Abstract
Enterotoxemia caused by Clostridium perfringens type D is one of the most prevalent clostridial diseases of sheep. The lesions of the acute form of this disease, particularly the cerebral lesions, are well characterized; however, detailed descriptions of the cardiac and pulmonary lesions are lacking. Here we describe cardiopulmonary lesions in experimental acute type D enterotoxemia in sheep and determine the role of epsilon toxin (ETX) in the development of these lesions. Four groups of 6 sheep were intraduodenally inoculated with either a wild-type C. perfringens type D strain; its etx knockout mutant, which is unable to produce ETX; the etx mutant complemented with the wild-type etx gene, which regains the ETX toxigenic ability; or sterile culture medium as a control. All sheep were subjected to postmortem examination within 24 hours of inoculation. Lesion scores were compared between groups for pulmonary edema; hydrothorax; ascites; hydropericardium; endocardial, myocardial and epicardial hemorrhages; microscopic lesions of acute myocardial degeneration and necrosis; and myocardial, endocardial, and epicardial edema, hemorrhage, and inflammation. Only sheep inoculated with the wild-type and complemented ETX-toxigenic bacterial strains developed cardiopulmonary lesions, which were present in varying degrees of severity and proportions. These lesions were not present in sheep inoculated with the etx mutant or in the negative control. We conclude that severe acute cardiopulmonary lesions in sheep with experimental enterotoxemia are associated with the capacity of the strains to produce ETX. These changes are likely contributors to the clinical signs and even death of affected animals.
Enterotoxemia caused by Clostridium perfringens type D is one of the most prevalent and economically important clostridial disease of domestic small ruminants. 26,28 C. perfringens type D produces alpha (CPA) and epsilon (ETX, encoded by the etx gen) toxins. 19,20,24 ETX is essential for virulence and responsible for the morbidity, mortality, and lesions caused by this agent in naturally susceptible hosts and rodent models. 9 Even though there is a good understanding of most lesions caused by ETX in ruminants, there are some knowledge gaps in the pathogenesis of type D enterotoxemia. ETX is produced in the intestine by C. perfringens type D during the exponential phase of bacterial growth. Abrupt dietary change, usually to rations rich in rapidly fermentable carbohydrates or lush forages, 28 has been regarded as the main predisposing factor for bacterial proliferation and ETX production in the intestinal environment. ETX is secreted into the intestinal contents as a poorly active prototoxin, which is then enzymatically cleaved into ETX by trypsin and other proteases, and/or by C. perfringens lambda protease; this cleavage increases its toxicity about 1000 times. 17 Additionally, some strains of C. perfringens are capable of activating ETX intracellularly by lambda-independent mechanisms and the active toxin can be released upon bacterial lysis in the stationary phase of bacterial growth. 12
The outcomes of type D enterotoxemia vary in different ruminant species. In goats, one of the major lesions caused by ETX is necrotizing enterocolitis. However, ETX exerts little if any histologically detectable changes in the intestine of sheep 28 or cattle. 6 Once produced in the intestine, ETX is absorbed through the intestinal wall and distributed systemically. Absorbed ETX specifically binds to an unidentified receptor on vascular endothelial cells and possibly other cell types, as well as myelinated nerve fibers of the central and peripheral nervous system. 5 Although the ETX receptor has not been definitely identified, the “myelin and lymphocyte” (MAL) protein has been proposed either as the ETX receptor or as part of the ETX receptor complex, as it is required for cellular binding and cytotoxic activity of ETX in hamster and canine cells in vitro and mice in vivo. 22 Whether this is the case in ruminants, the only animals in which spontaneous type D enterotoxemia is known to occur, has not yet been explored.
At the cellular level, ETX binding to the receptor results in heptamerization and formation of a transmembrane pore with cytotoxic effects that, when it occurs in endothelial cells, results in increased vascular permeability and edema. 8,17,29 In the brain, this perivascular and intramural vascular edema results in increased intracranial pressure and mechanical and hypoxic or ischemic damage to brain tissue that is thought to be responsible for the neurologic signs seen in sheep and cattle, and more rarely in goats with type D enterotoxemia. 10,18 However, a direct pore-forming action of ETX in other cell types is likely. Because of the systemic distribution of ETX, and the ubiquity of vascular endothelial cells throughout the organ systems, this toxin could potentially damage any vascularized tissue. However, lesions in type D enterotoxemia in sheep are seen most consistently in certain parts of the brain, and in the lungs and pericardial sac, 28 which suggests that the endothelial cell receptor for ETX might not be evenly distributed throughout the vasculature.
Although the lesions of acute type D enterotoxemia in sheep, 25 and particularly the cerebral lesions induced by ETX, 8,10 have been relatively well characterized, detailed descriptions of the cardiac and pulmonary lesions are lacking in the scientific literature. The aim of this work is to describe cardiopulmonary lesions in experimental acute type D enterotoxemia in sheep, induced by a wild-type C. perfringens type D strain and by its genetically modified derivatives, and to describe the association of ETX-producing strains with cardiopulmonary lesions. These findings complement our previous report on the neurologic lesions caused by these bacterial strains 10 and broadens the knowledge of heart and lung lesions produced by this agent.
Materials and Methods
Animals Experimental Inoculations
For this study, we used autopsy records, gross photographs, and formalin-fixed, paraffin-embedded hearts and lungs available from a previous study that involved experimental inoculation of sheep with isogenic strains of wild-type C. perfringens type D and 2 etx mutants. Details of the bacterial isolates, mutant preparation and the inoculation procedures have been published elsewhere. 9,13 Briefly, 24 sheep were randomly divided into 4 groups of 6 animals each. For 3 groups, animals were inoculated intraduodenally with 1 of 3 isogenic strains of C. perfringens type D: the wild-type strain CN1020 (group 1, sheep 1–6); its etx knockout mutant JIR4981, which is unable to produce ETX (group 2, sheep 7–12); or the etx mutant complemented in trans with the wild-type etx gene (JIR12604), which regained the ability to produce ETX (group 3, sheep 19–24). For the negative control group, sheep were inoculated with sterile tryptone glucose yeast agar medium (group 4, sheep 13–18). All procedures involving live animals were approved by the University of California–Davis Institutional Animal Care and Use Committee (Permit 16383).
After inoculation, all sheep were observed periodically until they died spontaneously or were euthanized due to severe clinical signs, which included tachypnea, dyspnea, ataxia, lethargy, depression, recumbency, blindness, paddling, and spastic convulsions. Sheep that survived to the end of the experiment, approximately 24 hours post-inoculation, were euthanized. Most animals inoculated with the wild-type strain (5/6, sheep 1–2 and 4–6), and with the etx-complemented strain (3/6 animals, sheep 19–21) developed clinical disease. None of the 12 sheep inoculated with the etx knockout strain or sterile culture medium developed clinical disease or died. 9 Details of sample collection and processing and results of the survival times, mortality, and overall clinical and pathological findings are published elsewhere, 9 and the neuropathological findings have been reported. 10
Gross Examination
A full autopsy was performed on every sheep and the following 7 gross lesions were scored: pulmonary edema, hydrothorax, ascites, hydropericardium, endocardial hemorrhage, myocardial hemorrhage, and epicardial hemorrhage. Lesions were scored based on their degree of severity, as absent (score 0), mild (score 1), moderate (score 2), and severe (score 3). The gross lesion scoring criteria are summarized in Supplemental Table S1.
Histopathologic Scoring: Heart
Formalin-fixed, hematoxylin and eosin (HE)-stained sections of heart and lung were examined and scored by one of the authors of this article (FG) in a blinded fashion. Two sections of left ventricular papillary muscles were obtained from each of the sheep in groups 1, 2, and 4. For animals in group 3, the following 11 samples of heart 4 were collected: sinoatrial node region, atrioventricular node region, pulmonary artery at the site of insertion in the right ventricular wall and pulmonary semilunar valve, right ventricular free wall and the parietal cusp of the tricuspid valve, right atrial appendage, left atrial appendage, left ventricular free wall and the parietal cusp of the bicuspid valve, left ventricular primary papillary muscle, left ventricular secondary papillary muscle, interventricular septum, and aortic semilunar valve at the site of insertion in the left ventricular wall.
The following 6 microscopic changes in the myocardium, endocardium, and/or epicardium were assessed on each heart section: acute cardiomyocyte degeneration or necrosis, edema, hemorrhage, inflammatory infiltrate (including the predominant type of inflammatory cells when present), fibrosis, and the presence of intrasarcoplasmic protozoal cysts. These microscopic changes were scored based on their severity and frequency from 0 to 3, in which score 0 indicated no lesions; score 1 indicated lesions that were mild, either focal or multifocal, but infrequent; score 2 indicated lesions that were moderate and/or multifocal but frequent; and score 3 represented severe and/or widespread lesions. Myocardial degeneration or necrosis was assessed as a continuum of the same category as follows: cardiomyocyte degeneration (ie, segmental sarcoplasmic hypereosinophilia and loss of transverse striations with nuclear hyperchromasia) was assigned a score of 1, and cardiomyocyte necrosis (ie, sarcoplasmic coagulation or fragmentation with nuclear pyknosis or karyorrhexis, with or without sarcoplasmic mineralization) were assigned scores of 2 or 3. The criteria used for scoring the main microscopic lesions are summarized in Supplemental Table S1.
Phosphotungstic acid hematoxylin (PTAH) stain and troponin C immunohistochemistry (IHC) were performed on selected heart sections of 8 animals from groups 1 (sheep 1, 4, and 5), 2 (sheep 7), 3 (sheep 19, 20, and 22), and 4 (sheep 13). For troponin C IHC, antigen retrieval was accomplished by incubating the slides in 0.1% protease (Sigma, P5147) at 37 °C for 15 minutes. A mouse monoclonal IgG2a antibody directed against human cardiac troponin C (clone #M5092922, catalogue 10-T78A, Fitzgerald Industries International) was used as a primary antibody. Detection used biotinylated goat anti-mouse antibody (Vector, BA9200) and ABC horseradish peroxidase kit (Vectastain Elite, Vector Labs, PK6100), with the substrate chromogen 3-amino-9-ethylecarbazole (AEC, Lab Vision, Thermo Scientific, TA-125-SA). Slides were counterstained with hematoxylin. All sections were used as internal positive controls, and serial sections that were incubated with normal mouse serum instead of the primary antibody were used as negative controls.
Lung
Sections of the caudal lung lobes from all the sheep were examined histologically and the degree of alveolar and interstitial edema (the latter including edema of the interlobular septa, perivascular and peribronchiolar connective tissue, and pleura) was assessed using a 4-tier grading system similar to that used for the heart.
Statistical Analyses
To assess differences in the proportions of sheep with or without lesions, one-tailed Fisher’s exact test was used (http://quantpsy.org/fisher/fisher.htm). Statistical analyses to assess differences between the scores of gross and microscopic lesions were performed using rank-based nonparametric tests, considering that parametric assumptions were not met. To assess differences in the scores for gross lesions, as well as the histologic lesions in the heart and lungs between all 4 groups (intergroup comparison), the Kruskal-Wallis test was applied, followed by the Dunn test for multiple pairwise comparisons. For the intergroup comparison, only the histologic lesions found in the 2 sections of left ventricular papillary muscles were used in group 3, because the other 9 cardiac regions examined in this group were not available in groups 1, 2, and 4. Thus, the intergroup comparison involved the same cardiac region in all sheep. The differences in the histologic scores for the cardiac lesions in the 11 different heart regions of sheep of group 3 (within-subject comparison) were analyzed by the Friedman test followed by the Wilcoxon signed-rank test for multiple pairwise comparisons. Results were expressed as median (minimum – maximum). Last, in the groups in which histologic lesions were observed (groups 1 and 3), the Spearman’s correlation coefficient (r) was calculated to estimate the association between various gross and microscopic lesions; results were interpreted as proposed by Taylor; 23 multiplicity adjustments were done using the false discovery rate correction. 2 All these analyses were performed in R v3.5.1 (R Core Team) and RStudio v1.1.447 (RStudio Team), and α = 0.05 was set for level of significance.
Results
Gross Lesions
The proportion of sheep with gross lesions and their severity scores were compared between experimental groups (Table 1). Gross lesions developed only in groups 1 and 3, which were inoculated with ETX-producing strains. The proportion of animals with gross lesions was significantly higher (P < .001) in these 2 groups (8/12) than in those inoculated with the etx knockout strain (group 2; 0/6) or sterile culture medium (group 4) (0/12).
Proportion of Sheep With Macroscopic Lesions in Groups Inoculated With Clostridium perfringens Type D Wild Type Strain (Group 1), etx Knockout Strain (Group 2), etx-Complemented Strain (Group 3), or Sterile Culture Medium (Group 4).
More animals developed gross lesions in group 1 (wild-type C. perfringens type D strain) versus group 3 (etx-complemented strain), but these differences were not statistically significant (P = .273). A significantly higher (P = .030) proportion of sheep developed hydrothorax in group 1 versus group 3; this lesion was not present in any of the animals in group 3. The proportion of sheep with pulmonary edema, hydropericardium, ascites, endocardial, epicardial, and myocardial hemorrhages did not differ significantly between groups 1 and 3 (P > .121).
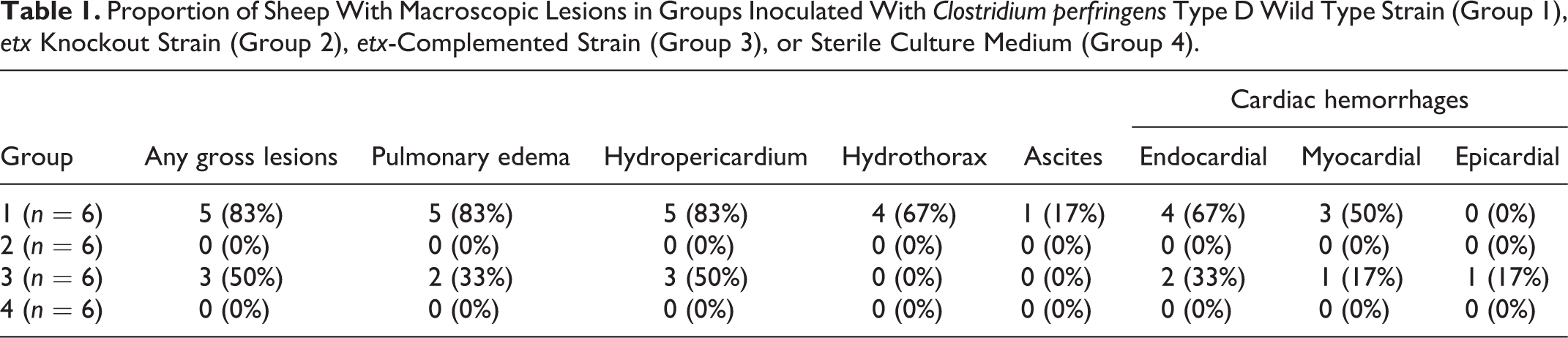
Hydropericardium was the most frequent gross lesion in sheep inoculated with ETX-producing strains, followed by pulmonary edema, endocardial hemorrhages, myocardial hemorrhages (Figs 1–4), hydrothorax, epicardial hemorrhages, and ascites. Hydropericardium varied from mild with no fibrin strands (sheep 19), to severe with (sheep 1) or without (sheep 20) fibrin strands, although it was moderate with fibrin in most sheep (2, 4–6, and 21).
Wild-type C. perfringens type D strain experimental infection, sheep.
The intergroup comparison of severity scores for gross lesions revealed that, overall, scores in sheep of groups 1 and 3 were higher than in those in groups 2 and 4. When the different gross lesions were evaluated separately, there were significant differences between groups for pulmonary edema (P = .006), hydrothorax (P = .003), hydropericardium (P = .005), and endocardial hemorrhages (P = .027; Fig. 5), but not for ascites (P = .4), myocardial (P = .075), or epicardial hemorrhages (P = .4).
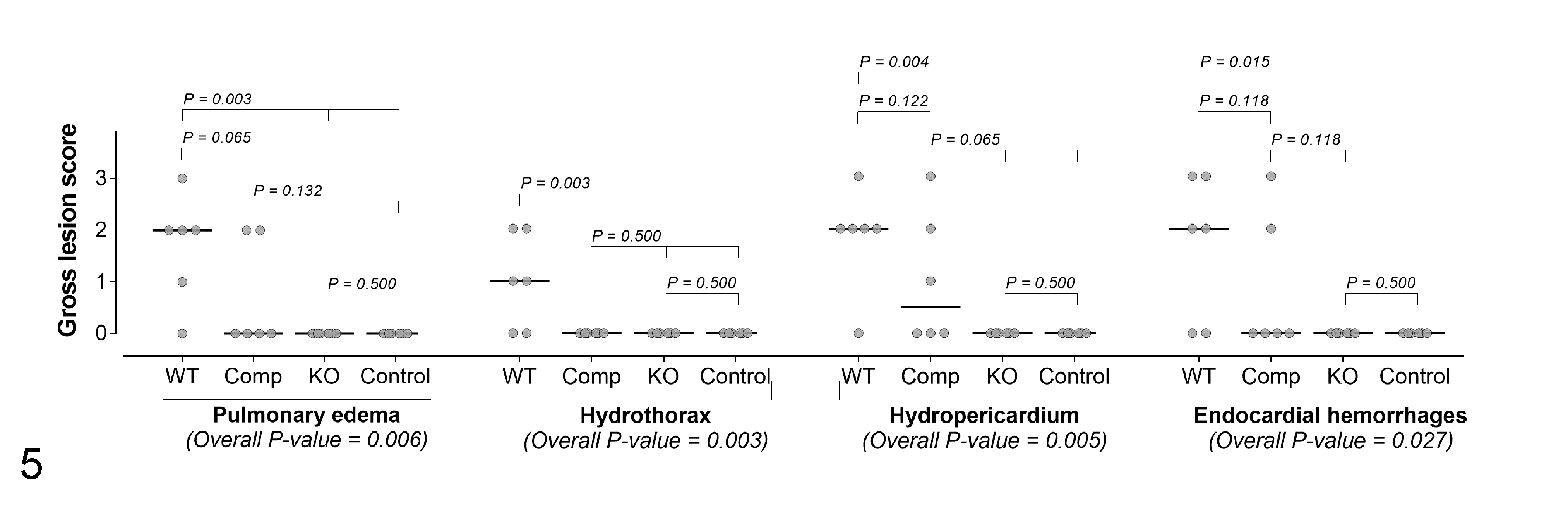
Intergroup comparison of selected gross lesions in 4 groups of 6 sheep inoculated with wild-type (WT) C. perfringens type D (group 1), etx-complemented strain (group 3, Comp), etx knockout (group 2, KO), and sterile culture medium (group 4, control). The grey circles represent the observed values and the horizontal bold black lines represent the medians.
Microscopic Lesions
As with the gross lesions, only sheep inoculated with ETX-producing strains (groups 1 and 3) developed microscopic lesions in the heart and lungs. The proportion of sheep with microscopic lesions (Table 2) was significantly higher (P < .001) in groups 1 and 3 inoculated with the ETX-toxigenic strains (8/12) than those inoculated with the etx knockout strain (group 2) or sterile culture medium (group 4; 0/12). The proportion of sheep with microscopic lesions did not differ significantly between groups 1 and 3.
Proportion of Sheep With Microscopic Cardiac and Pulmonary Lesions in Groups Inoculated With Clostridium perfringens Type D Wild Type Strain (Group 1), etx Knockout Strain (Group 2), etx-Complemented Strain (Group 3), or Sterile Culture Medium (Group 4).
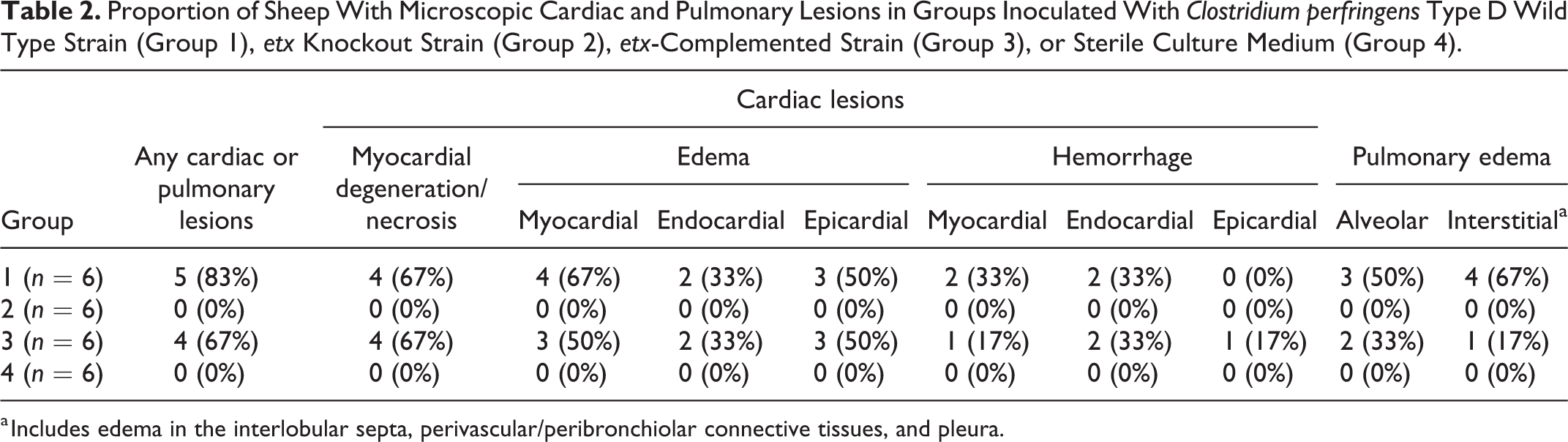
a Includes edema in the interlobular septa, perivascular/peribronchiolar connective tissues, and pleura.
Regarding changes in the heart considered to result from increased vascular permeability, cardiac edema and hemorrhages were observed only in sheep of groups 1 and 3. Myocardial edema was severe (score 3) to moderate (score 2) in group 1, and moderate to mild (score 1) in group 3. Extravasated eosinophilic (proteinaceous) fluid frequently expanded the interstitium, separated the cardiomyocytes, and surrounded the conduction system (Figs 6–9). Similar proteinaceous epicardial and endocardial edema affected sheep in groups 1 and 3 in proportions that did not differ significantly (P = .3) and were moderate (score 2) in both groups. Myocardial and endocardial hemorrhages were observed in similar proportions in sheep of groups 1 and 3 (P = .5) and were severe (score 3) to mild (score 1) in group 1, and moderate (score 2) in group 3 (Figs. 10, 11). None of the 12 sheep in groups 2 and 4 had microscopic myocardial, endocardial, or epicardial edema or hemorrhages.
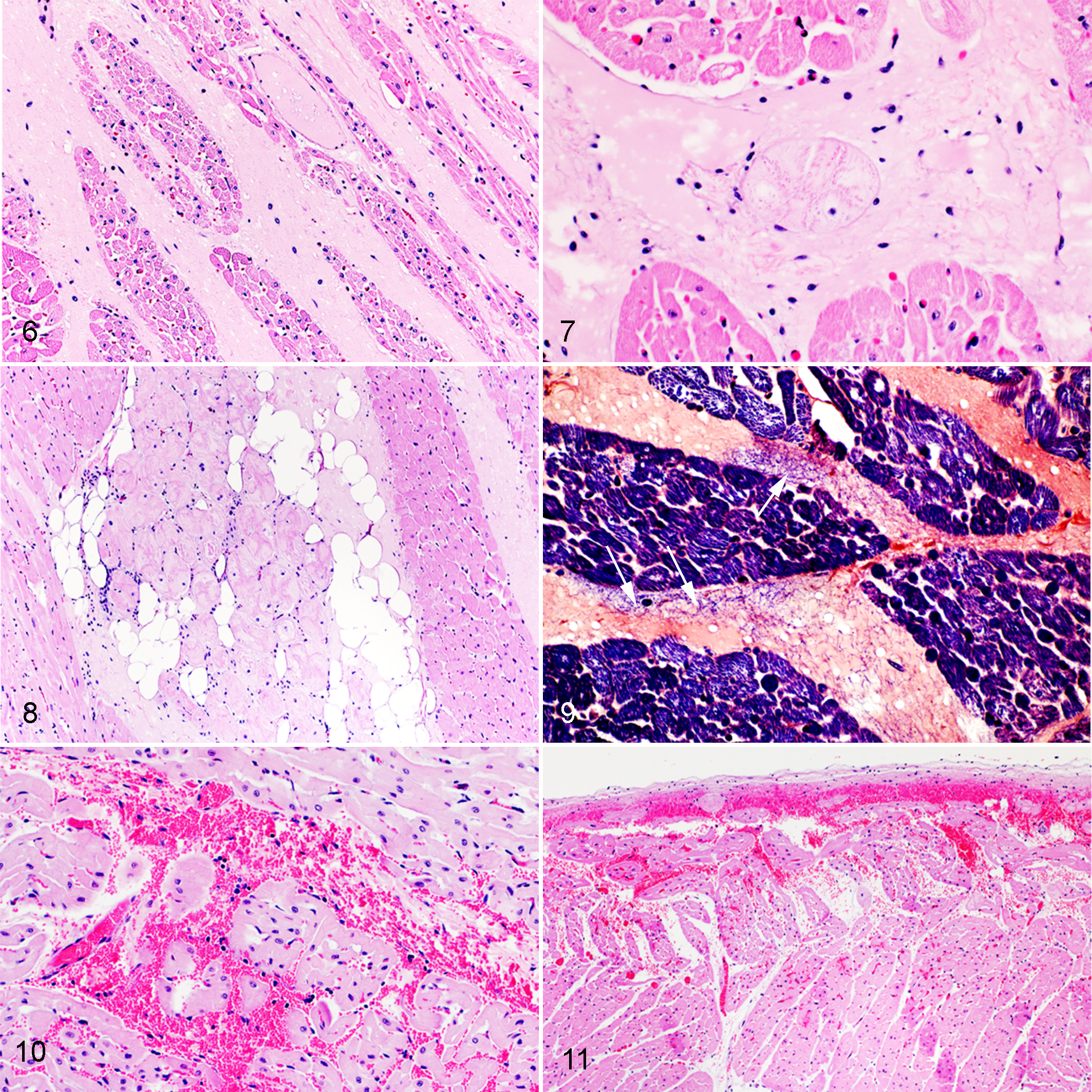
Experimental infection with epsilon toxin-producing strains of C. perfringens type D, heart, sheep.
Concerning degenerative and necrotizing myocardial fiber lesions, 4 of 6 sheep in group 1 had moderate (sheep 4 and 6) or severe (sheep 1 and 5) multifocal cardiomyocyte necrosis; frequently this lesion colocalized with myocardial edema and occasionally with myocardial hemorrhage (Figs. 12, 13). Three of 6 sheep in group 3 had moderate cardiomyocyte necrosis, while one sheep in this group had mild multifocal acute myocardial degeneration (score 1). Degeneration or necrosis occasionally affected individual or small groups of adjacent cardiomyocytes. The proportion of sheep with degenerative or necrotizing lesions in groups 1 and 3 did not differ significantly (P = .5). None of the sheep in groups 2 and 4 had degenerative or necrotizing myocardial lesions, and no sarcoplasmic mineralization was observed in any of the 24 sheep.
Experimental infection with wild-type C. perfringens type D strain, heart, sheep.
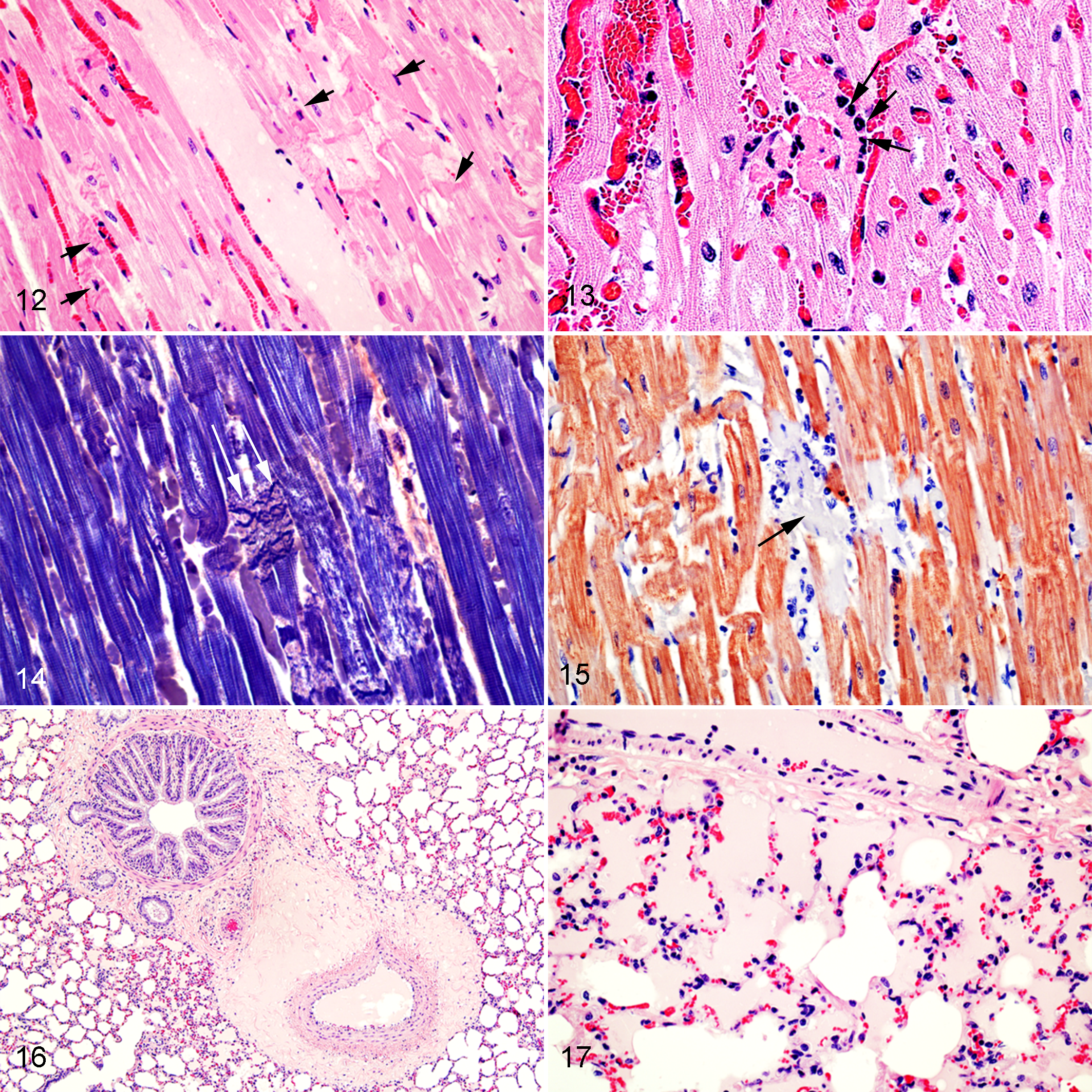
PTAH staining highlighted the loss of transverse striations and sarcoplasmic coagulation necrosis (Fig. 14), including the loss of transverse striations in degenerate cardiomyocytes in a sheep that had cardiomyocyte degeneration but no myocardial necrosis evident on HE-stained sections. Similarly, in the same sheep, troponin C IHC allowed for identification of cardiomyocyte necrosis or degeneration; affected cardiomyocytes showed marked segmental loss of sarcoplasmic immunoreactivity when compared to nonaffected adjacent cardiomyocytes (Fig. 15). However, similar changes of segmental loss of troponin C immunoreactivity and faint PTAH staining were also observed in some cardiomyocytes that did not have appreciable pathological changes on HE-stained sections in sheep of groups 2 and 4.
Regarding inflammatory lesions in the heart, in only 1 of the 2 animals with score 3 (severe) cardiomyocyte necrosis in group 1 (sheep 1), neutrophils occasionally infiltrated the endomysium surrounding necrotic myocytes (Figs. 13, 15) and rarely invaded the sarcoplasm of necrotic myocytes. Thirteen animals in all 4 groups had mild (11 cases) to moderate (2 cases) multifocal random aggregates of lymphocytes and histiocytes and 9 of these had scattered intrasarcoplasmic Sarcocystis-like protozoal cysts. No other cardiac microscopic lesions were observed in any of the animals from the 4 groups.
Histologic lesions were scored in 11 cardiac regions of animals in group 3. Of these lesions, only myocardial degeneration/necrosis was significantly different between regions (P = .029; Supplemental Table S2; Fig. 18). The atrioventricular node region had the highest scores for both lesions.
Microscopic pulmonary lesions, including both interstitial and alveolar edema (Figs. 16, 17), were seen only in sheep of groups 1 and 3 (Table 2). The proportion of sheep with microscopic pulmonary edema did not differ significantly between groups 1 and 3 (P = .3).
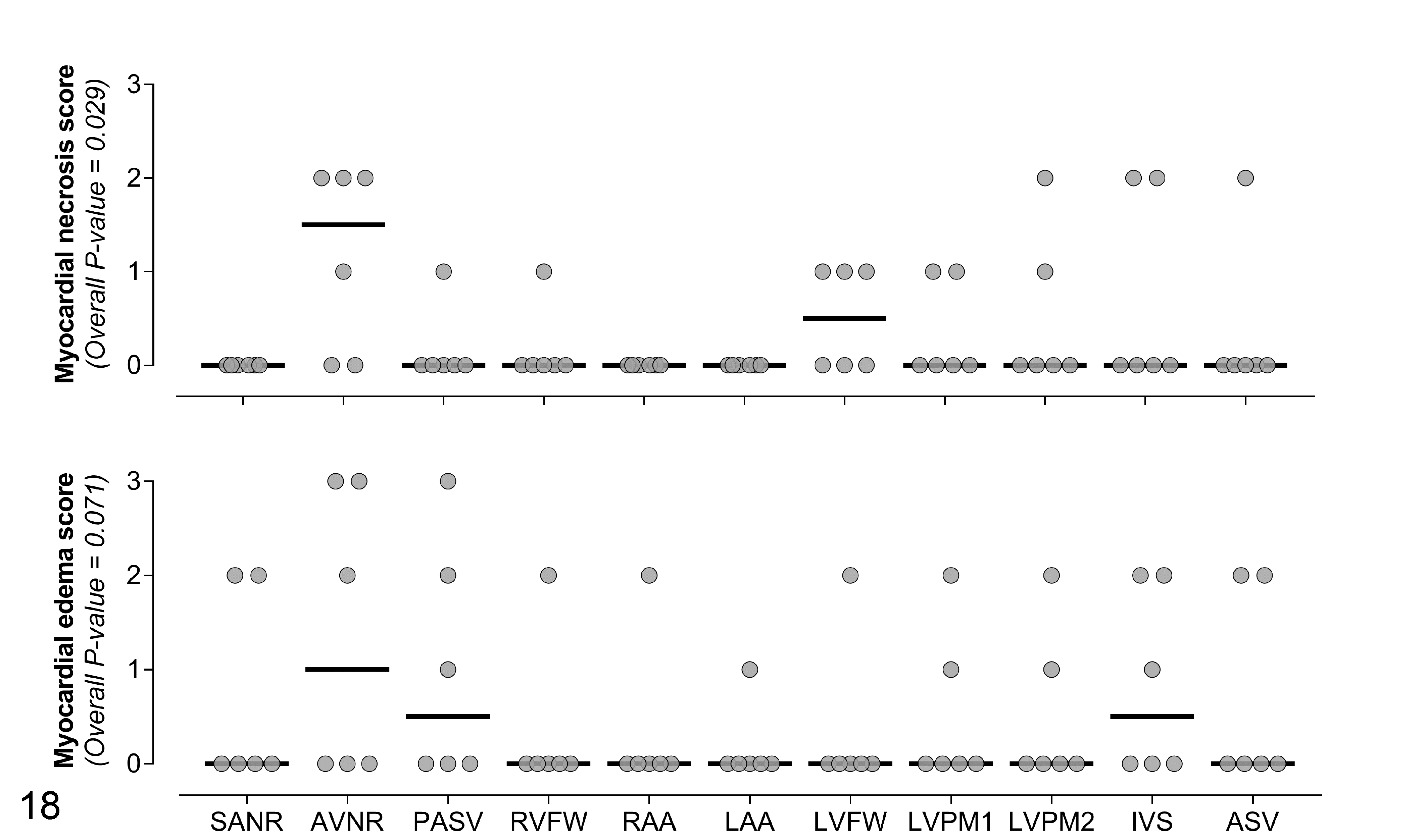
Within-subject comparison of scores for myocardial necrosis and myocardial edema in 11 cardiac regions of 6 sheep inoculated with the etx-complemented strain of C. perfringens type D (group 3). SANR, sinoatrial node region; AVNR, atrioventricular node region; PASV, pulmonary artery and semilunar valve at the site of insertion in the right ventricular wall; RVFW, right ventricular free wall; RAA, right atrial appendage; LAA, left atrial appendage; LVFW, left ventricular free wall; LVPM1, primary left ventricular papillary muscle; LVPM2, secondary left ventricular papillary muscle; IVS, interventricular septum; ASV, aortic semilunar valve at the site of insertion in the left ventricular wall. The grey circles represent the observed values and the horizontal bold black lines represent the medians.
The intergroup comparison of severity scores for histologic lesions showed that, overall, both cardiac and pulmonary lesions were higher in groups 1 and 3 than in groups 2 and 4 (Table 3). In sheep of groups 1 and 3 there was a strong positive correlation between the average scores for gross and histologic lesions (r = 0.891, P < .001; Supplemental Table S3).
Intergroup Comparison of Histologic Lesions in Groups Inoculated With Clostridium perfringens Type D Wild Type Strain (Group 1), etx Knockout Strain (Group 2), etx-Complemented Strain (Group 3), or Sterile Culture Medium (Group 4).

a Values are expressed as median (minimum – maximum).
b Lesions in the left ventricular papillary muscles were used for the intergroup comparison.
c,dDifferent letters in the same line indicate differences with P < .05.
Discussion
Our results confirm that the cardiac and pulmonary lesions described are associated with the capacity of the strains to produce ETX, since only animals inoculated with ETX-producing strains (groups 1 and 3), but not with the isogenic etx knockout mutant, developed gross or microscopic cardiac or pulmonary lesions.
In acute type D enterotoxemia of sheep, severe, usually lethal brain edema often develops. All 6 animals in group 1 developed moderate to severe microscopic vasogenic edema in the brain. Because 5 animals in this group that died or were euthanized due to severe clinical disease had cardiopulmonary lesions, 10 it is likely that the morbidity and mortality in this group was caused by cardiopulmonary or cerebral lesions or, more likely, a combination of these effects.
Of note, the one sheep of group 1 that was asymptomatic until the end of the experiment (24 hours after inoculation) and did not develop gross lesions had moderate vasogenic cerebral edema. 10 As clinical signs of experimental enterotoxemia in sheep can begin between 30 minutes and 26 hours after inoculation, 25 it is possible that this animal was within the incubation period when euthanized, or that clinical signs were minimal and therefore missed during clinical examination.
A somewhat different scenario was observed in group 3 animals, which were inoculated with the etx-complemented strain. In this group, 3 animals died or were euthanized due to severe clinical disease, and all 3 developed moderate to severe cardiopulmonary lesions. Interestingly, one sheep in this group had only hydropericardium and myocardial edema, but did not develop microscopic vasogenic edema in the brain, indicating that severe clinical signs necessitating euthanasia may have resulted from cardiac dysfunction. It is possible, however, that ETX directly affected cells other than microvascular within the central nervous system without light microscopic evidence of this damage. ETX has been shown to affect oligodendrocytes in mice, 15 and neurons in rats with subacute (48 hours) exposure. 7 Taken together, our findings suggest that cardiopulmonary lesions may contribute, in concert with cerebral lesions, to the morbidity and mortality in acute type D enterotoxemia.
One animal in group 3 (sheep 22) did not develop significant clinical signs or gross lesions when it was euthanized at the end of the experiment 24 hours post-inoculation. This animal had multifocal mild cardiomyocyte degeneration seen on HE-stained sections in 5 of the 11 cardiac regions examined, as well as loss of troponin C immunolabeling in the affected cardiomyocytes. This sheep also had severe perivascular cerebral edema in multiple areas of the brain. 10 This indicates that myocardial degeneration and severe brain perivascular edema can occasionally occur together as subclinical lesions.
Sudden death has been reported in type D enterotoxemia of sheep. 26,28 This was observed in one sheep in our study; an animal that died spontaneously approximately 10 hours after inoculation. However, the pathologic examination revealed a spectrum of gross and microscopic cardiopulmonary lesions that ranged from mild to severe, as well as severe perivascular edema in the brain. 10 This observation further supports the notion that in sheep with severe and widespread lesions, progression to death can occur very quickly. 25 In such cases death could be attributed to cerebral or perhaps cardiac dysfunction, given that even mild lesions in these organs could be life-threatening when vital structures of the brain or heart are involved.
Hydropericardium, most often with fibrin extravasation, was the most frequent gross lesion found in sheep inoculated with ETX-toxigenic strains (8/12 animals), followed by pulmonary edema (7/12 animals). Hydrothorax was present in 4/12 animals and was the only gross lesion for which a significantly higher number of sheep were affected in the group inoculated with the wild-type strain versus the etx-complemented strain, as none of the sheep inoculated with the latter developed this lesion. Hydrothorax was very strongly and positively correlated with interstitial pulmonary edema. This outcome was not unexpected as it probably results from plasma extravasation from the mediastinum, pleura, and pulmonary interstitium. Hydropericardium, pulmonary edema, and hydrothorax are frequently described gross lesions in type D enterotoxemia of sheep, both under experimental and natural conditions, 3,25,28 and they probably result from the action of ETX on the vasculature of the lungs/pleura, pericardial sac, and mediastinum. However, in animals with cardiac lesions, acute heart failure cannot be ruled out as contributors to these hemodynamic changes. Histologically, the eosinophilic appearance of the edema fluid in the lungs would support high-protein exudate due to increased vascular permeability over low-protein transudate expected in heart failure.
Endocardial and myocardial hemorrhages affected 6/12 and 4/12 sheep inoculated with the ETX-toxigenic strains, respectively, and the severity ranged from moderate to severe. It is not known if these hemorrhages result from effects of ETX on endothelial cells of the endocardium or of the subendothelial microvasculature. In this sense, a study that evaluated the effects of ETX on ovine, bovine, and caprine aortic endothelial cells in vitro found no morphological alterations suggestive of cytotoxicity or decreased viability, even when exposed to large ETX doses. 27 The authors discussed that the receptor for ETX might not be present in the endothelial cells of the aorta, while present in the endothelial cells of the microvasculature of the brain and other tissues, or that the action of ETX would require interactions with other cell types of the vascular wall in vivo. A recent study demonstrated a direct and dose-dependent cytotoxic effect on human cerebral microvascular endothelial cells exposed in vitro to ETX, 16 supporting previous in vivo conclusions that it is the endothelium of microvessels that appear to be predisposed to ETX damage. In this context, it has been demonstrated, in lambs, that ETX produced ultrastructural changes characterized by endothelial attenuation, condensation, and markedly increased electron density in myocardial, pulmonary, and renal interstitial capillaries, which resembled those found in ETX-exposed brain microvessels. 11 In our study, none of the sheep developed hemorrhages in the ascending aorta (not shown), even when severe endocardial hemorrhages were present. This further supports the notion that aortic endothelial cells are not susceptible to ETX in vivo. Additionally, no fibrin deposition or vegetative endocarditis was observed over the endocardium in any of the animals, as would have been expected if ETX had a significant cytotoxic effect on endocardial endothelial cells. Overall, it is suggested that changes in the endocardium are the result of altered permeability of the endocardial or subendocardial microvasculature rather than the endocardial endothelium per se.
With only a few exceptions, myocardial interstitial edema and cardiomyocyte necrosis colocalized in the same cardiac sections, and the degree of edema was usually equal or higher than for cardiomyocyte degeneration/necrosis in 5 of the 6 animals with these lesions from both affected groups. Furthermore, the correlation analysis indicated a moderate positive correlation between myocardial interstitial edema and cardiomyocyte necrosis. Taken together, these results suggest that focal myocardial necrosis seems to result from reduced localized perfusion (ischemia-hypoxia), although a direct effect of ETX on cardiomyocytes cannot be excluded, particularly in the animal with mild myocardial degeneration in the absence of histologically detectable myocardial edema.
Colocalization of myocardial necrosis and acute neutrophilic inflammation was observed in only one animal (sheep 1) in our study. The neutrophilic inflammatory infiltrate in this case was thought to be elicited by cardiomyocyte necrosis rather than being a primary event resulting from ETX toxemia. In humans with acute myocardial infarction, acute inflammation is used as a histologic criterion to determine the age of the infarction. 1 In the initial phase, acute myocardial necrosis without inflammation suggests lesions <8 hours old, while polymorphonuclear inflammation indicates a course of 8 to 24 hours. 14 Although this exact timeframe cannot be necessarily extrapolated to sheep, the general concept may apply in our cases. The lack of acute inflammation in most cases with myocardial necrosis in our study would suggest a very acute change (minutes to a few hours) with a slightly more protracted course in sheep 1.
The within-subject comparison of severity scores for histologic lesions in different cardiac regions in sheep of group 3 indicated that the highest scores for myocardial edema and necrosis were present in the atrioventricular node region. Because the atrioventricular node is crucial for the physiologic cardiac function, lesions in this region could determine arrhythmia or heart block and should be considered potentially significant. In cases of acute type D enterotoxemia, sampling this region for histologic examination could increase the chances of finding severe lesions, although lesions were not restricted to this anatomic region in most animals.
Troponin C immunoreactivity was markedly and segmentally reduced in cardiomyocytes that were clearly necrotic on HE-stained sections. However, because some nonnecrotic cardiomyocytes in animals inoculated with sterile culture medium or with the etx knockout strain also showed segmental loss of sarcoplasmic immunoreactivity, troponin C IHC is not recommended as a specific marker of cardiomyocyte degeneration or necrosis. A similar conclusion was reached with the use of troponin T IHC for the diagnosis of acute myocardial necrosis in humans, as unaffected or viable cardiomyocytes in control tissues showed non-specific patchy loss of immunoreactivity. 14
We conclude that cardiac lesions including myocardial interstitial edema, cardiomyocyte necrosis, and endocardial hemorrhages should be considered part of the spectrum of lesions of acute experimental type D enterotoxemia in sheep. In some cases, cardiac lesions themselves might be considered as a cause of severe clinical deterioration or death. As with other lesions of type D enterotoxemia, cardiac lesions are mediated by ETX and seem to result from microvascular damage and resulting marked increase in vascular permeability. Whether these cardiac lesions also occur in natural cases of type D enterotoxemia in sheep or other ruminants has yet to be determined. The atrioventricular node region showed higher severity scores of myocardial edema and necrosis, although lesions were not restricted to this anatomic location. Further studies should be performed on the pathogenesis of spontaneous diseases induced by ETX, particularly considering the potential detrimental health effects of ETX in humans. 21,30
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Giannitti_et_al - Cardiopulmonary Lesions in Sheep Produced by Experimental Acute Clostridium Perfringens Type D Enterotoxemia
Supplemental Material, Combined_supplemental_materials-Giannitti_et_al for Cardiopulmonary Lesions in Sheep Produced by Experimental Acute Clostridium Perfringens Type D Enterotoxemia by Federico Giannitti, Jorge P. García, Julian I. Rood, Vicki Adams, Joaquín I. Armendano, Juliann Beingesser and Francisco A. Uzal in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Karen Sverlow from CAHFS UC Davis for technical assistance with histology and immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Federico Giannitti acknowledges support from the Uruguayan “Agencia Nacional de Investigación e Innovación” (ANII) through mobility grant MOV_CA_2018_1_150021. This work was supported by Grant R01 AI056177 from the National Institute of Allergy and Infectious Diseases (NIAID). Research at Monash University was also supported by funding provided by the Australian Research Council to the Australian Research Council Centre of Excellence in Structural and Functional Microbial Genomics (Grant CE0562063).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
