Abstract
Pigs are considered one of the relevant animal models for ocular research as they share several histological and anatomical similarities with the human eye. With the increasing interest in juvenile animal models, this study aimed to describe the postnatal development of ocular structures in 16 Göttingen minipigs and 25 F2 domestic pigs, between birth and 6 months of age, using histopathology and immunohistochemistry against Ki-67, caspase-3, calbindin, glial fibrillary acidic protein, rhodopsin, and synaptophysin. All ocular structures in both pig breeds were incompletely developed at birth and for variable periods postnatally. Noteworthy histological features of immaturity included vascularization in the corneal stroma in neonatal Göttingen minipigs, increased cellularity in different substructures, remnants of the hyaloid vasculature, short and poorly ramified ciliary body processes, and a poorly developed cone inner segment. Increased cellular proliferation, highlighted by abundant Ki-67 immunolabeling, was observed in almost all developing structures of the pig eye for variable periods postnatally. Apoptosis, highlighted with caspase-3 immunolabeling, was observed in the retinal inner nuclear layer at birth and in the regressing hyaloid vasculature remnants. Immunohistochemistry against rhodopsin, synaptophysin, and calbindin demonstrated the short size of the developing photoreceptors and the immature cone inner segment morphology. Calbindin labeling revealed significant differences in the amount of positively labeled cone nuclei between the retinal area centralis and the non–area centralis regions. The elongation of Müller cell processes in the developing retina was shown with glial fibrillary acidic protein. In both pig breeds, the eyes reached histomorphological and immunohistochemical maturity at 6 months of age.
Over the last few decades, Göttingen minipigs have become increasingly used in preclinical toxicologic studies and translational research. 8,38,64,76,80 With their small size, easy handling, and anatomical and physiological features resembling humans, Gottingen minipigs have become an attractive animal model and an interesting nonrodent alternative to dogs and nonhuman primates. While the Göttingen minipig is generally preferred for practical reasons, domestic pigs offer specific advantages for orthopedic research and preclinical studies of medical devices in addition to being used for veterinary drug development. 85
The eye is the only special sense organ that is routinely evaluated by pathologists during preclinical general toxicity studies for the development of new drugs. 9 Therefore, a good understanding of the normal histology of this complex organ in every laboratory animal species is crucial. The anatomic and histologic similarities between the eyes of humans and pigs—including the ocular globe size, the holangiotic retinal vasculature, the absence of a tapetum, and the cellular composition and configuration of the retina—make the Göttingen minipig and the domestic pig excellent animal models for ocular research in various fields. 47,59,80,88 Pigs have been used not only for preclinical ocular studies but also to improve surgical procedures, develop ocular grafts, and to study several important veterinary and human eye conditions such as glaucoma, retinal detachment, and diabetic retinopathy, among others. 29,31,45,57,59,80
Notably, the pig is particularly relevant for retinal research because the porcine area centralis mimics to some extent the macular region in the human eye. 13,15,39,44,47,88 The macula and fovea in the retina of humans and nonhuman primates have been extensively characterized. The macula can simply be defined as a region of high ganglion cell density (except at the level of the fovea), while the fovea corresponds to a focal area within the macula that contains a significantly higher cone density and no rods. 11,40,42,90 In pigs, the area centralis (or visual streak) is a horizontally oriented region dorsal to the optic disc that is characterized by higher densities of ganglion cells and cone photoreceptors compared with other retinal regions. Unlike in humans and nonhuman primates, the high cone density area in pigs is not concentrated in a pit-shaped zone (the fovea) but rather corresponds to a wide horizontal area. 13,15,39,44,47,88 Although the normal ocular anatomy and histology in adult pigs have been described sporadically in the literature, information on the postnatal ocular histological development in neonatal and juvenile pigs is scarce or even nonexistent for most eye structures. 2,35,44,57,81,87 Histological descriptions of the area centralis in pigs are rare in the literature; more important, the postnatal histomorphological development of this important retinal region has not been previously documented in pigs, to our knowledge.
Juvenile animals in preclinical toxicologic studies have generated substantial interest over the last decades because of their potential value in evaluating the safety of new drugs intended to be administered clinically in children. 67,83 Indeed, the immaturity of different organs at birth and for various time periods thereafter in young animals and children can affect responses to xenobiotics in various ways that cannot be predicted using mature animals. 67,83 However, the lack of published histologic references or available age-matched controls for ocular tissues of neonatal/juvenile pigs complicates the pathologist’s evaluation of eye sections from young pigs in the preclinical toxicity field. Indeed, developmental features or immaturity must not be misinterpreted as drug-related changes. Among histological characteristics of organogenesis, cellular proliferation and apoptosis are 2 events that can be variably induced and/or inhibited by drugs, including in the eye. 25,30,34,53 This underscores the importance of characterizing the ocular developmental stage at birth and to understand if mitosis indicative of proliferation and apoptosis indicative of programmed cell death occur physiologically in the pig eye during the postnatal phase. Thus, this study aimed to provide detailed histologic and immunohistochemical reference information on the postnatal ocular development of Göttingen minipigs and F2 domestic pigs from birth to 6 months of age, with a special focus on the retinal area centralis region.
Materials and Methods
Animals
This study included 25 F2 domestic pigs obtained from the Diagnostic Service of the Faculty of Veterinary Medicine of the Université de Montréal, Saint-Hyacinthe, Quebec, Canada, which came from different pig farms throughout the province of Quebec and were submitted alive for necropsy. These animals were submitted for unrelated diagnostic reasons (mostly diarrhea cases), but a macroscopic ocular examination was performed to ensure that both eyes of F2 domestic pigs were grossly normal prior to being included in the study. The absence of ocular lesions was thereafter confirmed with histological evaluation by an American College of Veterinary Pathologists (ACVP)-certified pathologist. F2 domestic pig age groups included between 2 and 4 animals per group as follows: postnatal day (PND) 1 (n = 4), PND3 (n = 4), PND7 (n = 3), PND14 (n = 4), PND21 (n = 4), PND28 (n = 2), 2 months (n = 2), and 6 months (n = 2). Whenever available, equal numbers of males and females were included for each time point. F2 domestic pigs were anesthetized with a ketamine/xylazine mixture and euthanized via electrocution followed by exsanguination.
Göttingen minipigs (n = 16) were donated by Marshall BioResources USA and were of different ages as follows: PND1, PND7, PND14, PND21, PND28, 2 months, 3 months, and 6 months. Each time point included 2 animals, 1 female and 1 male. Göttingen minipigs were euthanized with sodium pentobarbital following general anesthesia with tiletamine/zolazepam mixture.
In addition, the eyes of male and/or female Göttingen minipigs at 6 (n = 2), 8 (n = 1), 9 (n = 2), 10 (n = 2), 12 (n = 3), and 24 (n = 1) months old were examined histologically and compared to identify the age at which all ocular structures reached histomorphological maturity, which was established at 6 months of age. Therefore, this time point was used as a reference for histologically mature ocular structures in Göttingen minipigs and F2 domestic pigs.
All procedures were conducted in accordance with the principles outlined in the current guidelines published by the Canadian Council on Animal Care (for F2 domestic pigs) and the Guide for the Care and Use of Laboratory Animals (for Göttingen minipigs).
Necropsy and Histology
For Göttingen minipigs, both eyes and optic nerves were fixed by immersion in neutral-buffered 10% formalin for 24 to 48 hours and then transferred to 70% ethanol until trimming. For F2 domestic pigs, one eye with the optic nerve was fixed in Davidson’s solution for 24 hours and one eye with the optic nerve was fixed in neutral-buffered 10% formalin for 24 hours, and then both eyes were transferred to 70% ethanol until trimming. Eyes were placed in fixative within 15 minutes after euthanasia. Prior to processing, each eye was consistently trimmed vertically (mid-axial) following a standardized protocol. A straight cut of the entire eye was performed starting caudally alongside the temporal aspect of the optic nerve and perpendicular to the ciliary artery/groove (through the sclera, retina, vitreous, lens, and finally the cornea in sequence; Fig. 1). The fixation and trimming methods used in this study followed the current recommended practices for the preparation of the eyes of large nonprimate laboratory animals for toxicologic pathological studies. 9 This trimming orientation allowed the sampling of both area centralis (or visual streak) and the non–area centralis areas of the pig retina. The area centralis in pigs corresponds to a horizontally oriented region of the retina located just dorsally to the optic disc and extends both nasally and temporally (Fig. 1). 15,32,39,88 Sections of 6 µm and 5 µm thickness, at the level of the optic disc and including a longitudinal portion of optic nerve, were used for hematoxylin and eosin (HE) staining and immunohistochemistry, respectively. Sections were mounted on TOMO slides for immunohistochemistry, and were dried overnight at 37 °C for HE staining or at 56 °C for immunohistochemistry to enhance tissue adherence to the slides. Routine HE-stained sections of both eyes were used for the histological description. For F2 domestic pigs, fixation in neutral-buffered 10% formalin and Davidsons’ fixative were compared. To compare the histology of the eyes of Göttingen minipigs and F2 domestic pigs, neutral-buffered 10% formalin-fixed sections were used. In this article, ocular structures were considered “histomorphologically mature” when they presented the same histological/morphological characteristics as the fully developed eye, that is, the eyes of pigs aged 6 months or older.
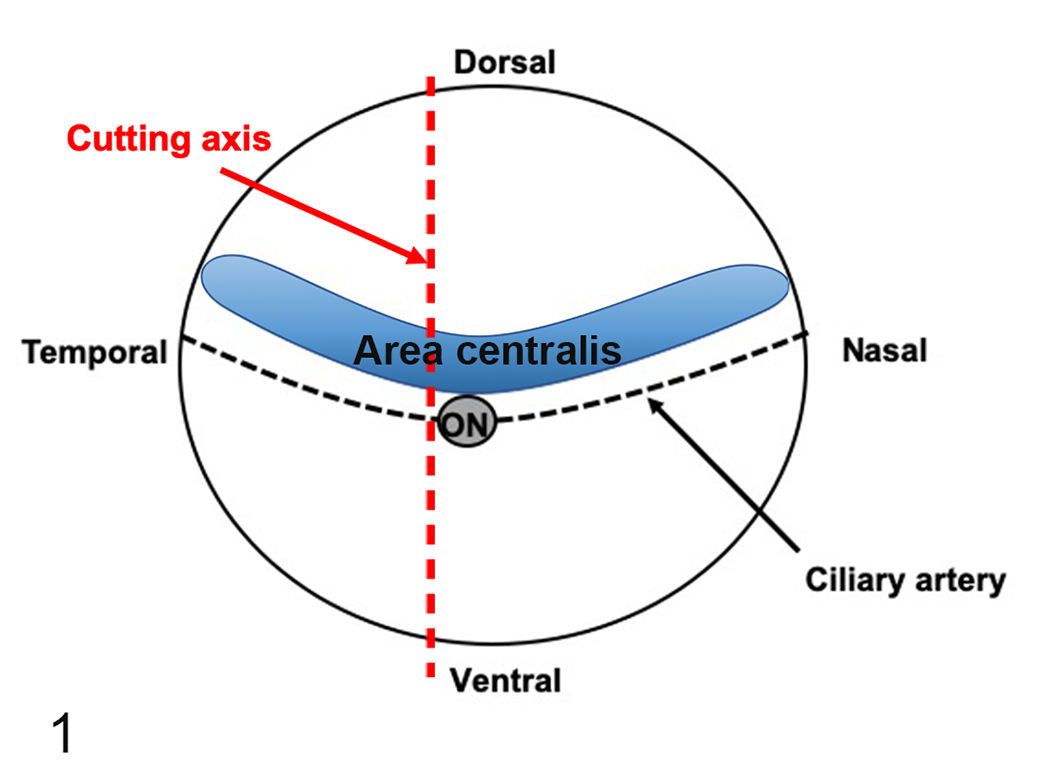
Illustration of the trimming axis for the eyes of Göttingen minipigs and F2 domestic pigs. A straight cut of the entire eye was performed starting caudally alongside the temporal aspect of the optic nerve (ON) and perpendicularly to the ciliary artery. The area centralis in pigs corresponds to a horizontally oriented region of the dorsal retina located just dorsally to the optic nerve/disc and extends both nasally and temporally.
Immunohistochemistry
For both Göttingen minipigs and F2 domestic pigs, immunohistochemistry was performed against Ki-67 (for cellular proliferation), 26 caspase-3 (for apoptosis and non-apoptotic activation of caspase-3), 22 synaptophysin (for synapses in the retina and inner segments of rods and cones), 27 glial fibrillary acidic protein (GFAP; for retinal glial cells), 28,46 rhodopsin (for rod photoreceptors), 2,35,56 and calbindin (for retinal horizontal cells and cone photoreceptors) 2,35,63,65 using 2 animals (only one eye per individual) per time point. All immunohistochemistry was performed on neutral-buffered 10% formalin-fixed tissue, except for Ki-67 in F2 domestic pigs, for which Davidson’s fixative was used. A semiautomatic immunohistochemistry method was performed using a Ventana Discovery XT stainer (version 10.22). A summary of the antibodies and conditions used with the Ventana Discovery XT immunostainer is presented in Table 1. Briefly, sections were deparaffinized, rehydrated, and antigens were unmasked via a heat-induced epitope retrieval technique using a Tris-based buffer at pH 8 to 8.5 (Cell conditioning 1 Cat. 950-124 from Ventana Medical Systems) for variable time and at different temperatures depending on the antigens (Table 1). Sections were then incubated in a 3% H2O2 solution (from the DABMAP kit cat. 760-124 or Chromomap DAB kit cat.760-159, both from Ventana Medical Systems) for 4 minutes to quench the endogenous peroxidase activity. Nonspecific binding sites were blocked with 5% normal goat serum (ab7481, from Abcam) for 32 minutes at room temperature for calbindin and Ki-67 markers. Primary antibodies were diluted in a diluent from Ventana Medical Systems (cat. 760-108) at the appropriate concentrations (Table 1), were manually added to the slides, and the slides were incubated for various time periods and at different temperatures depending on the antibody (Table 1). For all antibodies except for the rhodopsin antibody, sections were then incubated for 32 minutes at room temperature with a biotinylated goat anti-rabbit secondary antibody (BA1000 form Vector Laboratories) and the chromogenic reaction was performed using a streptavidin-biotin peroxidase detection system (DABMAP kit, cat. 760-124 from Ventana Medical Systems). For rhodopsin, tissue sections were incubated for 16 minutes at room temperature with an anti-mouse HRP secondary antibody (OmniMap cat. 760-4310 from Ventana Medical Systems) and the chromogenic reaction was performed using a DAB detection kit (Chromomap DAB kit, cat. 760-159 from Ventana Medical Systems). All slides were counterstained with hematoxylin.
Antibodies and Conditions Used for Immunohistochemistry on Pig Eyes.
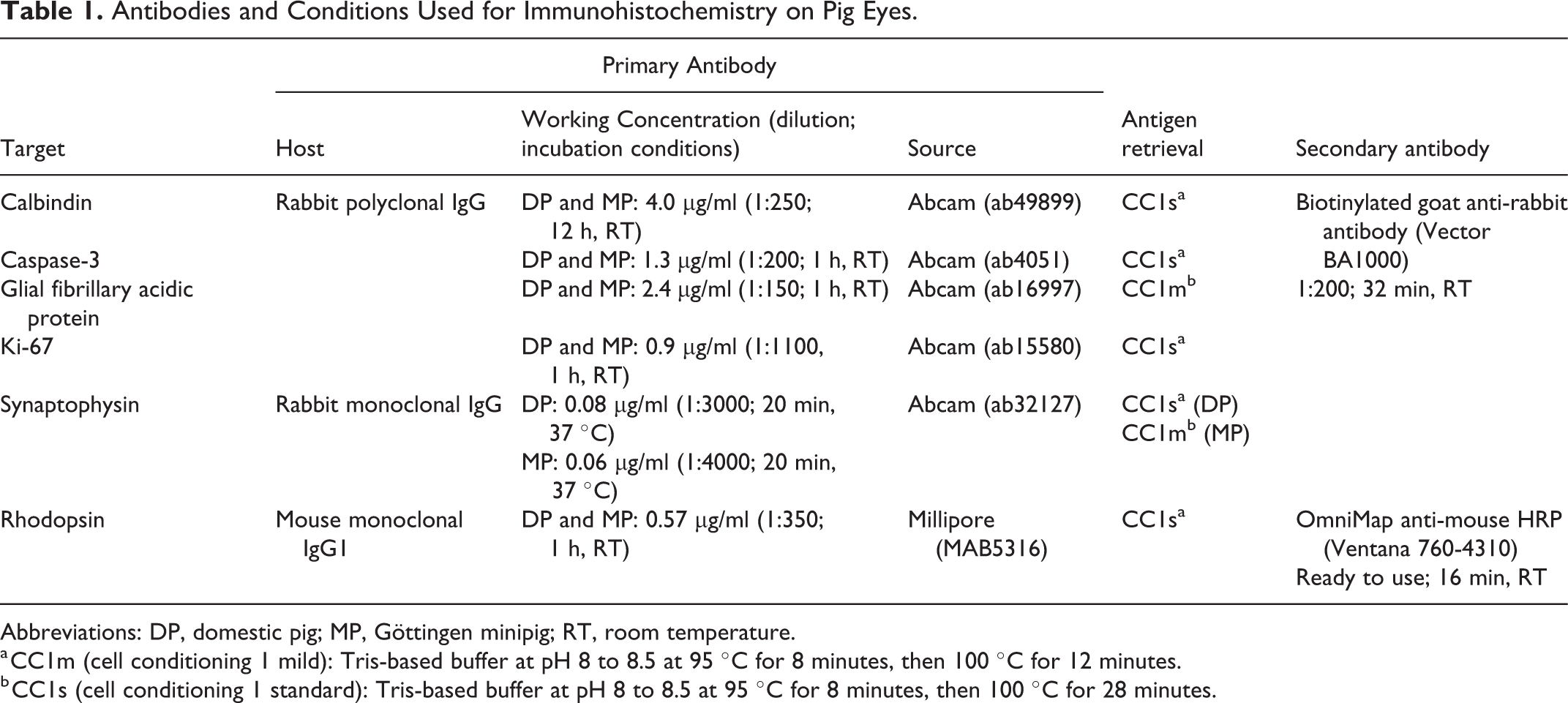
Abbreviations: DP, domestic pig; MP, Göttingen minipig; RT, room temperature.
a CC1m (cell conditioning 1 mild): Tris-based buffer at pH 8 to 8.5 at 95 °C for 8 minutes, then 100 °C for 12 minutes.
b CC1s (cell conditioning 1 standard): Tris-based buffer at pH 8 to 8.5 at 95 °C for 8 minutes, then 100 °C for 28 minutes.
All immunohistochemistry controls were performed on both Göttingen minipig and F2 domestic pig tissues and, for each marker, a negative and a positive control were included. The negative control was performed by substituting the primary antibody with the appropriate isotype-matched immunoglobulin (Ig) diluted to the same concentration as the working concentration of the primary antibody. Normal rabbit polyclonal IgG (ab37415) was used for calbindin, caspase-3, Ki-67, and GFAP; normal rabbit monoclonal IgG (ab172730) was used for synaptophysin; and normal mouse IgG1 (ab81216) was used for rhodopsin (all from Abcam). Positive tissue controls were performed on the following samples that had been fixed and processed with the same conditions as for the ocular tissues: the thymus was used for caspase-3 labeling, small intestine with Peyer’s patches and mesenteric lymph nodes were used for Ki-67 labeling, and ocular sections from an adult Göttingen minipig and an adult F2 domestic pig were used for calbindin, rhodopsin, synaptophysin, and GFAP. All isotype controls resulted in a complete absence of immunolabeling. Positive tissue controls showed specific positive immunolabeling in the expected appropriate structures or cells: apoptotic cells or apoptotic bodies in the thymus for caspase-3; dividing cells in intestinal crypts and Peyer’s patches or lymph nodes germinal centers for Ki-67; retinal ganglion cells, cone photoreceptors, horizontal cells, and outer plexiform layer for calbindin; synapses in the retinal inner and outer plexiform layers for synaptophysin; retinal glial cells and optic nerve for GFAP; and rod photoreceptors for rhodopsin.
Analysis of Histology and Immunolabeling
Because the ocular tissues from Göttingen minipigs and F2 domestic pigs were fixed and processed in 2 different facilities, preliminary standardization of immunohistochemistry was performed for every marker using the aforementioned positive tissue controls in order to establish the optimal protocols resulting in equivalent labeling patterns between the 2 pig breeds. The immunohistochemistry protocols used were identical for both pig breeds except for synaptophysin, for which a higher primary antibody concentration and longer antigen retrieval were needed in the domestic pig tissues in order to obtain the same positive labeling intensity as in the Göttingen minipig tissues. Furthermore, for F2 domestic pigs, since Ki-67 immunohistochemistry resulted in an equivalent labeling pattern regardless of the fixation method, Davidson’s-fixed ocular sections were used for this marker due to better preservation of morphology.
For histological and immunohistochemical evaluation of the retina in both Göttingen minipigs and F2 domestic pigs, 3 distinct regions were compared to determine if variations in histomorphological and/or immunolabeling pattern could be identified between these regions, namely, the ventral retina (ventral to the optic disc), the area centralis portion of the dorsal retina (just dorsal to the optic disc), and the non–area centralis portion of the dorsal retina (the portion between the area centralis and the dorsal ora serrata). The area centralis is easily identifiable histologically as the area of the dorsal retina (corresponding to approximately 1/5 of the dorsal retina, just dorsally to the optic disc), presenting a significantly higher ganglion cell density 15,32,88 compared to the rest of the ventral and dorsal retina. Of note, due to their specific triangular-square shape, 10 the inner segments of cone photoreceptors are easily recognizable in HE-stained histologic sections and by immunohistochemistry, even if negatively labeled with immunohistochemistry.
Analysis of Calbindin Immunolabeling of Cone Cells
Calbindin has been used to label different segments of cone photoreceptors in various species, including Göttingen minipigs. 2,17,18,35,41 Therefore, this marker was used to quantify the number of cone cell nuclei present in each retinal region (ie, the ventral retina, the area centralis portion of the dorsal retina, and the non–area centralis portion of the dorsal retina) for every Göttingen minipig included in this study (1 eye per animal). For each retinal region, pictures of 4 consecutive fields of view at 400× magnification were captured using an Olympus BX53 light microscope (Olympus) and a digital camera (Olympus DP22) linked to the Olympus CellSens imaging software. The length of the retinal segment in every picture was consistently 354 μm. Because the ventral retina and the dorsal non–area centralis regions are large areas, pictures were always taken within the middle third of the ventral and dorsal retina (relative to the optic disc), respectively, in order to evaluate the same regions in every animal. Then, the number of calbindin-positive nuclei located in the outermost layer of the outer nuclear layer (ONL) was counted per 354-μm-long retinal segment (or per microscopic field at 400×). Note that the pictures were taken without including the ganglion cell layer to remain blinded to the retinal region evaluated when doing the counting, and each picture was analyzed randomly and in a blinded manner (blinded to the animal ID and the retinal regions) by an ACVP-boarded pathologist.
For F2 domestic pigs, important variations in labeling intensity were noted in calbindin slides, which did not make them suitable for a proper quantitative evaluation, and therefore, only a qualitative evaluation of this marker was performed.
Statistical Analysis of Calbindin Counts
Statistical analysis was performed using the SAS 9.0 statistical software. Calbindin counts were square root transformed (
Statistical Analysis of the Effect of Age and Retinal Anatomic Site on Calbindin-Positive Nuclei Countsa in the Eyes of Göttingen Minipigs.
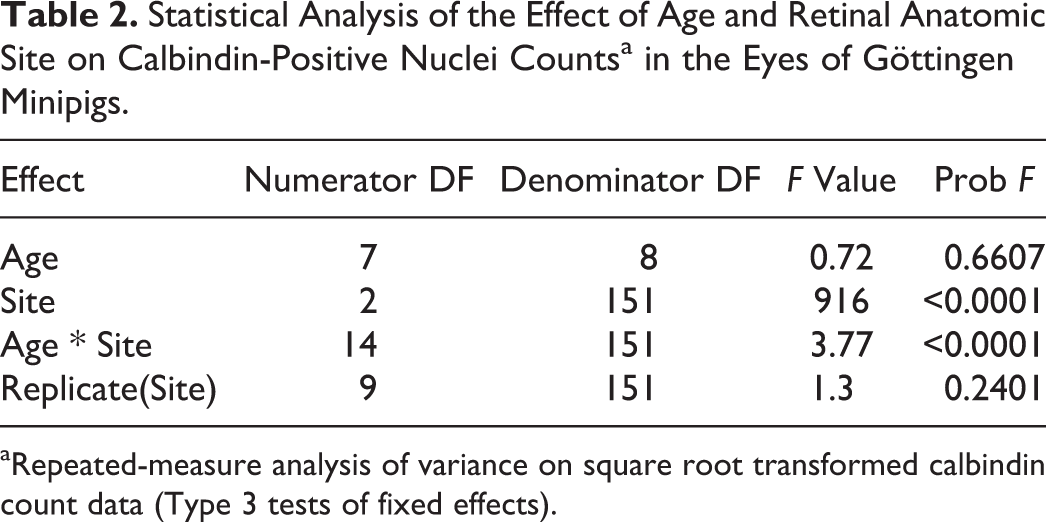
aRepeated-measure analysis of variance on square root transformed calbindin count data (Type 3 tests of fixed effects).
Results
Detailed histological and immunohistological descriptions of the postnatal ocular development of Göttingen minipigs and F2 domestic pigs are presented. Tables 3 and 4 summarize the postnatal time intervals when cellular proliferation and apoptosis were observed in specific ocular structures for both pig breeds, as characterized by the presence of immunopositive cells for Ki-67 and of caspase-3-positive apoptotic cells/apoptotic bodies, respectively. A summary of the ocular structures immunolabeled for calbindin, synaptophysin, rhodopsin, GFAP, and caspase-3 (non-apoptotic activation) is presented in Tables 5 and 6. The PND when each ocular structure reached histomorphological maturity for each pig breed is shown in Table 7.
Age Ranges When Pig Ocular Structures Presented Cellular Proliferation and/or Apoptosis (Part 1)a.
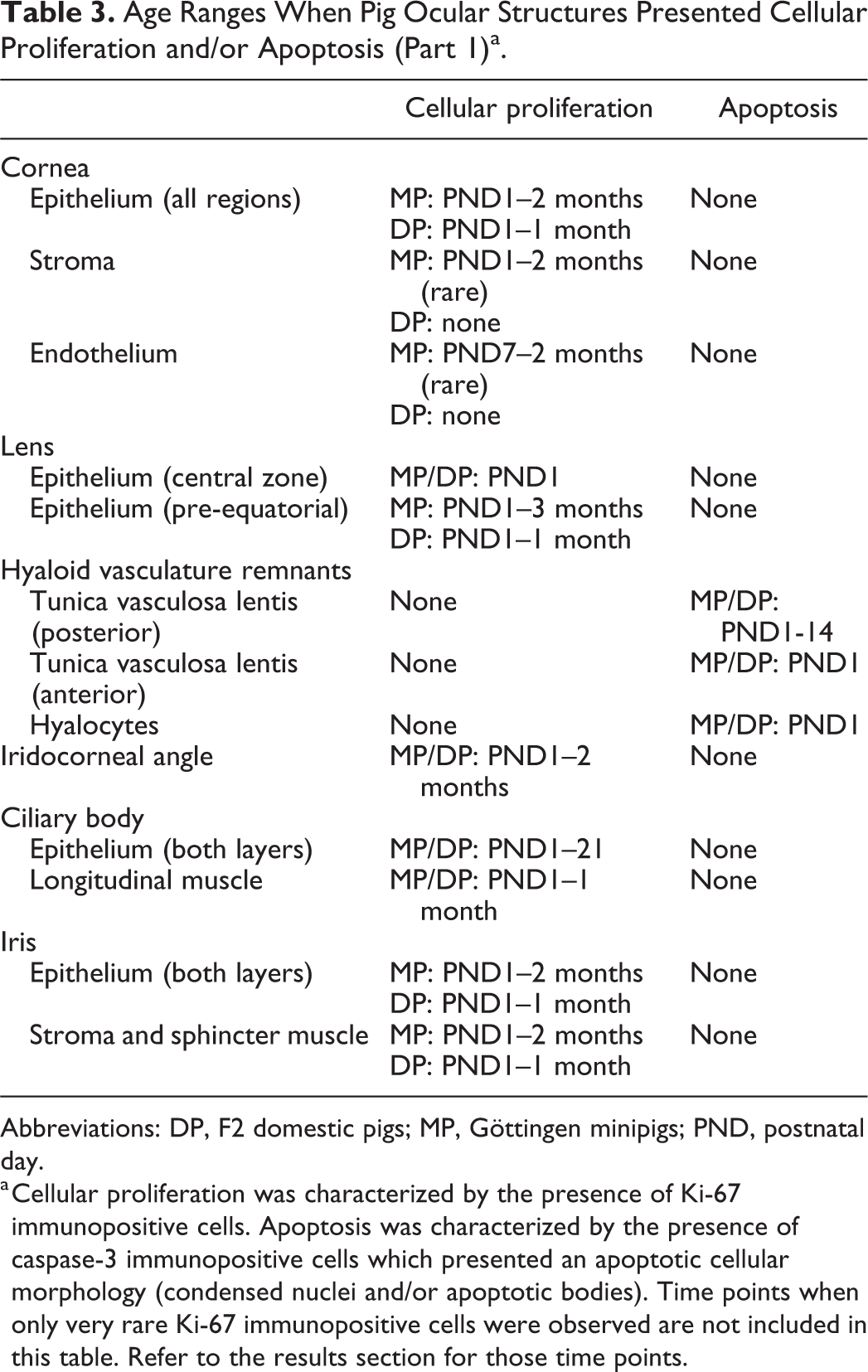
Abbreviations: DP, F2 domestic pigs; MP, Göttingen minipigs; PND, postnatal day.
a Cellular proliferation was characterized by the presence of Ki-67 immunopositive cells. Apoptosis was characterized by the presence of caspase-3 immunopositive cells which presented an apoptotic cellular morphology (condensed nuclei and/or apoptotic bodies). Time points when only very rare Ki-67 immunopositive cells were observed are not included in this table. Refer to the results section for those time points.
Age Ranges When Pig Ocular Structures Presented Cellular Proliferation and/or Apoptosis (Part 2)a.
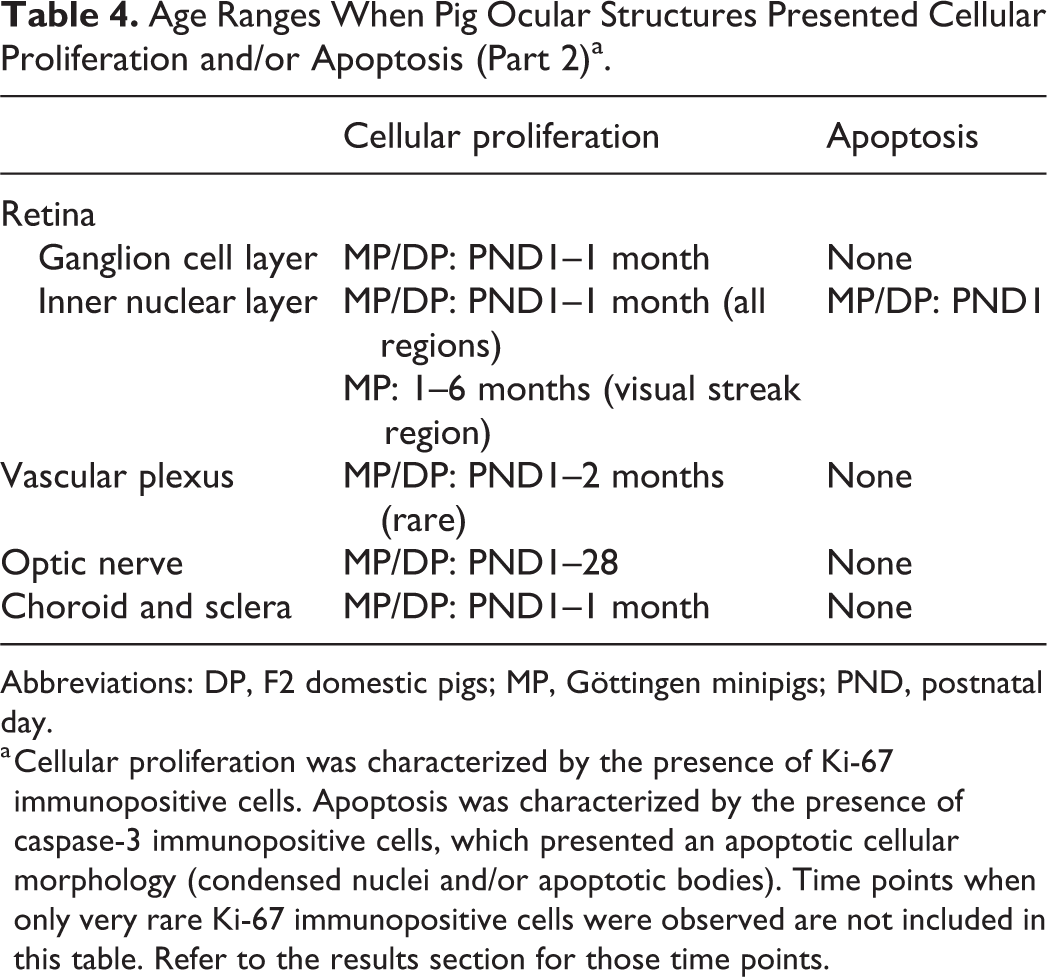
Abbreviations: DP, F2 domestic pigs; MP, Göttingen minipigs; PND, postnatal day.
a Cellular proliferation was characterized by the presence of Ki-67 immunopositive cells. Apoptosis was characterized by the presence of caspase-3 immunopositive cells, which presented an apoptotic cellular morphology (condensed nuclei and/or apoptotic bodies). Time points when only very rare Ki-67 immunopositive cells were observed are not included in this table. Refer to the results section for those time points.
Immunohistochemical Labeling of Synaptophysin, Rhodopsin, and Calbindin in Normal Eyes of Pigsa.
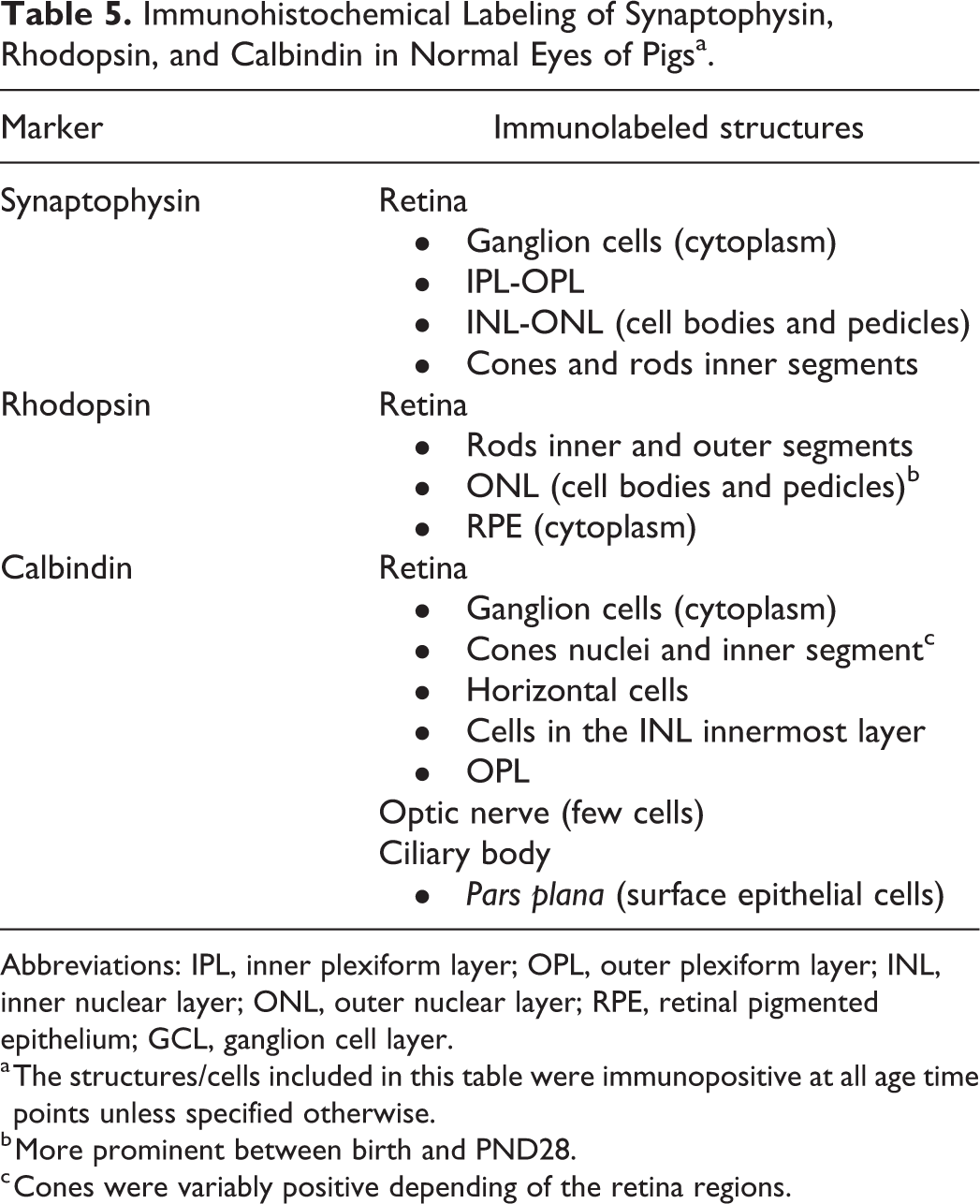
Abbreviations: IPL, inner plexiform layer; OPL, outer plexiform layer; INL, inner nuclear layer; ONL, outer nuclear layer; RPE, retinal pigmented epithelium; GCL, ganglion cell layer.
a The structures/cells included in this table were immunopositive at all age time points unless specified otherwise.
b More prominent between birth and PND28.
c Cones were variably positive depending of the retina regions.
Immunohistochemical Labeling of Glial Fibrillary Acidic Protein and Caspase-3 in Normal Eyes of Pigsa.
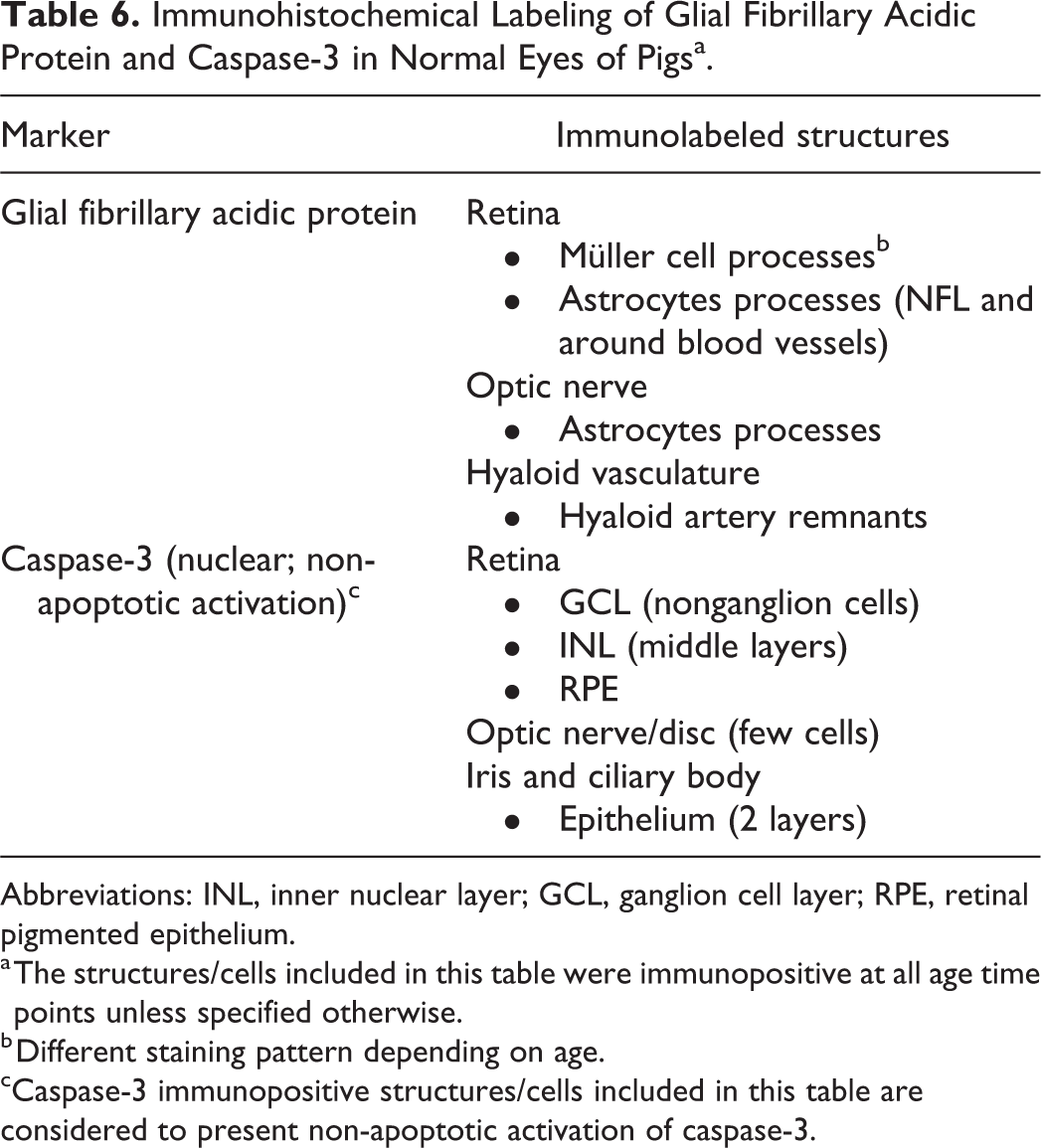
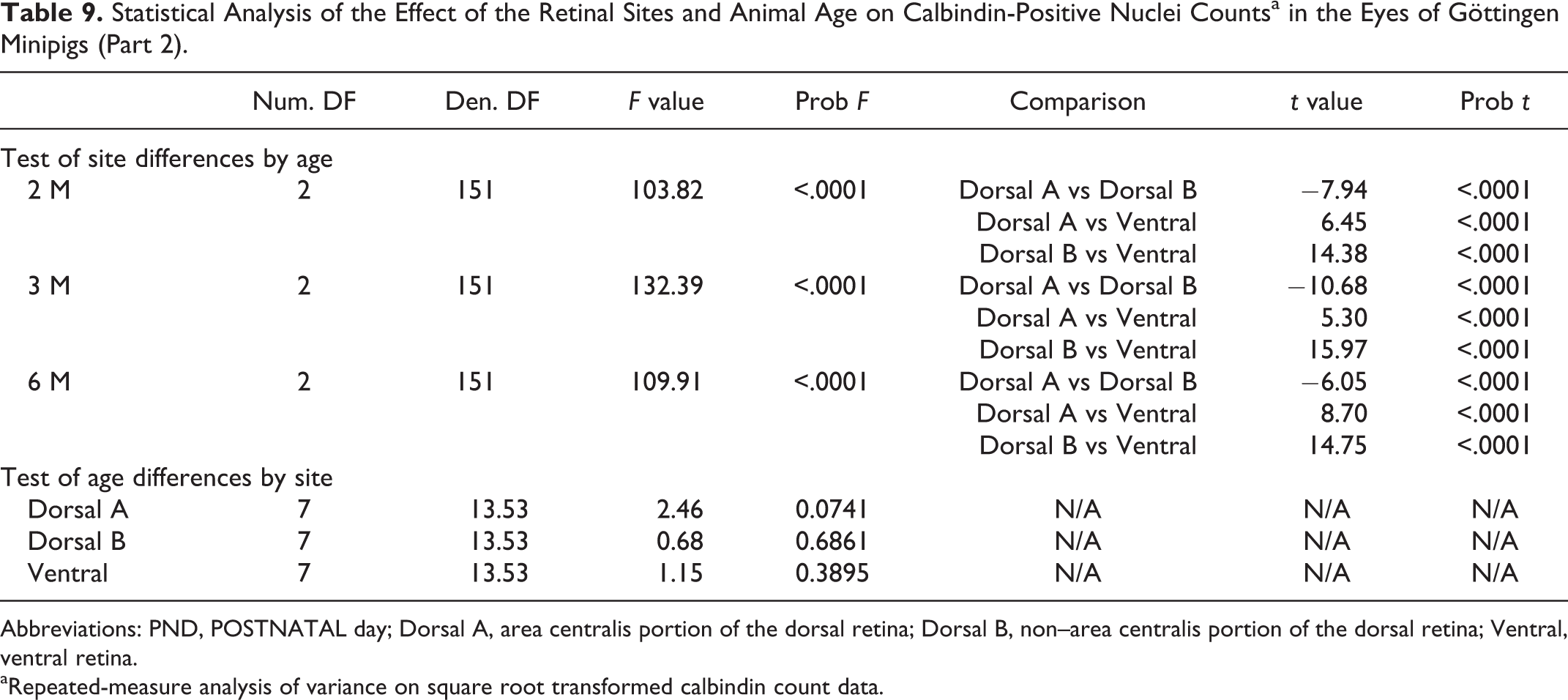
Abbreviations: INL, inner nuclear layer; GCL, ganglion cell layer; RPE, retinal pigmented epithelium.
a The structures/cells included in this table were immunopositive at all age time points unless specified otherwise.
b Different staining pattern depending on age.
cCaspase-3 immunopositive structures/cells included in this table are considered to present non-apoptotic activation of caspase-3.
Age of Histomorphological Maturity (or Complete Regression) of Various Ocular Structures in Göttingen Minipigs and F2 Domestic Pigs.
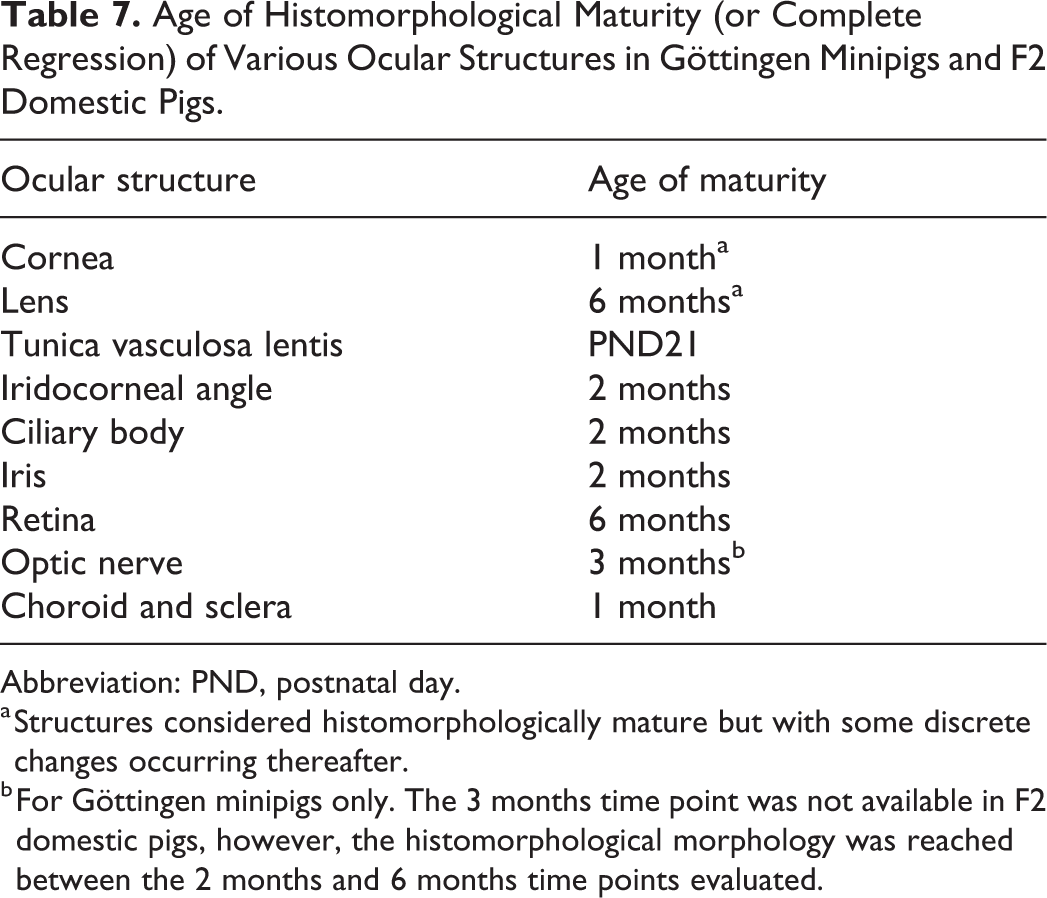
Abbreviation: PND, postnatal day.
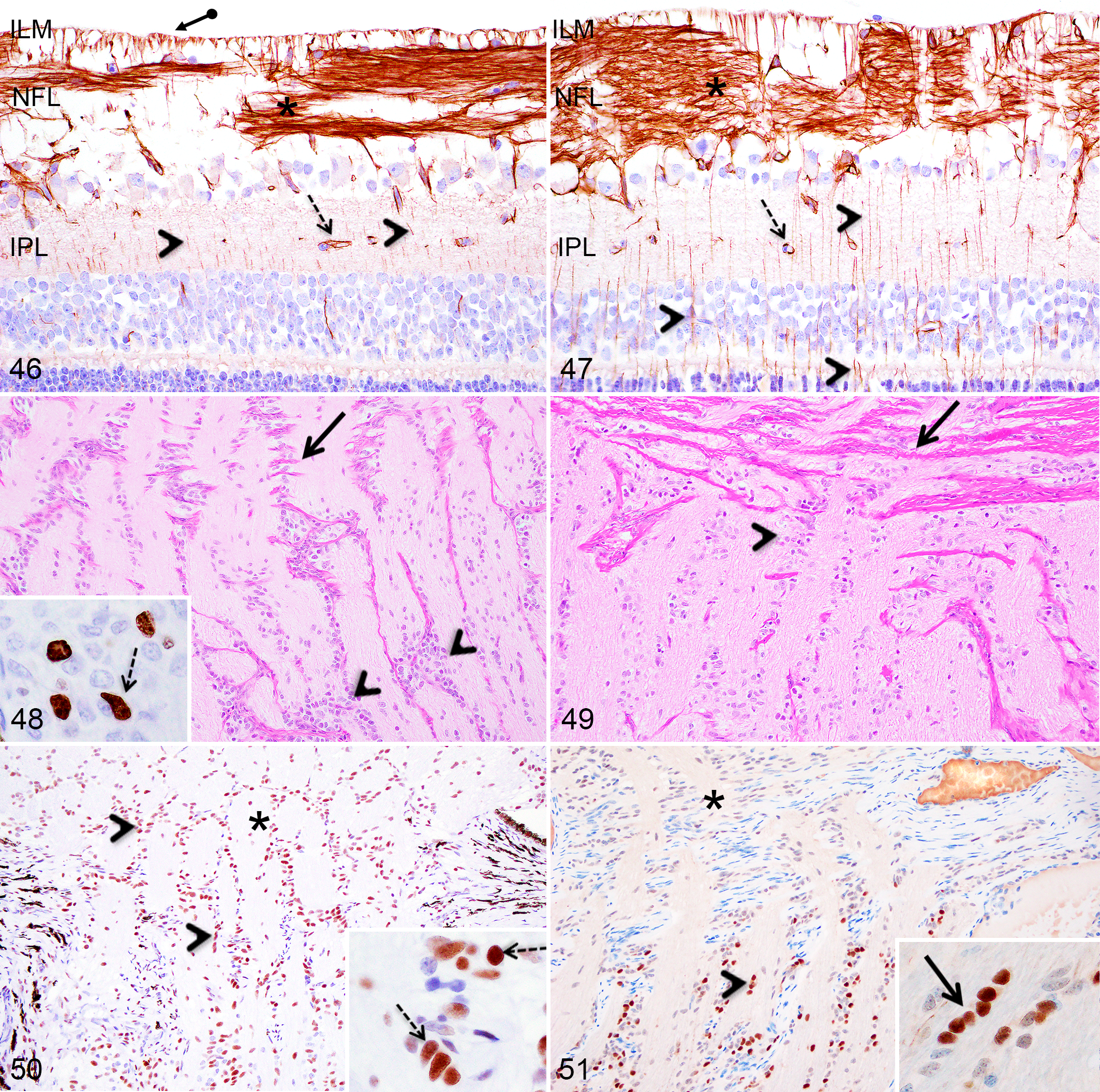
a Structures considered histomorphologically mature but with some discrete changes occurring thereafter.
b For Göttingen minipigs only. The 3 months time point was not available in F2 domestic pigs, however, the histomorphological morphology was reached between the 2 months and 6 months time points evaluated.
Cornea
Epithelium
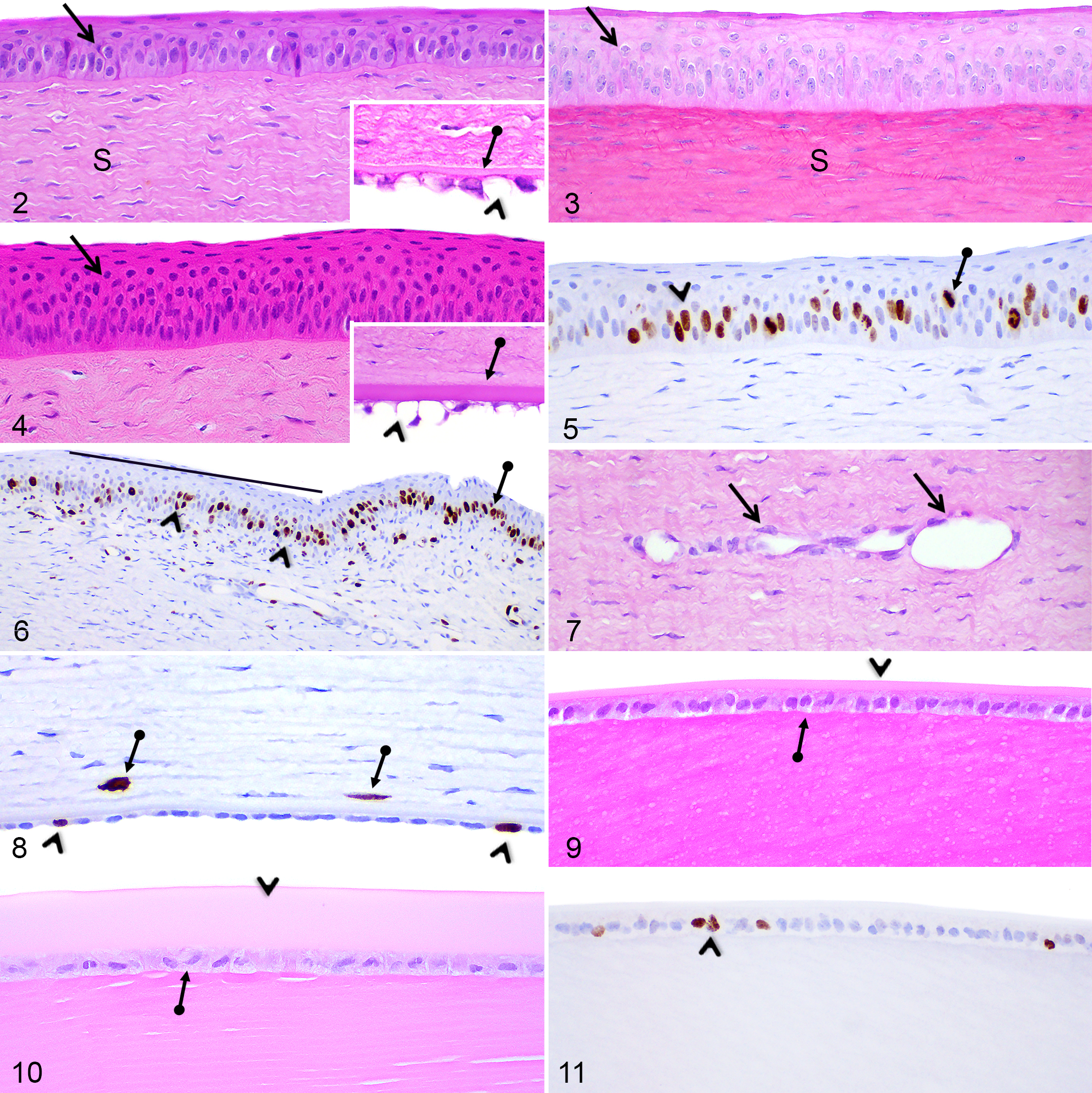
At PND1, the corneal epithelium consisted of a nonkeratinized stratified squamous epithelium composed of 3 to 5 cell layers in Göttingen minipigs (Fig. 2) and 4 to 7 cell layers in F2 domestic pigs (Fig. 3); the central and peripheral regions being more stratified (4–5 cell layers in Göttingen minipigs and 5–7 cell layers in F2 domestic pigs) than the limbal region (3–4 cell layers). For both Göttingen minipigs and F2 domestic pigs, the corneal epithelium gradually thickened and reached 8 to 10 cell layers in the central and peripheral regions and 6 to 7 cell layers in the limbal region at PND28 (Fig. 4), as in the adult retina.
Cornea and lens, Göttingen minipigs (MP) and F2 domestic pigs (DP).
From PND1 to 2 months in Göttingen minipigs and from PND1 to 1 month in F2 domestic pigs, numerous basal and suprabasal epithelial cells showed Ki-67 positive mitotic figures or cells with a diffuse nuclear labeling in the absence of mitotic figures (Figs. 5, 6). Positively labeled mitotic figures were mainly located in the basal cell layer but were also occasionally observed in the suprabasal wing cell layer. In both Göttingen minipigs and F2 domestic pigs, Ki-67 immunopositive epithelial cells were more abundant in the central region compared to the peripheral region. After 2 months, in Göttingen minipigs and 1 month in F2 Domestic pigs, Ki-67 immunolabeled corneal epithelial cells were sparse.
Stroma
The corneal stroma at PND1 in both pig breeds contained elongated stromal fibroblasts, termed keratocytes, similar to the adult cornea (Figs. 2, 3). At birth in Göttingen minipigs only, stromal capillaries extending parallel to the corneal epithelium and located at the mid-thickness of the corneal stroma were observed in the corneal central and peripheral regions (Fig. 7). From PND7 to 1 month, some capillaries remained visible in the peripheral region, but not in the central region. Central and peripheral stromal capillaries were not observed in F2 domestic pigs. Capillaries and small venules/arterioles were always observed in the limbal region in both pig breeds. From PND1 to 2 months in Göttingen minipigs only, rare Ki-67 immunopositive keratocytes were observed throughout the stroma (Fig. 8).
Endothelium
At birth in both pig breeds, endothelial cells in all corneal regions presented a rounded to oval nucleus and contained a large clear intracytoplasmic vacuole displacing the nucleus to the periphery of the cell (Fig. 2, inset). Endothelial cell nuclei gradually flattened to reach a mature elongated shape at PND21. From PND7 to 6 months, only rare endothelial cells in the central and peripheral corneal regions contained a clear intracytoplasmic vacuole, which was smaller compared to the vacuoles observed at PND1. At all time points in both breeds, limbal endothelial cells presented a peripherally located, round to oval nucleus and contained a large clear intracytoplasmic vacuole (Fig. 4, inset). In both Göttingen minipigs and F2 domestic pigs, a thin Descemet’s membrane was visible throughout the entire length of the cornea at birth (Fig. 2, inset) and gradually thickened until 6 months of age (Fig. 4, inset). From PND7 to 2 months in Göttingen minipigs, occasional Ki-67 immunopositive endothelial cells were noted in all regions of the corneal endothelium (Fig. 8). No Ki-67 immunopositive endothelial cells were noted in F2 domestic pigs.
Lens
Capsule
At birth, a thin capsule was detectable anteriorly and posteriorly in both pig breeds (Fig. 9). The capsule in both breeds gradually thickened until 6 months (Fig. 10) but remained slightly thinner at 6 months compared to the eyes of Göttingen minipigs aged more than 6 months.
Lens Subcapsular Anterior Epithelium
At birth, the lens subcapsular anterior epithelium corresponded to a simple cuboidal epithelium (Fig. 9) and a simple columnar epithelium in the pre-equatorial region. During the first month of age, the anterior epithelium and bow region were slightly more cellular (Fig. 9) compared to the that at 6 months old (Fig. 10). At PND1 in Göttingen minipigs and F2 domestic pigs, multiple epithelial cells immunopositive for Ki-67 (Fig. 11), including mitotic figures, were noticeable in the central region of the anterior subcapsular epithelium as well as in the pre-equatorial region. Very rare Ki-67-immunopositive epithelial cells showing diffuse nuclear labeling remained visible in the central epithelium until PND21 in Göttingen minipigs and until PND7 in F2 domestic pigs; while occasional Ki-67-immunopositive epithelial cells in the pre-equatorial region were visible until 3 months of age in Göttingen minipigs and until 1 month in F2 domestic pigs.
Lens Fibers
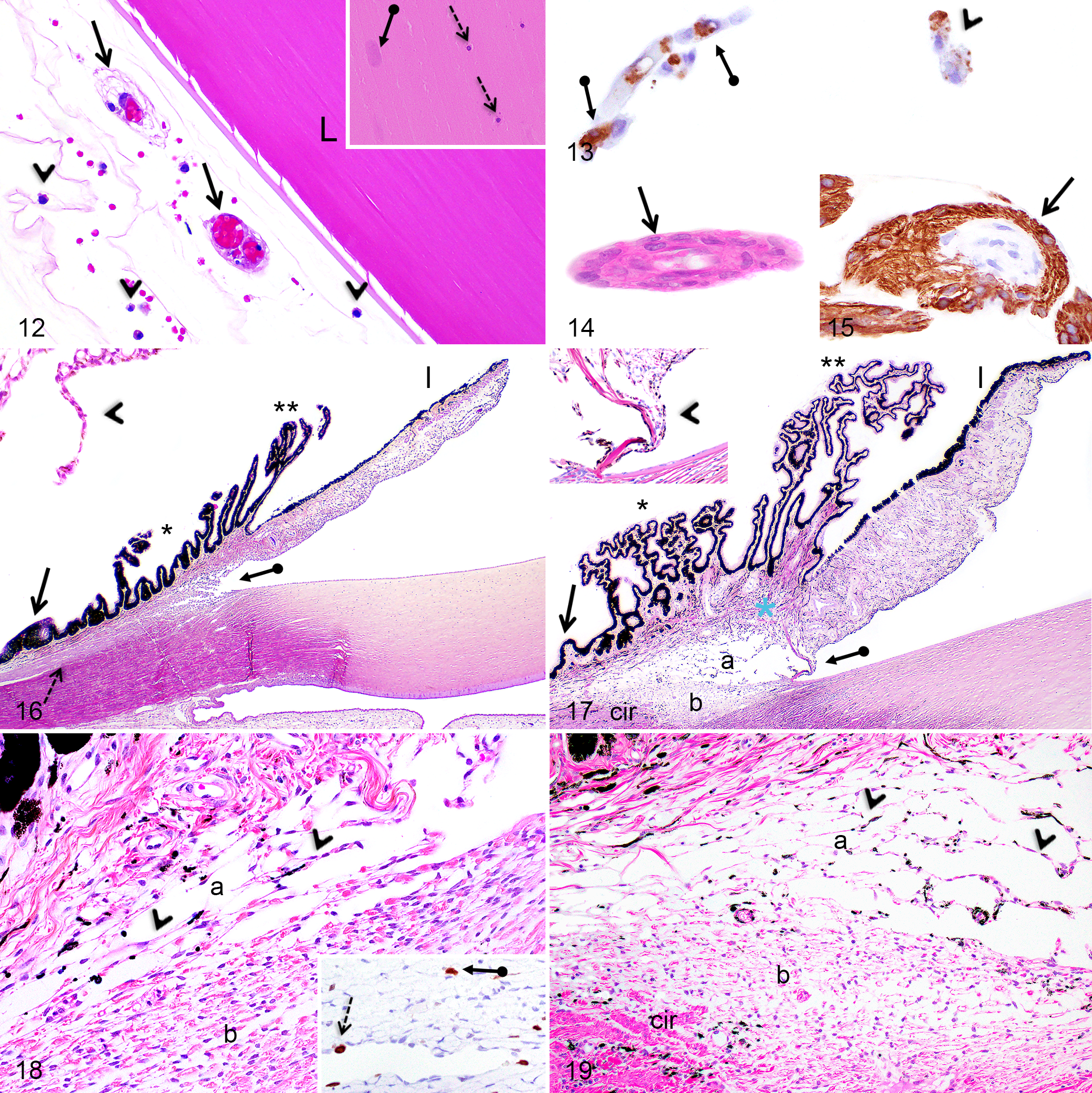
As the secondary fibers differentiated and internalized, their nuclei gradually disintegrated to form the organelle-free zone (OFZ), leaving behind small basophilic nuclear fragments toward the center of the lens (Fig. 12, inset). These nuclear fragments were easily noticeable at the edge of the OFZ from PND1 to 2 months in F2 domestic pigs and from PND1 to 3 months in Göttingen minipigs and were absent or rarely noted in some animals at 6 months. During the first 6 months of age, secondary lens fibers gradually accumulated and the OFZ widened. Disintegrating secondary lens fiber nuclei were not immunopositive for caspase-3.
Lens, hyaloid vasculature, anterior uvea and iridocorneal angle; Göttingen minipigs (MP) and F2 domestic pigs (DP).
Hyaloid Vasculature System
At birth in Göttingen minipigs and F2 domestic pigs, remnants of the tunica vasculosa lentis were observed alongside the anterior, lateral, and posterior aspects of the lens (Fig. 12) and they regressed completely first at the anterior pole of the lens at PND7 and lastly at the posterior pole of the lens at PND21 in both pig breeds. Furthermore, from PND1 to PND14, macrophages (termed hyalocytes) occupying the vitreous and the anterior and posterior chambers were commonly observed adjacent to the regressing hyaloid capillaries (Fig. 12). In both Göttingen minipigs and in F2 domestic pigs, endothelial cells composing the regressing tunica vasculosa lentis capillaries showed an apoptotic phenotype and were immunopositive for caspase-3 (Fig. 13) at the anterior pole of the lens at PND1 and at the lateral and posterior aspects of the lens from PND1 to PND14. At PND1, scattered hyalocytes were positive for caspase-3 or contained engulfed caspase-3-immunopositive apoptotic bodies (Fig. 13). Remnants of the hyaloid artery (Fig. 14) were noted in the posterior vitreous of some Göttingen minipigs from PND1 to PND14, but not in F2 domestic pigs, and these vestigial blood vessels were surrounded by GFAP-immunopositive processes (Fig. 15).
Iridocorneal Angle
The iridocorneal angle in pigs is divided into the ciliary cleft and the corneoscleral meshwork (Figs. 16, 17). The former is an extension of the anterior chamber and contains the pectinate ligaments anteriorly, which join the base of the iris to the limbal cornea, and multiple thin uveal cords posteriorly forming a loose trabecular meshwork. The corneoscleral meshwork is located in an internal wedge-shaped scleral sulcus and is composed of tightly compacted trabeculae. Melanin pigments are normally present in the pectinate ligament, uveal cords, and corneoscleral meshwork. The angular aqueous plexus (the analogue of the Schlemm canal in humans) is located externally to the corneoscleral meshwork and includes multiple endothelium-lined canals. At birth, in Göttingen minipigs and F2 domestic pigs, all components of the iridocorneal angle were identifiable but presented subtle signs of immaturity, except for the angular aqueous plexus, which appeared mature at birth.
Ciliary Cleft
At birth in both pig breeds, the ciliary cleft was narrow and contained few uveal cords lined by plump mesenchymal cells (Figs. 16, 18). Between birth and 2 months, the ciliary cleft progressively deepened and widened, the uveal cords became more numerous and composed of flattened mesenchymal cells, and the intertrabecular spaces became more prominent, as in adults (Figs. 17, 19). The pectinate ligament was visible at birth as a thin fibrovascular cord (Fig. 16, inset) and gradually thickened by the addition of collagen fibers until 2 months, when it became similar to adults (Fig. 17, inset).
Corneoscleral Meshwork
In Göttingen minipigs and F2 domestic pigs, the corneoscleral meshwork at PND1 was compact, more densely cellular, and the intertrabecular spaces were narrow (Figs. 16, 18). Between birth and 2 months of age, the intertrabecular spaces widened and the cellularity of the meshwork decreased to reach a similar histomorphology as in the adult (Figs. 17, 19).
From PND1 to 2 months in both pig breeds, occasional scattered endothelial cells lining the angular aqueous plexus canals and mesenchymal cells in the ciliary cleft’s uveal cords and in the corneoscleral meshwork showed multifocal Ki-67 nuclear labeling (Fig. 18, inset). At all ages, the pectinate ligament did not contain Ki-67 immunopositive cells.
Ciliary Body
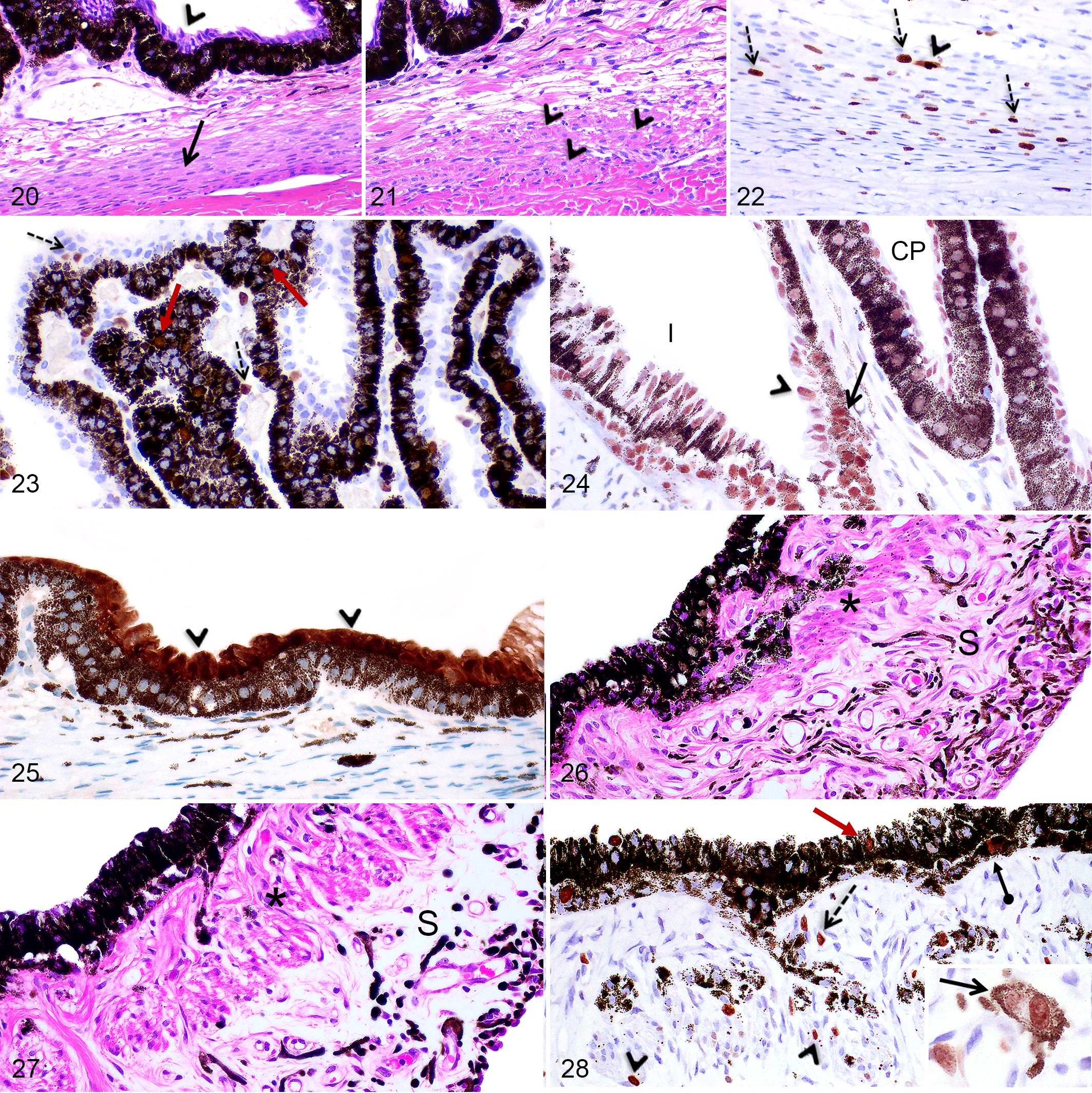
In pigs, the ciliary body is divided by the ciliary cleft into 2 portions. The inner portion is composed anteriorly of the ciliary processes, which are radially oriented, highly vascularized structures lined by a double-layered epithelium (termed pars ciliaris retinae), and posteriorly by the pars plana (the flat portion of the ciliary body; Figs. 16, 17). The ciliary processes were longer and thinner anteriorly and shorter and wider posteriorly toward the pars plana (Figs. 16, 17). The outer portion of the ciliary body consisted of 2 perpendicularly oriented groups of smooth muscles, located between the ciliary cleft/pars plana and the sclera (Figs. 16, 17). Longitudinally (or meridionally) oriented smooth muscle bundles extended parallel to the pars plana approximately from the ora serrata to the most posterior ciliary processes (Figs. 16, 20). Anterior to the longitudinal muscles, fewer and scattered circularly (or circumferentially) positioned smooth muscle bundles composed of one to few fibers were present adjacent or within the corneoscleral meshwork (Figs. 19, 21).
Ciliary body and iris, Göttingen minipigs (MP) and F2 domestic pigs (DP).
At birth in both pig breeds, the inner portion of the ciliary body was composed of short, thin, and poorly ramified ciliary processes presenting a well-differentiated double-layered epithelium (Fig. 20) and a scarce fibrous stroma. (Fig. 16). The ciliary processes became histomorphologically similar to adults after PND28. In both pig breeds, the longitudinal (meridional) portion of the ciliary muscles was well defined at birth but was narrow and more densely cellular (Figs. 16, 20). As this portion of the muscle thickened, the cellularity gradually decreased and reached a cellularity similar to the mature eye at 2 months of age. At birth, the circular (or circumferential) portion of the ciliary muscles was not identifiable while only individual smooth muscle fibers were noted at PND7 (Fig. 21). At 2 months, circular smooth muscle bundles were composed of 4 to 5 muscle fibers as in adults (Fig. 19).
In Göttingen minipigs and F2 domestic pigs, between PND1 and 1 month, abundant smooth muscle cells in the longitudinal muscles, but not in the circumferential muscle, were immunopositive for Ki-67 and included mitotic figures as well as diffuse nuclear labeling (Fig. 22). From PND1 to PND21 in both pig breeds, multiple epithelial cells of both layers of the ciliary epithelium and the pars plana as well as mesenchymal cells in the ciliary body stroma showed diffuse nuclear labeling for Ki-67 and labeling of mitotic figures (Fig. 23); thereafter, only rare Ki-67 immunopositive cells were observed. At all ages, nuclei of both layers of the ciliary body epithelium and the pars plana presented diffuse nuclear labeling for caspase-3 (Fig. 24) and solely the nonpigmented surface epithelial cells of the pars plana showed intense cytoplasmic and nuclear labeling for calbindin (Fig. 25).
Iris
At birth, the iris was thin and presented a poorly abundant and densely cellular stroma containing numerous mesenchymal cells (Figs. 16, 26). The sphincter and dilator muscles were identifiable at PND1, but the sphincter muscle was composed of fewer smooth muscle fiber bundles (Fig. 26) compared to the mature iris. The cellularity of the anterior stroma gradually decreased while the sphincter muscle thickened by the addition of new smooth muscle fibers until 2 months of age (Fig. 27), when the iris became histomorphologically similar to that of the adult iris.
From PND1 to 2 months in Göttingen minipigs and from PND1 to 1 month in F2 domestic pigs, Ki-67 immunopositive nuclei were observed in both layers of the posterior epithelium, in stromal mesenchymal cells and melanocytes, and in sphincter muscle cells (Fig. 28). Thereafter, only rare Ki-67 immunopositive cells were scattered in the anterior stroma. At all ages, the nuclei of the posterior epithelium, including the pigmented epithelial cells and the myoepithelial cells, showed diffuse nuclear labeling for caspase-3 (Fig. 24).
Retina
At birth in Göttingen minipigs and F2 domestic pigs, all layers of the retina were distinguishable in all retinal regions but were not histomorphologically mature. Postnatal developmental changes were noted until 6 months of age in both Göttingen minipigs and F2 domestic pigs, with no significant differences noted between both breeds. If not specified otherwise, the histological and immunohistochemical descriptions presented below are representative of all regions of the retina (central to peripheral; area centralis and non–area centralis regions). It is however understood that all layers of the retina become thinner toward the ora serrata.
Area Centralis Versus Non–Area Centralis Regions
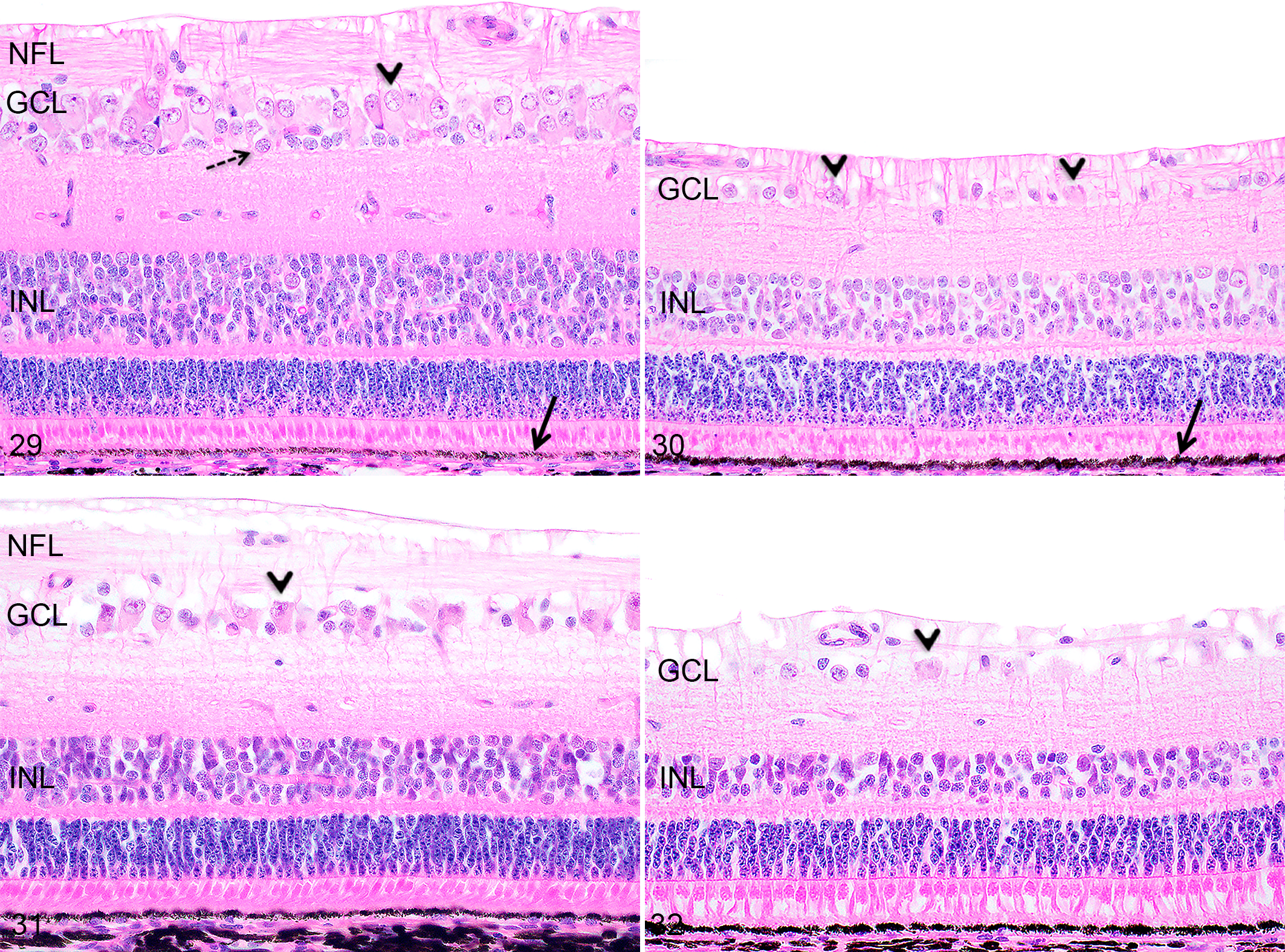
The area centralis region in Göttingen minipigs and F2 domestic pigs was easily identifiable at any age as a dorsally located (dorsal and adjacent to the optic disc) area characterized by a significantly higher ganglion cell density compared to the non–area centralis regions (the entire ventral retina and the retinal region located dorsal to the area centralis; Fig. 29–32). Regardless of the age and the size of the eye, the area centralis corresponded to approximately 1/5 of the dorsal portion of the retina. Other histomorphological differences noted between the area centralis and non–area centralis regions are described in detail below.
Retina, F2 domestic pigs (DP).
Nerve Fiber Layer (NFL) and Ganglion Cell Layer (GCL)
Histologically, the nerve fiber layer of the retina at birth did not differ significantly from the adult retina. At all time points, the NFL was thicker in the area centralis region compared to the non–area centralis regions, due to the higher density of ganglion cells in this region (Figs. 29–32). At birth, in Göttingen minipigs and F2 domestic pigs, both the area centralis and the non–area centralis regions were more densely cellular compared to the adult retina (Figs. 29–32); the area centralis at birth was composed of densely packed ganglion cells forming 2 to 3 cell layers (Fig. 29), while the non–area centralis regions contained one layer of ganglion cells arranged closely together (Fig. 30). The ganglion cell density in all retinal regions progressively decreased as the ganglion cells spread out until 6 months of age; at this age, the area centralis was composed of 1 to rarely 2 cell layers of ganglion cells (Fig. 31) and the non–area centralis regions corresponded to 1 discontinuous layer of sparse ganglion cells (Fig. 32). At all time points, ganglion cells were admixed with smaller non-ganglion cells (compatible with amacrine cells and astrocytes), which were more abundant at birth and PND7 (Fig. 29).
In Göttingen minipigs and domestic pigs, Ki-67-immunopositive non-ganglion cells and ganglion cells presenting a diffuse nuclear and/or a nucleolar labeling pattern were observed in the GCL from PND1 to 1 month (Fig. 33). At all time points, in both pig breeds, non-ganglion cells showed a diffuse nuclear labeling for caspase-3 without presenting an apoptotic phenotype (Fig. 34), and ganglion cells presented a diffuse finely granular cytoplasmic labeling pattern for calbindin (Fig. 35), while labeling for synaptophysin was cytoplasmic, with focal perinuclear marking (Fig. 36).
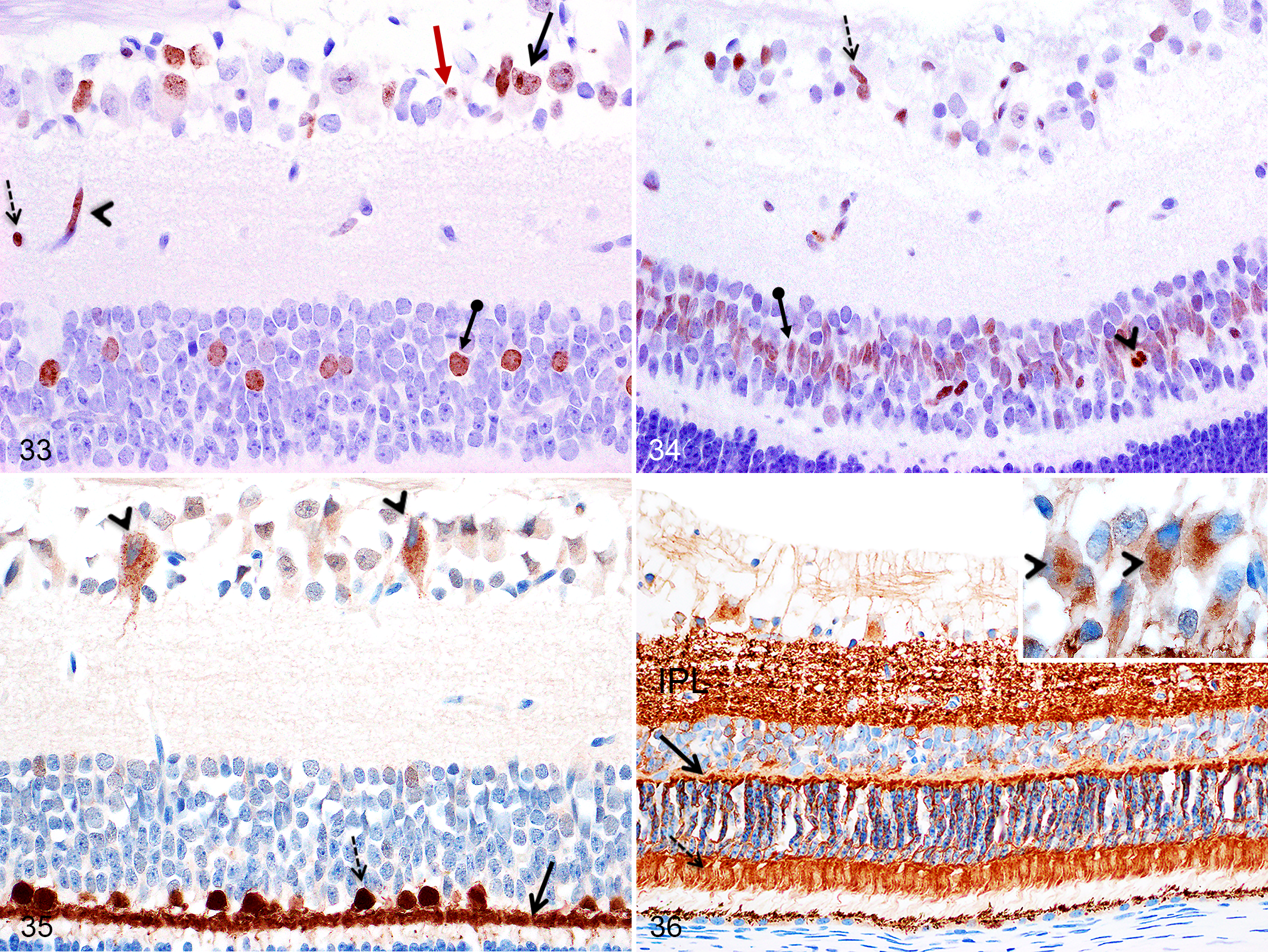
Retina, Göttingen minipigs (MP) and F2 domestic pigs (DP).
Inner Plexiform Layer (IPL)
At birth, the inner plexiform layer was histologically similar to the adult retina and the immunolabeling pattern for synaptophysin was equivalent throughout all time points (Fig. 36). In Göttingen minipigs from PND1 to 3 months and in F2 domestic pigs from PND1 to 1 month, rare cells scattered in the IPL showed diffuse nuclear Ki-67 immunolabeling (Fig. 33).
Inner Nuclear Layer (INL)
At all ages, the INL contained more cell layers in the area centralis region compared to the non–area centralis regions (Figs. 29–32). At birth in Göttingen minipigs and F2 domestic pigs, the INL was thicker than in the mature retina and was composed of approximately 8 to 10 cell layers in the area centralis region (Fig. 29), and 4 to 5 cell layers just dorsally to the area centralis and ventrally to the optic nerve (in the central region; Fig. 30). The number of cell layers in the INL decreased from PND1 to 6 months of age in all regions. At 6 months in the area centralis region, the INL was approximately 6 cell layers (Fig. 31) while the INL just dorsally to the area centralis and just ventrally to the optic disc (central region) was approximately 3 to 4 cells thick (Fig. 32).
From PND1 to PND28 in Göttingen minipigs and F2 domestic pigs, nuclei located in the middle layers of the INL showed diffuse nuclear immunolabeling for Ki-67 (Fig. 33); these immunopositive cells were numerous in the area centralis region while they were infrequent in non–area centralis regions of the retina. Thereafter, these Ki-67 immunopositive cells were not observed in the F2 domestic pigs, but in Göttingen minipigs from 2 months to 6 months, some Ki-67-immunopositive nuclei remained visible in the middle layers of the INL solely in the area centralis region. Only at birth (PND1), in Göttingen minipigs and F2 domestic pigs, rare apoptotic bodies strongly positive for caspase-3 were scattered in the INL (Fig. 34). At all ages, in both pig breeds, non-apoptotic cells in the middle layers of the INL and less commonly in the innermost cell layer showed diffuse nuclear labeling for caspase-3 (Fig. 34). In addition, at all ages, in both pig breeds, large nuclei located in the outermost layer of the INL (compatible with horizontal cells) as well as scattered smaller round nuclei at the innermost aspect of the INL were immunopositive for calbindin (Fig. 35). At any age in both pig breeds, immunolabeling for synaptophysin was observed in the cell body and pedicles of some cells present in all layers of the INL.
Outer Plexiform Layer (OPL)
At birth, the OPL was histologically similar to the adult retina. At all ages, the OPL was strongly immunopositive for calbindin (Fig. 35) and synaptophysin (Fig. 36) in both breeds.
Outer Nuclear Layer (ONL) and Photoreceptor Cell Layer
No significant differences in the ONL were noted between the area centralis and non–area centralis regions at all ages. At birth, the ONL (central region) was composed of 7 to 8 layers (Figs. 29, 30) and its thickness slightly decreased to 5 to 6 layers at 2 months of age (Figs. 31, 32). Of note, at any time point, the outermost layer was composed of slightly larger oval nuclei with a diffuse euchromatin, compatible with cone nuclei (Fig. 37).
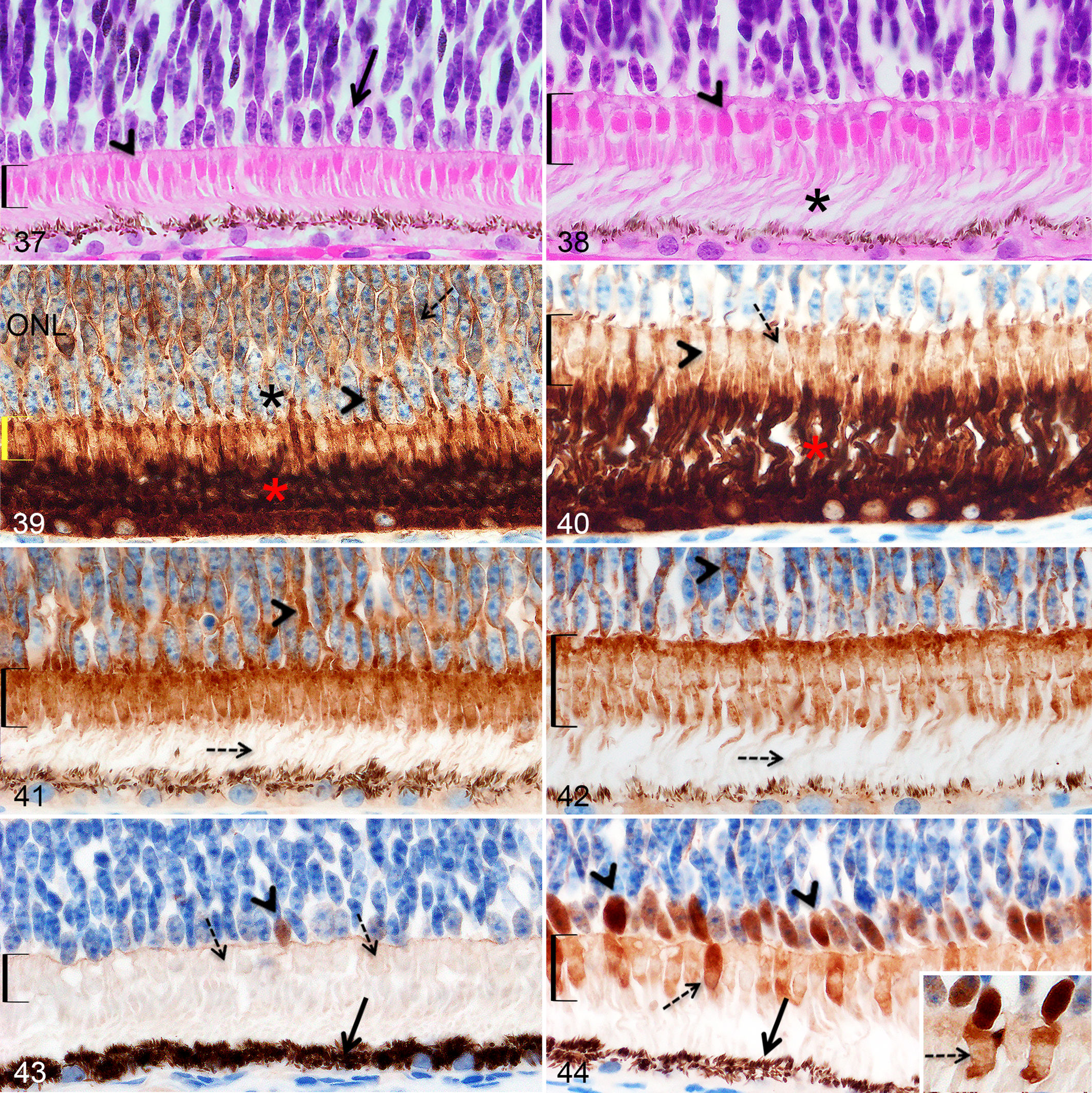
Retinal outer nuclear layer and photoreceptor layer, Göttingen minipigs (MP) and F2 domestic pigs (DP). All figures are at the same magnification. Brackets show the photoreceptor inner segments.
At birth, the photoreceptors were immature, which was illustrated on HE-stained sections by the striking differences in size and shape of the cone inner segments, the global shortness of the photoreceptors, and the poor delineation between the inner and outer segments compared to the photoceptors at 6 months, when the photoreceptors adopted a mature histomorphological aspect (Figs. 37, 38). The immunohistochemical labeling against rhodopsin, synaptophysin, and calbindin presented below allowed a more detailed description of the postnatal development of cones and rods.
The histological and immunohistochemical qualitative evaluations did not reveal perceptible histomorphological or density differences in the photoreceptor layer between the area centralis and non–area centralis regions at any time point (except for calbindin presented below).
Rhodopsin Immunolabeling
At any age, the rod outer segments stained intensely for rhodopsin while the inner segments were more mildly immunopositive (Figs. 39, 40). At birth in Göttingen minipigs and domestic pigs, rhodopsin immunohistochemistry highlighted the shortness of the rod outer segments (Fig. 39), which elongated until they became similar to the mature retina at PND21 in both pig breeds (Fig. 40). Rhodopsin labeling also highlighted the inner segments of rods, which can be challenging to identify with regular HE staining. In addition, at birth in both breeds, prominent immunolabeling for rhodopsin was present in the cell body and pedicles of rods in the ONL (Fig. 39); this labeling pattern decreased progressively until PND28, after which only very rare, scattered rods showed immunopositive cell bodies. In both pig breeds, displaced, positively stained, nuclei (with stained cell bodies) were present in the posterior half of the INL from birth to 2 months.
Synaptophysin Immunolabeling
At any ages in both pig breeds, synaptophysin immunolabeling was observed in the cell bodies and pedicles of both photoreceptors (in the ONL) and in the inner segments of both cones and rods, while the outer segments were not labeled (Figs. 41, 42). At birth in Göttingen minipigs and domestic pigs, synaptophysin immunohistochemistry further highlighted the short immature inner segments of both cones and rods: the inner segments of cones were narrow and oval (Fig. 37 for HE, Fig. 41 for synaptophysin labeling) instead of being wide and triangular to square as in the 6-month pigs (Fig. 38 for HE, Fig. 42 for synaptophysin labeling).
Calbindin Immunolabeling
Calbindin immunolabeling was observed solely in some cone nuclei in the outermost layer of the ONL (Figs. 43, 44) and variably in the inner segment of cones (Fig. 44). When cones nuclei were immunopositive, the inner segment immunolabeling varied from absent to intensely positive with no apparent predisposition for any region of the retina. When present, the inner segment labeling was characterized by a circular clear area which was present from birth and increased in size as the inner segment developed (Fig. 44).
The counts for calbindin-immunopositive nuclei located in the outermost nuclei row of the ONL were significantly different between each of the 3 evaluated regions (the ventral retina, the area centralis, and the non–area centralis dorsal retina) for all ages, except at PND1 when the ventral retina was not significantly different from the area centralis. The non–area centralis dorsal retina contained more calbindin-positive nuclei compared to the area centralis and to the ventral retina regions, and the number of calbindin positive nuclei in the area centralis region was higher than in the ventral region except at PND1 (Fig. 45, Tables 8, 9). No significant overall Age effect was observed for each site (P > .05). The P values obtained for the overall Site effect at each age (F test), the overall Age effect at each site (F test) and the between site comparisons at each age (t test) are included in Tables 8 and 9. The individual animal raw data for the calbindin-positive nuclei count in the retina are available in Supplemental Table S1.
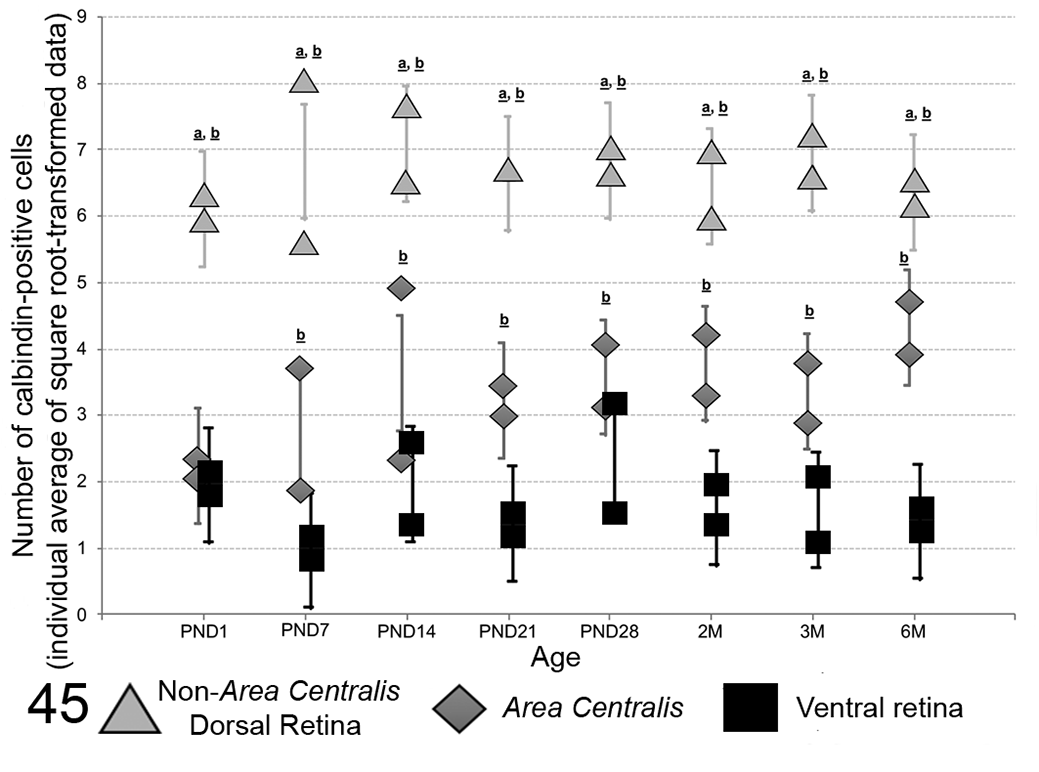
Distribution by age with 95% confidence interval (CI) of calbindin-immunopositive cone photoreceptor nuclei counts (individual average of replicates using square root transformed count data) for each retinal site. For all ages, the non–area centralis dorsal retina contained more calbindin-positive nuclei compared to the area centralis and to the ventral retina regions. Except at PND1, counts for calbindin-positive nuclei in the area centralis region were higher than in the ventral region (significantly different from area centralis:
Statistical Analysis of the Effect of the Retinal Sites and Animal Age on Calbindin-Positive Nuclei Countsa in the Eyes of Göttingen Minipigs (Part 1).
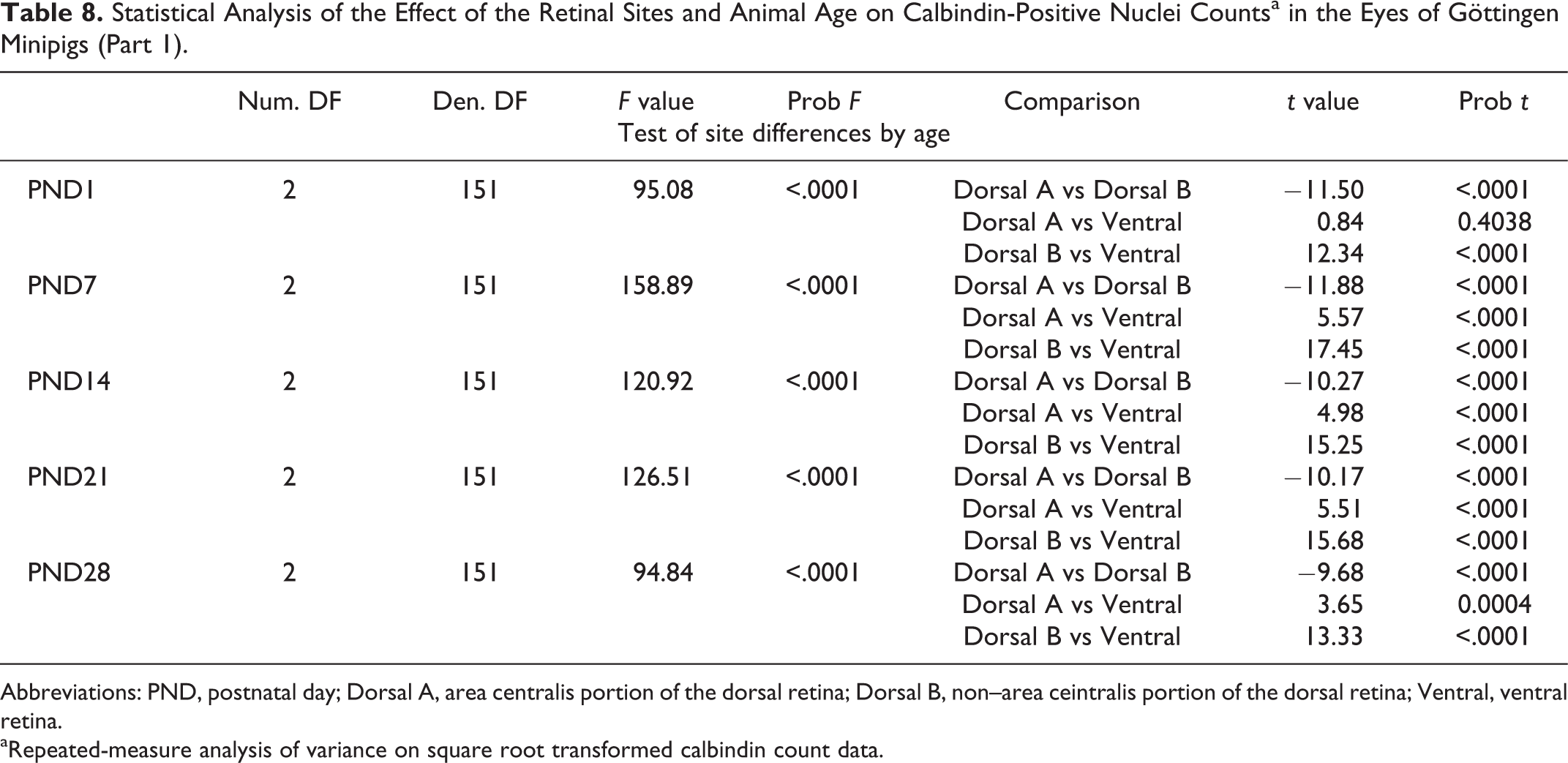
Abbreviations: PND, postnatal day; Dorsal A, area centralis portion of the dorsal retina; Dorsal B, non–area ceintralis portion of the dorsal retina; Ventral, ventral retina.
aRepeated-measure analysis of variance on square root transformed calbindin count data.
Statistical Analysis of the Effect of the Retinal Sites and Animal Age on Calbindin-Positive Nuclei Countsa in the Eyes of Göttingen Minipigs (Part 2).
Abbreviations: PND, POSTNATAL day; Dorsal A, area centralis portion of the dorsal retina; Dorsal B, non–area centralis portion of the dorsal retina; Ventral, ventral retina.
aRepeated-measure analysis of variance on square root transformed calbindin count data.
As the calbindin labeling intensity varied significantly between slides in F2 domestic pigs, a qualitative evaluation was performed. In properly stained sections, the same labeling pattern (same variations between all 3 regions) was observed in the eyes of domestic pigs.
Retinal Vascular Plexus
At birth in Göttingen minipigs and F2 domestic pigs, the superficial vascular plexus extending from the optic disc to the ora serrata within the nerve fiber layer was composed of small to medium-sized arterioles which were morphologically similar to the adult retina. Capillaries forming the inner and deep vascular plexus within the IPL, INL, and OPL were also visible at birth. Rare endothelial cells presenting nuclear immunolabeling for Ki-67 (Fig. 33) were scattered in both breeds from PND1 to 2 months.
Retinal Glial Cells (Müller Cells and Astrocytes)
Retinal glial cells, including Müller cells and astrocytes, were highlighted with GFAP. At birth, the entire inner limiting membrane (ILM) was composed of GFAP immunopositive processes that were perpendicularly arranged and presented a broad base (corresponding to Müller cell processes; Fig. 46). Müller cell end feet were wider in the peripheral retina compared to the central retina. At birth in both pig breeds, GFAP labeling of very fine discontinuous linear processes (compatible with Müller cell processes and cell bodies), traveling perpendicularly to the retinal cell layers, extended from the ILM to the IPL in the central retina (Fig. 46) and from the ILM to the middle of the INL in the retinal peripheral region. These GFAP positive processes became more prominent and elongated to reach approximatively the OPL in the central retina (as in adults) at 3 months in Göttingen minipigs (Fig. 47). At 1 month in both breeds, these processes reached the outer limiting membrane (OLM) in the peripheral retina. At all ages, Müller processes were always larger and crossed more cell layers in the peripheral retina compared to the central retina.
Retina and optic nerve, Göttingen minipigs (MP) and F2 domestic pigs (DP).
At birth, the NFL contained numerous horizontally oriented GFAP positive processes (astrocyte processes; Fig. 46), which were more abundant centrally than peripherally, as in adults. This labeling pattern remained similar throughout the different time points (Figs. 46, 47). At all ages, blood vessels of the retinal vascular plexus in the NFL, GCL, IPL and INL were surrounded by GFAP-positive astrocyte processes (Figs. 46, 47).
Retinal Pigmented Epithelium (RPE)
Interestingly, at all ages, the retinal pigmented epithelial cells in the ventral region of the retina and in the anterior 1/3 (closest to ora serrata) of the dorsal retina were significantly more pigmented compared to the remaining 2/3 of the dorsal retina (Figs. 29 and 30 and Figs. 43 and 44). At all ages, the nuclei of the epithelium showed diffuse nuclear labeling for caspase-3 and the cytoplasm was positive for rhodopsin.
Optic Nerve
At PND1, the optic nerve was composed of myelinated nerve fibers and was densely cellular, and the collagenous laminae of the lamina cribosa were poorly developed (Fig. 48). The lamina cribosa appeared to be of comparable thickness as in adults at PND28 in both breeds and the cellularity of the optic nerve became similar to adults at 3 months of age in the Göttingen minipigs (Fig. 49). At 2 months of age in domestic pigs, the optic nerve was still more densely cellular compared to the 6-month-old animals. At all ages in both breeds, the optic nerve and optic disk were composed of numerous, strongly GFAP positive astrocyte processes. In both pig breeds, Ki-67 positive cells with diffuse labeling of nuclei nucleoli and occasionally mitotic figures were numerous between PND1 and PND28, while they became sparser thereafter with no discernable mitotic figures (Fig. 48). In both breeds, some nuclei present in the optic nerve showed diffuse nuclear immunolabeling for caspase-3 (without presenting an apoptotic phenotype; Fig. 50) and/or calbindin (Fig. 51) at all ages. Caspase-3 immunopositive nuclei were also present in the optic disc, while no nuclei in this location were immunopositive for calbindin.
Incidental Findings in Retina and Optic Nerve
Different background or incidental changes were noted in juvenile Göttingen minipigs and F2 domestic pigs’ retina or optic nerve throughout the first 6 months of age.
In 2 Göttingen minipigs, near the optic disc, the optic nerve contained a poorly defined non-encapsulated nodule composed of GFAP-positive, disorganised fibrillar structures, reassembling nerve fibers, admixed with a heterogeneous population of randomly distributed cells (Fig. 52). Some cells presenting smaller nuclei showed intense immunolabeling for rhodopsin in the cell body and in linear structures attached to the cell body, suggestive of inner/outer segments of rod photoreceptors (Fig. 53). In addition, numerous cells with large round nuclei containing 1 to 2 prominent nucleoli presented intracytoplasmic immunolabeling for synaptophysin (Fig. 54), while other cells showed intense nuclear labeling for calbindin, similar to retinal horizontal cells (Fig. 55). Calbindin immunohistochemistry also revealed the presence of a positive fibrillar/granular material in the nodule’s stroma (Fig. 55).
Incidental findings, retina and optic nerve, Göttingen minipigs (MP) and F2 domestic pigs (DP).
Some level of cupping of the optic disc was observed at all ages (Fig. 56) in both pig breeds and without retinal or other pathological changes. In addition, retinal rosettes located in or adjacent to the optic disc were present randomly at various time points between birth and 6 months in both pig breeds (Fig. 57). Finally, scattered photoreceptor nuclei were displaced within the photoreceptors layer at various ages between birth and 6 months in both pig breeds.
Choroid and Sclera
Minimal changes occurred in the choroid after birth except for slight thickening and, between birth and PND28, the choroid was more densely cellular with plump nuclei compared to older time points. From PND1 to 1 month, numerous cells showed nuclear immunolabeling for Ki-67. Minimal changes occurred in the sclera after birth except a slight thickening. Scattered Ki-67 immunopositive scleral cells were occasionally observed between PND1 and PND28.
Discussion
As in human and nonhuman primates, pigs are born with open eyelids and relatively well-developed eyes compared to other laboratory animals, such as rodents and dogs, which are born with fused eyelids and markedly underdeveloped eyes. 87,89 Despite the more advanced ocular developmental stage in neonatal pigs compared to other commonly used lab animals and the mature look of some ocular structures at the first glance, the eyes of pigs at birth are still immature and continue to develop during the first 6 months of age. The HE histological evaluation has revealed sings of immaturity in every structure of the eye of both pig breeds at birth, which was further highlighted with Ki-67 immunohistochemical labeling showing the presence of cellular proliferation early in life in most structures. Immunohistochemistry against rhodopsin, synaptophysin, and calbindin highlighted the immature morphological features of the photoreceptor layer at birth and during the postnatal period, notably the global shortness of cones and rods seen with rhodopsin and synaptophysin labeling, and the poorly developed cone inner segment presenting a narrow and oval shape when stained with synaptophysin or calbindin. Calbindin labeling also revealed significant differences in the amount of positively stained cone nuclei between different regions of the pig retina. GFAP immunohistochemistry allowed to characterize the development of retinal glial cells, mostly the elongation of Müller cells processes after birth.
Cornea
The present study revealed that the cornea of both pig breeds was not fully developed at birth, as shown by the incomplete corneal epithelial stratification, the abundant Ki-67 immunopositive epithelial cells in the first weeks/months after birth, and the thin Descemet’s membrane.
The human corneal epithelium is 5 to 6 layers thick at birth, which correlates with the morphology of corneal epithelium in neonatal F2 domestic pigs, but not of Göttingen minipigs, which presented fewer corneal epithelial cell layers at birth. 77 The presence of prominent cytoplasmic vacuoles in the central and peripheral corneal endothelial cells at birth in both pig breeds was similarly described during the first month of life in Sprague Dawley rats. 89
Interestingly, stromal capillaries were observed in the central and peripheral regions of the cornea of multiple Göttingen minipigs, from birth to 1 month of age, inclusively, but not in the cornea of F2 domestic pigs. While corneal stromal vascularization is usually regarded as a pathological process, with neovascularization occurring in response to various insults, Göttingen minipigs included in this study did not present any sign of corneal pathologies. Stromal vascularization in the central and peripheral regions of the normal and healthy developing cornea has never been reported before in animals and human, to our knowledge, including during the prenatal corneal developmental stage. 21 Of note, limbal blood vessels are, however, normally present in animals and human. 19,21 It remained unclear whether the stromal vasculature in the corneal central and peripheral regions in Göttingen minipigs during the first month of life is physiologically involved in the corneal development or if it is a juvenile incidental/background finding specific to the Göttingen minipig. Nevertheless, this finding should be carefully interpreted in toxicological or diagnostic cases as it may not be indicative of the presence of a pathological process. There is evidence in the literature that vacuoles in the corneal endothelium can occur as a postmortem change. 84 However, knowing the minimal delay between the death of Göttingen minipigs and F2 domestic pigs included in this study and the time of fixation, the age-related progression in the distribution and size of these vacuoles and the concomitant age-associated transition in the shape of endothelial cells (from rounded to elongated) suggest that these vacuoles may be a characteristic of immature corneal endothelium. Further investigation would be required to confirm the precise nature of these vacuoles.
Numerous mitotic and nonmitotic Ki-67 immunopositive corneal central and peripheral basal epithelial cells during the postnatal corneal epithelium stratification process and in stromal keratocytes and endothelial cells has been described before in Sprague Dawley rats, but never in pigs. 52,89,96 In rodents, corneal epithelial stem cells were identified in all regions of the developing cornea 19 and the present study suggests the same.
Some results from this study suggest that the cornea of F2 domestic pigs at birth was more developed compared to the cornea of Göttingen minipigs, such as the more advanced epithelial stratification at birth, the shorter time points range with numerous Ki-67 immunopositive corneal epithelial cells noticeable, and the absence of Ki-67 proliferative stromal keratocytes and corneal endothelial cells in the F2 domestic pigs. Nevertheless, in both pig breeds, the corneal epithelium was fully stratified at PND28 and the Descemet’s membrane continued to thicken until 6 months of age, the age at which the cornea was considered histomorphologically mature.
Lens
As in humans, most of lens developmental processes in the Göttingen minipigs and F2 domestic pigs were completed during the gestational stages; nevertheless, subtle evidence of lens immaturity was noticeable at birth and thereafter for the first weeks to months of life in pigs. 48,87 Notably, the subcapsular anterior epithelium was slightly more densely cellular and contained multiple Ki-67 immunopositive proliferative epithelial cells for the first few weeks of life. The lens capsule at birth was markedly thin, and the centrally located OFZ was surrounded by multiple, small basophilic nuclear debris for the first 3 months in Göttingen minipigs and first 2 months in F2 domestic pigs. These small basophilic nuclear debris are compatible with the tardiest step of the secondary lens fiber denucleation process involved in the formation of the OFZ, as described in various species including in human. 4,23,89 As reported in the Sprague-Dawley rat, this nuclear fragmentation process was not associated with the activation of caspase-3 in secondary fibers, suggesting that this apoptotic executioner caspase is not required during this differentiation phenomenon, which has been debated in the literature. 54,89,91 Lens histomorphological maturity was considered reached when the central nuclear remnants disappeared, around 6 months of age in both breeds.
Hyaloid Vasculature
The hyaloid vasculature is critical for the development of the lens and the anterior segment of the eye. 16,24 While the involution of the entire hyaloid system is normally completed prenatally in human around the 35th to 36th gestation weeks, remnants of the tunica vasculosa lentis were noticeable in both pig breeds until PND21. 87 In addition, remnants of the hyaloid artery were present in the vitreous of Göttingen minipigs during the first 2 weeks of life, but not in F2 domestic pigs. Remnants of the tunica vasculosa lentis have been reported histologically up to 8 weeks in Göttingen minipigs, and remnants of the tunica vasculosa lentis and hyaloid artery were reported histologically up to the first week of life in Belgian Landrace domestic pigs. 24,87 The age discrepancies with the present study could be due to breed differences or to the difficulty of capturing such small vestigial structures in histologic sections, especially as the ocular diameter increased with age. Evaluating serial sections of the whole eye could have potentially revealed the presence of more hyaloid vascular remnants over a longer period of time postnatally.
In the present study, apoptotic caspase-3 immunopositive tunica vasculosa lentis endothelial cells and hyalocytes were noted at various time points, similar to what has been described in rats. 89 This is consistent with previous studies in rodents and humans suggesting a role for hyalocytes in the elimination of regressing capillaries and in the potential induction of hyaloid endothelial cells apoptosis. 50,58,95
Noteworthy, hyaloid artery remnants in Göttingen minipigs were surrounded by fine GFAP positive processes compatible with processes from astrocytes originating from the optic nerve and enveloping the hyaloid artery. Interestingly, there are evidence in humans with persistent fetal vasculature and in rodents with retained hyaloid vasculature that astrocytes abnormally envelop the hyaloid artery, preventing its physiologically programmed regression and therefore leading to its persistence after birth. 93,94,97 The hypothesis that astrocytes may be involved in the persistence of hyaloid vasculature postnatally in Göttingen minipigs would need further investigation.
Iridocorneal Angle
At birth, all the components of the ciliary cleft and corneoscleral meshwork composing the iridocorneal angle were identifiable but presented subtle signs of immaturity until approximatively 2 months in both pig breeds. The histological descriptions included in this study were similar to a previous histological and ultrastructural study of the iridocorneal angle of adult domestic pigs. 33,57 The anatomical and histological configuration of the iridocorneal angle of Göttingen minipigs and F2 domestic pigs are very similar to the angle of other nonprimate species, including dogs, cats, and horses. The angle in these species contains a stout pectinate ligament, numerous delicate uveal cords crossing a deep V-shaped ciliary cleft, and an angular aqueous plexus functioning as the aqueous outflow drainage system, which are not characteristics of the human and nonhuman primate angle. 5,69 –72 On the other hand, the size and shape of the corneoscleral meshwork in pigs more closely resembles the primate trabecular meshwork architecture compared to other laboratory animals. 57,70
Ciliary Body and Iris
The development of the iris and ciliary body in Göttingen minipigs and domestic pigs was not complete at birth. After birth, cellularity of the iridal stroma decreased, the iridal sphincter muscles became significantly more prominent, the ciliary processes elongated and became more numerous while important cellular proliferation occurred in the longitudinal muscles of the outer portion of the ciliary body. While the anatomy of the iris and ciliary processes is broadly similar among nonrodent, nonprimate mammalian domestic animals, the outer muscular portion of the ciliary body exhibits more anatomical variations. In pigs, the outer muscular portion of the ciliary body, characterized by a longitudinal and a circumferential smooth muscle, is significantly more developed than the ciliary musculature of rodents, which is almost nonexistent, but is not as prominent as the ciliary muscle of nonhuman primates and humans, which is composed of 3 differently oriented smooth muscles. 70,89
Notably, immunohistochemistry labeling revealed intranuclear activation of caspase-3 in both layers of the ciliary body and iris epithelium without any histological apoptotic phenotype, suggesting a non-apoptotic activation of caspase-3 in these cells. A similar non-apoptotic activation of caspase-3 in the superficial layer of the ciliary body epithelium of juvenile Sprague-Dawley rats has been described. 89 The possible implications of this non-apoptotic activation of caspase-3 in the iris/ciliary body epithelium are discussed below. In addition, the nonpigmented surface epithelial cells of the pars plana portion of the ciliary body epithelium in Göttingen minipigs and domestic pigs showed intense cytoplasmic and nuclear immunolabeling for calbindin, which to our knowledge has not been previously reported. Calbindin is a known marker for several neuronal cell types in the nervous system, including various specialized neurons composing the neuroretina. 14,35,41,46 Since the ciliary body epithelium derives embryologically from the neural tube (neuroectoderm) like the neuroretina, 6,62,70 and considering that the superficial epithelial layer of pars plana is the continuation of the neuroretina, we suspect that this epithelial layer segment shares similar cellular metabolic pathways, which could include the expression of calbindin. However, further research would be needed to confirm this hypothesis.
Retina and Optic Nerve
Despite the presence of distinct retinal cell layers, which may appear mature at birth in Göttingen minipigs and F2 domestic pigs, the retina of neonatal pigs showed evidences of immaturity, and various developmental histological and immunohistological changes occurred during the first 6 months of age. Of note, the ganglion cell layer density decreased in all retinal regions and the INL and ONL thinned with age, while the photoceptors layer markedly developed and GFAP-positive Müller cell processes became more prominent. Unlike commonly used laboratory animals, such as dogs and rodents, which are born with a markedly underdeveloped retina, 79,87,89 the developmental stage of the retina of pigs at birth closely resembles that of neonatal human and nonhuman primates. 82,87
Area Centralis Versus Non–Area Centralis Regions
To our knowledge, this is the first study in pigs that evaluated the postnatal development of the area centralis in neonatal and juvenile pigs and explored the possibility of histological and/or immunohistological variations between the area centralis and non–area centralis regions during the first 6 months of age. The main differences between the area centralis and non–area centralis regions noted histologically, apart from the variation in ganglion cell density, included significantly more numerous INL cell layers in the area centralis region, which was accompanied by more abundant Ki-67 immunopositive nuclei in the middle layers of the INL. Based on their location (middle layers), the Ki-67 positive cells in the INL may correspond to bipolar cells. 92
Importantly, despite previous reports that have showed, using retinal wholemounts, an increased density of cones in the area centralis region of pigs, 13,39 the qualitative histological and immunohistological (with rhodopsin and synaptophysin) evaluations of the photoreceptor layer performed herein did not show significant differences in the rods and cones distribution/density between the area centralis and non–area centralis regions. Therefore, a standard qualitative histological evaluation of photoreceptors may not be sensitive enough to identify subtle variations in the photoreceptor layer composition in the pig retina.
Nevertheless, significant differences in the number of calbindin-positive cone nuclei between the area centralis, the ventral and the dorsal non–area centralis regions were noted. Although calbindin has been used as a cone marker in different species including pigs, 2,35,37,41,63,65 calbindin was not expressed in all cones of the pig retina herein. As cones are easily recognizable with their unique inner segment morphology, differentiating calbindin-positive cones from negatively stained cones was readily feasible. For all ages, calbindin-labeled cone nuclei were significantly more numerous in the dorsal non–area centralis region compared the area centralis region and the ventral retina region. Furthermore, except at PND1, the ventral retina region contained the smallest number of calbindin-positive cones. At birth, the numbers of calbindin-positive cone nuclei were similarly low in the ventral and area centralis regions. Although not statistically significant, the results suggest an increasing trend in the number of calbindin-positive cone nuclei in the area centralis region with age (P = .0741), but not for the ventral and non–area centralis dorsal regions. Using a higher number of animals per age group would be interesting to validate if this increasing trend truly occurs.
Calbindin is a calcium-binding protein that is expressed in a variety of neurons in the nervous system, including in the retina of many species; however, its exact function remains unclear. 3,63,65 In this study, calbindin labeling was located in the nucleus and variably the inner segment of cones, but not in the outer segments, similar to what was described in nonhuman primate and previous studies in pigs. Studies in humans and various animals have shown that not all types of cones express calbindin and that there are expression variations between species. 2,17,18,35
Indeed, in macaque and marmoset, all short and medium-long wavelength cones contained calbindin, while calbindin was only present in medium-long cones in human. For both nonhuman primate and human, calbindin was absent in cones located in the foveal and parafoveal regions. 17,36,63,65 Also, cones in the retina of nocturnal animals do not express calbindin. 17,37,63,65 It is also known that the different types of cones are not equally distributed throughout different regions of the retina in humans and animals, including in pigs. 36,39,47 In pigs, the cone-rich area centralis area is mainly composed of medium-cones (more than short-cones) and the dorsal retina contains more numerous medium-cones than the ventral retina. 47 In humans, the cone-rich fovea is composed predominantly of long- and medium-cones. 11,47 Therefore, although more studies are needed to identify the specific subtype of cones that express calbindin in the pig retina, this study suggests that only a subset of cones express calbindin and that these cones are predominantly distributed in the dorsal retina. To our knowledge, this is the first study to document the distribution of calbindin-positive cones in the retina of pigs.
Photoreceptor Development
At birth in both pig breeds, the photoreceptor layer was histologically underdeveloped, which could be further appreciated with immunohistochemistry labeling for synaptophysin, calbindin, and rhodopsin. The latter marker highlighted the global shortness of rods inner and outer segments, while synaptophysin and calbindin emphasized the immature morphological features of cones inner segment, which was narrow and oval at birth and progressively widened until 6 months of age to finally adopt the typical wide and triangular/square shape. Although synaptophysin is usually used as a synapsis marker, this marker also specifically marks the inner segments of photoreceptors. The marked elongation of rods stained with rhodopsin after birth was also previously described in domestic pigs. 35
Retinal Glial Cell Development
Immunohistochemistry for GFAP did not reveal any labeling pattern differences with age for the immunostained astrocyte processes travelling in the NFL and surrounding capillaries of the retinal vascular plexus. However, GFAP-stained fine vertical linear processes reaching the ILM with wide “end-feet” (compatible with Müller cells processes), 46 were significantly thinner and shorter (crossing fewer retinal cells layers) at birth and elongated until 3 months in Göttingen minipigs (and until between 2 months and 6 months in domestic pigs, in which the 3 month time point was not available). GFAP activation in Müller cells is commonly associated with retinal injury; 7,33,45 however, in the present study, the increased labeling pattern was associated with normal postnatal development of the retina. This labeling pattern has been reported before when comparing normal pigs at birth and at 6 months of age. 46 The present study did not assess if this progression in the labeling pattern was the result of actual elongation of astrocytes processes or an increased GFAP expression.
Optic Nerve
The optic nerve was immature at birth, characterized by an increased cellularity, the presence of mitotic figures, and a poorly developed lamina cribosa. Calbindin immunohistochemistry labeling was noted in some nuclei in the optic nerve at all time points; however, the specific cell type expressing this protein is uncertain. Calbindin has been shown to be expressed in various subsets of neurons within the nervous system, including in the eye neuroretina, but its role remains unclear. 3,12,68
Retinal and Optic Nerve Cell Proliferation and Apoptosis
Mitotic figures were not noted in the retina of pigs after birth but were present in the optic nerve. Ganglion and non-ganglion cells in the GCL as well as some nuclei located in the middle layers of the INL (compatible with bipolar cells) presented a diffuse nuclear or nucleolar labeling pattern for Ki-67 for various time periods postnatally. In addition to labeling mitotic figures, Ki-67 immunohistochemistry detects cells present in all phases of the cell cycle, except in the G0 phase. Therefore, this marker also labels cells engaged in cellular activities such as DNA synthesis and growth as well as cells exiting the cell cycle, which may be occurring in the aforementioned immunopositive cells in the immature retina of pigs. 26,75 Caspase-3 immunopositive apoptotic bodies were only detected at birth in the INL. In a previous study in rats, the INL was the last layer to present caspase-3-positive apoptotic bodies during the postnatal development phase. 89
Retinal and Optic Nerve Background/Incidental Changes
Different background/incidental changes were observed in the retina and optic nerve of domestic pigs and Göttingen minipigs. Almost all ocular sections from both pig breeds, regardless of age, presented some degree of optic disc cupping, which has been described previously in the eyes of Göttingen minipigs as an artefact caused by excessive pressure on the eye or traction force on the optic nerve at necropsy. 55 Furthermore, several animals at various ages presented retinal rosettes which were always located in or just adjacent to the optic disc, unlike rosettes in rabbits which are more commonly observed in the peripheral retina. Incidental retinal rosettes have been described previously in rabbits, rodents, and dogs, but not in pigs. 51,66,74 Furthermore, the retina of both pig breeds at various ages presented displaced photoreceptors nuclei within the photoreceptor layer, which has also been reported as an incidental finding in normal rodents, nonhuman primate and human. 66,73
A peculiar background change was observed in 2 Göttingen minipigs (3 months and 6 months old), and not in F2 domestic pigs: the optic nerve, near the optic disc, contained a poorly defined and nonencapsulated nodule, which appeared to be composed of dysplastic and ectopic/displaced retinal neurons, as highlighted with immunohistochemistry markers. Rod photoreceptors and horizontal cells could convincingly be identified with rhodopsin and calbindin, respectively, while the presence of numerous large cells with cytoplasmic labeling for synaptophysin, resembling retinal ganglion cells, were also noted. This finding was observed randomly in 2 Göttingen minipigs out of 16 animals, but additional sectioning of the other subjects was not performed to confirm the true incidence of this finding and to investigate if it is present from birth.
Non-Apoptotic Activation of Caspase-3
Caspase-3 labeling of non-apoptotic cells (not presenting an apoptotic phenotype) was noted in the ciliary body and iris epithelium, in various specialized neurons of the retina, in the optic nerve, and in the retinal pigmented epithelium. The activation of caspase-3 without the induction of apoptosis has been associated with various cellular events associated with development and normal function of several cell/tissue types, but has mostly been described in neuronal cells in the brain. 20,43,49,60,61,78,86 There is one report documenting the non-apoptotic activation of caspase-3 in retinal ganglion cells of zebrafish. 43 The non-apoptotic activation of caspases in the nervous system is involved in neuronal differentiation, neurite outgrowth, dendrites branch formation, and synaptic plasticity. 43 Although not described before, the numerous caspase-3-positive neurons noted specifically in some layers of the INL of the pig retina may be engaged in similar cellular events. Furthermore, caspase-3 non-apoptotic activation was observed in the ciliary body and iris epithelium, which share the same embryological origin (the neural tube from the neuroectoderm) as the neuroretina and the optic nerve, in which a similar labeling pattern was observed. Similar physiological nonapoptotic capsase-3 activation mechanisms may occur in these immunopositive cell types of the eye. 6,62,70
Overall, the results from this study showed that the eyes of Göttingen minipigs and F2 domestic pigs are not fully developed at birth and still undergo histological and immunohistological developmental changes during the first 6 months of life, which involved some degree of cellular proliferation and remodeling. There was no significant histomorphological and developmental differences between both pig breeds, with the exception of the cornea of domestic pigs, which may be slightly more developed at birth compared to Göttingen minipigs. For both pig breeds, all ocular structures were considered histomorphologically mature at 6 months of age. The present study provided comprehensive information on the immunohistochemistry labeling patterns of various commonly used retinal markers in both pig breeds from birth up to 6 months of age. Moreover, this is the first study to describe histologically and immunologically the postnatal differences existing between the area centralis and non–area centralis regions of the retina in Göttingen minipigs and F2 domestic pigs, which included notably the specific distribution of calbindin-positive cone photoreceptors. The retina of pigs, with its area centralis region mimicking the human macula, may provide an interesting model to study macula-associated pathologies.
Supplemental Material
Combined_supplemental_materials-Vrolyk_et_al - Neonatal and Juvenile Ocular Development in Göttingen Minipigs and Domestic Pigs: A Histomorphological and Immunohistochemical Study
Combined_supplemental_materials-Vrolyk_et_al for Neonatal and Juvenile Ocular Development in Göttingen Minipigs and Domestic Pigs: A Histomorphological and Immunohistochemical Study by Vanessa Vrolyk, Marie-Josée Desmarais, Daniel Lambert, Julius Haruna and Marie-Odile Benoit-Biancamano in Veterinary Pathology
Footnotes
Acknowledgements
We are thankful to Dr Martine Denicourt from the Faculty of Veterinary Medicine of the Université de Montréal for donating neonatal F2 domestic pigs and to Nicole Navratil and Michelle Salerno from Marshall BioResources, North Rose, New York, for donating all Göttingen minipigs. We thank the technical staff from the Diagnostic Service of the Faculty of Veterinary Medicine of the Université de Montréal for the eye collection of F2 domestic pigs in necropsy as well as Jacinthe Cardin for her help with the fixation and sectioning of the eyes of F2 domestic pigs. We also thank the technical staff from Charles River Laboratories Montreal ULC for the eye collection and slides preparation for the Göttingen minipigs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported through grants from Mitacs, the Fonds du Centenaire of the Université de Montréal, and from Charles River Laboratories Montreal ULC.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
