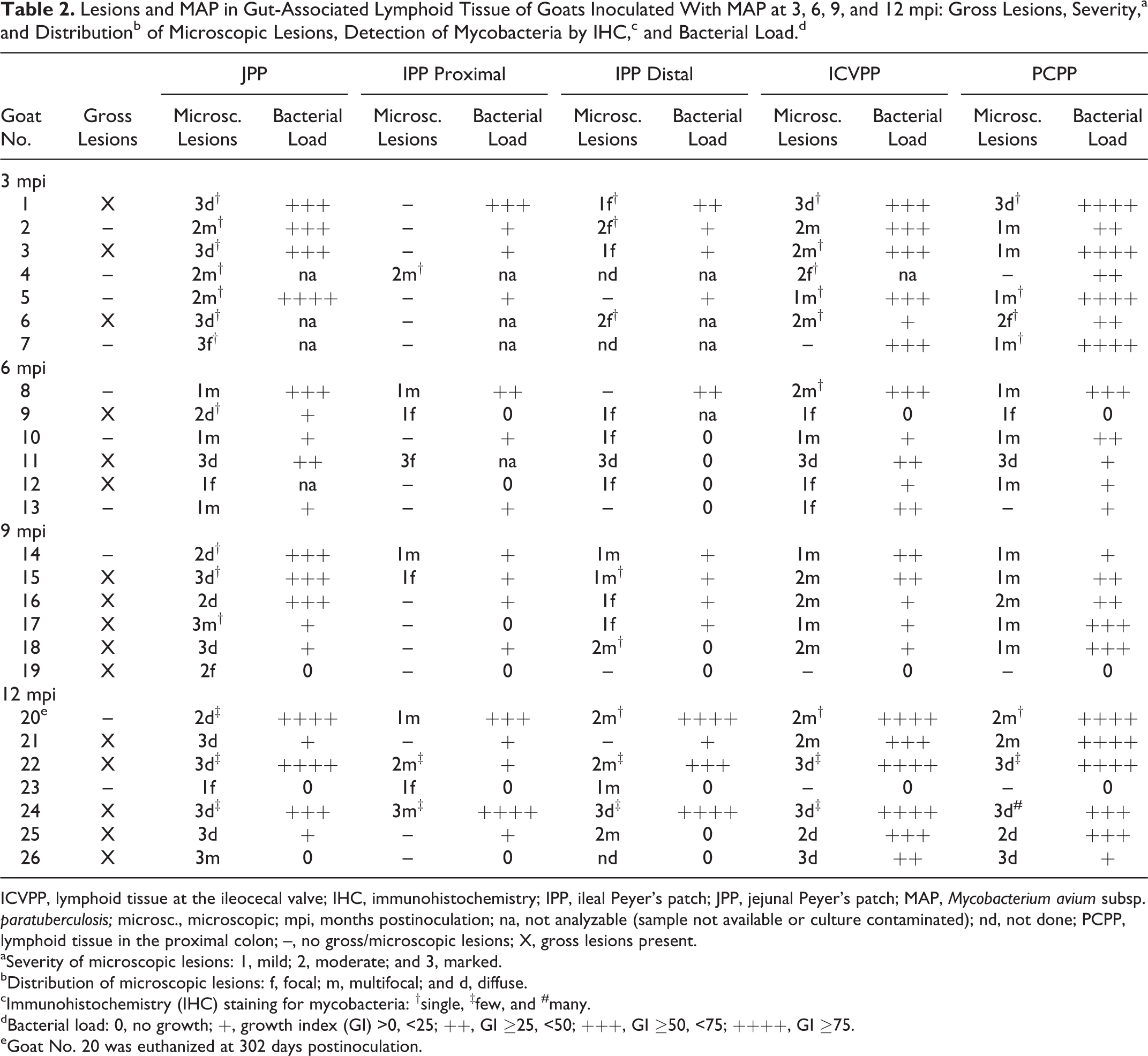
ICVPP, lymphoid tissue at the ileocecal valve; IHC, immunohistochemistry; IPP, ileal Peyer’s patch; JPP, jejunal Peyer’s patch; MAP, Mycobacterium avium subsp. paratuberculosis; microsc., microscopic; mpi, months postinoculation; na, not analyzable (sample not available or culture contaminated); nd, not done; PCPP, lymphoid tissue in the proximal colon; –, no gross/microscopic lesions; X, gross lesions present.
aSeverity of microscopic lesions: 1, mild; 2, moderate; and 3, marked.
bDistribution of microscopic lesions: f, focal; m, multifocal; and d, diffuse.
cImmunohistochemistry (IHC) staining for mycobacteria: †single, ‡few, and #many.
dBacterial load: 0, no growth; +, growth index (GI) >0, <25; ++, GI ≥25, <50; +++, GI ≥50, <75; ++++, GI ≥75.
eGoat No. 20 was euthanized at 302 days postinoculation.
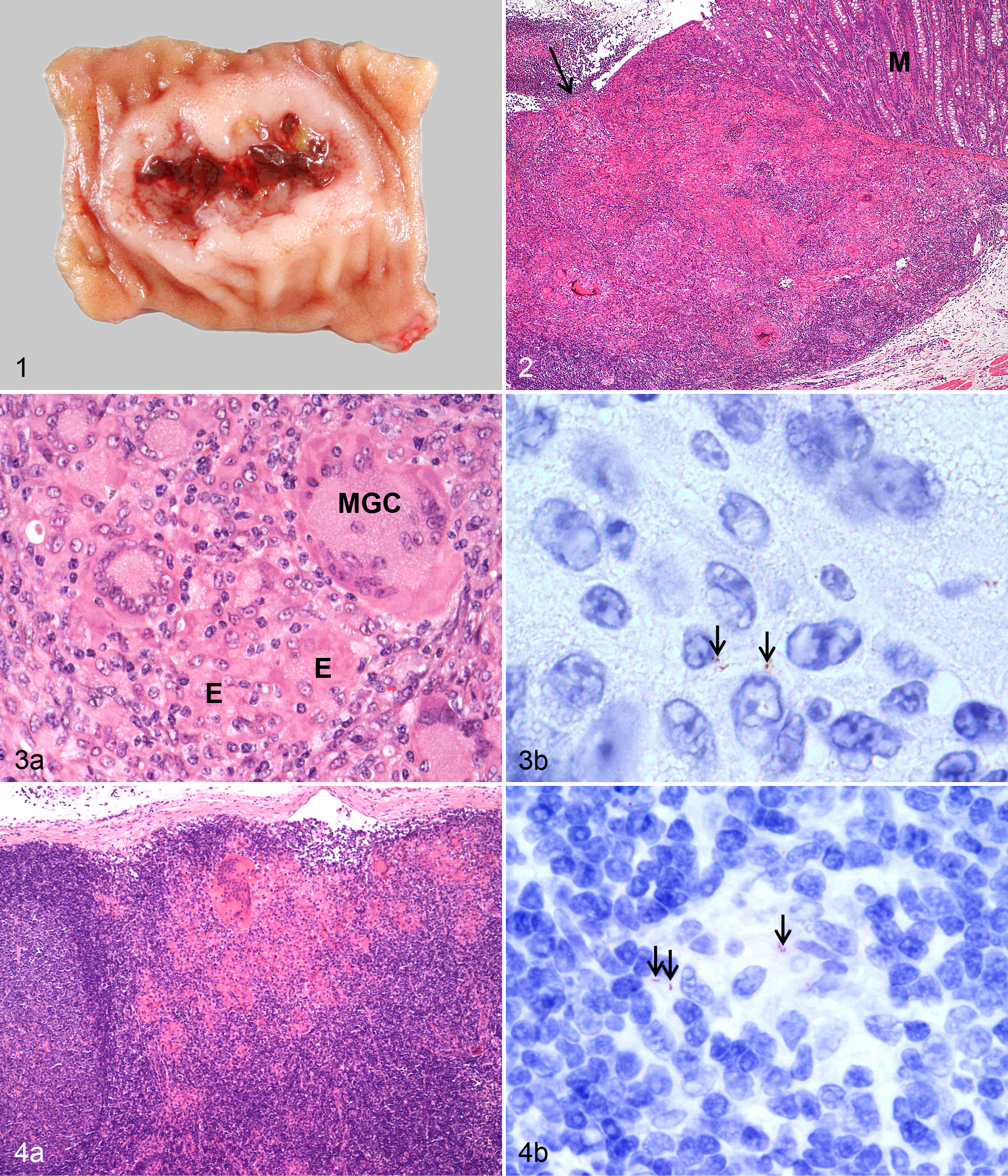
Histologic examination revealed granulomatous lesions in the intestines of all 7 goats. Lesions were restricted to GALT and adjacent mucosa in 5 goats (Fig. 2). Duodenal and cecal mucosa was focally affected in 1 goat each. In JPPs of 3 goats, multifocal to coalescing infiltrates of epithelioid cells, lymphocytes, and multinucleated giant cells (MGCs) were seen in descending frequency in interfollicular areas, domes, overlying lamina propria, and lymphoid follicles. In 4 goats, the architecture of JPPs was severely altered and partly to completely replaced by granulomatous infiltration with many and often degenerated MGCs, sometimes small foci of necrosis or calcification, neutrophils, and strands of connective tissue. Some lesions extended transmurally, resulting in mild lymphocytic and proliferative serositis. Lesions of comparable severity were observed at the ICVPP (Fig. 3a) and PCPP in 1 goat. Two goats had focally extensive lesions in the lymphoid tissue in the rectum. Mycobacteria were detected in low numbers in a few epithelioid cells and MGCs in all 7 goats by IHC (Table 2, Fig. 3b). This corresponded with a high to very high load of MAP in JPPs, ICVPPs, and PCPPs and a low to moderate load in the other intestinal sites (Tables 2 and 3).
Lesions and MAP in the Jejunum of Goats Inoculated With MAP at 3, 6, 9, and 12 mpi: Gross Lesions, Severity,a and Distributionb of Microscopic Lesions, Detection of Mycobacteria by IHC,c and Bacterial Load.d
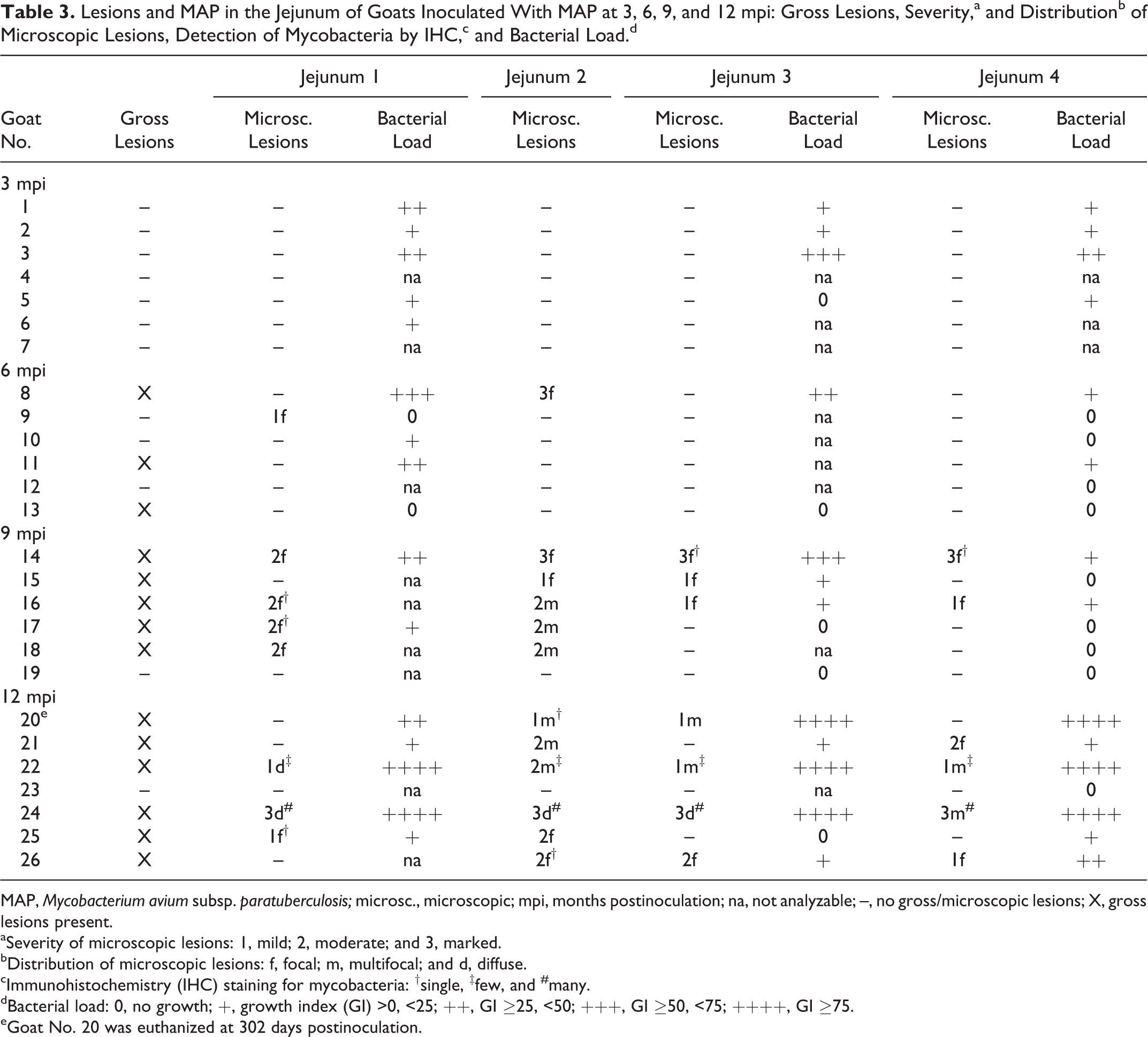
MAP, Mycobacterium avium subsp. paratuberculosis; microsc., microscopic; mpi, months postinoculation; na, not analyzable; –, no gross/microscopic lesions; X, gross lesions present.
aSeverity of microscopic lesions: 1, mild; 2, moderate; and 3, marked.
bDistribution of microscopic lesions: f, focal; m, multifocal; and d, diffuse.
cImmunohistochemistry (IHC) staining for mycobacteria: †single, ‡few, and #many.
dBacterial load: 0, no growth; +, growth index (GI) >0, <25; ++, GI ≥25, <50; +++, GI ≥50, <75; ++++, GI ≥75.
eGoat No. 20 was euthanized at 302 days postinoculation.
Lymph Nodes
J-LNs, ICV-LNs, and Co-LNs were of normal size and gross appearance, but histologic examination revealed focal to multifocal granulomatous infiltrates in the interfollicular areas close to the subcapsular sinus in 6 goats (Table 4, Fig. 4a). In 1 goat, lesions in the ICV-LN were confluent and extended deep into the paracortex. Mycobacteria were detected in low numbers in a few epithelioid cells and MGCs of 4 goats (Table 4, Fig. 4b). MAP was culturally isolated from all intestinal lymph nodes. High to very high bacterial load was detected in the ICV-LN of 5 goats and the proximal mesenteric LN of 3 goats (Table 4).
Lesions and MAP in Intestinal Lymph Nodes of Goats Inoculated With MAP at 3, 6, 9, and 12 mpi: Gross Lesions, Severity,a and Distributionb of Microscopic Lesions, Detection of Mycobacteria by IHC,c and Bacterial Load.d
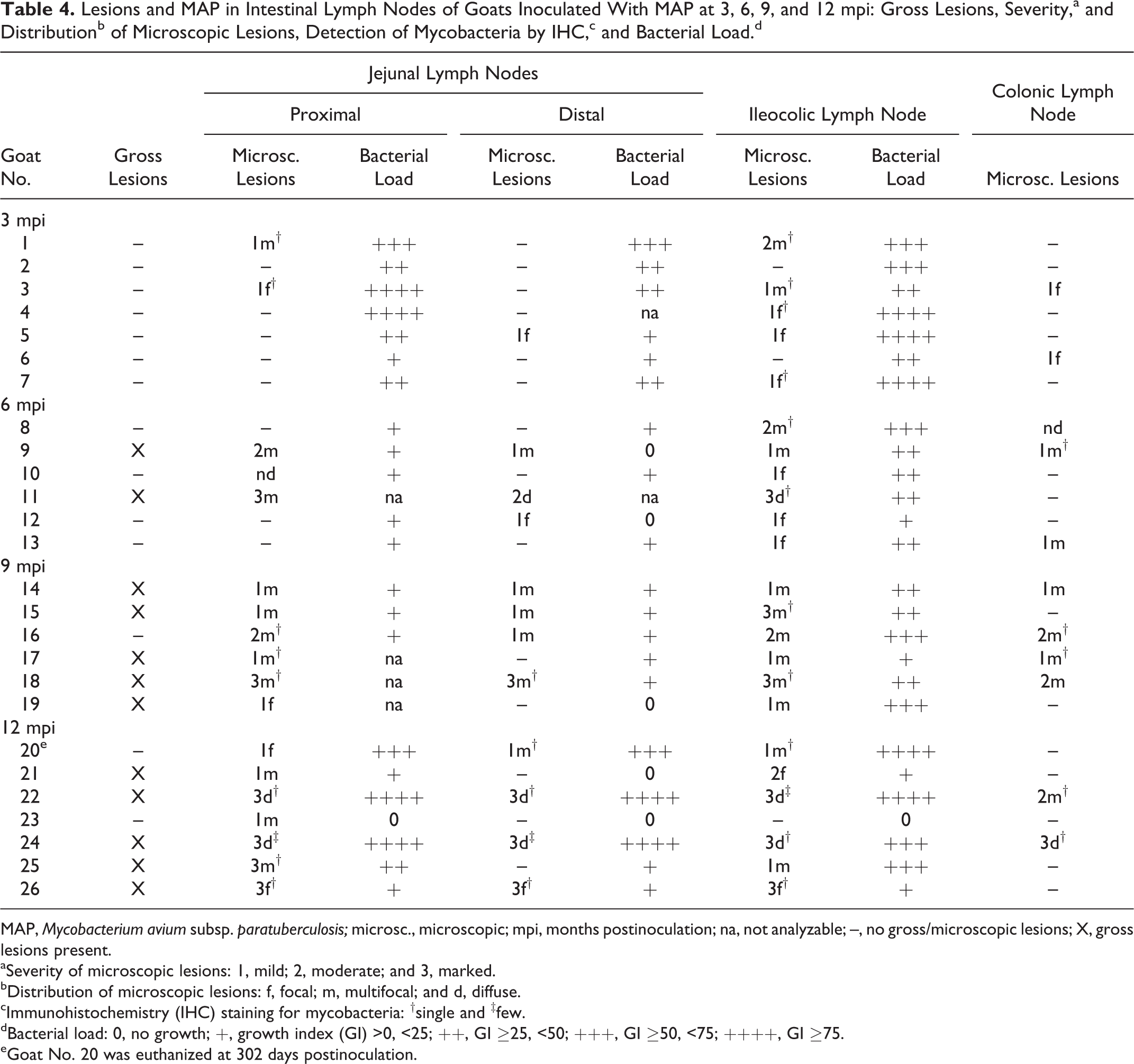
MAP, Mycobacterium avium subsp. paratuberculosis; microsc., microscopic; mpi, months postinoculation; na, not analyzable; –, no gross/microscopic lesions; X, gross lesions present.
aSeverity of microscopic lesions: 1, mild; 2, moderate; and 3, marked.
bDistribution of microscopic lesions: f, focal; m, multifocal; and d, diffuse.
cImmunohistochemistry (IHC) staining for mycobacteria: †single and ‡few.
dBacterial load: 0, no growth; +, growth index (GI) >0, <25; ++, GI ≥25, <50; +++, GI ≥50, <75; ++++, GI ≥75.
eGoat No. 20 was euthanized at 302 days postinoculation.
Other Organs
A focal infiltrate of epithelioid cells and MGCs was seen in the paracortex of a hepatic LN from 1 goat. Cultural isolation of MAP was possible in low amounts from the hepatic LN of 6 goats, superficial cervical LN of 4 goats, tonsil and liver of 3 goats each, and retropharyngeal LN of 2 goats.
Six Months Postinoculation (6 Goats)
Intestinal Tract
Macroscopic lesions in GALT and/or intestinal mucosa were seen in 5 goats (Tables 2 and 3). In 1 goat, all JPPs were severely thickened and ulcerated with regional lymphangitis; ICVPP and PCPP had a marked serositis. In addition, a few round or irregularly shaped, 2- to 5-mm areas of thickened mucosa sometimes centrally depressed (crater-like structures) were seen in the jejunum and proximal colon. Two other goats had mild multifocal serositis of JPPs. In 1 goat, 1-mm white firm nodules were scattered throughout JPPs and the IPP. In 2 goats, only crater-like structures in the jejunum were noted.
By histology, lesions were detected in GALT of all goats, but they varied markedly in severity (Table 2). In 3 goats, only mild focal to multifocal granulomatous infiltrates were present and were predominantly located in interfollicular areas. In the other 3 goats, lesions as described for the animals at 3 mpi were seen. In the goat with the severe gross lesions, GALT was partly to completely replaced by granulomatous infiltrates. The other 2 had multifocal to coalescing granulomatous infiltrates. In 1 goat, multiple granulomas with central necrosis and calcification were distributed throughout JPPs and the IPP. The crater-like structures in the jejunal mucosa were verified histologically in 1 goat (No. 8) only. They were not associated with GALT but consisted of areas of mucosa with severe atrophy of villi, crypt hyperplasia, diffuse lymphoplasmacytic infiltration, and multifocal to confluent foci of epithelioid cells and MGC in the lamina propria and submucosa. The crater-like appearance in the small intestine was due to the atrophy of central villi. By IHC, mycobacteria were detected in moderate to severe lesions in the GALT of 2 goats as described for the goats 3 mpi (Table 2). MAP was culturally isolated in low to moderate amounts from GALT and intestinal mucosa (Tables 2 and 3).
Lymph Nodes
Gross lesions were found as multiple, 1- to 3-mm foci of necrosis and calcification in J-LNs of 2 goats (Table 4). The ICV-LN of 1 goat had extensive areas of necrosis and calcification (Fig. 5). Histologic examination revealed focal to multifocal granulomatous inflammation in the mesenteric lymph nodes of all goats. Granulomatous infiltrates were small in 3 and more extensive in the other 3 goats, which had in addition granulomas with necrosis and calcification (Fig. 6). Mycobacteria were detected by IHC as granular structures in necrotic foci of 3 goats (Fig. 6, inset) and as single bacteria in epithelioid cells of 1 goat (Table 4). Bacterial load of MAP was low in J-LNs and moderate to high in ICV-LNs of all 6 goats (Table 4).
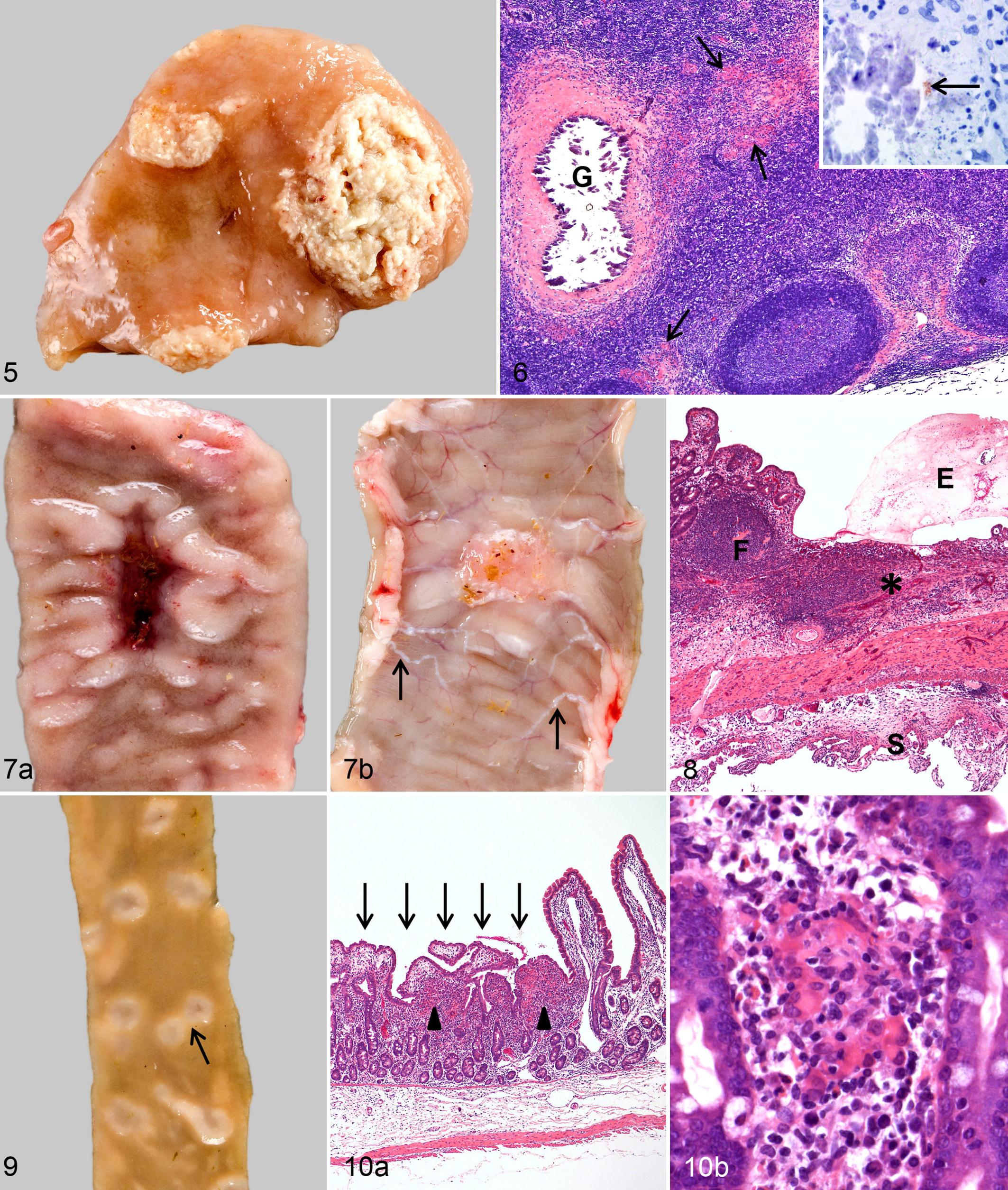
Other Organs
One goat had a few small granulomatous infiltrates in the liver and in the paracortex of the associated hepatic lymph node. MAP was culturally isolated in low amounts from the hepatic LN of 6 goats.
Nine Months Postinoculation (6 Goats)
Intestinal Tract
GALT
Macroscopic lesions were seen in 5 goats (Table 2). Multifocal small nodules were seen in 3 goats in the JPP and/or IPP. In 2 goats, all JPPs were deeply indented and surrounded by thickened nodular mucosa. Their surface was ulcerated in 1 goat (Fig. 7a). JPPs were easily located because of marked circumscribed fibrous serositis and regional lymphangitis (Fig. 7b).
Histologically, focal granulomatous infiltrate was seen in a JPP of 1 goat comparable to the findings in 3 goats at 6 mpi. The nodular thickening observed grossly in 3 goats consisted of multifocal to coalescing granulomatous infiltrates as described for the goats at 3 and 6 mpi. An increased number of MGCs and lymphocytes were present in 1 goat. In 2 goats, GALT was replaced by a severe transmural infiltrate of lymphocytes, plasma cells, and fibrous connective tissue with few diffusely interspersed epithelioid cells or multiple foci of epithelioid cells and MGCs (Fig. 8). There was a multifocal formation of lymphoid follicle-like structures. The overlying mucosa was either ulcerated or irregular without domes and villi and covered by attenuated cuboidal epithelium. Mesenteric lymphatics in the subserosa were surrounded by lymphohistiocytic infiltrates, and there was a severe circumscribed villous lymphohistiocytic to granulomatous serositis (Fig. 8). By IHC, mycobacteria were detected only as single bacteria in a few epithelioid cells in JPPs and the terminal IPP of 4 goats (Table 2). Cultural isolation revealed low amounts of MAP in the IPP of 5, low to moderate amounts in the ICVPP of 5, and high amounts in the JPP of 3 and in the PCPP of 2 goats (Table 2).
Intestine
Multiple, sometimes confluent crater-like structures as described for the goats at 6 mpi were found in the mucosa outside the GALT of 5 goats (Table 3, Fig. 9). They were numerous in the proximal and mid-jejunum and rare in the duodenum, distal jejunum, and ileum. Irregular, up to 6 mm in diameter mucosal thickening was seen in the cecum and proximal colon of 3 goats. Histologic lesions as described for the goats at 6 mpi were found in all 5 goats (Fig. 10a,b). By IHC, mycobacteria were detected in a few epithelioid cells in the lamina propria in the shortened villi of 3 of the 5 goats (Table 3). MAP was isolated in low amounts from the jejunal mucosa of 4 goats (Table 3).
Lymph Nodes
Mesenteric lymph nodes were enlarged in 5 goats (Table 4). Small 1- to 2-mm foci of calcification were seen in the subcapsular cortex of 2 goats and more extensive areas of necrosis and calcification in another 2 goats. By histology, granulomatous infiltrates were found in 6 and granulomas with necrosis and calcification in 4 goats. The cellular composition was mainly as described for 3 and 6 mpi. In 2 goats, numerous MGCs were seen in the subcapsular sinuses of the distal J-LN. By IHC, mycobacteria were detected in epithelioid cells or more frequently as focal labeling in the necrosis of at least 1 intestinal lymph node of 4 goats (Table 4). Bacterial load of MAP was low in the J-LNs and moderate to high in the ICV-LN of all 6 goats (Table 4). In 1 goat, MAP was culturally isolated only from the ICV-LN.
Other Organs
Three goats had a few small granulomatous infiltrates of epithelioid cells in the liver and 2 goats of epithelioid cells and MCGs in the hepatic lymph nodes. MAP was not isolated by culture or detected by IHC.
Twelve Months Postinoculation (7 Goats)
Intestinal Tract
GALT
Variable macroscopic lesions were seen in 5 goats (Table 2). In 1 goat, all JPPs were thickened with deep ulcerations (Fig. 11). In 3 goats, JPPs of the proximal and mid-jejunum were either deeply indented and covered by intact mucosa (Fig. 12) or extensively ulcerated. Small foci of calcification were present in the submucosa. Nodular thickenings were seen in 2 of the 3 goats. JPPs in 1 goat were interpreted as severely atrophic (Fig. 13). They were associated with a mild circumscribed villous serositis. The IPP was focally thickened in 2 goats and the ICVPP multifocally ulcerated, edematous, and thickened in 1 goat. Mild serositis was seen in the ICVPP of 2 and the PCPP of 4 goats.
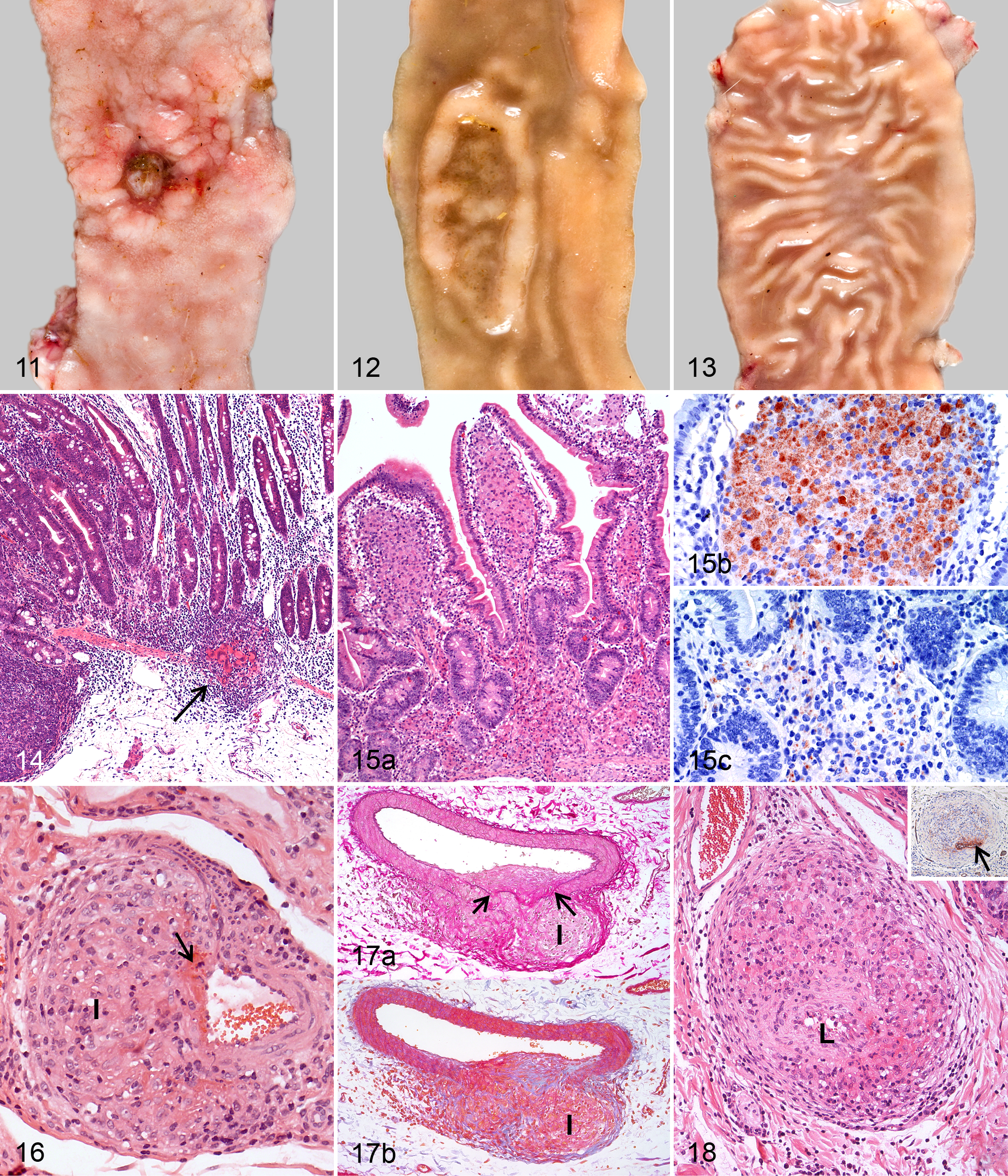
By histology, highly variable lesions were observed, and all goats were affected (Table 2). A few granulomas with central calcification and fibrous capsule were seen in 1 goat. Focal to multifocal infiltrates of few epithelioid cells and MGCs embedded in an extensive lymphoplasmacytic infiltrate predominated in 3 goats, as described for goats at 9 mpi (Fig. 14, Nos. 21, 25, and 26). In the atrophic JPPs found in 1 of the 3 goats, the organized lymphoid tissue was replaced by mild lymphoplasmacytic infiltration and fibrosis and covered by intact mucosa. Confluent to diffuse infiltrates of predominantly epithelioid cells and reduced numbers of lymphocytes and plasma cells were present in lesions of the remaining 3 animals. Numbers of lymphocytes and plasma cells were most mild in the goat (No. 24) with the most severe lesions. In this goat, multifocal necrosis and accumulation of neutrophils were found in the submucosa. By IHC, few or many mycobacteria were detected in the numerous epithelioid cells of 3 goats, while none could be demonstrated in the few epithelioid cells of the remaining 4 goats (Table 2).
Intestine
Gross lesions were seen in the small intestine of 6 and large intestine of 5 goats (Table 3). These were characterized by crater-like structures—as described for the goats at 6 and 9 mpi—in the small intestine of 5 goats; circumscribed mucosal thickening in the cecum and proximal colon of 3 goats; firm, white 1- to 5-mm nodules in the jejunum, ileum, cecum, and proximal colon of 3 goats; and segmentally thickened corrugated mucosa and lymphangitis with foci of necrosis and calcification in the small intestine of 2 goats (Nos. 22 and 24). In goat No. 24, the extensive necrosis of colonic lymph nodes had caused focal perforation of the distal colon with leakage of ingesta into the abdominal cavity and severe diffuse adhesive peritonitis.
By histology, lesions could be confirmed in all goats with gross lesions. Crater-like structures and circumscribed mucosal thickenings in the small and large intestine corresponded to those described for the goats at 6 mpi. Single mycobacteria were present in epithelioid cells of 2 of the 5 goats (Table 3). Small granulomas with central necrosis and calcification were present in the submucosa of 3 goats. Labeling for mycobacteria was detected in the granulomas of 1 goat. In the goats with segmentally thickened corrugated mucosa, confluent to diffuse infiltrates of epithelioid cells were seen in the lamina propria and submucosa (Fig. 15a). Mycobacteria were few in 1 of the goats but numerous in the other, with the highest number of mycobacteria in the villus tips (Fig. 15b) and markedly fewer in the basal lamina propria and submucosa (Fig. 15c).
Lymph Nodes
Mesenteric lymph nodes were enlarged in 5 goats with few 1- to 5-mm foci of necrosis and calcification in 2 and multifocal to extensive necrosis in 3 goats (Table 4). By histology, granulomatous infiltrates were seen in 7 goats and focal to multifocal granulomas with central necrosis and calcification, delineated in part by connective tissue, in 3 goats. Mycobacteria were detected in epithelioid cells and as granular deposits in the areas of necrosis.
Other Organs
Small foci of granulomatous inflammation were observed in the tonsil of 1 goat (No. 20), in the liver of 1 goat (No. 22), and in hepatic lymph node, liver, kidney, and lung of 1 goat (No. 24). In the last animal, a few mycobacteria were detected in epithelioid cells in the hepatic LN, liver, and lung by IHC.
Cultural Isolation of MAP
The results of cultural isolation of MAP were highly variable (Tables 2–4). MAP could not be isolated from any tissue of the animal with the mildest lesions. Low to moderate amounts of MAP predominated in 3 goats, with high bacterial load only in the ICVPP and PCPP. The bacterial load was high to very high in GALT, intestinal mucosa, and mesenteric LNs of the 3 remaining goats. MAP was also isolated from hepatic, superficial cervical, and retropharyngeal LNs of these 3 animals and from tonsil, liver, and spleen of 2.
Granulomatous Arteritis
Granulomatous arteritis of small- and medium-sized arteries was seen in the submucosa of the jejunum and colon in several goats at 6, 9, and 12 mpi (Table 2). The media were segmentally and frequently asymmetrically infiltrated by macrophages, epithelioid cells, lymphocytes, and occasionally few neutrophils in 5 goats (Nos. 12, 15, 17, 21, and 26; Figs. 16–18). Some granulomatous lesions were associated with fibrinoid necrosis of the vascular wall, leakage of erythrocytes, and activation and mitosis of endothelial cells (Fig. 16). The presence of an internal elastic membrane characteristic of arteries was shown in nonaffected segments of the vascular walls by special staining (Fig. 17a). Granulomatous infiltrates were delineated by mild fibrosis (Fig. 17b). The expansion of a granulomatous infiltrate resulted in some arteries having complete obliteration of the normal architecture and occlusion (Fig. 18). FVIII-positive remnants of endothelium and fragments of the internal elastic membrane helped to confirm that focal, circular, well-demarcated foci of granulomatous inflammation originated from arteries (Fig. 18, inset).
Discussion
The experimental infection of goat kids with MAP and the sequential autopsy of groups of 6 to 7 animals at predefined times postinoculation allow a detailed description of the progression of lesions and numbers of MAP within lesions over time. Throughout the entire observation period of 1 year, 25 of the 26 inoculated goats and all control goats were without clinical signs of paratuberculosis. Only 1 inoculated goat developed cachexia but no diarrhea at 12 mpi. Cachexia is a rather nonspecific clinical sign, but it has been described as a frequent finding in paratuberculosis of small ruminants. 6,13,20,24 Thus, most infected goats were in the clinically inapparent phase of paratuberculosis.
The MAP dose per animal administered in our study (100 mg bwm amounting to 2.6 × 108 cfu) was lower than doses applied in other experimental infection studies with goats. 15,26,39 Nevertheless, distinct macroscopic alterations were found in 20 of 26 inoculated goats and characteristic histologic lesions in all 26 inoculated goats. Lesions were attributed to the MAP infection because they were not seen in any of the control goats, and MAP was detected by culture. Macroscopic lesions occurred as early as 3 mpi predominantly in JPPs that were firm and thickened by granulomatous infiltration and had ulcerated surfaces. Mucosal ulcerations have been described in cases of naturally infected goats with diffuse intestinal lesions 19,24 and after experimental infection of goats with a mycobacterium isolated from patients with Crohn’s disease. 42 The ulcerations we observed were frequently accompanied by transmural inflammation, resulting in foci of circumscribed serositis along the small intestine. These may be the sites of origin of the strictures described in naturally infected goats. 19 The serositis facilitated a precise collection of altered intestine.
At 9 and 12 mpi, depleted Peyer’s patches (PPs) were covered by a simple epithelial lining instead of functional mucosa with villi, crypts, and domes. This was interpreted as repair of the mucosal ulceration and correlated with the regression of the inflammatory infiltrate in the PPs. The atrophic PPs observed at 12 mpi may represent a final stage of this development. In the goat that developed clinical signs of cachexia, there was no repair, but ulcers had perforated, causing diffuse chronic adhesive peritonitis. Although this is generally not associated with paratuberculosis in goats, similar findings were described in a recent study of naturally infected goats. 20
Variable findings have been reported concerning the preferential sites of lesions in the intestine of goats with paratuberculosis. Several studies found the jejunum most severely affected, 19,20,36,41 whereas others noted the distal small intestine. 25,40 In our investigation, predominantly JPPs in the proximal and mid-jejunum and the ICVPP were altered in subclinical paratuberculosis. This is substantiated by the results of cultural isolation of MAP at 3 mpi in which high amounts of MAP were isolated from JPPs and ICVPP, while only low amounts were found in IPP. Both JPPs and ICVPP represent sites of secondary lymphoid tissues, whereas the IPP that was less frequently affected is a combination of primary and secondary lymphoid tissues. 1,34 The increased frequency of lesions in JPPs and ICVPP may thus reflect variations in the uptake of luminal antigens between different sites of GALT. 18
At 3 mpi, lesions were mainly located in GALT, had rarely spread to intestinal lymph nodes, and were not detected in intestine outside GALT. This indicates that JPPs and ICVPP are the main ports of entry for MAP. An association between GALT and lesions of paratuberculosis has been described in several studies 9,16,17,25,36,41 and was related to the preferential but not exclusive uptake of MAP by M cells. 23,33,35 Small focal granulomatous infiltrates outside GALT and the cultural recovery of MAP may indicate that uptake of MAP may also occur via enterocytes, as has been shown by studies of ovine and murine intestine. 2,33 Similar focal granulomatous infiltrates seen in older, naturally infected goats, sheep, and cattle were interpreted as latent or persistent lesions. 7,14,30
It has been hypothesized that the translocation of MAP leads initially to a local inflammation in the GALT, which may spread locally due to suppressed local cell-mediated immune reactions, causing segmental lesions, and later disseminate to the lamina propria and mesenteric lymph nodes. 17,36 The fact that in the present study, moderate to very high quantities of MAP were isolated from jejunal lymph nodes and ICV-LN with mild lesions indicates that dissemination to local lymph nodes precedes tissue reactions.
Although individual animals could not be sequentially sampled in our study, the fact that all goats at 3 mpi had confluent to diffuse lesions in JPPs and individual goats at 6, 9, and 12 mpi had only small focal infiltrates indicates that regression of lesions may occur. This is further emphasized by the culture results showing reduced bacterial loads in most examined locations of the goats at 6 and 9 mpi.
There was no continuous progression in the extent and severity of lesions. Lesions were located at the same sites but were milder at 6 mpi compared with 3 mpi. A marked increase of severity and spreading to mucosa outside GALT and to mesenteric lymph nodes was noted at 9 mpi. The highest variation between individuals was seen at 12 mpi, varying from mild focal granulomatous infiltrates to severe diffuse lesions throughout the intestine. This confirms previous studies in sheep and goats that were difficult to interpret because of the low animal numbers per group. 17,25,36 In sheep, lesions were seen as early as 10 days postinoculation with multifocal to diffuse infiltrates at 3 mpi. 16,17 At 7 mpi, 1 lamb had small granulomatous infiltrates, while the other had extensive lesions with numerous AFB. 17 In goats, lesions were found in 1 study not before 3 mpi; at 4, 6, and 9 mpi; and not at 12 mpi, although this animal had seroconverted. 36 Another study reported epithelioid cell infiltrates with many AFB in JPP and IPP at 2 mpi and only small multifocal epithelioid cell infiltrates without AFB at 3, 4, 5, 6, and 9 mpi. 25 Differences between these studies might be due to differences in the dose of the inoculum and mode of inoculation, but the overall time course suggests that at some point during the subclinical infection, goats may control the multiplication of MAP, resulting in regression and healing of lesions, or an uncontrolled multiplication of MAP occurs and causes progression toward clinical disease. This divergence is most likely mediated by the immune system, but the triggering event is unclear. If the regression is not complete and MAP survives (eg, in foci of necrosis and calcification, which are frequent in mesenteric lymph nodes), exacerbation may occur as commonly seen in other mycobacterial infections. 44
Using IHC, most lesions were classified as paucibacillary, containing no or only very low numbers of mycobacteria. In contrast, high to very high amounts of MAP were culturally isolated from GALT and lymph nodes of 6 goats at 3 mpi and from intestinal mucosa, GALT, and lymph nodes of 3 goats at 12 mpi. This confirms previous studies, which revealed that IHC was markedly less sensitive than bacterial culture in detecting MAP-positive tissue 11,22 and is, therefore, very likely to underestimate bacterial load of tissue samples. The distribution and severity of histologic lesions in the clinically inapparent phase were not well reflected by the low number of mycobacteria detected by IHC.
Unique findings were the crater-like lesions in the small intestine of infected but not control goats, which were seen with increasing frequency with progression of time. Similar macroscopic lesions have been described in other experimentally infected goats at 9 mpi and 2 years postinoculation but were not microscopically examined. 25,41 The histologic examination revealed that they were not associated with isolated lymphoid follicles in the intestinal wall but affected the mucosa at random. The crater-like lesions became confluent in more severely affected goats; this may suggest that they are progenitors of the regional enteritis observed in paratuberculosis. 4
Another uncommon finding in paratuberculosis was the granulomatous arteritis in small- to medium-sized vessels in the submucosa of 5 goats at 6, 9, and 12 mpi. 5 There is one report of arteritis in cattle with diffuse multibacillary lesions, 14 but in our study, goats with mild, paucibacillary lesions were affected. Comparable lesions have been described in cases of intestinal tuberculosis in vessels in the submucosa of the ICVPP and small- and medium-sized vessels in the mesentery, causing occlusion of arteries, ulcerations, perforations, and strictures, and in cases of Crohn’s disease in humans. 8,21,43 As in the present investigation, granulomatous vasculitis was often demonstrated in these cases by special stains only. Thus, it may have been misinterpreted in previous reports as focal granulomatous infiltrates, especially when the lumen was occluded.
Granulomatous arteritis may be an additional mechanism of dissemination of MAP and MAP-infected cells both locally in the intestine and systemically. In the intestinal tract, MAP-infected emboli may become trapped in the capillary network in the lamina propria of the villi. This would explain the preferential localization of granulomatous infiltrates in the villous core. 14,17 The vascular branching may also explain the initially focal lesions in the intestine outside GALT.
As reported in calves, cultural isolation of MAP from tonsils, retropharyngeal and superficial cervical lymph nodes, and liver and hepatic lymph nodes at 3 mpi might be due to a direct colonization of these organs. Tonsils were found to be a common portal of entry for MAP in addition to the small intestine in calves. 29 Since isolation was less frequent in goats at 6 and 9 mpi, there was apparently little progression of infection after the initial colonization in these extraintestinal tissues. Dissemination to numerous extraintestinal tissues was observed in the goats with confluent to diffuse granulomatous intestinal lesions at 12 mpi, as described for sheep and goats. 6,19
In conclusion, the presented model of experimental infection of goats with MAP allows new insights into the pathogenesis of paratuberculosis and reveals extensive interactions between MAP and the host organism early after infection without any clinical evidence of disease. These initial interactions take place in GALT and are most likely crucial for further progress of the infection. The vascular lesions observed in the intestinal wall may be essential for the initiation of the regional enteritis. Similarities with Crohn’s disease in humans make this model interesting for comparative studies.
Footnotes
Acknowledgements
We thank the animal facility at FLI, Jena, for their expert help; Jörg Otto for his support with autopsies; Wolfram Maginot for the photographic documentation and the layout of the pictures; Sabine Lied and Lisa Wirker for the paraffin histology and special stains; and Sandy Werner for preparation of the inoculum and cultural isolation of MAP from tissues.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Animal Diseases Fund of Thuringia, Hesse, Mecklenburg-Western Pommerania, Lower Saxony, Rhineland-Palatinate, and Baden-Wuerttemberg.
