Abstract
Cross-species infection with ovine herpesvirus 2 (OvHV-2) in cattle causes malignant catarrhal fever (MCF). MCF may involve the central nervous system (CNS) with necrotizing arteritis and/or vasculitis described to be unique to MCF and discriminatory compared to other viral CNS infections. However, a systematic histopathological characterization of the neural form of MCF in cattle is lacking. We examined medulla oblongata (
Keywords
Malignant catarrhal fever (MCF) is a multisystemic disease of Artiodactyla caused by members of the gammaherpesvirus family. Cross-species infections from natural hosts, in which infection is asymptomatic, to dead-end hosts causes generally severe clinical infections with fatal outcome. In the European, Australian, and North and South American cattle population, sheep-associated MCF due to cross-species transmission of the endemic ovine herpesvirus 2 (OvHV-2) is the most frequent variant. 17,18,24,25,39 Transmission from sheep to cattle is a result of direct or indirect contact between the 2 animal species but is considered to be inefficient with an estimated incidence of only 0.6‰. 23 Clinical MCF in cattle usually occurs sporadically with little to moderate economic impact for farmers, while morbidity and associated economic losses may be high in other farmed dead-end host species. 3,7,27,32
Persistent high fever (>40 °C) in association with mucosal ulcerations and bilateral keratitis with corneal opacity are considered to be cardinal clinical signs of MCF but affected animals may also suffer from neurological deficits, cutaneous lesions, or diarrhea. 4,8,30 Due to sporadic incidence, governmental surveillance activities to monitor MCF in the cattle population are not implemented in Switzerland. However, MCF was detected in European syndromic surveillance programs and accounts for about 2.8% to 3.8% of investigated cases in passive bovine spongiform encephalopathy (BSE) surveillance programs targeting animals with neurological signs (unpublished data). 2,21
Until recently, the method of choice for reliably diagnosing MCF was histopathological examination of various organs including kidney, liver, urinary bladder, oral mucosa, cornea or conjunctiva, and brain. 8,24 Typical histopathological findings in these organs include lymphohistiocytic and necrotizing vasculitis especially of small- and middle-sized arteries as well as lymphohistiocytic infiltrates with lymphoblasts in nonlymphoid organs, paracortical hyperplasia of lymph nodes, and degeneration or necrosis of lymphoid follicles and various epithelia. Necrotizing vasculitis/arteritis typically affecting the carotid rete mirabile, the brain, and the kidneys is considered to be the pathognomonic lesion of MCF. In the brain, lymphohistiocytic meningoencephalitis may occur 17,24,25,39 with sporadic descriptions of neuronal degeneration or necrosis, gliosis, and neuronophagia mimicking neurotropic viral encephalitis. 12,13 Consequently, lack of discrimination of neural MCF may result in underestimation of MCF prevalence. Additionally, a reliable demarcation of neural MCF from notifiable encephalitic diseases including bovine herpesvirus 1 (BHV1) infection, rabies, Aujeszky’s disease, louping ill, tick-borne encephalitis (TBE), and bovine virus diarrhea/mucosal disease (BVD/MD) 1,5,6 is indispensable. Discrimination may be difficult when examination is restricted to the brain (brainstem) and a full-body necropsy is not available, which is frequently the case in BSE surveillance. In such cases, molecular confirmation may be required for definitive diagnosis of MCF. Polymerase chain reaction (PCR) tests for detection of OvHV-2 nucleic acid in tissue samples and blood are suggested by the World Organization of Animal Health (OIE) for a definitive diagnosis of MCF. 24
Because clear pathological criteria of neural MCF are lacking, the aim of this study was to systematically describe the histopathology of bovine neural cases in which OvHV-2 was confirmed by quantitative polymerase chain reaction (qPCR) in the brain, with a focus on vasculitis and neuronophagia. Additionally, in situ hybridization (ISH) and immunohistochemistry (IHC) were performed to characterize the target cells of OvHV-2 in the brain.
Materials and Methods
Case Definition
MCF was defined based on the repeated positivity of brain samples in a quantitative OvHV-2 PCR with a cycle quantification (Cq) <40. Within the framework of diagnostic pathology and BSE surveillance studies between 1995 and 2020 at the Division of Neurological Sciences, Vetsuisse Faculty, University of Bern, 125 animals (75 cattle with histopathological evidence of nonsuppurative meningoencephalitis and 50 cattle without histopathological lesions) were screened for the presence of OvHV-2 DNA by qPCR. Neurological signs were reported in 38/75 animals with nonsuppurative meningoencephalitis. Presence of neurological signs was unknown in 37/75 animals with nonsuppurative meningoencephalitis and 50 animals without brain lesions. Sixteen animals with nonsuppurative meningoencephalitis (of which neurological signs were reported in 9; Table 1) and 2 animals without lesions scored repeatedly positive in qPCR and were included in this study. Most of these 18 animals were included in 2 previous studies. 37,40 Formalin-fixed and paraffin-embedded (FFPE) brain tissues from the cases were retrieved from the archive of the Division of Neurological Sciences. The brain was the only tissue examined in this study. Subsets of brains were tested for the presence of other viruses including astroviruses (17/18, all negative), bovine herpesvirus 6 (15/18, 1 positive animal [case 6], Cq 31.1), Borna disease virus (7/18, all negative), bovine polyomavirus 2 (7/18, all negative), rabies virus (7/18, all negative), flaviviruses (7/18, all negative), paramyxoviruses (16/18, all negative), and bovine retrovirus CH15 (7/18, all negative). 37,40
Clinical and Pathologic Findings and Quantification of OvHV-2 in the Brain of 18 Cattle.
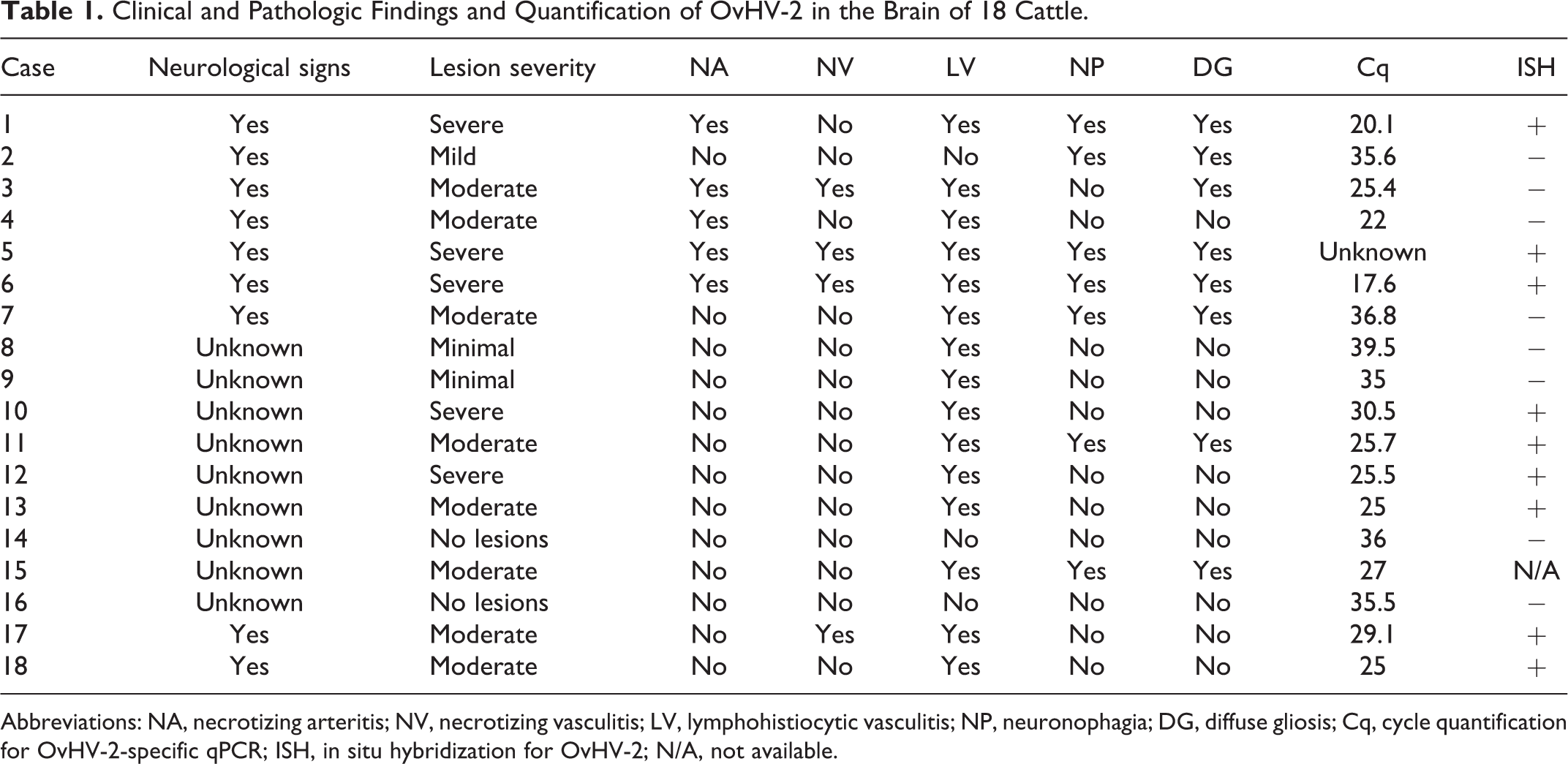
Abbreviations: NA, necrotizing arteritis; NV, necrotizing vasculitis; LV, lymphohistiocytic vasculitis; NP, neuronophagia; DG, diffuse gliosis; Cq, cycle quantification for OvHV-2-specific qPCR; ISH, in situ hybridization for OvHV-2; N/A, not available.
Samples
Of the 18 cases, FFPE tissue blocks of multiple brain regions (including medulla oblongata, midbrain, hippocampus, thalamus, basal nuclei, cerebrum) were available in 9 cases, while in 8 cases only FFPE tissue blocks of the medulla oblongata were accessible (Table 2). Unfortunately, from one animal, only hematoxylin and eosin–stained sections of medulla oblongata were available precluding additional investigations. Blood (case 5), frozen pooled samples of obex, frontal lobe and thalamus (cases 17 and 18), or frozen samples of the medulla oblongata (remaining cases) were used for DNA extraction.
Presence of Lesion in Brain Regions of OvHV-2-Positive Cattle.
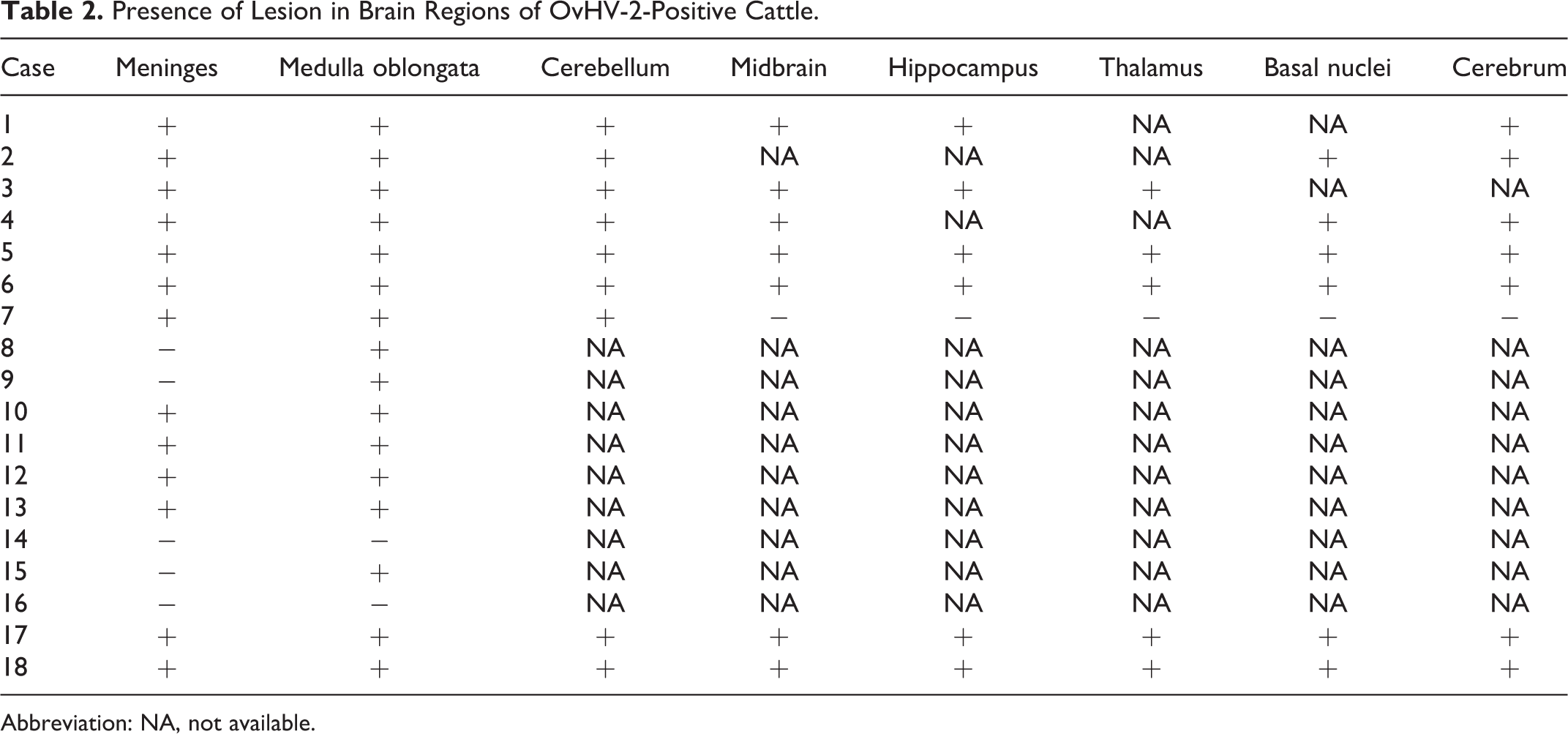
Abbreviation: NA, not available.
Fresh frozen and FFPE brain tissue of animals that had been previously tested negative for OvHV-2 by next generation sequencing (NGS) and qPCR 40 and FFPE brain tissue of a healthy slaughtered animal served as negative controls for qPCRs and ISH.
DNA Extraction and qPCR
For each animal, DNA extraction was carried out using the DNeasy Blood and Tissue Kit (Qiagen) according to the manufacturer’s instruction. Extracted DNA was used for quantitative taq-man probe-based PCR with a previously published primer-probe combination. 36 The assays were run with an end volume of 25 µl and contained 1× Path-ID qPCR Master Mix (Thermo Fisher Scientific Inc), 240 nM of the forward primer (GAGAACAAGCGCTCCCTACTGA), 600 nM of the reverse primer (CGTCAAGCATCTTCATCTCCAG), 80 nM of the probe (FAM-AGTGACTCAGACGATACAGCACGCGACA-BHQ1), and 10 µl extracted DNA. Cycling conditions were adjusted to 2 minutes at 50 °C, 10 minutes at 95 °C followed by 40 cycles of 15 seconds at 95 °C and 1 minute at 60 °C. QPCRs were run either on a 7300 Real-time PCR System (Thermo Fisher Scientific Inc) or on a CFX96TM Real Time System (BioRad). Accordingly, for following Cq determination the Sequence Detection Software (version 1.4, Thermo Fisher Scientific Inc) or the FX Maestro software (version 4.1.2433.1219, BioRad) was used with an auto-calculation of the baseline threshold. A suitable cutoff value for positive scoring was evaluated previously and was set to 40 reaction cycles. 40 Animals with the lowest Cq value or in which OvHV-2 was already confirmed in NGS 40 served as positive controls for following qPCR reactions. Additionally to the above-mentioned negative controls, a water sample was carried along as a nontemplate control in every reaction.
Histopathological Examination
Histopathological analysis was performed on hematoxylin and eosin–stained slides. For each case, vascular lesions were classified as lymphohistiocytic vasculitis if there was infiltration of the vascular wall with lymphocytes and macrophages without apparent fibrinoid vessel wall necrosis, and/or as necrotizing vasculitis if there was fibrinoid vessel wall necrosis of any type of vessel, and the presence of neutrophils, with or without perivascular protein leakage and hemorrhages. Lesions were separately recorded as necrotizing arteritis if fibrinoid vessel wall necrosis of arteries was unambiguously observed. Hence, 1 to 3 types of vasculitis might have been recorded per case. Additionally, neuronophagia (neuronal degeneration or necrosis, nodular gliosis) and diffuse gliosis (diffusely increased number of astrocytes and microglia) were recorded. Lesion severity was categorized in “no lesions” when no inflammatory lesions were observed in the brain, “minimal” when few inflammatory cells were found around vessels without forming complete perivascular cuffs, “mild” when few vessels were surrounded by complete perivascular cuffs of one cell layer with or without gliosis and neuronophagia, “moderate” when multifocal perivascular cuffs with 2 to 3 layers of inflammatory cells were present with or without gliosis and neuronophagia, and “severe” when perivascular cuffs exceeded 3 layers of inflammatory cells and the distribution was multifocal or coalescing.
In Situ Hybridization
In 17 animals, brain tissue was available for in situ detection for OvHV-2. Of every animal, FFPE brain slides with the most severe lesions were chosen for ISH. In animals with only medulla oblongata available, this region was used. ISH was performed with the RNAscope system (Advanced Cell Diagnostics) using the RNAscope 2.5 Detection Kit-Brown (Advanced Cell Diagnostics) according to the manufacturer’s instructions. A probe targeting OvHV-2 ORF25 and ORF50 (Advanced Cell Diagnostics; Cat No. 501091) was used in the assays. Counterstaining was performed with Mayer’s hemalum solution (Merck KGaA) and the slides were mounted with Aquatex (Merck KGaA) mounting medium. FFPE tissue slides of one animal that displayed a strong nuclear ISH staining in a pilot staining experiment served as a positive control for the following screening (case 6). Negative controls included tissues of animals tested negative for OvHV-2 by qPCR and NGS 40 or a healthy slaughtered animal.
Immunohistochemistry
Available FFPE tissue slides of all animals with neuroparenchymal lesions and 3 animals without neuroparenchymal lesions (cases 3, 4, and 12) were additionally labeled with antibodies against CD3 and CD20 by IHC to investigate the parenchymal distribution of T and B lymphocytes, respectively. IHC labeling was performed as described elsewhere 28 with minor modifications. For both IHC runs, antigen retrieval was performed using the pH6 Target Retrieval Solution of the RNAscope System (Advanced Cell Diagnostics) as described by the manufacturer and the polyclonal rabbit anti-human CD3 antibody (Dako Denmark A/S) was diluted 1:200 in Dako Antibody Diluent (Dako Denmark A/S). Signal detection was performed with the Mouse and Rabbit Specific HRP/DAB (ABC) Detection IHC kit (ab64264; Abcam Plc) according to the manufacturer’s instructions. The slides were counterstained with Mayer’s hemalum solution, and finally mounted with Aquatex (Merck KGaA) mounting media.
CD3+ T lymphocytes were counted in perivascular cuffs and brain parenchyma of 10 fields of view and, due to lower abundancy, in glial nodules of 5 fields of view of a size of 0.159 mm2 each (400× magnification) on a Zeiss Axio Scope.A1 microscope (Carl Zeiss Microscopy GmbH). The ratio of CD3+ cells to the total number of nuclei present in the fields of view was calculated. FFPE tissue slides of an animal with negative results for OvHV-2 in qPCR and NGS 40 were incubated with rabbit immunoglobulin fraction (solid-phase absorbed; Dako Denmark A/S) and served as a negative control.
Combined In Situ Hybridization and Immunofluorescence
A combination of fluorescent ISH and immunofluorescence was performed on one positive animal with neuroparenchymal brain lesions (case 6) as described elsewhere 15 in order to identify the infected cell type and their distribution in the brain parenchyma. Briefly, ISH was performed with the RNAscope 2.5 Detection Kit-Red (Advanced Cell Diagnostics) according to the manufacturer’s instructions to the point of signal detection with the above-described OvHV-2 probe (Advanced Cell Diagnostics; Cat No. 501091). Then, immunofluorescence was performed with polyclonal rabbit anti-human CD3 antibody (1:200 in Dako Antibody Diluent at 4 °C overnight; Dako Denmark A/S), polyclonal rabbit anti-human CD20 antibody (1:100 in PBS-T for 1 hour at room temperature; Thermo Fisher Scientific Inc) and polyclonal rabbit anti-ionized calcium-binding adapter molecule 1 (Iba-1) antibody (1:500 in PBS-T for 1 hour at 37 °C; WAKO). Following incubation with the primary antibodies, slides were washed 3 times in PBS-T and incubated with fluorescent secondary antibodies (Alexa Fluor 488 goat anti-rabbit; Abcam Plc) diluted 1:1000 in PBS-T and DAPI BioChemica (AppliChem GmbH) diluted 1:10 000 in PBS-T for 1 hour at room temperature. After a further washing step, slides were mounted with Glycergel, Aqueous Mounting Medium (Dako Denmark A/S) and correlation analysis was performed on an Olympus Fluoview FV3000 Confocal Laser Scanning Microscope (Olympus Europa). As a negative control, a FFPE tissue slide of an animal that was tested negative for OvHV-2 in both qPCR and NGS 40 was incubated with the OvHV-2-specific probe and unspecific rabbit immunoglobulin fraction (Dako Denmark A/S). The slide was treated with detection protocols as described above.
Results
In the 18 cases that were positive by OvHV-2 qPCR, histopathological examination of the brain revealed a broad spectrum in the neuropathological phenotype. Interestingly, no histopathological changes were observed in 2/18 animals (11%). In the other 16/18 animals (89%), lesion severity ranged from minimal to severe. Lesion phenotypes included primarily vascular changes such as lymphohistiocytic vasculitis or necrotizing vasculitis and arteritis, but also neuronophagia, or diffuse reactive gliosis. In cases with several brain regions available, no clear regional targeting of lesions was observed (Table 2). Neurological signs were reported in 9/18 animals (50%) but not further described, whereas no clinical history was available in the remaining 9/18 animals (50%). The presence of neurological signs, lesion severity, lesion type, as well as results of the OvHV-2-specific qPCR and ISH are provided for every animal in Table 1.
Inflammatory changes were most commonly moderate or severe. Vasculitis was the most common pathological presentation observed in neuroparenchymal and meningeal vessels, most commonly affecting small sized vessels, but also larger arteries and veins. The most frequent type of vasculitis was a lymphohistiocytic infiltration of the vascular wall and Virchow-Robin space (15/18 animals, 83%) with occasionally few neutrophils (Fig. 1). In 4/15 animals with lymphohistiocytic vasculitis (27%), necrotizing vasculitis (ie, fibrinoid vessel wall necrosis and the presence of neutrophils) was additionally present in the neuroparenchyma (Fig. 2) and meninges. Hence, the overall prevalence of necrotizing vasculitis in this study was 22% (4/18). Intriguingly, necrotizing arteritis (ie, fibrinoid vessel wall necrosis of arteries), which is considered to be a pathognomonic lesion of MCF, was observed in only 5/18 of the animals (28%) and mainly affected meningeal but not neuroparenchymal arteries (Fig. 3). Indicators of vascular damage and increased blood brain barrier permeability (hemorrhages, perivascular protein leakage) was present in 2 additional animals out of 15 with lymphohistiocytic vasculitis (13%; Fig. 4). In 7 of the 16 brains with inflammatory lesions (44%), signs of neuronophagia such as nodular gliosis with or without neuronal degeneration were observed (Fig. 5); all of these were accompanied by diffuse gliosis (Fig. 6). On the other hand, neuronophagia was associated with vasculitis in 6/7 animals (86%, Table 1). In one of these animals, however, only minimal meningeal arteritis was observed additionally to the neurotropic changes.
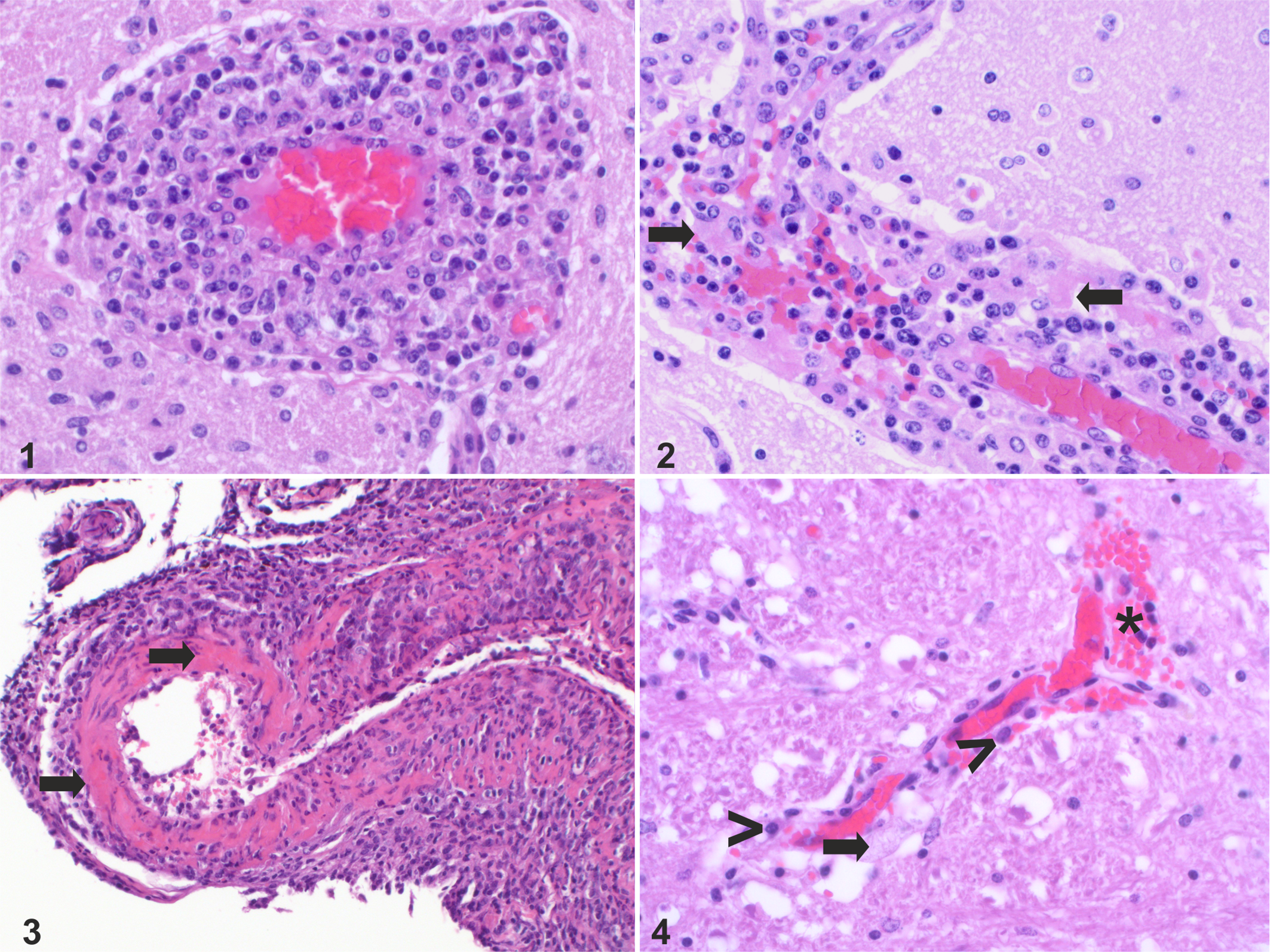
Ovine herpesvirus 2 infection, brain, cow. Hematoxylin and eosin.
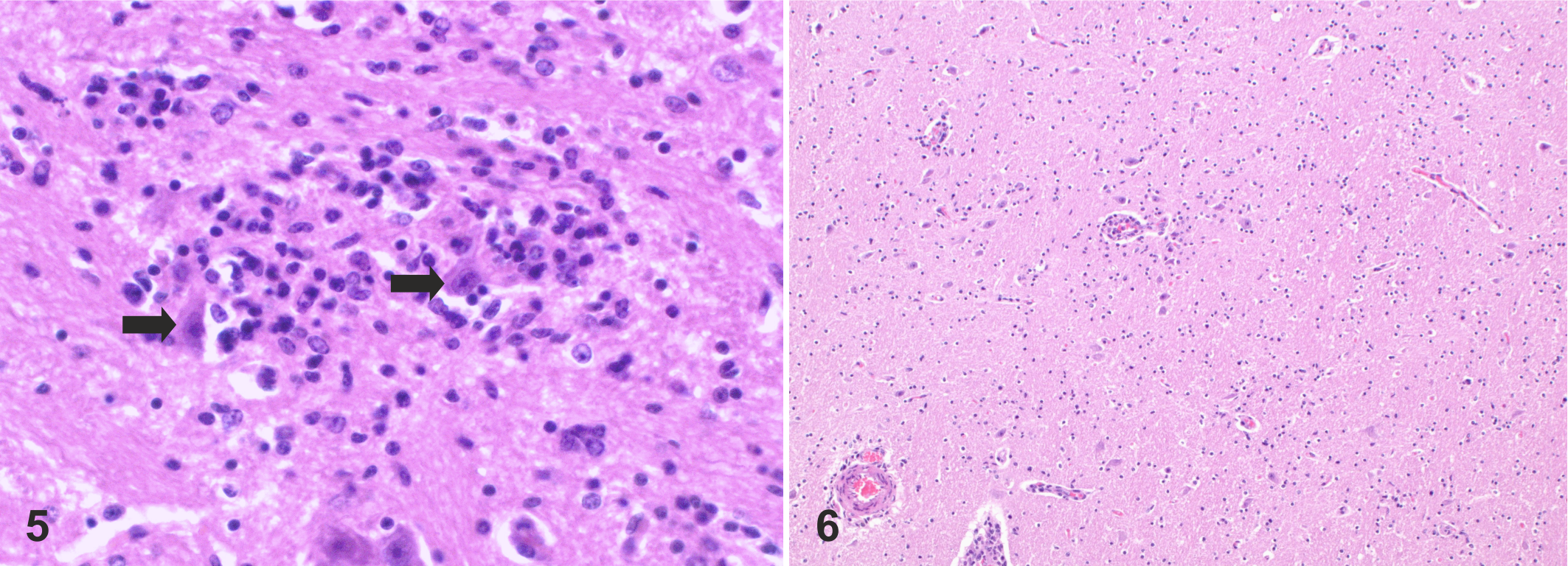
Ovine herpesvirus 2 infection, brain, cow. Hematoxylin and eosin.
Cq values of the OvHV-2-specific qPCR ranged between 17.6 and 39.5. Comparison of Cq values of the OvHV-2-specific qPCR with the severity of histopathological lesions in the brain showed low Cq values in animals with moderate and severe lesions (mean 27.0 and 23.4, respectively) and high Cq values in animals with mild, minimal, or no lesions (mean 35.7, 37.3, and 35.6, respectively; Table 3). Hence, the severity of brain lesions was related to viral genome copy numbers. Only one animal with moderate lesions exceeded a Cq value of 35. The Cq value was not available for one animal with severe lesions, as this animal was not tested in our laboratory.
Relationships Among Severity of Histologic Lesions, Cycle Quantification (Cq) Values for OvHV-2-Specific qPCR, and In Situ Hybridization (ISH) for OvHV-2 in 18 Cattle.
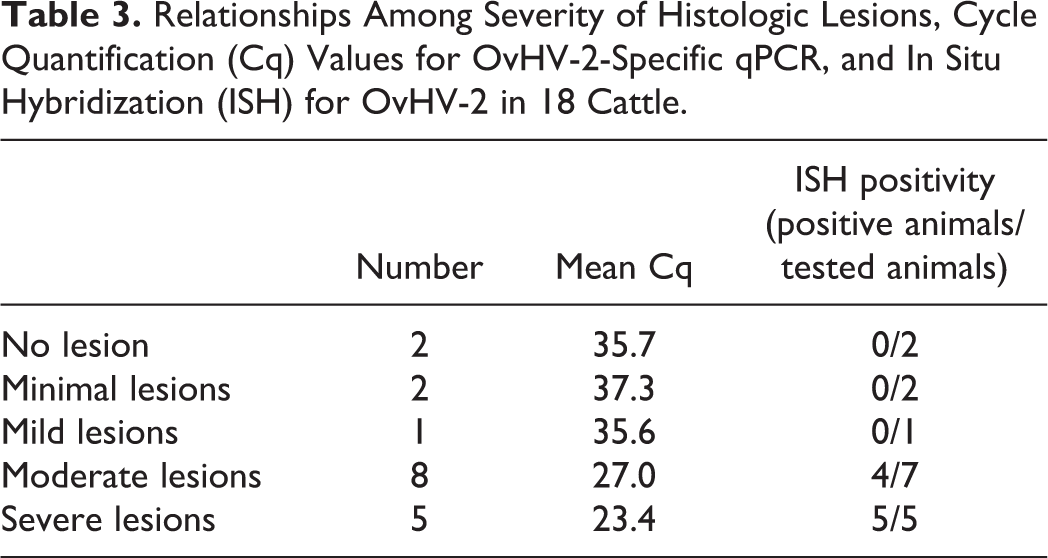
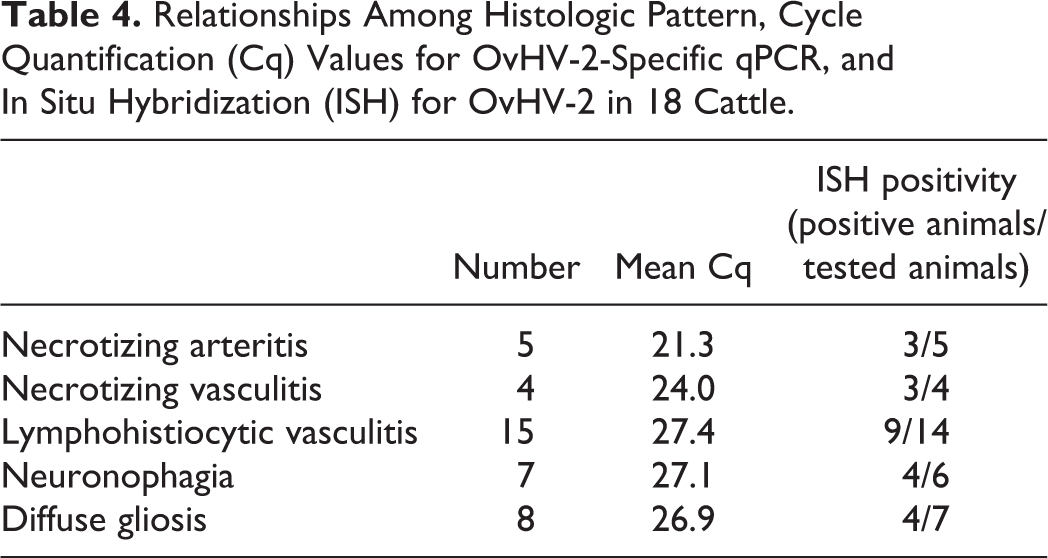
When comparing the Cq values of different histopathological categories, the lowest mean Cq value (21.3) was observed in animals with pathognomonic necrotizing arteritis (Table 4).
Relationships Among Histologic Pattern, Cycle Quantification (Cq) Values for OvHV-2-Specific qPCR, and In Situ Hybridization (ISH) for OvHV-2 in 18 Cattle.
Using ISH, OvHV-2 was detected in FFPE tissue slides of 9/17 of tested animals (53%). Positive signal consisted of predominantly intranuclear brown granules and was mainly seen in leukocytes of perivascular cuffs. Occasionally, in animals with a strong ISH signal in the brain, OvHV-2 nucleic acid was observed in neuroparenchymal cells with morphological characteristics compatible with lymphocytes either in the neighborhood of vessels or in areas of nodular gliosis (Figs. 7–10). Double staining with cell markers in one animal exhibiting neuronal damage confirmed the infected cells to be exclusively CD3+ T lymphocytes (Figs. 24–27). OvHV-2 nucleic acids were not observed in neurons or in Iba1+ microglia and macrophages. Interestingly, OvHV-2 was detected in situ in all 5 animals with severe lesions and in 4/7 with moderate lesions. In animals without lesions or with minimal and mild lesions, OvHV-2 nucleic acid was not detected by ISH. Still, 3/7 animals with moderate lesions remained negative in ISH (Table 3). In 1 of these 3 brain samples, the Cq value was also comparably high (36.8), whereas 2 of these 3 animals had very low Cq values (22 and 25.4; Table 1).
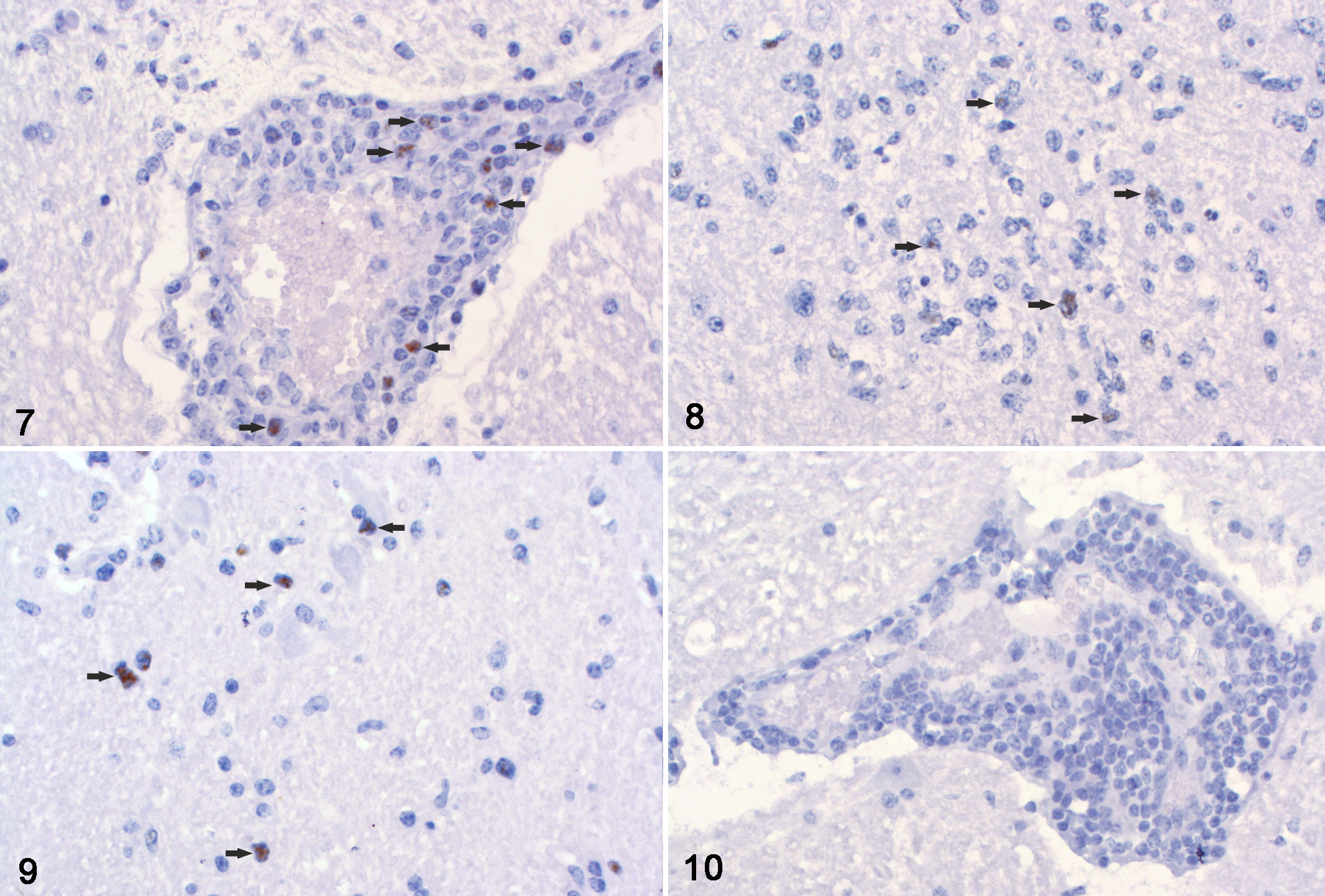
Ovine herpesvirus 2 (OvHV-2) infection, brain, cow. Chromogenic in situ hybridization using an OvHV-2-specific probe.
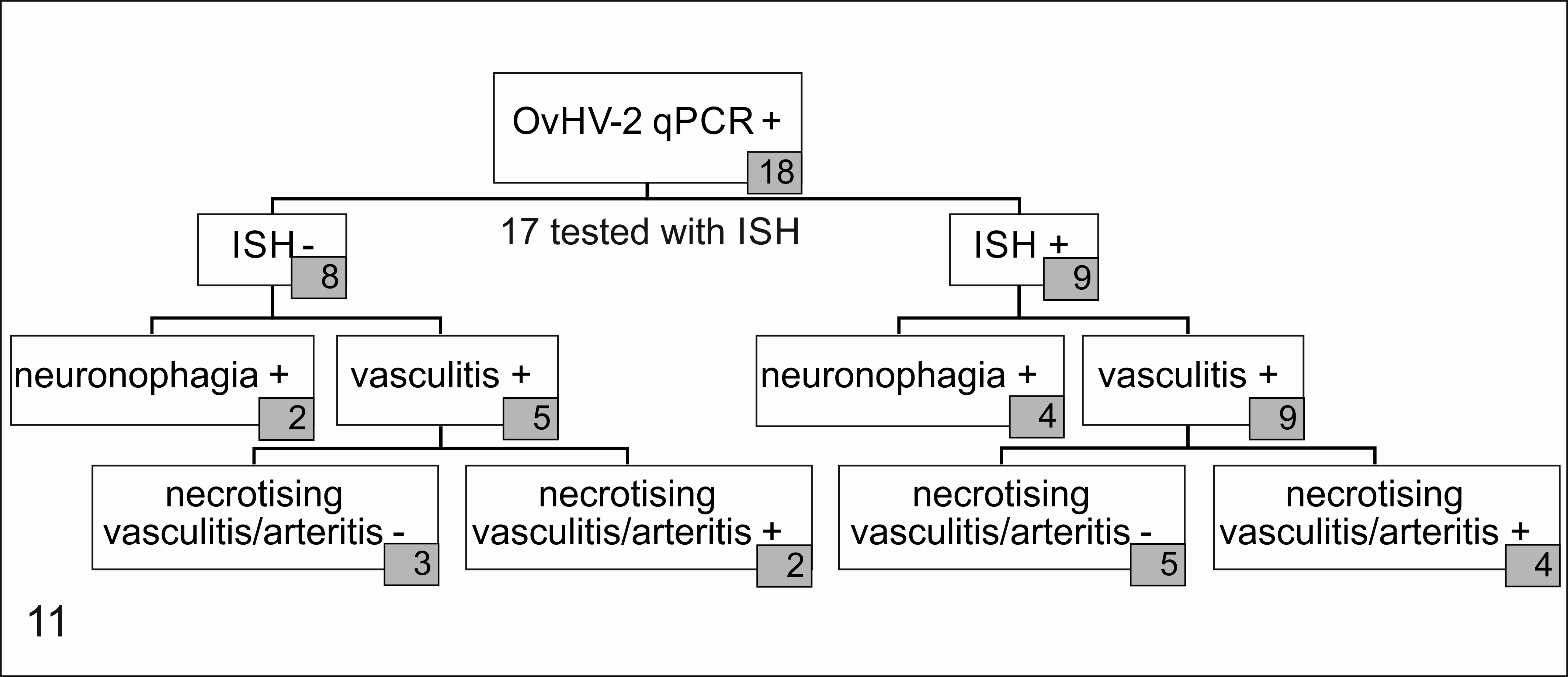
Frequencies of necrotizing arteritis/vasculitis and neuronophagia and the relationship to in situ hybridization (ISH) results in brain tissues of 18 cattle.
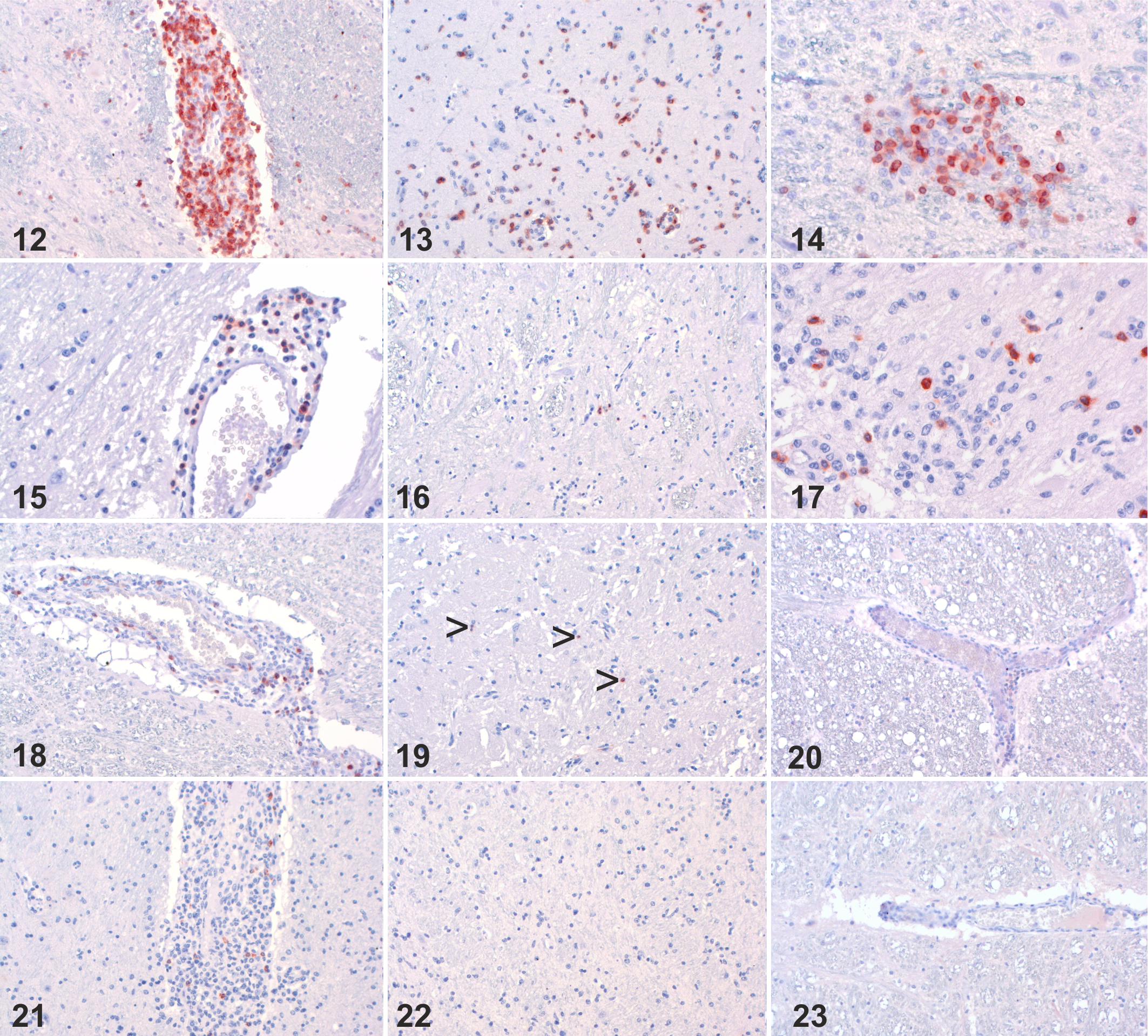
Ovine herpesvirus 2 (OvHV-2) infection, brain, cow. Immunohistochemistry for CD3 (Figures 12–20) and CD20 (Figures 21–23) (IHC).
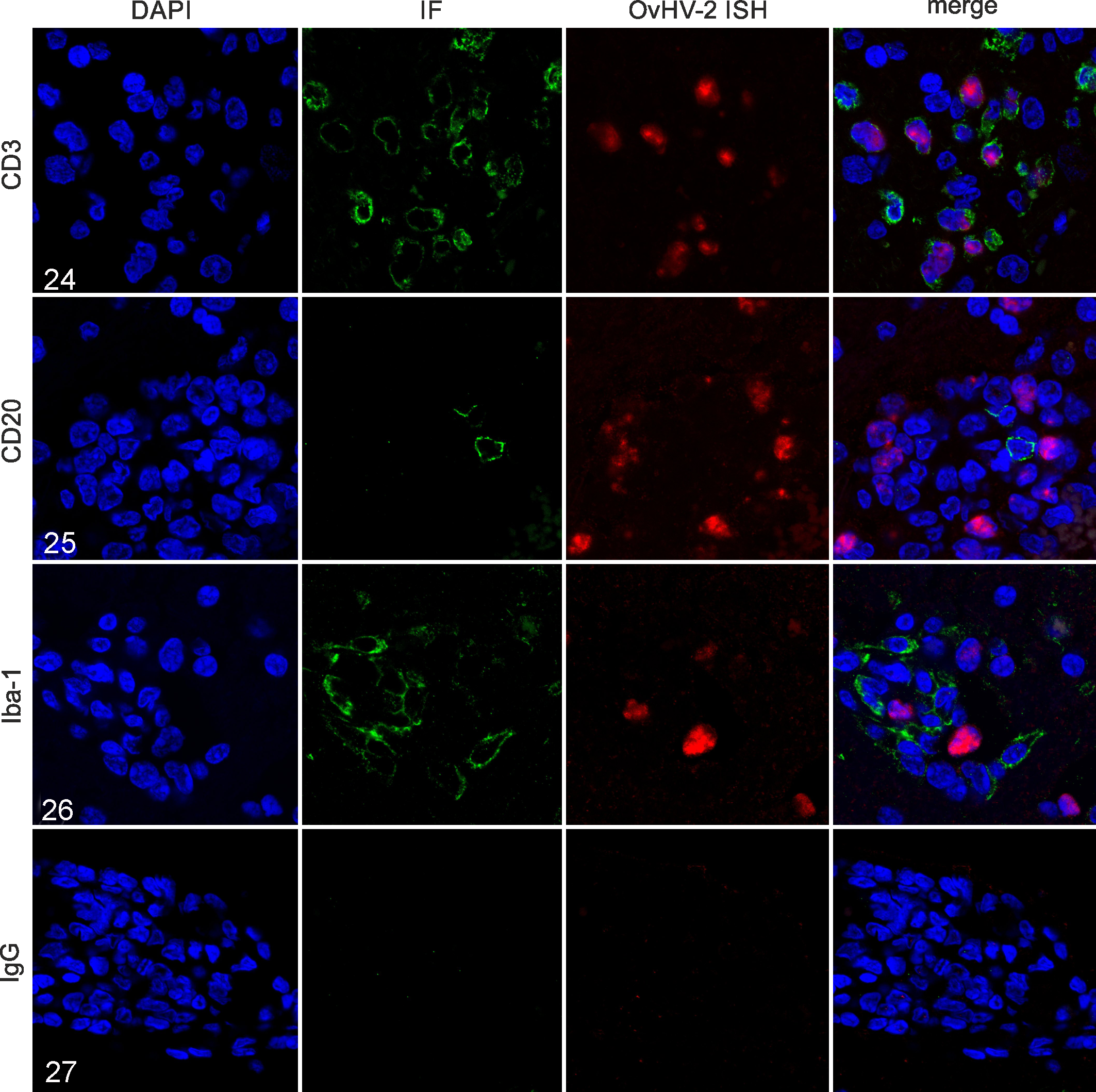
Ovine herpesvirus 2 (OvHV-2) infection, brain, cow. Combination of fluorescent OvHV-2 in situ hybridization (ISH) using an OvHV-2-specific probe, with immunofluorescence using different leukocyte markers or rabbit IgG as a negative control. Nuclei are stained blue (DAPI), OvHV-2 nucleic acid is stained red, and leukocyte markers are stained green.
All 9 animals with positive ISH results had a lymphohistiocytic vasculitis in the brain. However, necrotizing vasculitis and/or arteritis (ie, with fibrinoid vessel wall necrosis) was additionally observed in only 4/9 of these animals (Fig. 11). Of the 6 tested animals that showed signs of neuronal damage (ie, neuronal degeneration or necrosis, gliosis, and neuronophagia), OvHV-2 nucleic acid was detected by ISH in 4 and was absent in 2 (Fig. 11, Table 4). However, the ISH-negative animals with neuronal changes had Cq values >35 in the OvHV-2-specific qPCR and, hence, only had low quantities of OvHV-2 nucleic acid in the brain.
Overall, the quantity of ISH-positive cells varied substantially from a high density of positive cells to only scattered positive cells in perivascular cuffs. While the strongest ISH signals were observed in animals with severe lesions and low Cq values, some animals with severe brain lesions exhibited only few virus-infected cells in ISH.
The distribution of B and T lymphocytes was evaluated by IHC in all 6 animals with vascular and neuroparenchymal lesions in the brain (for which FFPE material was available) and in 3 animals with vascular lesions but without clear neuronal involvement (Fig. 12–23). In cases with low Cq values (ie, larger amounts of virus) and severe lesions including neuronal damage (
Combined ISH for OvHV-2 and immunofluorescence for leukocyte markers was done for one animal with neuroparenchymal brain lesions and low Cq value (Fig. 24-27). In this animal, infiltrating T lymphocytes were infected with OvHV-2 as revealed by simultaneous detection of OvHV-2 nucleic acid and CD3+ (Fig. 24). In contrast, neither B lymphocytes nor Iba-1-positive macrophages/microglia were infected with OvHV-2 (Figs. 25, 26).
Discussion
In this study, we compared the histopathology of 18 brain samples that were confirmed as OvHV-2 positive by qPCR. The phenotype and severity of histopathological lesions in the brain varied substantially among investigated animals, and no clear targeting of specific anatomic regions of the brain was observed. A pathognomonic necrotizing vasculitis and arteritis as described in textbooks 17,39 was missing in the majority of cases. Lymphohistiocytic vasculitis was by far the most frequent lesion pattern (15/18) and neuronophagia occurred frequently as well (7/18). Thus, MCF cannot be reliably diagnosed based on histology of the brain only, and confirmation by detection of either viral DNA in tissue and/or blood or specific antibodies in the serum is indicated when pathognomonic lesions are missing in the brain and full-body necropsy is not available. 24 “Head-only” submissions are practiced in BSE and rabies surveillance activities and, due to the sporadic character of MCF, diseased animals might be submitted as BSE or rabies suspect cases when no clear systemic and MCF-specific signs are detectable.
The observation of neuronal damage in the brain of several animals was somewhat puzzling. In 2 cases with neuronophagia, no vasculitis or only minimal vasculitis was observed. In most of the animals with neuronal damage, OvHV-2 nucleic acid was demonstrated by ISH. In cases in which OvHV-2 was not detected in situ, degradation of nucleic acids due to prolonged formalin fixation needs to be considered. 35 Investigation in one animal detected viral nucleic acid exclusively in CD3+ T cells, but not in CD20+ B lymphocytes, Iba-1+ microglia/macrophages, or in neurons. Although the final cause for neuronal damage and neuronophagia during OvHV-2 infection remains unclear, several mechanisms might be involved: (1) Co-infection with a yet-unknown neurotropic virus that was not detected by our methods. However, such a high rate of co-infections appears unlikely. (2) Misdirected activity of the immune response toward neurons due to epitope spreading, bystander effects, or molecular mimicry as described in betaherpesvirus infection. 14,34 Although it remains controversial whether or not dysregulation of uninfected cytotoxic T lymphocytes is involved in the pathogenesis of MCF, 18,30,31 autoimmunity could be suggested by the numerous CD3+ cells in glial nodules around neurons, of which few contain OvHV-2. (3) Latent neuronal OvHV-2 infection which remained undetected by our ISH technology. 11 Latency has been described in OvHV-2 infection 9,10,13,20 and latent neuronal infection occurs in the related gammaherpesvirus, Kaposi’s sarcoma virus. 38 However, undetected latent infection appears unlikely to be responsible for neuronal damage as latency is a viral mechanism for immune evasion with minimal protein expression and is unlikely to be associated with an inflammatory reaction by the host. 16
Brain lesions were present in 89% of OvHV-2 positive animals, which is consistent with previous studies that report central nervous system lesions in up to 97% of cattle with confirmed MCF. 22,33 Interestingly though, 2 animals, in which the clinical and neurological status was unknown, did not exhibit any histological changes in the brain. Therefore, it remains unclear how to interpret PCR-confirmed cases of MCF without histological involvement of the brain. Despite the high fatality rate of MCF, cows rarely may recover from MCF infection and remain chronically PCR-positive or might be subclinically infected. Recovery from and subclinical infection with OvHV-2 was found more frequently than previously assumed, especially in areas with high MCF prevalence. 19,26,29 Therefore, a positive OvHV-2 PCR result may not be relevant for the diagnosis in these animals. Whether the 2 animals without cerebral lesions were persistently infected, however, remains unknown. Indeed, both animals had very high Cq values in the brain suggesting absence of active viral replication and thus potentially indicating a persistent infection, which was further supported by negative ISH results. However, only the medulla oblongata of both animals was available for histological examination, and therefore, it cannot be excluded that lesions affecting other brain regions were missed. The same holds true for other organs as no full-body necropsies were performed, which makes a definite conclusion on the disease status and the cause of death of the animals impossible. Additionally, it cannot be fully excluded that qPCR was falsely positive in these 2 animals, for example, due to contamination in the lab, as Cq values were above 35 and ISH did not detect any OvHV-2 nucleic acid. This is also valid for cases with inflammatory lesions but high Cq values and lack of ISH detection. Even though a linear increase of Cq values was detected up to cycle 40 in 5-fold dilution series of a strongly positive OvHV-2 sample in one of our previous studies, 40 extensive validations of sensitivity and specificity of the qPCR have not yet been performed.
In conclusion, we identified a variety of neuropathological changes in MCF in cattle. MCF may mimic neurotropic viral infection, while pathognomonic necrotizing arteritis may be absent. Hence, validated molecular tests are warranted for the diagnosis of OvHV-2 in nonsuppurative encephalitis without pathognomonic vascular lesions. At the same time, we demonstrated that in situ detection of OvHV-2 with ISH on the brain is inconsistent and should be supported by PCR results. In one animal with neuroparenchymal brain lesions, OvHV-2 was exclusively present in CD3+ T lymphocytes, but not in neurons or microglia. At the same time, increased infiltration of CD3+ T lymphocytes in perivascular cuffs, brain parenchyma, and glial nodules was associated with neuronal damage and low Cq values. These observations suggest molecular mimicry as a possible pathomechanism of MCF in the brain and encourage further research regarding potential cross-reactive viral proteins.
Footnotes
Acknowledgements
We would like to acknowledge the veterinarians for submission of bovine brains within the framework of neurological disease surveillance. We are grateful for the technical support by Elsbeth Keller-Gautschi and Camille Monney.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by the Federal Food Safety and Veterinary Office.
