Abstract
Runting stunting syndrome (RSS) in commercial chickens has been reported worldwide, and although several studies have attempted to clarify the cause and describe the lesions, there are gaps in knowledge of the epidemiology, pathogenesis, and etiology. The study objective was to use commercial chicks naturally affected by RSS to describe the histologic changes of RSS in all segments of the small intestine in chicks of different ages and to identify viral gene sequences in affected chicks and their association with histologic lesions. Chicks lacking clinical signs but from the same houses and from unaffected houses were used as controls. The average weight of affected chicks was significantly lower than expected for their flocks. Macroscopically, the small intestines had paler serosa, with watery, mucoid, or foamy contents and poorly digested food. Histologic lesions were characterized by necrotic crypts, crypt dilation, and flattening of the crypt epithelium. Histomorphometry of the intestines revealed villous atrophy especially in the jejunum and ileum. Histologic changes in other organs were not observed. Random next-generation sequencing of total RNA extracted from formalin-fixed paraffin-embedded tissues detected avian nephritis virus, avian rotavirus, and picornavirus in jejunal segments from 7-day-old chicks. No viruses were detected in the jejunum of 1-day-old chicks. Detection of picornaviral reads was significantly associated (P < .05) with histologic lesions of RSS. Sequence analysis of the picornavirus revealed genetic similarity with the genus Gallivirus. Using in situ hybridization for galliviral nucleic acid sequences, the signal was associated with crypt lesion severity, although signal was detected both in chicks with and without RSS.
Keywords
Runting stunting syndrome (RSS; also known as malabsorption syndrome) is an enteric disease of chickens that results in severe economic losses due to decreased weight gain, worse feed conversion, and a nonuniform flock. 3 RSS have been reported in several countries including the United States, 34,49 the Netherlands, 40 Germany, 30 Hungary, 33 Croatia, 4 Poland, 43 Iran, 26 Jordan, 39 Pakistan, 35 and Brazil. 24 RSS affects mostly young chickens up to 3 weeks old, 36 but usually occurs around the first 7 days of age. 14,30 The main clinical signs are diarrhea with undigested food, decreased body weight gain with growth retardation, abnormal feather development, and high morbidity. 16,32,46 At necropsy, the small intestines are paler and may contain watery to mucoid contents with poorly digested food and excessive gas. 14,35,36,48 Microscopically, there is dilation of the crypt lumen with flattening of the crypt epithelium (so-called cystic enteritis) 17 that is followed by villous atrophy and fusion with decreased goblet cells in the small intestine. 14,30,35,46
The cause of RSS is not well understood and many viruses have been described in association with this syndrome including chicken astrovirus, 10,13,14 avian nephritis virus (ANV), 7,10,34,38 chicken parvovirus, 10,15,33,48 avian reovirus, 10,34 avian rotavirus (AvRV), 31,34 and infectious bronchitis virus. 10,12 Recently, other viruses have been detected as possible agents of cystic enteritis, such as birnavirus, 19,21,27 calicivirus, 10,19,45 fowl adenovirus group-I, 8,19,24,38 and other members of the families Picornaviridae, 10,19 Siphoviridae, Myoviridae, 10 Anelloviridae, and Circoviridae as well as CRESS-DNA virus. 19 It is suggested that RSS may be caused by one or more viruses.
Most of the above-mentioned studies surveyed for viruses in the feces (and thus may include the normal viral microbiota) and did not characterize the histologic lesions associated with the different intestinal segments. Only one of those studies performed histopathological examination, limited to the duodenum and pancreas. 28 No previous studies colocalized viral nuclei acid or antigen with the lesions, nor associated the histologic lesions with growth retardation. The objective of this study was to describe the histologic changes in all segments of the small intestine in chicks of different ages that were naturally affected with RSS. A further objective was to identify viral nucleic acid sequences in affected chicks by next-generation sequencing (NGS) and characterize their association with histologic lesions by in situ hybridization.
Materials and Methods
Samples
Two hundred and forty-three chicks were collected from 4 different farms and 6 different flocks and houses during 2016 and 2017. The commercial farms were located in 3 different municipalities in the state of Minas Gerais (Pará de Minas, Nova Serrana, and São Sebastião do Oeste). These farms were selected because of (1) a previous history of RSS and/or history of economic loss resulting from flocks with growth stunting and inefficient weight gain in the first week of age, (2) re-use of litter (to investigate environmental persistence), and (3) producer permission for sampling sufficient numbers of animals. The farms were visited to evaluate the environment, management, clinical signs, mortality rate, and aspects of production. At this time, 15 to 30 chicks including 3 to 5 healthy chicks were sampled. Chicks were females, males, or both, ranging from 1 to 11 days old (Table 1). One-day-old chicks were collected from the hatchery before being housed. Four- and 5-day-old chicks were not collected. A total of 213 chicks were examined. Limitations imposed by the producers prevented uniform collection across all farms.
Overview of Chicks Sampled for Analysis. Farms and Houses of Each Flock With the Number and Age of Collected Chicks.
The selected flocks contained chicks with clinical signs characteristic of RSS, such as apathy, growth retardation, diarrhea, and abnormal feather development. Chicks without clinical signs from the same flock were also collected as controls. Chicks (n = 100) from each house were randomly weighed (MF Filizola digital model precision scale) to obtain the flock’s average weight. In the Sector of Animal Pathology of the Veterinary School of the Universidade Federal de Minas Gerais (UFMG), each chick was weighed individually on the same digital scale and euthanatized by cervical dislocation according to the guidelines on euthanasia Resolution 1000/2012 of Federal Council of the Veterinary Medicine. The procedures in this study were performed with approval by the Animal Experimentation Ethics Committee of Universidade Federal de Minas Gerais (Protocol 62/2017).
After euthanasia, a complete necropsy was performed on all chicks and macroscopic changes were recorded. Duodenum, jejunum, ileum, and other organs were collected and fixed for 48 hours in neutral-buffered 10% formalin. Approximately 21 small intestinal samples were collected from each bird: 7 cross sections each of duodenum, jejunum, and ileum (2 cross sections of the cranial, 3 cross sections of the middle portion, and 2 cross sections of the caudal portion). Spleen, thymus, cloacal bursa, liver, lung, heart, kidney, ventriculus, proventriculus, pancreas, cecal tonsils, and cecum were also collected.
Histopathology
The fixed tissues were embedded in low-melt paraffin and 5-μm-thick sections were obtained. The sections were stained with hematoxylin-eosin (HE) following routine procedures and analyzed under a light microscope. 20 All 3- to 11-day-old chicks (n = 213) were histologically examined. Slides of each segment of small intestine (duodenum, jejunum, and ileum) contained approximately 7 sections, and the section with most severe lesions was used to count the crypts and classify the inflammatory cells, as described below. Artifacts and mucosa-associated lymphoid tissue were not counted. For crypt assessment, all crypts were counted as either normal, dilated, or necrotic. The cross sections were graded based on the percentage of dilated and necrotic crypts: marked (31% or more of crypts were affected), moderate (15% to 30% of crypts), or mild (less than 15% of crypts, but more than 3 dilated or 4 necrotic crypts). Chicks with 3 or less dilated crypts or 4 or less necrotic crypts were considered normal. The inflammatory cells of the same section were classified based on the cell type, intensity, and distribution. Inflammatory cells were classified into heterophils, macrophages, lymphocytes, and plasma cells. The intensity was determined as previously described with modifications 47 to an estimated average of the number of inflammatory cells present in high power fields (400×): mild = 0 to 10 cells, moderate = 11 to 20 cells, and marked = more than 20 cells. Multiple 400× fields were used to evaluate the entire circumference of each cross section, and the cross section was scored with the category that was most prevalent for that cross section. The distribution of inflammation was classified as focal, multifocal, multifocal to coalescing, or diffuse. In addition, the other organs of these chicks were also evaluated for the presence of histologic lesions.
Morphometry
Intestinal morphometric analysis was performed for the duodenum, jejunum, and ileum from 7-day-old chicks of farm Y flock 3 House B. These chicks were selected because these animals had higher intensity of lesions and they had sufficient numbers of blocks that lacked histologic artifacts. Thus, 6 chicks with intestinal lesions consistent with RSS (independent of the number of crypts with necrosis or dilatation; see Results for description) and 6 with normal intestines were selected (12 animals total; considered the minimum sample size for statistical significance). Morphology indices included the villus height (from the tip of the villus to the crypt opening) and crypt depth (from the base of the villus to the base of the crypt) to evaluate the intensity and distribution of villous atrophy. Twenty-five villi and crypts were measured for each animal with the software Leica Application Suite (LAS) v4.2 Leica. Crypts were selected to ensure that they could be measured from base to the top without artifact.
Variables of villous height and crypt depth of each segment were compared between healthy and diseased chicks. To determine statistical significance, the villous height and crypt depth of each segment were submitted to analysis of variance (Fisher test at 5% probability) using STATA software.
Samples for Molecular Tests
A subset of the jejunal samples used for histopathology were used for molecular testing. The samples were from Farm Y/Flock 2/House B (7 days old) and Farm Y/Flock 3/House B (1, 3, and 7 days old) to check the permanence of the virus in the environment. Samples from Farm Z/Flock 6/House F were used as control (3 days old). Twelve samples with marked histologic crypt lesions (31% or more of affected crypts) were selected as RSS samples (six 7-day-old chicks from each of the 2 affected flocks), and 13 samples lacking histologic changes were selected as controls (two 7-day-old chicks from each of the 2 RSS-affected flocks, 3 unaffected 1-day-old chicks from the hatchery, 3 unaffected 3-day-old chicks from one of the RSS-affected flocks, and two 3-day-old chicks from the unaffected control farm). For each sample, twenty 5-μm-thick tissue sections were collected in one 1.5-ml centrifuge tube. The microtome and the blade were decontaminated with 70% ethanol and RNase Away (Sigma) before and after collecting tissue sections from each sample to avoid cross-contamination between samples. After deparaffinization by CitriSolv (VWR International), total RNA was extracted using the RNeasy FFPE (formalin-fixed paraffin-embedded) Kit (Qiagen) as per manufacturer’s instructions. RNA was quantified using the Qubit RNA HS Assay Kit on a Qubit fluorometer 3.0 (ThermoFisher Scientific), and the purity of RNA was determined by NanoDrop Spectrophotometer 2000/2000c (ThermoFisher Scientific). Fragment size of RNA was measured using the RNA 6000 Pico kit on an Agilent Bioanalyzer instrument (Agilent Technologies) as per manufacturer’s instructions.
DNA libraries for NGS sequencing were prepared using the KAPA Stranded RNA-Seq Library Preparation Kit for Illumina platforms (Kapa Biosystems) following manufacturer’s instructions. First and second strand DNA were synthesized with random primers and then the cDNA fragments were marked and A-tailed before ligation of unique barcode adapters, which allowed for multiplexing of samples into a single sequencing run. Bead-purified adapter-ligated libraries were amplified by PCR (polymerase chain reaction; 10 cycles) using the library amplification master mix provided in the kit. The quantification of dsDNA concentration in libraries was performed using the dsDNA High Sensitivity Assay Kit (ThermoFisher Scientific) on a Qubit fluorometer 3.0 (ThermoFisher Scientific). The average DNA fragment size in each library was measured using the High Sensitivity DNA kit in the Agilent Bioanalyzer. To facilitate uniform clustering, libraries with an average DNA fragment size of 240 to 300 bp and a concentration greater than 3 ng/µl were used. Libraries were diluted to 4 nM, pooled by equal volume (5 μl), denatured with NaOH (0.2 N final concentration), incubated for 5 minutes at room temperature, and diluted to 20 pM concentration with chilled HT1 hybridization buffer (Illumina). The library was diluted to a final concentration 10 pM with the same buffer. A control library (3% PhiX174, Illumina) was added and the libraries were chilled on ice.
Next-Generation Sequencing and Analysis
The paired-end sequencing was conducted on an Illumina MiSeq instrument using a 300 cycle (2 × 150) MiSeq Reagent Kit v2 (Illumina). After automated cluster generation in MiSeq, the sequencing reads were de-multiplexed based on their unique adapter.
The raw sequencing data were analyzed using the Galaxy platform interface 1 and partially utilizing a previously described bioinformatics workflow. 11 Briefly, raw reads quality was assessed using FastQC 2 and any residual adapter sequences were trimmed using Cutadapt v1.6. 22 Trimmed reads from the same farm were pooled together and were mapped against the Gallus gallus and PhiX174 reference genomes using BWA-MEM v0.2.1 in order to identify host and control library reads. 18 Mapped host and control reads were filtered out using the Filter sequences by mapping v0.0.4 tool. 6 The remaining read pairs were joined with PEAR v0.9.6.0. 18 De novo assembly was performed using the MIRA assembler v3.4.1. 5 The assembled contigs were blast-searched against the NCBI nt database (blastn). The best hit was identified as chicken/Hungary/Pf-CHK1/GV/2013 (GenBank Accession Number KT880666).
To obtain a larger final consensus, a reference-based assembly approach was used. The raw reads, after adapter trimming and filtering out the host and library control reads, were pooled, and imported into Geneious v. 9.1.8 (Biomatters Ltd). Pair reads were merged and target-based assembly using the complete genome sequence of chicken/Hungary/Pf-CHK1/GV/2013 (KT880666, best blast hit of de novo assembled contigs) as a reference was performed. The “High Sensitivity” settings of the Geneious mapping tool were utilized. The consensus yielded after each mapping run was used as a reference for a consequent mapping run using the same settings, for a total of 5 iterations. The final consensus was blast-searched against the NCBI nt database (blastn).
In Situ Hybridization
In situ hybridization (ISH; RNAScope, Advanced Cell Diagnostics) was used to assess for presence of galliviral genome fragments in FFPE intestinal sections (gallivirus was targeted based on the sequencing results). The preliminary sequence data (partial genomes) from each flock described above were used to design proprietary probes (Advanced Cell Diagnostics) that would identify the genomes of the de novo assembled virus. To avoid false negative results, the probes were designed to not hybridize to the genomes of the following previously described poultry picornaviruses: NC_039235.1 (Chicken megrivirus strain chicken/B21-CHV/2012/HUN), NC_028964.1 (Chicken gallivirus Pf-CHK1/GV), NC_024770.1 (Chicken gallivirus 1 isolate 518C), and NC_018400.1 (Turkey gallivirus). A probe for chicken ubiquitin C mRNA (UBC) was used as a positive control probe, and a probe mixture for Bacillus subtilis strain SMY methyglyoxal synthase (mgsA), dihydrodipicolinate reductase (dapB) and oxidized nucleotide pyrophosphohydrolase (ypjD) was used as a negative control. Cross-hybridization was reduced by performing in silico quality control analysis. 45
ISH was performed on 3 animals that were NGS-positive for galliviral reads and 3 animals that lacked galliviral reads from the following groups: FarmY/Flock2/HouseB (both 7 days old), FarmY/Flock3/HouseB (2 positive: 7 days old; 2 negative: 3 days old and 7 days old). Samples were selected based on flock diversity and the presence/absence of detectable galliviral reads by NGS. RNAScope® 2.5 HD-Red ISH was performed on FFPE jejunal sections according to manufacturer’s protocol. Briefly, 5-μm-thick sections were incubated in a dry oven for 1 hour at 60 °C, deparaffinized in xylene for 10 minutes, and then washed in 100% ethanol for 2 minutes. Endogenous peroxide was quenched with manufacturer-supplied hydrogen peroxide for 10 minutes at room temperature (RT). Antigen was retrieved by incubation for 15 minutes at 98 to 102 °C followed by protease Plus (Advanced Cell Diagnostics) at 40 °C for 15 minutes in a HybEZ hybridization oven (Advanced Cell Diagnostics). The slides were incubated with the probes at the ACD-recommended concentrations for 2 hours at 40 °C. Slides were washed 2 times with 1× Wash Buffer (Advanced Cell Diagnostics) for 2 minutes at RT then incubated with a series of compounding chromogenic amplifiers with different temperature and incubation time: Amp1 at 40 °C for 30 minutes, Amp2 at 40 °C for 15 minutes, Amp4 at 40 °C for 15 minutes, Amp5 at RT for 30 minutes, and Amp 6 at RT for 15 minutes. Slides were washed 2 times (in 1× Wash Buffer) between each amplifier incubation. Slides were then incubated for 10 minutes with the chromogen at RT and the quick washed with distilled water. Staining was visualized on a light microscope after counterstaining with 50% hematoxylin (Vector lab) × 2 minutes, drying, and coverslipping with EcoMount (BioCare Medical). For each block, one section was incubated with the anti-GV probe, one was incubated with an irrelevant probe (anti-DapB; negative control probe), and one was incubated with an anti-chicken ubiquitin probe (positive control probe). An archived tissue block from a purpose-raised chicken from an unrelated previous study was used as negative control tissue.
To determine the quantity of positive signal, all crypts were counted (with and without positive signal) and the percentage of crypts with positive signal was calculated and classified as 1 of the 4 categories: absent = none positive signal, mild = 1% to 25% of crypts with positive signal, moderate = 26% to 70% of crypts, and marked = more than 70% of crypts with positive signal.
Statistical Analysis
Weight measures, counts of crypts in the cross-section of duodenum, jejunum, ileum, and intensity of the inflammatory cells were entered into a spreadsheet (Microsoft Office Excel 2007), organized by age and presence (or absence) of lesions. Averages, standard deviations, or relative frequencies of these variables were calculated. To evaluate the normality of quantitative variables, Shapiro-Wilk test and graphical analysis applying quantile-quantile plots were performed. The t test was used to determine the significant differences between groups for variables with normal distribution, and for the variables with nonnormal distribution, the Wilcoxon test or Kruskal-Wallis test were performed. Fisher’s exact test was applied to evaluate significant associations between categorical variables.
The Kruskal-Wallis nonparametric test was used, followed by the Dunn test to check the differences between the number of total lesions (crypts with dilation and necrosis and the type of inflammatory cells) in the small intestine, and to compare dilated or necrotic crypts by age. The Wilcoxon nonparametric test was performed to check the difference between proportion of crypts marked in ISH of chicks with and without histologic lesions.
Spearman’s correlation test was performed to verify the correlation between lesioned crypts and the intensity of the inflammatory cells. Pearson’s correlation test was used to analyze the correlation between the body weight and the number of necrotic and dilated crypts.
P values less than .05 were considered statistically significant in all data analysis that was carried out in Stata software version 15 (StataCorp, 2017, Stata Statistical Software: Release 15).
Results
History and Clinical Signs
The broilers were of the Cobb500 breed. The chicks were housed in a negative-pressure chicken dark-house (houses B and C) or in a conventional, positive-pressure house (houses A, D, E, and F). The number of chicks in each house varied from 30 000 to 40 000. Chicks were supplied with nipple drinker and mash feed ad libitum. They were reared in houses made of concrete with a deep rice paddy husk litter. Hygienic precautions included separate clothes and boots at the entrance of the house but did not include a footbath disinfection barrier. Cats or dogs were not present and rodents had adequate control. Chicks were vaccinated against infection bursal disease (IBD), Newcastle disease, and Marek’s disease. These vaccines were administered in ovo or at 1 day of age.
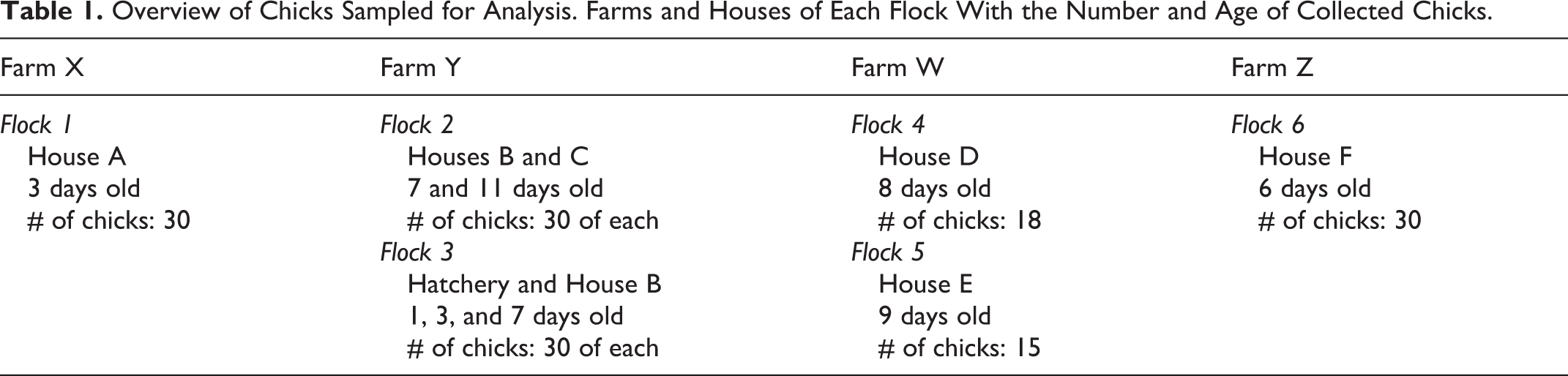
The standard weight (ie, weight divided by age) of 3- to 9-day-old sick chicks was statistically lower than standard weight of 3- to 9-day-old healthy chicks (t test, P < .05; Fig.1). A negative Pearson’s correlation was observed (P < .05) between the standard weight of affected chicks and the number of necrotic crypts (r = −0.151, P < .05), but not between the standard weight and the number of dilated crypts.
Standard weights of the healthy chicks (n = 42) and those affected by runting and stunting syndrome (RSS; n = 64). Standard weight is calculated as body weight (grams) divided by age (days). The boxes show the median and quartiles; whiskers show the highest and lowest values. • outliers. P < .05, t test. *Boxes denoted by a different letter indicate significant differences (P < .05).
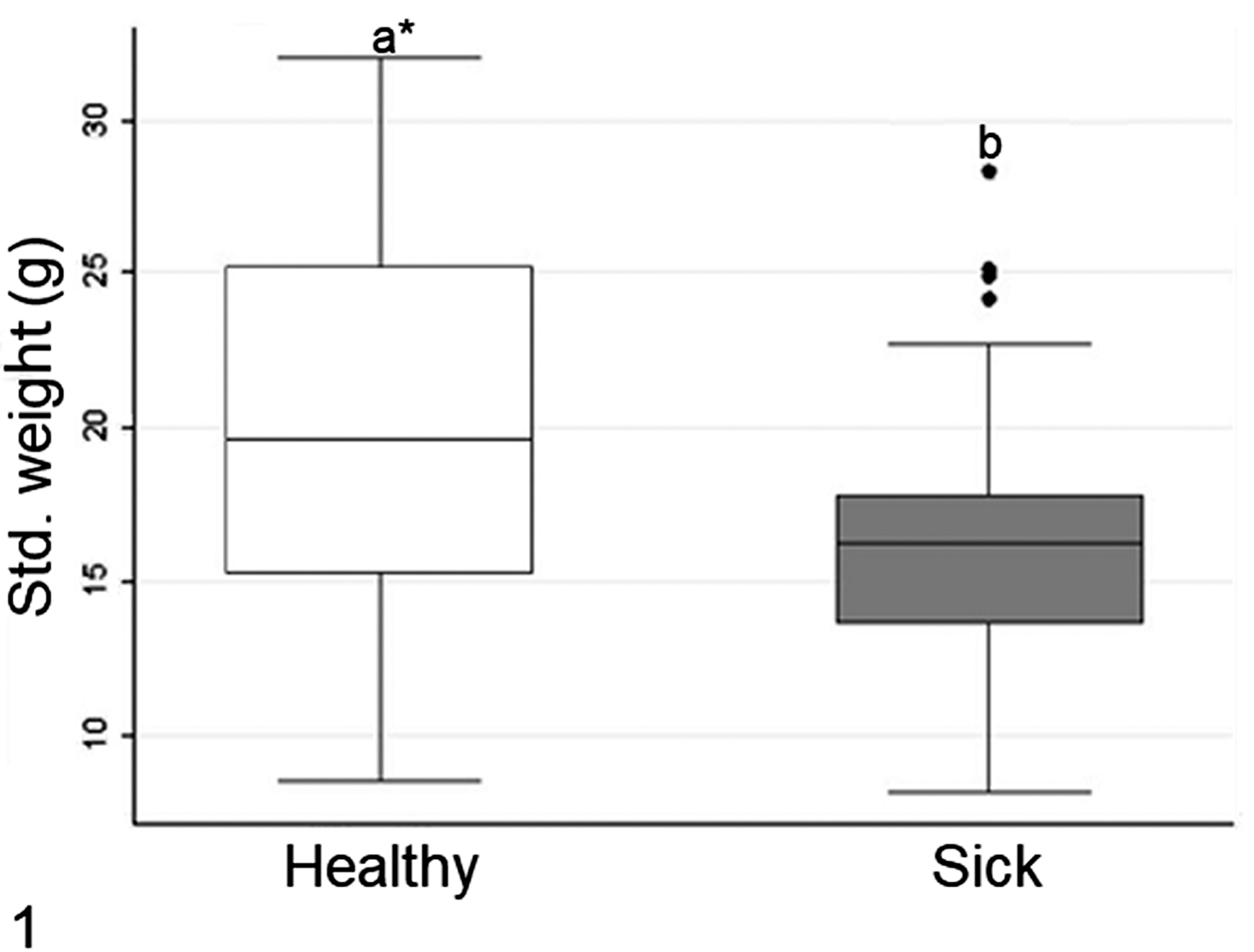
The flocks with chicks 3 days old or older presented characteristic clinical signs of RSS. The main clinical signs were growth retardation (Fig. 2) and apathy (Fig. 3), followed by poorly digested food in the feces, diarrhea, and rarely with abnormal feather development. The mortality rate of these flocks in the first week varied from 0.5% to 1.9%.
Runting stunting syndrome, chicks. Figure 2. Eleven days old, house C from farm Y. There is growth retardation of the diseased (left) compared to the healthy (right) chick.
Gross Pathology
At necropsy, sick chicks had paler small intestinal serosa than unaffected chicks. The intestines were frequently distended by gas and contained watery or foamy content (Fig. 4) with undigested food (Fig. 5). Some chicks had hyperemia in the serosa of small intestine. Omphalitis or yolk sac infections were sporadically detected, as expected, in chicks with or without RSS. No other diseases were diagnosed.
Histopathology
None of the 3-day-old chicks (n = 60) had significant crypt changes (ie, 3 or fewer dilated or necrotic crypts were considered normal/insignificant) or inflammation greater than the mild score. Of the 6- to 11-day-old chicks, 45/153 had no histologic lesions, with 28 of these having growth retardation but lacking lethargy and diarrhea. All of these healthy chicks had normal crypts and only mild inflammation. However, the other 108 six- to 11-day-old chicks had histologic lesions and clinical signs typical of RSS. Considering all 3 intestinal segments (n = 324) from 108 chicks with lesions, 63 chicks had severe lesions in the duodenum, 101 had severe lesions in the jejunum, and 66 had severe lesions in the ileum. These histologic lesions included mild to marked villous atrophy with fusion, and crypts that were dilated and/or necrotic (Figs. 6, 7). Of these 324 segments with lesions, the necrotic crypts were predominant in 158 (48.8%) segments characterized by karyorrhexis and karyolysis of enterocytes and generally with loss of crypt architecture and conformation (Fig. 8). Dilated crypts were found in 166 (51.2%) segments and histologically the crypts were lined by flattened enterocytes and the lumen was filled with cellular debris and heterophils (Fig. 9).
The chicks (n = 108) usually had lamina propria expanded by mild and multifocal infiltrates of one or more type of inflammatory cells (heterophils, macrophages, and/or lymphocytes and plasma cells; Fig. 10). Positive Spearman’s correlations were observed (P < .05) between the number of affected crypts and the intensity of inflammatory cells (r = 0.477). Most of segments classified as mild inflammation were predominately infiltrated by macrophages (286/324; 88.3%). The most common infiltrate in segments classified as moderate or marked was lymphoplasmacytic (146/324; 45.1%). In addition, according Kruskal-Wallis test, there was significant difference (P < .05) in the number of dilated crypts between sections with minimal, mild, and moderate inflammatory cell infiltration. For necrotic crypts, a difference (P < .05) was only found between cases with minimal and mild inflammatory cell infiltrate. The macrophages contained intracytoplasmic microvacuoles and were mainly located next to the necrotic/dilated crypts. Heterophils were often located in the middle third of the villi or next to the affected crypts. Lymphocytes and plasma cells did not have a specific location. No relation was found between age and intensity of inflammatory cells infiltration.
In all cases of 6- to 11-day-old chicks with histologic lesions, the jejunum was the segment most affected by crypt necrosis and dilatation (P < .05). Significant differences were found in the number of necrotic crypts between duodenum, jejunum, and ileum (P < .05), as well as the number of dilated crypts. Moreover, both the jejunum and ileum had significantly different levels of crypt necrosis as compared to crypt dilation (P < .05), whereas no differences between crypt necrosis and dilation were detected in the duodenum (P > .05; Fig. 11).
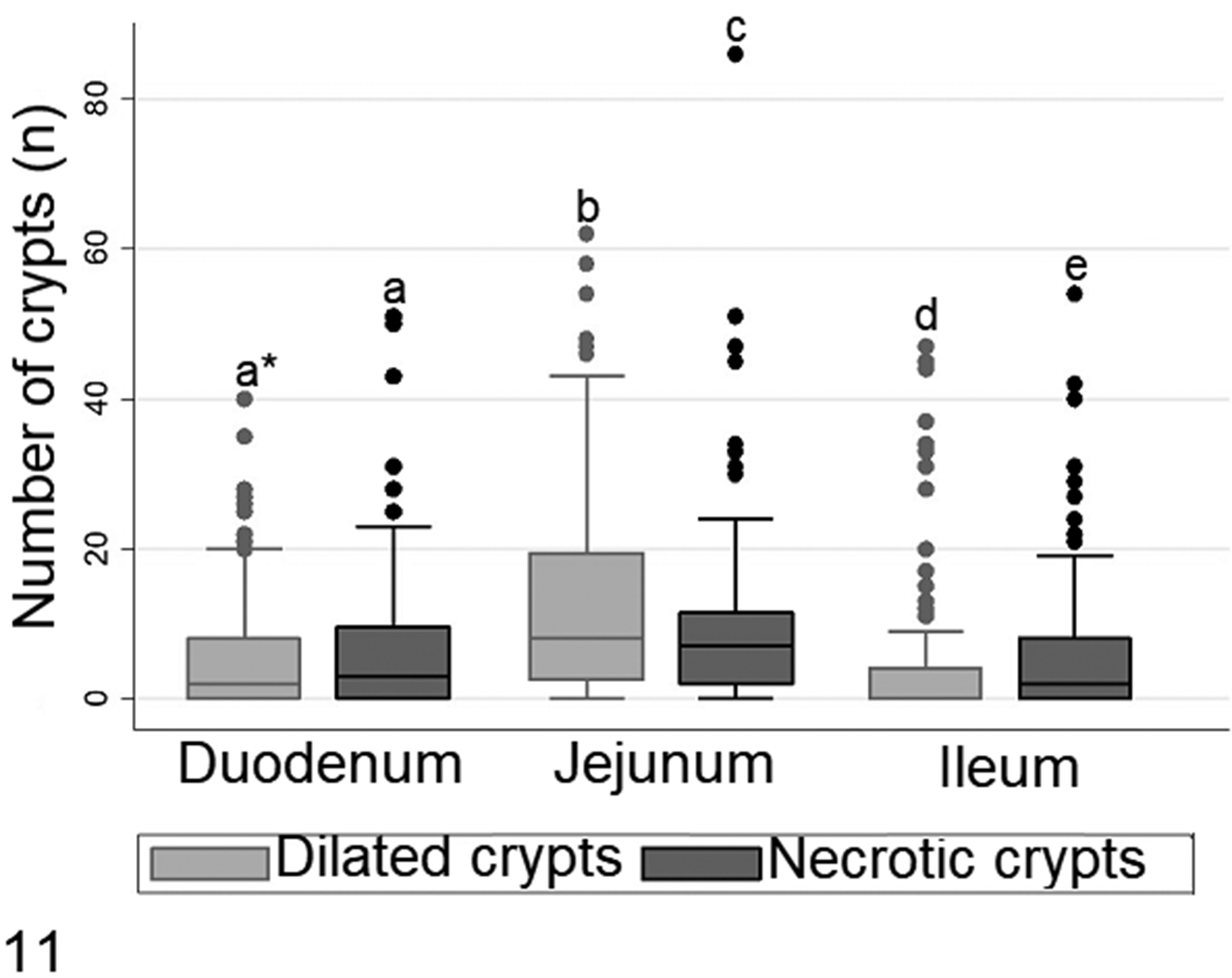
Number of dilated and necrotic crypts per intestinal segment, from 108 six- to 11-day-old chicks with histologic lesions of RSS. The boxes show the median and quartiles; whiskers show the highest and lowest values. • outliers. *Boxes denoted by a different letter indicate significant differences (P < .05), Dunn test.
According to Kruskal-Wallis test followed by Dunn test, crypt necrosis was significantly highest in day 6, and then tapered in severity out to day 11 (Fig. 12). In contrast, crypt dilation reached its peak in days 7 and 8, which were statistical different (P < .05) in relation with other ages (Fig. 13).
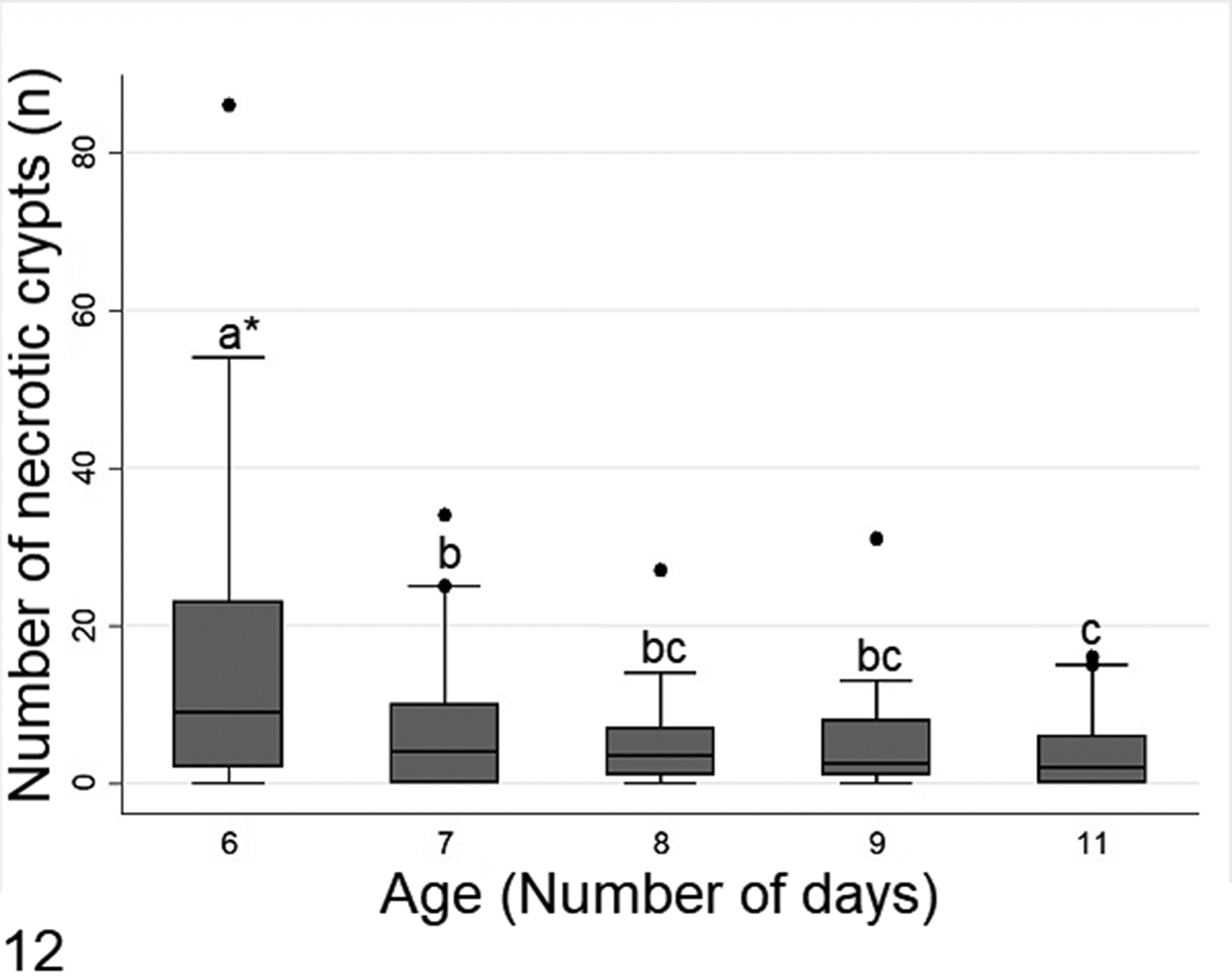
The number of necrotic crypts in diseased chicks of different ages. The data are from all intestinal segments in 108 six- to 11-day-old chicks with histologic lesions of RSS. *Boxes denoted by a different letter indicate significant differences (P < .05), Dunn test.
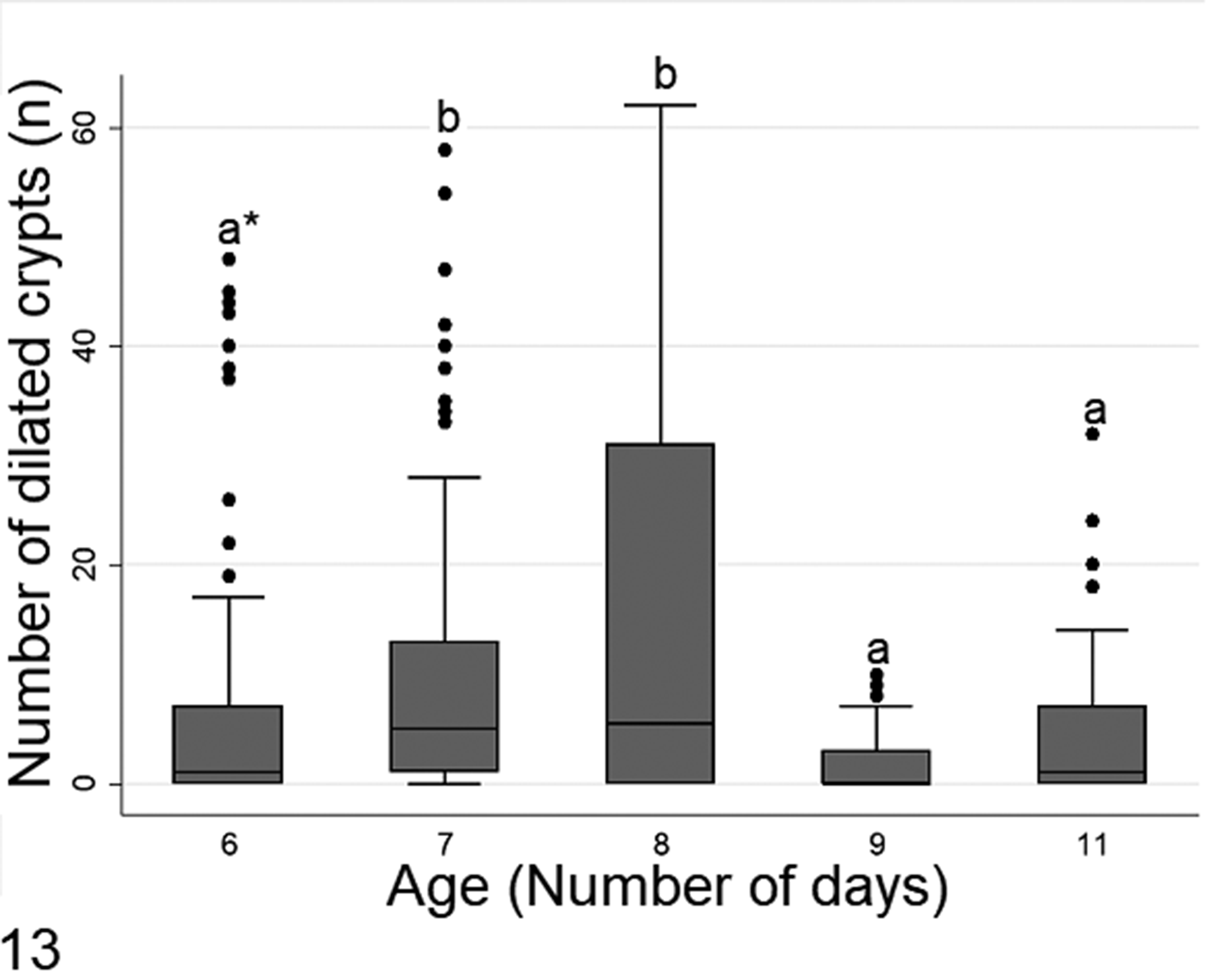
The number of dilated crypts in diseased chicks of different ages. The data are from all intestinal segments in 108 six- to 11-day-old chicks with histologic lesions of RSS. *Boxes denoted by a different letter indicate significant differences (P < .05), Dunn test.
The spleen, thymus, liver, lung, heart, kidney, ventriculus, proventriculus, pancreas, cecal tonsils, and cloacal bursa lacked histologic lesions. While not as extensively characterized compared to the small intestine, the ceca of seven 7-day-old chicks had mild dilated and/or necrotic crypts with mild and multifocal infiltration of heterophils and/or lymphocytes and plasma cells. Rarely these cases had inflammation without crypt changes. The chicks with cecal lesions usually had moderately to markedly dilated crypts in the jejunum.
The duodenal villi from 7-day-old chicks were significantly longer in sick chicks compared with healthy chicks. In contrast with duodenal results, the jejunal and ileal villi were shorter in sick chicks compared to healthy chicks. Furthermore, the duodenal and ileal crypts were significantly longer in sick chicks (Fig. 14).
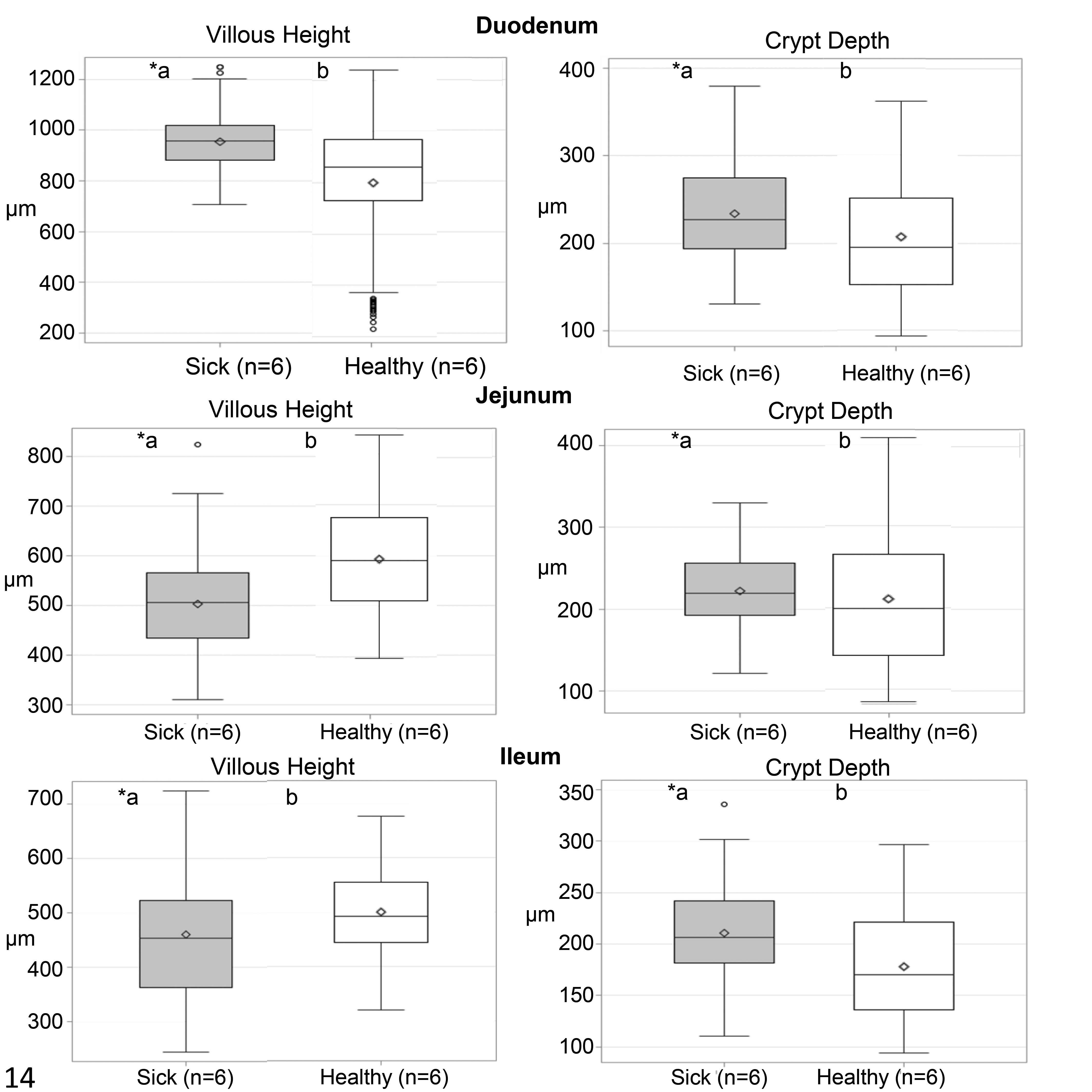
Villus height and crypt depth in different small intestinal segments from healthy chicks (n = 6) and those with histologic lesions of RSS (n = 6). The boxes show the median and quartiles; whiskers show the highest and lowest values. ○ outliers. ◊ average. *Boxes denoted by a different letter indicate significant differences (P < .05), Fischer test.
Next-Generation Sequencing
The total RNA concentration from 25 FFPE samples varied between of 62 and 2321.4 ng/μl. The 260/280 ratios were 1.9 to 2.04, and the RNA integrity number (RIN) values were 1.4 to 2.7.
Random NGS of total RNA extracted from FFPE tissues was performed using jejunum only because this segment was the more affected one and the selected ages were 3 and 7 days old. Avian nephritis virus, avian rotavirus, and picornavirus were identified. In the 3-day-old chicks only avian nephritis virus was detected, whereas the 7-day-old chicks had avian rotavirus, avian nephritis virus, and picornavirus (Table 2). No viruses were detected in the 1-day-old chicks.
Viruses Identified by Next-Generation Sequencing in Chicks From Each Location.
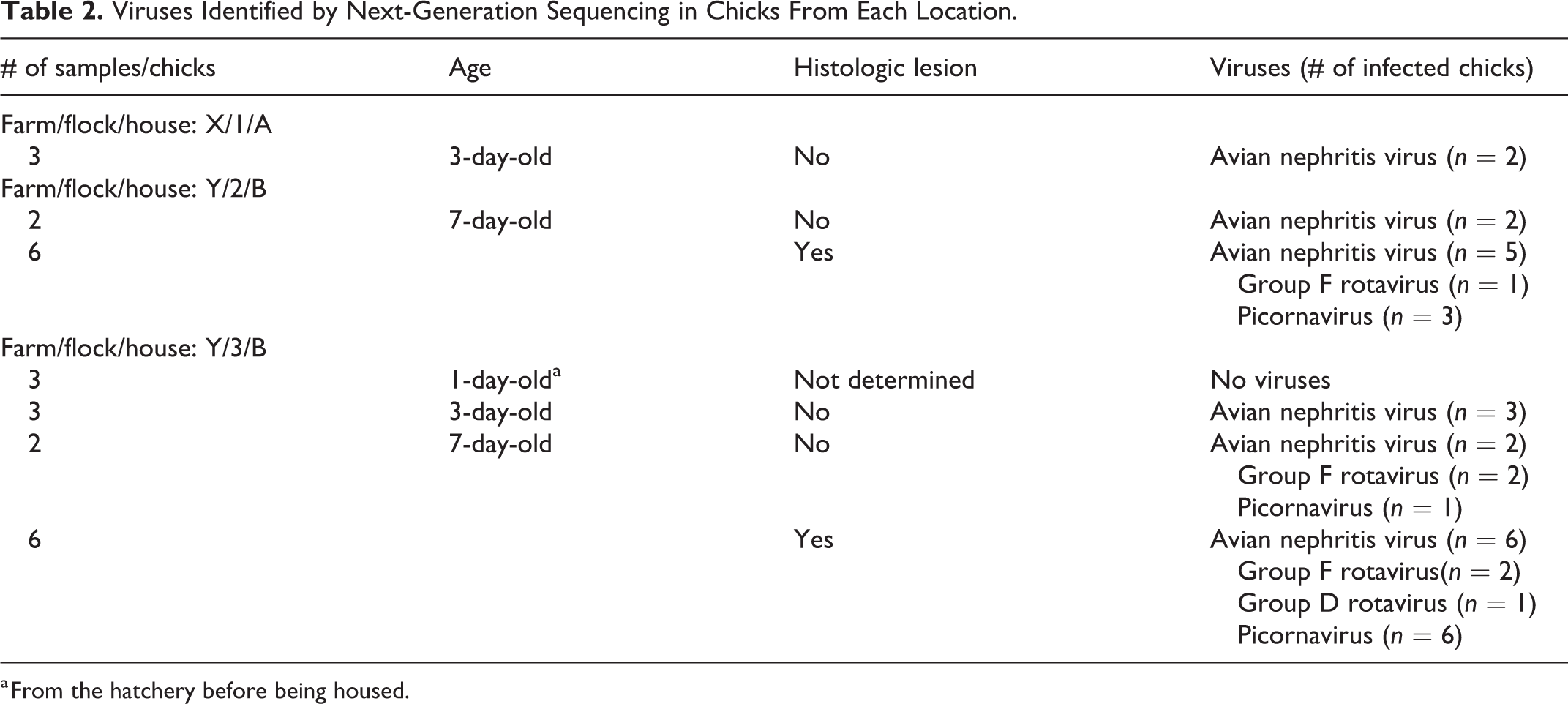
a From the hatchery before being housed.
The permanence of viruses in the environment was evaluated by chicks collected from 2 different flocks (2 and 3) and housed in the same house (B) at different times. The first 7-day-old chicks housed had avian rotavirus, avian nephritis virus, and gallivirus. The chicks housed later were collected at 1 day of age and lacked NGS-detectable viruses. By 7 days of age, this second flock in the same house was positive for the same viruses from the previous flock.
The described viral reads were identified in chicks with histologic lesions and in chicks with no RSS lesions. Fisher’s exact test showed that only the picornavirus RNA presence was statistically significant (P < .05) in chicks with lesions. Sequencing yielded 22 743 342 raw reads. After quality and filtering out the host and control library reads, 123 433 read pairs remained. These reads were pooled and were used in the assembly workflow. The final consensus, obtained from 560 mapped reads was 8604 bp in length, and contained 4 contigs: 983 bp, 209 bp, 1089 bp, and 4653 bp. The final consensus blastn results showed that the identified virus was distantly related to chicken picornaviruses isolated in different countries. The best hit (a novel picornavirus chicken/CHK1/USA/2010, JF424824) was 82.79% identical; however, as only partial genome of chicken/CHK1/USA/2010 was available, the query-hit alignment was only 3210 bp long. The second and the third best hits were chicken/Hungary/Pf-CHK1/GV/2013 (KT880666) and chicken/HongKong/518C/2010 (KF979337) with 75.45% and 75.21% nucleotide identity, respectively. The identity results of each of the 4 contigs searched separately are presented in Table 3.
Sequence Identity Results for Each of the Separate Contigs in the Final Consensus.
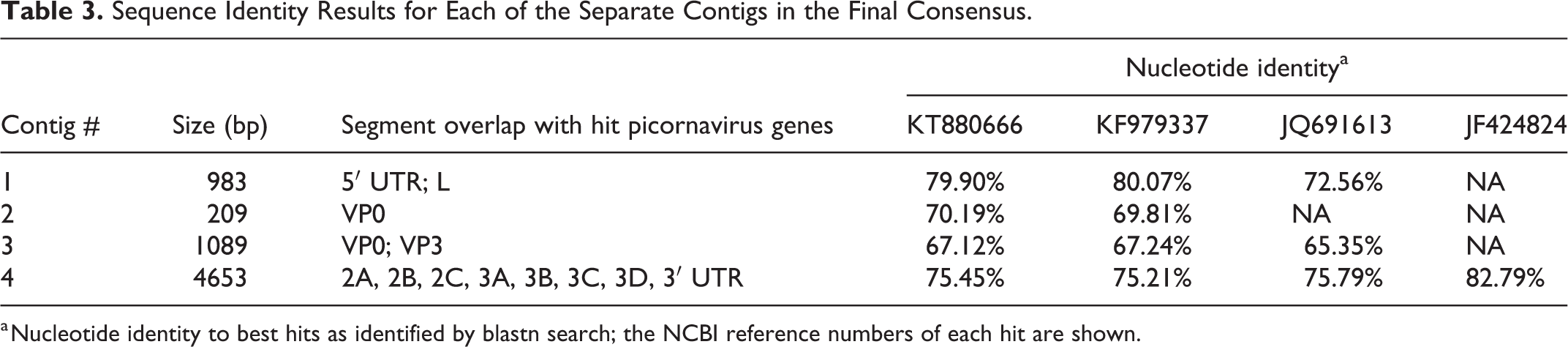
a Nucleotide identity to best hits as identified by blastn search; the NCBI reference numbers of each hit are shown.
In Situ Hybridization
In situ hybridization was performed using 6 blocks/chicks at 7 days old that were used for NGS to associate presence or absence of lesions with galliviral infection. All chicks were ISH positive for gallivirus in the epithelial cells, including the three that were negative in the NGS and lacked histologic lesions. The chicks without histologic lesions had minimal to mild positive signal, while the chicks with necrotic crypts had moderate to marked positive signal. A significant difference was found between the proportion of crypts with positive signal in the ISH in chicks with (Fig. 15) and without (Fig. 16) lesions according Wilcoxon test (P < .05). The location of positive signal for GV by ISH was multifocal in the mucosa, primarily epithelial cells of apparently normal crypts and adjacent lamina propria (Fig. 17), and in the lamina propria associated with lymphoid follicles (Fig. 18). Positive signal was not visualized in the necrotic or dilated crypts. No positive signal was observed in control tissues (archival, experimental tissues sections unrelated to this study).
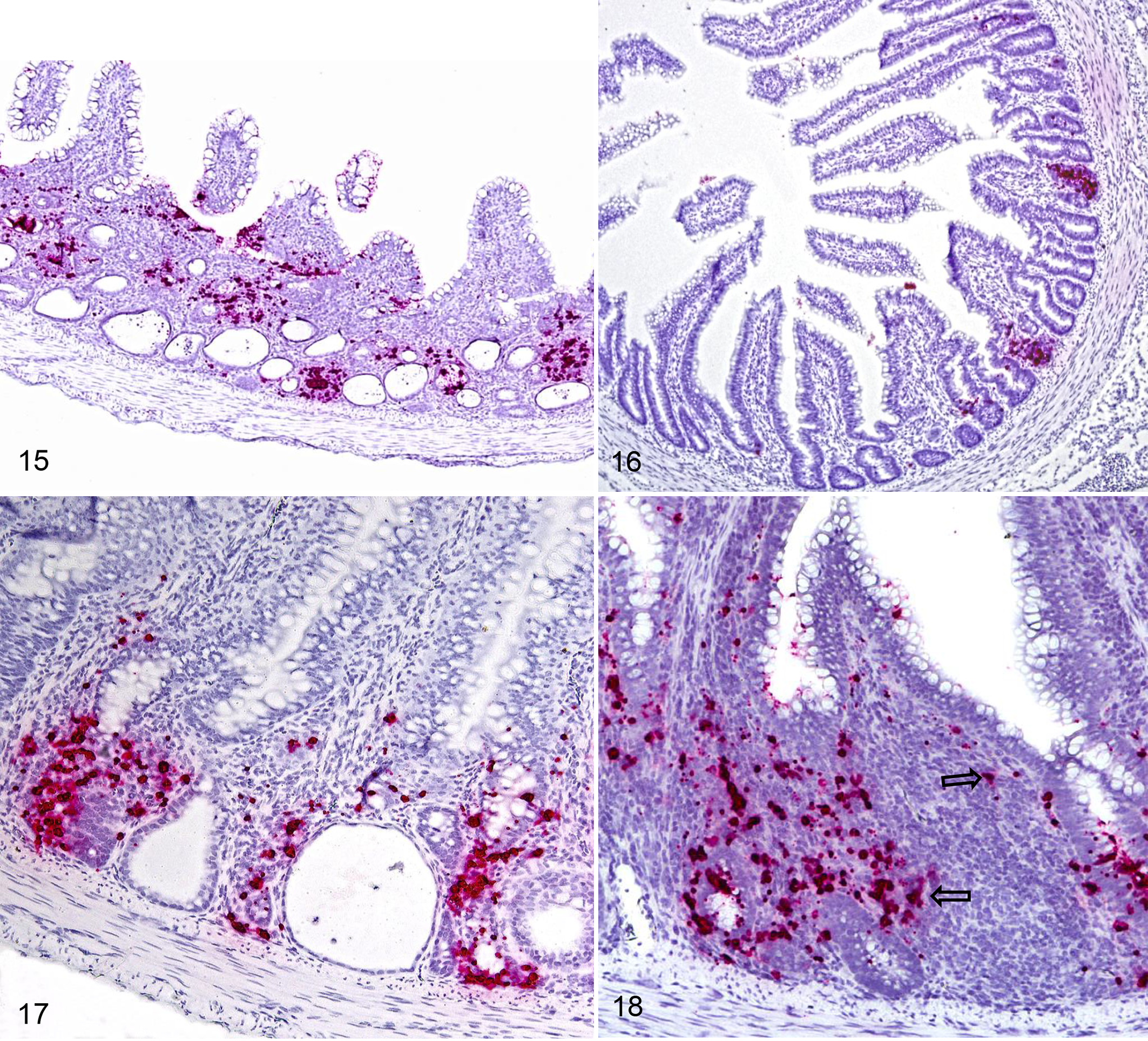
Jejunum, chicks. In situ hybridization for gallivirus.
Discussion
In order to assess the role of viruses in naturally occurring RSS, the clinical features and histopathology were characterized, NGS was used to identify viral reads associated with RSS, and ISH was used to determine the location of virus in affected and unaffected intestinal tissues. Consistent with previous reports, 12,25,35 poor growth and feed conversion were the major clinical signs in the present study. Dilated and necrotic crypts with villous atrophy and mixed inflammatory infiltrates were histologically identified throughout all sections of the small intestine. Several viruses were identified using NGS; however, only gallivirus was statistically associated with the presence or absence of RSS histologic lesions. Subsequently, gallivirus nucleic acid was detected in diseased intestine by ISH, and the signal in the intestine was statistically associated with lesion severity.
While nonspecific, the classical clinical signs of RSS in chicks have been described as growth retardation, apathy, diarrhea, and poor feathering. 16,24,30,46 Lack of efficient weight gain is the most significant clinical sign, due to the economic losses associated with flock-wide poor feed efficiency. In this study, the standard weight of 3- to 9-day-old chicks affected by RSS was significantly lower than that of healthy chicks. This is important, as the weight at 7 days age has a positive relation with the broiler weight at slaughtering age. 37 Losses such as those observed in affected chicks are detrimental to the performance of the broilers and are consequently responsible for considerable economic loss for the producer, even with the low mortality rate that is typically found with RSS.
As expected from a disease with poor weight gain and diarrhea, the gastrointestinal tract is the tissue with significant lesions in RSS. 12,25,35 Macroscopic findings in this study included intestinal serosal pallor, gas-distention with watery/foamy content and undigested food and, in some cases, hyperemic intestinal serosa, consistent with previous reports. 36,40,47 Some authors described atrophy of lymphoid organs such as cloacal bursa, thymus, and spleen, 17,29 but in the present study, these findings were absent.
Similar to the gross findings, histologic changes of RSS were focused on the small intestinal mucosa. Histologic lesions included atrophy and fusion of villi, dilated crypts (cysts), and necrosis of crypt epithelial cells, which have been previously described. 14,30,46 Similar to other enteric diseases, intestinal epithelial damage is considered the lesion responsible for the poor weight gain, and thus the significant economic losses. Other lesions of RSS include degeneration and vacuolation of pancreatic acinar cells with fibrosis, proventriculitis with glandular degeneration and infiltrate of macrophages and lymphocytes, 35,40 and atrophy of cloacal bursa. 35 These lesions were absent in the current study.
The jejunum was the segment of small intestine most significantly affected by RSS. 36,40,44,47 Although the jejunum was the most damaged segment, our study showed that the duodenum and ileum are also severely affected, so they should not be ignored for diagnosis. Changes in the cecum were also identified, which was different from a previous report, 30 but the changes were mild and considered clinically insignificant. In all small intestinal segments, the number of necrotic crypts was significantly greater in 6-day-old chicks compared with other ages and the crypt dilation peaked 1 to 2 days later. This pattern of crypt necrosis peaking on or before day 6, followed by peak crypt dilation at days 7 and 8, suggests that the virus causes the necrosis, probably during the replication phase, and cell debris accumulates resulting in crypt dilatation. 36,44 A study of RSS demonstrated that degeneration of enterocytes from the crypt occurred from day 3 postinfection, with remarkable cystic distension of crypts and marked villous atrophy at days 5 and 7 postinfection, 47 findings similar those found in the present study.
As expected with the aforementioned, crypt changes were associated with a significant reduction of villus height in the jejunum and ileum. The reduction in jejunal villus height has been described as a finding in chicks affected by RSS. 36,44 Only one study with avian rotavirus performed morphometry in chicks between 5 and 14 days old, and identified marked villous atrophy extending from cranial jejunum to the ileum with predominance of lesions in the middle to distal small intestine. 30 The villous atrophy is probably a result of epithelial necrosis and inhibition of cell proliferation at the base of crypts which prevents the migration of these cells to the tip of the villus. 36
Our findings of increased duodenal villous height and increased depth of the duodenum and ileum crypts could indicate regeneration in these segments following injury. It was not possible to evaluate chronologically the type and intensity of the lesion in the small intestine, since our study performed morphometry only in 7-day-old chicks, which was after peak crypt necrosis.
In the present study, the lamina propria of the small intestine was usually expanded by infiltrates of heterophils, macrophages, and/or lymphocytes and plasma cells. Previous research identified infiltration predominated by macrophages and lymphocytes in small intestine; 41,45 however, at least one RSS experimental study described a heterophilic infiltration that was most pronounced at day 3 postinfection and reduced by day 5 postinfection. 47 The role of inflammatory cells in chicks affected by RSS is not clear. The current study showed that the sections with more severe crypt lesions also had a higher intensity of inflammatory cells.
Next-generation sequencing of jejunal segments from 3- and 7-day-old chicks revealed multiple viruses in RSS-affected and unaffected chicks including avian nephritis virus, avian rotavirus, and gallivirus. Avian nephritis virus and avian rotavirus were previously reported in Brazil 8,9,23,24,28 and in other countries. 7,10,19,30,34,38,43,44 Similar to our findings, some researchers found avian nephritis virus in chicks at 3 and 7 days of age, but avian rotavirus was only detected from chicks in the second week of age, 8 diverging from our study that found avian rotavirus in chicks at 7 days of age. This age differential 34 may explain why avian rotavirus was not as frequent as avian nephritis virus in the current study. Regardless, avian nephritis virus was detected in healthy and diseased chicks as well as with avian rotavirus in a low frequency, so it was difficult to establish direct relationships.
The absence of viruses in 1-day-old chicks suggest that the viruses studied in the present work were absent from the jejunum at this age; however, the detection limit of NGS in FFPE is not well established. Some authors suggested vertical transmission of avian nephritis virus because it was detected in broiler chicks in the first week of age 8,42 and others found avian rotavirus in hatchery. 34 The presence of the same viruses (avian rotavirus, avian nephritis virus, and gallivirus) from those found in the previous flock from the same house suggest that these viruses are environmentally stable and can remain in the environment even with good hygiene and sanitation. This is consistent with what is known about astroviruses, rotaviruses, and picornaviruses and their relative stability in the environment and highlights the need to reconsider chicken litter reuse. 39
Last, picornavirus reads were detected in the current study. Statistical tests in the current study showed that the detection of picornavirus by NGS was significantly associated with histologic lesions (P < .05). Previous research in Brazil detected numerous reads from picornaviruses and other viruses in pools of 3- to 5-week-old chicken feces presenting clinical signs characteristic of RSS but it was not possible to associate any particular virus with the syndrome. 19 In another metagenomic study from the United Kingdom, picornaviruses were among the most abundant viruses identified at the family level, from both healthy and sick chicks. 10
While these previous metagenomic studies identified picornaviruses in the feces, none had performed in situ investigations. ISH findings in the current study verified gallivirus (Family Picornaviridae) in the mucosa of RSS-affected and unaffected jejunum and in NGS-positive and NGS-negative sections. These animals may lack RSS disease due to relatively minimal galivirus replication. The reasons for the failure to detect gallivirus by NGS in tissues from chicks without lesions remain to be elucidated, but it may be due to the lower amount of virus, as suggested by the ISH association, and the relative insensitivity of NGS from FFPE tissues as compared to RNAScope ISH. While the signal was often localized away from the necrotic areas, the proportion of ISH-positive crypts was significantly higher in chicks with histologic lesions than in those without. Thus, it indicates an association of gallivirus levels and RSS and suggests a possible role in the pathogenesis of RSS.
Conclusions
Histopathology identified lesions that would be consistent with the clinical signs typical of RSS. Growth retardation with weight loss was the major clinical sign in the present study, confirming that there is economic loss for the producer. Histologic changes were absent in 3-day-old chicks and present at 6 days to 11 days of age. The jejunum was most frequently affected, although lesions are also found in the duodenum and ileum. Histologic lesions characteristic of RSS included dilated and necrotic crypts with inflammatory infiltrate mainly composed of mononuclear cells associated with the lesioned crypts. The crypt dilatation occurred after crypt necrosis suggesting it is a result of crypt necrosis. Villous atrophy was also observed and it was more significant in the jejunum and ileum. Therefore, the jejunum from 6- to 11-day-old chicks may be the best segment to diagnose RSS by histopathology. RSS has been associated with several viruses, making it difficult to identify the true cause. While several viruses were identified in this study through NGS, only gallivirus was statistically associated with RSS occurrence based on the presence or absence of histologic lesions. Gallivirus nucleic acid was detected in disease intestine by ISH, and the signal in the intestine was statistically associated with lesion severity. While gallivirus was more often localized outside of necrotic crypts, this association suggests a potential role for a gallivirus levels in the severity of RSS. It is possible that RSS is a multifactorial disease that includes one or more viral causes and as yet unidentified predisposing causes that result in more severe lesions with the viral infection. Thus, it is important to associate molecular tests with histopathology, since viruses may be present in the intestine without associated pathologic changes.
Footnotes
Acknowledgements
We are thankful to Kelsey Young for her technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The first author received a research fellowship from Coordenação de aperfeiçoamento de Pessoal de Nível Superior—Brazil (CAPES)—Finance Code 001, and National Council for Scientific and Technological Development (CNPq). This research was supported by the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG), Project Grant Number APQ-04398-16; the FAPEMIG-University of Georgia Research Mobility Grant Program; and the Department of Pathology at the University of Georgia. This work was also partially supported by the United States Department of Agriculture Clinical Research Information System (Grant 6612-32000-072-00D).
