Abstract
A clear distinction can be made regarding the susceptibility to and the severity of lesions in young lambs when compared to adult sheep. In particular, there are important differences in the lesions and tropism of Rift Valley fever virus (RVFV) in the liver, kidneys, and lymphoid tissues of young lambs. A total of 84 lambs (<6 weeks old), necropsied during the 2010 to 2011 Rift Valley fever (RVF) outbreak in South Africa, were examined by histopathology and immunohistochemistry (IHC). Of the 84 lambs, 71 were positive for RVFV. The most striking diagnostic feature in infected lambs was diffuse necrotizing hepatitis with multifocal liquefactive hepatic necrosis (primary foci) against a background of diffuse hepatocellular death. Lymphocytolysis was present in all lymphoid organs except for the thymus. Lesions in the kidney rarely progressed beyond hydropic change and occasional pyknosis or karyolysis in renal tubular epithelial cells. Viral antigen was diffusely present in the cytoplasm of hepatocytes, but this labeling was noticeably sparse in primary foci. Immunolabeling for RVFV in young lambs was also detected in macrophages, vascular smooth muscle cells, adrenocortical epithelial cells, renal tubular epithelial cells, renal perimacular cells, and cardiomyocytes. RVFV immunolabeling was also often present in capillaries and small blood vessels either as non-cell-associated viral antigen, as antigen in endothelial cells, or intravascular cellular debris. Specimens from the liver, spleen, kidney, and lungs were adequate to confirm a diagnosis of RVF. Characteristic lesions were present in these organs with the liver and spleen being the most consistently positive for RVFV by IHC.
Keywords
Rift Valley fever (RVF) is a viral hemorrhagic disease caused by a single-stranded RNA virus of the Bunyavirales order, Phenuiviridae family. The virus, first isolated in 1931 during an investigation into an outbreak among sheep in the greater Rift Valley of Kenya, causes severe disease in both humans and animals. 6 Subsequently, epidemics have been reported in most countries in Africa. 22 The disease also spread to the Arabian peninsula and caused large epidemics in Saudi Arabia and Yemen. 28 RVF was also reported in Madagascar and later in the Comoros and Mayotte. 3,14,30 The disease mainly affects sheep, but cattle, goats, African buffaloes, camelids, and other wild animals are also reportedly affected. 23 The virus is primarily transmitted by mosquitoes and experimentally competent vector species have been identified in Europe and the United States, raising concern about the risk of introduction of RVF virus (RVFV) into these RVF-free regions. 15,33
The first description of RVF in Kenya reported heavy mortalities in newborn lambs, a marked rise in the mortality rate of ewes, and an increase in abortions. 6 The authors remarked that the liver lesions in young lambs were much more severe than in adult sheep and that extensive hepatic necrosis only rarely occurred in adult sheep. Daubney et al 6 also noted that lesions in the kidneys of young lambs differed from those of adult sheep. Multifocal acute renal tubular injury (nephrosis) in adult sheep with RVF was confirmed in subsequent research. 6,11,12,18,20,24 However, in lambs, the kidney lesion rarely progresses beyond tubular epithelial cell degeneration, characterized by cell swelling and karyolysis. 6 In addition to extensive hepatic necrosis, studies of natural infections in young lambs also reported gallbladder edema, pleural effusions, lung edema, and widespread hemorrhages in many organs, including occasional enteric hemorrhage. 4,6,27 Varying degrees of pyknosis and karyorrhexis in lymphoid tissues of newborn lambs that died of RVF were also reported. 4
Experimentally, the incubation period of RVF in 1- to 5-day-old lambs is 12 to 24 hours. 7 This is followed by a marked febrile response (41–42°C) and rapid progression to death within 24 to 72 hours. 7 The initial lesion in the liver of 1- to 5-day-old lambs consists of small aggregates of neutrophils in the sinusoids that develop between 12 and 24 hours after inoculation. 7 Between 24 and 36 hours, small random foci of hepatocellular necrosis, referred to as primary foci, develop. 7 These foci involve only a few hepatocytes at 24 hours after exposure and evolve into larger, more circumscribed areas of liquefactive necrosis in the ensuing 6 to 12 hours. 4 Within primary foci, the hepatocytes and infiltrating leukocytes disintegrate completely, causing a loss of tissue and cellular profiles, and present as fine basophilic karyorrhectic particles against a background of homogeneous eosinophilic debris. 4,7 Shortly after development of the primary foci, diffuse destruction of the remaining hepatocytes occurs. 4 A number of authors have described primary foci in young lambs and concluded that it is pathognomonic for RVFV. 4,7,9
The cellular tropism of naturally occurring RVF has only been studied using immunohistochemistry (IHC) in 4 newborn lambs wherein viral antigen was reported in the liver, spleen, lymph nodes, and lungs. 35 Previously, we described the lesions and reported the cellular tropism of natural RVFV infection in adult sheep from the same outbreak in South Africa. 18 RVF is much more acute in young lambs than in older sheep, and there are important differences in the lesions and tropism, particularly the liver, kidneys, and lymphoid organs of young lambs. Therefore, the principal aim of the present study was to describe the quantitative and qualitative pathomorphology of RVFV infection in diverse organs from a large number of naturally infected lambs and contrast this with previously published results for adult sheep. 18 Further aims were to ascertain the extent to which virus can be detected in different organs and establish the diagnostic significance of certain tissues and specific histologic features of RVF in young lambs.
Materials and Methods
Case Selection
All specimens originated from the carcasses of naturally infected lambs necropsied during the 2010 to 2011 RVF outbreak in South Africa that affected mainly sheep. 23 This work was done with approval from the Animal Ethics Committee of the University of Pretoria (clearance certificate V096-16). Supplementary material in support of the results presented in this article is available from the authors upon request.
Tissue samples were collected from sheep, including young lambs, as described previously. 18 Briefly, tissues from 1034 necropsied animals were collected by government officials and the principal investigator (L.O.), preserved in 10% neutral buffered formalin, and embedded in paraffin, and hematoxylin and eosin–stained histologic sections were routinely prepared. Included were at least 173 young lambs up to 6 weeks of age. From this collection, cases were excluded when the level of autolysis was severe, when only 1 organ was submitted, or when the liver was not available for study. A total of 84 cases were suitable for study, of which 71 cases were classified positive for RVF based on results of histopathology, reverse transcription quantitative polymerase chain reaction (RT-qPCR), and/or IHC. A parallel interpretation of tests was chosen to maximize sensitivity, and therefore, any animal with at least 1 positive test result was selected for study. In addition, animals that died as part of the outbreak and were shown to be infected with RVFV were presumed to have died of RVF even if lesions were absent. Negative controls included in this study were 13 lambs that died of natural infections during the outbreak, were examined for RVFV as part of the government campaign to monitor the spread of RVF, but tested negative.
Diagnostic Tests
Nucleic acid extractions and RT-qPCR on fresh liver specimens for 46 of the cases from the 2010 outbreak were performed at the Biotechnology PCR Laboratory of the Agriculture Research Council–Onderstepoort Veterinary Institute as described elsewhere. 19 None of the cases included from the 2011 outbreak (n = 16) were tested using RT-qPCR.
Immunohistochemistry with additional controls was performed as described in the original validation study. 19 Briefly, labeling for the detection of RVFV antigen was performed using polyclonal antibody to RVFV (mouse ascitic fluid), prepared at the National Institute for Communicable Diseases (Johannesburg, South Africa) as described. 25,32 Previously, 40 controls were tested that included 8 RT-qPCR- and IHC-positive RVF cases that occurred in 2009 and 32 additional negative tissue controls that had liver lesions that resembled RVF but resulted from other causes. 19 The controls were tested with a range of relevant and irrelevant antibodies—namely, polyclonal antibody to RVFV, polyclonal antibody (mouse ascitic fluid) to Wesselsbron virus (antibody to Wesselsbron virus), monoclonal antibody (mouse ascitic fluid) to bovine herpesvirus 1, and rabbit polyclonal antiserum to equine herpesvirus 1. 19 The antibody to Wesselsbron virus, which was used as the negative reagent control antibody in this study, was similarly prepared at the National Institute for Communicable Diseases, and validated for IHC at the Faculty of Veterinary Science, University of Pretoria. 25,34 In addition, to confirm that intravascular immunoreactivity observed in this study was due to RVFV antigens, tissue sections were sequentially tested with the antibodies to RVFV and Wesselsbron virus.
The immunohistochemistry method included microwave antigen retrieval in citrate buffer (pH 6.0), blocking of endogenous peroxidases with 3% hydrogen peroxide, incubation with the anti-RVFV primary antibody at 1:500 dilution for 1 hour, and rabbit anti-mouse secondary antibody (F0232; DakoCytomation, Glostrup, Denmark) followed by detection with a standard avidin-biotin peroxidase system, Vectastain Elite ABC-HRP Kit (PK-6100; Vector Laboratories, Burlingame, CA), NovaRED peroxidase substrate (SK-4800; Vector Laboratories), and hematoxylin counterstain.
Examination of Tissues
Histomorphological features in all available organs were systematically recorded and reviewed within the context of lesions associated with RVFV infection. Two authors (L.O., S.J.C.) reviewed 20% of the cases together to validate data collection methods and the remainder were examined by the principal investigator (L.O.).
Lesion severity in the liver, spleen, and kidney was scored using a qualitative scale as mild, moderate, or severe. The number of primary foci in the liver specimens was scored on a semiquantitative scale as isolated (<5), scattered (between 6 and 10), or widespread (>10). All liver samples measuring between approximately 5 mm2 and 20 mm2 were examined. Immunolabeling for RVFV antigen was scored on a semiquantitative scale as isolated, scattered, or widespread labeling, as previously described. 18
The number and types of organs available for study varied considerably by case with liver (n = 84; inclusion criterion), spleen (n = 64), lung (n = 63), and kidney (n = 56) being the specimens most often submitted. Additional tissue specimens included 40 heart, 30 gastrointestinal tract, 21 lymph node, 19 brain, 10 thymus, 8 adrenal, 3 gallbladder, and 3 skin specimens. Gastrointestinal tract specimens included tongue (n = 5), forestomach (n = 3), abomasum (n = 15), small intestine (n = 19), and large intestine (n = 1).
Statistical Analysis
Categorical data were presented as proportions and 95% mid-P confidence intervals. Data from ordinal scales were described using the median and interquartile range (25th to 75th percentiles). Results for the ordinal scales were compared between the liver and other organs using Wilcoxon signed rank tests. Correlations between scaler data were estimated using Spearman’s ρ and categorical data using Cohen’s κ. Statistical analyses were performed in freeware (Epi Info, version 6.04; Centers for Disease Control and Prevention, Atlanta, GA, USA) and commercial statistical software (IBM SPSS Statistics Version 25; SPSS, Inc, an IBM Company, Chicago, IL). Significance was set as P < .05.
Results
Overview
The organs most consistently positive for RVFV by IHC were liver and spleen (Table 1). Viral antigen was also easily identified in lung, kidney, brain, lymph node, thymus, and small intestine. Positive labeling was typically fine diffuse to coarse granular cytoplasmic labeling. Vascular labeling was especially prominent in blood vessels, both as non-cell-associated antigen, as well as cell-associated antigen in endothelial cells and vascular smooth muscle cells. Endothelial/non-cell-associated intravascular viral antigen was often present in the brain, lymphoid organs, and the lung (Table 2), whereas labeling in vascular smooth muscle cells was most commonly present in the kidney (Table 3). Immunolabeling was not observed in sequential slides incubated with antibody to Wesselsbron virus instead of RVFV (Supplemental Figs. S1–S4). In addition, typical histological lesions and RVFV antigen were not detected in any of the tissues from the 13 cases included as negative tissue controls (Supplemental Figs. S5–S8). Immunohistochemistry was negative for RVFV antigen in all samples from negative control cases.
Frequency of Viral Antigen in Different Organs and Comparison to Quantity of Immunolabeling in Liver, for 71 Young Lambs With Natural Rift Valley Fever Virus Infection.
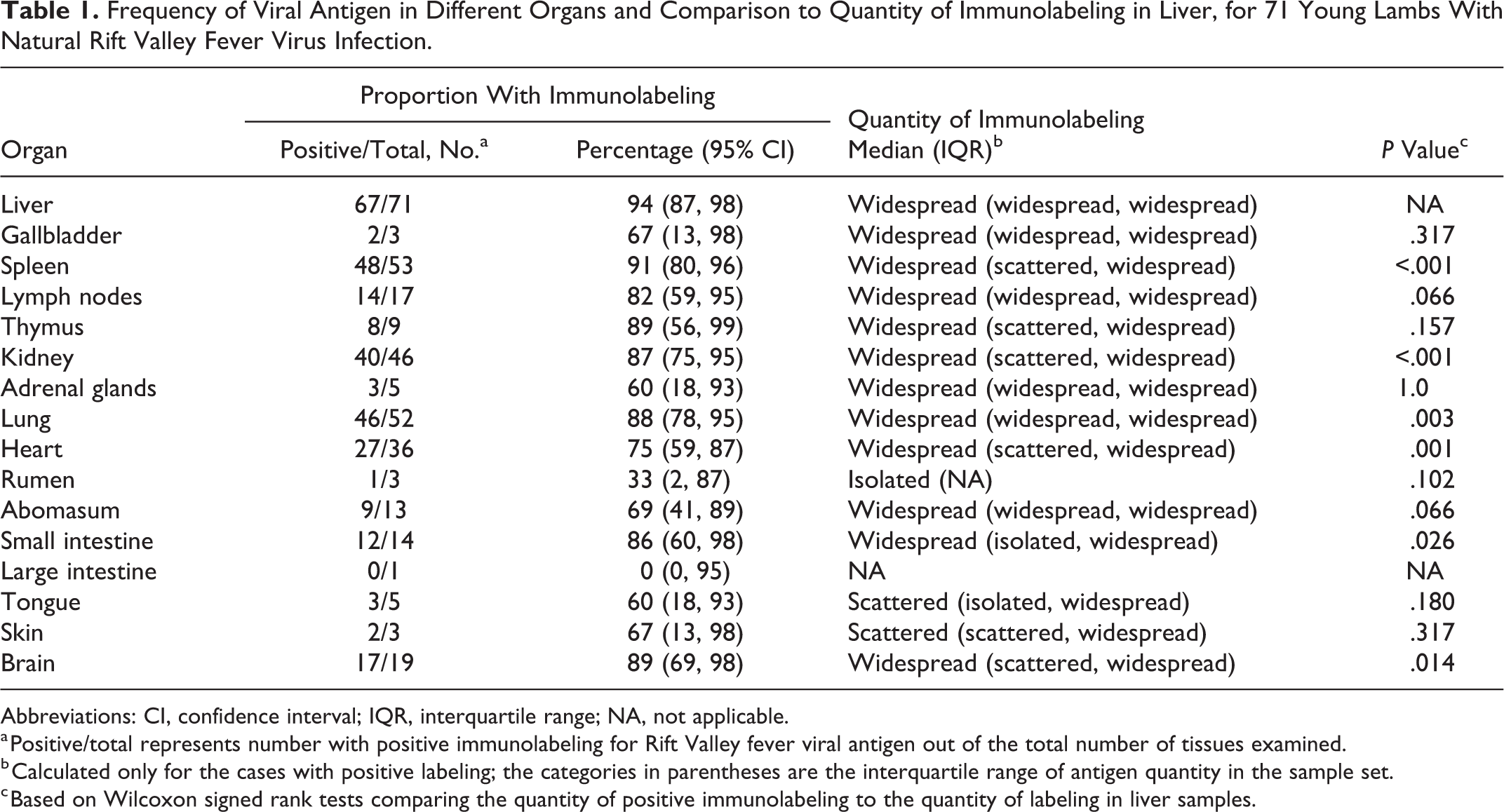
Abbreviations: CI, confidence interval; IQR, interquartile range; NA, not applicable.
a Positive/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues examined.
b Calculated only for the cases with positive labeling; the categories in parentheses are the interquartile range of antigen quantity in the sample set.
c Based on Wilcoxon signed rank tests comparing the quantity of positive immunolabeling to the quantity of labeling in liver samples.
Frequency of Non-Cell-Associated Viral Antigen in Blood Vessels or Antigen in Vascular Endothelial Cells or Intravascular Cellular Debris, for Young Lambs With Natural Rift Valley Fever Virus Infection.
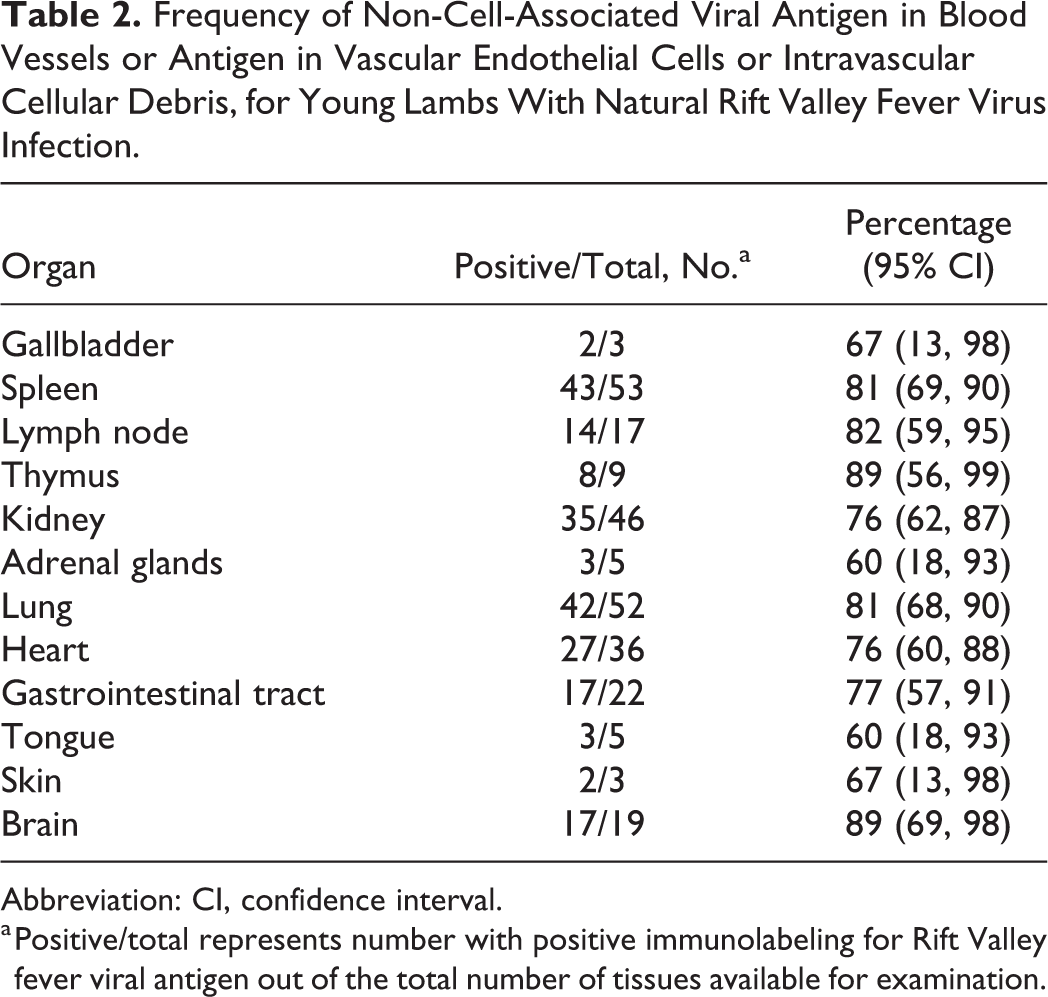
Abbreviation: CI, confidence interval.
a Positive/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues available for examination.
Frequency of Viral Antigen in Vascular Smooth Muscle in the Organs of Young Lambs With Natural Rift Valley Fever Virus Infection.a

Abbreviation: CI, confidence interval.
a Positive/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues available for examination.
Liver
Macroscopically, the livers were often swollen and friable. They were either light yellow or yellowish-brown due to diffuse necrosis or dark red due to severe congestion (Figs. 1, 23). Necrotic foci could be distinguished in some, while in other cases, congestion or diffuse color changes masked necrosis.
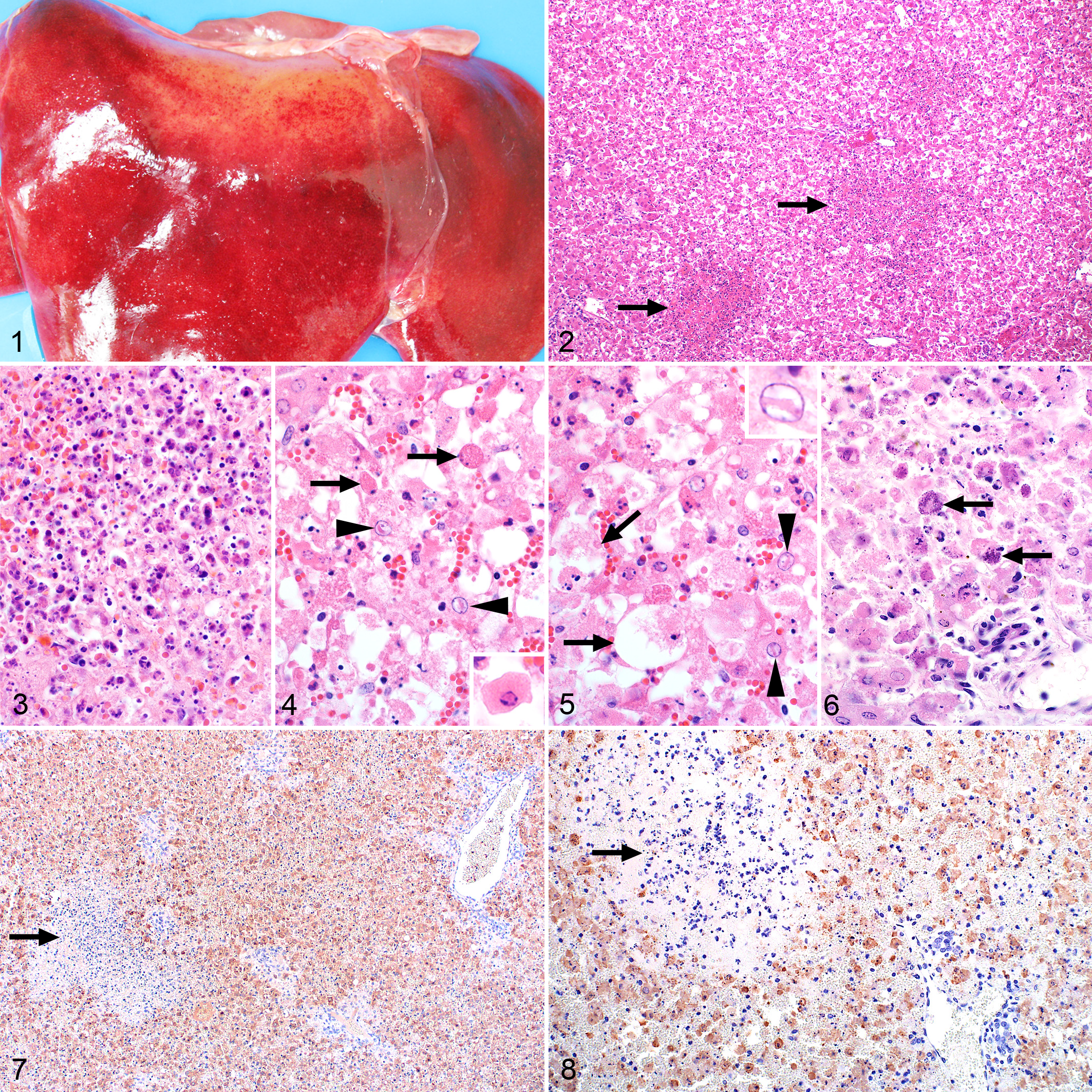
Microscopically, diffuse necrosis accompanied by an infiltrate of predominantly degenerate neutrophils with fewer macrophages was present in 65 of 71 (92%) of the cases with available specimens. Primary foci were present in the liver specimens from 60 of 71 (85%) cases and were isolated (30/71), scattered (16/71), or widespread (13/71). These foci were randomly distributed, well-demarcated areas of liquefactive necrosis, consisting of remnants of completely disintegrated hepatocytes, fine nuclear fragments, and a sparse infiltrate of degenerate neutrophils and macrophages in a collapsed reticular network (Figs. 2, 3 and Suppl. Figs. S1a–S3a).
Few surviving hepatocytes were present, and invariably these showed degenerative (hydropic) changes. The orderly arrangement of hepatocytes into plates was markedly disrupted and sinusoids were difficult to discern. Hepatocytes with condensed hypereosinophilic cytosol and marginalized chromatin, pyknosis, or karyorrhexis (Councilman bodies or acidophilic bodies) were present (Fig. 4). Other hepatocytes were swollen and rounded, with the nucleus completely disintegrated (karyolysis) or only small nuclear fragments remaining (karyorrhexis) (Fig. 5). Some hepatocytes appeared to have collapsed due to rupture of the cell membrane. Limiting plate hepatocytes were necrotic, and the connective tissue in the portal tracts was often disrupted. Hemorrhage or severe congestion featured prominently in most cases. Intranuclear inclusion bodies (Figs. 4, 5) were identified in 27 of 71 (38%) of the specimens and mineralization of hepatocytes (Fig. 6) in 15 of 71 (21%). Necrosis in the liver was positively correlated with necrosis in the lymph nodes (ρ = 0.515; P = .034) but not the spleen (ρ = 0.116; P = .409).
RVFV antigen was widespread and easily identified in 67 of 71 (94%) of the cases. Most RVFV immunolabeling in the liver was within hepatocytes (Fig. 7). Within primary foci, labeling was primarily present in scattered cellular debris. In addition, viral antigen was always sparse in primary foci compared to the intense labeling in the surrounding liver parenchyma (Fig. 8; Suppl. Figs. S1a, S2a, and S3a).
Four cases, positive by RT-qPCR, were negative by IHC in all the available organs. Mild hepatocellular injury with severe acute renal tubular injury was present in 1 of these cases while another had moderate centrilobular hepatic necrosis that was considered to have resulted from anemia. In this latter case, hemonchosis was mentioned in the records. Lesions were absent from the liver and the kidneys of the other 2 cases.
Gallbladder
Edema and occasional hemorrhages were present macroscopically in the examined gallbladders. Histologically, in 2 of the 3 cases with available specimens, IHC labeling was present in capillaries and small blood vessels either as non-cell-associated viral antigen or as antigen in endothelial cells or intravascular cellular debris.
Spleen
Gross lesions were uncommon in the spleen with capsular and subcapsular hemorrhages only occasionally present. Microscopically, splenic necrosis was a prominent feature in 50 of 53 (94%) of the cases and was either mild (n = 9), moderate (n = 15) or severe (n = 26). Generally, the periarteriolar lymphoid sheath (PALS) was a narrow zone of clearly identifiable cells, while in most cases (51/53), follicular germinal centers, mantle cell layers, and marginal zones were poorly developed (Fig. 9). The latter was interpreted as age appropriate for young lambs. Therefore, necrosis mainly involved lymphocytes in the red pulp and the peripheral aspects of the PALS (Fig. 9). Necrosis was present in both the red and white pulp in 47 of 48 (98%) of the cases that tested positive for RVFV antigen by IHC and characterized by the prominent presence of cellular debris in addition to occasional tingible-body macrophages. In the red pulp, necrosis was mild, moderate, and severe in 7 of 48 (15%), 17 of 48 (35%), and 23 of 48 (48%) cases, respectively. Necrosis in the white pulp was graded as mild, moderate, or severe in 18 of 48 (38%), 14 of 48 (29%), and 15 of 48 (31%) of the IHC-positive cases, respectively.
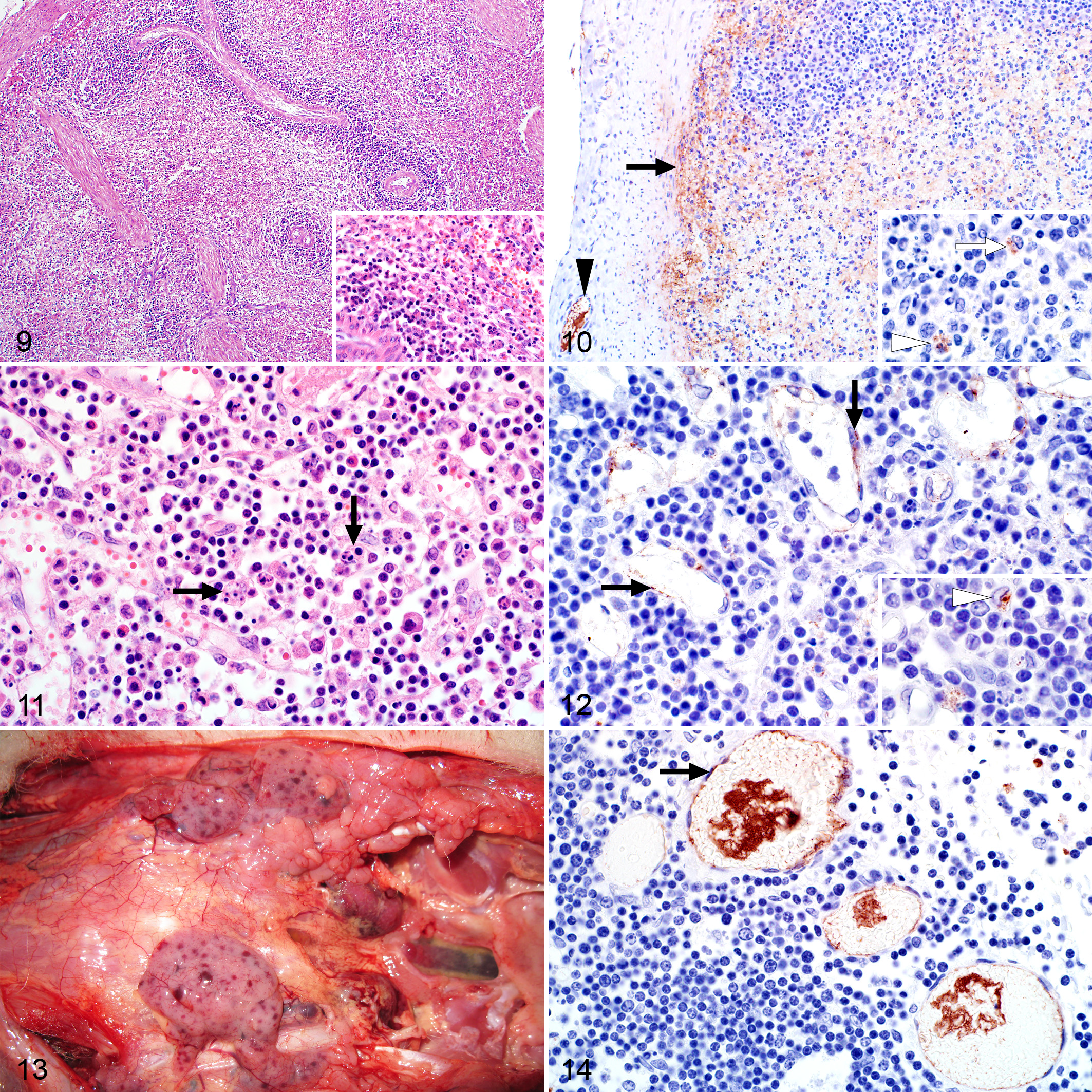
Splenic infection with RVFV was detected by IHC in 48 of 53 (91%) cases, and antigen was widespread (n = 33), scattered (n = 9), or isolated (n = 6). Viral antigen was commonly present in the red pulp in association with cellular debris (Table 4). In 41 of 53 (77%) of the cases, labeling was especially prominent in the subcapsular red pulp and in small blood vessels and occasionally smooth muscle within the capsule (Fig. 10 and Suppl. Fig. S4a). Non-cell-associated antigen and antigen in endothelial cells and cellular debris were also often present in small blood vessels. Mononuclear cells, morphologically consistent with macrophages, in the red pulp or at the periphery of the PALS were also positive in 45 of 53 (85%) cases. Occasionally, cells identified as tingible-body macrophages (Fig. 10) contained RVFV antigen (14/53 cases).
Distribution of Viral Antigen in Different Zones of the Spleen, for Specimens With Positive Immunolabeling for Rift Valley Fever Virus in Young Lambs.
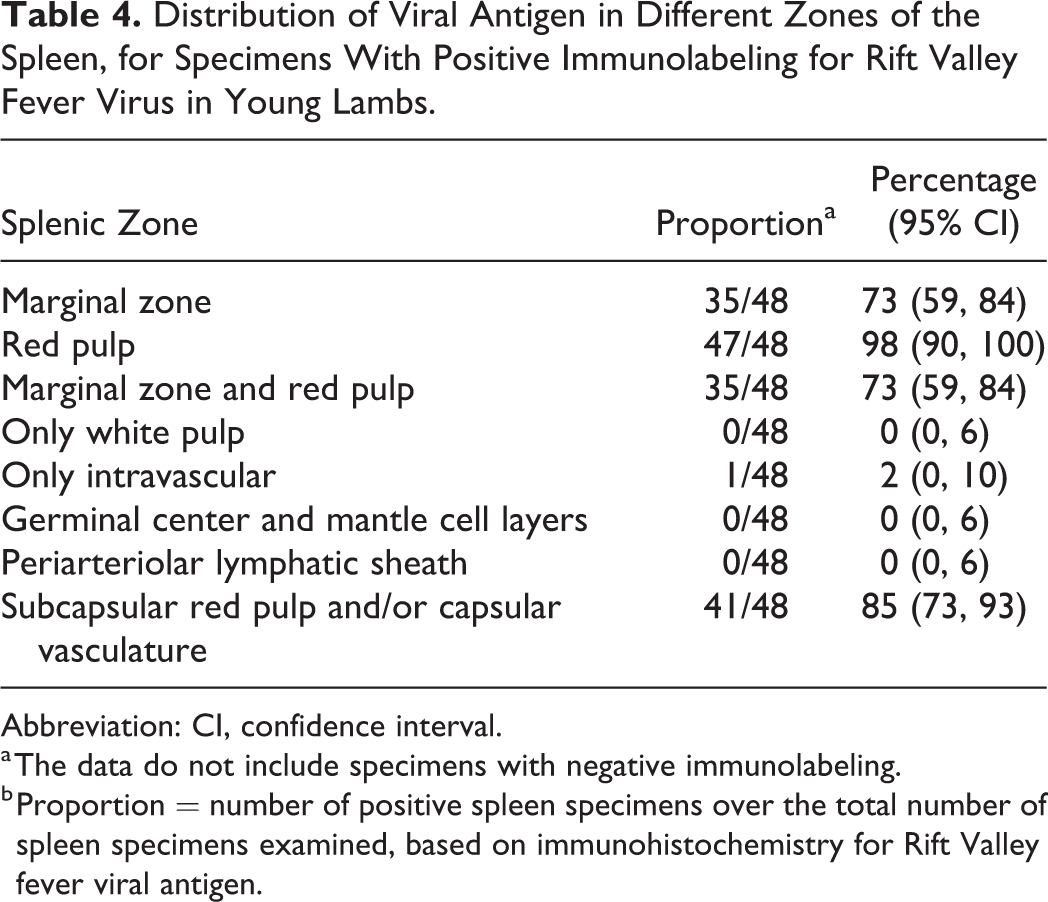
Abbreviation: CI, confidence interval.
a The data do not include specimens with negative immunolabeling.
b Proportion = number of positive spleen specimens over the total number of spleen specimens examined, based on immunohistochemistry for Rift Valley fever viral antigen.
Lymph Nodes
Macroscopically, lymph nodes were enlarged, edematous, and congested. Mesenteric lymph nodes were especially affected. Microscopically, there were variable degrees of lymphocytolysis and lymphoid depletion in 15 of 17 (88%) of the cases. Similarly to the spleen, follicular germinal centers were not well developed in most cases, and apoptotic lymphocytes were scattered throughout the cortex and the medullary cords (Fig. 11). In the cases that tested positive for RVFV antigen, lymphocytolysis was mild, moderate, and severe in 2 of 14 (14%), 8 of 14 (57%), and 4 of 14 (29%) of cases, respectively. Necrosis in lymph nodes was positively correlated with splenic necrosis (ρ = 0.577; P = .050).
Of the cases with available lymph node specimens, 14 of 17 (82%) were positive for RVFV antigen with widespread labeling in most cases (n = 11). Viral antigen was present in small blood vessels and capillaries in endothelial cells (Fig. 12) or as non-cell-associated virus (Suppl. Fig. S5a). In the sinusoids, mononuclear cells, morphologically consistent with macrophages, were also positive for viral antigen (Fig. 12).
Thymus
Petechiae or ecchymoses present in the thymus (Fig. 13) were confirmed microscopically as being in the interstitium in 3 of 9 (33%) cases with available specimens. However, lymphocytolysis that was frequently present in the other lymphoid organs was absent in the thymus. In addition, viral antigen was confined to capillaries and small blood vessels as non-cell-associated viral antigen, antigen in endothelial cells, or intravascular cellular debris (Fig. 14 and Suppl. Fig. S6a).
Kidney
Gross kidney lesions were uncommon other than occasional capsular or cortical petechiae or ecchymoses. Microscopically, severe multifocal acute tubular epithelial injury lacking significant associated inflammation was present in 11 of 46 (24%) cases. Mild or moderate injury occurred in 5 of 46 and 4 of 46 cases, respectively. Characteristic degenerative changes in tubular epithelial cells included fine vacuolation and swelling as well as impingement of the tubular lumen. This was occasionally accompanied by pyknosis or karyolysis, with detachment of a few cells from the basement membrane. Marked acute renal tubular epithelial necrosis was absent. There were no tubular epithelial lesions in the remaining 26 of 46 (57%) of the kidney specimens, even though 23 of these tested positive by IHC. In addition, the glomeruli appeared less densely cellular than normal and scattered pyknosis, and karyorrhexis was present within the glomeruli of 37 of 46 (80%) cases (Fig. 15). Nuclear debris was also often present in the interstitial capillaries, and pyknosis and karyorrhexis in the glomeruli were positively correlated (ρ = 0.896, P < .001) with the presence of nuclear debris in the interstitial capillaries. The presence of pyknosis and karyorrhexis in the glomeruli was also significantly associated with the detection of viral antigen in the glomeruli (ρ = 0.309, P = .0346).
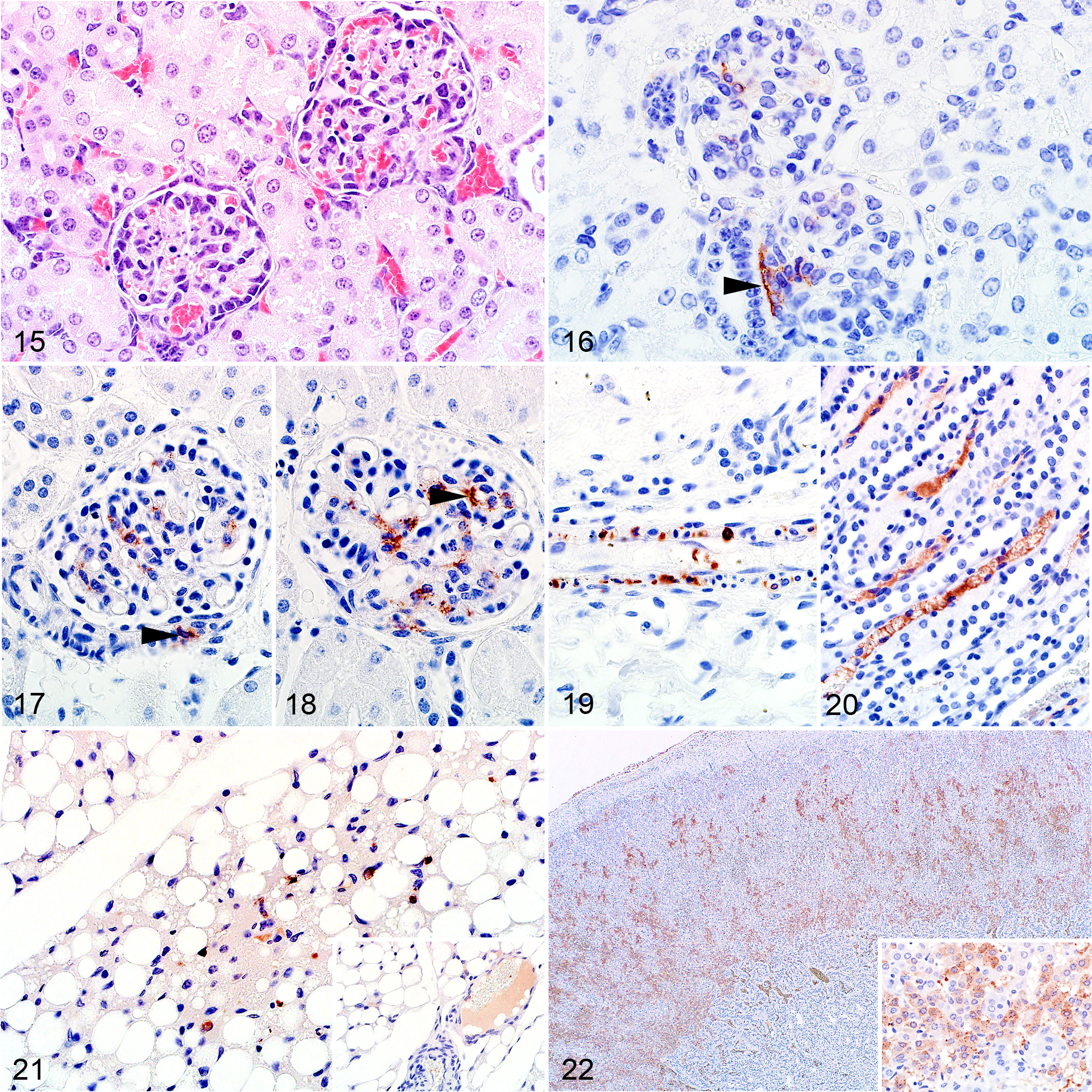
Of the cases with available kidney specimens, 40 of 46 (87%) were positive for RVFV antigen. In the positive cases, labeling was present in both the cortex and medulla. Glomerular labeling was present in 37 of 40 (93%) cases. Viral antigen was often present at the vascular pole of the glomerulus opposite the macula densa, within a small group of cells that are likely juxtaglomerular and extraglomerular mesangial cells (lacis cells) (Fig. 16 and Suppl. Fig. S8a). Cells of the macula densa were not affected. Labeling was also present in smooth muscle cells in the efferent or afferent arteriole (Fig. 17) or vascular endothelial cells (Fig. 18). In 26 of 40 (65%) cases, viral antigen was also present in smooth muscle cells of small blood vessels in the cortex or at the corticomedullary junction (Fig. 19). Labeling of tubular epithelium was present in 8 of 40 (20%) of the IHC-positive cases, and viral antigen was isolated (n = 6) or scattered (n = 2). Labeling was predominantly in proximal tubules (n = 6), less frequently in distal tubules (n = 3), and absent from any other levels of the nephron. Labeling in the glomeruli was significantly associated with the presence of viral antigen in the tubules (ρ = 0.112; P = .099).
Viral antigen was present in the cortical interstitial capillaries in 32 of 46 (70%) of the cases where glomerular labeling was also present, and this association was significant (ρ = 0.715; P < .001). Viral antigen was also present in the medullary interstitial capillaries (Fig. 20 and Suppl. Fig. S7a) in 31 of 46 (67%) of the cases, and the presence of viral antigen in the cortical interstitium was significantly associated with presence of viral antigen in the medullary interstitium (ρ = 0.748; P < .001). In both cortical and medullary interstitial capillaries, viral antigen was present as non-cell-associated antigen, in vascular endothelial cells or within cellular debris. In interstitial cortical capillaries, viral antigen was also present in mononuclear leucocytes. In 2 cases, viral antigen was not present in the glomeruli, tubules, or the cortical interstitial capillaries but instead occurred predominantly in medullary interstitial capillaries. Viral antigen was also present in capillaries and small blood vessels in the perirenal adipose tissue (Fig. 21).
Adrenal Gland
Lesions were infrequent, with a few necrotic cells present in the zona fasciculata in only 1 of 5 cases. RVFV antigen was present in secretory cells in all zones of the cortex in 3 of 5 cases (Fig. 22). Labeling was also present in capillaries and small blood vessels in the capsule and in periadrenal adipose tissues. Lesions and viral antigen were absent from the adrenal medulla.
Lung
Marked pulmonary edema and congestion accompanied by mild to moderate hydrothorax were common findings (Fig. 23). Microscopically, intra-alveolar and interstitial edema was present in 39 of 52 (75%) of the cases (Fig. 24). Inflammation, varying from mild to severe, was present in the alveolar capillaries in all cases and consisted of predominantly mononuclear cells. Pyknosis and karyorrhexis were often present in the alveolar septa and pulmonary blood vessels (Fig. 24). Hemorrhage was present in 5 of 52 (10%) of the cases.
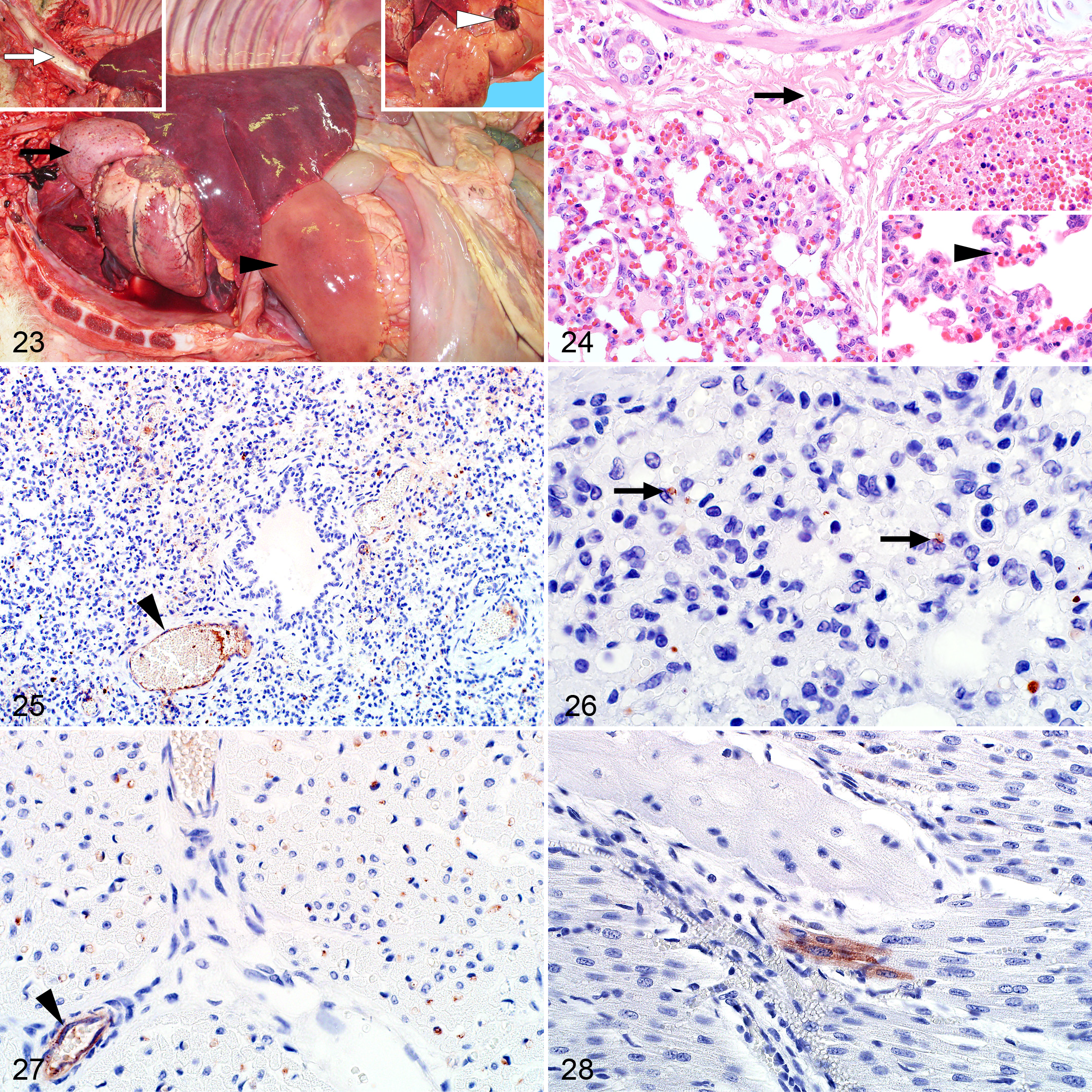
Labeling was widespread and easily identifiable in 46 of 52 (88%) of the cases (Fig. 25). Viral antigen was present in mononuclear cells, histomorphologically consistent with macrophages, within the interstitial capillaries of 43 of 52 (83%) cases (Fig. 26). Labeling was also present as non-cell-associated antigen or antigen associated with endothelial cells or cellular debris in small blood vessels and capillaries in 42 of 53 (79%) of the cases (Fig. 25 and Suppl. Fig. S9a). Antigen was also present in pulmonary vascular smooth muscle cells in 1 case.
Heart
Macroscopically, epicardial and endocardial hemorrhages as well as mild to moderate hydropericardium were present in most cases. However, histomorphological lesions attributable to RVFV infection were not present in the cardiac parenchyma in any of the cases with available specimens.
RVFV antigen was present in the heart of 27 of 36 (75%) of the cases. Viral antigen was typically non-cell-associated or within endothelial cells of the small blood vessels and capillaries (Fig. 27 and Suppl. Fig. S11a) and widespread in most cases (19/27). This included capillaries in pericardial adipose tissue. In 3 cases, viral antigen was also present in the smooth muscle cells of small blood vessels, and in 5 cases, single or small aggregates of cardiomyocytes were positive for viral antigen (Fig. 28 and Suppl. Fig. S10a). In 1 case, viral antigen was present in Purkinje fibers.
Gastrointestinal Tract
Peritoneal and abomasal mucosal hemorrhages were occasionally noted macroscopically. Microscopically, blood was sometimes present in the lumen of the abomasum (4/15; 27%) and the small intestine (2/19; 11%). Rare foci of necrosis were present in the lamina propria of the small intestine. In contrast, karyorrhexis of cells in the lamina propria, probably lymphocytes, was common. Karyorrhexis and pyknotic cellular debris was also present in lymphoid follicles in the Peyer’s patches (Fig. 29).
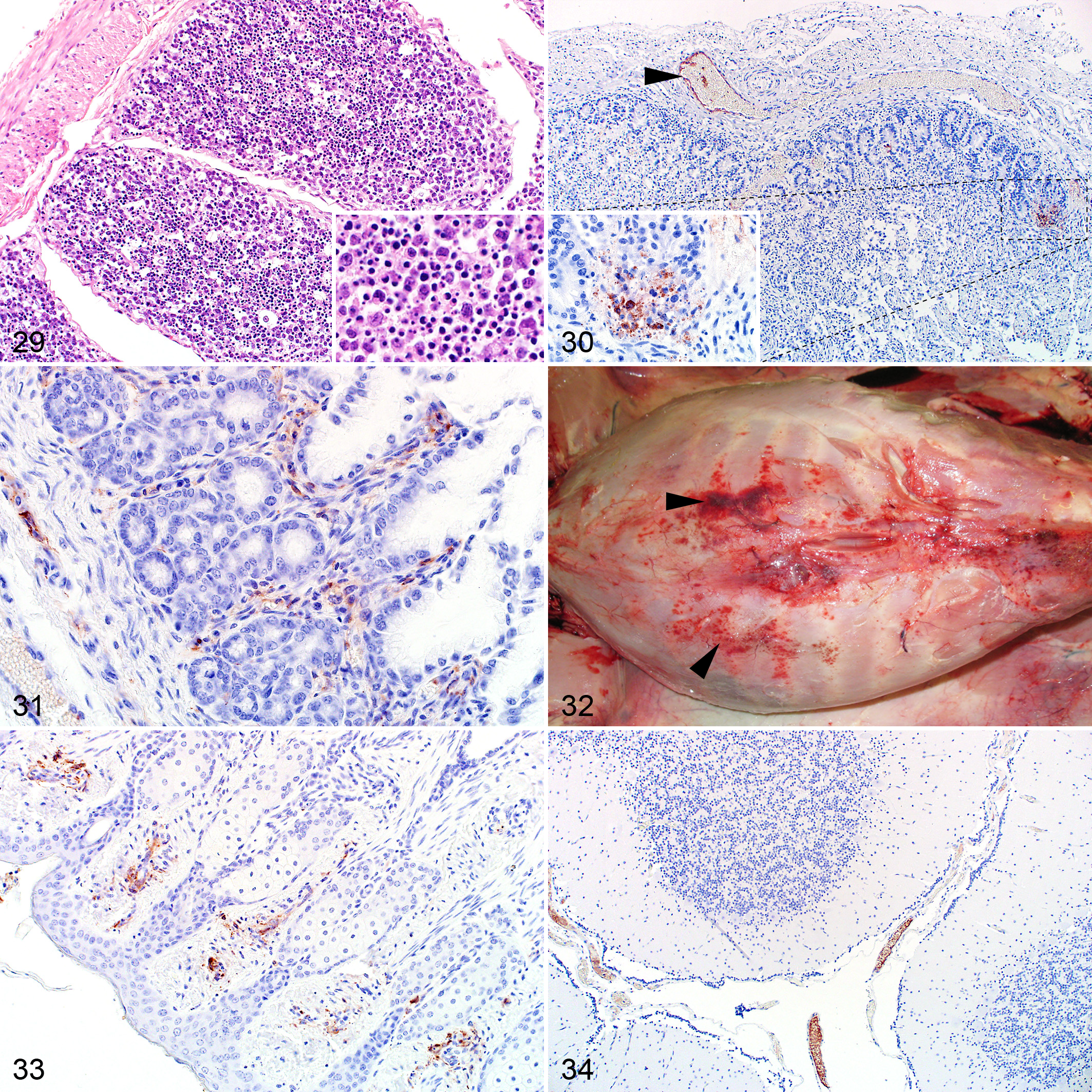
Viral antigen was present in vascular endothelial cells in small blood vessels and capillaries in the rumen, abomasum, small intestine, and tongue (Figs. 30, 31). In the small intestine, labeling was also associated with cellular debris in the lamina propria and present in small groups of mucosal epithelial cells.
Subcutis and Skin
Macroscopically, hemorrhages were commonly present in the subcutis (Fig. 32). Viral antigen was present in the superficial dermis in association with cellular debris or mononuclear cells. Positive labeling was also intravascularly associated with cellular debris or vascular endothelial cells (Fig. 33).
Nervous System
The only histological lesion present in tissues from the central nervous system was mild to moderate edema. Viral antigen was detected in 17 of 19 (89%) cases with available specimens. Positive labeling was mainly present in vascular endothelial cells or cellular debris in capillaries and small blood vessels in the meninges (Fig. 34 and Supp. Fig. S12a). Occasionally, viral antigen was present in capillaries in the white or gray matter but never within the brain parenchymal cells. Non-cell-associated viral antigen was often present within lumens of blood vessels. Peripheral nerve bundles, occasionally encountered in tissue sections, had viral antigen in their capillary endothelial cells; antigen was absent from nerve fibers in these locations.
Discussion
The severity and pathomorphology of RVF in young lambs are different from that in adult sheep. In particular, the lesions in the liver, kidneys, and lymphoid tissues differ remarkably. In addition, the distribution of RVFV antigen differs from what we previously reported in adult sheep from the same outbreak and in many instances from the same infected flock. 18 Therefore, the principal aims of this study were to describe the tissue tropism and target cells of RVFV in diverse organs from a large number of naturally infected young lambs less than 6 weeks old and to summarize the gross, histopathological, and immunohistochemical findings relative to results from previous research. 18 A further aim was to determine the extent to which virus can be detected in different organs, establish the diagnostic importance of different tissues, and characterize specific histologic features of RVF in lambs. The 2010 to 2011 RVF outbreak in South Africa provided a valuable opportunity to study the pathology and cellular tropism of RVFV in 71 young lambs and compare findings with previous reports.
Similarly to the adult cases published previously, the frequencies of lesions and immunolabeling could be an overestimate of the general population of RVF cases since organs were not consistently sampled in these field cases and there might have been a tendency to sample grossly abnormal organs. 18 Another limitation is that the exact age at which the pathomorphological appearance of lesions in young lambs begins to resemble those seen in adults is not known. In this study, the oldest lamb with histologic lesions and immunolabeling resembling that of young lambs was 6 weeks old. Unfortunately, there were no lambs with ages recorded as 7 or 8 weeks. The age at which lesions become less severe probably depends on the maturity of the immune system, nutritional status of the lamb, breed, farming practices, and perhaps the virus strain. Therefore, pathologists confronted with cases of RVF could encounter variations in the relationship between animal age and the lesions and pattern of immunolabeling.
Early in the 2010 outbreak, it was reported that veterinarians conducting necropsies on a number of lambs did not recognize the possibility of RVF until they came upon a carcass with clearly distinguishable miliary necrotic foci in the liver or hemorrhages in the abomasum, because these lesions are important features of RVF in South Africa (personal communication; 2010). Macroscopic lesions can be nonspecific in young lambs with RVF, because the characteristic liver lesions are sometimes obscured by congestion or discoloration of the liver due to diffuse necrosis or autolysis. Lesions suggestive of vascular endothelial injury can include hemorrhages in many organs, including the subcutis; hydropericardium, hydrothorax, and ascites; marked diffuse pulmonary congestion and edema; and edema in other organs, including the lymph nodes, gallbladder, or the mucosa of the abomasum. Previous research has reported that necrotic foci in the liver of 1- to 5-day-old lambs infected with RVFV can be obscured by general hepatic discoloration. 7 In that study, hemorrhages appeared late in the disease course and the only other lesions were lymphadenomegaly and histologically appreciated depletion of the white pulp of the spleen. Therefore, circumspection is required when conducting necropsies on young lambs in RVFV endemic areas.
Previous research also reported that liver lesions do not occur in young lambs prior to 24 hours after virus exposure but that virus can be detected in blood and liver samples as early as 12 hours postexposure. 5,7 Four lambs in the present study tested positive on RT-qPCR of the liver but were negative on IHC. The necrotizing hepatitis typical of RVF in young lambs was also absent in these cases. These lambs were possibly infected with RVFV and developed a viremia but died of other complications (eg, systemic inflammatory response syndrome) before lesions could develop in the liver and virus became detectable by IHC. Whatever the reason for the disparate test results, this demonstrates that the investigator, when confronted with high mortalities, should examine and sample multiple lambs if possible and might, on occasion, encounter discordant test results.
The pathogenic mechanism for RVFV-induced necrotizing hepatitis is likely hepatocellular death and the death of Kupffer cells or infiltrating macrophages by both apoptosis and lytic cell death mechanisms, including pyroptosis and necroptosis. The histomorphological changes in hepatocytes, described in previous research, concur with modern descriptions of apoptosis, and apoptosis was demonstrated in mice using terminal deoxynucleotidyl transferase dUTP nick-end labeling. 4 –7,31 In addition, the presence of NSs filaments in the nucleus induces DNA damage responses and causes cell cycle arrest, p53 activation, and apoptosis. 1 However, in the present study, many hepatocytes in RVFV-infected lambs also displayed histomorphological features of lytic cell death mechanisms, which are characterized by cell swelling, rupture of the plasma membrane, and cellular collapse. 2,16 Research has shown that RVFV induces the formation of nucleotide-binding domain, leucine-rich-containing family, and pyrin domain-containing-3 (NLRP3) inflammasomes that activate caspase 1, leading to the maturation of interleukin 1β and interleukin-18 and the induction of pyroptosis. 10 However, pyroptosis is considered a primary cellular response following the sensing of potentially damaging insults, whereas necroptosis is mostly observed as a backup cell death defense mechanism that is triggered when apoptosis is hindered. 13 It has been demonstrated that RVFV triggers apoptosis mainly through activation of caspase 8, but that the NSm protein of the virus suppresses apoptosis of target cells to ensure the efficient release of progeny virus within the first 24 hours after infection. 37 Therefore, the majority of the progeny virus is released prior to virus-induced apoptosis. Influenza A virus infection elicits concomitant activation of parallel pathways of mixed-lineage kinase domain-like protein (MLKL)–driven necroptosis and Fas-associated protein with death domain (FADD)–mediated apoptosis, while the NLRP3 inflammasome promotes host immune defense against influenza A virus infection, causing pyroptosis of infected epithelial cells, macrophages, and dendritic cells in the lung. 17,21 Macrophages also undergo NLRP3 inflammasome-mediated pyroptosis in other viral infections, including dengue virus and hepatitis C. 29,38 Macrophages play an important role in RVF pathogenesis, and conceivably, some of the cellular debris seen in necrotic foci in RVFV infection is composed of the remnants of macrophages. Therefore, it would seem that cell death morphology in the liver in RVFV-infected lambs is more heterogeneous than previously suspected, and the possibility that multiple cell death pathways are involved should be further explored.
Diagnostically, foci of liquefactive hepatic necrosis, referred to as primary foci in previous reports, are a very useful feature of RVFV infection in young lambs. 4,7 The term liquefactive necrosis is preferred since the primary foci are not typical of coagulative necrosis where the architecture of the hepatic plates is preserved, and features of other recognized forms of necrosis are absent. Previous research reported that lesions in the liver of lambs commenced as small aggregates of neutrophils in the sinusoids followed by the death of small groups of hepatocytes. 7 In lambs, this occurred at 24 to 36 hours after inoculation. Cell death progressed to the point where only remnants of completely lysed hepatocytes remained within a collapsed reticulum framework. Myriads of small nuclear remnants were scattered throughout the lesion, and infiltrating neutrophils and macrophages underwent degenerative changes and likewise disintegrated. The areas of necrosis enlarged but remained discrete or rarely became confluent, and they were accompanied by an infiltrate of more neutrophils and macrophages. Previous research also reported that between 36 and 48 hours postinfection, lesions were absent from the remainder of the liver parenchyma and hepatocytes outside the primary foci were histologically normal. 7 Precipitously, though, during the ensuing hours, all but a few hepatocytes underwent cell death so that the primary foci were a prominent feature against a background of widespread cell death. From previous research, it would seem that the initial sequence of events in adult sheep is the same, and histomorphologically random foci of cell death in adult sheep also consist of disintegrated hepatocytes and leucocytes, with infiltrating neutrophils and macrophages. 18 However, diffuse hepatocellular death rarely occurs in adult sheep. Qualitatively, lytic cell death with collapse of the reticulum framework was also less apparent in adult sheep. 18 Therefore, primary foci against a background of diffuse hepatocellular death is a striking diagnostic feature in young lambs. In addition, the mild portal edema and inflammation, characteristic of RVF in adult sheep, were not observed in young lambs likely due to disruption of the portal tract connective tissue by diffuse hepatic necrosis. 18
Previous research reported that the primary foci were more useful diagnostically than intranuclear inclusion bodies, with primary foci found in 100% of cases and nuclear inclusions in only 49%. 4 The present study had a similar ratio of 2:1 for primary foci (59/71) and nuclear inclusions (27/71). However, primary foci were present in only 86% of the cases in the present study. In most cases, only 1 liver specimen was submitted and only a single mixed-organ paraffin block was prepared for most cases to limit costs, so the liver specimens were often quite small. Also, it was previously reported that lesions in the liver can differ between specimens taken from the same animal. 18 Easterday et al 7 also reported that primary foci were difficult to find in some lambs. It is less likely that necrosis would be absent or overlooked, but primary foci might be missed if only 1 very small specimen is available for histologic examination. Therefore, sampling multiple liver lobes and combining fewer organs in a block might improve the odds of identifying this very useful diagnostic feature in naturally infected lambs.
Lymphocytolysis was present in all lymphoid organs except for the thymus, where resident T lymphocytes were completely unaffected in all cases. This strengthens a previous hypothesis that B lymphocytes, although not directly infected, are preferentially targeted in RVF. An alternative explanation is that developing lymphocytes in the thymus might be protected from the effects of RVFV infection by the blood-thymus barrier. Lesions and labeling in the spleen of young lambs also differed considerably from that of adult sheep. Previous research demonstrated that adult sheep generally had mature secondary follicles, and necrosis was most obvious in the germinal centers, mantle cell layers, marginal zones, and peripheral zones of the PALS of the white pulp (ie, B-cell-rich areas). 18 In the present study, lymphoid follicles were poorly developed (age appropriate in lambs), and necrosis primarily involved lymphocytes in the red pulp with fewer necrotic lymphocytes observed in the peripheral zones of the PALS. However, lymphocytolysis occurred in both white and red pulp at an equal frequency. Previously in adult sheep, positive labeling was observed more readily in the white pulp, particularly in the marginal zone. In young lambs, labeling was widespread and easily identifiable in the red pulp and the splenic capsule. In most of the lamb cases, labeling was observed in small blood vessels in the capsule, and foci of intense labeling were often present in the subcapsular red pulp. Labeling in or beneath the capsule was not seen in any of the adult cases sampled during the same outbreak. 18 Therefore, part of the splenic capsule should always be included in samples from young lambs.
Multifocal acute renal tubular injury, characterized by tubular epithelial cell pyknosis, karyorrhexis, and karyolysis and accompanied by intratubular cellular and proteinaceous debris and detachment of the epithelium from the basement membrane, which has been previously reported in adult sheep, was absent in young lambs. 18 Instead, in agreement with earlier research, some lambs (20/46) had varying degrees of degenerative (hydropic) changes in tubular epithelial cells. 4,6 Daubney et al 6 also reported that death ensues too rapidly following RVFV infection in lambs to permit the development of extensive lesions. In contrast, the previously reported pyknosis and karyorrhexis present in the glomeruli and interstitial capillaries, similar to previous findings in adult sheep, was a useful diagnostic feature. 4,18
In RVFV antigen-positive adult sheep, labeling was often present in the renal tubules (33/55; 60%) and widespread in 9 of these, whereas labeling was isolated or scattered when present at all in lambs (8/40; 20%). 18 Conversely, in the kidneys, viral antigen was more often present in vascular smooth muscle cells in young lambs (26/46; 57%) compared to adult sheep (5/83; 6%). 18 In young lambs, smooth muscle labeling was present in small arteries in the cortex, in the efferent or afferent arterioles, and in arcuate arteries at the corticomedullary junction. In the adult cases, the labeling was only present in the muscular tunic of arcuate arteries at the corticomedullary junction. 18 Viral antigen was also more commonly observed in endothelial cells in capillaries and small blood vessels in the young lambs than in adult sheep. Possibly young lambs do not survive long enough for virus to spread much beyond the blood vasculature in the kidney, and due to the much higher viremia in lambs, RVFV seemingly infects smooth muscle cells and endothelial cells more extensively. 7,8
Another prominent feature in the kidney was viral antigen in a small group of cells opposite the macula densa. This was interpreted as antigen occurring in the perimacular cells that include juxtaglomerular and granular extraglomerular mesangial cells. Smooth muscle cells in the afferent arterioles transition into granular extraglomerular mesangial cells at this location. Juxtaglomerular mesangial cells have also been demonstrated to be modified smooth muscle cells capable of contracting or relaxing in response to vasoactive agents. 26 Kidney specimens from 2 of 83 adult sheep examined retrospectively had a similar pattern of labeling. 18 In addition, van der Lugt et al 35 also found RVFV antigen in mesangial cells in 2 of 12 newborn lambs. In mice, it was experimentally demonstrated that adeno-associated virus 9 exclusively targets the juxtaglomerular apparatus and specifically cells opposite the macula densa. 39 It is possible that vascular smooth muscle cells and the renal perimacular cells share receptors that make them both permissible for RVFV infection.
Infection of granular extraglomerular mesangial cells might suppress renin production, which could have a deleterious effect on the regulation of glomerular filtration rate and renal blood flow. Infection of aldosterone-secreting cells in the zona glomerulosa of the adrenal gland could potentiate this effect. RVFV infection of secretory cells in all zones of the adrenal cortex seemed to be more pronounced in young lambs compared to adult sheep, in which it was most prominently seen in secretory cells of the zona fasciculata. 18 The normal physiologic response to hypotension might be suppressed due to RVFV infection and, combined with vascular leakage, could exacerbate shock. In Dengue hemorrhagic fever, the most severely affected children develop dengue shock syndrome due to excessive depletion of intravascular volume as a result of plasma leakage, accompanied by only minor bleeding manifestations. 36 Furthermore, RVFV infection of glucocorticoid-producing cells in the zona fasciculata of the adrenal gland could also decrease the metabolic availability of glucose and fatty acids, further increasing the likelihood of a fatal outcome. However, additional studies would be needed to validate these speculations.
Previous research reported that the peak level of virus in the blood of young lambs is consistently greater than in adults. 8 This concurs with the present investigation, where viral antigen was generally widespread and easily detectable in the vasculature of young lambs, while in adult sheep, labeling could be difficult to find in organs other than the liver. 18 Previous research in young lambs also reported that by 24 hours postinfection, virus titers were high in the liver, spleen, lymph nodes, kidneys, lungs, and brain, and the authors inferred that the virus in the tissues was largely in the blood of these organs. 7 Findings in the present study support that conclusion, since viral antigen was often present in capillaries and small blood vessels as non-cell-associated viral antigen or antigen associated with intravascular cell fragments. This was especially noticeable within the splenic capsule; interstitial tissues of the kidney, heart, lungs, and lymphoid tissues; meninges of the brain; adipose tissue; and the lamina propria and submucosa of the abomasum and small intestine. Therefore, samples from the liver and spleen should be sufficient to confirm a diagnosis of RVFV in young lambs using PCR or IHC (Table 5). Samples from the lung and kidney would be useful additional samples for histopathology, because pyknosis and karyorrhexis in the capillaries of the glomeruli and pulmonary interstitium were often present. Samples from the thymus and brain, although likely to test IHC positive for viral antigen in RVFV-infected lambs, do not have any characteristic histologic lesions and therefore were not considered essential for confirming the diagnosis.
Preferred Diagnostic Specimens From Young Lambs for Investigation of Rift Valley Fever Using Histopathology and Immunohistochemistry.
Abbreviation: RVFV, Rift Valley fever virus.
Conclusion
The principal histologic lesion observed in young lambs naturally infected with RVFV was diffuse necrotizing hepatitis with prominent foci of liquefactive necrosis (primary foci). Hepatic lesions were typically accompanied by lymphocytolysis in lymphoid tissues with the notable exception of thymus as well as pyknosis and karyorrhexis in the glomerular and pulmonary interstitial capillaries. In addition, lesions in the kidney rarely progressed beyond hydropic change and occasional pyknosis or karyolysis of tubular epithelial cells. Therefore, histological review of tissues from lamb deaths has an important role in RVF diagnosis.
Liver and spleen specimens were the most consistently positive for RVFV antigen and were adequate to confirm or exclude a diagnosis of RVFV in most cases using either PCR or IHC. Specimens from the lungs and kidneys were useful additional samples due to their characteristic histologic lesions. Rarely, PCR-positive lambs did not have typical histological lesions or viral antigen detectible by IHC in their tissues. However, RVFV causes high mortality in young lambs, and provided adequate samples are taken from multiple cases, the diagnosis should be straightforward.
The presence of RVFV antigen in vascular smooth muscle cells, vascular endothelial cells, adrenocortical epithelial cells, and renal perimacular cells was more prominent in the young lambs in this study compared to the adult sheep cases from the same outbreak that were studied previously. We hypothesize that these cells are more permissible to RVFV infection in very young animals, which would worsen clinical outcomes, such as hepatic and renal failure and shock, and contribute toward an increased likelihood of a fatal outcome to infection.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Odendaal_et_al - Lesions and Cellular Tropism of Natural Rift Valley Fever Virus Infection in Young Lambs
Supplemental Material, Combined_supplemental_materials-Odendaal_et_al for Lesions and Cellular Tropism of Natural Rift Valley Fever Virus Infection in Young Lambs by Lieza Odendaal, A. Sally Davis, Geoffrey T. Fosgate and Sarah J. Clift in Veterinary Pathology
Footnotes
Acknowledgements
We thank staff of the histopathology laboratory of the Department of Paraclinical Sciences, Section of Pathology, especially Rephima Phaswane and Naomi Timmerman for their assistance, knowledge, input, and support. We thank colleagues at the National Department of Agriculture, Fisheries and Forestry, Directorate of Veterinary Services for providing access to original diagnostic data and colleagues at IDEXX laboratories (South Africa) for the specimens. We also thank Antoinette Odendaal for preparing all the panels for this manuscript. The authors are also grateful for the support of Professor Estelle Venter from the College of Public Health, Medical and Vet Sciences, James Cook University, Australia, and the Department of Veterinary Tropical Diseases, Faculty of Veterinary Science of the University of Pretoria.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose receipt of the following financial support for the research, authorship and/or publication of this article: The Institutional Research Theme, Biotechnology and the Management of Animal and Zoonotic Diseases, of the University of Pretoria and the Agricultural Sector Education Training Authority, South Africa. Publication of this study was funded by the Department of Paraclinical Sciences of the Faculty of Veterinary Science, University of Pretoria and the Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, of Kansas State University.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
