Abstract
Immunohistochemistry allows the localization of proteins to specific regions of the nephron. This article reports the identification and localization of proteins in situ within normal canine, feline, and mouse kidney by immunohistochemistry; maps their distribution; and compares results to previously reported findings in other species. The proteins investigated are aquaporin 1, aquaporin 2, calbindin D-28k, glutathione S-transferase-α, and Tamm-Horsfall protein. Aquaporins are integral membrane proteins involved in water transport across cell membranes. Calbindin D-28k is involved in renal calcium metabolism. Glutathione S-transferase-α is a protein that aids in detoxification and drug metabolism. The role of Tamm-Horsfall protein is not fully understood. Proposed functions include inhibition of calcium crystallization and reduction of bacterial urinary tract infection. The authors’ findings in the dog are similar to those in other species: Specifically, the authors localize aquaporin 1 to the proximal convoluted tubule epithelium, vasa recta endothelium, and descending thin limbs; aquaporin 2 to collecting duct epithelium; and calbindin D-28k within distal convoluted tubule epithelium. Glutathione S-transferase-α has variable expression and is found in only the renal transitional epithelium in some individuals, in only the proximal straight tubules in others, or in both locations in others. Tamm-Horsfall protein localizes to thick ascending limb epithelium. These findings are similar in the cat, with the exception that aquaporin 1 is located in glomerular podocytes, in addition to proximal convoluted tubule epithelium, and glutathione S-transferase-α is found solely within the proximal convoluted tubule within all kidney samples examined. The mouse kidney is almost identical to the dog but expresses glutathione S-transferase-α in the glomeruli only.
Immunohistochemistry (IHC) utilizing aquaporin 1 (AQP1), aquaporin 2 (AQP2), glutathione S-transferase-α (GST-α), calbindin D-28k, and Tamm-Horsfall protein (THP) antibodies has been used to localize these proteins to segments of the nephron in various species, including human and rat kidneys.6,13,19,23,25–27,30,39,40 Identification of specific segments of the nephron can be challenging histologically. AQP1, AQP2, THP, and calbindin D-28k have been used as nephron segment markers and utilized in double-labeling IHC to more exactly localize other proteins within the nephron of humans and rats. 21 By localizing proteins of interest within the nephron, we can further elucidate the function of these proteins in renal physiology and pathophysiology and may be better able to identify nephron segments in disease states.
Our study establishes the in situ location of proteins that have a distribution well documented in other species and identifies these proteins as nephron segment markers in the dog, cat, and mouse kidney. The proteins evaluated include AQP1 for localization of the proximal convoluted tubule (PCT), AQP2 for the collecting ducts, GST-α for the PCT, calbindin D-28k for the distal convoluted tubule (DCT), and THP for the thick ascending limb (TAL) of the loop of Henle.6,13,19,23,25–27,30,39,40
Materials and Methods
Five disease-free kidneys from each of the 3 species (dog, cat, and various strains of sentinel mice) were obtained from recently euthanized animals and formalin fixed. Tissues were routinely processed and paraffin embedded. Four-micrometer sections were placed on positive-charged slides. Sections were deparaffinized with xylene and rehydrated by a series of graded baths in alcohol and phosphate buffered saline buffer.
Heat-induced epitope retrieval was performed on all slides, except those for THP IHC. 31 Sections for heat-induced epitope retrieval were heated to 125°C, under pressure, for approximately 1 minute in target retrieval solution (‘low pH [6.0] Dako, Carpinteria, CA, for AQP1; high pH [9.0] Dako, for AQP2, calbindin d28-k, and GST-α). Nonspecific antibody binding was blocked for 5 minutes in Background Sniper (Biocare Medical, Concord, CA). Endogenous peroxidase activity was quenched in 3% hydrogen peroxide. The samples were washed with Tris-buffered saline.
All samples were incubated with their primary antibody overnight at 4°C. The following primary antibodies were used: rabbit anti-human AQP1 polyclonal antibody (1:500, Chemicon, Temecula, CA), rabbit anti-rat AQP2 affinity-purified polyclonal antibody (1:500, Millipore, Billerica, MA), anti-human GST-α monoclonal antibody (1:50, Novocastra, Bannockburn, IL), anti-bovine calbindin D-28k monoclonal antibody (1:1000, Sigma-Aldrich, St Louis, MO), and sheep anti-human Tamm-Horsfall glycoprotein polyclonal antibody (1:2500, Chemicon).
Sections for THP immunostaining were then incubated with an HRP-labeled anti-sheep secondary antibody for 1 hour at 4°C. 31 All other slides were incubated with Envision+ System (Dako). Immunoreactive complexes were identified with diaminobenzidine and the sections counterstained with hematoxylin.
Renal tubules and other microstructures were identified on the basis of light microscopic features and anatomical location. In the renal cortex, PCTs have a greater external diameter than DCTs, and the luminal epithelium has a brush border, giving the tubular lumen a “star-shaped” appearance. 33 These features are absent in the DCT. 33 These morphologic features are carried into the medullary rays, where the PCT becomes the straight portion of the proximal tubule. 33 Also within the medullary rays is the beginning portion of the DCT, known as the TAL, sharing the morphologic features of cortical DCT. 33 Running in parallel with these structures are the collecting ducts. These tubules are similar in diameter to proximal tubules but have smaller, cuboidal epithelium with prominent cell borders, exhibit more nuclei per cross section, and do not have a brush border. 33 Thin limbs are within the medullary rays and identified by their moderately sized lumen and thin, low, cuboidal to squamous epithelium. Small blood vessels throughout the kidney are thin-walled, endothelial-lined structures containing erythrocytes. The more prominent vessels in the medullary rays, running in tandem with renal tubules, are the vasa recta.
Western blot analysis to confirm appropriate species cross-reactivity was also performed. Freshly collected kidney tissue from all 3 species was flash frozen and stored at –80°C. Later, RIPA lysis buffer (Santa Cruz Biotech, Santa Cruz, CA) and a protease inhibitor cocktail (Sigma-Aldrich) were added to small pieces of thawed homogenized renal tissue. Centrifuged supernatants were aliquoted and stored at –20°C until thawed for immediate analysis.
Protein concentration was quantified using a modified Lowry technique. The amount of protein loaded onto premade SDS gels (Bio-Rad Laboratories, Hercules, CA) varied by the protein to be isolated and species of animal. Gels were run for 1 hour at 180V, after which gels were transferred to PVDF membranes (Millipore) overnight at 35V in a 4°C cold room. Membranes were then blocked in 5% milk buffer for 1 hour prior to incubating with the primary antibody for 3 hours. Membranes were incubated with an appropriate biotinylated secondary antibody in 5% milk buffer for 1 hour. For THP blots, a directly conjugated rabbit anti-sheep HRP secondary antibody (Abcam, Cambridge, MA) was used. 31 Endogenous peroxidase activity was blocked using hydrogen peroxide, after which the remaining samples were incubated for 30 minutes in streptavidin–horseradish peroxidase. Protein bands were identified using a chemiluminescent kit (Thermo Scientific, Rockford, IL).
Results
Aquaporin 1
Staining for AQP1 was positive in the canine kidney within the luminal epithelial cells of the PCT and proximal straight tubules (PST), the vasa recta endothelium, the descending thin limbs (DTLs) of the loop of Henle, and erythrocyte membranes (Figs. 1, 2). Within the outer cortical PCT, staining was intense along the cells’ apical margin with a less intense, finely granular cytosolic component (Fig. 1). Within the inner cortical medullary rays, AQP1 stained the luminal cells of the PST. These cells had strong staining along their basolateral margin and even more intense staining along the apical margin. There was only faint cytosolic staining in these cells. Nuclei did not stain. The endothelium of the vasa recta was intensely stained. Within the medulla, intermittent thin limbs showed strong, diffuse staining of their luminal cells (Fig. 2). The thin limbs that stained for AQP1 are likely to be the DTLs, based on localization of AQP1 to the DTLs in other species.3,7,23,26,27
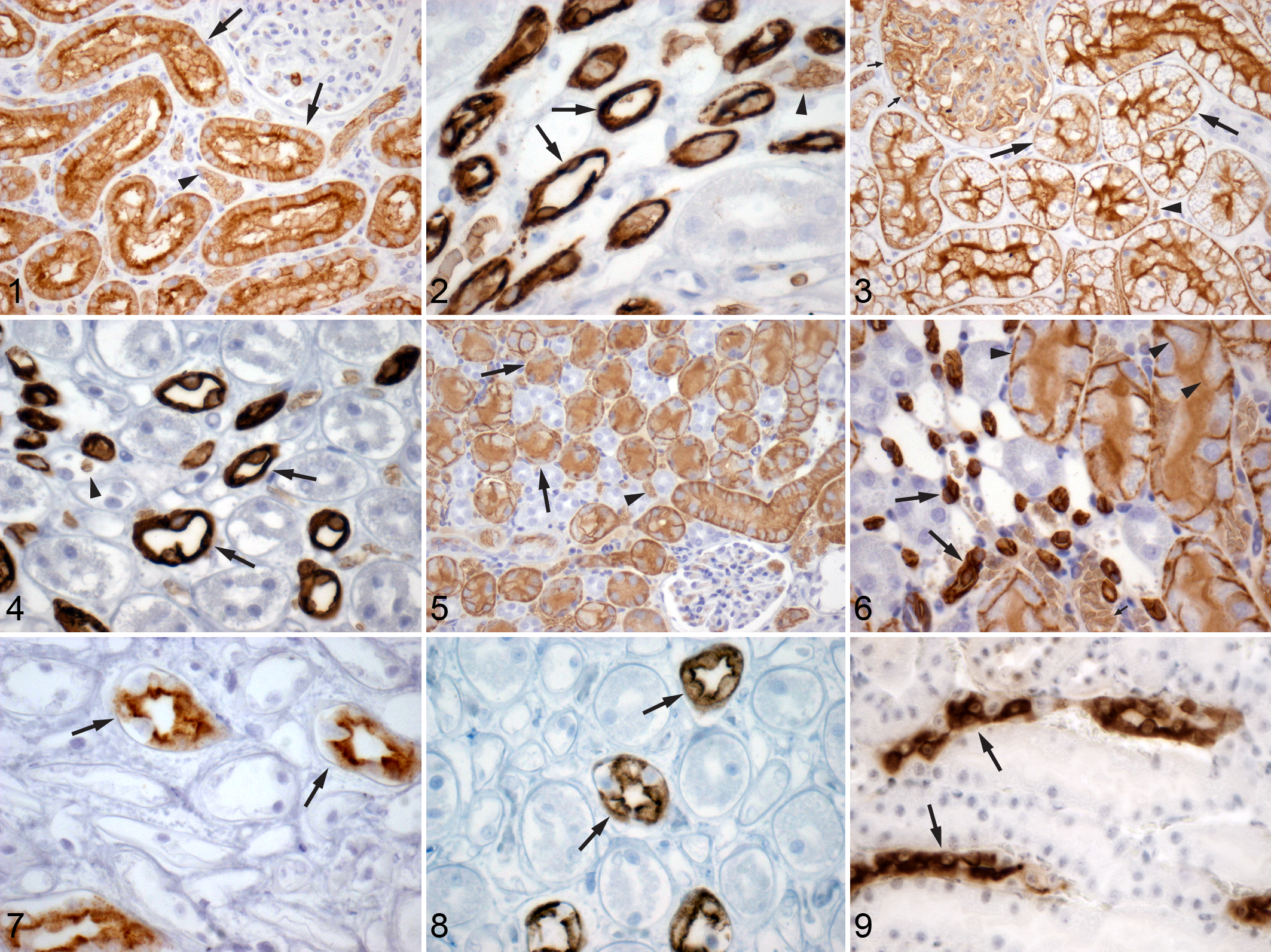
Findings were similar in the renal epithelium of the cat with some minor differences. Staining for AQP1 was strong in the PCT luminal cells but was equally intense along the basolateral and apical membranes in the outer cortex (Fig. 3). There was no staining noted in the cytoplasm or nuclei of these cells. Glomerular podocytes also stained positive for AQP1 in cats only (Fig. 3). As seen in the dog, cat PST epithelium within the medullary rays stained more intensely than in the outer cortex along the basolateral and apical membranes of the luminal cells. The endothelium of the vasa recta stained strongly for AQP1. Within the medulla, intermittent thin limbs were intensely stained, similar to previously reported positive staining of the DTLs in other species (Fig. 4).3,7,23,26,27
AQP1 staining in the murine kidney was identical to that described above for the dog (Figs. 5, 6). This is in keeping with previous reports in rat and mouse kidneys.26,27,41,43
Aquaporin 2
Immunostaining for AQP2 appeared similar within the cortex and medulla of the dog. AQP2 intensely stained the apical border of intermittent luminal cells of the collecting ducts (consistent with the principal cells) (Fig. 7). There was also less intense, granular staining of the cytoplasm of these cells. The nuclei did not stain for AQP2. No other regions of the nephron had AQP2 staining.
In the cat, intermittent luminal cells, the principal cells, of the collecting duct stained for AQP2 (Fig. 8). In contrast to the dog, the staining pattern within the cortex was both basilar and apical, becoming primarily apical as the collecting ducts progressed into the medulla. The cat also had less intense, granular staining of the cytoplasm. As in the dog, no other regions of the nephron had AQP2.
Positive staining for AQP2 was seen along the borders of the principal cells of the collecting duct and diffusely throughout the cytoplasm in the mouse kidney.
The findings in our species are similar to reports in other mammalian species, including rats and mice (Fig. 9).26,32,38,39
Glutathione S-transferase-α
GST-α had variable expression within the dog kidney. Interestingly, the transitional epithelium lining the renal papilla of some dogs was positive for GST-α staining along the cell borders and diffusely throughout the nucleus (Fig. 10 ). Less intense cytosolic staining is also visible. In some kidney sections with positive GST-α staining of renal transitional epithelium, no staining was found in the tubules (Fig. 11). Kidney sections from other dogs demonstrated similar positive GST-α staining in the epithelium of PST within the medullary rays with only rare staining of PCT epithelium (Fig. 12). The amount and intensity of staining in the PST is also variable among individuals. These findings are in contradiction with a previously published study examining the location of GST isoenzymes in the dog kidney, which found only isoenzymes μ and π, not α, within renal tubules. 5
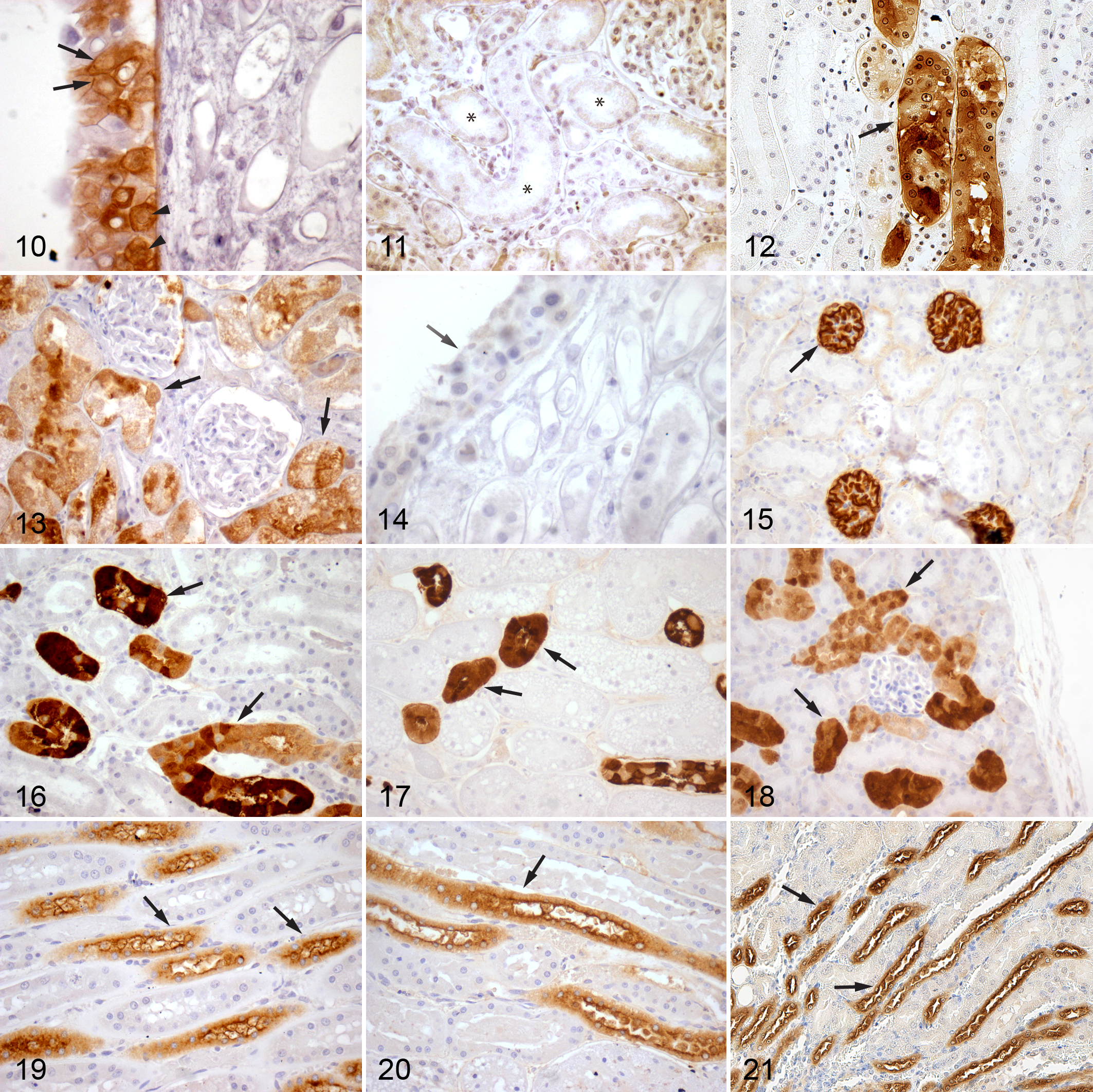
The epithelial cells of the PCT in the feline kidney had diffuse, cytoplasmic GST-α, as well as nuclear localization (Fig. 13). However, there was variable intensity of immunostaining among segments of the PCTs. No staining of renal transitional epithelium was observed (Fig. 14).
Contradictory to previous reports in rats and mice, GST-α was not identified in the PCTs of our mouse kidneys.8,15 Instead, the glomeruli stained intensely positive for GST-α (Fig. 15), and no other part of the renal tubule stained positive for the enzyme.
Calbindin D-28 k
Calbindin D-28k was present in the DCT of the dog, cat, and mouse (Figs. 16–18, respectively). In the dog and mouse, the tubules in the outer cortex stained the strongest. The inner cortex and outer medulla had lesser numbers of stained tubules. The intensity of the staining of these tubules was somewhat variable in all regions of the kidney. Additionally, there was variability in intensity of individual epithelial cells stained within the same tubular segment. Cellular localization was both cytosolic and nuclear. This staining pattern is consistent with previously reported staining of the DCTs, connecting tubules, and, to a lesser extent, the proximal collecting ducts in other species.14,40
Staining for calbindin D-28k was almost identical in the cat kidney. However, within the outer cortex, there was even more variability in the intensity of individual cell staining within the same tubule. Some cells did not stain at all, while others varied from faint to intense in their expression of calbindin D-28k.
Tamm-Horsfall Protein
The distribution of THP was identical in all 3 species. The epithelia of the TAL in the loop of Henle were intensely stained, while there was less intense staining along cells lining the DCT (Figs. 19–21). THP was expressed along the apical border of the epithelial cells in both the TAL and DCT. There was also diffuse cytoplasmic staining in these cells. Nuclei did not stain. No other regions of the nephron stained positive for THP.
The renal distribution of AQP1, AQP2, GST-α, calbindin D-28k and THP is presented in schematic diagram form for all three species investigated here (Figs. 22–24).
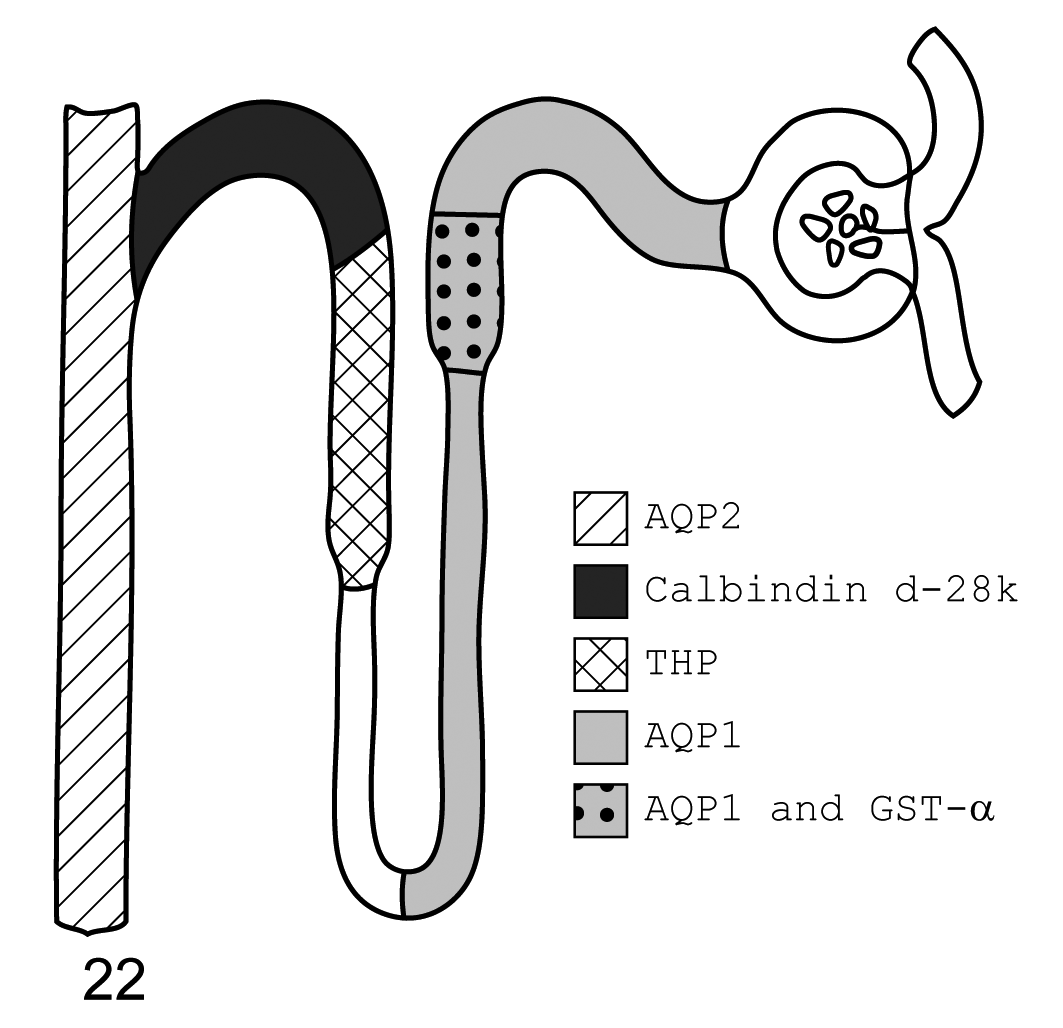
Dog. Diagram of immunohistochemistry localization of aquaporin 1, aquaporin 2, glutathione S-transferase-α (GST-α), calbindin d-28k, and Tamm-Horsfall protein. From left to right, aquaporin 2 is found in the collecting ducts, calbindin d-28k in the distal convoluted tubule, Tamm-Horsfall protein in the thick ascending limb, aquaporin 1 throughout the descending thin limb and proximal convoluted tubule, and GST-α is localized to the proximal straight tubules of the loop of Henle. Some dogs did not stain positive for GST-α at this site. Not represented is the variable staining of renal transitional epithelium and rare proximal convoluted tubule epithelial cells for GST-α seen in some dogs.
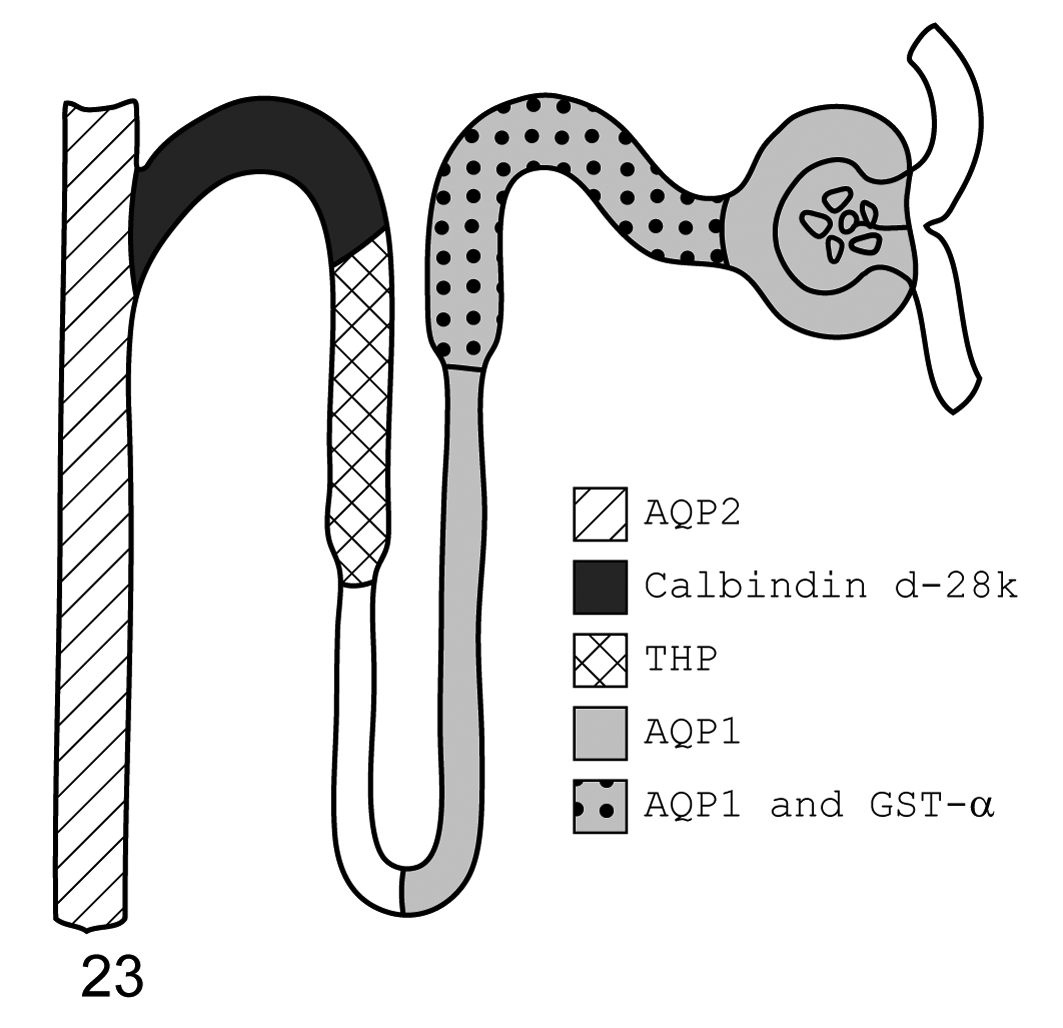
Cat. Diagram of immunohistochemistry localization of aquaporin 1, aquaporin 2, glutathione S-transferase-α, calbindin d-28k, and Tamm-Horsfall protein. From left to right, aquaporin 2 is found in the collecting ducts; calbindin d-28k in the distal convoluted tubule; Tamm-Horsfall protein in the thick ascending limb; aquaporin 1 throughout the descending thin limb, proximal convoluted tubule, and glomerulus (podocytes); and glutathione S-transferase-α in the proximal convoluted tubule.
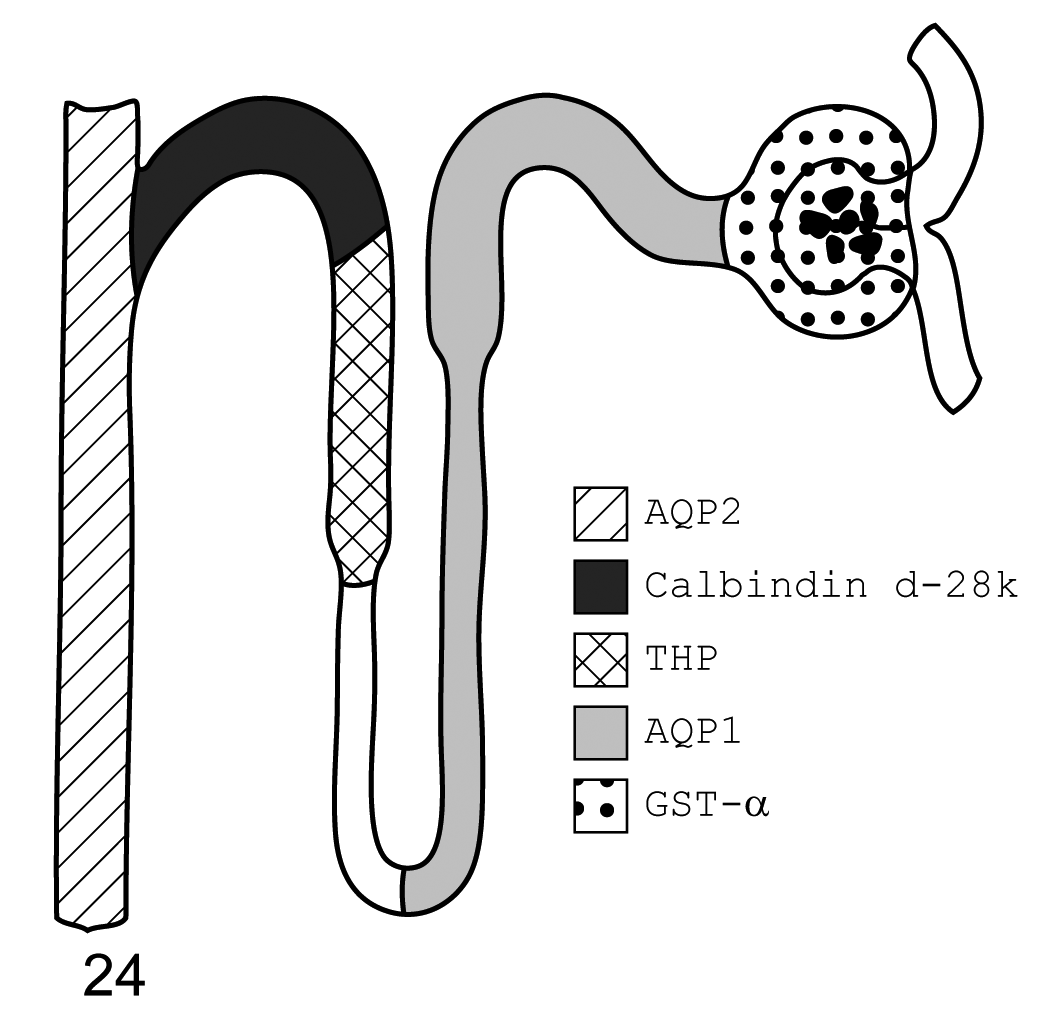
Mouse. Diagram of immunohistochemistry localization of aquaporin 1, aquaporin 2, glutathione S-transferase-α, calbindin d-28k, and Tamm-Horsfall protein. From left to right, aquaporin 2 is found in the collecting ducts, calbindin d-28k in the distal convoluted tubule, Tamm-Horsfall protein in the thick ascending limb, aquaporin 1 in the descending thin limb and proximal convoluted tubule, and glutathione S-transferase-α in the glomerulus.
Western Blot Analysis
Western blot analysis of homogenized kidney tissue from all 3 species was performed for AQP1, AQP2, GST-α, calbindin D-28k, and THP to further validate species antibody cross-reactivity.
Immunoblotting of dog, cat, and mouse kidney tissue confirmed the presence of 2 protein species of approximately 25 and 37 kDa when probed with rabbit anti-human AQP1 polyclonal antibody (Chemicon), which is consistent with molecular masses of the unglycosylated (28 kDa) and glycosylated (35–60 kDa) forms of AQP1 previously reported (data not shown).
Immunoblotting using anti-human GST-α monoclonal antibody (Novacastra, Bannockburn, IL) confirmed the presence of 2 bands of approximately 17 and 25 kDa in the kidney homogenate of all 3 species. Previous studies have shown a molecular weight of 26 to 31 kDa for GST-α in rabbits, rats, and humans.
Bands of approximately 25 kDa were found using immunoblotting with mouse anti-bovine calbindin D-28k monoclonal antibody (Sigma-Aldrich) in kidney homogenate in all 3 species. As the name implies, calbindin D-28k is known to have a molecular weight of 28 kDa.
AQP2 and THP could not be isolated via immunoblotting.
Discussion
Aquaporin 1 and Aquaporin 2
AQP1 was the first aquaporin discovered in the early 1990s. It is the water channel that contributes to water balance within many tissues and organs, especially in the kidney, where AQP1 is vital to water absorption by the PCT.4,6,25–27
Since the discovery of AQP1, 9 other aquaporin isoforms have been discovered in the kidney, including AQP2.4,6,25–27 AQP2 is the chief target for vasopressin in the collecting duct. Vasopressin induces a shuttling mechanism that moves AQP2 from intracellular vesicles to apical plasma membranes.32,35,39 Once AQP2 has relocated to the apical plasma membrane, water can be reabsorbed through the water channel, concentrating urine.32,35,39 Conversely, in the absence of vasopressin, AQP2 is endocytosed back into the cytoplasm, and water is excreted in the urine.32,35,39
AQP1 and AQP2 have been studied extensively in human medicine, primarily relying on rat and mice models. Understanding the influence of various disease states on these renal proteins has greatly increased our understanding of renal water balance. For example, late-phase polyuria associated with cisplatin toxicity results from the loss of AQP1 from the PCT, as well as the loss of AQP2 from the collecting ducts, leading to the inability to concentrate urine. 18 Another study shows that the reduction of AQP1 and AQP2 in ischemic acute renal failure models probably leads to the lack of urine concentrating ability after the insult. 43 Changes in AQP1 and AQP2 expression in the kidney and their excretion in the urine can also be affected by extrarenal diseases, such as congestive heart failure and liver failure. 16,44
AQP1 has been localized to the kidney in humans, rats, and mice, as well as the musk shrew and house sparrow. 3,7,22,26,27 Specifically, AQP1 is found along the apical and basolateral membrane of luminal cells within the PCT. The highest expression is in S3, the straight portion of the proximal tubule that descends into the outer medulla. AQP1 is also found within type II DTL epithelium and, to a lesser extent, in type III and type I DTL epithelium and the descending portion of the vasa recta. 3,25,27 Type II DTL epithelium is found in long-looped nephrons within the cortical labyrinth. This specialized epithelium has tall epithelium, short and blunt microvilli, and small numbers of organelles. Type I and type III DTL epithelium is simpler in structure and found in short-looped nephrons and within the inner medulla, respectively.
Less research on aquaporin expression has been done in dogs and cats. AQP1 expression was evaluated via immunoblotting in tissue from Shiba Inu dogs. AQP1 was found in the lung, kidney, and spleen of these dogs. 28 To our knowledge, however, the exact location and characterization of AQP1 within the dog kidney have not been previously reported, nor has any information regarding renal AQP1 expression in felines been reported. We localized AQP1 within the dog and cat to sites reported for other species: the PCT, DTLs, and vasa recta (Figs. 1–4). Interestingly, the cat also expressed AQP1 in the glomerular podocytes, indicating a potential difference in renal water management for the cat (Fig. 3). Additionally, we reconfirmed the presence of mouse AQP1 in these locations (Figs. 5, 6).
AQP2 has also been identified in many mammalian and avian species, with most research investigating human, rat, and mouse kidney tissue. 3,20,22,32,38,39,42 Renal expression of AQP2 in dogs and cats has not been reported. Our findings are identical to those of previously studied species. We found that AQP2 is located in the principal cells of the collecting duct and, to a lesser extent, the connecting tubules in both dogs and cats (Figs. 7, 8). These findings were reconfirmed in mice (Fig. 9). Specifically, AQP2 is found chiefly along the apical border of the principal cells but can also be seen within the cytosol.
The localization and characterization of renal AQP1 and AQP2 expression confirm the role of these proteins in water regulation in dogs, cats, and mice. Similar distribution of AQP1 and AQP2 in these species would be expected given that the basic physiology of water management is likely conserved between most mammalian species. This confirmation is the initial step in evaluating the roles of AQP1 and AQP2 in both renal and extrarenal diseases of cats and dogs. Additionally, these proteins can reliably serve as in situ markers for their respective regions of the nephron, which can be difficult to distinguish from other tubules histologically.
Glutathione S-transferase-α
Glutathione S-transfersases are a family of enzymes found in various tissues, especially the liver and kidney. They are integral in detoxification of many substances, including cyclosporine, other cytotoxic drugs, chemicals, and carcinogens. 10,13,15 There are a few different classes of the enzyme (GST-α, GST-μ, GST-π) that have unique locations within human and rat kidneys. 10,13,15
GST-α is not found in the urine of normal, healthy individuals but is found in the urine of individuals that have incurred renal tubular damage prior to an increase in serum creatinine. 10 Specifically, increased GST-α levels have been seen in urine from individuals with the following disorders affecting the PCT epithelium: cyclosporine-induced PCT damage, acute tubular necrosis, renal transplant infarct, ischemia, cisplatin toxicity, gentamicin toxicity, and post–heavy metal exposure. 10,13,15 There is promise in its use as an early marker of acute renal disease or to monitor individuals undergoing certain medical treatments, although additional research is needed. 10,13,15,17,45
GST-α is present in the cells of the PCT of rabbits, rats, and humans. 13 In humans, the cellular localization of GST-α is described as cytoplasmic and nuclear within individual cells. 13 Minimal GST-α has also been noted in medullary tubules believed to be the thin limbs of the loops of Henle in humans. 13
Distribution was quite different in the dog kidney. The transitional epithelium along the renal papilla showed vibrant, strong cytoplasmic and nuclear GST-α expression in some individuals (Fig. 10). Overall, sections of dog kidney did not demonstrate significant GST-α staining in the PCT (Fig. 11). Only rare PCT epithelial cells stained positive in some individuals. However, some kidney samples showed staining of the PST epithelium within in the medullary rays (Fig. 12). Two kidneys (from different individuals) had staining of both the renal transitional epithelium and PST epithelium.
To date, few studies concerning GST expression in dog renal tissue have been performed. Bohets et al identified the presence of isoenzymes GST-μ and GST-π in homogenized dog kidney tissue and the cytosol of an established dog renal cell line using affinity chromatography and immunoblotting. 5 In contrast to our results, this study found no GST-α in the canine kidney.
The variable but positive staining of the transitional epithelium and PST epithelium in our study suggests that there may be individual or breed variation in GST isoenzyme expression within renal tissues. This theory for variation in individual GST-α expression may be further substantiated by the findings of researchers investigating the role of P450 and GST-α in trichloroethylene renal toxicity in rats. Cummings et al found that their strain of male F344 rats varied from other previously studied rats in that they possessed GST-α in both their PCT and DCT, and they suggested that this variation in GST-α distribution may be due to the strain of rat being used. 8
In contrast to the dog, the feline kidney showed expression of GST-α similar to that previously reported in humans and rats. Staining was shown in all PCT cells but was of variable intensity between individual cells (Fig. 13). Renal papilla transitional epithelium did not demonstrate the presence of GST-α in the cat (Fig. 14). Also, individual variation in feline renal expression of GST-α was not seen in this study.
The mouse kidney also had strikingly different results than expected. Rats have been utilized extensively for GST research, and while glutathione S-transferase class expression appears slightly variable among rat strains, GST-α remains confined to renal tubules. 8,10,11,15 In the mouse kidney, glomeruli stained positive for all individuals sampled, and no tubular staining was appreciated (Fig. 15). Again, these findings in the murine kidney, as well as the dog kidney, highlight the importance of understanding species variations in tissue protein expression, especially before relying on them as markers for disease.
Calbindin D-28k
Calbindin D-28k is a calcium-binding protein with a very strong affinity for calcium. 14,30,34 In the kidney, there are several theories on the role of calbindin D-28k, and it is therefore possible that the protein has multiple functions within this organ alone. 14,30,34,40 Although approximately 60% of calcium is reabsorbed in the proximal tubule, it is in the DCT that selective calcium reabsorption occurs. 14 1,25-Dihydroxyvitamin D3 is made in the proximal tubule, but its actions take place in the distal tubule where calbindin is expressed. 14 This has long formed a strong association between selective calcium reabsorption and calbindin. As renal calbindin expression appears dependent on and influenced by many factors, including 1,25-dihydroxyvitamin D3, PTH, and calcitonin, it seems certain that the protein is involved in calcium regulation, but the exact involvement is yet unknown. 14 Some theories suggest calbindin acts as a calcium shuttle or as a buffer (keeping intracellular calcium below a toxic level) or plays a role in the basolateral membrane calcium pump of renal epithelial cells. 14
Calbindin D-28k is found in many tissues in many species, including various reptiles, amphibians, fish, chicks, rats, rabbits, monkeys, and humans. 30,34,40 The protein has been commonly found in kidney, intestine, pancreas, and bone in many of these species. 30,34,40 It has also been heavily studied in retinal and brain tissue from various species, including the cat and dog. 9,12 Its expression and function appears to be dependent on vitamin D metabolites in some tissues, as in the kidney, but not in all. 9,12,14,30,34,40 Therefore, the functions of calbindin D-28k could be widely different within these tissues.
In previous studies, calbindin D-28k has been localized to the DCT of several mammalian species, including rabbits, rats, and humans. 14,40 In these species, the staining is seen diffusely in the cytosol and nucleus. Calbindin D-28k has also been found in the connecting tubules and in the collecting ducts with cytosolic and nuclear expression. 30,34,40 There is progressively less expression of calbindin in these regions. 30,34,40 However, there does appear to be some species variation in the abruptness of this change. 30,34,40 It should also be noted that a 1998 toxicology study examining the nephrotoxic effects of cyclosporine A on dogs did localize calbindin D-28k to renal tubular epithelium via IHC but did not go as far as to state which renal tubules stained positive for calbindin D-28k. 1
In our species of interest, the dog, cat, and mouse, renal expression of calbindin was similar to that seen in other species. Within the DCTs, intermittent expression of calbindin D-28k was seen in all 3 species (Figs. 16–18). Staining was cytosolic and nuclear in the renal epithelial cells of the DCT. Some cells in the DCT showed no positive staining. It is difficult histologically to definitively differentiate the DCT from the connecting tubules, and it is certainly possible that some of the tubules with positive staining are indeed connecting tubules. The collecting ducts do not contain positively stained cells.
Tamm-Horsfall Protein
THP, a glycoprotein also called uromodulin, is the most abundant protein found in urine and is thought to have many functions. 19,37 Possible functions include regulation of renal cytokines and defense against urinary tract infection, specifically targeting type 1 fimbriated Escherichia coli. 2,29 The protein also appears to help protect against the formation of calcium oxalate crystals. 24 It has also been implicated as a contributor to tubulointerstitial renal disease and cast nephropathy in multiple myeloma patients. 37
A THP-like protein has been studied in dogs specifically because of the specie’s urinary excretion of vitamin A. 31,36 Humans reabsorb retinol, but dogs (and it is thought carnivores in general) excrete retinol and retinyl esters in their urine. 31,36 These products are hydrophilic and need a carrier in urine, which has recently been shown to be a Tamm-Horsfall-like protein. 36 In the study of this canine phenomenon, THP has been localized to the luminal cells of the TAL of the loop of Henle in the outer medulla and the cells composing the cortical DCT. 31,36 In previous reports, immunohistochemical staining was most intense at the apical margins of cells in the TAL. 31,36 Within the DCT, positive cells appeared intermittently and had a homogenous cytoplasmic expression. 31,36 Similar findings have been found in humans. 15
We obtained identical results in our dog, cat, and mouse kidneys to those described above in other species (Figs. 19–21).
Western Blot Analysis
Species antibody cross-reactivity was confirmed for our antibodies via Western blot analysis. Homogenized tissue was used from dog, cat, and mouse kidney and probed with each antibody used for IHC.
When probed with rabbit anti-human AQP1 polyclonal antibody (Chemicon), 2 species of approximately 25 and 37 kDa were found in dog, cat, and mouse tissue. These proteins’ molecular weights are consistent with the molecular weight of the unglycosylated (28 kDa) and glycosylated (35–60 kDa) forms of AQP1. These findings, coupled with IHC localization that is strongly compatible with reports in other species, give strong evidence for species-specific cross-reactivity of the anti-AQP1 antibody used in this study.
Immunoblotting using anti-human GST-α monoclonal antibody (Novacastra) confirmed the presence of 2 protein species of approximately 17 and 25 kDa in the kidney homogenate of all 3 species. Previous studies have shown a molecular weight of 26 to 31 kDa for GST-α in rabbits, rats, and humans. The 25-kDa protein identified could easily be GST-α, especially given the intense staining seen in the feline kidney that is consistent with previous reports of GST-α localization. As we were using whole homogenized renal tissue and not cell cultures, we hypothesize that some proteins may have been further cleaved, resulting in a lower molecular weight protein fragments of 17 kDa.
Immunoblotting using anti-bovine calbindin D-28k monoclonal antibody (Sigma-Aldrich) on homogenized kidney tissue from all 3 species yielded consistent results for dog, cat, and mouse tissue. A single band of 25 kDa was found for all 3 animal species. This protein is compatible with the known 28-kDa molecular weight of calbindin D-28k. The strikingly consistent IHC localization of calbindin D-28k to the DCT, as reported in various other species, gives further evidence of good species cross-reactivity of our anti-calbindin D-28k antibody.
Unfortunately, our attempts at immunoblotting were not as successful for AQP2 and THP. Despite many attempts, definitive bands could not be identified with our anti-rat AQP2 antibody probing of homogenized tissue. The failure of immunoblotting may be due to the fact that antibodies are conformationally dependent and the epitopes could vary in arrangement from more linear in tissue homogenate as compared to more complex tertiary folding in tissue sections. However, the consistent and robust localization of AQP2 to the collecting ducts of all 3 species is in strong accordance with that reported in other species and strong evidence of species antibody cross-reactivity in tissue section.
Supporting cross-reactivity of our antibody for THP is a previous study in which the same antibody was used for identifying THP in the urine of dogs via Western blotting. 36 Additionally, our IHC staining consistently localized THP to the TAL of all 3 of our species of interest.
One disadvantage to evaluating histologic sections only is that nephron segment identification is based solely on morphology. For example, the descending versus ascending thin limbs of the loop of Henle cannot be definitively identified histologically. As some of the thin limbs did not express AQP1, it can be assumed that these sections represent the ascending thin limbs, while the thin limbs that did express AQP1 are likely the DTLs, based on findings in other species. 3,25,27 Additionally, the connecting tubules cannot be differentiated reliably from the collecting duct morphologically. It is certain that the collecting ducts express AQP2, but we must suppose, on the basis of information from other species, that the connecting tubules also express some AQP2. 3,20,22,32,38,39,42 Electron microscopy with immunolabeling could help to more definitively localize and characterize the distribution of renal AQP1 and AQP2 localization in our samples.
To our knowledge, with the exceptions of THP, AQP1, and calbindin D-28k in the dog and AQP1 and AQP2 in the mouse, the proteins examined here have not been localized to the kidneys of our species. Additionally, except for THP in the dog and AQP1 and AQP2 in the mouse, these proteins have not been localized in situ within the dog, cat, and murine kidney.
We have localized 5 renal proteins in 3 different species using IHC and examination by light microscopy. AQP1 was found in the luminal epithelial cells of the PCT and the thin limbs of the loop of Henle within the dog, cat, and mouse kidney. AQP2 was expressed exclusively in the collecting ducts of all species. GST-α had rare staining in the cells of the canine PCT but was highly expressed in PST epithelium and renal transitional epithelium, with variable expression among individuals. The cat demonstrated strong expression of GST-α within the PCT, while murine glomeruli stained positively. Calbindin D-28k was localized to the DCT of all species. Finally, THP was highly expressed in the TAL of the loop of Henle, with lesser expression in the DCT, in all species.
The renal proteins identified and discussed above are quite varied in their function within the kidney. Describing their location in these additional species may aid further study of their behavior in these animal species. Additionally, some of these proteins can now serve as reliable renal nephron segment markers to help definitively identify segments of the nephron histologically. Further study of these renal proteins’ IHC localization in different diseased states is warranted. Results of such studies may prove invaluable in several ways. Foremost, in drug research and development, researchers may be able to utilize these IHC techniques to definitively identify what part of the renal tubule is affected in toxicologic studies. If the expression of these proteins in the urine of healthy and diseased animals can be studied, it may also be possible to correlate the location of renal damage to specific proteins found in urine, which could negate the need for renal biopsies in some cases. Additionally, antibodies to these proteins can be used in double-labeling IHC to localize other proteins of interest to specific regions of the nephron, without utilizing more costly and time-consuming methods. 21 With the localization of AQP1, AQP2, GST-α, calbindin D-28k, and THP in the dog, cat, and murine kidney, many interesting avenues of study are now opened.
Footnotes
Acknowledgements
We would like to thank Jessica Prenni and Hataichanok Scherman for their invaluable help with Western blotting. Also, we are appreciative of Jay Oaks for his help designing the figures seen in this article.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received funding for portions of this project from a grant awarded by Colorado State University’s College of Veterinary and Biomedical Sciences’ College Research Council.
