Abstract
Microcystic hamartoma was detected as a tumorlike mass in the left caudal lung lobe of a 12- year-old mixed-breed dog. Histologically, the mass was characterized by microcysts of various sizes that mimicked alveoli and were surrounded by thin fibrous septal tissue. However, unlike the adjacent lung parenchyma, bronchial or bronchiolar trees were absent, and the septal vascular channels were extremely underdeveloped. Immunohistochemically, the cells lining the microcysts were consistently positive for cytokeratin but not for vimentin, whereas the septal fibroblast-like cells were negative for cytokeratin and positive for vimentin. Electron microscopy confirmed that the microcysts were lined with a layer of type I and type II mature pneumocytes. This is the first description of the detailed morphologic features of microcystic hamartoma.
Keywords
Hamartoma refers to a benign tumorlike nodule composed of an overgrowth of mature cells and tissues native to the organ in which it occurs. 4 Pulmonary hamartomas reported in domestic animals are of cartilaginous, 1 vascular, 6 bronchiolar, 7 and alveolar 2 3 origin. Hamartomas of alveolar origin seen in foals have been called polyalveolar lobe or lobar hypertrophy. 2 3 The affected lobes showed a tumorlike mass and were characterized histologically by an excessive number of alveoli. In the lungs of humans with tuberous sclerosis, a hamartomatous proliferation of type II pneumocytes occurs frequently and is called micronodular pneumocyte hyperplasia. 5 The lesions are recognized as multiple well-demarcated nodules up to 8 mm in diameter. In a report of primary mesenchymal or mixed-cell-origin lung tumors in four dogs, one case designated as microcystic hamartoma was composed of well-differentiated fibrous and epithelial tissue. 8 However, the detailed histologic features were not described. Here, we describe the histologic, immunohistochemical, and ultrastructural features of a microcystic hamartoma in the lung of an adult dog.
A tumorlike mass was incidentally found in the left caudal lung lobe of a 12-year-old mixed-breed dog by roentgenogram and computed tomography. Surgical excision of the affected lobe was performed by thoracotomy. The excised lobe including the mass was fixed in 10% neutral buffered formalin. Fixed samples of the mass and the adjacent lung parenchyma were embedded in paraffin, cut at 5 μm, and stained with hematoxylin and eosin (HE), Masson trichrome for collagen fiber, and Elastica von Gieson for elastic fiber. Frozen sections of small pieces of the mass were subjected to oil red O and Sudan black B stains for lipids. For immunohistochemistry, paraffin-embedded sections were stained using a streptavidin–biotin–peroxidase system with monoclonal antibodies against human smooth muscle actin and porcine vimentin (Dako A/S, Glostrup, Denmark) or with polyclonal antibodies against bovine keratin and bovine S-100 protein (Dako, Carpinteria, CA). For vimentin, smooth muscle actin, and keratin stains, sections were incubated in phosphate-buffered saline at 121 C for 20 minutes. For electron microscopy, small pieces of the formalin-fixed mass were postfixed in 1% osmium tetroxide, embedded in epoxy resin, and stained with uranyl acetate and lead citrate.
The grayish white, solid mass was 2 cm diameter and was located in the lung parenchyma just beneath the pleura. It was coarsely lobulated and noncystic and was sharply delineated from the underlying lung parenchyma, which was collapsed and congested.
Histologically, the mass was composed of microcystic spaces of various sizes that mimicked the alveoli (Fig. 1). The microcystic spaces were lined with a layer of small round or flat cells and often contained the amorphous substances positive for both oil red O and Sudan black B stains and macrophages phagocytosing them (Fig. 2). Among the microcysts, there were thin septa composed of moderate numbers of fibroblast-like cells, a few blood vessels, and various numbers of collagen fibers but no elastic fibers. Lung parenchyma adjacent to the mass was without apparent fibrous demarcation and had collapsed alveoli, moderate congestion, and chronic bronchitis.

Lung; dog. Hamartoma with microcystic spaces of various sizes and septal fibrous tissue. HE. Bar = 63 μm.
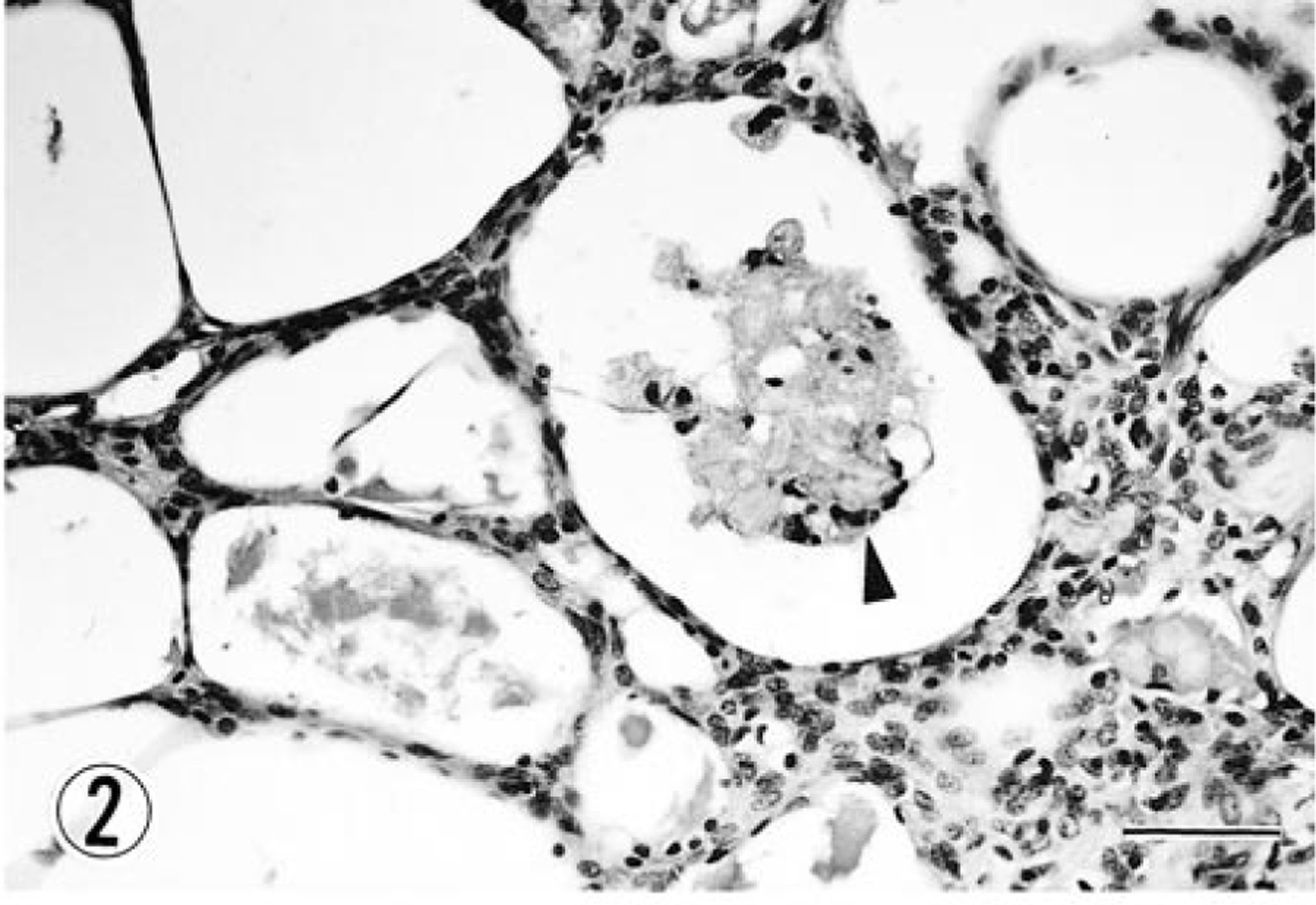
Lung; dog. Higher magnification of Fig. 1 showing amorphous substances and macrophages (arrowhead) in microcyst. HE. Bar = 20 μm.
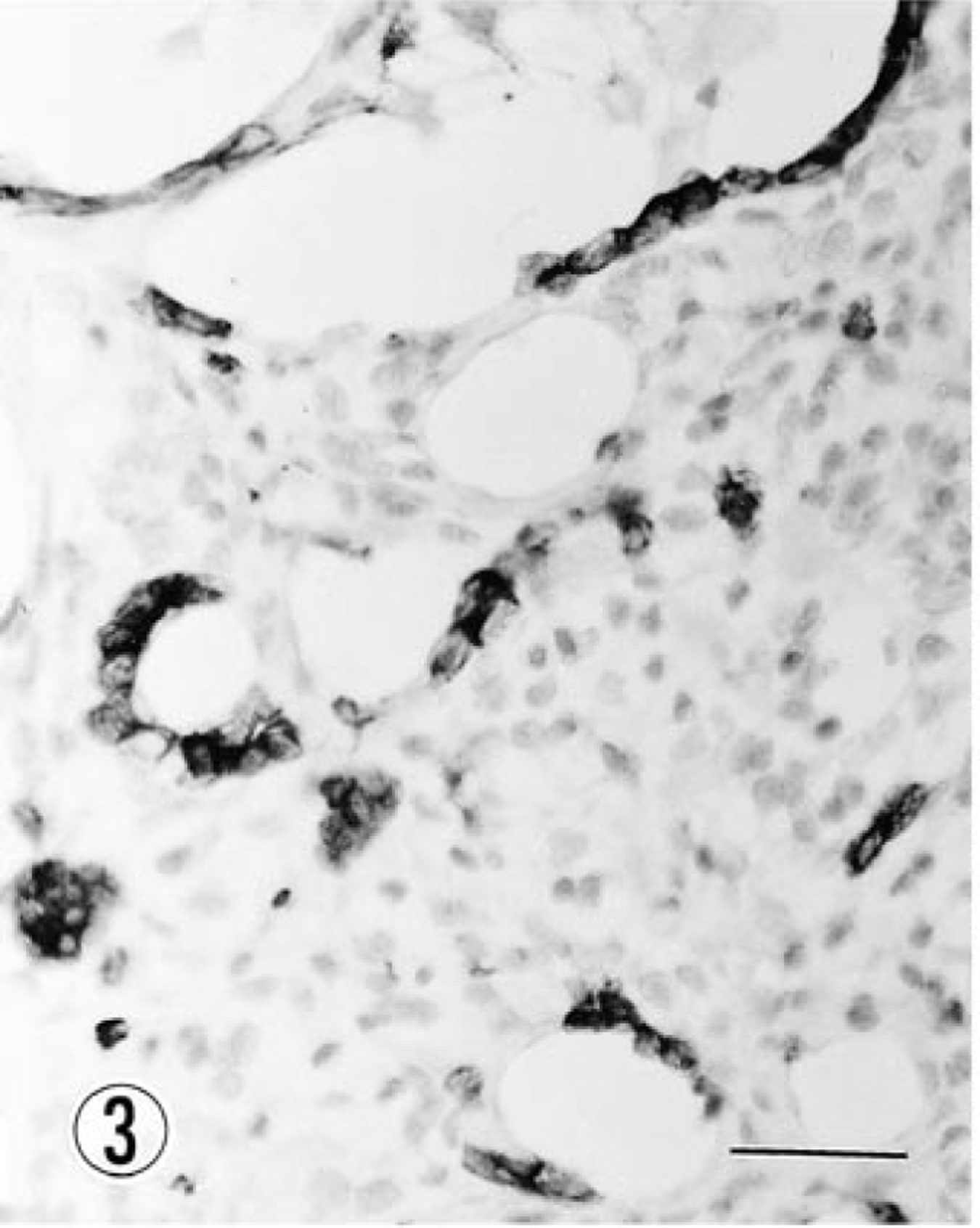
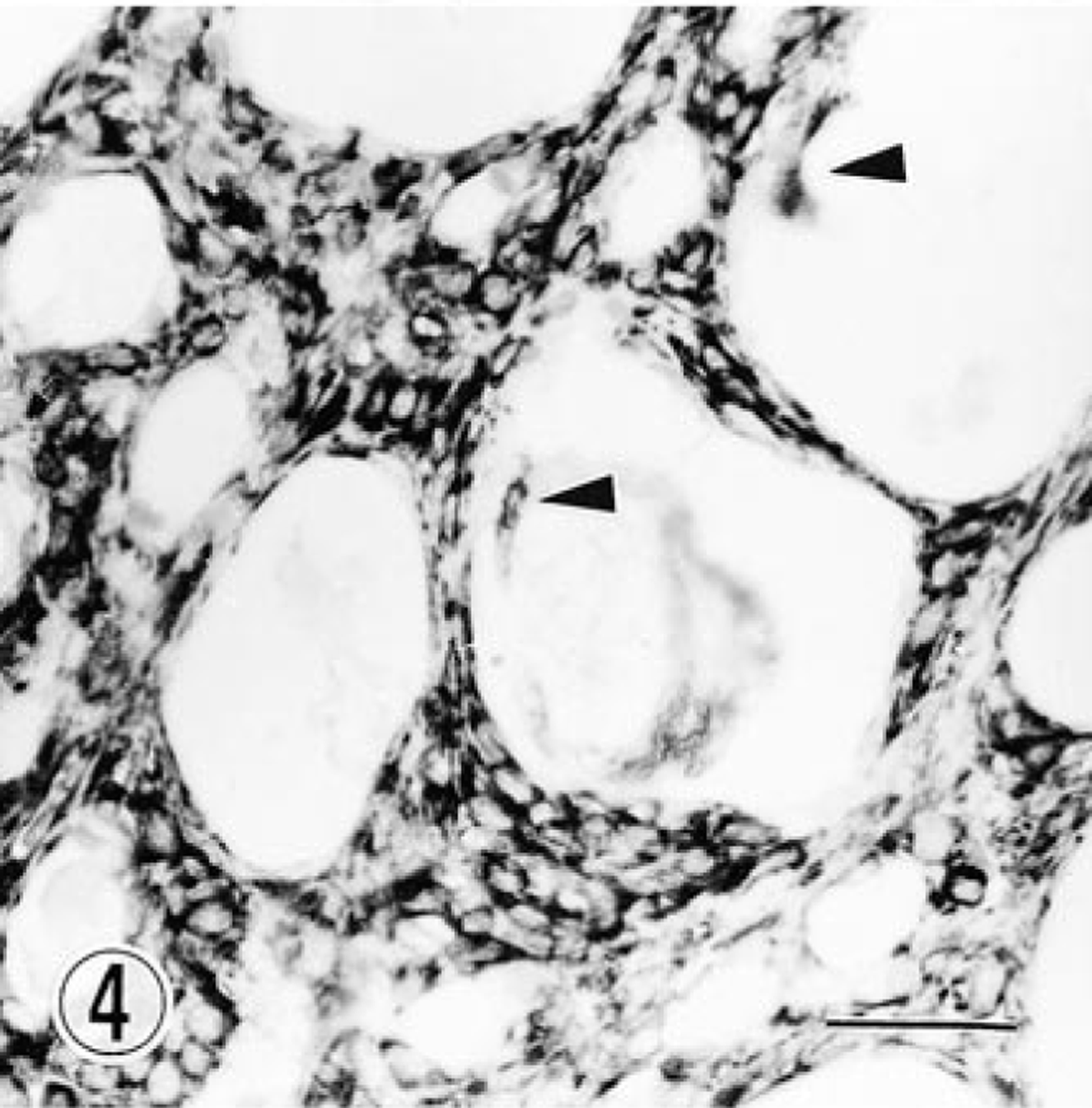
Immunohistochemically, cells lining the microcysts were consistently positive for cytokeratin (Fig. 3) and negative for vimentin, whereas fibroblast-like cells were positive for vimentin (Fig. 4) but not for cytokeratin. Very few fibroblast-like cells showed a positive reaction to smooth muscle actin; microcyst-lining cells were consistently negative for smooth muscle actin. There were no cells reactive to S-100 protein.
Lung; dog. Microcyst-lining cells showing strong positivity for cytokeratin. Immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 12 μm.
Lung; dog. Vimentin-positive fibroblast-like cells. Positive reaction is also seen in macrophages (arrowheads) in microcysts. Immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 12 μm.
Electron microscopy revealed that the cysts were lined with cells corresponding to type I and type II pneumocytes. Type I cells had an extremely large amount of attenuated cytoplasm, which was rich in ribosomes and pinocytotic vesicles. It was difficult to find the nuclei because they were scarce in the thin sections. Type II cells had dark cytoplasm with a large round or oval nucleus (Fig. 5). These cells were mainly characterized by various numbers of round lamellar bodies in the cytoplasm (Fig. 5). Both types of cells rested on a thin and continuous basal lamina, which formed a complete separation between the inner microcyst lining and the septal connective tissue. The major element in the septum was fibroblast-like cells which had moderately developed rough endoplasmic reticulum, dispersed ribosomes, some pinocytotic vesicles, and a few mitochondria. Bundles of collagen fibers intervened between those cells.
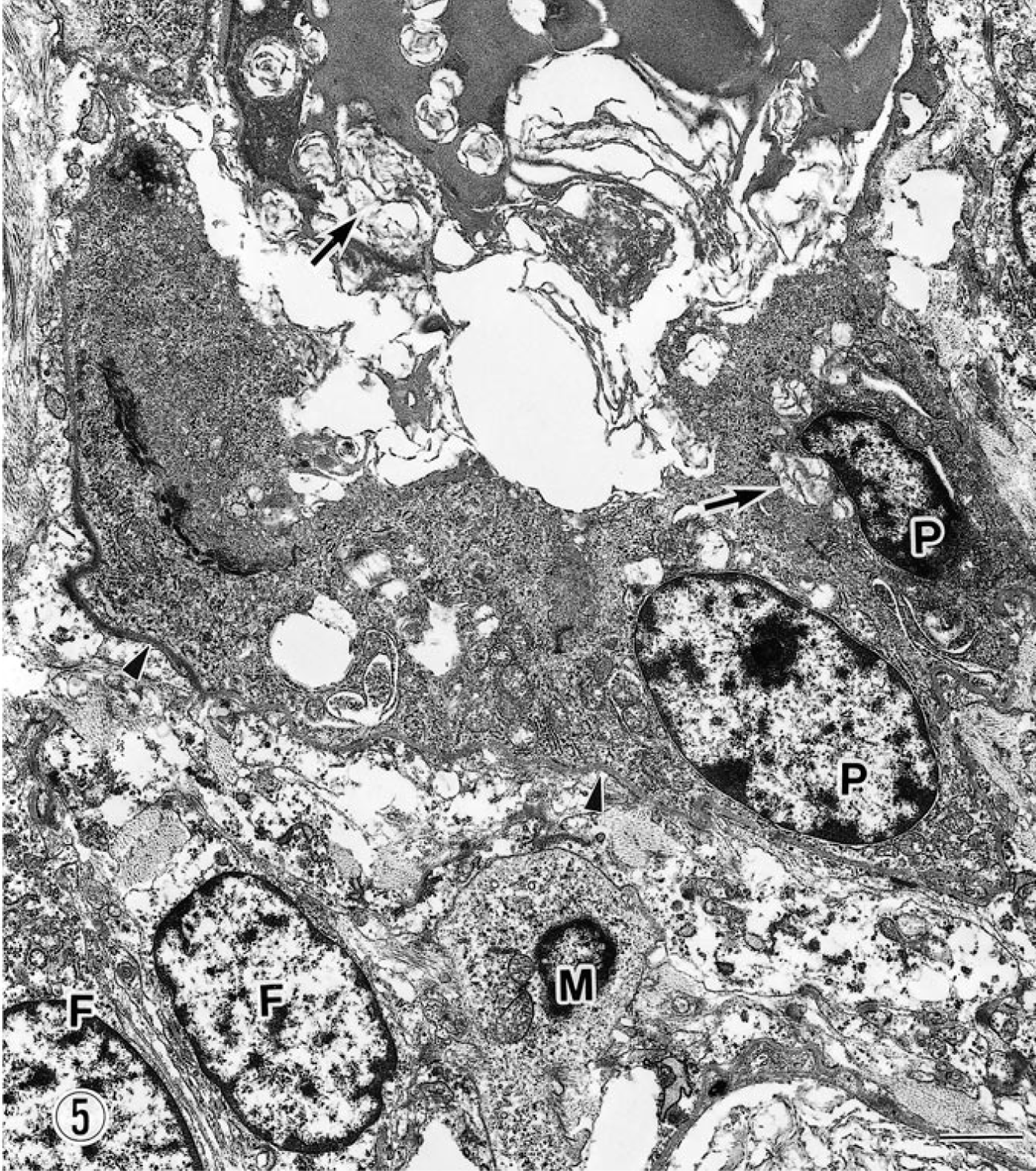
Lung; dog. Type II pneumocytes (P) are surrounded by basal lamina (arrowheads). Fibroblast-like cells (F) and macrophages (M) are seen in the septum. Arrows indicate intracytoplasmic and intramicrocytic lamellar bodies. Uranyl acetate and lead citrate. Bar = 2 μm.
It is often impossible to distinguish between hamartomas and benign neoplasms. Pulmonary hamartoma with mixed involvement of lipocytes and epithelial cells (lipomatous hamartoma) or fibrous and epithelial cells (microcystic hamartoma) was documented in two dogs as a benign neoplasm. 8 In this dog, despite the gross tumorlike appearance of the mass no neoplastic changes were seen in any type of cells in the mass, although fibroblast-like cells in the septum appeared to be somewhat undifferentiated. The mass was considered a tumorlike malformation composed of an abnormal combination of normal cell and tissue components of the lung. These changes also fulfill the diagnostic criteria of a hamartoma.
Another major difference between this mass and normal lung tissue or other hamartomatous lesions of alveolar origin 2 3 5 was the complete absence of bronchi and bronchioles. These microcysts were blind sacs that had developed with no connection to the tracheobronchial tree of the lung and thus were nonfunctional in gaseous exchange, although structurally they resembled alveoli. This lack of function accounts for the abnormal accumulation of a surface-active material (surfactant) secreted by type II pneumocytes within the microcysts. In addition, the septal constituents in this hamartoma differed from those of normal alveolar wall in the following respects: 1) a paucity of vascular channels, 2) a high population of fibroblast-like cells, and 3) an absence of elastic fibers. This malformation in the lung may have occurred at the stage of organogenesis in the embryo and may have continued to grow to some extent in postnatal period, although its exact histogenesis remains unclear.
Footnotes
Acknowledgements
We thank Dr. Y. Nomura, Azabu University, Kanagawa, for his helpful discussion.
