Abstract
Lesions of D-galactosamine (D-GalN)-induced hepatotoxicity resemble those of human acute viral hepatitis. This study investigated hepatic mesenchymal cells including hepatic stellate cells (HSCs) and myofibroblasts in D-GalN-induced hepatotoxicity. Rats, injected with D-GalN (800 mg/kg body weight, once, intraperitoneally) were examined on post single injection (PSI) at 8 hours and days 1 to 5. Lesions consisting of hepatocyte necrosis and reparative fibrosis were present diffusely or focally within the hepatic lobules on PSI days 1 and 2, and then the injury recovered on PSI days 3 and 5. Myofibroblasts expressing vimentin, desmin, and α-smooth muscle actin (α-SMA) were present in the lesions. Double immunofluorescence showed that myofibroblasts reacted simultaneously to vimentin/α-SMA, desmin/α-SMA, and desmin/vimentin; furthermore, myofibroblasts reacting to vimentin, desmin, and α-SMA also co-expressed glial fibrillary acidic protein (GFAP), a marker of HSCs. Additionally, GFAP-expressing myofibroblasts reacted to nestin and A3 (both are markers of immature mesenchymal cells). Cells reacting to Thy-1, a marker for immature mesenchymal cells, also appeared in fibrotic lesions. In agreement with the myofibroblastic appearance, mRNAs of fibrosis-related factors (TGF-β1, PDGF-β, TNF-α, Timp2, and Mmp2) increased mainly on PSI days 1 and 2. Myofibroblasts with expression of various cytoskeletal proteins were present in diffuse or focal hepatic lesions, and they might be derived partly from immature HSCs and from immature mesenchymal cells.
The liver is a unique organ with an extraordinary capacity to regenerate after injury. Hepatic stellate cells (HSCs) serve as the first line of defense against immune and inflammatory challenges by producing various cytokines, thereby attracting inflammatory cells from the systemic circulation and lymphoid organs. 21,45
Chronic viral hepatitis may lead to development of cirrhosis and hepatocellular carcinoma. 46,47 Relevant animal models are required for better understanding the pathogenesis of human viral hepatitis. 4 The D-GalN model of liver injury is characterized by multifocal degeneration and coagulative necrosis with infiltration of inflammatory cells, with reaction of M1-/M2-macrophages. 38,43 Thus, D-GalN-induced hepatic lesions are considered to resemble those of human virus hepatitis. Reparative fibrosis concomitantly occurred in D-GalN-induced hepatic injury via proliferation of mesenchymal cells. 3,8 Hepatic lesions in D-GalN-induced hepatotoxicity are different from those seen in thioacetamide and carbon tetrachloride–induced hepatotoxicity in which hepatocyte injury and fibrosis occur in the centrilobular area. 16,18
HSCs reside between liver sinusoidal endothelial cells and hepatocytes and represent 5% to 8% of all liver-constituting cells. 40 In physiological condition, HSCs store vitamin A, synthesize extracellular matrices (ECMs) and matrix-degrading metalloproteinases, and regulate sinusoidal blood flow. 40 In injury, HSCs may transdifferentiate into α-smooth muscle actin (α-SMA)-expressing myofibroblasts capable of producing collagen and fibrogenic cytokines, which contribute to reparative fibrosis that may progress to cirrhosis. 23,29,34,37 HSCs are considered to be the main cell of origin of myofibroblasts in hepatic fibrosis. 3,9,44 Myofibroblasts may also be generated from interstitial fibroblasts and pericytes with immature mesenchymal nature, or by epithelial-mesenchymal transition (EMT) of tubular epithelial cells in renal fibrosis. 7
Previously, we reported the participation of M1-/M2-macrophages in the D-GalN acute rat liver model. However, cellular analysis of hepatic mesenchymal cells developed in D-GalN-induced liver injury remains to be investigated. The objective was to characterize HSCs and myofibroblasts in D-GalN-induced rat hepatotoxicity with diffuse and focal lesions using markers (A3 and Thy-1) for immature mesenchymal cells, because myofibroblasts may be generated from immature mesenchymal cells. 7
Materials and Methods
Experimental Design
Twenty-four, 6-week-old, male F344 rats (110-120 g body weight, Charles River) were used in this experiment. Rats were housed in an animal room maintaining at 20 to 22 °C temperature with 12 hours light-dark cycle, and fed a standard diet (DC-8, CLEA Japan) and tap water ad libitum. After 1-week acclimatization, 20 rats in the experiment group were injected intraperitoneally with D-GalN (Sigma Aldrich, dissolved in 0.9% NaCl at a dose of 800 mg/kg body weight) as previously described. 38 The remaining 4 rats served as controls and were injected with an equal volume of 0.9% NaCl through the same route (intraperitoneally), and they were sacrificed on post single injection (PSI) day 1. Four rats were examined on each PSI time point at 8 hours and on 1, 2, 3 and 5 days. This experiment complied with Osaka Prefecture University guidelines for animal care and was approved by the local ethic committee (No. 30-2).
Histopathology and Immunohistochemistry
All rats were euthanized under isoflurane anesthesia, and liver tissues collected were fixed in 10% neutral buffered formalin (NBF), or periodate-lysine-paraformaldehyde (PLP) solution which was further processed by PLP-Amex (acetone, methyl-benzoate and xylene) method as previously described. 38 NBF-fixed tissues were dehydrated, embedded in paraffin, and then cut at 4-µm thicknesses. Formalin-fixed, deparaffinized sections were stained with hematoxylin and eosin (HE) for histopathological examinations and with azan-Mallory stain for collagen deposition.
PLP-fixed tissue sections were used for immunohistochemical analyses with primary antibodies listed in Table 1. After pretreatment, Histostainer (Histofine, Nichirei Bioscience Inc) was used for the staining. Briefly, the tissue sections were incubated with 5% skimmed milk in phosphate buffered saline (PBS) for 10 minutes, followed by 1-hour incubation with primary antibodies. Then, the sections were treated with 3% H2O2 in PBS for 15 minutes and were incubated for 30 minutes with horseradish peroxidase-conjugated secondary antibody (Histofine simple stain MAX PO; Nichirei Inc). Positive reactions were visualized with 3,3′-diaminobenzidine (DAB; Nichirei Inc) for 10 minutes and the slides were counterstained lightly with hematoxylin. As negative controls, tissue sections were treated with mouse non-immunized serum instead of the primary antibody.
Primary Antibodies Used for Single Immunohistochemical Labelling.
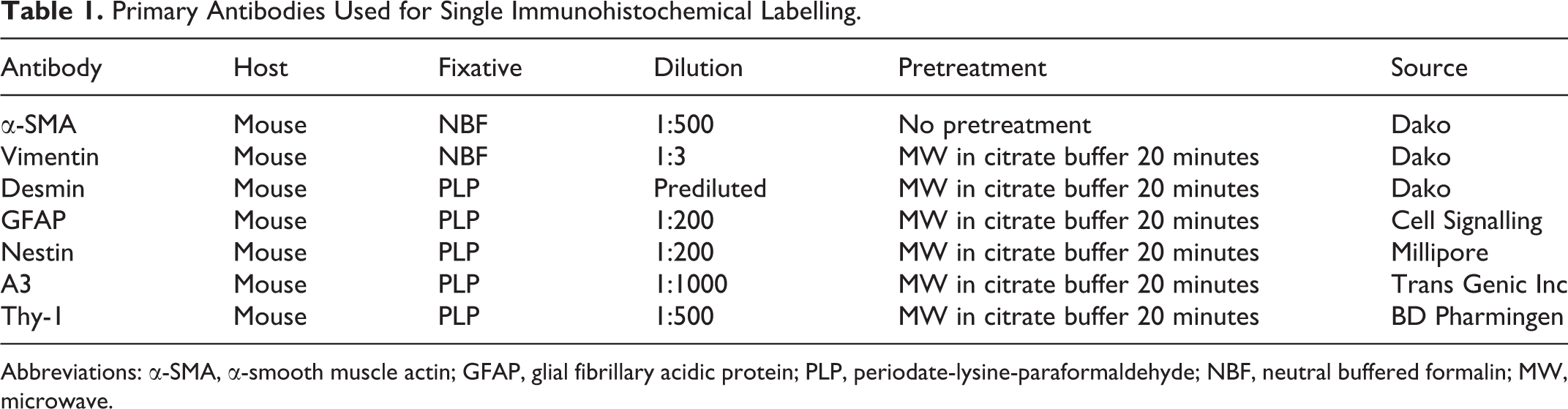
Abbreviations: α-SMA, α-smooth muscle actin; GFAP, glial fibrillary acidic protein; PLP, periodate-lysine-paraformaldehyde; NBF, neutral buffered formalin; MW, microwave.
Double Immunofluorescence Staining
To investigate the co-localization of cell specific antigens, double immunofluorescence was carried out using cryostat tissue sections (10-µm thick) from D-GalN-treated rats on PSI day 2. The combinations in the dual immunofluorescence were vimentin/α-SMA, desmin/α-SMA, desmin/vimentin, α-SMA/GFAP, vimentin/GFAP, desmin/GFAP, nestin/GFAP, and A3/GFAP. The sections were fixed with acetone/methanol (1:1) for 10 minutes at 4 °C, air-dried for 30 minutes, and washed with PBS. Sections were blocked with 10% normal goat serum for 30 minutes and treated with primary antibodies overnight at 4 °C. Then, sections were washed and allowed to react with secondary antibody goat anti-mouse IgG conjugated with Alexa 488 or Alexa 568 (Invitrogen) for 45 minutes at room temperature. The sections were incubated with second primary antibody followed by fluorescent dye-conjugated secondary antibody. Alexa 568 non-labeled α-SMA, GFAP, and vimentin were used for combination of vimentin/α-SMA, desmin/α-SMA, desmin/vimentin, α-SMA/GFAP, vimentin/GFAP, desmin/GFAP, nestin/GFAP, and A3/GFAP. The slides were cover slipped with Fuluoro-KEEPER antifading reagent containing 4′,6-diamidino-2-phenylindole (DAPI; Nakarai Tesque) for nuclear staining and analyzed by a virtual slide scanner (VS-120, Olympus).
Real-Time Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
Liver samples were immersed in RNA later (Qiagen GmbH) and stored at −80 °C until use. Total RNA from the liver tissue was extracted by using SV total RNA isolation system kit (Promega) according to manufacturer’s instructions. The concentration of RNA was determined using a nanodrop 1000 spectrophotometer (Thermo Scientific) and 2.5 µg of total RNA was reverse-transcribed to cDNA by using a superscript VILO reverse transcriptase (Invitrogen). Real-time PCR was conducted by using TaqMan gene expression assays (Life Technologies) in a PikoReal Real-time PCR System (Thermo Scientific). Probes used are shown in Table 2. The mRNA expression was normalized against that of 18S rRNA as an internal control. The data were calculated using the comparative Ct method (ΔΔCt method).
Probes Used in the Real-Time RT-PCR.
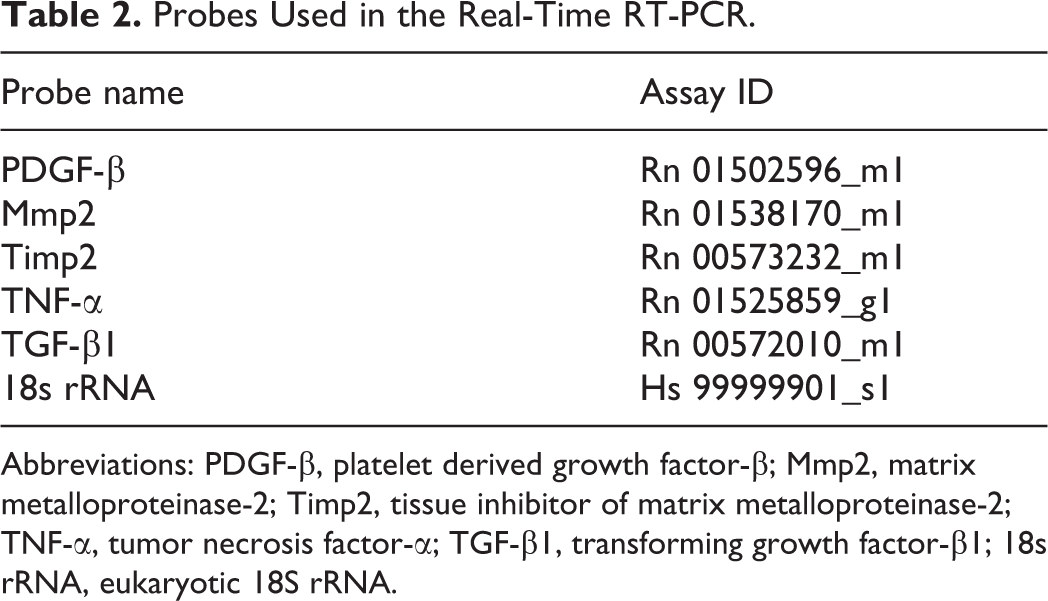
Abbreviations: PDGF-β, platelet derived growth factor-β; Mmp2, matrix metalloproteinase-2; Timp2, tissue inhibitor of matrix metalloproteinase-2; TNF-α, tumor necrosis factor-α; TGF-β1, transforming growth factor-β1; 18s rRNA, eukaryotic 18S rRNA.
Histopathological Evaluation
Cells showing a distinct immunopositive-reaction for α-SMA, vimentin, desmin, GFAP, nestin, Thy-1, and A3 in the lesion areas were semiquantitatively analyzed with scoring criteria (number and intensity) as follows: − = no positive cells (0–1 positive cell/40× field), ± = slightly positive (2–5 positive cell/40× field), + = positive (6–10 positive cell/40× field), 2+ = moderately positive (10–15 positive cell/40× field), 3+ = highly positive (>15 positive cell/40× field). Liver sections from 3 rats were examined at each examination point. The percentage of double immunofluorescence for vimentin/α-SMA, desmin/α-SMA, desmin/vimentin, α-SMA/GFAP, vimentin/GFAP, desmin/GFAP, nestin/GFAP, and A3/GFAP were calculated per 0.2 mm2 of tissue in the lesion areas.
Data Analyses
Data are shown as mean ± standard deviation (SD), and statistical analysis was performed using Dunnett’s multiple comparison tests. Significance was considered at P < .05.
Results
D-GalN-Induced Hepatic Lesions
In control livers and livers at PSI 8 hours after D-GalN administration, there were no lesions. On PSI days 1 and 2 after D-GalN administration, liver lesions consisting of hepatocyte degeneration and coagulative necrosis (as a single cell and foci of several cells) were seen diffusely or focally within the hepatic lobules (Fig. 1, arrowhead, inset). In addition, inflammatory cells were seen in the lesions (Fig. 1, arrow). 38 Mesenchymal cells with collagen deposition, demonstrable with azan-Mallory staining, simultaneously appeared on PSI days 1 and 2, being more prominent on PSI day 2. The collagen deposition was irregularly seen near injured hepatocytes or focally within the lobules (Fig. 3, arrowhead, inset) as the lesions did not specifically affect the perivenular or periportal areas. On PSI days 3 and 5, the lesions recovered to control levels (Figs. 2, 4). 38 Collectively, D-GalN-induced hepatic lesions were characterized by multifocal lesions consisting of simultaneous development of hepatocyte degeneration/necrosis, inflammatory cell infiltration, and reparative fibrosis.
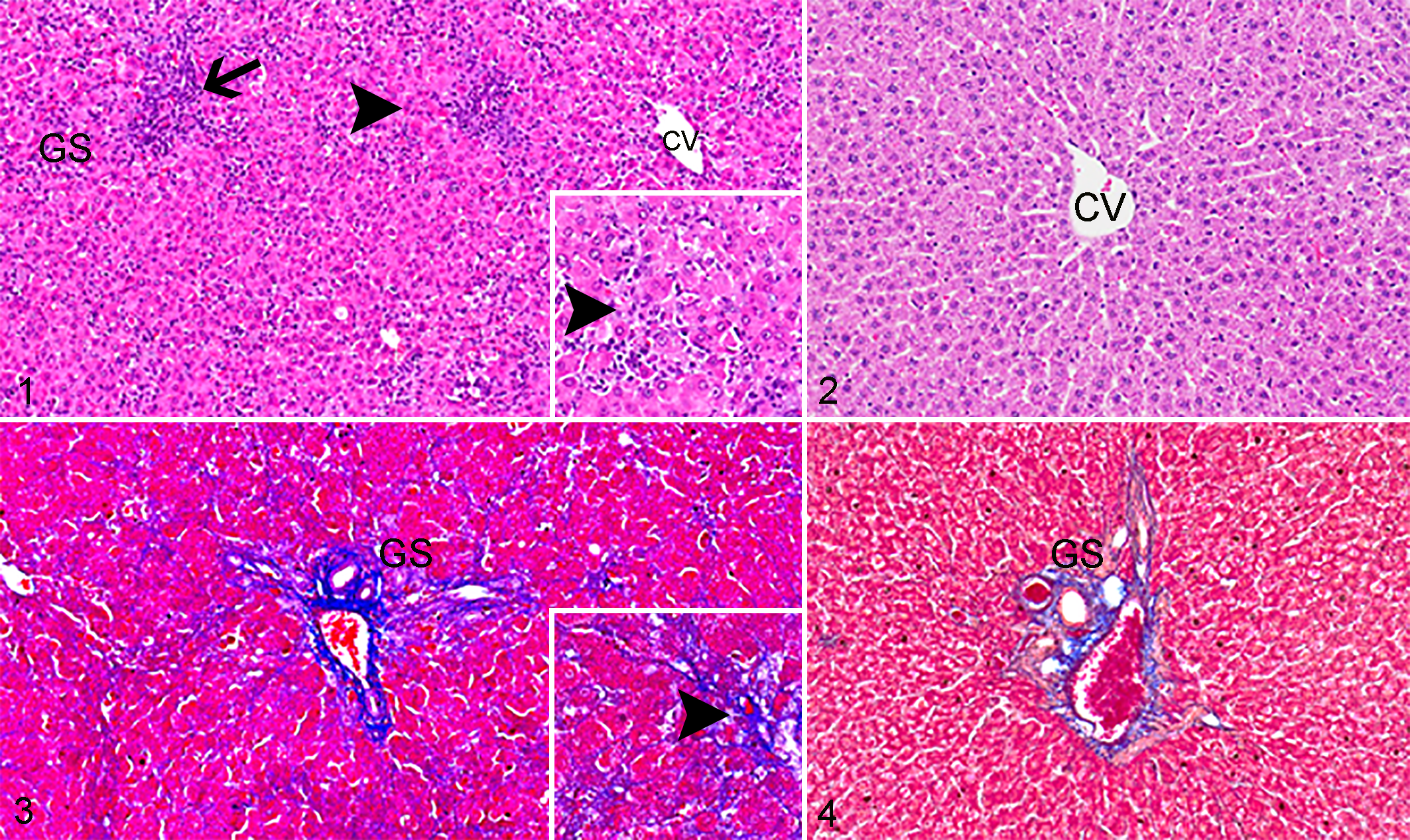
D-galactosamine (D-GalN)-induced liver injury, liver, rat. CV; central vein, GS; Glisson’s sheath.
Single-Label Immunohistochemistry for α-SMA, Vimentin, Desmin, and GFAP
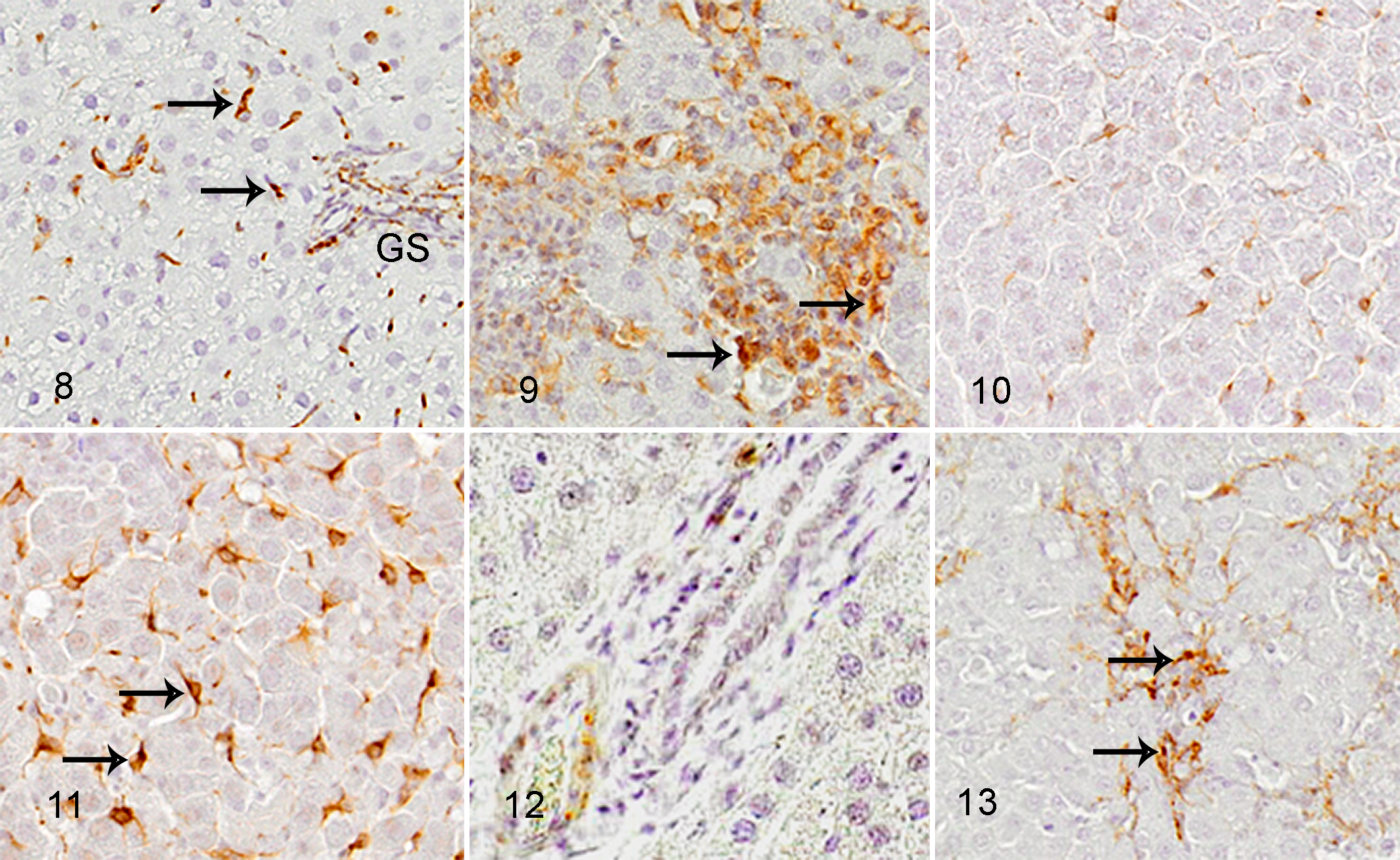
In control livers, mesenchymal HSCs positive for α-SMA (Fig. 5) and desmin (Fig. 12) were not seen, whereas HSCs reacting to vimentin (Fig. 8) and GFAP (Fig. 10) were present along the sinusoid, likely representing HSCs. 44
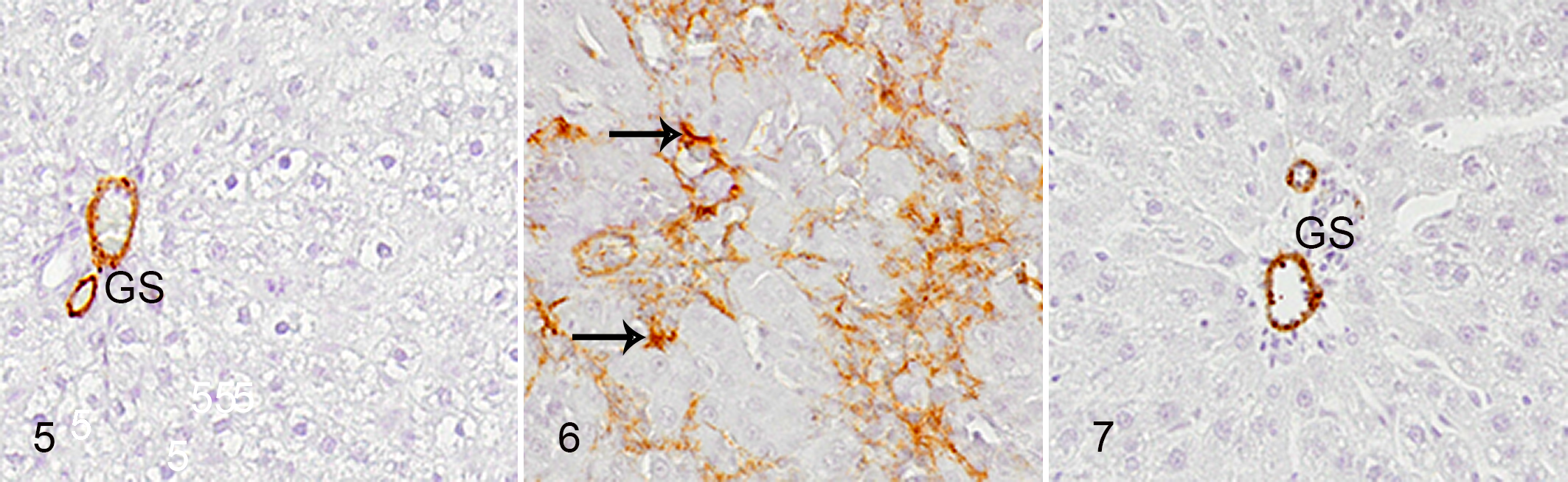
Liver, rat. Immunohistochemistry for α-SMA.
Liver, rat. Immunohistochemistry (IHC).
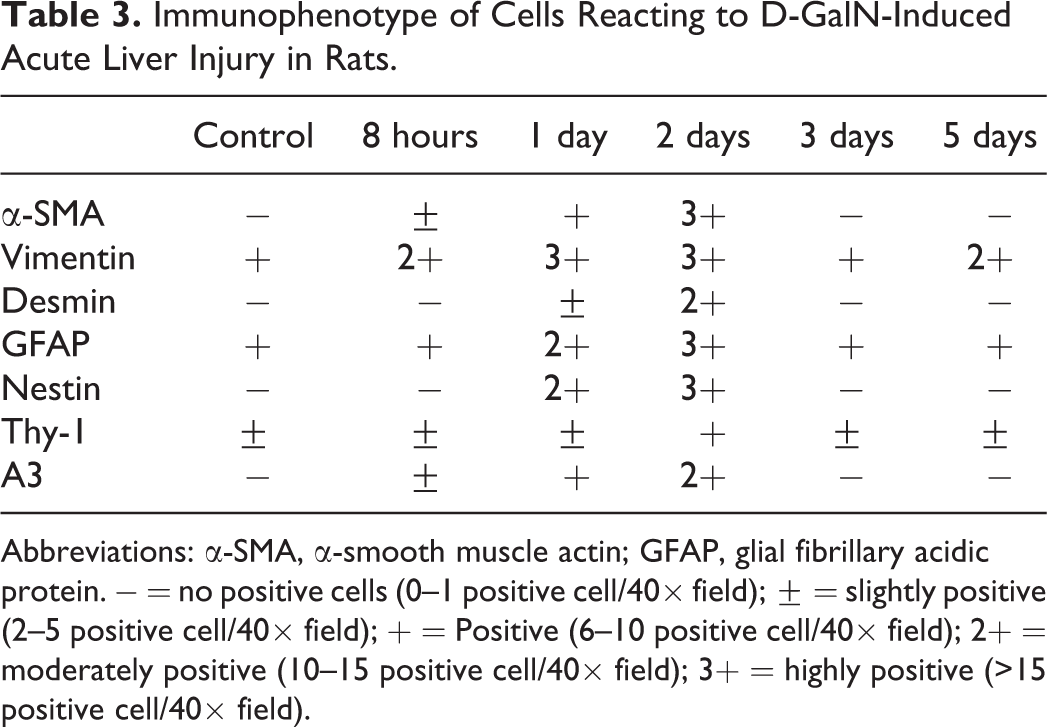
In D-GalN-induced hepatic lesions, mesenchymal cells reacting to vimentin (Fig. 9) began to increase on PSI day 1. Additionally, mesenchymal cells reacting to α-SMA (Fig. 6), desmin (Fig. 13), and GFAP (Fig. 11) increased on PSI days 1 and 2 and were maximal on PSI day 2. The cells reacting to these markers were seen diffusely or focally within the lobules, particularly in areas with collagen deposition. Some cells reacting to GFAP were located along the sinusoid, indicating activated HSCs (Fig. 11). Other cells reacting to α-SMA (Fig. 6), vimentin (Fig. 9), and desmin (Fig. 13) were present as clusters of several cells, indicating the development of myofibroblasts; these cells were round to spindle-shaped or stellate. On PSI days 3 and 5, α-SMA-positive reaction was seen only in the vascular wall smooth muscle similar to that in controls (Fig. 7). Other mesenchymal cells reacting to these markers decreased quickly, except for increased number of vimentin-positive cells on PSI day 5 (Table 3).
Immunophenotype of Cells Reacting to D-GalN-Induced Acute Liver Injury in Rats.
Abbreviations: α-SMA, α-smooth muscle actin; GFAP, glial fibrillary acidic protein. − = no positive cells (0–1 positive cell/40× field); ± = slightly positive (2–5 positive cell/40× field); + = Positive (6–10 positive cell/40× field); 2+ = moderately positive (10–15 positive cell/40× field); 3+ = highly positive (>15 positive cell/40× field).
Double-Label Immunofluorescence for Vimentin, α-SMA, Desmin, and GFAP
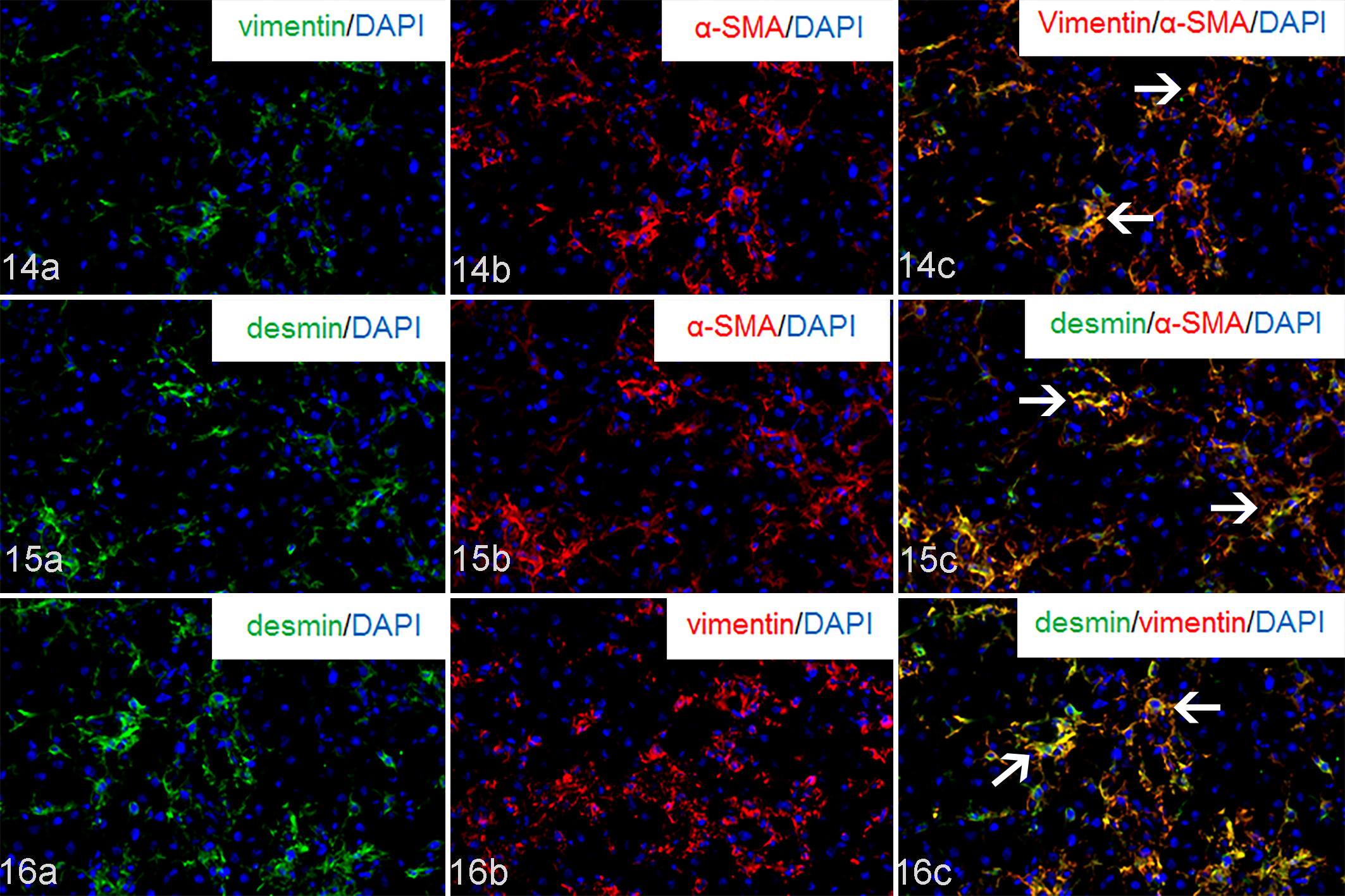
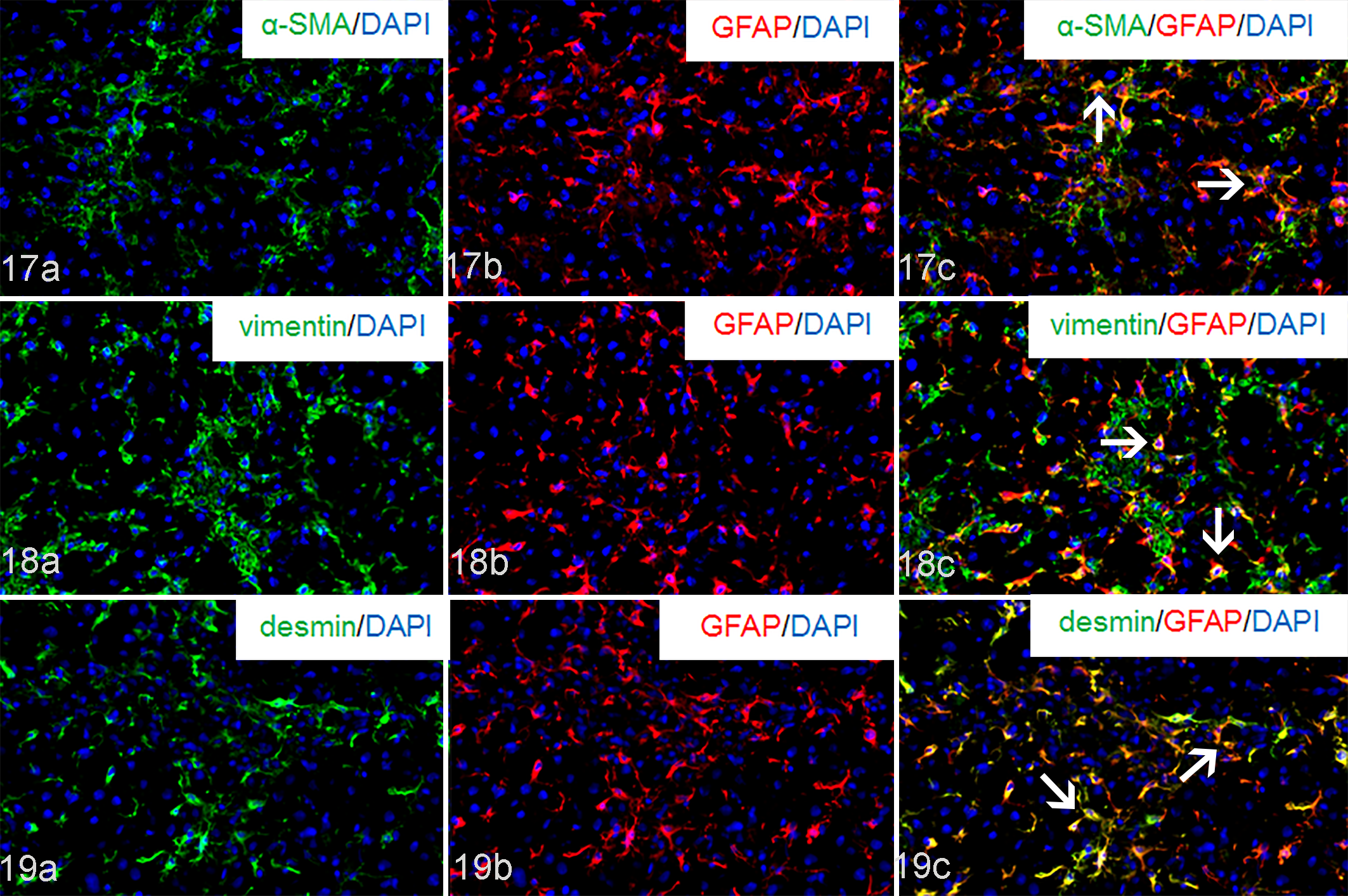
Since myofibroblasts express vimentin, desmin, and α-SMA during development, 7 liver samples on PSI day 2 were studied using double immunofluorescence. The percentage of mesenchymal cells reacting to vimentin/α-SMA (Fig. 14a–c), desmin/α-SMA (Fig. 15a–c), and desmin/vimentin (Fig. 16a–c) were 82%, 92%, and 86%, respectively (Supplemental Fig. S1). In hepatic fibrosis, GFAP-expressing mesenchymal cells may be regarded as myofibroblasts derived from HSCs, because HSCs express GFAP in normal liver. 2 In the present fibrotic lesion, the percentage of cells that were double-positive for α-SMA/GFAP (Fig. 17a–c), vimentin/GFAP (Fig. 18a–c), and desmin/GFAP (Fig. 19a–c) per GFAP-single positive cells was 74%, 64%, and 79%, respectively (Supplemental Fig. S2).
D-GalN-induced injury, liver, rat. Double immunofluorescence on post single injection day 2. Arrows indicate double-positive cell reaction. 4′,6-diamidino-2-phenylindole (DAPI) for nuclear stain.
D-GalN-induced liver injury, liver, rat. Double immunofluorescence on post single injection day 2. Arrows indicate double-positive cell reaction. 4′,6-Diamidino-2-phenylindole (DAPI) for nuclear stain.
Immunoreactivity of Immature Mesenchymal Cell Markers to Thy-1, A3, and Nestin
Thy-1, nestin, and A3 were used as markers of immature mesenchymal cells 31,48 to investigate the possible derivation of myofibroblasts in D-GalN-induced liver lesions. Round or spindle-shaped cells reacting to nestin and A3 were seen in the affected areas on PSI days 1 and 2, with the greatest on PSI day 2 (Figs. 20, 21; Table 3). A small number of Thy-1-positive cells were seen in the portal space in control livers. In D-GalN-induced liver lesions, the positive cells slightly increased in the affected areas mainly in portal space (around the Glisson’s sheath) PSI on day 2 (Table 3); Thy-1-positive cells were also located around the interlobular bile ducts (Fig. 22).
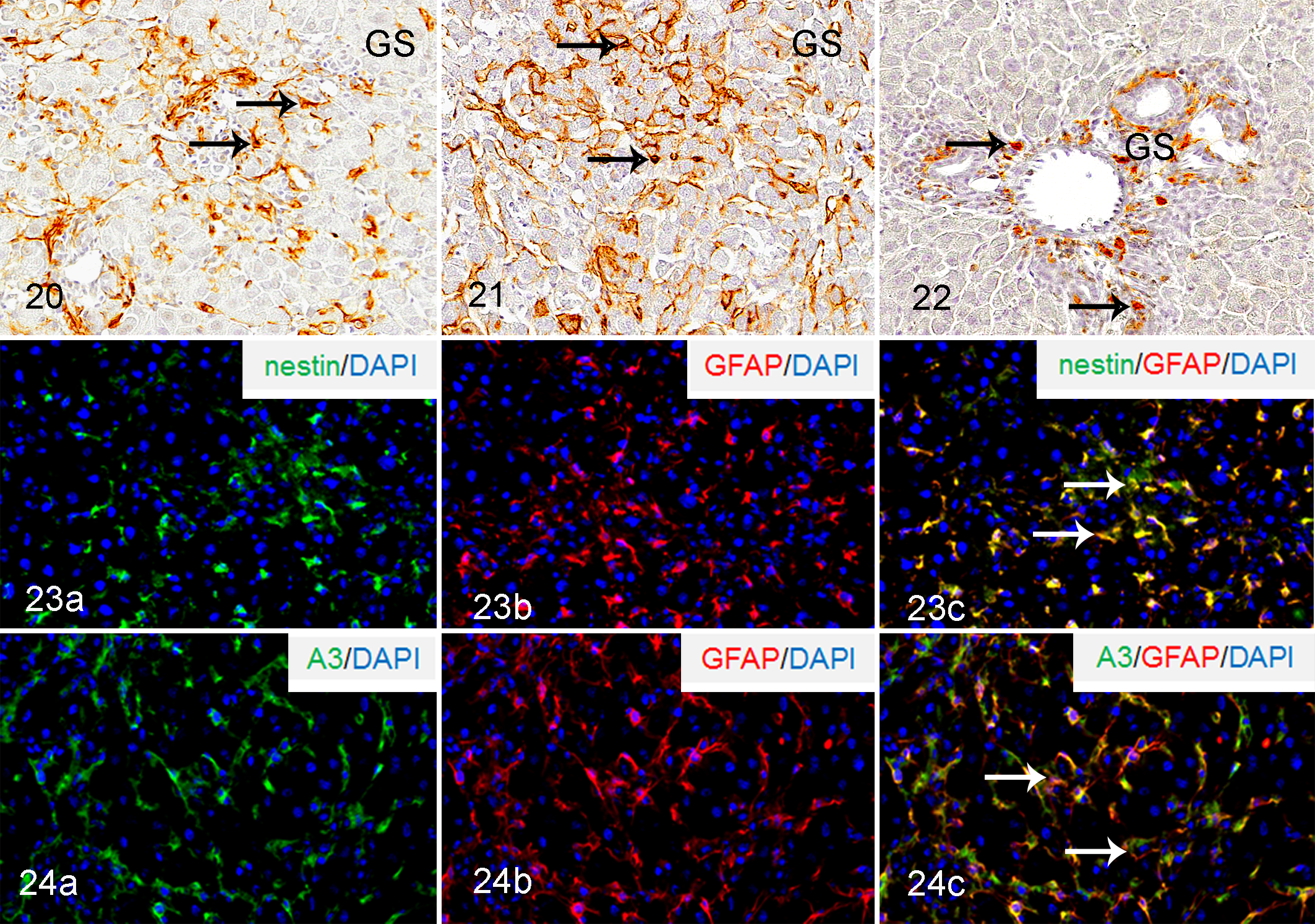
D-GalN-induced liver injury, liver, rat; on post single injection day 2.
By double immunofluorescence staining, 67% and 76% of nestin- and A3-positive cells showed co-expression for GFAP, respectively, on PSI day 2 (Figs. 23a–c and 24a–c; Supplemental Fig. S3). Double immunofluorescence for GFAP and Thy-1 could not be performed, since the isotype of both antibodies was mouse IgG1.
Fibrosis-Related Cytokine Gene Expression
TGF-β1, PDGF-β, and TNF-α mRNAs were significantly increased on PSI days 1 and 2 (Figs. 25–27), and Timp2 mRNA was significantly increased on PSI day 2 (Fig. 28). Furthermore, mRNA expression of Mmp2 showed a consistent significant increase during the observation period from PSI 8 hours and day 5 (Fig. 29).
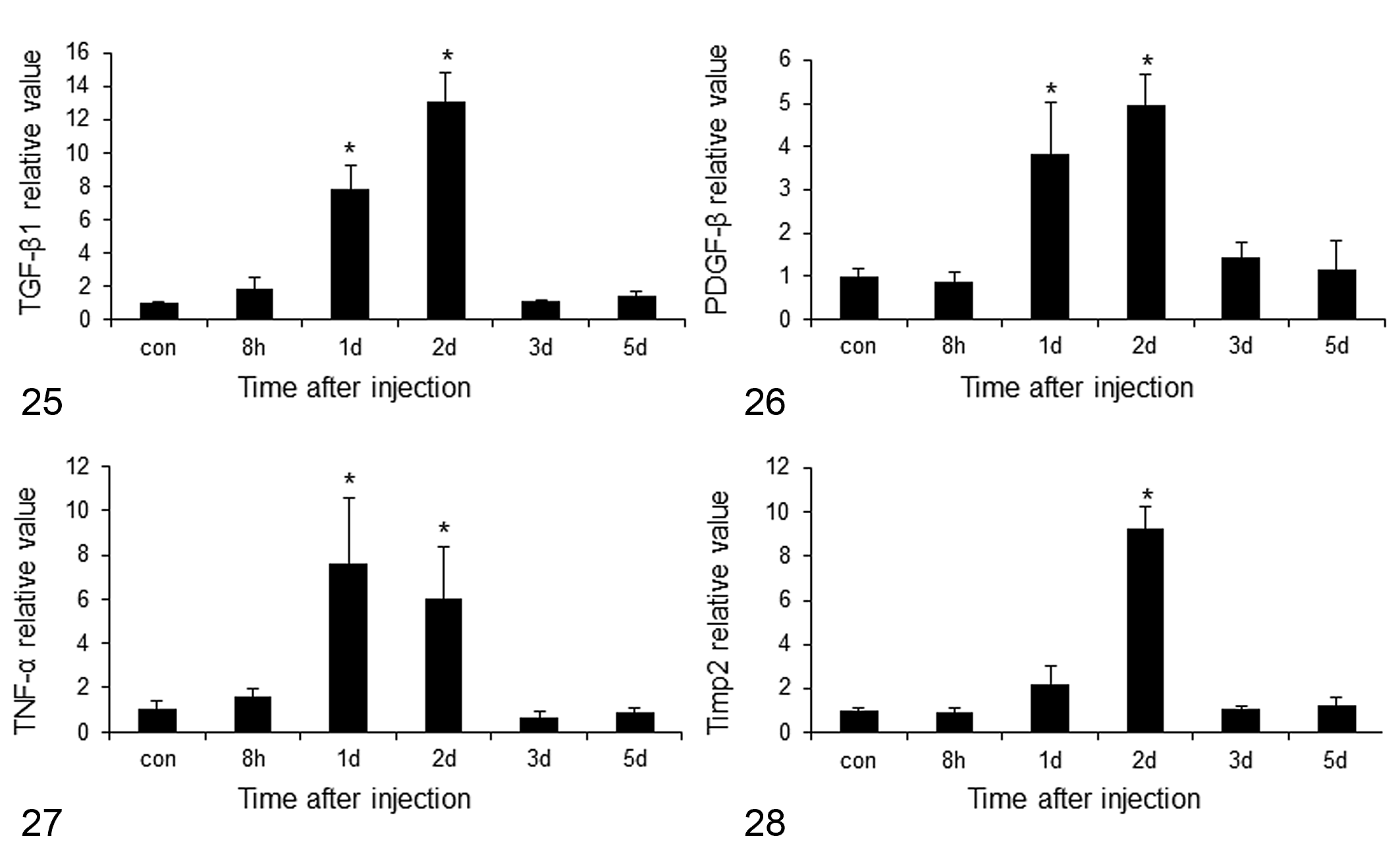
mRNA expression of myofibroblasts and mesenchymal cell-related factors in rats at different times after single injection of D-GalN.
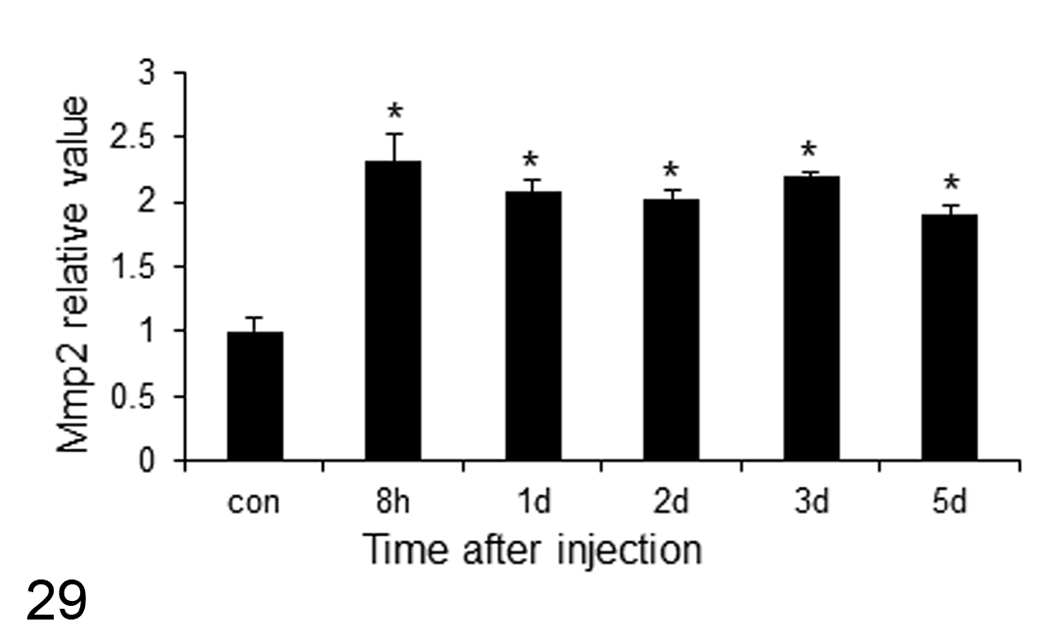
mRNA expression of Mmp2 (matrix metalloproteinase-2) at different times after single injection of D-GalN. Con, control rats. Data were normalized to 18S mRNA level. n = 4 per group; *, P< 0.05 versus control, by Dunnett’s test.
Discussion
Histopathology of D-GalN-Induced Rat Liver Lesions
D-GalN is experimentally used for induction of hepatic lesions in rats and mice. 27,50 The D-GalN-induced hepatic lesions are different from those caused by thioacetamide and carbon tetrachloride, in that the latter 2 chemicals induce hepatocyte injury exclusively in the perivenular area. On the other hand, D-GalN-induced hepatic lesions were characterized by multifocal degeneration and coagulation necrosis within the lobules, showing no special areas such as the perivenular and periportal areas. In perivenular hepatic lesions induced by thioacetamide and carbon tetrachloride in rats and mice, the fibrosis also commonly occurs in the affected perivenular areas, of which the lesion is characterized by focal appearance of myofibroblasts and collagen deposition. 11,17 Interestingly, the collagen depositions in D-GalN-induced hepatic lesions were seen irregularly within the lobules, presumably resulting from the multifocal distribution of the primary lesions. In human acute virus hepatitis, there is also multifocal necrosis/degeneration of hepatocytes and subsequent inflammation/reparative fibrosis exclusively within the lobules. 2,36 Thus, the distribution of multifocal lesions consisting of necrosis/degeneration of hepatocytes and inflammation within the lobules seen in the present D-GalN-induced liver injury are similar to that reported in human acute virus hepatitis. 36,38
Properties of Mesenchymal Cells in D-GalN-Induced Rat Liver Injury
α-SMA expression is the most reliable marker of activated HSCs after hepatocyte injury, and also of myofibroblasts formed in fibrogenesis. 6,15 Additional markers of developing myofibroblasts are vimentin, an intermediate filament protein expressed mainly by mesenchymal cells, 12 and desmin, a subunit of intermediate filament in cardiac, skeletal, and smooth muscle. 33 It has been considered that HSCs could transform into myofibroblasts in liver injury. 9,10,14 In the present D-GalN-induced hepatic lesions, mesenchymal cells reacting to α-SMA, vimentin, and desmin were diffusely or focally observed within the lobules on days 1 and 2; these were most numerous on PSI day 2 and decreased on days 3 and 5 to control levels. The appearance of α-SMA-positive HSCs in D-GalN-induced liver injury is also similar to that in human viral hepatitis. 19 Furthermore, double immunofluorescence analysis of D-GalN-induced hepatic lesions on PSI day 2 showed that approximately 82% and 92% of α-SMA-expressing cells reacted simultaneously to vimentin and desmin, respectively, and approximately 86% of vimentin-expressing mesenchymal cells co-expressed desmin. Thus, myofibroblasts in the D-GalN-induced hepatic lesions could express α-SMA, vimentin, or desmin in varying degrees, as well as with combinations of these cytoskeletal proteins. Myofibroblasts in cutaneous fibrosis show immunophenotypes for α-SMA and vimentin, but not desmin, 22 suggesting differences in cellular characteristics of myofibroblasts between cutaneous fibrosis and hepatic fibrosis.
GFAP expression is considered as an authentic marker for quiescent HSCs in normal liver; additionally, in liver injury, activated HSCs which may be regarded as myofibroblasts reacted to GFAP. 30,44 In the present D-GalN-induced hepatic lesions, GFAP-positive cells were most frequent on PSI day 2; the diffuse or focal distribution of GFAP-positive cells was similar to those of myofibroblasts reacting to α-SMA, vimentin, and desmin. The cells reacting to GFAP were fewer on PSI days 3 and 5, like the kinetics of myofibroblasts reacting to α-SMA, vimentin, and desmin. Similarly, GFAP-positive cell accumulation was seen in foci of fibrosis in the perivenular areas in thioacetamide-induced hepatic lesions. 44 Double immunofluorescence analysis of D-GalN-induced hepatic lesions on PSI day 2 showed that 64%, 79%, and 74% of GFAP-expressing cells co-expressed vimentin, desmin, and α-SMA, respectively. Collectively, it was considered that myofibroblasts expressing α-SMA, vimentin, and desmin might be generated from HSCs originally expressing GFAP. In cutaneous and renal fibrosis, myofibroblasts reacting to GFAP were not seen. 22,49 On the contrary, pancreatic stellate cells, which are located around acinar cells in the pancreas, express GFAP, and myofibroblasts in pancreatic fibrosis are known to express GFAP, indicating the derivation of pancreatic stellate cells to myofibroblasts. 13,25 The cellular features of pancreatic myofibroblasts may be similar to those of hepatic myofibroblasts in their expression of α-SMA, vimentin, desmin, and GFAP.
TGF-β1 is produced mainly by infiltrating macrophages reactive to injury. 39,42 In liver injury, PDGF-β is secreted by resident Kupffer cells and inflammatory cells. 32 TGF-β1 and PDGF-β are important for induction and proliferation of myofibroblasts. 24 TNF-α is a pro-inflammatory cytokine and has immune-regulatory properties; 28,35 this factor could induce inflammatory cells which are able to produce factors such as TGF-β1 and PDGF-β. In the present D-GalN-induced hepatic lesions, mRNA expression of TGF-β1, PDGF-β, and TNF-α were increased simultaneously on days 1 and 2 when hepatocyte lesions were first seen. It is likely that development of myofibroblasts from HSCs might be related to increased expression of these fibrogenic factors. Increase in TNF-α and TGF-β1 mRNA was similarly reported in human viral hepatitis. 41
Generally, Mmp2 and Timp2 have been documented to be produced by activated HSCs after injury. 20 In the current D-GalN-induced hepatic lesions, mRNA expression of Timp2 increased on PSI day 2, and increased Mmp2 mRNAs was persistently seen from PSI 8 hours on to PSI day 5. Mmp2 may be associated with degradation of tissues in liver injury and in fibrotic lesions, and its function is inhibited by Timp2. Increased level of Timp2 on PSI day 2 might be related to that of Mmp2 at early stages when hepatic lesions began to be seen on PSI days 1 and 2; these factors could contribute to the development of hepatic lesions in the D-GalN-injected rats. In addition, increased expression of Mmp2 mRNA, which was still seen on PSI days 3 and 5, might have been related to continuous remodeling of hepatocytes after injury, although there were no significant changes in HE-stained sections on PSI days 3 and 5.
Possible Derivation of Myofibroblasts From Mesenchymal Immature Cells
A3 was generated as an antibody recognizing immature mesenchymal cells such as stem cells in the bone marrow and vascular pericytes. 48 Nestin is an intermediate filament and is expressed by neuronal stem cells 31 and activated HSCs in rats. 44 In the present D-GalN-induced hepatic lesions, mesenchymal cells reacting to A3 and nestin were increased in the affected areas on PSI days 1 and 2, indicating the participation of immature mesenchymal cells in the lesion development. Furthermore, the double immunofluorescence staining using samples on PSI days 2 showed that A3 and nestin were expressed in 76% and 67% of GFAP-positive cells, respectively. Hepatic myofibroblasts co-expressing A3/GFAP or nestin/GFAP might be derived from HSCs with more immature mesenchymal nature; such cells are presumed to be partly recruited from the bone marrow, because A3 labels mesenchymal stem cells in rat bone marrow. 48 Recently, it has been reported that multilineage-differentiating stress-enduring (Muse) cells, a stem cell generated from the bone marrow, may participate in lesion development in distant organs, and that Muse cells may also contribute to functional recovery after tissue injury. 26
Thy-1 is a glycoprotein expressed in mesenchymal stem cells in the bone marrow and vascular pericytes, and its antibody has been used as a marker for immature mesenchymal cells. 1,5 In control livers, a few Thy-1-positive cells were present in the interlobular bile ducts in the portal space (Glisson’s sheath). A small number of Thy-1-reacting mesenchymal cells were seen in and around the portal space in the D-GalN-injected rats on PSI day 2. In cutaneous fibrosis, Thy-1-positive cells have been considered to transdifferentiate into myofibroblasts appearing in the fibrotic lesions. 8,22 Participation of Thy-1-positive immature mesenchymal cells has not been reported in perivenular fibrosis after liver injury induced by thioacetamide. 44
In conclusion, the present study showed that there was a dynamic participation of mesenchymal cells in hepatic lesions induced by D-GalN. The mesenchymal cells showed myofibroblastic immunophenotypes for α-SMA, vimentin, and desmin in varying degrees, or in combination. The hepatic myofibroblasts might be derived partly from HSCs with immature nature co-expressing GFAP/A3 and GFAP/nestin or Thy-1-positive immature mesenchymal cells. Additionally, fibrosis-related factors such as TGF-β1, PDGF-β, TNF-α, Mmp2, and Timp2 were related to development of hepatic fibrosis in which myofibroblasts appeared diffusely or focally. The immunophenotypic characteristics of hepatic myofibroblasts, which were clarified by the present study, are useful for understanding the pathogenesis of hepatic lesions, because the present D-GalN-induced hepatic lesions resemble liver lesions of human acute viral hepatitis. In addition, the present immunophenotypical analyses for myofibroblasts may give some clues for pathological studies on hepatic mesenchymal cells.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Rahman_et_al - Characterization of Immature Myofibroblasts of Stellate Cell or Mesenchymal Cell Origin in D-Galactosamine-Induced Liver Injury in Rats
Supplemental Material, Combined_supplemental_materials-Rahman_et_al for Characterization of Immature Myofibroblasts of Stellate Cell or Mesenchymal Cell Origin in D-Galactosamine-Induced Liver Injury in Rats by Nahid Rahman, Mizuki Kuramochi, Takeshi Izawa, Mitsuru Kuwamura and Jyoji Yamate in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by JSPS KAKENHI Grant Numbers 26292152 and 19H03130 (to Yamate), by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) from AMED and Grand Numbers 3P18 am 0101123 (to Yamate), and by the Otsuka Toshimi Scholarship Foundation (Number 17-S42, 18-S32 to N. Rahman).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
