Abstract
Two groups of six 8-week-old pigs were challenged with 1×109 cfu Brachyspira (Serpulina) pilosicoli or Serpulina intermedia daily for 3 consecutive days to study the pathology of porcine colonic spirochetosis by scanning electron microscopy (SEM) and fluorescent in situ hybridization (FISH) with oligonucleotide probes targeting ribosomal RNA specific for B. pilosicoli and the genus Brachyspira/Serpulina. Six pigs served as noninoculated controls. The animals were euthanatized successively between postinoculation days 14 and 24. B. pilosicoli was reisolated in feces from all of the inoculated pigs; however, only two pigs developed transient watery diarrhea. S. intermedia was reisolated from four of the inoculated pigs, but clinical signs were not observed. Gross examination of the B. pilosicoli–infected pigs revealed dilated large intestines with a hyperemic mucosa, whereas the large intestines of the S. intermedia–inoculated pigs and the control pigs appeared normal. SEM examination of B. pilosicoli–infected pigs revealed degenerated epithelial cells and spirochetal colonization of the colonic mucosa in four pigs. By FISH, B. pilosicoli cells were found colonizing and invading the surface epithelium and the crypts in all the pigs. Spirochetal crypt colonization markedly exceeded the occurrence of spirochetes on the mucosal surface. SEM examination of S. intermedia–inoculated pigs revealed no abnormalities, and Serpulina cells were detected only sporadically in the otherwise normal-appearing mucosa of four pigs by FISH. The results provide further evidence that B. pilosicoli is associated with colitis in pigs, although the gross lesions are mild. The spirochete is capable of colonizing the large intestine, inducing mucosal damage, invasion of the crypt and surface epithelium, and focal infiltration of the lamina propria. In addition, the study shows the applicability of FISH for specific identification of B. pilosicoli in formalin-fixed tissue.
Keywords
Several reports have associated weakly β-hemolytic intestinal spirochetes (WBHIS) with colitis in growing pigs,3,7,9,13,17,18,23,26,35 and the condition is described as porcine intestinal spirochetosis or porcine colonic spirochetosis, whereas swine dysentery is caused by the strongly β-hemolytic spirochete Brachyspira (formerly Serpulina) hyodysenteriae.14,34 Based on phenotypic and genotypic criteria, WBHIS can be divided into four different groups,11,12,25,31,33 and one of these has been assigned to a new species: Brachyspira (formerly Serpulina) pilosicoli.41 This spirochete is commonly isolated from pigs with diarrhea and colitis.12,29,36 Another group of WBHIS, Serpulina intermedia,33 has also been isolated from pigs with diarrhea although exact pathologic changes have not been described.4,12 The genus Serpulina (S. hyodysenteriae, S. pilosicoli, and S. innocens) has recently been unified with the genus Brachyspira.30
Experimentally, spirochetal infection with colitis and diarrhea has hitherto only been induced in pigs after oral inoculation of B. pilosicoli 35,37,39 and a WBHIS classified as Brachyspira (formerly Serpulina) innocens.3 Microscopically, the infection is characterized by a variable increase in the number of mononuclear cells and neutrocytes in the colonic mucosa followed by slight edema. The luminal epithelium may be affected by necrosis, and spirochetes can be found within crypts dilated by mucus. Extensive colonization and attachment of spirochetes by one end to the luminal epithelium is consistently observed in natural infection and has been excellently revealed by scanning electron microscopy (SEM).17 In experimental infections with B. pilosicoli end-on attachment of spirochetes has, however, only been observed focally in a few animals, and there has been no description of SEM changes.7,39 End-on attachment of spirochetes to epithelial cells (intestinal spirochetosis) has also been reported in natural cases of human, avian, and canine intestinal spirochetosis8,10,15,38,42 and has been used as a pathogenicity criterion for human, canine, and porcine isolates of B. pilosicoli in chick models.27,28,40 Thus, the ability of the spirochetes to attach end-on to the luminal epithelium may be an important virulence mechanism.
The objectives of this investigation were to study the pathogenesis of experimental infections with B. pilosicoli and S. intermedia with attention to SEM changes, including end-on attachment of the spirochetes to the epithelium. The association of the spirochetes with the large intestinal mucosa was furthermore studied by fluorescent in situ hybridization (FISH) with oligonucleotide probes targeting ribosomal RNA (rRNA) specific for B. pilosicoli and the porcine members of Brachyspira/Serpulina.
Materials and Methods
Animals and experimental design
Eighteen 8-week-old pigs weighing 20–23 kg (crossbreeds of Danish Landrace and Yorkshire) were obtained from a high-health herd known to be free from Brachyspira (Serpulina) hyodysenteriae. In addition, neither B. pilosicoli nor S. intermedia had been isolated from the herd previously. The animals were housed in separate pens with concrete floors without bedding and were fed a commercial 18.5% protein feed (62% barley, 21% soybean) mixed with 50% crushed wheat. Six animals were inoculated with 1×109 cfu B. pilosicoli by stomach tube daily for 3 consecutive days. Similarly five animals were inoculated with 1×109 cfu S. intermedia, whereas a sixth noninoculated animal was placed in the same pen. The last six animals were kept as nonchallenged controls. The pigs were observed daily for clinical signs of intestinal disease, including a fecal scoring: +++: watery feces; ++: semiliquid feces; +: semisolid feces; and 0: normal feces. Euthanasia and necropsy of the inoculated pigs were performed successively between postinoculation days 14 and 24, whereas the control pigs were euthanatized and necropsied between days 14 and 18.
Microbiology
The B. pilosicoli and the S. intermedia for inoculation were Danish field isolates from two herds with diarrheal problems among 8- to 12-week-old pigs. Spirochetes for inoculation were cultured on fastidious anaerobic agar with 5% bovine blood and suspended in physiologic saline just before inoculation. The isolates were identified biochemically as described by Fellström et al.12 In addition, the identity of the B. pilosicoli isolate was determined by polymerase chain reaction analysis as described by Leser et al.25
Fecal samples of the inoculated pigs were collected daily, whereas a sample of the large intestine was taken at necropsy from all the animals. The samples were inoculated on a selective medium (tryptic soy agar with 5% bovine blood, rifampicin (0.0125 g L−1), spectinomycin (0.2 g L−1), vancomycin (0.05 g L−1), and colistin (0.0125 g L−1). Growth was recorded after 3, 6, and 8 days at 37 C under anaerobic conditions (80% N2, 10% CO2, 10% H2). Subculturing was performed on fastidious anaerobic agar with 5% bovine blood.
Histology
After examination for the presence of gross lesions, tissue samples for light microscopic examination were taken from duodenum, mid jejunum, ileum (20 cm from the ileocecal valve), cecum, proximal colon, and distal colon at the top of the spiral. The tissue samples were fixed in 10% buffered formalin, dehydrated in alcohol, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Sections of the large intestines were furthermore stained by the Warthin-Starry (WS) silver stain method and were used for FISH.
FISH of formalin-fixed tissue sections
Sections of the large intestines were used for FISH with the following oligonucleotide probes: a genus-specific Brachyspira (porcine Serpulina spp.) probe SER1410: 5′-GTCATTCCATCGAAACATA-3′; a specific probe for B. pilosicoli Pilosi209: 5′-GCTCATCGTGAAGCGAAA-3′ (the Brachyspira [Serpulina] probes and their specificity are described by Boye et al.5); a probe general for the bacterial domain EUB338: 5′-GCTGCCTCCCGTAGGAGT-3′ (Amann et al.2); and an unspecific control probe NON-EUB338: 5′-CGACGGAGGGCATCCTCA-3′.1 The labeled probes were purchased from Hobolth DNA Syntese, Hiller⊘d, Denmark. The oligonucleotide probes were synthesized by standard phosphoramidite chemistry. The oligonucleotides were 5′ labeled with an aminohexyl linker and were subsequently conjugated to either fluorescein isothiocyanate (Peninsula Laboratories, Belmont, CA) or the isothiocyanate derivative CY3 (cyanine dye CY3.29-OSu; Biological Detection Systems, Pittsburgh, PA).
Prior to hybridization, the sections were deparaffinated in xylene and kept in ethanol for 10 minutes. Before the hybridization solution was applied, the sections were circumscribed with a hydrophobic PAP-pen (Daido Sangyo Co. Ltd, Tokyo, Japan). The hybridization was carried out at 37 C with 30 μl of hybridization buffer (100 mM Tris [pH 7.2], 0.9 M NaCl, 0.1% sodium dodecyl sulfate) and 75 ng of probe for 16 hours in a moisture chamber. Then the samples were washed in 100 ml of prewarmed (37 C) hybridization buffer for 15 minutes and subsequently in 100 ml of prewarmed (37 C) washing solution (100 mM Tris [pH 7.2], 0.9 M NaCl) for 15 minutes. The samples were rinsed in water, air dried and mounted in Vectashield (Vector Laboratories, Burlingame, CA) for microscopy.
Epifluorescence microscopy
A Leica DMRB microscope equipped for epifluorescence with a 100-W mercury lamp and a double filter set G/R513803 was used to visualize red and green fluorescence simultaneously. Micrographs were taken with a Leica MPS 32 camera using Kodak Ektachrome Elite 400 film.
Electron microscopy
For SEM, tissue samples were taken from cecum, proximal colon, and colon at the flexura sigmoidea. The tissue samples were fixed in a mixture of 75% alcohol, 20% undiluted formalin, and 5% acetic acid. The samples were dehydrated in acetone and critical point dried in a Balzer CPD 020 critical point dryer. The tissue samples were coated under vacuum with gold-palladium using a Polaron SC 7640 Sputter Coater and examined in a JSM 840A scanning electron microscope. After SEM examination, the samples were transferred to 10% buffered formalin and processed for histologic examination.
Results
Clinical signs and microbiology
Diarrhea of variable frequency and severity among the B. pilosicoli–inoculated pigs was observed between postinoculation day 9 and until the last animal was euthanatized at postinoculation day 24, as shown in Table 1. The watery and semisolid feces had no signs of blood or increased amounts of mucus. The feces of the S. intermedia–inoculated and the control pigs remained normal throughout the study period.
Experimental Brachyspira pilosicoli and Sepulina intermedia infections in pigs. Feces score at different postinoculation days and culturing of Brachyspira/Serpulina from the feces.

∗s = reisolation of B. pilosicoli or S. intermedia from feces, s∗ = isolation of B. innocens; +++ = watery feces, ++ = semiliquid feces, + = semisolid feces; X = euthanasia; § = euthanasia due to lameness.
Microbiologically, B. pilosicoli was reisolated in feces from all of the inoculated pigs (Table 1). Reisolation from five of the six pigs between postinoculation days 2 and 4 probably represented a normal transit of the intestine. Thereafter B. pilosicoli was frequently reisolated from the pigs between postinoculation days 8 and 24, indicating spirochetal colonization of the large intestine of the pigs. S. intermedia was only sporadically reisolated from four of the inoculated pigs between postinoculation days 7 and 17, thus indicating colonization of the large intestine. In addition, B. innocens was isolated once from Nos. 8 and 9. WBHIS were not isolated from the control animals.
Gross findings
At necropsy, the stomachs of both the inoculated and the control pigs were normally filled by feed, and the small intestines appeared normal. The large intestines of the B. pilosicoli–inoculated pigs were slightly hyperemic, distended by gas and watery to semisolid contents. The muscular tonus of the large intestinal wall was increased. However, increased thickness of the wall was not evident. The colonic content was greenish to greenish-gray and without evidence of blood or increased mucus production. The colonic mucosa of Nos. 4 and 5 appeared glistening, with intensive focal hyperemia and with the presence of small (3–5 mm), adherent solid nodules of plant material. The focal hyperemia and the plant nodules were mainly dispersed throughout the mid spiral region of colon. Necrosis was not observed in the hyperemic mucosa or in the mucosa beneath the nodules after removing the plant material.
The gross lesions in the large intestines of the other B. pilosicoli–inoculated pigs were similar although the extension and seriousness were less severe. The colonic lymph nodes were of normal size and consistency. Gross lesions in other organs were not observed.
The large intestines of the S. intermedia–inoculated and the control pigs appeared without abnormalities and without adherent nodules of plant material.
Histology and FISH
The pathologic findings in the pigs, including FISH, are summarized in Table 2. Histopathologically, spirochetal colitis of variable severity was present in all segments of the colon of the B. pilosicoli–inoculated pigs although the lesions were most prominent in the mid spiral region, while the cecal and distal colonic mucosa appeared normal. The lesions were most significant and extensive in Nos. 4 and 5 and were morphologically characterized by focal erosions and increased extrusion of the surface epithelial cells, slight edema and infiltrate of mononuclear cells in lamina propria, and elongated crypts with loss of epithelial cells as shown in Fig. 1 compared with the large intestine of the control pigs, which all appeared normal (Fig. 2). WS stain revealed spirochetes colonizing the mucosa and mucus layer. The spirochetes were identified as B. pilosicoli by FISH in all the examined sections using probe Pilosi209. Areas of the mucosa from Nos. 4 and 5 with multiple focal erosions and severe sloughing of epithelial cells were characterized by massive occurrence of B. pilosicoli colonizing the epithelial surface and the mucus layer. The spirochetes were randomly oriented to the surface and invading the intercellular spaces between degenerated epithelial cells down to the basement membrane (Fig. 3). Similar but less intensive epithelial changes and sparse spirochetal colonization of the mucosal surface were seen in other areas of colon corresponding to the macroscopic mild hyperemia in Nos. 2–5, although the crypt openings were filled with spirochetes (Fig. 4). An evident palisade-like end-on attachment of densely packed spirochetes to the luminal epithelium was observed only in pig No. 5 between two crypt openings. The area of the mucosal surface with end-on attachment was found underneath adherent food nodules. A sporadic colonization by B. pilosicoli cells and moderate elongation of the crypts characterized the changes in the mucosa with only slight macroscopic signs of colitis. B. pilosicoli cells in the mucus layer on the luminal surface were seen rarely in Nos. 1 and 6.
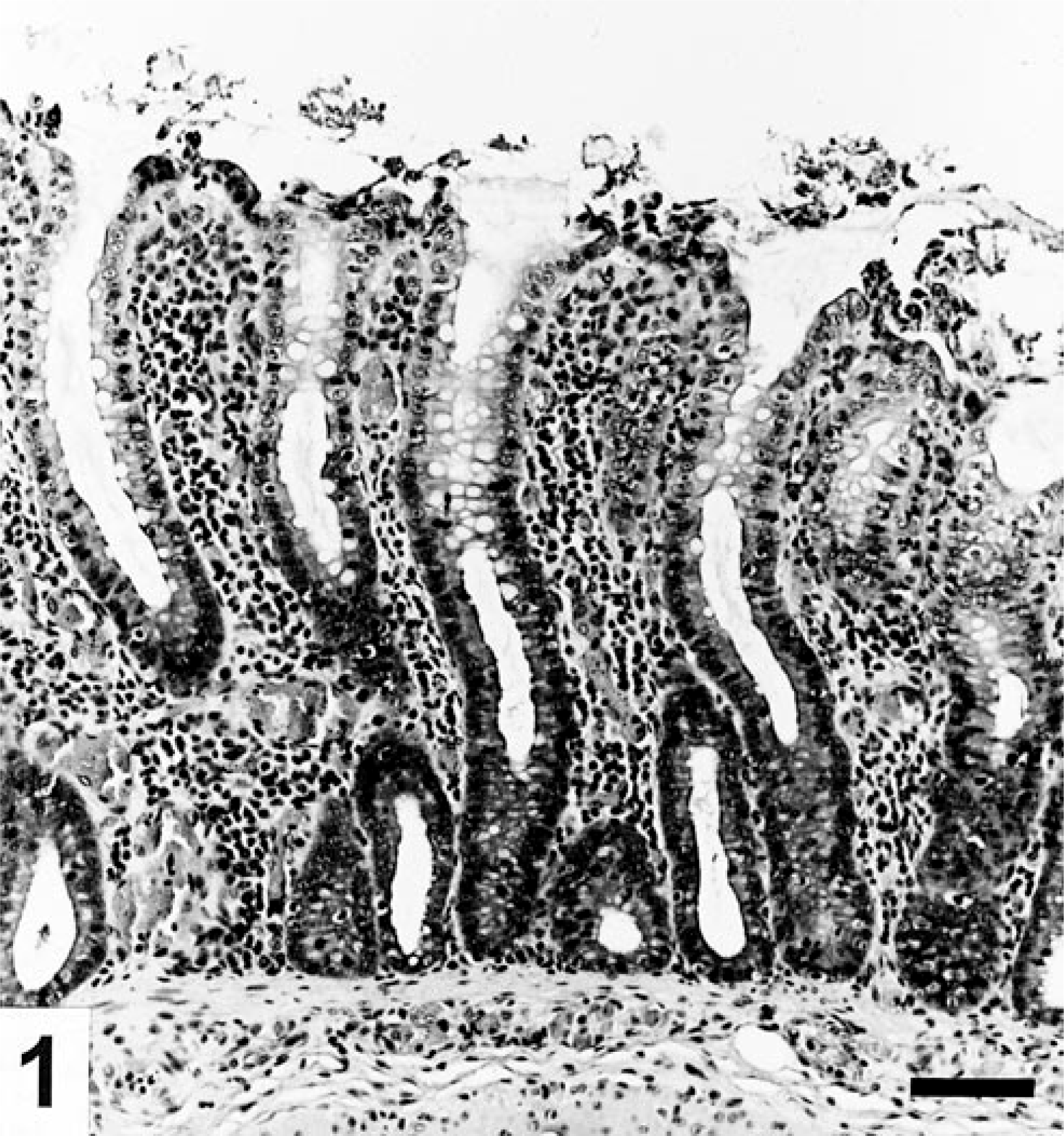
Colon; pig No. 4 inoculated with B. pilosicoli. Catarrhal colitis with degeneration and necrosis of epithelial cells in the extrusion zone between the crypt openings, elongation of the crypts, and a predominant mononuclear cell infiltrate in lamina propria. HE. Bar=50 μm.
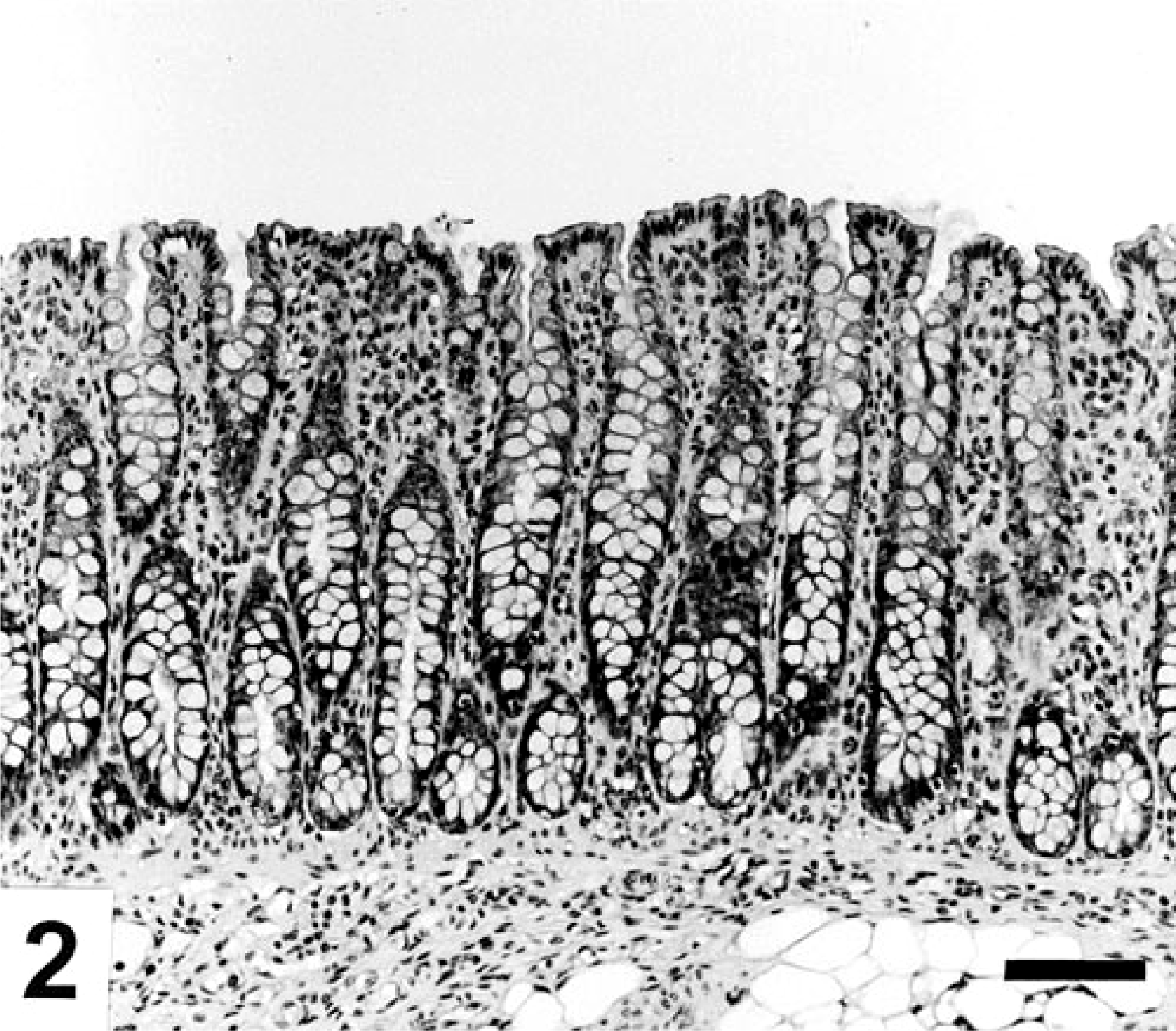
Colon; noninoculated pig with a normal appearing mucosa. HE. Bar=50 μm.
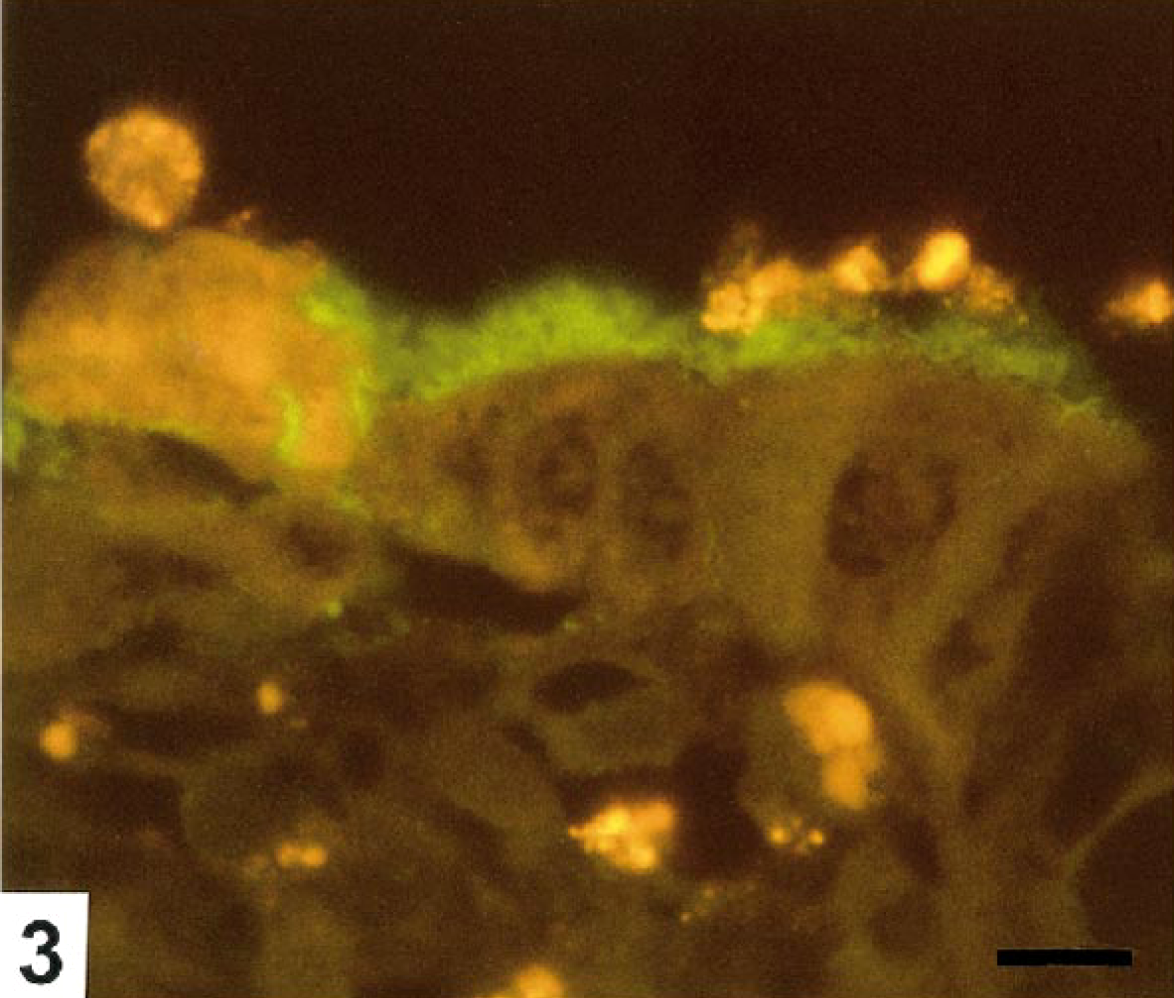
Colon; pig No. 4. Large numbers of randomly oriented B. pilosicoli (green) colonize the surface epithelium, the mucus layer, and necrotic epithelial cells in an extrusion zone. FISH. Bar=10 μm.
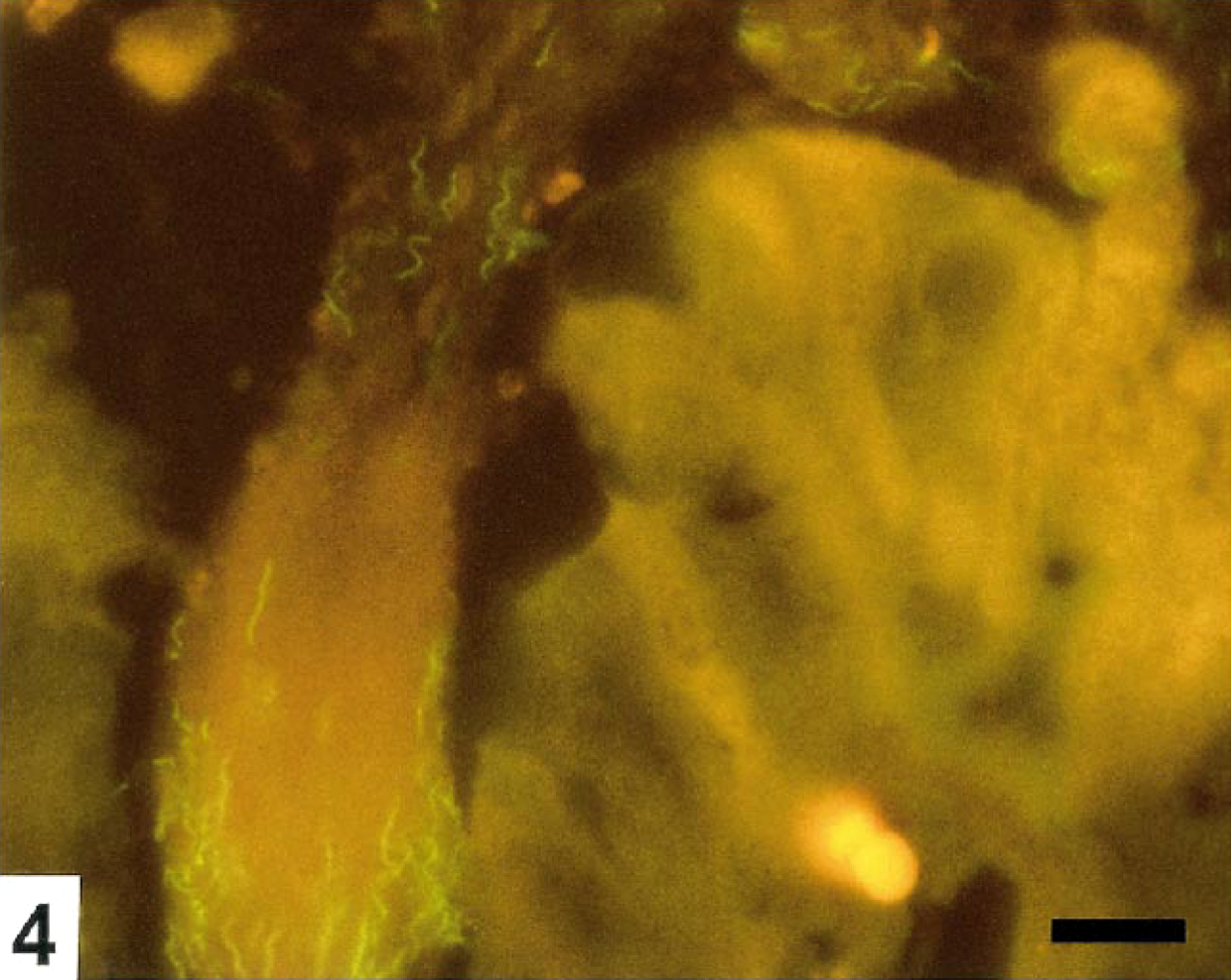
Colon; pig No. 5. Moderate number of B. pilosicoli colonizing the mucus in a crypt opening. A few necrotic cells are seen in the surface epithelium. FISH. Bar=10 μm.
Experimental Brachyspira pilosicoli and Serpulina intermedia infections in pigs. Pathology and FISH.∗
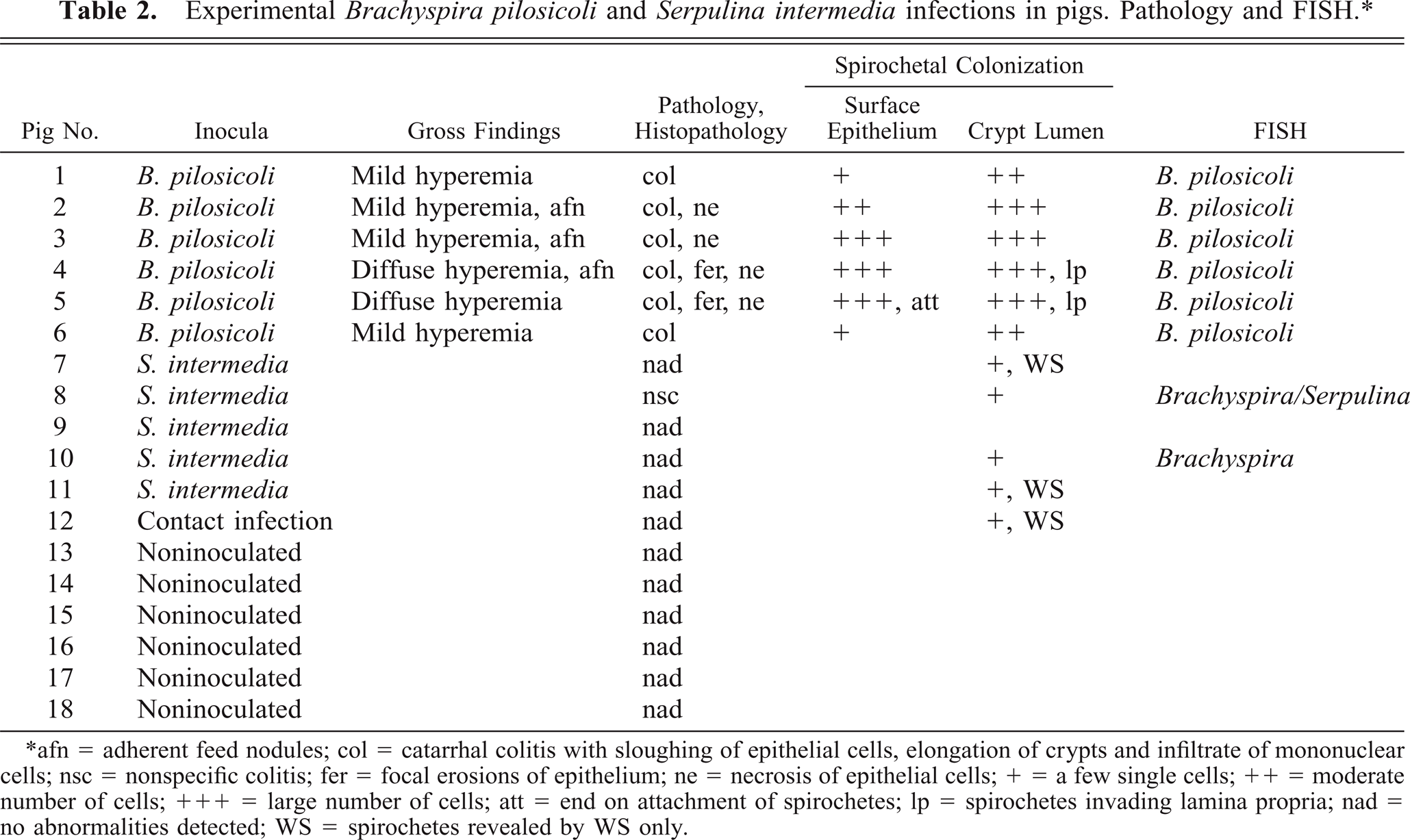
∗ afn = adherent feed nodules; col = catarrhal colitis with sloughing of epithelial cells, elongation of crypts and infiltrate of mononuclear cells; nsc = nonspecific colitis; fer = focal erosions of epithelium; ne = necrosis of epithelial cells; + = a few single cells; ++ = moderate number of cells; +++ = large number of cells; att = end on attachment of spirochetes; lp = spirochetes invading lamina propria; nad = no abnormalities detected; WS = spirochetes revealed by WS only.
The colonic crypts were diffusely hyperplastic and elongated, slightly to moderately dilated, and lined by columnar to cuboidal epithelium. B. pilosicoli diffusely colonized the mucus, even deep in the crypts. Invasion of degenerated and necrotic crypts cells by single or small clusters of B. pilosicoli were seen together with invasion of the viable crypt epithelium along the lateral cell boundaries. Figure 5 illustrates invasion of the epithelium in a moderately B. pilosicoli colonized crypt. The crypt changes and colonization by B. pilosicoli were observed throughout the mucosa but most intensively in the highly hyperemic areas of the colons of Nos. 4 and 5. The degree of the crypt colonization far exceeded the presence of B. pilosicoli on the mucosal surface. The mucosal invasion by B. pilosicoli was usually restricted to the epithelium; however, in areas with erosions and under the adherent food nodules, the spirochetes were commonly found infiltrating the adjacent superficial parts of the lamina propria (Fig. 6), which in addition were heavily infiltrated with monocytes, lymphocytes, plasma cells, and a moderate number of neutrocytes. Accumulation of neutrocytes and other cellular debris in the crypts were rare.
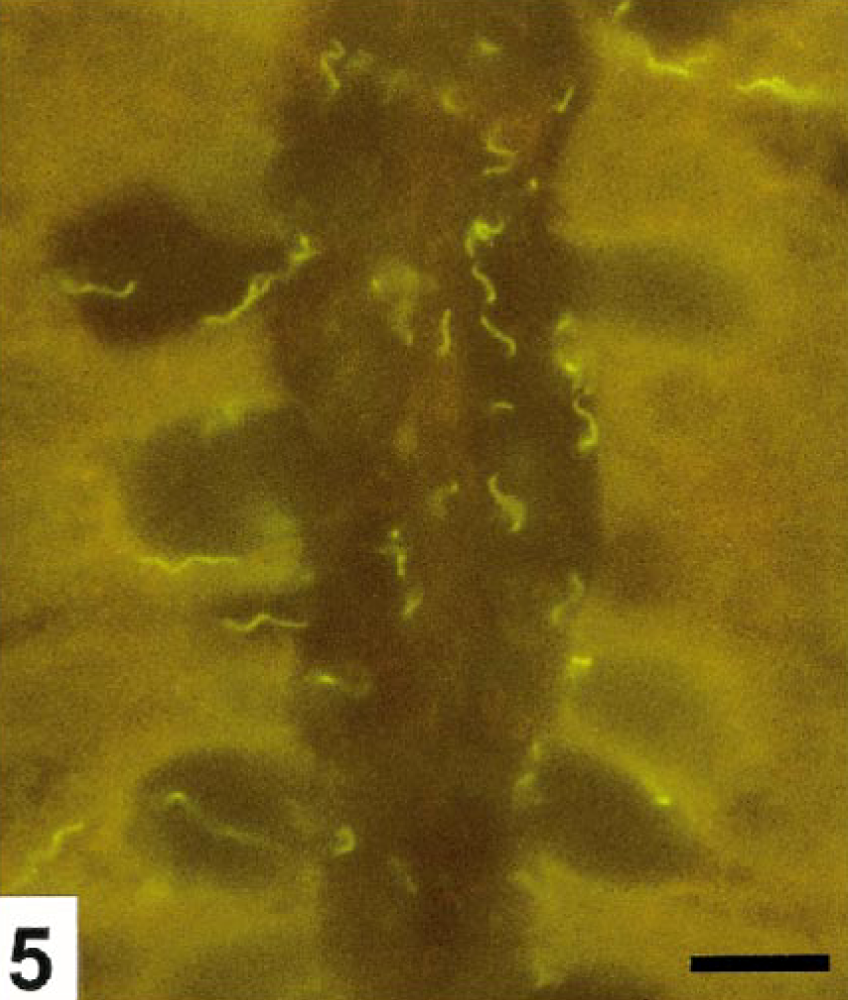
Colon; pig No. 5. Crypt colonized by B. pilosicoli. The spirochetes are seen within goblet cells and invading the crypt epithelium along the lateral cell membranes. FISH. Bar=10 μm.
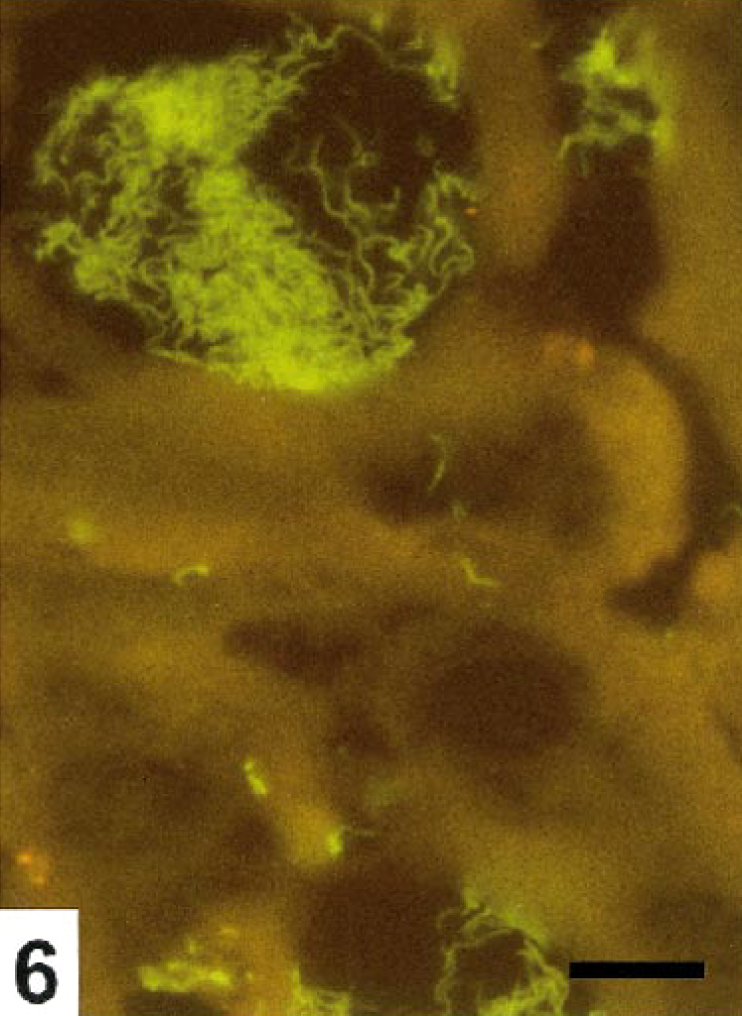
Colon; cross-sections of crypts, pig No. 4. B. pilosicoli excessively colonizes the crypts and infiltrates the adjacent lamina propria. FISH. Bar=10 μm.
Histologic examination of the S. intermedia–inoculated pigs revealed no significant pathologic changes. The crypt depth and the epithelial lining in the large intestines were similar to that observed in the control pigs. However, single spirochetes were occasionally seen in the crypts of all the S. intermedia–inoculated pigs, and in one case, a small cluster of spirochetes was found in a crypt but without epithelial changes. The spirochetes hybridized only with the SER1410 probe (data not shown), thus indicating that they belonged to the genus Serpulina but were different from B. pilosicoli. Spirochetes were not observed in the large intestines of the control animals either by WS staining or by FISH with the probe SER1410.
SEM
Focal erosions and other epithelial lesions were seen in areas of the colon corresponding with the hyperemia found postmortem in the B. pilosicoli–infected pigs. In the extrusion zones between the crypts, dome-shaped degenerated and necrotic epithelial cells with sparse and disrupted microvilli were bulging from the surrounding epithelium (Fig. 7). Varying numbers of uniform, slender spirochetes were seen in crypt openings and colonizing extrusion zones in these parts of the colon in four animals (Nos. 2–5). However, intensively colonized crypt openings were exclusively seen in the midspiral region of colon. Multiple spirochetes were seen in close contact to necrotic epithelial cells and were invading the intercellular space between necrotic and degenerated cells (Fig. 8), although epithelial necrosis was also observed in areas with only a moderate number of spirochetes (Fig. 9). In erosions with complete loss of epithelial cells between the crypt openings, the spirochetes formed a mat of randomly oriented cells on the denuded basement membrane. Spirochetal colonization of the luminal epithelium was consistently correlated to a concurrent intensive colonization of the crypts by B. pilosicoli cells as also demonstrated by FISH. In addition to the spirochetal colonization of the extrusion zones, spirochetes were found longitudinally on the surface of the epithelium, including halfway down in goblet cells in the crypts. Even in areas with thousands of spirochetes in the crypt openings, the surrounding epithelium was usually only colonized by a low number of longitudinally lying spirochetes, and specific end-on attachment of spirochetes to the luminal epithelium was not verified by SEM in any of the pigs. Large areas of the cecal and colonic mucosa with only slight macroscopic signs of typhlitis or colitis were covered by a thin mucus layer, and the epithelium appeared intact with only occasional evidence of a few degenerated cells in the extrusion zones in those parts of the mucosa free from mucus. Scanning of the mucosa after removing the nodular plant material from the fixed tissue samples was impossible.

Colon; mucosa, pig No. 5 inoculated with B. pilosicoli. Multiple, dome-shaped degenerated and necrotic epithelial cells (★) in the extrusion zones between the crypt openings. Slender spirochetes (arrows) are seen in the crypt openings and colonizing the extrusion zones with necrotic epithelial cells. SEM. Bar=20 μm.
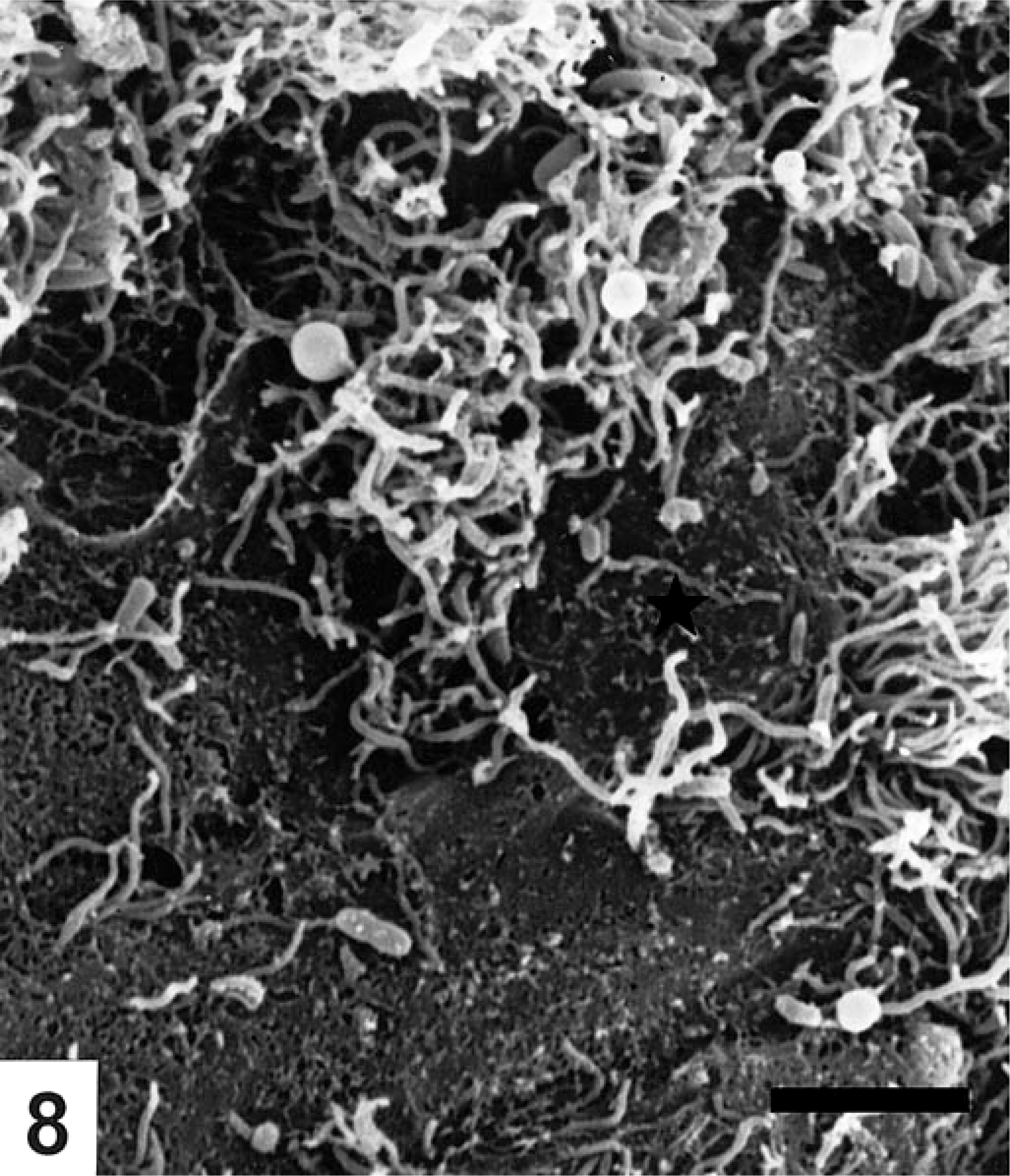
Multiple spirochetes invade the intercellular space around a degenerated epithelial cell with disrupted microvilli (★). Colon from pig No. 4 inoculated with B. pilosicoli. SEM. Bar=5 μm.
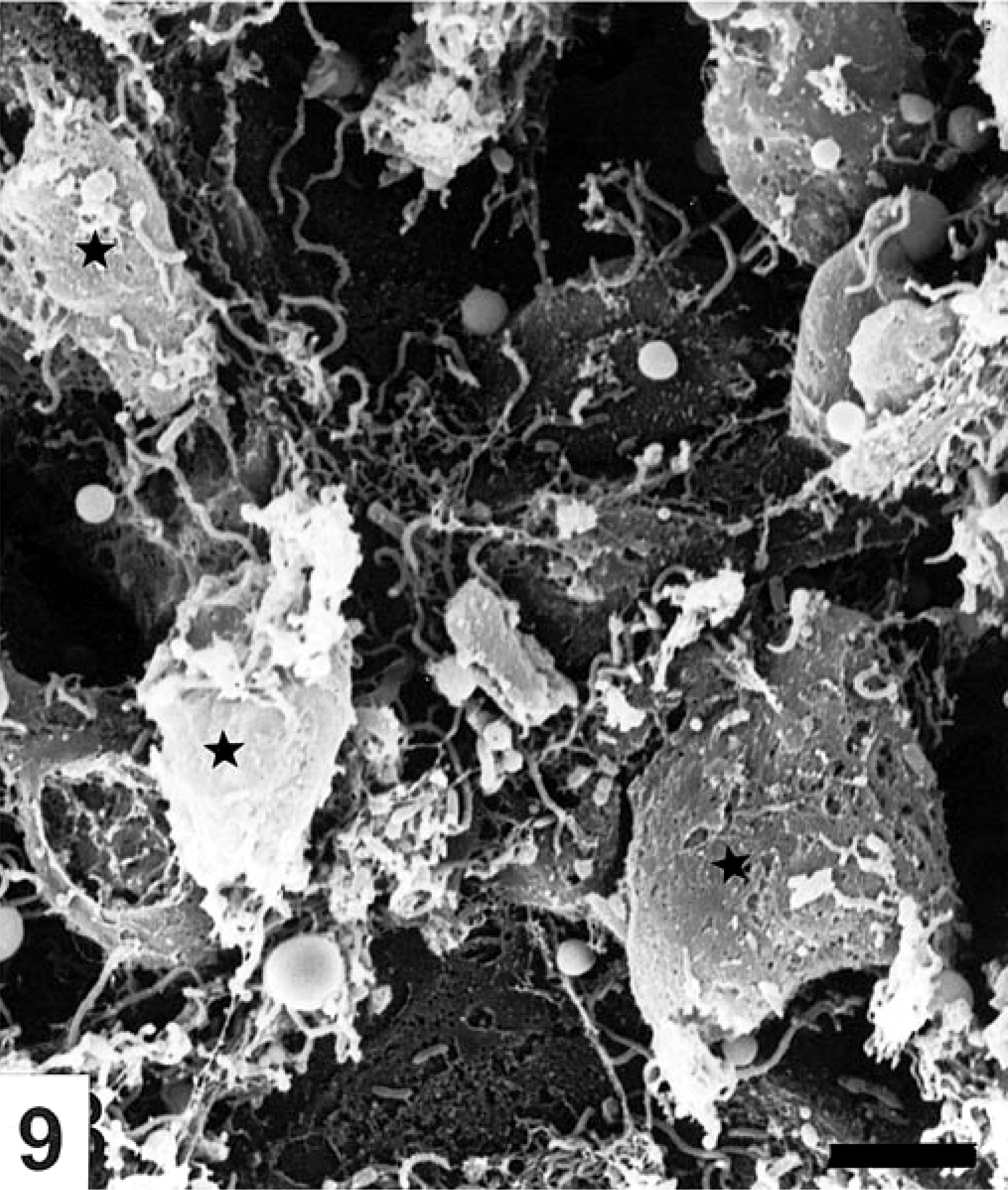
Degeneration and extrusion of epithelial cells (★) in an area with only a moderate number of spirochetes. Colon from pig No. 4 inoculated with B. pilosicoli. SEM. Bar=5 μm.
The large intestinal mucosa of the S. intermedia–inoculated and control pigs appeared uniform and without abnormalities by SEM. Spirochetes were not observed.
Discussion
The spirochete B. pilosicoli has recently emerged as a significant problem in intensive pig-farming systems worldwide.7,11,29,36,41 Results of this study demonstrate that oral inoculation of growing pigs with a Danish isolate of B. pilosicoli can induce clinical signs and macroscopic changes similar to those observed in naturally occurring infection and further confirm the pathogenicity of B. pilosicoli as shown in a few previous experimental studies from the UK, Australia, and the USA.7,35,36,37,39 Thus, detection of the spirochete in situ or by culture from cases of naturally occurring porcine colitis is likely to be significant, particularly in the absence of other intestinal pathogens.
The incubation period of between 9 and 24 days in the clinically affected animals was longer than that reported previously (3–9 days).7,35,37 Challenging 5-week-old pigs with an Australian isolate of porcine B. pilosicoli, Trott et al.41 observed diarrhea developing on the second postinoculation day in two of 12 animals followed by diarrhea in another two animals on postinoculation days 8 and 10. When necropsied on postinoculation day 11, moderate microscopic changes were observed only in the four pigs that developed diarrhea. In the present study only transient watery diarrhea in two pigs was observed between postinoculation days 9 and 20 among all the B. pilosicoli–infected pigs. In the study by Trott et al.41 and in our study, each pig was dosed with approximately 1×109 to 1×1010 cfu B. pilosicoli daily for 3 consecutive days. This is an infective dose that probably far exceeds the amount of spirochetes ingested in natural infection. A comparable dose of B. hyodysenteriae usually causes serious mucohemorrhagic diarrhea within 5 to 9 days after inoculation.14,20,34,35
The gross lesions in pigs with B. pilosicoli infection are relatively mild compared with those occurring in swine dysentery, although diphtheritic lesions, multifocal erosions, and mucoid colitis have been described in previous experimental studies.35,37,39 Even though significant morphologic changes were present microscopically, the most serious gross lesions in this study were hyperemia of the mucosa. Similarly, absence of significant gross lesions has been reported in field cases indicating that histologic examination of colons from pigs with diarrhea is relevant in cases that lack gross lesions.17,32
SEM examination showed significant B. pilosicoli infection in four pigs. Spirochetal colonization and attachment to the colonic surface began along the extrusion zone in the intercrypt areas, sometimes proceeding to a coalescing spirochetal mat on the denuded basement membrane in areas with epithelial erosions. Spirochetal invasion of the epithelium was seen in the intercellular junctions between necrotic and degenerated cells, whereas end-on attachment of the spirochetes to the mucosal surface was not obvious. Similar spirochetal colonization of the mucosal surface has been reported in SEM studies on natural porcine,17,32 human,15 avian,38 and canine42 intestinal spirochetosis and in chick models with B. pilosicoli 27,28,40; however, in these reports clusters or more diffuse masses of spirochetes in end-on attachment to the epithelium were predominant. To ensure the absence of end-on attachment and that the spirochetes were not detached during the SEM procedure, the observations were compared with the spatial distribution of B. pilosicoli as seen by FISH. Even the huge masses of spirochetes on the mucosal surface appeared to be randomly oriented, and differences in surface colonization between the FISH and SEM observations were not confirmed. End-on attachment was only seen by FISH between two crypt openings in one pig. Furthermore, the comparison demonstrated that the colonization of the crypts was more extensive and significant than surface colonization. B. pilosicoli colonized the mucus in the colonic crypts and caused colitis in all six inoculated pigs. The spirochetes were observed invading the crypt epithelium, particularly the goblet cells, causing cell degeneration, necrosis, and inflammation and initiating regenerative epithelial hyperplasia resulting in crypt elongation similar to mucosal and cryptal changes described for experimental swine dysentery.1,14,20 Evidence of spirochetal invasion of the crypt epithelium has been observed in experimental B. pilosicoli infection37 in pigs, but not as commonly as it has been reported in natural infections.17,18,32 In previous challenge studies, B. pilosicoli–associated colitis has histologically been observed in 25 (4/12) and 83% (15/18 and 5/6) of the pigs.35,37,39 Variation in the phenotype of B. pilosicoli, diet, and origin of the pigs are possible factors that may influence the outcome of the infection. In swine dysentery, B. hyodysenteriae colonizes the intestinal mucosa by association with intestinal mucus with no specific orientation or attachment to the epithelial surface1,20; however, Glock and Harris14 found spirochetes embedded in and invading through the apical surface of epithelial cells. While end-on attachment of spirochetes (B. pilosicoli) to the luminal epithelium forming a false brush border has previously almost consistently been reported in natural colitis, the same phenomenon has been observed in only a few pigs with experimentally induced B. pilosicoli infection.7,39 We observed focal end-on attachment in only one pig, and in a large survey of field cases of porcine colitis, Thomson et al.36 did not find end-on attachment of spirochetes in pigs from 44 herds with evident spirochetal colitis and isolation of B. pilosicoli, indicating that the attachment phenomenon is uncommon. Besides the spirochetal colonization of the mucosa, a concurrent infection with the protozoan Balantidium coli, which has been associated with diarrhea and colitis in humans, is frequently reported in natural as well as in experimental infection, and it is possible that the organism may facilitate the development of mucosal lesions.7,24,35,39
Experimental infection with an isolate of S. intermedia resulted in reisolation of the organisms from four of five animals between days 7 and 15 after inoculation, indicating that transient colonization of the intestines succeeded, but neither clinical signs nor macroscopic or microscopic morphologic changes were observed during the study. Little information is available in the literature concerning the association of S. intermedia with colitis, but the spirochete has occasionally been isolated from growing pigs with diarrhea.4,12 S. intermedia has also been reported from chickens in natural as well as experimental infections.10 Thus, the importance of S. intermedia in the pathogenesis of porcine colitis and in colitis of other species has to be investigated further.
We used FISH targeting rRNA for in situ detection of B. pilosicoli and spirochetes belonging to the genus Serpulina as previously described.5 Oligonucleotide probes targeting rRNA are attractive because rRNA is naturally amplified in growing cells.6 The result of this study showed further the applicability of the method for identification of B. pilosicoli in formalin-fixed samples. Specific identification or distinguishing between the different spirochete species in situ, e.g., in pathogenicity studies and as a diagnostic tool, is essential because of the occurrence of apathogenic spirochetes (B. innocens, Serpulina murdochii) in the large intestine of normal, healthy pigs.19,21,33 Thus, demonstration of silver-stain–positive spirochetes in the lumen of the intestine, even with colitis, is only suggestive of porcine colonic spirochetosis.7 Moreover, concomitant colonization of the large intestine with more than one Serpulina species has been reported, and an accurate identification of the different spirochetes in situ is desirable.12,18,29 Distinction between intestinal spirochetes based on morphologic criteria is possible using transmission electron microscopy, but the ultrastructural differences are subtle and may not be reliable diagnostic aids in clinical specimens.16 Due to strong cross-reactions between the different species of Serpulina, development of immunohistochemical methods based on monoclonal antibodies for specific detection of spirochetes in situ is difficult.22,43 By the FISH technique, even single cells present in or on the mucosa can be detected in formalin-fixed tissue samples used for histology. This study shows the applicability of the method for identification of B. pilosicoli in pathogenesis studies.
Footnotes
Acknowledgements
We thank A. R. Pedersen, U. L. Andreasen, and S. M. Jensen for technical assistance. This work was supported by the Research Secretariat of the Danish Ministry of Food, Agriculture and Fisheries, project number SUN94-3 and MIL97-SVS-1.
