Abstract
Naked mole-rats (NMRs) are common in the managed care of zoos and valuable models for aging research. Limited information on NMR neuropathology is available despite many studies regarding their aging physiology. Histologic sections of brain from 27 adult (5–27 years old) NMRs from 2 zoos were reviewed to determine presence or absence of lesions associated with advanced age in humans and other mammals. A majority (23/27; 85%) of NMR brains had cerebral cortical neuronal changes with rounded or angular neurons, cytoplasmic vacuoles containing pale yellow pigment, periodic acid–Schiff (PAS)-positive granules and green autofluorescence, compatible with lipofuscinosis. Less severe lesions were present in cerebellar Purkinje cells, medulla, and hippocampal neurons. The hypothalamic neuropil of all NMRs had scattered variably sized PAS-positive granules and 10 (37%) had larger round bodies consistent with corpora amylacea. The youngest NMRs, 5 to 7 years old, generally had minimal or no cerebrocortical lesions. Further studies will help understand brain aging in this long-lived species.
Keywords
Naked mole-rats (
In addition to serving their use as animal models, NMRs are commonly housed in managed care within zoological institutions where individuals carry out eusocial behavior within colonies in seminatural environments as they age. Retrospective evaluations of postmortem data from zoo-housed NMRs have documented common lesions and age-related diseases as well as the initial reports of cancer in this species. 7,8,22 However, there is a paucity of information regarding spontaneous central nervous system lesions of NMRs. Although efforts to standardize necropsy protocols for NMRs in zoological settings are ongoing, there is substantial variability in quality and completeness of brain sections in archival case materials. Furthermore, the presence of common, age-related changes such as neuronal lipofuscinosis, if not severe, is inconsistently reported since it is found in many aged zoo animals. The goal of this study was to determine if NMRs have any brain lesions that correlate to those found in aged humans, laboratory rodents, and other animal species.
Histologic sections of brains from 27 adult (16 male and 11 female) worker (nonbreeding) NMRs housed at 2 zoological institutions were evaluated, and identified changes were compared with neurodegenerative and other age-related changes reported in humans and other animal species, including laboratory rodents.
18,25
For inclusion in the study, histologic sections from formalin-fixed, paraffin-embedded samples of the cerebral cortex, hippocampus, thalamus, medulla, and cerebellum had to be available. Evaluation was standardized in reference to the brain atlas of the naked mole-rat
24
and of a comparable species, the African mole-rat (
In evaluation of hematoxylin and eosin (H&E)-stained sections, most NMR brains (23/27; 85%) had a mild to moderate degree of cerebral cortical neuronal changes with small, angular or rounded neurons, cytoplasmic vacuoles (some containing pale yellowish pigment), dispersed Nissl substance, and irregular hyperchromatic nuclei. The lesions spanned the outer and inner layers of the cerebral cortex (Figs. 1 –3), often more severe in the inner zones but without specificity to identifiable cortical anatomical regions. Other cortical neurons appeared normal. In several brains, dark neuron artifact was found focally extending from the cortical surface and outer to inner cortical neuronal layers. The affected neurons were distinguished from dark neuron artifact based on their location (middle and inner cortices, distant from cortical surfaces), distribution, involvement of individual neurons, and heterogeneity of changes in nearby neurons. 14 Affected neurons (without dark neuron artifact) had numerous PAS-positive intracytoplasmic granules (Fig. 4), which were often acid-fast, and usually exhibited green autofluorescence (wavelength range: 470 nm excitation, 525 nm emission) confirming lipofuscin accumulation (Fig. 5). Compared to cerebral cortical neurons, those in the hippocampus, thalamus, medulla oblongata, and cerebellar Purkinje cell layer had much fewer PAS-positive granules and less autofluorescence. Most (4/7) of the younger (2–7 year old) NMRs had no or minimal neuronal changes, including autofluorescence, in the cerebral cortex or elsewhere (Fig. 6).
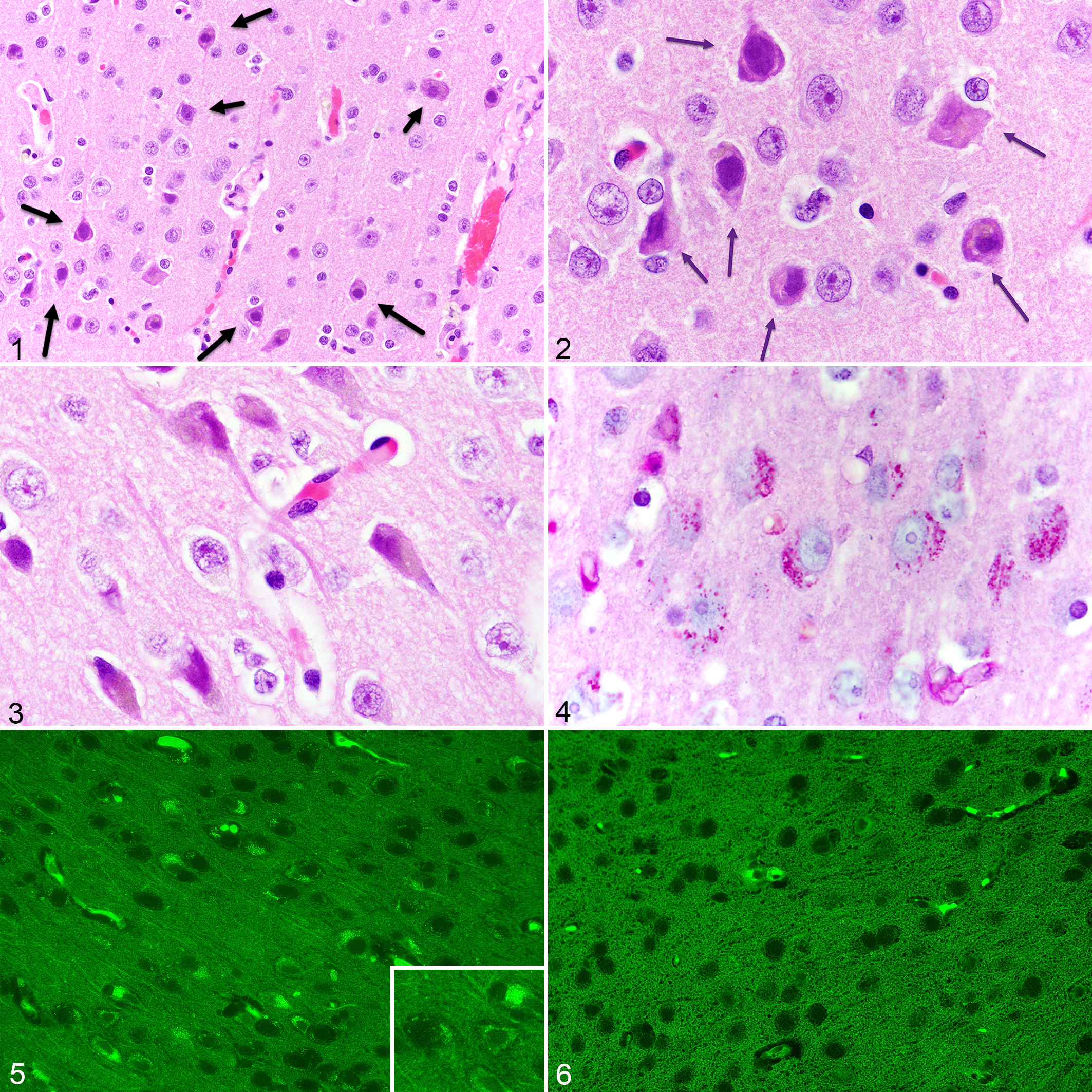
Cerebral cortex of aged naked mole-rats.
In HE-stained sections, the periventricular and paraventricular hypothalamic nuclei of 10 (37%) NMRs contained multiple extracellular variably sized (up to 20 µm diameter), round, amphophilic to grey, glassy to dull, globular bodies (Fig. 7). These were PAS-positive (Figs. 8, 9) and reminiscent of polyglucosan bodies or corpora amylacea. 2,5 Also present in 26/27 (96%) NMRs (including the youngest NMR), few to numerous, 2 to 10 µm diameter, intensely PAS-positive, extracellular granules were scattered throughout the neuropil but not seen in HE-stained sections (Figs. 8, 9). In both HE and PAS stains, the bodies had a substructure reminiscent of a soccer ball. Corpora amylacea and granules were faintly positive (violet) with cresyl violet acetate and had metachromatic (pale purple to violet) staining with toluidine blue (Fig. 10), but were not acid-fast or autofluorescent. Similar PAS-positive granules were also found in in the lateral hypothalamus and globus pallidus in a few NMRs but most sections did not include these areas anatomic structures. There was no evidence of amyloid plaques or neurofibrillary tangles in the HE-stained histologic sections of NMR brains, in contrast to other aging animal models 25 and like previous reports investigating NMR neurodegenerative pathophysiology. 11,12
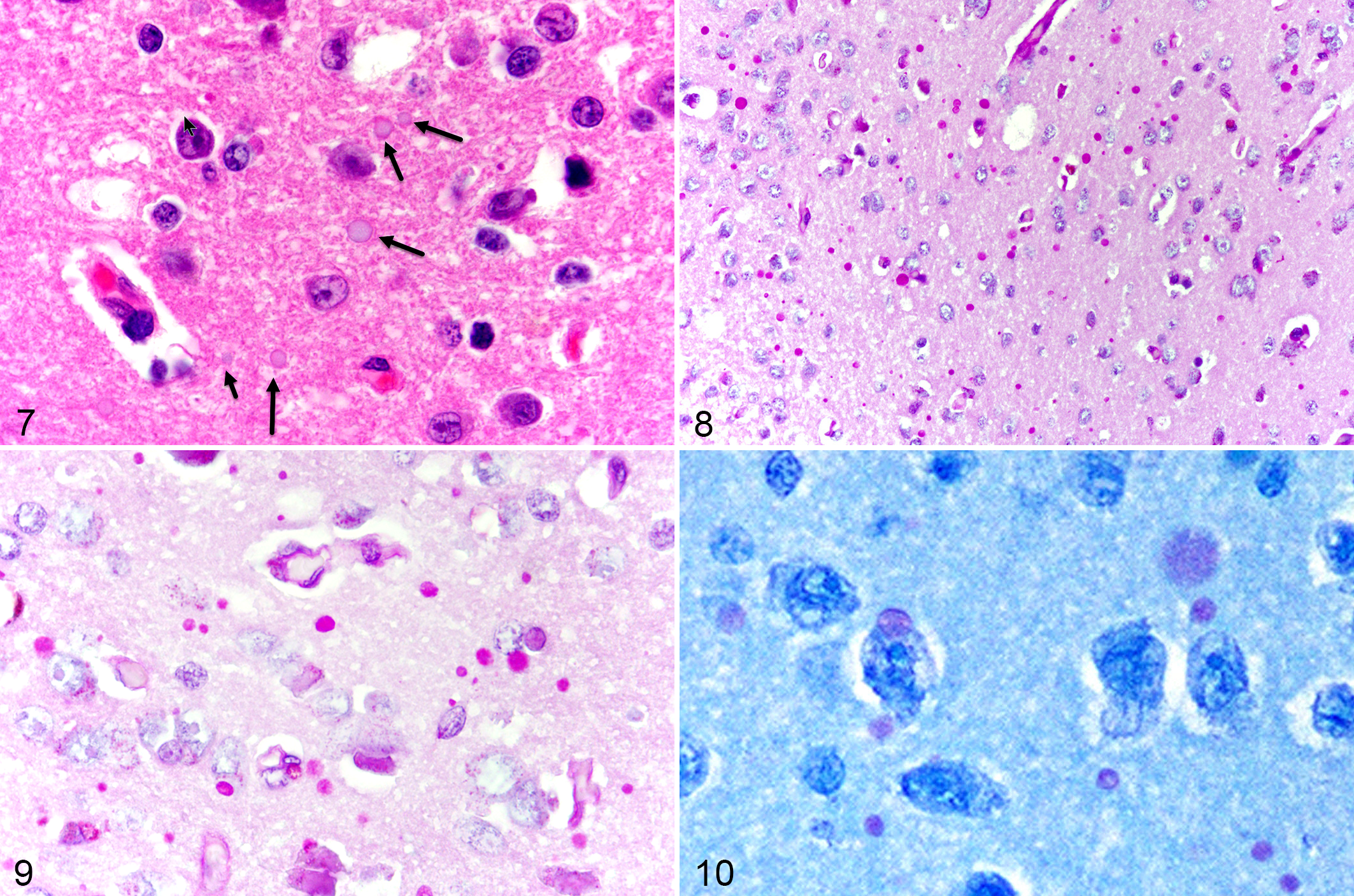
Hypothalamus, aged naked mole-rats.
From this small retrospective analysis of zoo-housed NMRs, it is evident that these long-lived rodents develop histologic changes reminiscent of those found in humans and animals of advanced age and also occurring in some neurodegenerative diseases. 5,17,18,25 Though exact ages of some of these individuals were not known, this descriptive report includes the first documentation of autofluorescent neuronal granules and corpora amylacea in NMR brains. Furthermore, the distribution, affected cell types, and frequency of lipofuscin granules were important findings considering the recognized differences among humans, nonhuman primates, and other animals, and the potential utility of NMRs as models for neurobiology-focused aging studies. 15,16,18,19
Neuronal granules have been reported in the brains of laboratory rodents and are often considered incidental but age-related findings in controlled studies. 21,25 These granules encompass several different types of degradation products and are often biomarkers for neurodegenerative lesions like amyloid plaques and neurofibrillary tangles. 16 Periodic acid–Schiff-positive, autofluorescent neuronal granules were found in a large percentage (96%) of the examined NMR brains of varying ages, strongly suggestive of lipofuscin accumulation. Lipofuscinosis has been previously reported in NMRs specifically in neurons, adrenal cortical cells, and cardiomyocytes, 7,10 though neither of these studies utilized PAS reaction or assessed pigments for autofluorescence. Lipofuscin may be caused by lipid peroxidation that occurs during cellular metabolism and accumulates with age. 18 Old laboratory rats have lipofuscin in different regions of the brain with the highest rate of accumulation in the hippocampus and lesser in the cerebral cortex and thalamus. 19 Unlike in rats, cortical PAS-positive neuronal granules in aged mice are not typically autofluorescent, including those found in Purkinje cells and spinal neurons. 16,21 Like humans and chimpanzees and in contrast to old lab rats, these NMRs had very little evident lipofuscin accumulation in Purkinje cells, suggesting a possible neuroprotective mechanism in this brain region. 15,19 As such, NMRs have been shown to have neuronal changes not necessarily correlated with reported high levels of oxidative damage and accumulation of amyloid-β. 11,12 Lipofuscin accumulation may serve as a correlate to other oxidative stress markers and can be analyzed within the brain in situ. 1,12
An unexpected finding was the presence of presumed polyglucosan bodies or corpora amylacea in the hypothalamus of 37% of examined NMRs based on routinely stained sections. PBGs are inclusions within neurons, axons, or glia (particularly astrocytes) that are composed of glycoproteins and other yet unknown components. 2,5 Polyglucosan bodies are common in aged humans and animals and are typically referred to as corpora amylacea when found in individuals lacking other signs of neurodegenerative disease or when found in certain cell types. 2,5,17,25 Corpora amylacea are distinguished from other types of polyglucosan bodies found in genetic neurologic diseases, like Lafora bodies, based on distribution, affected cell types, staining and immunostaining characteristics. 2,5,17 However, the significance of corpora amylacea in the examined NMRs is unclear, because these bodies are age-associated but not associated with other degenerative lesions. Immunohistochemical evaluation to assess for corpora amylacea and other polyglucosan bodies is recommended in future neuropathology-based aging studies.
To the authors’ knowledge, this is the first detailed examination of histologic age-related changes in the brains of NMRs. Prospectively, efforts should be made to achieve optimal coronal sections of well-preserved brain tissue by reflecting the skull cap prior to immersion fixation. Brain removal after fixation will help avoid excessive handling and dark neuron artifact. In most cases, PAS reaction was the most useful to detect and better evaluate the distribution of neuronal and glial cytoplasmic granules and corpora amylacea. Furthermore, assessing routinely stained brain sections for autofluorescence was worthwhile to determine extent of lipofuscin accumulation. Based on these findings, complete and standardized histologic evaluation of the brain should be considered when performing postmortem examinations of NMRs. Future controlled studies incorporating histologic changes in brain tissues utilizing tissue markers of neurodegeneration, lipofuscin accumulation, and oxidative damage will provide valuable insights of the aging brain in this long-lived rodent.
Footnotes
Acknowledgements
The authors would like to thank Renee Walker and staff of the University of Illinois Veterinary Diagnostic Laboratory Histology Lab for slide preparation, special staining and immunohistochemistry. We also thank Kenton Kerns of The National Zoo for providing colony information, Dr. Bob Garman for invaluable histopathology consultation, Drs. Cristy Rocio Gonzalez Barrientos and Thomas Wilhelm Westermann for their valuable comments, Drs. Shiko Kimura and Henrique Dias for help with the fluorescence microscope, Drs. Kali Holder, Neel Aziz, and Tim Walsh for pathology support and other Zoological Pathology Program pathologists and residents for initial postmortem evaluations. Thanks to Dr. Jaime Landolfi for reviewing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
