Abstract
A 5-month-old female Great Dane puppy was treated for hematochezia, tenesmus, and rectal prolapse by resection of a 10-cm-long segment of colon and rectum. Grossly, the colorectal segment had diffuse mucosal and submucosal thickening with multiple polypoid nodules. The histologic diagnosis was colorectal hamartomatous polyps with ganglioneuromatosis. Duplication of PTEN was detected by quantitative multiplex polymerase chain reaction testing. The presence of 2 hamartomatous colorectal lesions with PTEN mutation is similar to human Cowden syndrome.
In human medicine, pediatric colorectal polyps are recorded in hamartomatous polyposis syndromes—juvenile polyposis syndrome, Peutz Jeghers syndrome, and the PTEN (phosphatase and tensin homolog) hamartoma tumor syndrome, which includes Cowden syndrome—that share autosomal dominant inheritance, increased risk of colon cancer, and complications such as rectal bleeding, intussusception, and bowel obstruction.1,6,15 Diffuse intestinal ganglioneuromatosis is recorded in Von Recklinghausen syndrome, multiple endocrine neoplasia type IIb, and Cowden syndrome.8,10 Diffuse colorectal ganglioneuromatosis and ganglioneuroma have both been reported in dogs.5,11,12 However, concurrent hamartomatous polyposis and ganglioneuromatosis have not been reported in nonhuman animals. We report colorectal hamartomatous polyposis and ganglioneuromatosis with PTEN duplication in a Great Dane puppy.
Case History
A 5-month-old female Great Dane puppy was presented with tenesmus, hematochezia, and rectal prolapse. A 10-cm-long segment of the colon and rectum was resected and submitted for pathologic evaluation. Grossly, the colorectal wall was diffusely thickened (mean thickness, 20 mm). The mucosal surface was distorted by about 20 polypoid nodules, 2 to 8 mm in diameter, which protruded into the lumen.
Methods
Portions of the formalin-fixed surgical specimen were processed routinely for histologic examination. Paraffin sections, 3 μm in thickness, were stained with hematoxylin and eosin (HE). Immunohistochemistry was performed using a standard avidin–biotin–peroxidase complex method, as well as markers for neural cells (NeuN, dilution 1:800, mouse monoclonal, Chemicon, Billerica, MA), S-100 protein (dilution 1:4000, Dako, Carpinteria, CA), glial fibrillary acidic protein (GFAP; dilution 1:5000, Dako), and proliferation (Ki67, dilution 1:50, Dako). Brain tissue was used as the positive control for NeuN, S-100 protein, and GFAP. The primary antibodies were omitted for the negative controls.
Genomic DNA was extracted from formalin-fixed, paraffin-embedded (FFPE) tissue, using the NucleoSpin Tissue XS Kit (Qiagen, Courtaboeuf, France), according to the manufacturer’s protocol. First, direct sequencing was used to search for single-nucleotide substitutions or small rearrangements of the canine PTEN gene. Primers were designed according to GeneBank reference sequences (NM_001003192 for mRNA, NC_006608 for genomic sequence) for polymerase chain reaction (PCR) amplification and sequencing of coding exons and their intronic boundaries. Because NC_006608 contains a large gap sequence, encompassing exon 1 and its promoter, only the last 10 exons were evaluated. Both PTEN and a second candidate gene, BMPR1A (bone morphogenic protein receptor 1A), were searched for large rearrangements by quantitative multiplex PCR of short fragments (QMPSF). A set of primers was designed to simultaneously amplify exons 5 (122 base pairs [bp]), 8 (137 bp), and 4 (180 bp) of PTEN, as well as exons 2 (102 bp), 6 (130 bp), and 11 (113 bp) of BMPR1A and exon 14 (78 bp) of MLH1 (mutL homolog 1), used as a control. Histologically normal FFPE tissues (colon, rectum, liver, kidney, tongue, and blood platelets) from healthy dogs were used as controls.
Results
Histologically, the colon and rectum had severe diffuse mural thickening with polypoid mucosal nodules (Fig. 1). The polypoid nodules consisted of tubular epithelial proliferation in abundant stroma, infiltrated by lymphocytes, plasma cells, and macrophages (Fig. 2). Many colonic glands were so dilated by mucus and cellular debris that the lining epithelium was flattened. Besides well-differentiated colonic glands lined by columnar epithelium with numerous goblet cells, less differentiated glands (Fig. 3) were lined by crowded cuboidal epithelial cells with few goblet cells and increased immunohistochemical labeling for the proliferation marker, Ki67. Some glands extended through the muscularis mucosae into the submucosa.
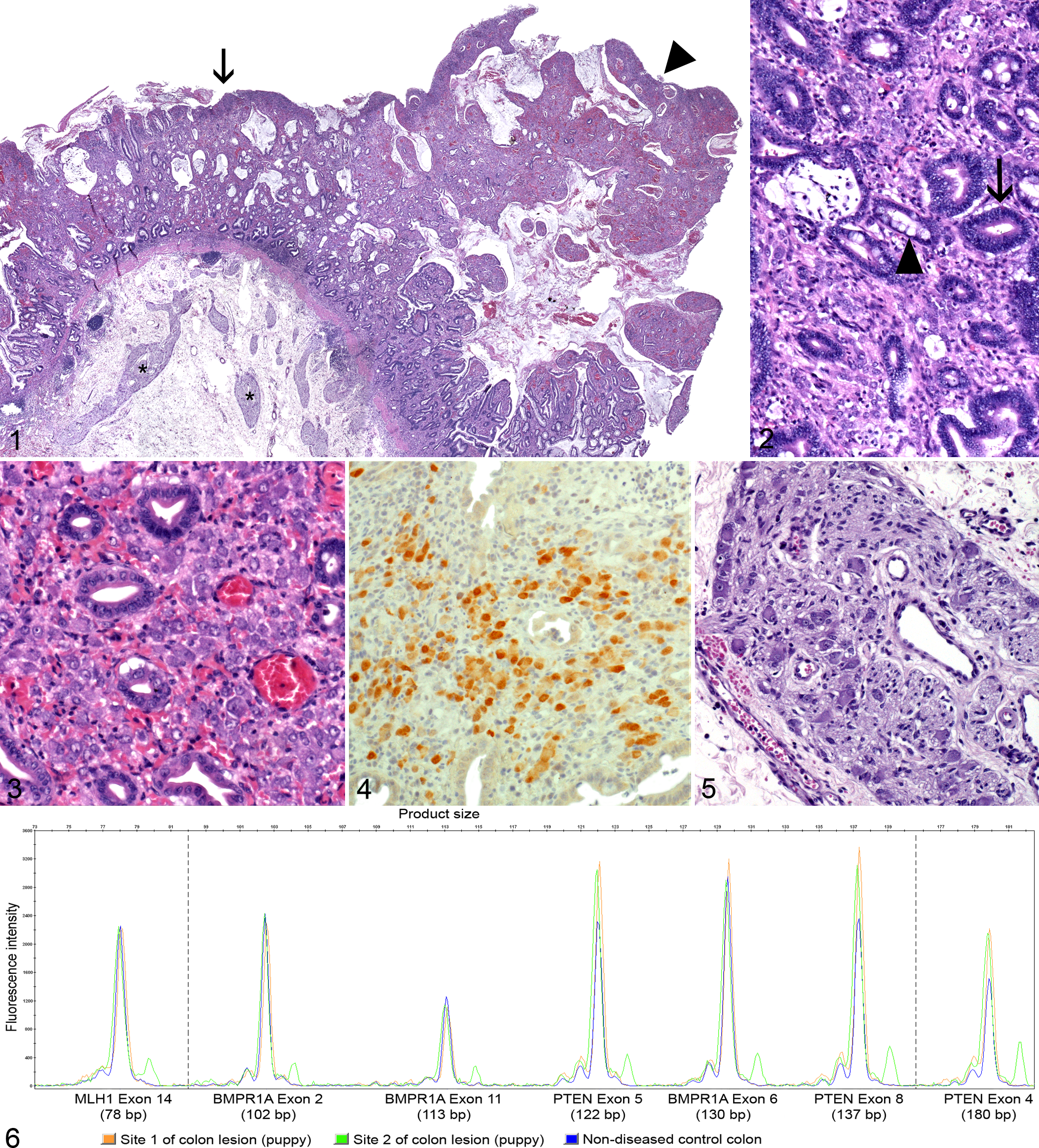
Numerous ganglionic neuronal cell bodies, up to 40 μm in diameter, were scattered individually or in clusters through the colorectal lamina propria (Fig. 3). These ectopic (mucosal) neuronal cell bodies were labeled by NeuN immunohistochemistry (Fig. 4). Absence of GFAP or S-100 protein immunoreactivity indicated a lack of accompanying glial cells. Profiles of submucosal (Fig. 5) and myenteric plexuses were increased in size and number.
The mucosal epithelial proliferation was diagnosed as colorectal hamartomatous polyposis; the presence of ectopic ganglion cells in the colorectal mucosa, together with hyperplastic and hypertrophic enteric plexuses, was classified as diffuse ganglioneuromatosis. The 3 amplified exonic products (exons 4, 6, 8) of PTEN were duplicated (Fig. 6). No large rearrangement was detected in BMPR1A.
Discussion
A hamartoma is a tumor-like lesion composed of a disorganized overgrowth of mature cells and tissues in their normal anatomic location. Human colorectal hamartomatous polyps are encountered in rare pediatric hamartomatous polyposis syndromes, inherited as autosomal dominant traits, 15 and characterized by gastrointestinal polyps and symptoms such as rectal bleeding and prolapse. Each syndrome has distinct histologic features, organ involvement, and germline mutation. Peutz-Jeghers syndrome is caused by mutation of the STK11 gene (serine/threonine kinase 11), and it consists of gastrointestinal (usually, small intestinal) hamartomatous polyps, with smooth muscle proliferation, mucocutaneous hyperpigmentation, and increased risk of stomach, small bowel, colon, pancreas, breast, ovary, uterus, and lung cancer.1,6,15 Juvenile polyposis syndrome is characterized by multiple polyps without smooth muscle proliferation and an increased risk of colorectal cancer. Germline mutations of SMAD4 (SMAD family member 4), BMPR1A, and ENG (endoglin) genes, involved in the transforming growth factor β signaling pathway, have been identified.1,3,14,15 Polyps in Cowden syndrome are histologically indistinguishable from those of juvenile polyposis syndrome but are caused by mutation of PTEN. The PTEN gene acts as a negative regulator of the AKT pathway; loss of PTEN function alters cell cycle regulation, cell adhesion and motility, angiogenesis, and cell size.3,15
In this Great Dane puppy, the clinical signs of rectal bleeding and prolapse, the colorectal location of the hamartomatous tissue, and the histologic features all bear a striking resemblance to the polyps of juvenile polyposis syndrome and Cowden syndrome. Colorectal polyps have been described in middle-age or older dogs 2 but not, to our knowledge, in a puppy.
Diffuse ganglioneuromatosis is defined as a poorly demarcated, multinodular to diffuse, intramural to transmural proliferation of ganglion cells of the enteric autonomic nervous system.1,5,6,10 Human hereditary intestinal ganglioneuromatosis is associated with 3 main entities. First, multiple endocrine neoplasia type IIb is caused by mutations of the RET proto-oncogene, involved in growth and differentiation of neural crest-derived tissues.8,10,13 Second, Von Recklinghausen disease, associated with NF1 (neurofibromin 1) gene mutations, is characterized by neurofibromas along peripheral nerves. 1 The gastrointestinal tract involvement in 25% of patients is manifested as localized neurofibromas or diffuse mural ganglioneuromatosis. 15 Finally, diffuse ganglioneuromatosis is described in Cowden syndrome, in association with colorectal hamartomatous polyps.
Three cases of ganglioneuromatous proliferation have been reported in young dogs. Colonic ganglioneuromatosis was reported in a 12-month-old Lhasa Apso, but lesions were restricted to the submucosa and the tunica muscularis and unaccompanied by mucosal epithelial proliferation. 5 An 18-month-old Australian Terrier had 2 rectal mucosal ganglioneuromas. 11 A jejunal ganglioneuroma was confined to the tunica muscularis in an 18-month-old Doberman Pinscher. 12
The coexistence of 2 distinct hamartomatous colorectal lesions—polyps and diffuse ganglioneuromatosis—has not been reported in veterinary medicine. Mutation of one gene (PTEN) could account for both lesions, as described in human Cowden syndrome. 9 No such mutation was identified in this case; however, neither exon 1 nor its promoter was evaluated, because they are not spanned by the sequences in public databases. Thus, a point or frameshift mutation cannot be ruled out. Because of this limitation and assuming that 2 hits would be required if PTEN were involved, we investigated a possible loss of the second allele by QMPSF. This method has been used to detect large rearrangements in DNA extracted from human FFPE specimens. 7 A second candidate gene, BMPR1A, was evaluated. Our results rule out a whole gene deletion for PTEN and BMPR1A. However, duplication of PTEN was detected in the 3 exons examined (exons 4, 5, 8). The duplication could correspond to a copy number variation (CNV), unrelated to the lesions, as suggested by a recent study in which CNV was reported in a large region encompassing PTEN of canine chromosome 26. 4 Alternatively, this duplication could have disrupted the open reading frame and led to an aberrant transcript.
Human patients with Cowden syndrome are at greater risk of colon, breast, thyroid, and endometrial cancer. This dog could not be evaluated for recurrence or development of additional colorectal lesions, because it died of gastric dilation and volvulus 1 year after colorectal resection/anastomosis and necropsy was not permitted.
Footnotes
Acknowledgements
We thank Catherine Guereaud, Lydie Guigand, and Bernard Fernandez, Department of Pathology, Nantes-Atlantic College of Veterinary Medicine, Food Science and Engineering, for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
