Abstract
Many preclinical studies in investigative dermatology are performed preferably in pigs because pig skin is more similar to human skin than is rodent skin. A frequently used model is allergic contact dermatitis (ACD); however, this T-cell—mediated skin condition so far is not well characterized in pigs. The present study is aimed at the evaluation of morphologic and immunohistochemical features of experimentally induced acute ACD in Göttingen minipigs using 2,4-dinitrofluorobenzene (DNFB) as a hapten. Eight minipigs were sensitized with 10% DNFB and challenged 2 weeks later at different sites with 1% DNFB. In addition to clinical examinations, cutaneous blood flow was quantified by laser Doppler velocimetry (Periflux PF3). These examinations were performed before challenge and 8, 24, 48, and 72 hours after challenge. Skin biopsies were taken at the same time points, fixed, sectioned, and stained with Giemsa for histologic evaluation, or with mouse anti-swine monoclonal antibodies (CD1, CD2, CD4, CD5, CD8, CD25, CD45, MHCII) and with one mouse anti-human monoclonal antibody (CD62E) cross-reacting with swine for immunohistochemical evaluation. Positively stained cells were counted per square millimeter of epidermis and dermis by using a video image analyzing system (Videoplan Kontron). Erythema and cutaneous blood flow peaked at 24 hours. The major epidermal changes most pronounced at 48 hours were acanthosis, spongiosis, intracellular edema, exocytosis, and abscesses mainly containing neutrophils and mononuclear cells (MNC). Perivascular infiltrates of MNC as well as neutrophils and eosinophils were the most significant dermal changes, with peak levels at 24–48 hours. In biopsies taken before challenge, CD1+ dendritic cells were found in similar numbers and locations as MHCII+ cells in the epidermis. In the epidermis the maximum CD1+ cell decrease occurred at 24 hours whereas in the dermis the maximum increase in CD1+ stained cells was seen at 72 hours. The dermal infiltrate (CD2+, CD5+, CD25+, and CD45+) was most dense at 48 hours. Between 8 and 48 hours more CD4+ were present than CD8+ cells, whereas at 72 hours CD4+ and CD8+ cells were similar in numbers. These findings closely resemble changes in human ACD. Therefore, DNFB-induced ACD in Göttingen minipigs is considered to be an appropriate animal model to study immunopathologic mechanisms and pharmacologic intervention.
Keywords
Preclinical research in biomedical science uses laboratory animals to profile new compounds for possible uses as drugs. Therefore, laboratory animals serve either as models for the assessment of pharmacodynamic effects or, in advanced stages of drug development, for the identification of untoward effects (toxicities) of drug candidates. Therefore, the results obtained from animal pharmacology or animal toxicology studies should be reliably predictive for the situation in humans. In general, this applies to animal models that resemble human conditions. For example, experimentally induced diseases in animals that closely mimic diseases in humans are trustworthy models. Allergic contact dermatitis (ACD) is such an example because humans and animal species such as cats and dogs can suffer from similar cutaneous hypersensitivity reactions and ACD can be induced experimentally in humans and animals. 52 Therefore, numerous investigations on experimentally induced ACD are performed by investigative dermatologists using mice in order to explore immunopathogenetic mechanisms and to obtain new insights into the prophylaxis or treatment of this skin condition, and possibly in other T-cell–mediated inflammatory skin diseases. 19 The use of mice has numerous advantages over using larger animals, which in turn has resulted in many available investigative tools for mice that are not accessible to this extent for studies in other species. However, with regard to the morphology and physiology as well as to the response to epicutaneously applied drugs, murine skin is very different from human skin, whereas pig skin is more similar. 3 29 31 38 43 For example, topical cyclosporine A is highly active in rodent ACD but inactive in domestic pigs and humans because permeability of rodent skin is higher than in humans or swine. 25 37 51 For previously mentioned reasons domestic pigs or minipigs are increasingly used for preclinical studies in investigative dermatology, which in turn has resulted in increased availability of new investigative tools such as monoclonal antibodies to cell surface markers or cytokines. 17 24 34 40 46 49 55 59 61 In addition, the possible use of genetically altered pigs as organ donors for humans created tremendous interest in swine immunology as an investigative tool.
The present study is an effort to characterize clinical, morphologic, and immunohistochemical features of experimentally induced ACD in Göttingen minipigs and to compare the findings with changes in human ACD. Göttingen minipigs have been used because they are biologically well characterized, have a pale, lightly pigmented skin, and are convenient to handle. 18 Additionally, they are bred commercially on a large scale for use in biomedical research.
This similarity of skin anatomy, physiology, and immunology as well as the availability of immunologic reagents allowed the development and characterization of this appropriate animal model for studying immunopathologic mechanisms and pharmacologic intervention. 36 37 39 The study is also a contribution to the immunohistochemical characterization of normal skin of Göttingen minipigs. The test sites of 2,4-dinitrofluorobenzene (DNFB)-induced acute ACD were studied by clinical examination, measurement of microvascular blood flow, and by histological, immunohistochemical, and histomorphometric analyses 8, 24, 48, and 72 hours after elicitation of the hypersensitivity reaction.
Materials and Methods
Laboratory animals
Eleven female Göttingen minipigs were obtained from Ellegaard Göttingen Minipigs Aps (Dalmose, Denmark) at age 3 months. The animals were maintained at the Novartis Research Institute, Vienna, Austria, individually in pens with 2-m2 floor area in a separated room under conventional standardized conditions (22 ± 2 C room temperature, 50 ± 10% relative humidity, 12 hour day (150 lux)/12 hour night, and 10 changes of air/hour). Pelleted food (SMP MOD (E) PL produced by Special Diets Services, Witham Essex, UK) was given in amounts of 280–480 g per day per animal. At the time of experimentation the animals were approximately 6 months old and had a body weight of 15.0–18.3 kg. Eight animals were used for studies of ACD; three animals remained unsensitized and were used as controls to examine possible changes caused by the hapten used for the elicitation of the hypersensitivity reactions in the eight sensitized animals. For experimental manipulations the animals were transferred into hammocks and handled without sedation except for the collection of biopsies when the animals were sedated with ketamine (20 mg/kg IM) and xylazine (2 mg/kg IM).
Experimentation license
The conditions of housing and care of the animals as well as the performance of the experiments were in agreement with the Austrian law of animal experimentation. The study protocol was approved by the Bundesministerium für Wissenschaft, Forschung und Kunst (GZ 68.205/63-Pr/4/95).
Induction of ACD
On day 1, the animals were sensitized with 10% DNFB (Merck, Darmstadt, Germany). The hapten was dissolved in acetone:DMSO:olive oil (5:1:3, v/v/v) and applied to four areas on the outer aspect of the pinnae and medial aspect of the thigh, bilaterally (in total 500 µl). Challenge reactions were elicited with 40 µl of DNFB (1%, prepared in acetone:olive oil (8:1.9; v/v) applied topically to each of the four test sites (4 × 2 cm) arranged bilaterally on the dorsolateral area of the shaved back. DNFB at 1% has been shown in preliminary studies to be the minimal concentration to cause regularly gross inflammatory changes in sensitized Göttingen minipigs (unpublished observation). The test sites were challenged either 8, 24, 48, or 72 hours before biopsy for simultaneous collection of all tissue samples in order to avoid distress to the animals by repeated anesthetic procedures. Test sites from different areas were used for each time point.
Examination of the test sites
Before challenge and immediately before collection of the skin biopsies (i.e., 8, 24, 48, and 72 hours after challenge) the test sites were examined clinically. Intensity and extent of inflammatory erythema as well as swelling and induration were scored with 0 (normal finding) to 4 (severe) allowing a combined maximal score of 12 per designated site. In addition, dermal microvascular blood flow was noninvasively assessed with the flowmeter PeriFlux PF3 (Perimed KB, Stockholm, Sweden), utilizing the Doppler shift, that is the frequency change that laser light undergoes when interacting with objects (blood cells) in motion. For histomorphologic examination three adjacent biopsies (6 mm in diameter) were collected from each test site. One sample was fixed in 10% neutral buffered formalin, trimmed, embedded in methacrylate (Kulzer & Co, Wehrheim, Germany), sectioned at 2 µm, and stained with Giemsa (Merck). The other two samples were immersed in O.C.T. compound (Miles Laboratories, Elkhart, IN, USA) and frozen in 2-methylbutane cooled in liquid nitrogen and stored at −80 C until 5-µm frozen sections could be prepared for immunostaining. Frozen sections were mounted on poly
List of monoclonal mouse anti-swine antibodies used for phenotyping of cells in skin sections.

CD = Cluster designation.
A.S. = Armin Saalmüller, Bundesuntersuchungsanstalt für Viruskrankheiten bei Haustieren, 72001 Tübingen, Germany; K.H. = Karin Haverson, Division of Molecular and Cellular Immunology, Department of Veterinary Clinical Sciences, University of Bristol at Langford, Langford BS18 7DY, UK.
Mouse anti-human (cross-reactive).
Histomorphologic evaluation of Giemsa-stained sections
For comparative evaluation and semiquantitative assessment of the histopathologic changes and cellular infiltration of the epidermis and the dermis, a catalogue of changes (listed in Tables 2, 3) and a five-point scoring procedure were used, where 0: no changes; 1: minimal changes, isolated inflammatory cells in few microscopic fields (0.035 mm2 at 250×); 2: mild changes, few inflammatory cells in several microscopic fields; 3: moderate and focal changes, few to numerous inflammatory cells in nearly all microscopic fields; 4: severe and extended changes, numerous inflammatory cells in almost all microscopic fields; 5: very severe and extended changes, inflammatory cells en masse in all visual fields. One section from each animal at each time point (in total eight per time point) was examined.
Summary of pathologic changes in normal control sites (0 hours) and in 2,4-dinitrofluorobenzene (DNFB) allergic contact dermatitis (ACD) sites 8, 24, 48, and 72 hours after elicitation of ACD: changes semiquantified by scores† (median (range), n = eight per time point).
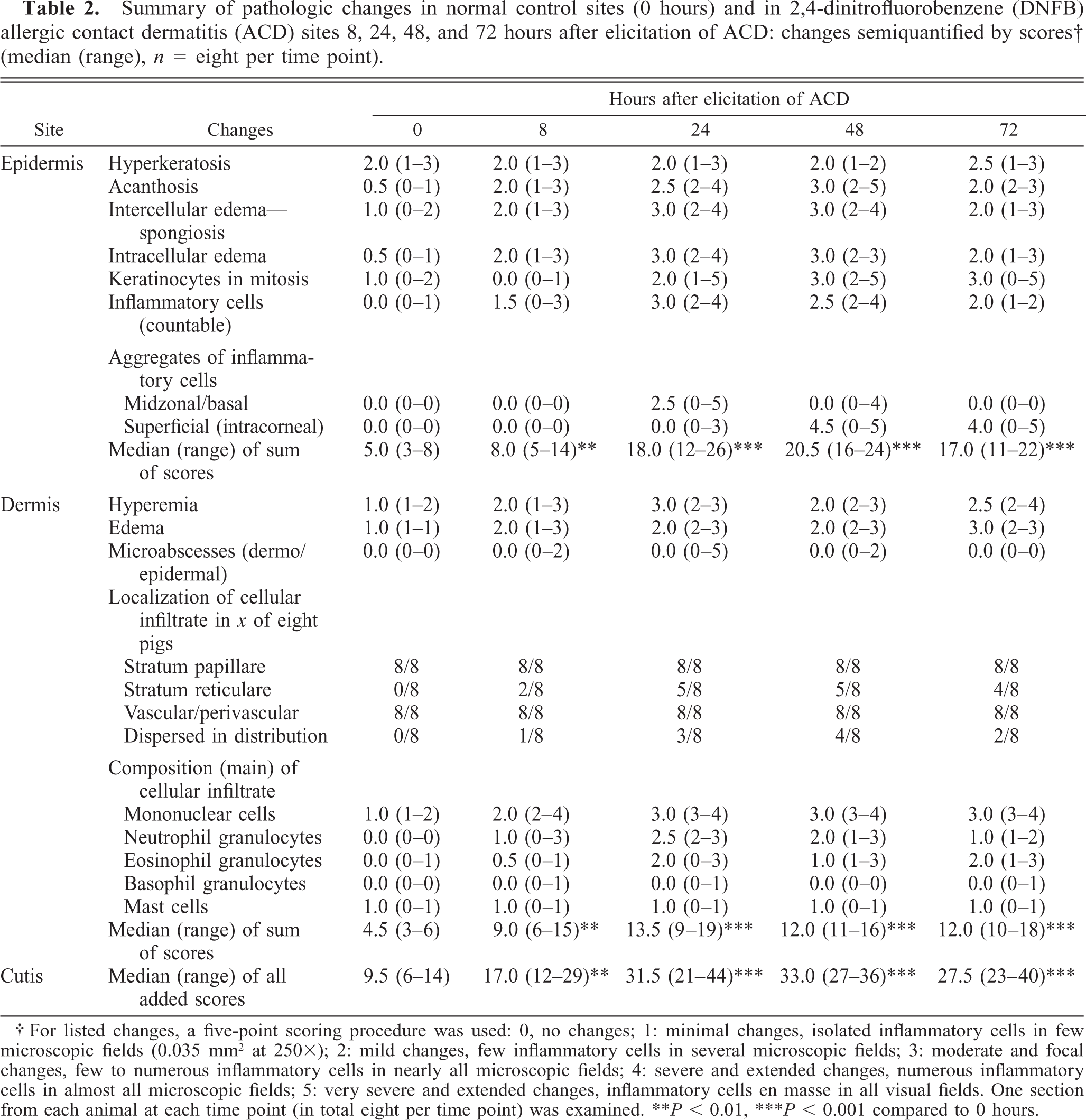
For listed changes, a five-point scoring procedure was used: 0, no changes; 1: minimal changes, isolated inflammatory cells in few microscopic fields (0.035 mm2 at 250X); 2: mild changes, few inflammatory cells in several microscopic fields; 3: moderate and focal changes, few to numerous inflammatory cells in nearly all microscopic fields; 4: severe and extended changes, numerous inflammatory cells in almost all microscopic fields; 5: very severe and extended changes, inflammatory cells en masse in all visual fields. One section from each animal at each time point (in total eight per time point) was examined. ∗∗P, 0.01, ∗∗∗P, 0.001 compared to 0 hours.
Summary of pathologic changes in normal control sites (0 hours) and 2,4-dinitrofluorobenzene (DNFB) control sites 8, 24, 48, and 72 hours after application of 1% DNFB to unsensitized animals: changes semiquantified by scores∗ (median (range), n = three per time point).
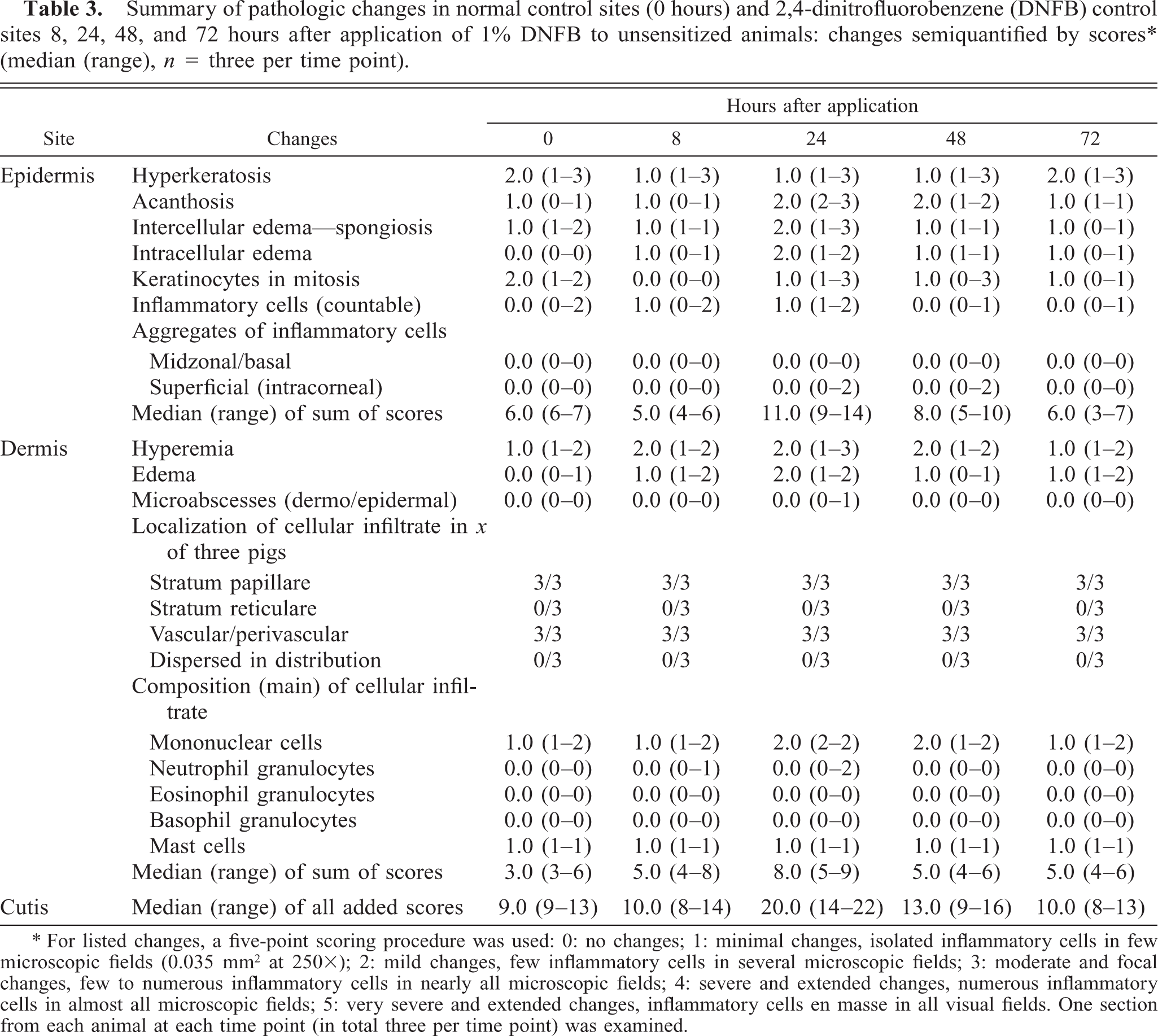
For listed changes, a five-point scoring procedure was used: 0: no changes; 1: minimal changes, isolated inflammatory cells in few microscopic fields (0.035 mm2 at 250X); 2: mild changes, few inflammatory cells in several microscopic fields; 3: moderate and focal changes, few to numerous inflammatory cells in nearly all microscopic fields; 4: severe and extended changes, numerous inflammatory cells in almost all microscopic fields; 5: very severe and extended changes, inflammatory cells en masse in all visual fields. One section from each animal at each time point (in total three per time point) was examined.
Histomorphometric analyses
Cell counts in formalin-fixed or cryosections were performed at 250× magnification with an image analyzer (Videoplan, Kontron, Munic, Germany). Eosinophils and mast cells (only intact cells) in Giemsa-stained sections were counted in a dermal area of 3.5 mm2 (approximately 100 microscopic fields) and their density per square millimeter was reported as 0 (0–3 cells), 1 (3–15 cells), 2 (15–30 cells), 3 (30–100 cells), 4 (100–500 cells), and 5 (>500 cells). The immunohistochemically stained cells in the epidermis and in the dermis were separately counted. In each section an epidermal area of approximately 0.4 mm2 and a dermal area of 1.8 mm2 was examined (i.e., about 12 epidermal and 50 dermal microscopic fields in each section). CD1+ cells were also counted in the epidermal area superlying a 1 mm epidermal basal layer.
Controls
For evaluation of the histologic baseline status one skin biopsy was collected and prepared as outlined from each animal from the dorsolateral area of the back 4 weeks before the principal study. These samples have been named normal control sites. In addition, three animals that had not been sensitized to DNFB were treated topically with 1% DNFB and examined in the same way as the sensitized and challenged animals in order to assess the primary irritative potential of 1% DNFB macroscopically and histologically in normal animals. These test sites have been named DNFB-control sites in contrast to the DNFB-ACD sites representing sites with DNFB-induced ACD. The antibodies against the cell surface markers have been tested previously on sections of lymph node and spleen samples from Göttingen minipigs by avidin–biotin complex (ABC) staining to ensure reactivity and specificity. Skin sections were also incubated first with irrelevant isotype-matched antibodies for controls. The microscopic examination was performed in a coded fashion without knowledge of the origin and time point of the biopsy.
Statistics
In order to achieve normal distribution and homogeneous variances the data were logarithmically transformed before statistical analysis. Data from DNFB-control sites and DNFB-ACD sites at different time points were compared with the corresponding 0-hour values (normal control) using paired t-tests. Resulting P values were adjusted for multiple comparisons with the method of Hochberg–Bonferroni.
Results
Gross changes
Dermatitis due to contact hypersensitivity was observed at an earliest of 8 hours after challenge with clinical signs of erythema and swelling. The changes were initially patchy and mild, in contrast to full development where all test sites were uniformly and intensively reddened, elevated, and edematous or slightly indurated. The changes peaked at 24 hours after challenge (Fig. 1); thereafter regression occurred as shown by scores in Fig. 2. Three days after challenge the DNFB-ACD sites were flat again; however, erythema was still pronounced. Vesicles, crusts, or other secondary lesions were not observed. Changes in measured microvascular blood flow coincided with the gross changes (Fig. 3). DNFB-control sites (i.e., sites treated with 1% DNFB in nonsensitized animals) showed a mild transient erythema without swelling only in one of three animals (Fig. 2). The blood flow peaked in these DNFB-control sites 8 hours after treatment (Fig. 3).
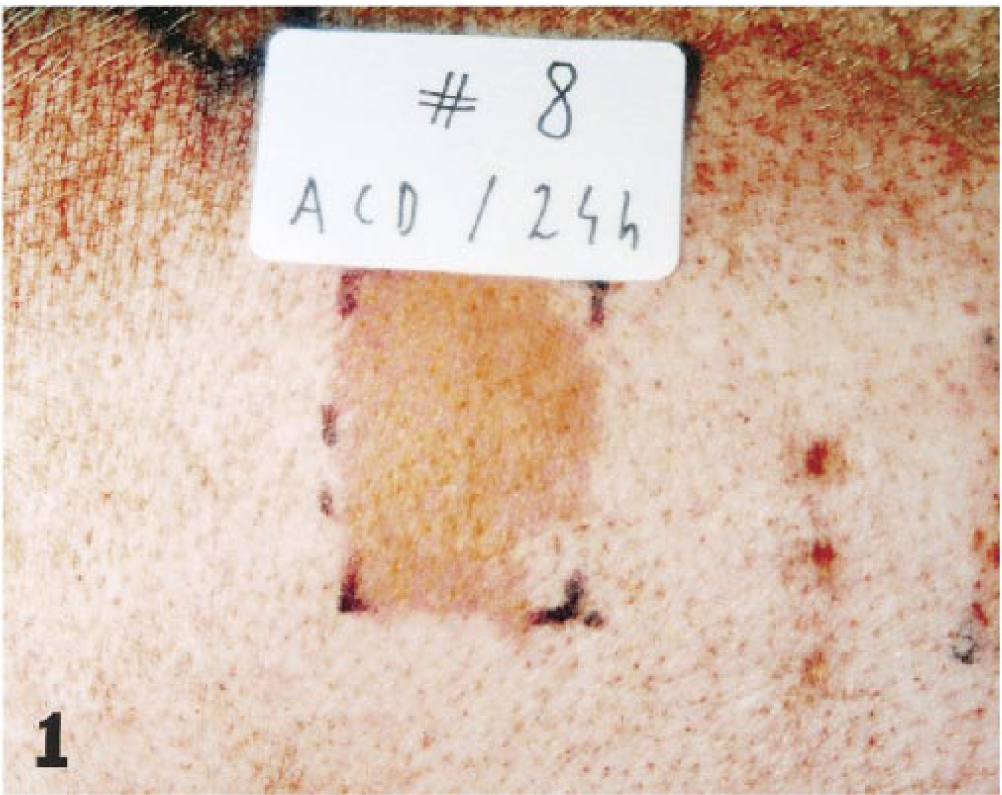
Gross changes of allergic contact dermatitis on the dorsolateral back of pig No. 8 24 hours after elicitation with 2,4-dinitrofluorobenzene: severe erythema, slight edema.
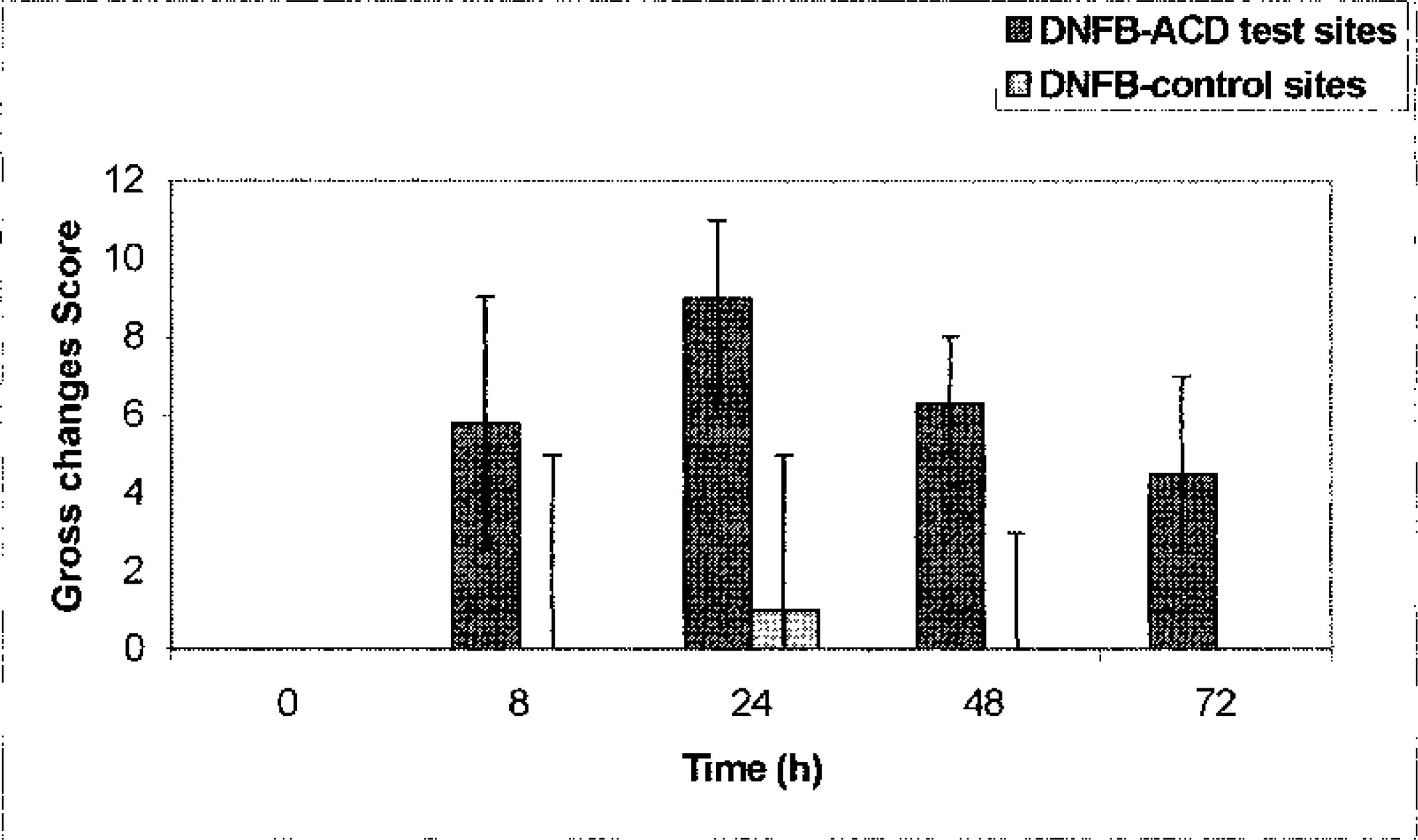
Changes in gross lesions of 2,4-dinitrofluorobenzene (DNFB) allergic contact dermatitis sites (▪) and in DNFB-control sites (;x9). 0 before challenge. Intensity and extent of inflammatory erythema as well as the swelling and induration were scored with 0 (normal finding) to 4 (severe) allowing a combined maximal score of 12 per site. Values: median (range).
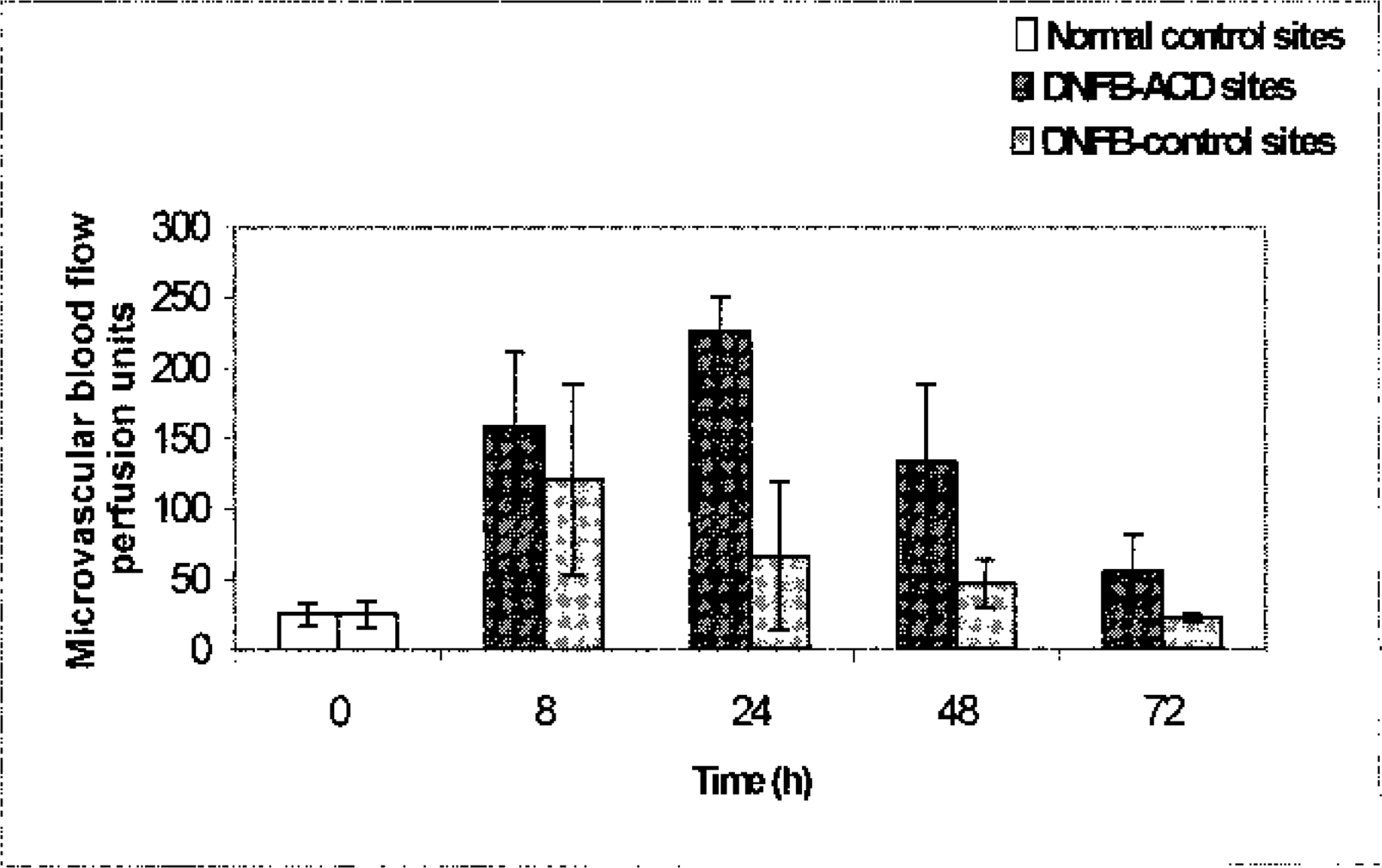
Changes in dermal microvascular blood flow in 2,4-dinitrofluorobenzene (DNFB) allergic contact dermatitis sites (▪) and in DNFB-control sites (;x9). 0: before challenge (normal control sites, □). Values: mean (perfusion units) ± SD.
Histopathology
In sections that were prepared for the assessment of the base line status (normal controls) in animals used for ACD (Table 2, column 0 hours), signs of mild scattered inflammatory reactions occasionally have been observed; in particular foci of keratinocytes with intracellular edema, hyperemia, and tiny, superficial perivascular infiltrates composed of mononuclear cells have been seen. The scoring of the epidermal and dermal findings resulted in a median score of 9.5 (range: 6–14). The changes seen in DNFB-ACD sites 8 hours after elicitation of the hypersensitivity reaction consisted of a mild hyperplastic epidermis with spongiosis and inflammatory cells in the lower layers. In the uppermost portion of the dermis, vasodilatation, perivascular edema, and cellular infiltration were present, overall scored at 17.0 (12–29). Subsequently, these changes increased and reached maximal intensity 24–48 hours after challenge (scores 31.5 [21–44] and 33.0 [27–36], respectively). The epidermis showed pronounced acanthosis associated with many mitotic or vacuolated keratinocytes and moderately heavy spongiosis. The inflammatory cells evident throughout the epidermis were still countable during the first 24 hours after challenge. However, by 48 hours they increased to uncountable numbers and were tightly packed in aggregates in the upper epidermis or in intracorneal abscesses in most sections. Dermal changes had also increased with neutrophil microabscess with central leukocytoclasia frequently present at the dermoepidermal junction. Perivascular and scattered inflammatory cells were also present in the reticular area of the dermis. The inflammatory cells were mainly mononuclear cells with some neutrophils and eosinophils. The proportion of granulocytes was greatest at 24 hours. The mean number of eosinophils per square millimeter of dermis increased from 2.0 ± 1.7 in normal controls before challenge to 5.3 ± 4.9 (8 hours), 27.5 ± 18.5 (24 hours), 17.1 ± 10.3 (48 hours), and 20.4 ± 13.6 (72 hours after challenge). Most of the eosinophils were intact and showed no signs of degranulation. No changes in numbers were seen in DNFB-control sections, which contained 0.64 ± 0.5 eosinophils. Compared to the infiltration of eosinophils, which peaked at 24 hours, the number of mast cells per square millimeter of dermis remained unchanged and ranged from 4.6 ± 2.2 (before challenge) to 5.4 ± 2.2 3 days after challenge (data not shown in Table 2). Basophils were very rare and observed only in sections from 24-hour DNFB-ACD sites. By 72 hours a general tendency towards regression already occurred, which resulted in lower score values. Small intracorneal abscesses, acanthotic changes of the epidermis, and a reduction in cellular infiltration in the superficial and deep dermis indicated the preceding acute inflammation. Figures 4–6 show representative changes 8 and 24 hours after elicitation of ACD.
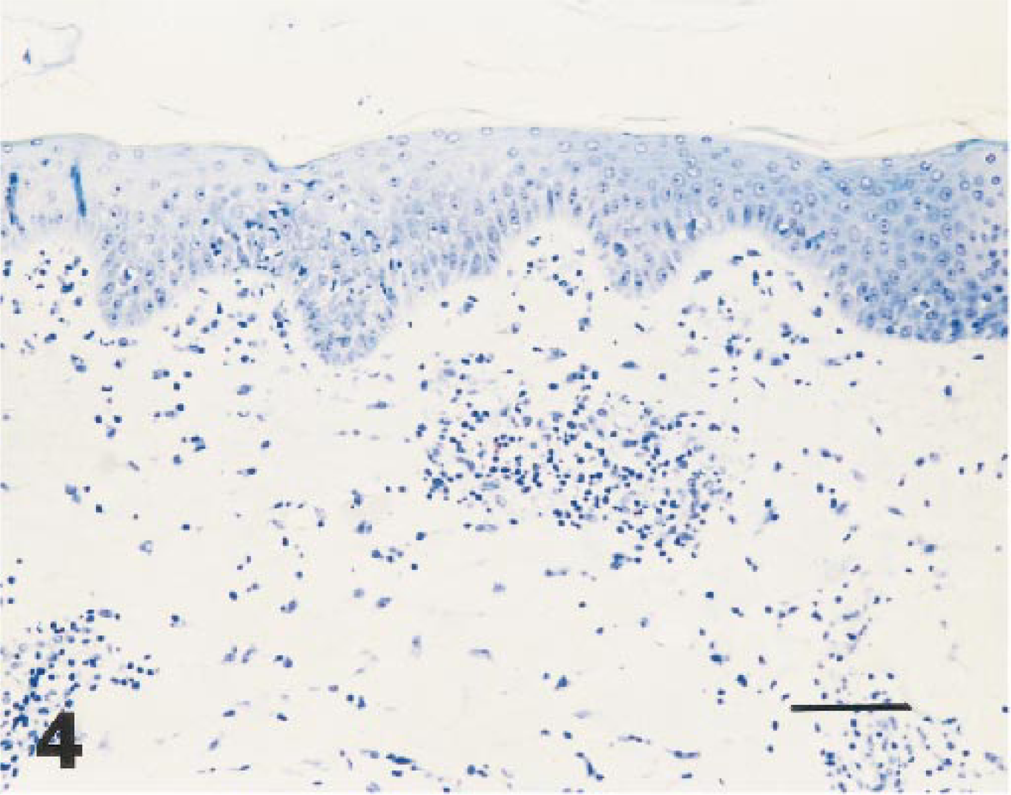
Skin; 2,4-dinitrofluorobenzene allergic contact dermatitis site, 8 hours after elicitation: mild exocytosis, spongiosis, and perivascular infiltrate. Semithin section. Giemsa staining. Bar = 28 µm.
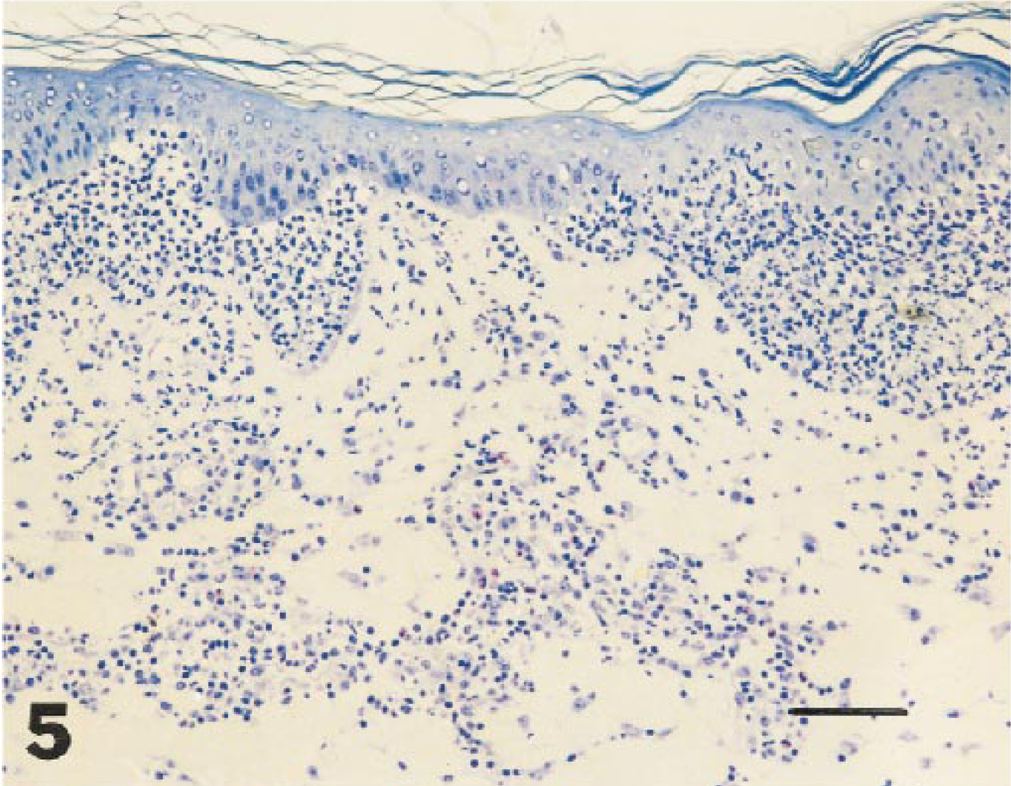
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 24 hours after elicitation: dense inflammatory cell infiltrate in the dermis (mainly perivascular) and epidermis (basal). Semithin section. Giemsa staining. Bar = 28 µm.
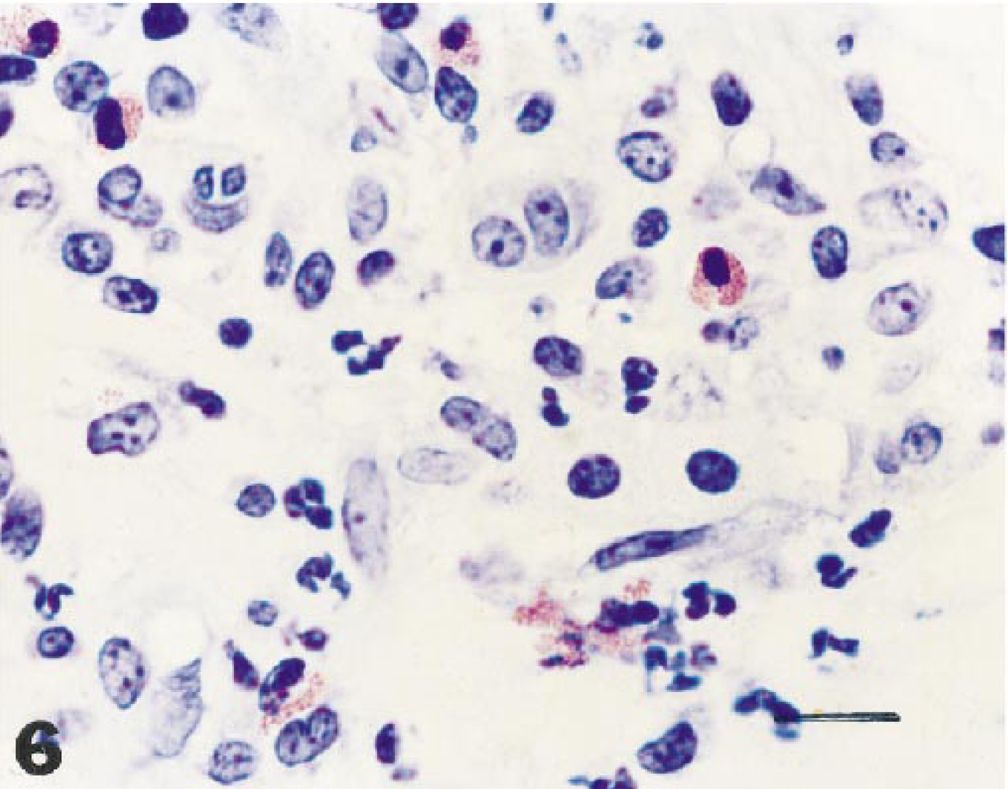
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 24 hours after elicitation, detail from Fig. 5: neutrophils, eosinophils, and lymphocytes predominate in the inflammatory infiltrate. Semithin section. Giemsa staining. Bar = 4.4 µm.
The three animals used for the assessment of the primary irritative potential of 1% DNFB (DNFB-control sites) revealed the same baseline status as the eight animals used for the examination of ACD (Table 3). Although only three sectioned specimens were used for each time point, microscopic examination revealed some changes that were very similar in quality to the hypersensitivity reaction. However, the changes clearly differed in intensity and time course and were not statistically significantly different from normal control sites. The primary irritant contact dermatitis caused by 1% DNFB was much milder and reduced in duration compared to ACD elicited also by 1% DNFB, had its greatest development after 24 hours, and had resolved by 72 hours as shown simplified by scores in Fig. 7.
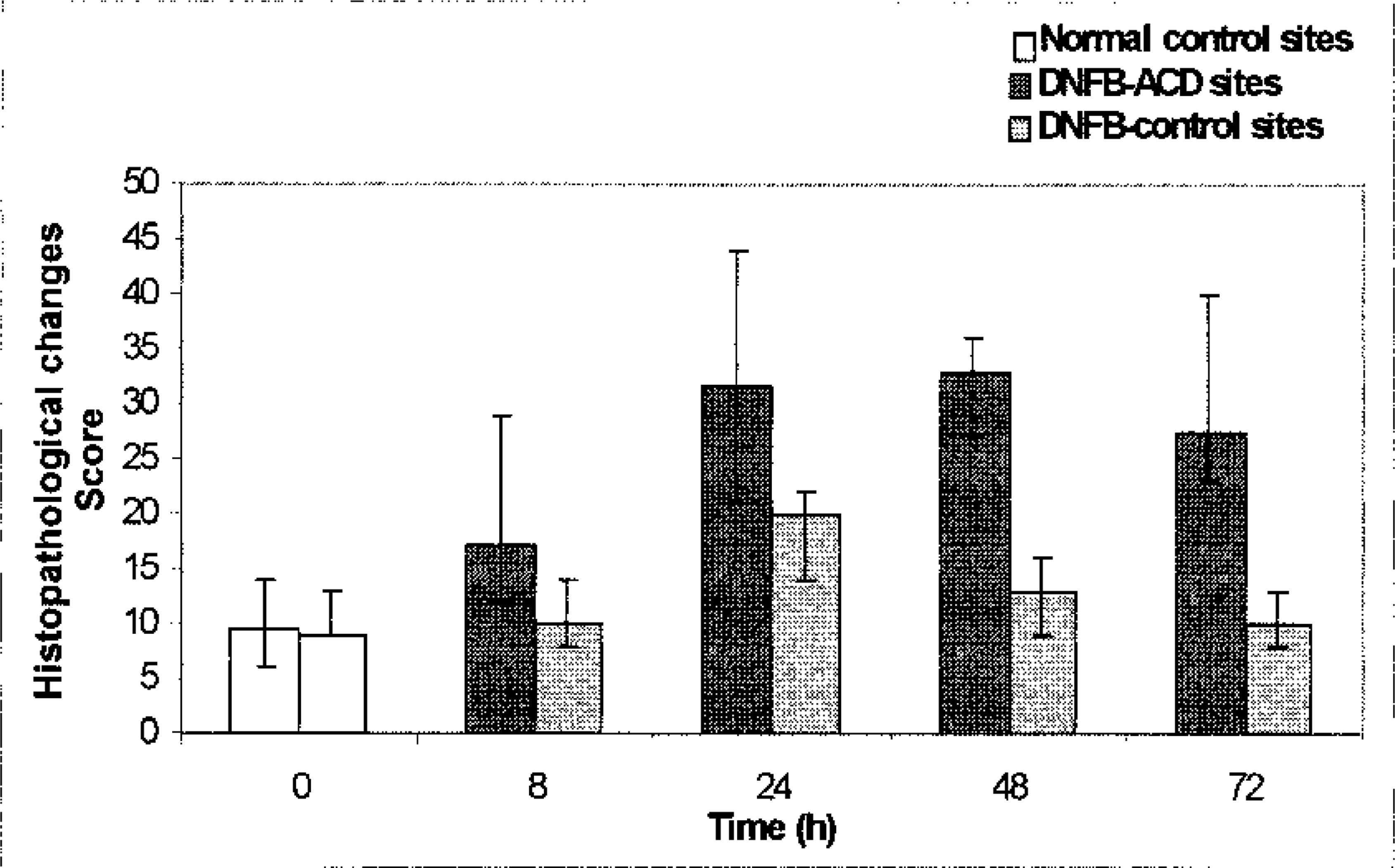
Changes in histopathologic epidermal and dermal lesions (quantified by five-point scoring procedure) in 2,4-dinitrofluorobenzene (DNFB) allergic contact dermatitis sites (▪) and in DNFB-control sites (;x9). 0: before challenge (normal control sites, □), Values: median (range).
Immunochemistry
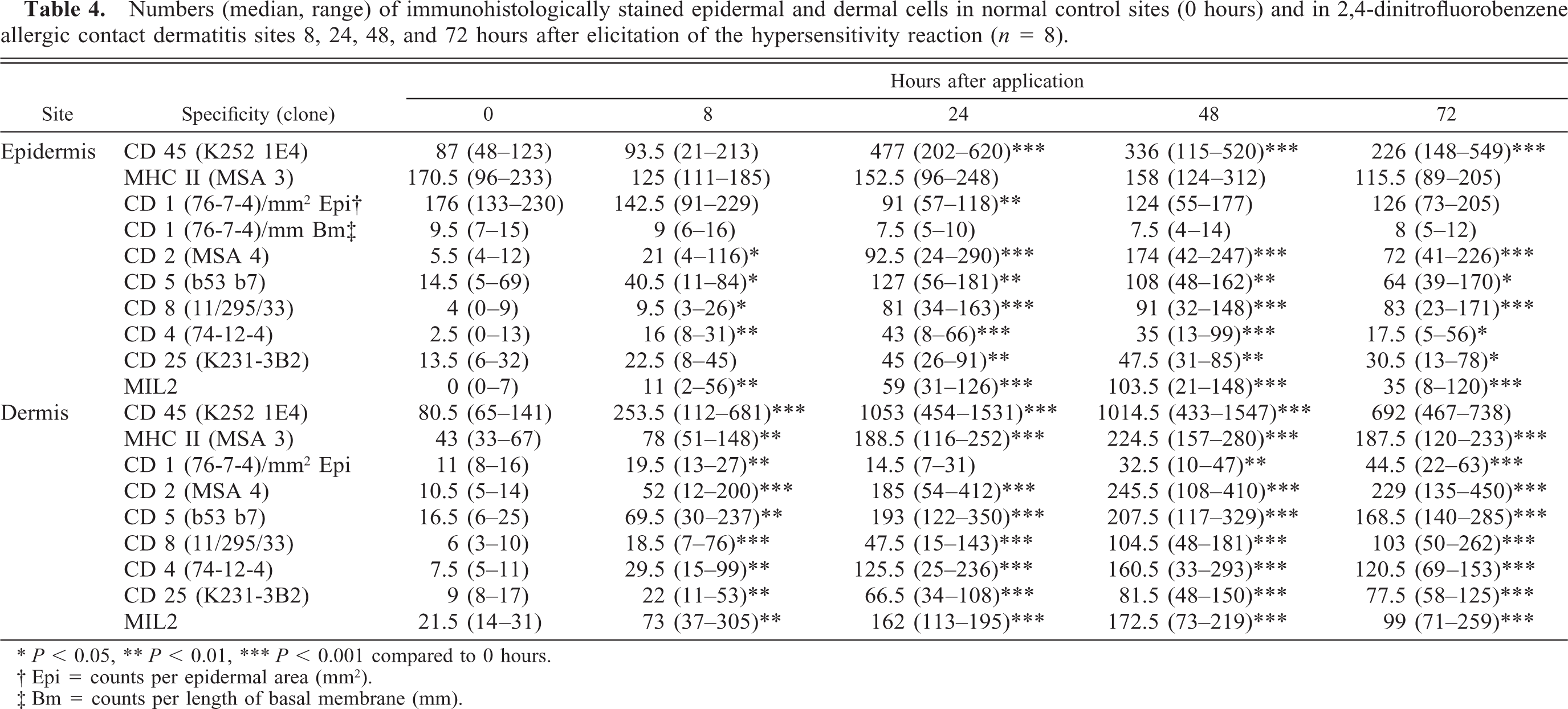
In specimens collected for determination of the baseline status (normal control sites), CD1-, MHC class II-, CD2-, CD4-, CD5-, CD8-, CD25-, CD45-, and MIL2-positive cells were observed in the basal and suprabasal layers of the epidermis. CD1+ and MHC class II+ were similar in location and number. In the dermis the most abundant cells were CD45+ cells, followed by MHC II+ cells, MIL2+, CD5+, and CD1+ cells, which were less dendritic than the epidermal CD1+ cells. Similar to the epidermal condition, the number of dermal CD5+ cells was higher than those of the CD2+ cells and the sum of CD4+ and CD8+ cells; CD4+ and CD8+ cells were similar in frequency. CD25+ cells were also present. In general, the frequency of immunohistochemically stained cells was higher in the upper dermis than in the deeper dermis and individually very different as can be deduced from the large range of values shown in Tables 4 and 5 (column 0 hours).
Numbers (median, range) of immunohistologically stained epidermal and dermal cells in normal control sites (0 hours) and in 2,4-dinitrofluorobenzene allergic contact dermatitis sites 8, 24, 48, and 72 hours after elicitation of the hypersensitivity reaction (n = 8).
P, 0.05, ∗∗P, 0.01, ∗∗∗P, 0.001 compared to 0 hours.
Epi = counts per epidermal area (mm2).
Bm = counts per length of basal membrane (mm).
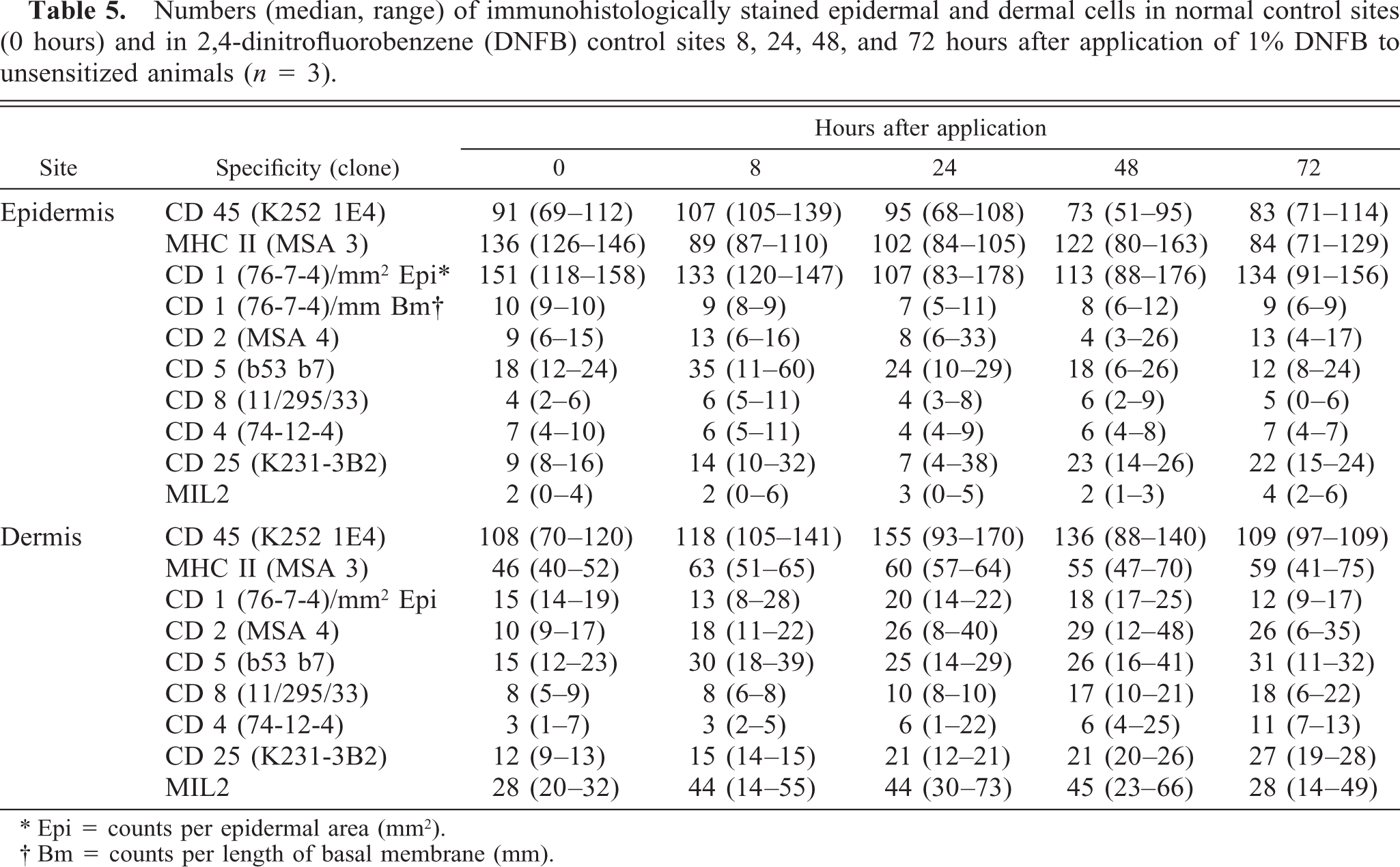
Numbers (median, range) of immunohistologically stained epidermal and dermal cells in normal control sites (0 hours) and in 2,4-dinitrofluorobenzene (DNFB) control sites 8, 24, 48, and 72 hours after application of 1% DNFB to unsensitized animals (n = 3).
Epi = counts per epidermal area (mm2).
Bm = counts per length of basal membrane (mm).
Immunohistochemical characterization of cells in the epidermis of DNFB-ACD sites, revealed a decrease in CD1+ cells. The number was lowest 24 hours after challenge (−48%, P < 0.01, cells per mm2 epidermis) and still below baseline values at 72 hours (−28%). The numbers of class II+ cells were similar in all biopsies. A clear and statistically significant increase in CD45+, CD2+, CD5+, CD4+, CD8+, CD25+, and MIL2+ cells was observed, which, in general, peaked from 24 to 48 hours and declined 72 hours after elicitation. Only numbers of CD8+ cells plateaued from 24 to 72 hours. The changes related to the baseline values were in CD2+ 32-fold at 48 hours, in CD8+ 20- to 23-fold at 24–72 hours, and in CD4+ cells 17-fold at 24 hours. CD4+ cells were clearly less frequent in the epidermis than CD8+ cells 24–72 hours after challenge. CD5+ and CD25+ cells increased by a factor of nine and four, respectively. Cells positive for CD45 increased fivefold (maximum at 24 hours; Table 4, Fig. 8). In comparison, infiltration of the epidermis was most pronounced 24–48 hours after challenge and dermal infiltration was most severe from 48 to 72 hours after elicitation.
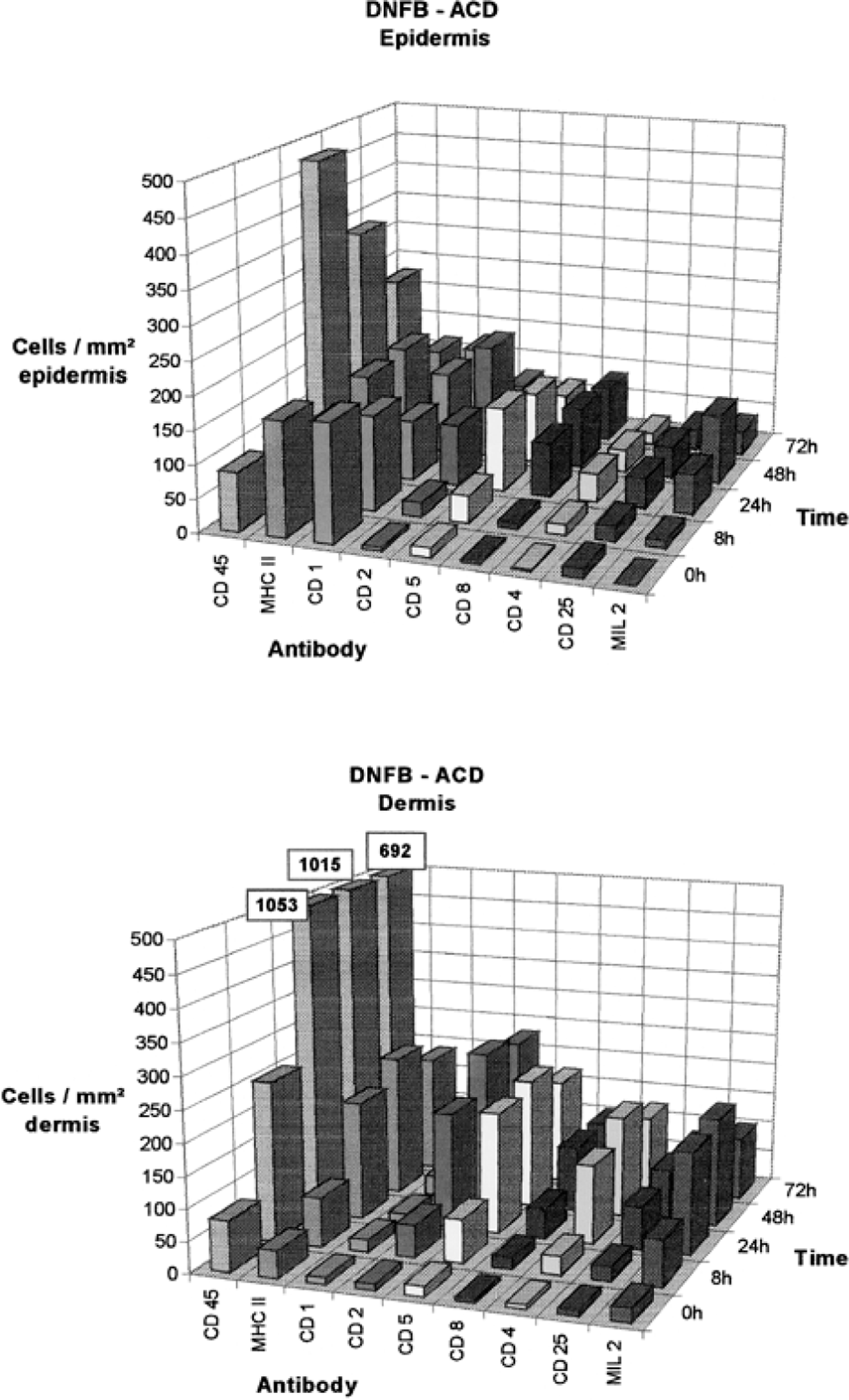
Summary of changes in epidermal and dermal cells expressing the markers CD45, MHC class II, CD1, CD2, CD5, CD8, CD4, CD25, or MIL2 in sections from 2,4-dinitrofluorobenzene allergic contact dermatitis sites: histomorphometric determination of positively stained cells at various time points after elicitation; 0: before challenge (normal control sites). Median of cells per square millimeter of epidermis (upper panel) and dermis (lower panel).
Numbers of dermal CD1+ cells were similar to normal controls 24 hours after challenge and increased by factors of two (8 hours), three (48 hours), and four (72 hours), respectively. MHC class II+ cells showed an increase and were most frequent at 48 hours (fivefold). Most CD2+ (23-fold increase) and CD5+ cells (13-fold increase) were observed at 48 hours. CD4+ and CD8+ cells increased by factors of 17–21 but differed in infiltration kinetics. CD4+ cells preceded CD8+ cells in infiltration and peaked at 48 hours, whereas infiltration of CD8+ cells was most pronounced at 48 and 72 hours. Infiltration kinetics of CD25+ cells were similar to CD8+ cells (ninefold at 48 and 72 hours). MIL2+ and CD45+ cells were most frequent at 24 and 48 hours and increased by factors of 8 and 13, respectively (Table 4, Fig. 8).
In DNFB-control sites of the three unsensitized animals treated with 1% DNFB no statistically significant changes in numbers of the examined cell types were observed. The increase in labeled epidermal or dermal cells was only up to fourfold in median values at maximum compared to normal control sites. Tendencies towards increase were observed in epidermal CD5+ cells (8 hours) and CD25+ cells (48 hours, 72 hours), although numbers of CD45+ cells were very similar at all time points. In the dermis, the CD45+ cells (24 hours, 48 hours), CD2+ cells (24–72 hours), CD5+ cells (8–72 hours), CD8+ cells (48 hours, 72 hours), CD25+ cells (24–72 hours), and MIL2+ cells (8–24 hours) cells appeared to be slightly increased (Table 5, Fig. 9).
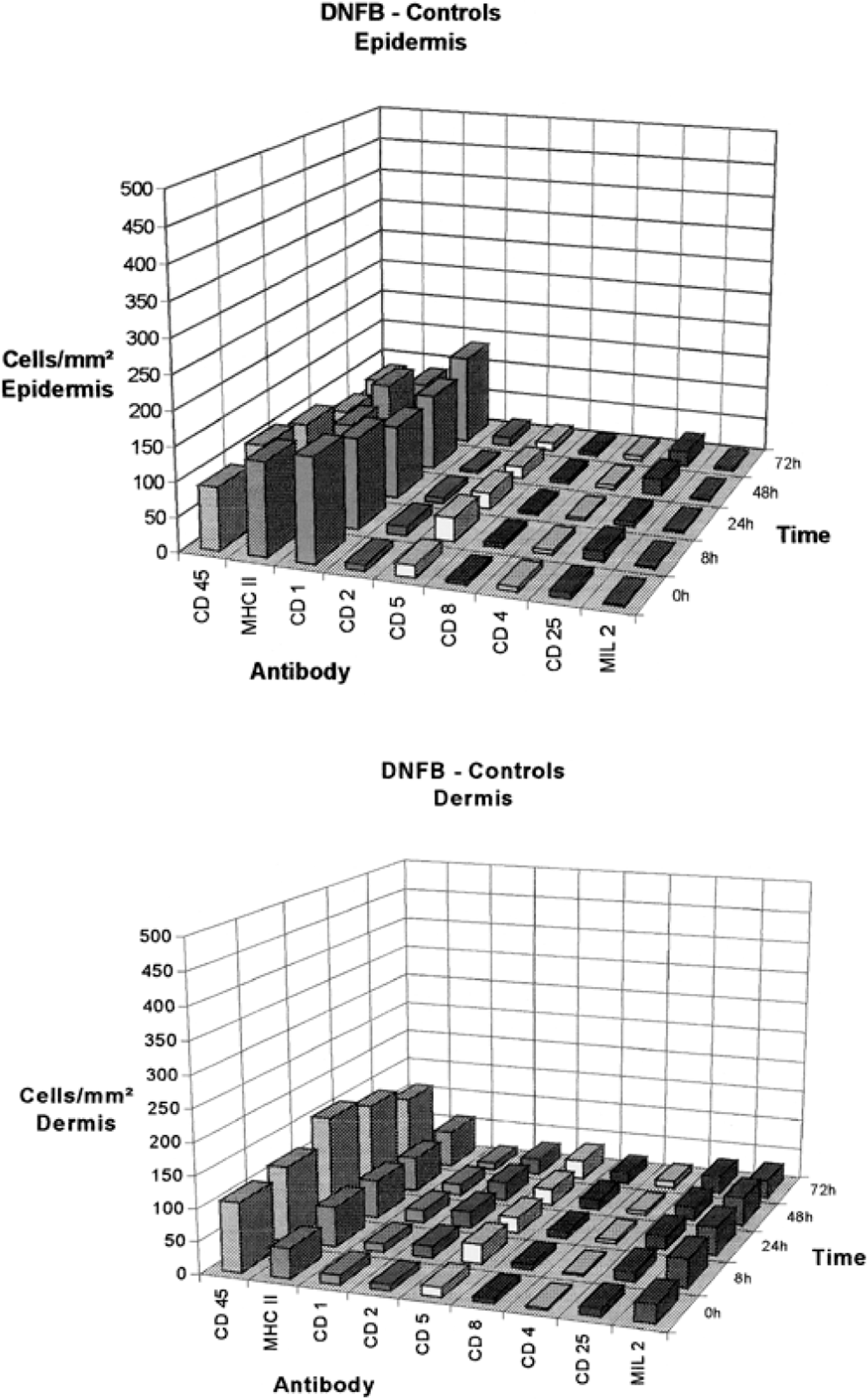
Summary of changes in epidermal and dermal cells expressing the markers CD45, MHC class II, CD1, CD2, CD5, CD8, CD4, CD25, or MIL2 in sections from 2,4-dinitrofluorobenzene (DNFB) control sites: histomorphometric determination of positively stained cells at various time points after application of DNFB; 0: before application (normal control sites). Median of cells per square millimeter of epidermis (upper panel) and dermis (lower panel).
Staining for the endothelial and inducible adhesion molecule E-selectin showed some increase in intensity in biopsies collected later than 8 hours after application of 1% DNFB, both in DNFB-ACD and DNFB-control skin samples. Representative examples of immunohistochemical staining (CD4+, CD5+, CD8+, CD25+, CD45+, and E-selectin+) of sections from DNFB-ACD sites are shown in Figs. 10–15.
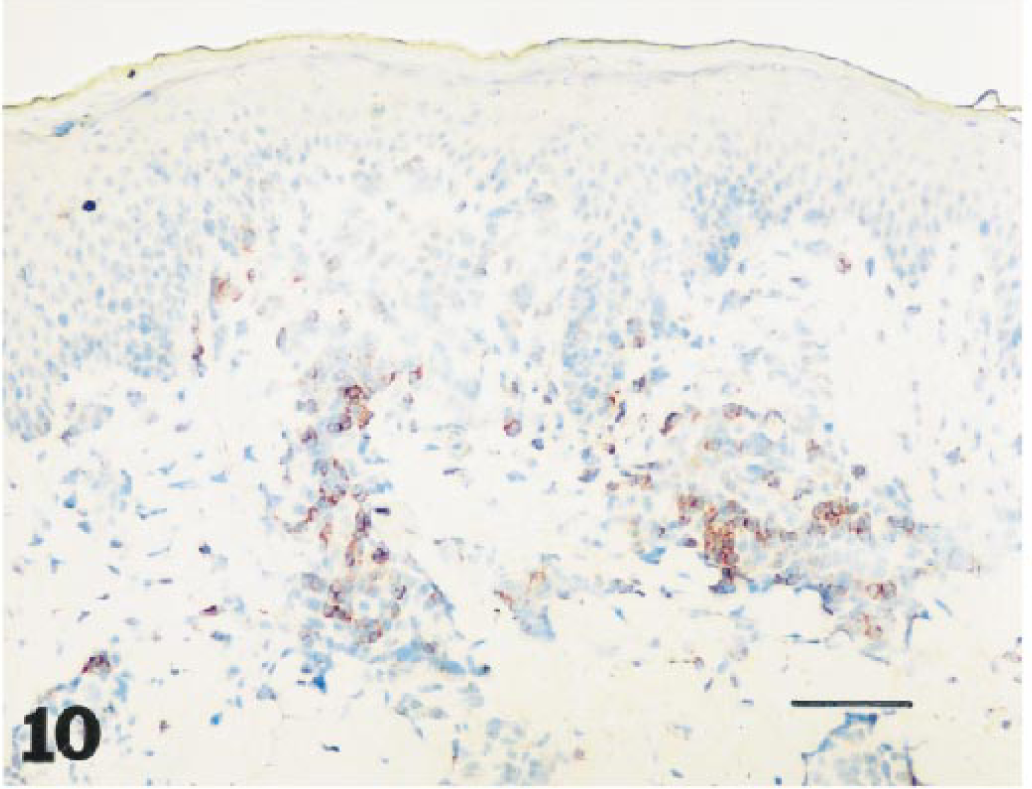
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 24 hours after elicitation: clusters of CD4+ T-cells, in increased numbers perivascular. Frozen section. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 28 µm.
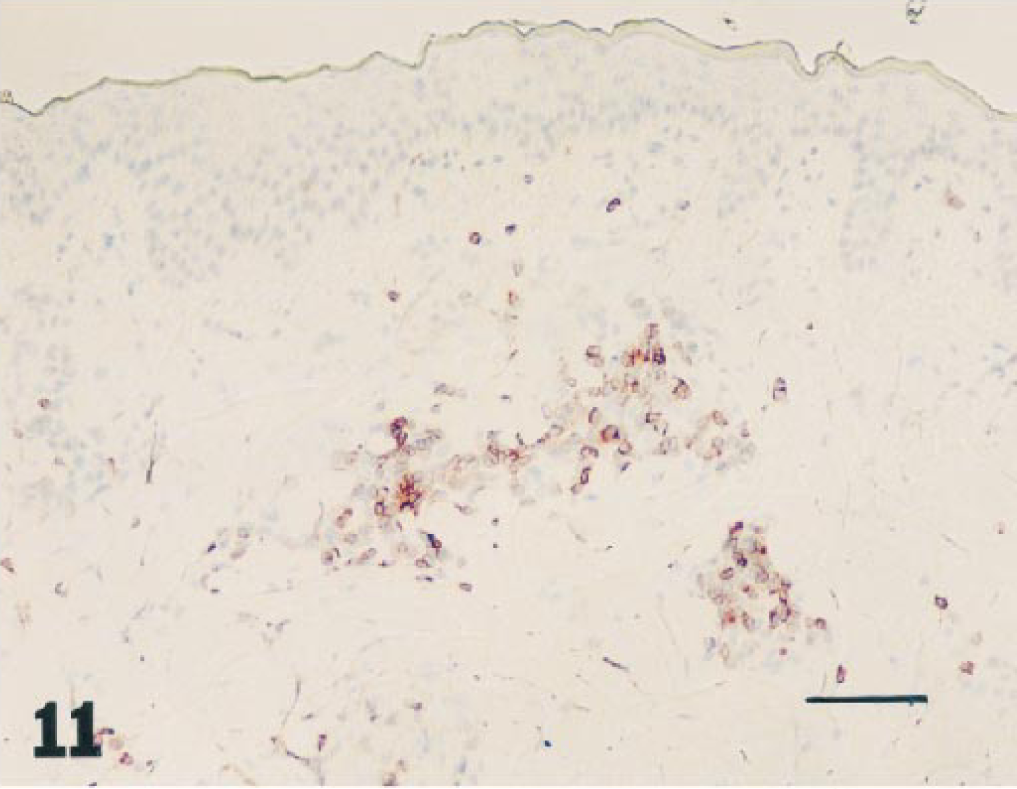
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site 8 hours after elicitation: CD5+ cells within the perivascular infiltrate. Frozen section. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 28 µm.
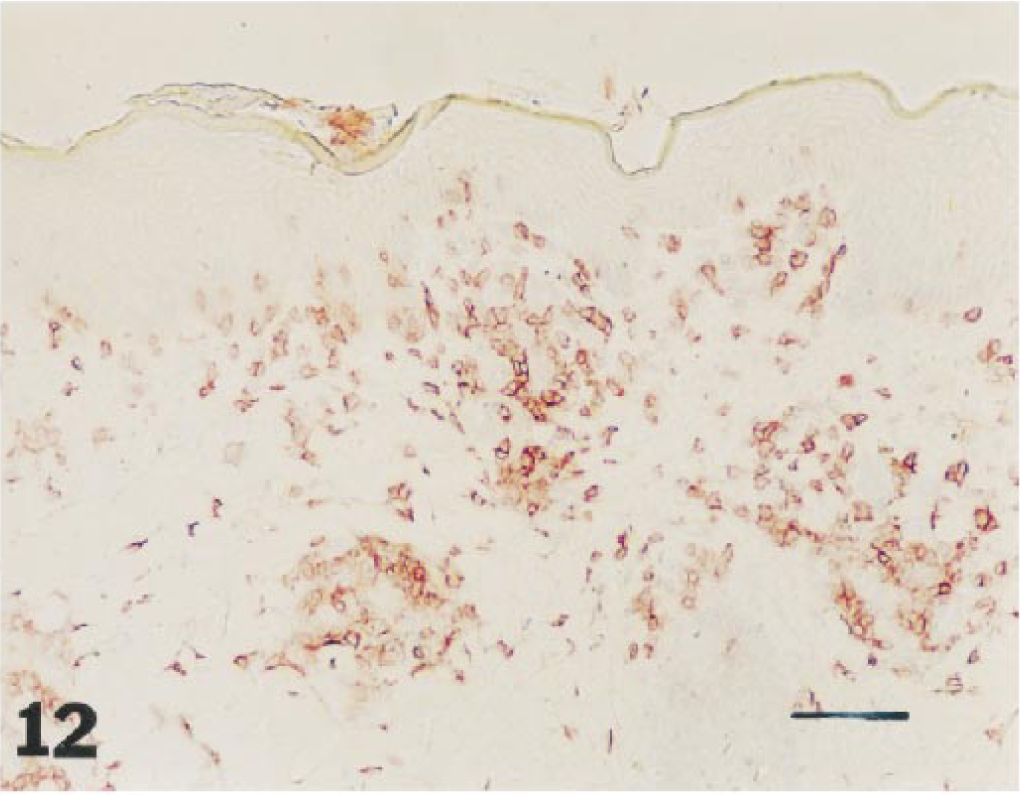
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 72 hours after elicitation: dense infiltrate of CD8+ cells in the upper dermis. Frozen section. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 28 µm.
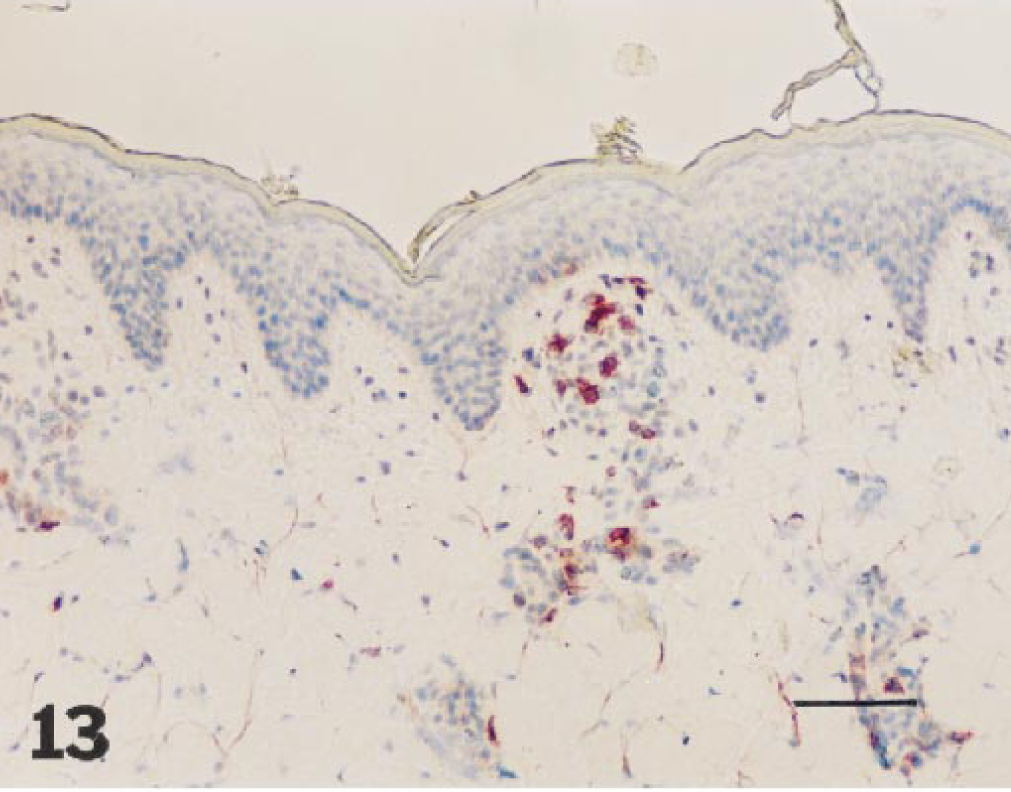
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 72 hours after elicitation: perivascular infilrate contains many CD25+ cells. Frozen section. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 28 µm.
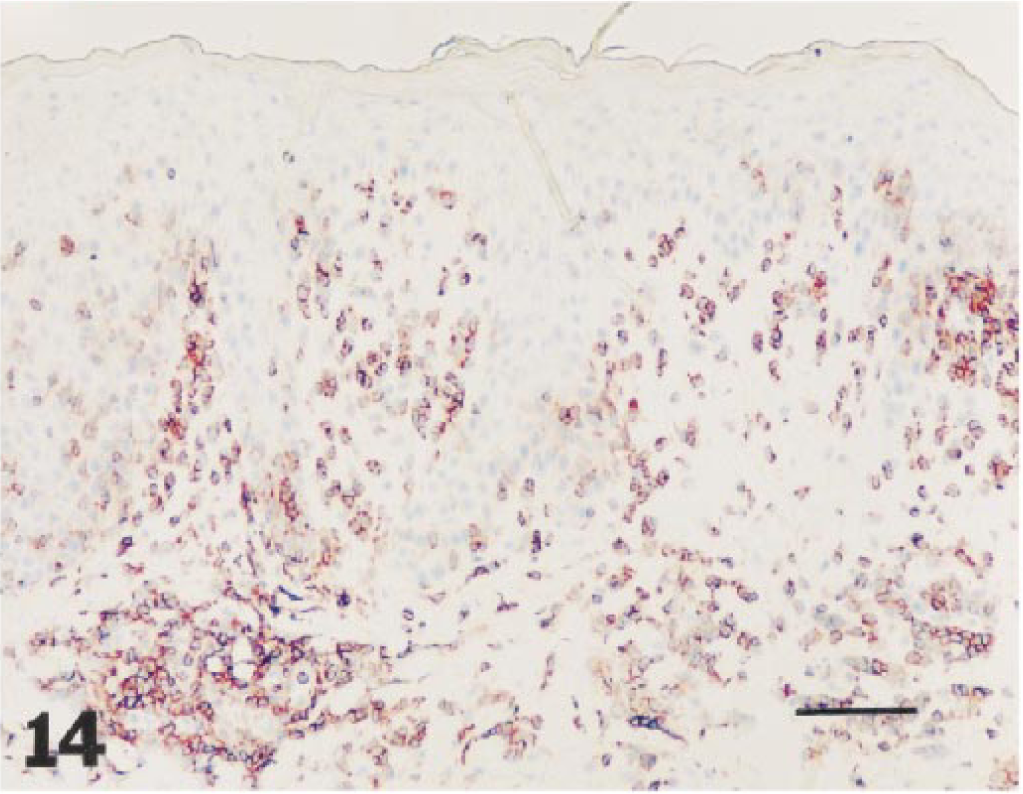
Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 24 hours after elicitation: numerous CD45+ cells in the epidermal and dermal infiltrate. Frozen section. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 28 µm.

Skin, 2,4-dinitrofluorobenzene allergic contact dermatitis site, 72 hours after elicitation: pronounced endothelial staining with anti-E-selectin. Frozen section. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 28 µm.
Discussion
The present study revealed that skin exposure to 1% DNFB has caused in all sensitized Göttingen minipigs an acute hypersensitivity reaction associated with a pronounced erythema and swelling of all test sites. These changes were fully developed 24 hours after challenge and diminished thereafter. Because potent sensitizers such as DNFB can also cause primary irritant contact dermatitis we used three nonsensitized animals for DNFB controls. In contrast to the findings in the sensitized animals, 1% DNFB caused only very mild changes in one of the three pigs 8 hours after application, indicating that the described features in the eight sensitized animals were caused by hypersensitivity reactions. The major objective of the study was to characterize the changes of acute ACD in pigs and to compare acute ACD in this model to that observed in human beings. The observed gross changes were identical with the stadium erythematosum et oedematosum in humans. 13 The phases stadium vesiculosum et bullosum, which is in severe cases of hand and foot contact eczema often followed by stadium madidans (i.e., oozing phase), and the stadium squamosum were not observed in the experimentally induced ACD of Göttingen minipigs. The skin conditions at the test sites on the dorsolateral back of the animals possibly did not allow the formation of these changes although the inflammatory reaction in general was pronounced.
The histopathologic changes at the DNFB-ACD sites resembled very closely human acute ACD in terms of physical features and kinetics of the lesions. Mild acanthosis, intra- and intercellular edema, exocythosis, and microabscesses in the epidermis were common findings. However, intraepidermal vesicles often seen in human patch test reactions have not been observed in porcine samples. 45 The formation of vesicles possibly was missed by the selected time points of biopsies because intracorneal abscesses were already present 24 hours after challenge and may have resulted from preceding intraepidermal vesicles filled with inflammatory cells. The dermal changes in the porcine samples also mirrored the human condition. Hyperemia, edema, and vascular and perivascular infiltration by inflammatory cells, predominately composed of mononuclear cells intermingled with neutrophil and eosinophil granulocytes, peaking 48–72 hours after challenge were observed. 10–12, 32 The involvement of mast cells, which are known as disease-intensifying elements in ACD from studies in mice, cannot be deduced directly from the present study, because their numbers remained relatively constant over time. 16 28 Onset of edema possibly was associated with mast cell activation and degranulated cells possibly were rapidly replaced by intact cells maintaining cell numbers constant.
The histologic changes in the DNFB-control sites were clearly distinct from the DNFB-ACD changes in intensity and time course. Mild reactive changes occurred to DNFB, which has concentration-dependent irritative potential, and probably also occurred to the mild mechanical trauma of cleaning and shaving of the sites because minimal background changes were also observed in the normal control sites collected 4 weeks before the principal investigation.
For immunocytochemical characterization of changes in ACD, we first phenotypically characterized cells in normal pig skin tissue (normal controls). In the epidermis CD1+ and MCH class II+ cells were observed in similar numbers and locations and most likely represented Langerhans cells. 8 These antigen-presenting cells are bone marrow derived and should therefore also be CD45+. 27 However, the numbers of epidermal CD45+ cells were 33–51% less than the CD1+ or MHC class II+ cell counts. This phenomenon was also observed by Foster et al. 15 in normal human epidermis, assuming a low expression of CD45 molecules on Langerhans cells. CD5+ cells were more frequently observed than CD2+ cells in the epidermis of normal control sites. Because the antibody b53b7 labels CD5+ T-cells as well as null cells, NK cells, and B cells the real number of CD5+ T-cells was masked. 47 However, because the specificity of the b53b7 antibody is higher than the specificity of the MAS4 (anti-CD2) antibody for null cells, which has in turn a higher specificity than the b53b7 antibody for NK cells, one can conclude that the increased number of CD5+ cells is due to costained null cells (phenotypically CD2−, CD4−, CD8−, TCRγδ+) because B cells are not present in normal epidermis. 4 7 53 57 Further supporting these arguments is our finding that in spleen sections of the Göttingen minipigs very few positively stained cells were seen with b53b7 in B cell areas, and also the report that only 10–30% of the B cells are CD5+ in pigs. 1 The sum of CD4+ and CD8+ cells was clearly less than the number of CD5+ cells, indicating again that null cells/TCRγδ+ cells with a high affinity to epithelial tissue in pigs have contributed to the high number of CD5+ cells. 4 This is also in agreement with studies performed by Saalmüller et al. 48 Considering null cells/TCRγδ+ cells as the most frequent subset of T-cells, porcine epidermis possibly differs from human skin with TCRαβ+ T-cells as the prominent cell type. 14 54 However, these findings need verification using multiple staining with specific antibodies or mRNA analyses with reverse transcription–polymerase chain reaction of epidermal sheets. A high prevalence of TCRγδ+ cells, in the skin has also been described in young lambs. 23 Like humans, epidermal CD4+ and CD8+ cells were almost equal in numbers, and both types together were slightly more frequent than CD2+ cells. 15 Because in pigs the four different types of CD4+CD8−, CD4−CD8+, CD4+CD8+, and CD4−CD8− T-cells are known and the CD4−CD8− cells have been labeled with the anti-CD5 antibody (b53b7), the quota of CD4+CD8+ is approximately 25%. These cells probably represent functionally T memory cells. 56 62 The number of CD25+ cells was higher than the numbers of the CD4+, CD8+, or CD2+ cells; therefore, cells other than activated CD4+ and CD8+ cells, which are also present in human epidermis, may have reacted with the antibody against the α chain of the IL2 receptor. 14 More likely, candidates are the γδ+T-cells rather than Langerhans cells because null cells also express CD25 and the numbers of CD25+ cells correspond approximately to the difference in CD5+ and CD2+ cells, whereas the numbers of CD1+ and CD25+ did not match. 21
In contrast to the epidermis, the dermis contained clearly fewer MHC class II+ cells than CD45+ cells. The CD1+ cells by shape are considered to represent dermal dendritic cells. With regard to the numbers and ratio of CD2+, CD5+, CD4+, CD8+, and CD25+ cells, the dermal condition was very similar to the epidermal milieu, except for the ratio of CD4+ to CD8+ cells, which was slightly in favor of the CD4+ cells in the dermis. This observation is also known from studies in human skin. 14 Positive staining for E-selectin was observed in all samples and is possibly related to its constitutive expression in dermal endothelial cells as reported for human cells, although induction by mechanical stimuli (cleaning and shaving) might also be considered. 9 20
In DNFB-ACD sites pronounced and statistically significant changes in cellularity were observed. A massive increase in CD45+ cell numbers occurred in the epidermis and dermis, which was most pronounced 24–48 hours after challenge. This finding is similar to the human ACD condition and corresponds to the inflammatory cell infiltration seen in Giemsa-stained sections. 45 The number of epidermal CD1+ cells (cells per mm2) decreased during the elicitation phase in pig samples as described in human specimens. 6 Because contradicting reports also exist describing an increase or at least no changes in CD1+ numbers in human samples we also counted the numbers of CD1+ cells per millimeter of epidermal basal membrane. Again, although not statistically significant, the number was less compared with normal control sites. 33 The numbers of MHC class II+ cells remained more or less constant, suggesting that emigrating Langerhans cells (positive for CD1 and MHC class II) were replaced by immigrating MHC class II+ monocytes or macrophages. Eight hours after challenge more CD5+ cells were seen in the epidermis than CD2+ cells or CD4+ and CD8+ cells taken together. As outlined previously, this difference may be due to infiltration with null/γδ+T-cells. This cell type has been described in lambs as a prominent T-cell population in the elicitation phase of DNCB-induced contact hypersensitivity. 26 After 48–72 hours CD2+ cells were more frequent than CD5+ cells and this difference resulted probably from NK cells, which are also stained by the antibody MSA4 reacting with CD2 (Saalmüller, personal communication). CD8+ cells dominated over CD4+ in the epidermis from 24 to 72 hours after challenge. An increase also occurred in CD25+ cells, which exceeded in numbers CD4+ cells, indicating that CD8+ T-cells or null/γδ+T-cells were also positive for CD25. An increase in activated CD25+ T-cells has also been observed in human biopsies from patch test reactions. 2 The antibody MIL2, which is reported to label monocytes, macrophages, and neutrophil granulocytes, did not react with aggregated inflammatory cells in the epidermis. Therefore, these cells may have lost their surface markers because of the onset of necrosis or autolysis within these intraepidermal microabscesses.
An increase also occurred in dermal cells positive for CD45, MHC class II, and CD1; the latter cell type probably represents dermal dendritic cells, which are also increased in human ACD. 60 A general feature of the inflammatory infiltrate in ACD is the high proportion of T-cells. 2 35 42 44 This characteristic has also been observed in porcine samples containing high numbers of CD2+, CD5+, CD4+, CD8+, and CD25+ cells. The ratio of dermal CD2+ and CD5+ cells was similar to the epidermal condition and NK cells may have also been labeled with the anti-CD2 antibody MSA4. In samples collected up to 48 hours after challenge, CD4+ cells were more frequently observed than CD8+ cells and were similar in numbers at 72 hours. This observation was also made in human biopsy studies, indicating that in the early efferent phase CD4+ cells dominate, whereas in later stages CD4+ and CD8+ cells were equal in number. 41 50 Because the number of CD25+ cells in the present samples also increased with time, similar to the CD8+ cells, we believe that CD8+ cells may have also expressed the IL-2 receptor. Staining with the MIL2 antibody labeled dispersely distributed cells in the dermis and only a few cells within the vascular/perivascular infiltrate; the significance of this finding is unclear and may be related to undefined specificity of the antibody. In agreement with findings in human ACD, increased reactivity for E-selectin was also noticed in the course of ACD in pig. 5 9 20
For comparison, minor and statistically not significant changes in cellularity were observed in the DNFB-control sites from three unsensitized pigs treated with 1% DNFB. A slight decrease occurred in CD1+ and MHC class II+ cells, probably due to emigration of Langerhans cells or to effects interfering with the expression of these surface markers. The number of T-cells remained almost constant, except a slight increase in CD5+ cells over CD2+ cells, which, again, is likely due to additional staining of null/γδ+T-cells with the anti-CD5 antibody. These cells may have also been labeled with the anti-CD25 antibody resulting in slightly elevated numbers of CD25+ cells. The immunohistologic staining of dermal cells correlates with the Giemsa staining, showing a mild unspecific inflammatory reaction against the DNFB application.
In summary, the present study revealed a close relationship between human and porcine ACD with regard to clinical, histologic, and immunohistologic findings. Therefore, ACD in pigs seems to be a very relevant animal model for humans. Increased numbers of null/γδ+T-cells and extrathymic CD4+CD8+ cells may be peculiarities to pig skin.
Footnotes
Acknowledgements
We wish to thank Dr. Saalmüller for supplying the antibodies and Elisabeth Kowalsky and Hermann Fahrngruber for their excellent technical assistance.
