Abstract
The naked mole-rat (NMR;
Naked mole-rats (NMRs;
As a eusocial species, NMRs live in large colonies with a division of labor into reproducers (breeders), including a single queen and 1 to 3 breeding males, and subordinate workers. 7,8,11,18,30 Workers are typically smaller and functionally sterile, and they reportedly have shorter life spans than do breeders, as they are subject to different predation risks. 13,18,20,30 After 6 months of age, subordinate NMRs are capable of becoming breeders and will fight to establish dominance and breeding status if the current queen dies, often resulting in fatal injuries. 18,19,31 NMRs are also highly xenophobic and will kill foreign individuals or conspecifics under certain circumstances. 1,9,17 –19 Due to their interesting appearance and natural history, NMRs have been popular zoo exhibit animals since the 1980s. Husbandry practices for NMRs have evolved over the years; however, veterinary care opportunities remain limited due to their eusociality and the inability to return individuals to the colony following diagnostic procedures or treatments. 17,19 Furthermore, individual identification of zoo-housed NMRs is generally not part of husbandry protocols, as it is difficult in the seminatural, large colonies found in zoologic settings. 17,19
In a retrospective study, necropsy and histopathologic data from 138 adult NMRs from 8 separate colonies housed at a zoologic institution were reviewed and common lesions tallied and described.
19
Interestingly, histologic changes in the kidney were prevalent in this cohort (127 individuals; 92%). Of these renal lesions, 57% were considered part of a chronic degenerative process and referred to as
Renal disease is a common finding in mice and rats of long-term toxicology and aging studies.
10,21,24,27,36
–39
In fact, some spontaneous renal diseases of aged rodents are utilized as models of human renal disease.
48
CPN or old rat nephropathy is a disease phenomenon described with high frequency in control rats of carcinogenicity studies and is most prevalent in old (>12 month) male Sprague-Dawley and F344 rats.
21,22,24,27,37,38
Similar to CPN in rats, there are typical age-related renal lesions in various strains of inbred and noninbred mice (termed
Materials and Methods
As described in our previous retrospective study of zoo-housed NMRs, 19 gross necropsy and histopathology reports were reviewed from 138 adult dead or euthanized NMRs from May 1996 to July 2011. Following review of representative cases of those NMRs with renal lesions, criteria were developed to calibrate pathology nomenclature, microscopic diagnoses, and interpretation of findings. Specific histologic changes and diagnoses of the kidney were tallied for males, females, and queens. Unless otherwise noted, tissues sections were fixed in 10% neutral buffered formalin, paraffin embedded, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Additional representative sections of kidney representing mild, moderate, and severe cases were further evaluated following staining with Jones methenamine silver (JMS), periodic acid–Schiff (PAS), and Masson trichrome (MT) to better evaluate glomerular (JMS and PAS) and fibrotic (MT) changes. Furthermore, Prussian blue staining was completed in rare cases to determine iron composition of pigments. A classic case of rat CPN was used as a comparison for NMR renal histologic lesions (Suppl. Figs. 1–6).
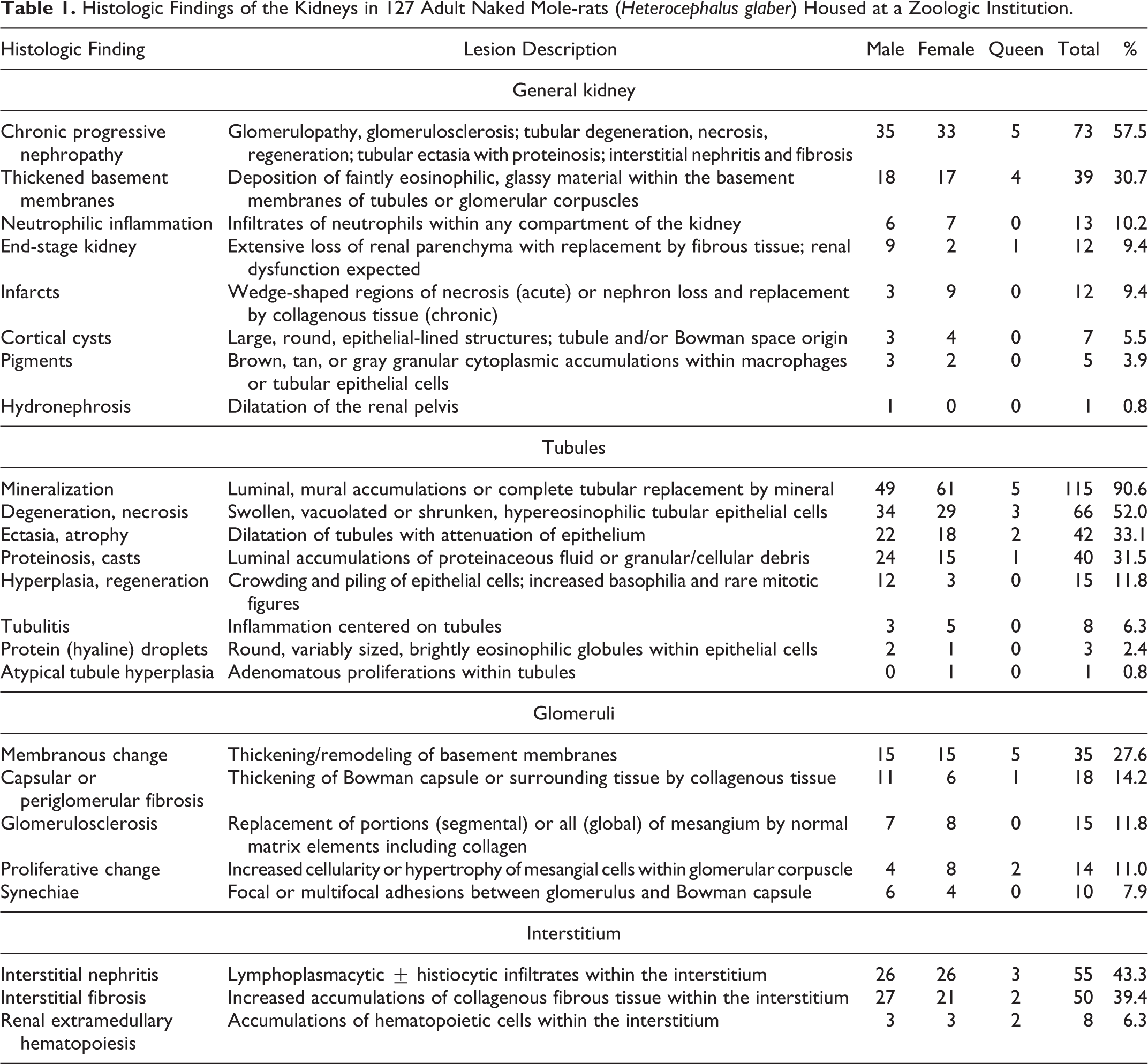
Histologic findings were grouped into general diagnoses and specific microscopic changes based on compartment of the kidney: general (>1 compartment affected), tubules, glomeruli, and interstitium (Table 1). General kidney lesions included chronic nephritis/CPN, thickened basement membranes, neutrophilic inflammation, end-stage kidney disease, infarcts, cortical cysts, pigments, and hydronephrosis. Tubular lesions included degeneration/necrosis, atrophy/ectasia, proteinosis/casts, hyperplasia, epithelial protein droplets, and tubulitis. Glomerular lesions included glomerular membranous change, glomerular proliferative change, periglomerular/capsular fibrosis, synechiae, and glomerulosclerosis. Interstitial lesions included interstitial nephritis, interstitial fibrosis, and extramedullary hematopoiesis.
Histologic Findings of the Kidneys in 127 Adult Naked Mole-rats (
Results
During a 15-year period, 138 zoo-housed adult NMRs that were found dead (
At necropsy, grossly evident renal disease was reported in 16.7% of NMRs, although histologic changes were found in 127 of 138 (92%). Of these 127 individuals, 71 (55.9%) were female (including 7 queens), and 56 (44.1%) were male. Incidences of individual lesions and diagnoses were compared between sexes (females including queens) via a risk ratio (ie, relative risk) analysis, and there was no evident sex predilection. In addition, there was no apparent sex bias regarding severity of renal disease. In the approximately 17% of NMRs with gross renal disease, affected kidneys were diffusely pale tan and either slightly enlarged or shrunken with multiple pinpoint to 2-mm-diameter cystic structures (microcysts) and/or an irregular knobby to granular cortical surface. Those NMRs with gross lesions had correlative histologic changes. Following microscopic evaluation, an additional 40% of NMRs that lacked grossly evident disease had lesions of varying severity, which in combination led to a diagnosis of chronic nephropathy.
Secondary lesions classically associated with renal disease—such as uremic gastropathy or glossitis, parathyroid hyperplasia, and fibrous osteodystrophy—were not found. Mineralization of soft tissues and/or parenchymal organs was reported in 25 (19.7%) NMRs with renal lesions. Of these, 13 had diagnosed calcinosis cutis, which in many cases, was attributed to dietary administration of vitamin D
19
: 10 were male and 3 were female, including 2 queens, although only 1 was reported to have end-stage renal disease. In addition to cutaneous mineralization, some of these NMRs had multicentric mineralization affecting great vessels (
General Kidney
Over 57% of NMR kidneys had a range of concurrent and presumably related histologic changes, which in combination led to the diagnosis of CPN or chronic nephritis (Figs. 1–18). Nephropathy severity ranged from mild (Fig. 1) to severe (Fig. 3). Lesions included many, but not always all, of the following: thickened basement membranes of glomerular corpuscles and tubules; membranous and/or proliferative glomerular change with rare synechiae, sclerosis, and obsolescence and limited periglomerular fibrosis; tubular ectasia and proteinosis with casts and cyst formation; tubular degeneration, necrosis, regeneration, and hyperplasia; interstitial mixed accumulations of lymphocytes, plasma cells, fewer macrophages and neutrophils; variable hemorrhage; and interstitial fibrosis, which was best appreciated with MT stain (Figs. 2, 4). Of the 7 queens, 5 (71.4%) had renal lesions compatible with chronic nephropathy.
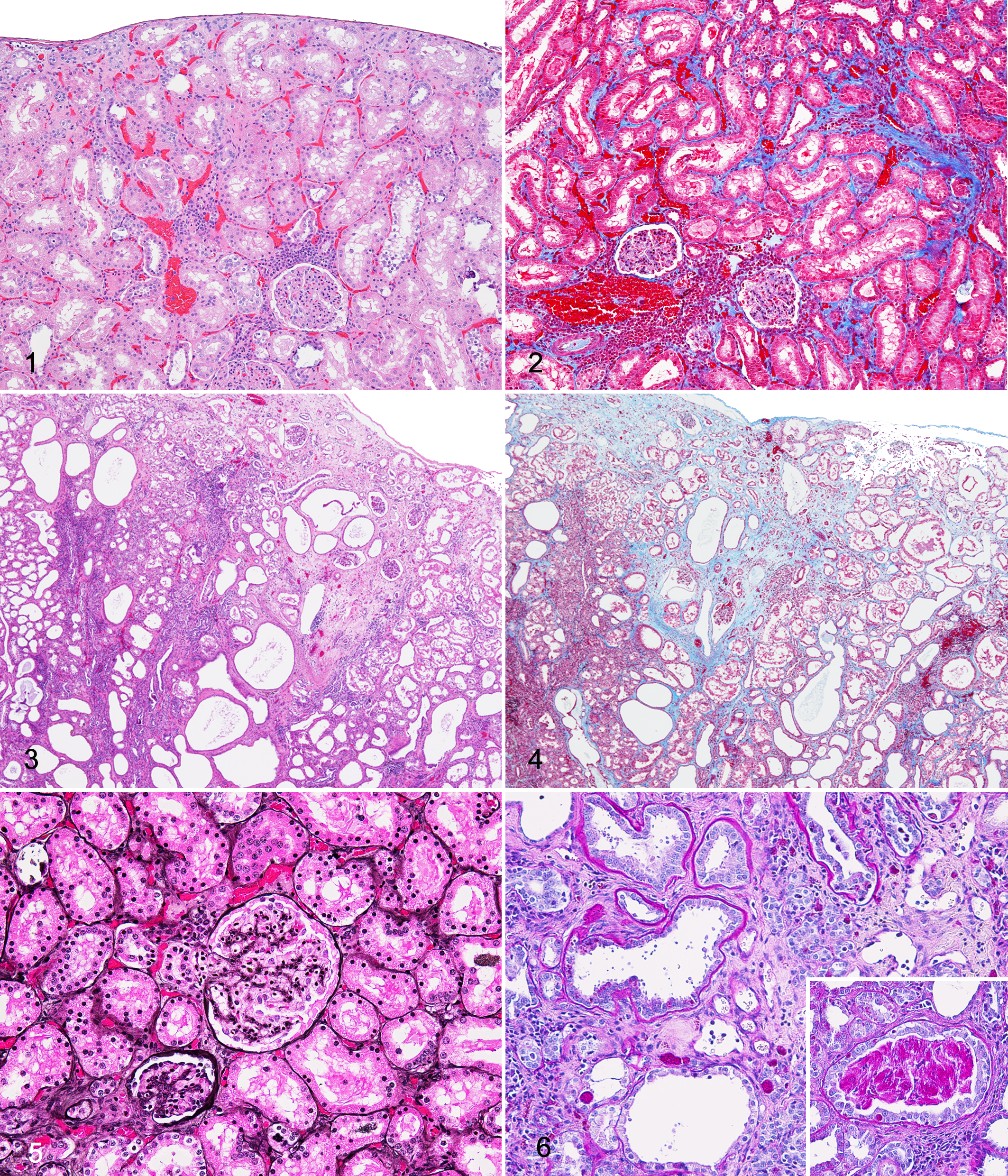
Several cases (9.4%) had lesions compatible with end stage renal disease, in which large portions of the functional parenchyma were severely compromised or lost and replaced by fibrous tissue with inflammatory infiltrates. All of these NMRs were those also diagnosed with severe CPN. Twelve (9.4%) NMRs had evidence of generally chronic infarction characterized as well-demarcated, wedge-shaped regions of fibrosis and loss of nephrons, although not all NMRs with infarcts had significant nephropathy (Figs. 3, 4).
Approximately 30% of the NMRs had kidneys with evident thickening of basement membranes of the glomeruli (Figs. 13, 18) and/or the tubules (Fig. 9). Though predominantly noted in the glomeruli in HE-stained sections, thickened basement membranes were better visualized with JMS and PAS stains (Figs. 5, 6, 14, 15).
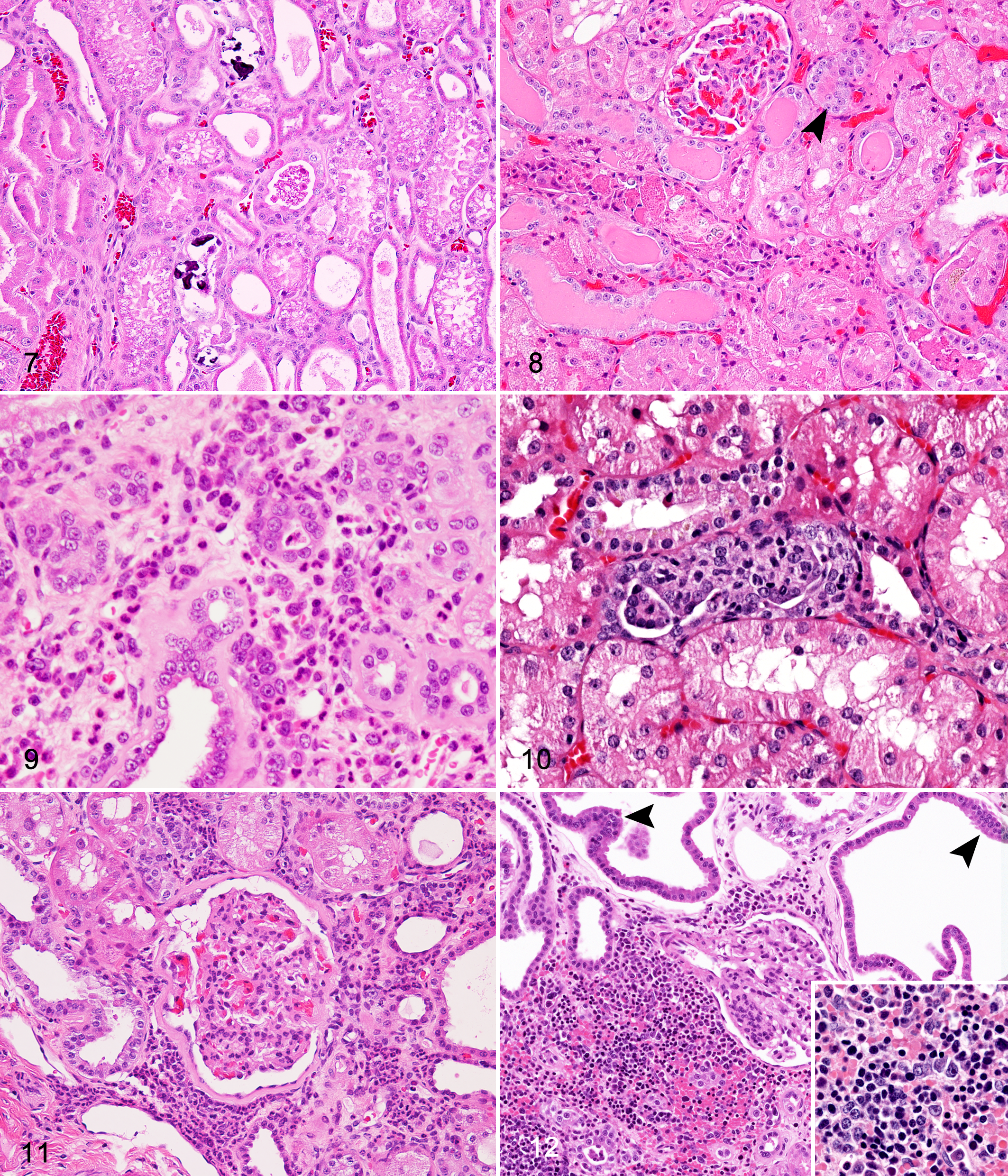
Roughly 10% of NMRs had neutrophilic inflammation reported as a component of their renal disease. A majority of the cases had overall low to moderate numbers of neutrophils mixed with interstitial mononuclear infiltrates, and fewer had mild neutrophilic tubulitis, possibly associated with renal tubular necrosis (Figs. 9, 11). Three NMRs had evidence of embolic nephritis associated with septicemia; these animals had necrosuppurative inflammation.
Cortical cysts were described in about 5% NMRs and were limited to the cortex. The cysts were composed of large, round, thin-walled structures lined by attenuated epithelial cells with variable luminal fluid and debris. The origin of the cysts was not determined, although they were considered to have developed from severely dilated tubules or Bowman capsules. Large cortical cysts could be seen grossly and were often associated with more severe and chronic cases of CPN.
Macrophages (Fig. 9) and renal tubular epithelial cells both had cytoplasmic pigments, which were tan-gray and/or golden to dark brown and granular (Figs. 8, 10). In general, pigments of tubular cells were consistent with lipofuscin, and negative Prussian blue staining was supportive. Cytoplasmic material was better visualized in PAS-stained sections, and it is possible that overall pigment accumulation was underreported upon routine (HE) evaluation (Fig. 6). Interstitial macrophages and few tubular epithelial cells had positive Prussian blue staining, indicative of hemosiderin. Hemosiderin-laden cells were most commonly described in NMRs with more severe interstitial nephritis, in which hemorrhage was a component, as well as in cases of chronic renal infarction.
Hydronephrosis was a rare finding in these NMRs: it was reported in a single male with large renal cysts, suggesting some degree of postrenal obstruction, although there were no abnormalities reported in the urinary bladder or tract.
Tubules
Approximately 90% of NMRs had evidence of renal tubular mineralization; however, >28% lacked any additional kidney lesions. Tubular mineralization was characterized by luminal accumulations of deeply basophilic, granular, or fragmented angular material that often obscured tubular epithelium with maintenance of the basement membrane (Figs. 1, 7).
Tubular degeneration and necrosis were almost exclusively noted together and therefore considered to be related processes. Degeneration manifested as epithelial swelling, vacuolation, and pallor of the cytoplasm (Figs. 7, 8). Necrosis was evident by shrunken, hypereosinophilic, and karyorrhectic, pyknotic, or karyolytic epithelial cells with variable cell sloughing and associated cellular and granular casts (Fig. 8). Some affected tubules also had multifocally to segmentally denuded basement membranes or attenuation of remnant epithelial cells.
Tubular ectasia and atrophy were common (33.1%) and prominent features in those NMRs with CPN. Multifocally to regionally, tubules were moderately to severely dilated and frequently tortuous with attenuation of epithelium, and they contained luminal material that consisted of scant to abundant protein fluid (see next section) or cellular/granular debris (Figs. 3, 4, 6, 7, 11, 12). Tubular atrophy, typically seen concurrently with ectasia, was characterized by shrunken, often misshapen tubules (Figs. 5–9).
In many cases (31.5%), ectatic and lesser-affected renal tubules contained variably eosinophilic, homogenous material interpreted as proteinaceous fluid (Figs. 7, 8). Frequently noted in conjunction with protein fluid was clumped, granular to globular brightly eosinophilic (HE) material that was highly visible with PAS staining (Fig. 6, inset; Fig. 7). As a common counterpart to tubular degeneration and necrosis, granular or cellular casts were described as eosinophilic granular and amorphous accumulations with and without enmeshed karyorrhectic cellular debris, respectively (Figs. 5, 7, 8).
Renal tubular regeneration and hyperplasia were noted almost exclusively in those NMRs with nephropathy (11.8%). The 1 remaining case had interstitial fibrosis and renal tubular mineralization but lacked significant tubular degeneration and necrosis. Tubular regeneration and hyperplasia were often reported congruently and were associated with tubular injury, as evidenced by degeneration and necrosis, and interstitial fibrosis. Tubules considered to be undergoing regeneration had multifocally to segmental attenuation, hypertrophy, and hyperplasia of the epithelium (Figs. 7–9, 12). Tubular hyperplasia was indicated by increased basophilia of epithelial cells, which were typically enlarged (hypertrophy) and crowded with variable piling upon an intact basement membrane (Figs. 7–9, 12). Some hyperplastic tubules were tortuous or nodular. In select cases, these latter characteristics were bordering on a diagnosis of atypia, although mitoses and megalocytosis were rarely noted.
Reported previously in our review, 19 1 NMR with mild chronic nephropathy had a small nodule of atypical tubular hyperplasia composed of multiple papillary proliferations of haphazardly arranged, basophilic tubular epithelial cells that extended slightly beyond normal tubule borders, with no prominent basement membrane, mild atypia, and slight compression of adjacent parenchyma. This NMR had other rare atypical profiles of tubular epithelium with crowding, piling, papillary projections, and increased basophilia (Fig. 10).
As mentioned above, tubulitis was typically a neutrophilic process with luminal accumulations and disruption of the tubular epithelium by intact and degenerate neutrophils (Fig. 9). This was a fairly uncommon finding (6.3%) in this NMR cohort.
Hyaline or protein droplets were seen in 2.4% of cases, specifically in 2 males and 1 queen. In 2 of these cases, there were no reported glomerular lesions; instead, significant tubular injury was found, including degeneration, necrosis, and proteinosis. Upon PAS staining of select cases, renal tubular protein accumulations were pronounced and therefore likely underreported per routine HE staining (Fig. 6).
Glomeruli
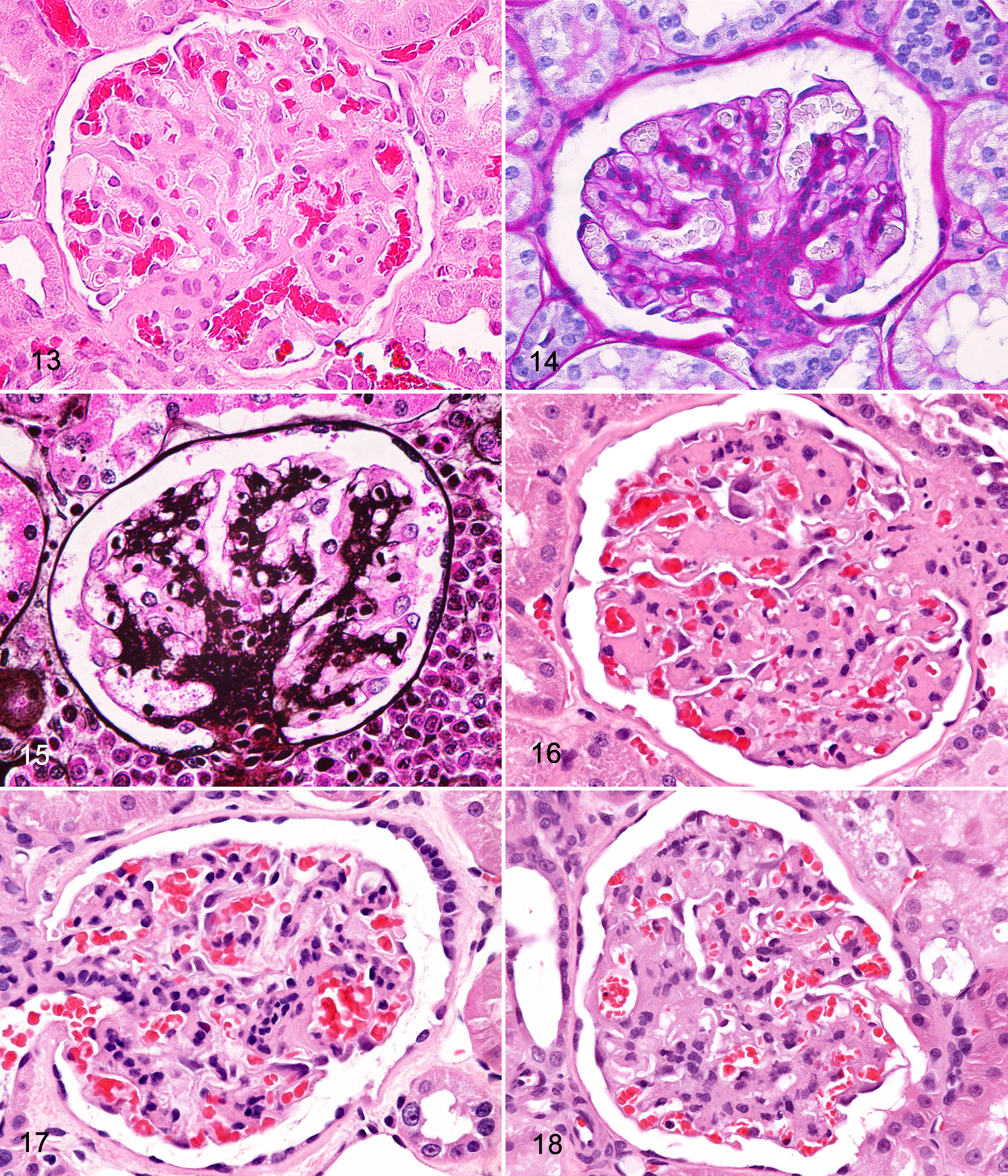
All glomerular changes were based on histologic evaluation of sections stained with HE except for those indicated. In this study,
Notably, proliferative change was seldom noted in the absence of membranous change; therefore, the more accurate diagnosis in these NMRs was considered membranoproliferative glomerulopathy. Affected glomeruli had segmental to global hypercellularity with enlarged, frequently crowded mesangial cells (Figs. 17, 18). Occasionally, inflammatory infiltrates were also reported and interpreted as glomerulonephritis, although, more often than not, infiltrates were seen concurrently with proliferative change and classified as 1 diagnosis in this study.
Glomerulosclerosis, noted in 11.8% of NMRs, was characterized by segmental to global replacement of corpuscles with mesangial matrix elements including collagen, the latter of which was referred to as
For most cases, fibrosis of the Bowman capsule and/or periglomerular tissue was found together with glomerulopathy and/or sclerosis (Figs. 11, 16, 17). In a few instances, capsular fibrosis was found in glomeruli with only thickened basement membranes and/or multifocal synechiae. Of these, interstitial fibrosis was commonly noted and appreciated on MT-stained sections.
Focal or multifocal adhesions of the glomerular visceral epithelium to the parietal epithelium of Bowman capsules were found in 8% of NMR kidneys. Generally, synechiae were associated with glomerulopathy (Fig. 18), sclerosis, and obsolescence, although in rare cases, these lesions were found in glomeruli with thickened basement membranes and capsular fibrosis only, without evidence of significant membranous, proliferative, or sclerotic change.
Interstitium
Interstitial nephritis was a frequent finding in these NMRs (43.3%). In these kidneys, the interstitium contained variable mononuclear infiltrates, predominantly lymphocytes, plasma cells, and macrophages, with less frequent neutrophils (Figs. 1, 3, 9, 11). This lesion was found almost exclusively in cases of CPN, although not all cases of CPN had significant interstitial nephritis. Severe nephropathy cases also had variable interstitial hemorrhage associated with interstitial inflammation, tubular destruction, and hemosiderin-laden macrophages.
Similar to interstitial nephritis, fibrosis was seen only in CPN cases with rare exception. Interstitial fibrosis was characterized as accumulations of variably cellular and collagenous fibrous tissue that separates nephrons and appears as bright blue by MT staining (Figs. 2, 4). In severe cases, fibrous tissue surrounded and partially to completely replaced injured tubules (Fig. 9).
Extramedullary hematopoiesis was noted in 6.3% NMRs and consisted of interstitial clusters of erythroid, myeloid, and, rarely, thrombocyte precursors (Fig. 12, inset). Except for 1 case, renal extramedullary hematopoiesis was found in NMRs with concurrent CPN.
Discussion
Our retrospective study of pathology data exposed chronic nephropathy as a common age-related and potentially health span–limiting disease process in a zoo-housed NMR population. These data also highlighted striking similarities as well as some important distinguishing characteristics among mouse, rat, and NMR renal disease. Although functional data were not available, based on the proportions of parenchyma affected, renal insufficiency was suspected in those individuals with severe cases of chronic nephropathy and end-stage kidney disease. Furthermore, our study demonstrated the importance of histopathology as a component of the postmortem exam, as renal disease was not reported grossly in 40% of those NMRs with microscopically detected chronic nephropathy.
Due to the eusocial nature of NMRs as described above, it was not feasible to collect urine or blood from individuals to determine renal function utilizing tests such as serum blood urea nitrogen and creatinine, as well as urine-specific gravity and protein:creatinine ratio. However, extensive functional testing has been completed in mice and rats, and those results correlated to histologic changes. 6,21,27,38,41,47 NMR renal functional capacity may be estimated by the severity of lesions with some assertion, particularly in severe and end-stage disease states. Despite this, the clinical manifestations and significance of purported renal dysfunction were not assessed and cannot be extrapolated from laboratory rodents or other species due to the well-described resilience of NMRs and their unique physiologic mechanisms.
Prior to this study, reports of renal disease in NMRs were rare. Edrey et al described limited renal disease in their analysis of 2 extremely old (29–30 years) individuals. 20 The remarkably advanced age of these NMRs may have been a result of relative kidney health. As such, some of the more severe end-stage renal lesions noted in this survey of adult NMRs could have contributed to a shortened health span and possibly premature death. 1,27,39 As the NMR continues to be heralded as an age-defying animal, it is critical to identify and describe potentially age-related, spontaneous lesions, which may affect aging studies. 4,5,13,15,35
The similarities between NMR and old male rat nephropathy were profound (Suppl. Fig. 2); however, certain characteristics of NMR nephropathy suggested different underlying mechanisms (Suppl. Figs. 1, 3–6). One of the first signs of CPN in rats is proteinuria, considered a sequela to glomerular injury and dysfunction. 6,22,38 In old male rats with severe CPN, a significant glomerular lesion is sclerosis with segmental to global replacement of the corpuscle by collagenous matrix tissue (Suppl. Figs. 1, 3, 4). 21,22,27,38 Glomerulonephritis is a common component of mouse chronic nephritis seen in several inbred strains during aging or long-term toxicology studies. 10,21,39,48 However, in the examined NMR cohort, even those individuals with severe chronic nephropathy, glomerular changes were not as prevalent as tubular and interstitial lesions, and they were predominantly characterized as membranous glomerulopathy (Table 1; Figs. 13–15). These findings suggest that the glomeruli may not be the initial site of injury within the nephron, as is speculated in rat CPN and mouse chronic nephritis. 6,21,24,48 Of note, Bowman capsule dilatation—which can be found in mouse and rat glomerulopathies and cystic renal disease—was not a consistent finding in NMR nephropathy, though possibly associated with cortical cysts in this cohort. 21
Renal tubular mineralization was found in a large percentage of NMRs, some of which did not have concurrent renal lesions. This finding is similar to reports in young rats, which can develop mineralized tubules at 2 to 3 months of age and have no other microscopic changes. 38 Furthermore, female rats of several strains have been shown to exhibit tubular mineralization, whereas male rats lack this lesion. Estrogen levels have been implicated, since tubular mineralization can be prevented by ovariectomy and administration of exogenous estrogens to castrated females and males induces mineralization. 38 There was no significant difference in the prevalence of tubular mineralization between male and female NMRs. In addition, renal calcinosis has been detected in some mice strains used in aging studies. 10 As previously speculated, 19 select cases of NMR renal tubular mineralization may be representative of dystrophic and metastatic (ie, uremic) calcification, since these individuals had concurrent renal disease with some degree of functional compromise. However, approximately 28% of NMRs with renal tubular mineralization lacked additional renal lesions.
It is well documented that NMR calcium metabolism and homeostasis are different from that of other animals, as they have no dietary requirement for vitamin D. 16,44 Furthermore, mineralization of skin and soft tissues (calcinosis cutis), heart, kidneys, and other parenchymal organs can be induced by dietary administration of vitamin D, both experimentally and spontaneously in captive NMR populations. 19,40 Although calcinosis cutis was reported in several individuals in this cohort—which in severe cases was attributed to temporary, inadvertent administration of vitamin D in the diet—there was no direct correlation with renal tubular mineralization or renal disease in general. Based on these findings, there appears to be disturbances in mineral homeostasis in some NMRs with renal lesions; however, without data to determine renal function, including serum calcium and phosphorus levels, mineralization cannot be attributed to renal insufficiency. As such, common lesions secondary to chronic renal disease found in other species—for example, uremic gastropathy, fibrous osteodystrophy, and parathyroid hyperplasia—were not noted in this NMR cohort.
Tubular regeneration and hyperplasia were common in these NMRs: typically seen as a component of more severe CPN cases and in those regions of the kidney with fibrosis and evidence of previous infarction. However, there were some instances in which tubular hyperplasia was noted in lesser-affected kidneys (Fig. 10). Of these, a single NMR had tubular hyperplasia with convincingly atypical characteristics, similar to the atypical tubular hyperplasia described in rat strains and adenomas found in old mice. 21,26,28,42 Renal tubular hyperplasia, atypical hyperplasia, and adenoma have been well documented in Fischer 344 rats with CPN. 21,26,28,42 In these studies, the incidence of renal tubular hyperplasia, adenoma, and adenocarcinoma increased significantly with higher grades of CPN. In our study population, atypical tubular hyperplasia was found in 1 (0.7%) individual, whereas CPN was diagnosed in >50% NMRs; therefore this correlation does not appear to exist in NMRs. Of note, fibroblasts isolated from NMRs exhibit contact inhibition and secrete high-molecular-mass hyaluronan, both of which are considered critical to cancer resistance. 34,43,46 It is plausible that hyperplastic and neoplastic responses are not seen in severe cases of NMR CPN due to these antiproliferative phenomena.
As noted previously, NMR chronic nephropathy had similarities to the mouse renal disease in terms of early glomerular changes and end-stage disease syndromes. 21 Interestingly, NMRs from this study did not have high incidence of glomerular amyloidosis as seen in various inbred mouse strains. 10,21 In addition to amyloidosis, other common disease states, including hydronephrosis and male urologic syndrome, were virtually absent from the examined NMR cohort.
Differences in endogenous (eg, genetics, sex) and exogenous factors (eg, husbandry parameters, diet, hydration) influence the development or lack of renal lesions in laboratory rodents. 21,29,41,47,48 Particularly in regard to diet, housing, and husbandry, CPN in rats has been linked to high-protein diets and stressful (ie, crowded) housing conditions. 29 C57BL/6 mice from separate suppliers have been shown to significantly differ in the incidence of renal disease. 41 Our study highlights renal lesions found in 8 NMR colonies housed at a single institution, maintained in the same conditions, and fed the same diet. Therefore, it would be prudent to evaluate NMRs from other institutions to (1) determine if similar renal disease exists in other NMR colonies and (2) examine the role of these factors on the development on NMR nephropathy. Of note, NMRs at zoologic institutions in the United States originate from a finite number of individuals; therefore, lack of genetic diversity may have resulted in increased susceptibility to renal disease. 11,30,31
NMRs from these zoo-housed colonies demonstrated renal lesions similar to, but also distinct from, those reported in laboratory rodent strains used in aging studies. Despite their status as models for extreme longevity and lengthy health span, NMRs are susceptible to potentially clinically significant age-related renal disease, as our study demonstrates. Regarding the pathogenesis and underlying mechanisms of NMR nephropathy, much is to be learned about the inciting and contributing factors. However, with both zoo-housed and research colonies more popular than ever, the opportunity to study renal disease in NMRs is absolute.
Footnotes
Acknowledgements
We thank the Chicago Zoological Society, Brookfield Zoo veterinarians, and Fragile Kingdom animal support staff, in particular Melissa Hamby and Mark Warneke, for providing case materials and animal husbandry information; Renee Walker and staff from the Histology Laboratory of the University of Illinois Veterinary Diagnostic Laboratory for slide preparation. Finally, we thank Kerrie Allen, Brian Johnson, and Erin McCarty of the University of Washington Histology and Imaging Core and the Comparative Pathology of Aging Program and Lena Lim of the Anatomic Pathology Laboratory for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
