Abstract
Since axonal injury (AI) is an important component of many veterinary neurologic disorders, we assessed the relative ability of a panel of antibodies (amyloid precursor protein, 3 subunits of neurofilament protein, protein gene product 9.5, ubiquitin, and synaptophysin) to detect axonal swellings or spheroids. Abundant axonal spheroids found in necrotic internal capsule foci produced in 4 sheep by chronic Clostridium perfringens type D epsilon neurotoxicity provided a model system in which to evaluate this important diagnostic tool. There was heterogeneous labeling of subsets of spheroids by the respective antibodies, suggesting that, in order to detect the complete spectrum of AI in diagnostic cases, a range of antibodies should be used, not only when spheroids are plentiful but also when they are few in number or incompletely developed. The application of insufficient markers in the latter cases can potentially lead to the contribution of AI to lesion pathogenesis being underappreciated.
There is now a range of antibodies available to detect axonal injury (AI), and we wished to compare the efficacy of these antibodies in detecting the complete range of AI manifest as axonal swellings or spheroids. In order to test these antibodies, we selected 4 cases of focal symmetrical encephalomalacia (FSE) produced by clostridial epsilon toxin, in which there were abundant spheroids in, and more particularly in the periphery of, necrotic foci in the internal capsule.
Clostridium perfringens type D enterotoxemia of sheep spans a disease continuum ranging from a peracute clinical course with sudden death at the most severe end of the spectrum to chronic intoxication with a more protracted presentation. The latter chronic form occurs when lower doses of toxin are absorbed from the intestine into the systemic circulation, or sheep are partially immune. This often leads to the development of macroscopic, bilaterally symmetrical, malacic (necrotic) foci in certain areas of predilection in the brain (termed FSE), such as the basal ganglia, internal capsule, thalamus, subcortical white matter, substantia nigra, hippocampus, and cerebellar peduncles. 4,5,12
The FSE cases were supplied by Agriculture Victoria Research, Melbourne and Gribbles Veterinary Pathology, Adelaide, and the bilaterally symmetrical necrotic foci found in these cases conformed to the neuroanatomic pattern of FSE lesions, which are considered to be diagnostic for type D neurotoxicity. 8 The brains from these naturally occurring field cases were immersion-fixed in 10% neutral buffered formalin, coronal sections paraffin-embedded, and 6 µm sections cut and stained with hematoxylin and eosin.
Five discrete tracts in the internal capsule were selected from each of the 4 FSE cases for analysis, and necrotic foci were considered to be similarly aged, based on the predominantly neutrophilic attendant inflammatory reaction. While each of the 5 white matter tracts from the 4 FSE cases contained different numbers of spheroids, only tracts composed of similar numbers of spheroids in hematoxylin and eosin–stained sections were compared. For each immunomarker, the number of spheroids labeled, and the robustness of their immunoexpression, was overall graded + to ++++ (Table 1).
Immunoreactivity of different markers of axonal injury in the internal capsule of 4 sheep with focal symmetrical encephalomalacia.

a Five internal capsule tracts were analyzed for each case.
b Overall spheroidal immunoreactivity graded + to ++++, encompassing the number of spheroids detected and robustness of expression.
The battery of immunohistochemical markers of AI used was amyloid precursor protein (APP), neurofilament protein (NFP; light, medium, and heavy NFP subunits of 68, 160, and 200 kDa molecular weight, respectively), protein gene product 9.5 (PGP 9.5), ubiquitin, and synaptophysin. APP is a constitutively expressed, highly conserved, 100 to 140 kDa transmembrane glycoprotein that is normally transported anterograde by fast axoplasmic transport. It is the most sensitive, early marker of AI, accumulating rapidly within 1 to 3 hours of an insult, proximal to the site of injury. 1,2 APP only labels injured axons as the amount of APP in uninjured axons is insufficient to be detected by routine light microscopy. 11 NFPs are primary cytoskeletal components of axons and consist of 3 related proteins, termed light, medium, and heavy, and are visualized in normal axons. Each NFP has the same rod domain. However, NFP-M and NFP-H have side-arms of different length and phosphorylation status, which control the axon diameter, and after AI, NFP dephosphorylation results in rapid proteolysis of these side-arms. The degree of phosphorylation correlates inversely with the velocity of transport of NFPs, with NFP-L immunopositive spheroids forming early, and NFP-M and NFP-H swellings later, after the initial insult. Each isoform may be either phosphorylated or non-phosphorylated, and antibodies to NFP are specific for only one of these two configurations; 1,3 the anti-NFP antibodies used in the present study recognized phosphorylated-neurofilament proteins. Ubiquitin is a small (8.6 kDa), highly conserved, and ubiquitously expressed protein that is transported in a slow manner and targets proteins for both non-lysosomal degradation via the ubiquitin-proteasome system and the autophagy lysosomal pathway. Normal axons do not exhibit ubiquitin immunoreactivity in formalin-fixed, paraffin-embedded material. Ubiquitin immunopositivity of spheroids suggests that structures within the reactive swellings are recognized as being abnormal within the nerve microenvironment and, accordingly, are targeted for degradation. 3,7 PGP 9.5 is a soluble, 27 kDa, cytoplasmic protein, also known as ubiquitin carboxyl-terminal hydrolase (UCH-L1), which is a member of the ubiquitin hydrolase family of proteins. It functions as an ubiquitin carboxyl-terminal hydrolase isoenzyme. Synaptophysin is a calcium-binding glycoprotein (38 000 kDa), which is the most abundant integral membrane protein constituent of synaptic vesicles of neurons. It is moved by fast axoplasmic transport and is present in virtually all neurons in the brain that engage in synaptic transmission. 3
For immunohistochemistry, the following monoclonal and polyclonal antibodies were used. The monoclonal antibodies were APP (gift from Professor Colin Masters, clone 22C11), 1:1000 dilution; NFP-H (Sigma, cat. N5389, clone NE14), 1:40,000 dilution; NFP-M (Sigma, cat. 34-1000, clone RMP14.9), 1:2000 dilution; NFP-L (Sigma, cat. N5139, clone NR4), 1:3000 dilution; and PGP9.5 (Novus Biologicals, cat. NB600-1160, clone 31A3), 1:2000 dilution. The polyclonal antibodies used were ubiquitin (Abcam, cat. ab7780), 1:4000 dilution; and synaptophysin (Abcam, cat. ab14692), 1:3000 dilution. In brief, sections were dewaxed using xylene and then rehydrated through alcohols. Sections were treated with methanol/hydrogen peroxide for 30 minutes. All sections were then rinsed twice in phosphate-buffered saline (PBS; pH 7.4) for a further 5 minutes each wash. Antigen retrieval was performed using citrate buffer (pH 6). Slides were allowed to cool and washed twice in PBS (pH 7.4). Nonspecific proteins were blocked using normal horse serum for 30 minutes. Antibodies were then applied at the above listed dilutions at room temperature overnight. The following day, the sections were given 2 washes in PBS, then a biotinylated anti-mouse secondary (Vector Laboratories, cat. BA-2000) for the monoclonal antibodies and a biotinylated anti-rabbit secondary (Vector Laboratories, cat. BA-1000) was applied for 30 minutes at room temperature. Following 2 PBS washes, the slides were incubated for 1 hour at room temperature with a streptavidin-conjugated peroxidase tertiary (ThermoScientific, cat. 21127). Sections were then visualized using diaminobenzidinetetrahydrochloride (DAB), washed, counterstained with hematoxylin, dehydrated, cleared, and mounted on glass slides. A negative control omitting the primary reagent, as well as a positive control showing the normal pattern of expression of the antigen in question, were run with each batch of slides.
The comparative analysis of brain sections labeled with the different antibodies revealed a complex pattern of axonal spheroid immunolabeling, with different subsets often being labeled by different antibodies. In addition to variability in the total number of spheroids detected by each antibody, there was also diversity in the size of spheroids labelled and their labeling intensity. However, as shown in Table 1, while immunolabeling of axonal spheroids differed between the different antibodies tested, it was consistently similar for each antibody in all 4 FSE cases. Accordingly, Figures 1 to 8 show immunolabeling of spheroids from one of the internal capsule tracts selected for comparison between cases.
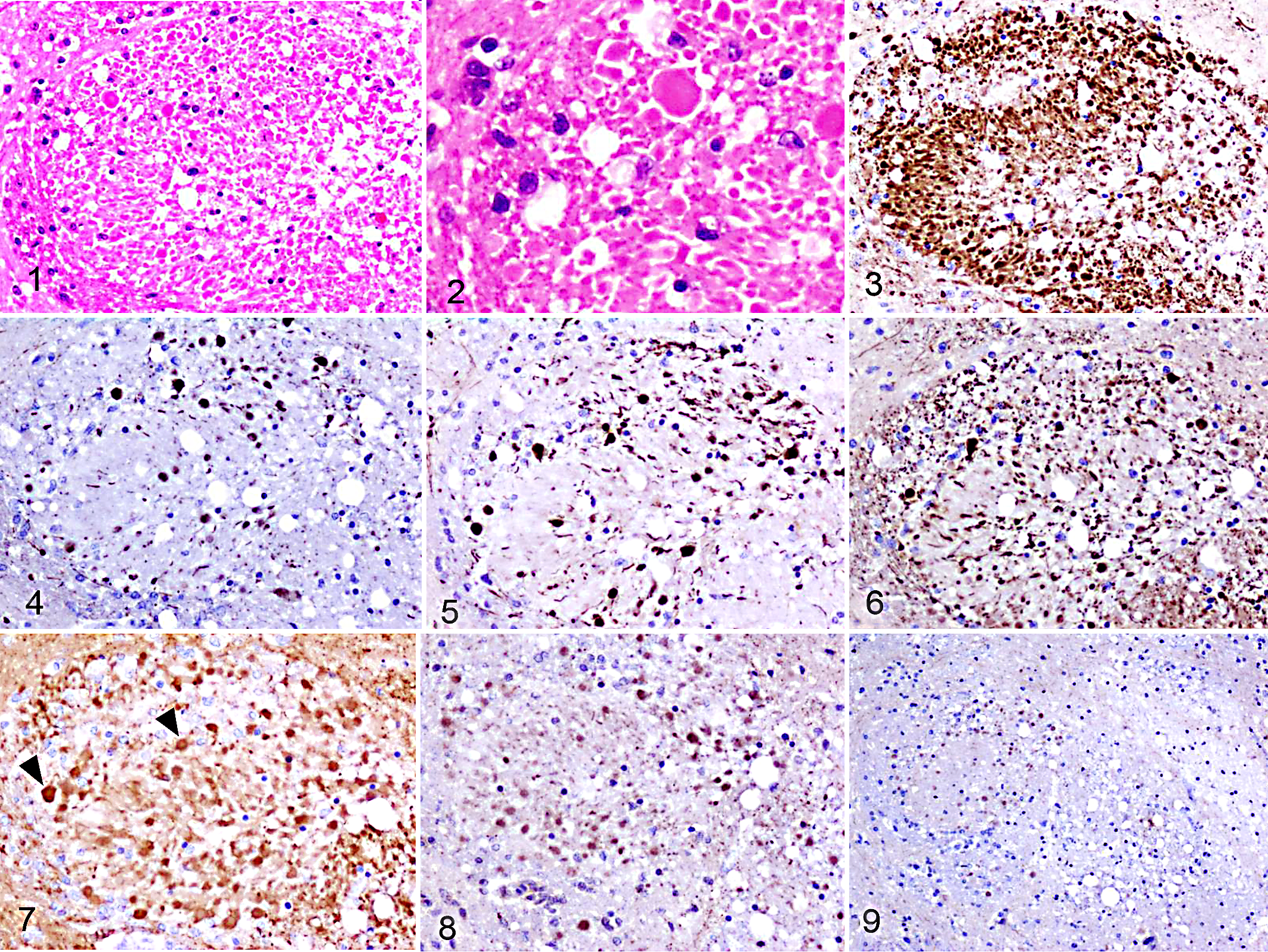
Focal symmetrical encephalomalacia, internal capsule, sheep.
The hematoxylin and eosin–stained section of the selected white matter tract showed numerous axonal spheroids of varying size (Figs. 1, 2). APP (Fig. 3) always detected many more axonal spheroids, both large and small, than other markers, and the immunolabeling was strong and easily visualized. NFP immunolabeling varied according to the subunit examined. NFP 68 (Fig. 4) and NFP 200 (Fig. 5) detected larger spheroids. NFP 200 labelled a few more spheroids than NFP 68. NFP 160 (Fig. 6) labelled a few large spheroids, but also a subset of many smaller spheroids. PGP 9.5 (Fig. 7) labeled a similar number of spheroids to NFP 200, but the immunolabeling was much weaker. Ubiquitin (Fig. 8) detected fewer spheroids than PGP 9.5, with similar intensity to the latter. Spheroidal labeling with synaptophysin (Fig. 9) was scant. Spheroids immunopositive to the different antibodies were largely confined to the immediate vicinity of necrotic foci, with only an occasional spheroid found at a distance from the necrotic area in the respective coronal sections, these being detected more readily with APP and NFPs.
In more advanced FSE cases in which there was lytic necrosis and replacement by invading macrophages, the latter had phagocytosed not only myelin basic protein-positive myelin debris (Fig. 10) but also APP (Figs. 11, 12) and NFP (Fig. 13) immunopositive axonal fragments. This engulfment of fragments of damaged axons seems, to the best of our knowledge, to be a novel finding in this disease.
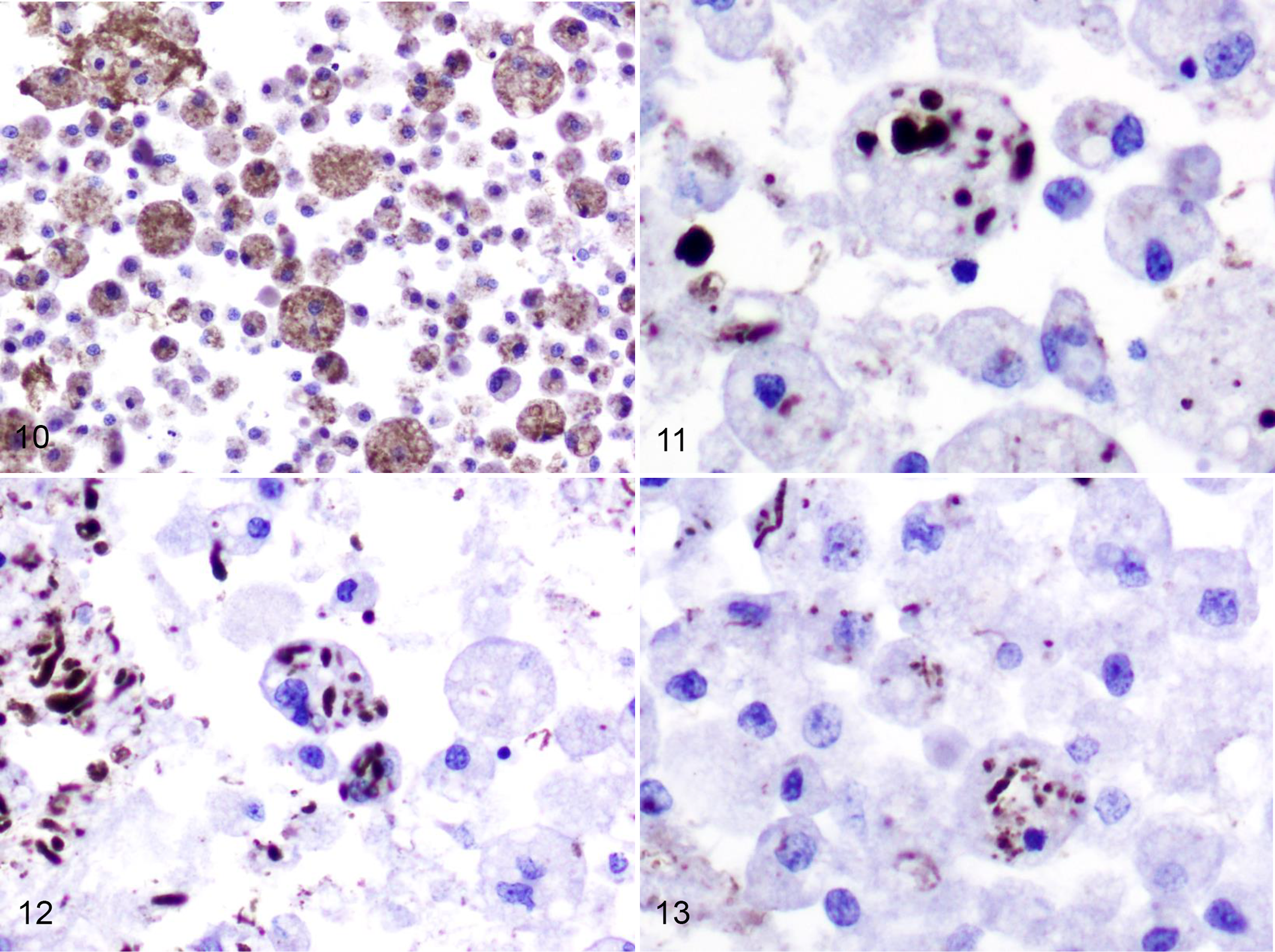
Focal symmetrical encephalomalacia, internal capsule, sheep.
Axonal spheroids in the tracts selected for analysis varied greatly in size, morphology, and intensity of immunolabeling, suggesting that they were at different stages of temporal evolution and/or of different aggregated protein composition. It is now known that axonal degeneration may continue for a long period of time, even up to a year, after the initial insult. 1 Since the internal capsule contains numerous ascending and descending myelinated axons of varying caliber, and subserving very different functions, the morphologic expression of AI probably depends, in part, at least, on the functional state of the axons that are damaged at the time of the injurious insult.
The heterogeneity of immunolabeling of axonal spheroids by different markers of AI is influenced by the rate at which the respective molecular markers are transported along axons. Vesicular molecules such as APP and synaptophysin are moved by fast axoplasmic transport and, accordingly, could be expected to form spheroids at the site of axonal disruption more rapidly than the relatively slowly transported cytoskeletal and cytosolic proteins such as NFP, ubiquitin, and PGP 9.5. Moreover, within an individual axon, the nature of the injury may vary at different sites along the nerve fiber, leading to differential accumulation of transport dysfunction markers. There is also experimental evidence that some injured axons may not be irreversibly damaged, 10 with a subset of axonal swellings capable of being repaired. APP-immunopositive axons, for example, seen in histologic sections are now considered to be probably a mix of reversible and irreversible AI, 6 and both APP-immunopositive and APP-immunonegative axonal spheroids may be found in a given lesion. In spheroids not labeled by APP, there may have been insufficient time for cytoskeletal disruption, and subsequent impairment of axonal transport, to occur and result in enough APP accumulation to be visible by light microscopy. Alternatively, APP immunoreactivity could have been lost due to spheroidal dissolution.
In veterinary pathology, the immunohistochemical profile of axonal spheroids has been best examined in cases of neuroaxonal dystrophy (NAD), a neurologic disorder in which axonal spheroids are numerous in the brain. 7 In ovine NAD, 7 there was great diversity of spheroidal protein expression using different signatures of impaired axonal transport (APP, NFP-H, NFP-M, ubiquitin, and synaptophysin), but the relative efficacy of these markers in detecting axonal spheroids was not assessed. This heterogeneity was interpreted as being a reflection of the different stages of their formation. In canine NAD cases, one study 9 found axonal spheroid immunopositivity to NFP-L, synaptophysin, ubiquitin, and tau, while another 8 showed variable immunoreactivity to NFP-L, NFP-M, and NFP-H, tau, HSP70, ubiquitin, synapse-associated proteins (synaptophysin, syntaxin-1, and SNAP25), α- and α/β-synuclein, and the calcium-binding proteins calbindin, calretinin, and parvalbumin. The efficacy of spheroidal immunodetection was not evaluated in the first canine case, but in the second, most spheroids in almost all brain regions were strongly immunopositive to NFP, α/β-synuclein, HSP70, and ubiquitin, while reactivity to α-synuclein, syntaxin-1, SNAP25, and the calcium-binding proteins was much more restricted in neuroanatomical distribution.
In conclusion, the findings in these FSE cases showed that a diverse range of AI, presenting as axonal spheroid formation, was detected by the different immunohistochemical markers used. The heterogeneity of AI found confirms that multiple markers need to be used in order to detect the full spectrum of AI present in a given brain lesion, and inappropriate marker selection may significantly underestimate both the total amount of AI present and its contribution to a given neural lesion. While APP is currently the most reliable marker of AI, no single immunomarker can capture the total amount of AI present, with different subsets of damaged axons often being labeled by different antibodies. The range of immunolabeling of axonal spheroids resulting from disruption of axonal transport is mechanistically largely a reflection of the different stages of their temporal development and differing mixes of accumulated proteins.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
