Abstract
Rift Valley fever (RVF) is a mosquito-borne disease that affects both ruminants and humans, with epidemics occurring more frequently in recent years in Africa and the Middle East, probably as a result of climate change and intensified livestock trade. Sheep necropsied during the 2010 RVF outbreak in South Africa were examined by histopathology and immunohistochemistry (IHC). A total of 124 sheep were available for study, of which 99 cases were positive for RVF. Multifocal-random, necrotizing hepatitis was confirmed as the most distinctive lesion of RVF cases in adult sheep. Of cases where liver, spleen, and kidney tissues were available, 45 of 70 had foci of acute renal tubular epithelial injury in addition to necrosis in both the liver and spleen. In some cases, acute renal injury was the most significant RVF lesion. Immunolabeling for RVFV was most consistent and unequivocal in liver, followed by spleen, kidney, lung, and skin. RVFV antigen-positive cells included hepatocytes, adrenocortical epithelial cells, renal tubular epithelial cells, macrophages, neutrophils, epidermal keratinocytes, microvascular endothelial cells, and vascular smooth muscle. The minimum set of specimens to be submitted for histopathology and IHC to confirm or exclude a diagnosis of RVFV are liver, spleen, and kidney. Skin from areas with visible crusts and lung could be useful additional samples. In endemic areas, cases of acute renal tubular injury should be investigated further if other more common causes of renal lesions have already been excluded. RVFV can also cause an acute infection in the testis, which requires further investigation.
Keywords
Rift Valley fever (RVF) virus (RVFV) was first reported in sheep in 1931 in the Rift Valley of Kenya and is a zoonotic disease that causes considerable morbidity and mortality in ruminants and occasionally humans. 11,33 It is caused by a mosquito-borne, single-stranded RNA virus of the Bunyavirales order, Phenuiviridae family (formerly the Bunyaviridae family), genus Phlebovirus. Epidemics of RVF are more and more frequent in Africa and the Middle East and generally occur during years of abnormally high rainfall when sufficient numbers of susceptible livestock congregate in areas containing RVFV-infected mosquitoes. 7,19,41 Livestock trade contributes to the spread of the disease into disease-free areas, thereby expanding the geographical distribution of RVF. 7 Susceptibility to RVFV varies with age, with mortality rates typically 90% or higher in young lambs and 20% to 30% in older lambs and ewes. 15 Adult sheep often die without showing any definite signs, and abortion in ewes is often the only indication of infection in a flock. 15
Few studies of the gross and histopathological lesions in sheep that died of natural RVFV infection have been published. 9,11,38,39,43 These studies reported that sheep, including lambs that die from natural RVFV infection, have the most significant lesions in the liver but that lesions can also occur in other organs, including the spleen, lymph nodes, kidneys, lungs, heart, adrenal glands, gall bladder, skin, and digestive tract. 9,11,38,43 Necrotic foci are distributed more or less evenly throughout the liver and can coalesce to form diffuse necrotic lesions in lambs. 11 Additionally, adult sheep occasionally have marked renal tubular epithelial degeneration and necrosis. 11,16,17,31 Schultz 38 reported that the lesions of RVF in South African sheep differed from those previously described by Daubney et al. 11 Daubney et al 11 had little to remark about microscopic lesions in the spleen and lungs, whereas Schultz 38 reported necrosis in the white pulp of the spleen and edema, emphysema, and hemorrhages in the lungs of the South African cases. Schultz 38 also reported cortical and medullary hemorrhages in the adrenal glands and subcutaneous hemorrhages, whereas Daubney et al 11 did not mention adrenal or subcutaneous lesions. However, neither Daubney et al 11 nor Schultz 38 reported the total number of animals or the numbers in the various age groups that were examined.
The cellular tropism of RVFV was studied using immunohistochemistry (IHC) in 8 experimentaly infected and 4 naturally infected newborn lambs, wherein extensive hepatocyte necrosis and a progressive increase in viral antigen in the liver was reported. 43 Viral antigen was also reported in a few cells in the spleen, lymph nodes, kidneys, and lungs. In a study of naturally infected sheep, RVFV antigen was identified by IHC in liver from 1 sheep and autolysed spleen from another sheep. 39 In a single experimentally infected 3- to 4-month-old sheep, viral antigen was most prominent in the liver and the spleen, with lesser amounts also detected in the adrenal glands and within renal tubular epithelial cells and luminal debris in the kidney. 16 In a trial of a RVFV vaccine candidate, viral antigen was reported in the liver, spleen, lymph nodes, kidney, lungs, and adrenal glands of the unvaccinated sheep. 17 Viral antigen-positive macrophages or dendritic cells were detected in the lungs and medullary sinuses of mesenteric lymph nodes. There was also evidence of viral antigen in renal glomeruli and rarely in the adrenal cortex. In a study involving 40 lambs, of which 24 were experimentally infected with RVFV, antigen was observed in hepatocytes and mononuclear phagocytes in the liver, spleen, and lymph nodes as well as endothelial cells in lymphoid organs. 45 RVFV antigen has also been observed in the skin of 2 experimentally infected sheep. 44 Notwithstanding, in all these studies, the tropism and target cells of RVFV were only partly revealed, making future observations in similar studies difficult to contextualize.
The aim of this study was to describe the tissue tropism and target cells of RVFV in a large number of naturally infected sheep, specifically 99 sheep from the 2010 South African RVFV outbreak. A further aim was to determine the extent to which virus can be detected in different organs and summarize gross, histopathological, and immunohistochemical findings in the present study relative to results from previous research.
Materials and Methods
Outbreak Description
In the summer of 2010, South Africa had an extensive outbreak of RVF affecting livestock and humans. 29 A diagnosis of RVF was confirmed using reverse transcription (real-time) quantitative polymerase chain reaction (RT-qPCR) and in selected cases also confirmed by virus isolation and IHC. The outbreak affected mainly sheep (13 117 cases reported), but cattle, goats, African buffaloes, camelids, and other wild animals were also reportedly affected. 35
Case Selection
All specimens originated from the carcasses of naturally infected sheep necropsied during the 2010 RVF outbreak in South Africa. This work was done in agreement with the Animal Ethics Committee of the University of Pretoria (clearance certificate V096-16). Supplementary material in support of the results presented in this article is available from the authors on request.
Tissue specimens were collected from hundreds of sheep during the outbreak and submitted to the Agriculture Research Council-Onderstepoort Veterinary Institute for testing, including RT-qPCR on 1617 specimens. Tissues collected in formalin from 1034 necropsied animals were submitted for histopathological examination. These necropsies were conducted by employees of the South African State Veterinary Services and private veterinary practitioners. With permission from the State Veterinary Services, the principal author conducted necropsies on 97 animals that included 37 adult sheep. Tissue specimens from these cases were collected in 10% neutral buffered formalin and paraffin embedded. Ultimately, FFPE tissue specimens were available from 306 adult sheep. From this collection, cases were excluded when the level of autolysis was severe or only 1 organ was available for study. A total of 124 cases were suitable for study, of which 99 cases were classified positive for RVF with 1 or more positive test results for histopathology, RT-qPCR, and/or IHC. Sheep that died of natural infections during the outbreak and examined for RVFV as part of the government campaign to monitor the spread of RVF but tested negative were included as controls (n = 25). Immunohistochemistry has a sensitivity and specificity of 97.6% and 99.4%, histopathology of 94.6% and 92.3%, and RT-qPCR of 97.4% and 71.7%, respectively. 29 A parallel interpretation of tests was chosen to maximize sensitivity, and therefore, any animal with at least 1 positive test result was selected for study.
Diagnostic Tests
Nucleic acid extractions and RT-qPCR on fresh liver for 81 of the cases were performed at the Biotechnology PCR Laboratory of the Agriculture Research Council-Onderstepoort Veterinary Institute as described elsewhere. 29 Embedding, sectioning, and hematoxylin and eosin (HE) staining of formalin-fixed tissues were done according to the standard operating procedures of the Section of Pathology, Department of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria. Immunohistochemical labeling for the detection of RVFV antigen was performed using polyclonal hyperimmune mouse sera (National Institute for Communicable Diseases, Johannesburg, SA) as described previously. 29 Briefly, the immunoperoxidase method included microwave antigen retrieval in citrate buffer (pH 6.0), incubation with the anti-RVFV primary antibody at 1:500 dilution for 1 hour and rabbit anti-mouse secondary antibody (F0232, DakoCytomation, Glostrup, Denmark), followed by detection with a standard avidin-biotin peroxidase system, Vectastain Elite ABC-HRP Kit (PK-6100, Vector Laboratories, Inc, Burlingame, CA, USA), NovaRED peroxidase substrate (SK-4800, Vector Laboratories, Inc), and hematoxylin counterstain.
Examination of Tissues
Histomorphological features in all available organs were systematically recorded and reviewed within the context of lesions associated with RVFV infection. Two authors (LO, SJC) reviewed 20% of the cases together to validate data collection methods, and the remainder were examined by a single author (LO). Collected data were validated by the independent review of 30% of the cases by another author (ASD) prior to finalizing the data set for analysis.
The number and types of with liver (n = 124) organs available for study varied considerably by case, with liver (n = 124), spleen (n = 110), and kidney (n = 102) being the specimens most often submitted. In some cases, additional tissue specimens were available: 87 lung, 67 heart, 40 gastrointestinal tract, 23 gall bladder, 20 lymph node, 13 skin, 13 brain, 11 adrenal, 10 uterus, and 2 testis specimens. Gastrointestinal tract specimens included forestomach (n = 5), abomasum (n = 24), small intestine (n = 20), large intestine (n = 2), and tongue (n = 12).
Lesion severity in the liver, spleen, and kidney was scored using a qualitative scale as mild, moderate, or severe (Supplemental Tables S1, S2, and S3). Immunolabeling for RVFV antigen was scored on a semi-quantitative scale as isolated, scattered, or widespread labeling. In the liver, spleen, and kidney, an area of tissue measuring approximately 5 mm2 was evaluated. For all other tissues where labeling tended to be isolated (gall bladder, lung, heart, intestines, tongue, adrenal glands, skin, lymph nodes, uterus, and testis), all the available tissue was examined. Isolated labeling was defined as positive signal that was only rarely present in the organ parenchyma and involved a total cell count of less than 20. These could be single cells or multifocal groups of cells. In cases with scattered labeling, positive signal was occasionally present in single cells or groups of cells, and the total cell count was between 21 and 100. Widespread labeling was defined as positive signal involving greater than 100 cells in the examined area of tissue.
Statistical Analysis
Categorical data were described as frequencies, proportions, and 95% mid-P exact confidence intervals (CI). Semi-quantitative data were described using the median and interquartile range. Paired categorical data were compared using McNemar’s chi-square tests. Semi-quantitative data were compared between the liver and all other organs using Wilcoxon rank sum tests. Correlations between semi-quantitative variables were estimated using Spearman’s rho (ρ). Statistical analyses were performed in available software (Epi Info, version 6.04, CDC, Atlanta, GA, and IBM SPSS Statistics Version 23, International Business Machines Corp, Armonk, NY, USA) and results interpreted at the 5% level of significance.
Results
Overview
Immunolabeling for RVFV was most consistent and unequivocal in liver, followed by spleen, kidney, lung, and skin (Table 1). Positive labeling was cytoplasmic and typically fine diffuse to coarse granular. Labeling was widespread in 68 of 82 (83%) of the liver specimens that tested positive for RVFV by IHC. In comparison, labeling in the spleen, kidney, and lung was only widespread in 9 of 63 (14%), 11 of 55 (20%), and 6 of 40 (15%) of the IHC-positive cases, respectively, and difficult to find in many cases. Notably, 6 of 11 (55%) of the skin specimens were positive for RVFV antigen. Neither histological lesions nor RVFV antigen were detected in any tissues from the central nervous system (n = 13) or in any tissues of the 25 cases included as negative controls.
Frequency of Viral Antigen in Different Organs and Comparison to Quantity of Immunolabeling in Liver, for 99 Sheep With Rift Valley Fever Virus Infection.a
a Pos/total represents number with positive immunolabeling for Rift Valley fever viral antigen out of the total number of tissues examined. CI, confidence interval; IQR, interquartile range; NA, not applicable.
b Calculated only for the cases with positive labeling; the categories in parentheses are the interquartile range of antigen quantity in the sample set.
c Based on Wilcoxon rank sum tests comparing the quantity of positive immunolabeling to the quantity of labeling in liver samples. P value corresponds to McNemar’s chi-square test comparing the proportions of positive immunolabeling when no cases had detectable labeling.
Liver
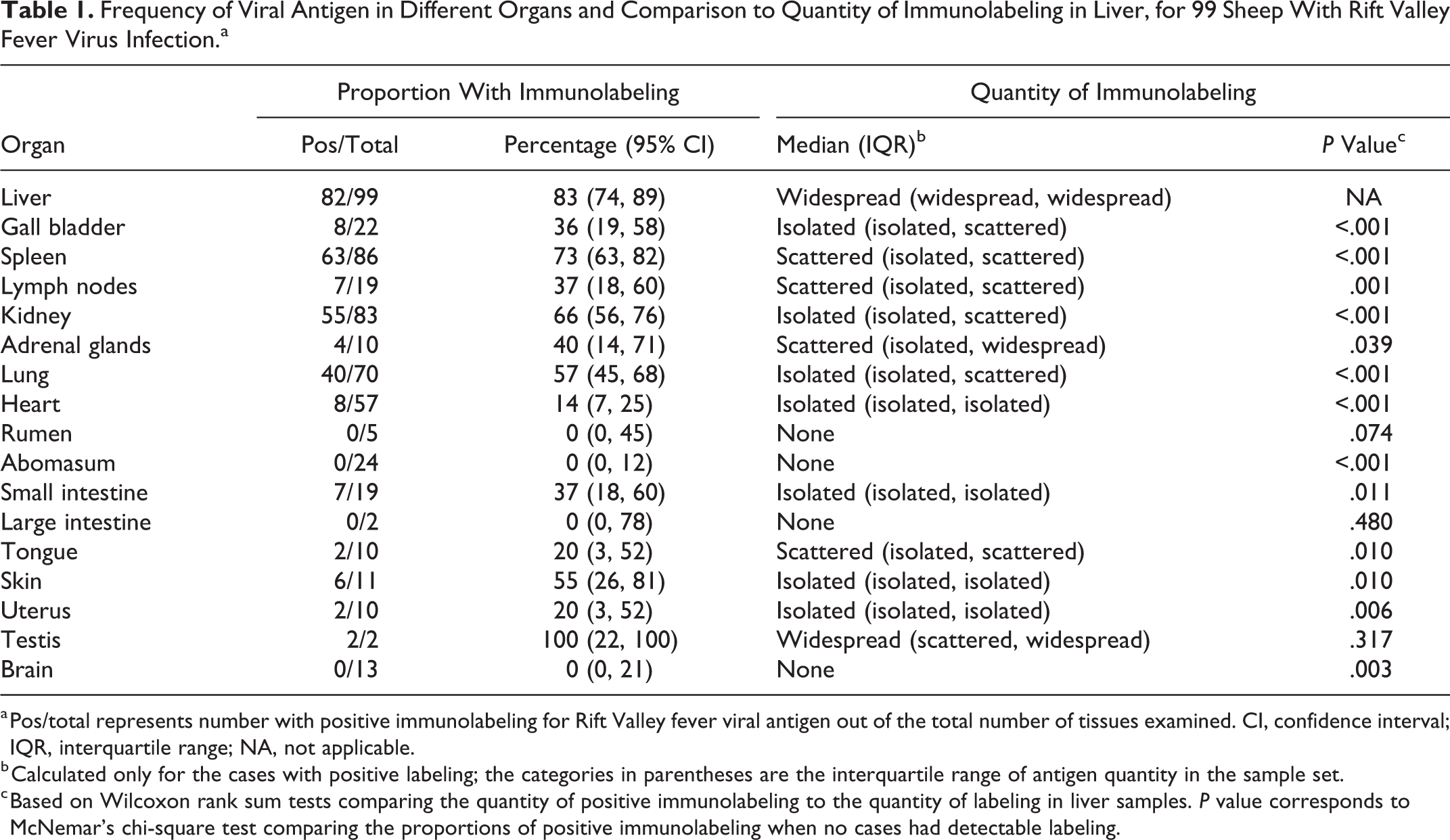
Macroscopically, the liver often had a mottled red appearance (Fig. 1) due to the presence of innumerable ill-defined necrotic foci. Microscopically, necrotic foci were situated irregularly throughout the lobule, and their distribution varied from multifocal to focally extensive to confluent bridging. Necrosis was generally accompanied by hemorrhage (Fig. 2) and a mild to moderate infiltrate of neutrophils and macrophages. Typically, the portal tract interstitium was expanded by mild edema and low numbers of mononuclear cells exhibiting occasional karyorrhexis (Fig. 3). The bile ducts were uninvolved. Many hepatocytes had features of apoptosis (Figs. 3, 4) that included dissociation of cells, cellular shrinkage and rounding, hypereosinophilic cytoplasm, pyknosis and karyorrhexis, and fragmentation of cells into multiple smaller apoptotic bodies. Adjacent hepatocytes often had varying degrees of micro- or macro-vesicular degeneration and anisokaryosis. Scattered hyperplastic Kupffer cells were occasionally present within adjacent sinusoids. Filamentous or oval eosinophilic intranuclear inclusions associated with nuclear chromatin marginalization were unequivocally observed in the hepatocytes of only 7 cases (Fig. 4).
Necrosis was absent in the liver specimens from 10 of 99 (10%) cases. Of these, 7 were positive by RT-qPCR. Notably, for the other 3 cases, no histological lesions or immunohistochemical labeling were present in the liver or in the spleen of 2 cases with available specimens; however, scattered (n = 2) or widespread (n = 1) RVFV antigen and acute tubular injury were present in the kidneys, and 2 of these 3 cases were also positive by RT-qPCR of the liver.
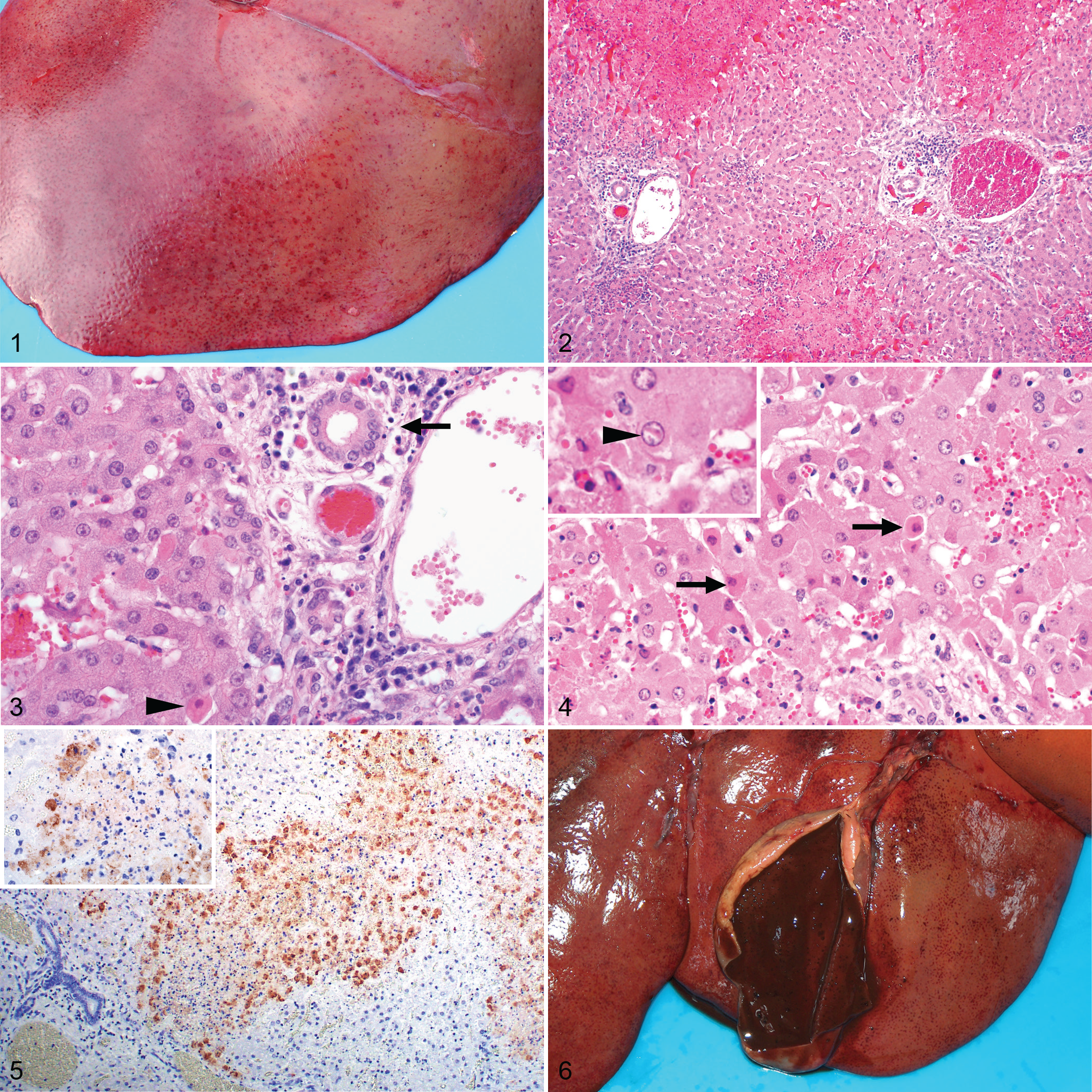
The severity and distribution of hepatic necrosis varied considerably among cases with 34, 23, and 32 specimens classified as mild, moderate, or severe necrosis, respectively. Most often, the patterns of necrosis (Table 2) were predominantly centrilobular (27/99), predominantly confluent centrilobular to midzonal (27/99), or multifocal random (23/99). However, necrosis was never limited to 1 particular zone and was found in multiple zones whenever necrosis was recorded. Instead, there could be confluent centrilobular necrosis with a few midzonal and periportal foci, or patchy involvement of the centrilobular zones with prominent necrosis midzonally and in the periportal areas, or a random distribution of necrotic foci throughout the lobule, or massive necrosis involving the entire hepatic lobule. Additionally, foci of necrosis were invariably present in 1 or more of the periportal zones, specifically affecting hepatocytes of the limiting plate.
Predominant Histologic Patterns of Liver Necrosis in 99 Sheep With Rift Valley Fever Infection.a
a Proportion represents number affected out of the total number of tissues examined. CI, confidence interval.
Hepatocytes were the predominant target of RVFV infection in the liver (Fig. 5), with IHC labeling widespread to scattered (82/99) and easily identified in most cases. Diffuse, fine to coarse granular labeling was present in the cytoplasm of hepatocytes, and viral antigen-positive cytoplasmic fragments were frequently present within the sinusoids and central veins. Areas of hepatocellular death were thought to become devoid of immunolabeling as lesions became more extensive. Positively labeled Kupffer cells and degenerate neutrophils were sparse. Immunolabeling was not detected in nuclei or in association with intranuclear inclusion bodies.
Gall Bladder
Hemorrhage and edema were occasionally present macroscopically in the gall bladder (Fig. 6). Microscopically, 10 of 22 (45%) cases with available specimens had foci of necrosis and hemorrhage. Typically, hemorrhage was prominent in the serosa or adventitia, but sometimes it extended into the muscularis externa or the lamina propria. Foci of necrosis, present in the lamina propria, serosa, or adventitia, had a mild to moderate infiltrate of mononuclear cells and degenerate neutrophils. There was no significant correlation between liver necrosis and gall bladder necrosis (ρ = 0.132; P = .549).
Immunolabeling for RVFV antigen was present in 8 of 22 (36%) of the cases. The labeling was generally in or around capillaries or small blood vessels and included cellular debris, neutrophils, or mononuclear cells. Areas of necrosis did not contain viral antigen in 3 of 22 cases, and in 1 of 22 cases, the labeled cells were not within areas of necrosis.
Spleen
Gross lesions were uncommon in the spleen, with capsular and subcapsular petechiae present occasionally. Microscopically, there were varying degrees of necrosis in the white and red pulp. This was a prominent feature in 64 of 86 (74%) of the cases, giving the majority of the specimens a paucicellular appearance histologically (Fig. 7). Necrosis was most apparent in the germinal centers, mantle zones, marginal zones, and peripheral zones of the periarteriolar lymph sheaths (PALS) of the white pulp and was characterized by the presence of cellular debris and tingible-body macrophages (Fig. 8). Necrosis was mild (21/63), mainly involving the follicular germinal centers; or moderate (25/63), with prominent necrosis of the germinal centers and the mantle zone and some involvement of the marginal zone and peripheral zone of the PALS; or severe (18/63), with prominent involvement of the follicles, marginal zones, and peripheral zones of the PALS. Necrosis was never prominent in the T-cell rich area of the PALS adjacent to the tunica media of the central artery. Lymphocytolysis in the red pulp was typically far less prominent and generally occurred as single cell necrosis.
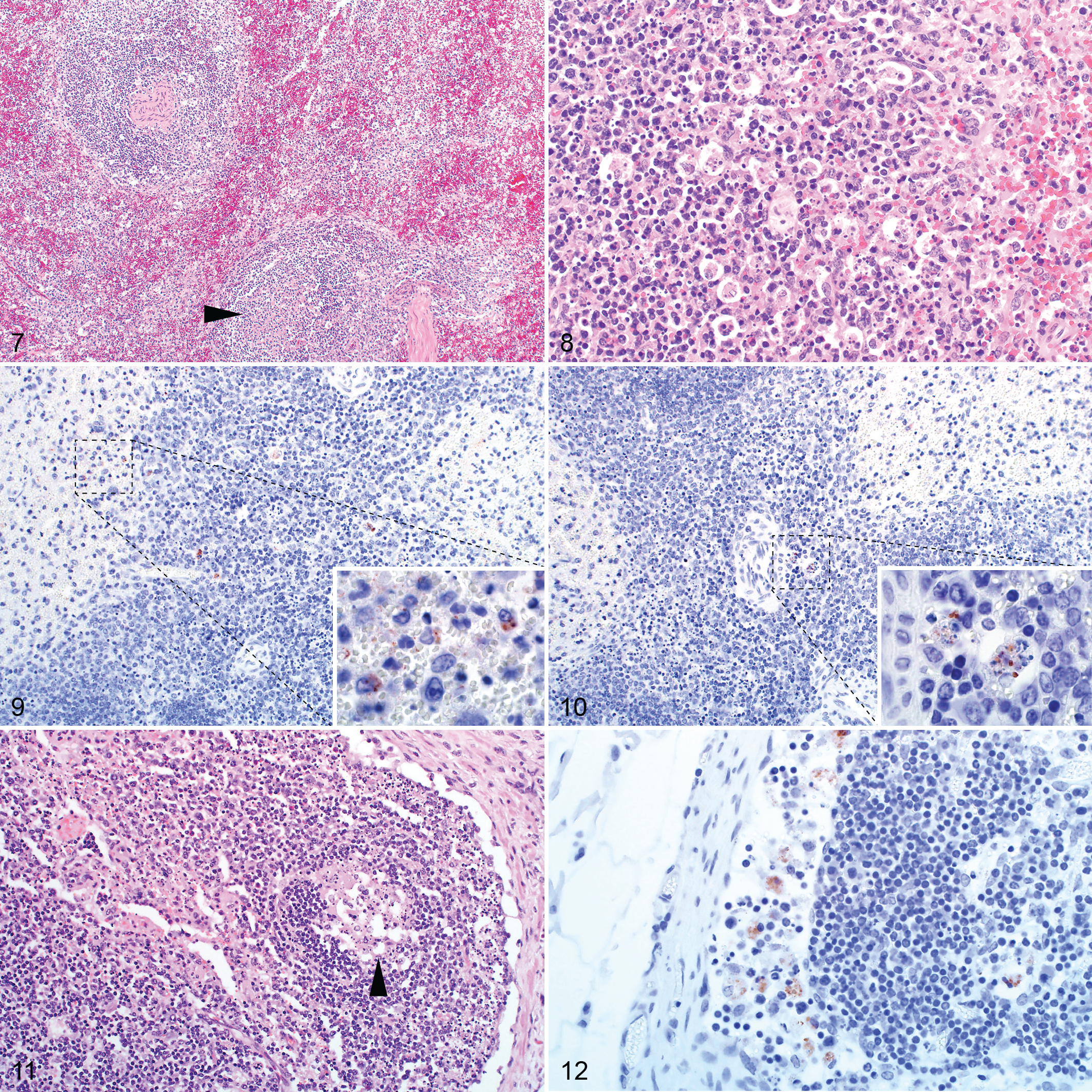
Splenic infection with RVFV was detected by IHC in 63 of 86 (73%) cases with available specimens. Viral antigen was either isolated (n = 30), scattered (n = 24), or widespread (n = 9), and the quantity of labeling was significantly associated with the degree of splenic necrosis (ρ = 0.505, P < .001). Viral antigen was mainly in cellular debris or in large mononuclear cells that had abundant, often clear cytoplasm and oval to polygonal, often slightly eccentric nuclei, histomorphologically identified as macrophages (Fig. 9). Positive labeling was observed more readily in the white pulp, particularly in the marginal zone, which was positive in 59 of 63 (94%) of the cases with IHC-positive spleen specimens (Table 3). The red pulp was positive in 58 of 63 (92%) of the cases with IHC-positive spleen specimens. Interestingly, viral antigen was absent from the white pulp in 4 specimens and instead was widespread only in the red pulp in these cases. In 21 of 63 (33%) of the cases with IHC-positive specimens, sparse labeling was present in cells in the follicles. These cells were polygonal with abundant cytoplasm and large nuclei and contained phagocytosed cellular debris in the cytoplasm, consistent with tingible-body macrophages (Fig. 10). However, not all cells identified as tingible-body macrophages contained RVFV antigen.
Distribution of Viral Antigen in Different Zones of the Spleen for Specimens With Positive Immunolabeling for Rift Valley Fever Virus in Sheep.a
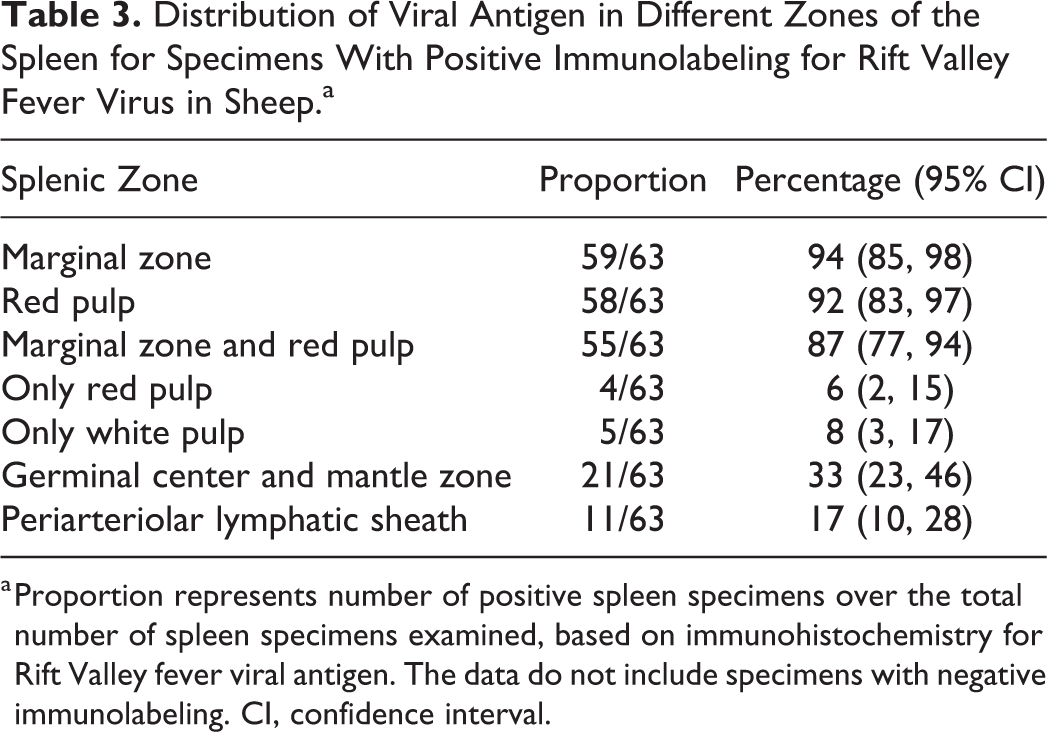
a Proportion represents number of positive spleen specimens over the total number of spleen specimens examined, based on immunohistochemistry for Rift Valley fever viral antigen. The data do not include specimens with negative immunolabeling. CI, confidence interval.
Notably, splenic necrosis was absent from 22 of 86 (26%) of the cases classified as positive for RVF. Test results for these cases varied with 13 testing negative for viral antigen by IHC but positive on RT-qPCR and/or liver histology. Of these cases, 4 of 22 had either widespread (n = 3) or isolated (n = 1) positive labeling for RVFV antigen in the spleen as well as widespread necrosis and viral antigen in the liver. A further 2 of 22 cases had no detectible necrosis or viral antigen in the liver or the spleen but were IHC-positive in the kidney. The remaining 3 cases with no detectible necrosis or labeling in the spleen had necrosis and viral antigen in the liver.
Lymph Nodes
Macroscopically, lymph nodes were enlarged, edematous, and congested. Mesenteric lymph nodes were often especially affected. Microscopically, there was lymphoid necrosis characterized by varying degrees of follicular lymphocytolysis and cortical lymphoid depletion in 10 of 19 (53%) of the cases with available specimens. Necrosis in lymph nodes was significantly associated with splenic necrosis (ρ = 0.532, P = .023). In most cases and similar to the spleen, lymphocytes within follicular germinal centers were especially depleted (Fig. 11). In some cases, there was also mild to moderate depletion of the mantle zone lymphocytes accompanied by scattered lymphocytolysis in the interfollicular cortex and the paracortex. In severe cases, the entire cortex was depleted, and no distinction could be made between follicles, interfollicular cortex, and paracortex.
Of the cases with available lymph node specimens, 7 of 19 (37%) were positive for RVFV antigen. Viral antigen was present in scattered mononuclear cells histomorphologically consistent with macrophages in the subcapsular, paratrabecular, and medullary sinuses (Fig. 12).
Kidney
Gross lesions in the kidney were uncommon other than occasional capsular or cortical petechiae or ecchymoses. Microscopically, multifocal acute tubular epithelial injury lacking significant associated inflammation was frequently present (68/83 cases). Severe injury was present in 30 of 83 (36%) of the cases while moderate and mild injury occurred in 14 of 83 (17%) and 24 of 83 (29%), respectively. There were no lesions in the remaining 15 kidney cases even though 6 of these tested positive by IHC. Occasionally, hemorrhages were present in the glomeruli or the interstitium. Scattered nuclear pyknosis and karyorrhexis was present in the glomeruli and accompanied by occasional neutrophils in 63 of 83 (76%) of the cases (Fig. 13). Glomeruli often appeared less densely cellular than normal. Acute tubular injury was significantly associated with the degree of pyknosis and karyorrhexis in the glomeruli (ρ= 0.267, P = .015) and the amount of viral antigen detected (ρ = 0.227, P = .039). Similar to the glomeruli, nuclear debris was often present in the interstitial capillaries. Tubular injury was characterized by tubular epithelial cell pyknosis, karyorrhexis, and karyolysis, with detachment of these cells from the basement membrane (Fig. 14).
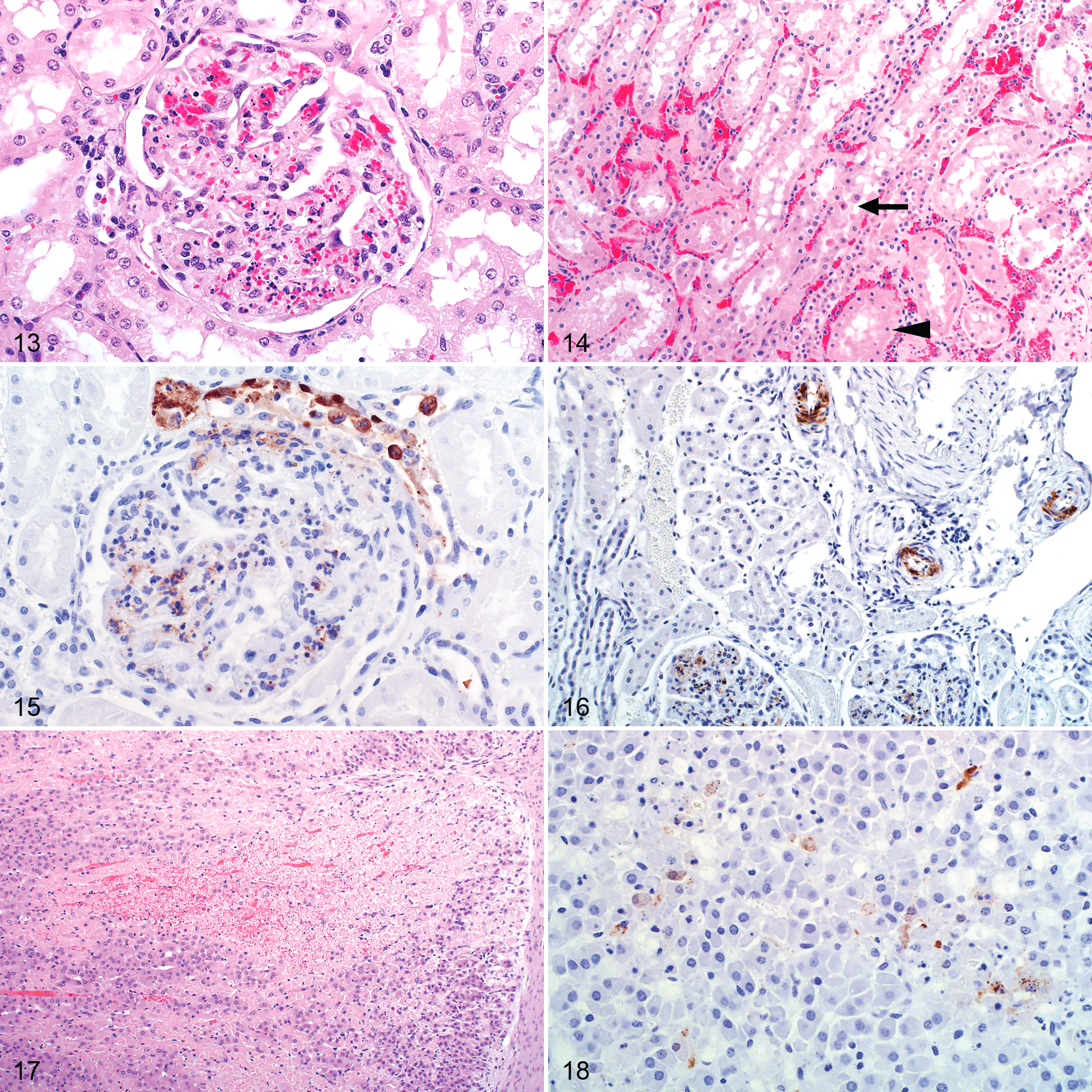
RVFV antigen was present in the kidney of 55 of 83 (66%) of the cases with available specimens. In the positive cases, viral antigen was typically in the cortex with 41 of 55 (75%) having antigen in the glomeruli, 33 of 55 (60%) in the tubules, and 22 of 55 (40%) in both (Table 4). More specifically, viral antigen was in glomerular and interstitial capillaries, tubular epithelial cells, and vascular smooth muscle cells (Figs. 15, 16). In the glomeruli, viral antigen was in mononuclear cells, neutrophils, cellular debris, or extracellularly in the capillaries. Viral antigen was present in the interstitial capillaries in 36 of 41 (88%) of the cases where glomerular labeling was also present, and labeling in the glomerular capillaries was significantly associated with labeling in the interstitial capillaries (ρ = 0.789, P < .001). In the interstitial capillaries, viral antigen was present in mononuclear cells, cellular debris, or extracellularly. Within tubules, labeling was in the cytoplasm of epithelial cells and predominately in the proximal convoluted tubules (Table 5). Labeling in the interstitial capillaries was significantly associated with labeling in tubular epithelial cells (ρ = 0.282, P = .010). There was also an association between labeling in the glomerular capillaries and tubular epithelial cell labeling (ρ = 0.281, P = .010). Necrotic cells in areas of acute tubular injury were not consistently positive for viral antigen. Labeling was never present simultaneously in both proximal tubules and collecting tubules. In 3 cases, viral antigen was not present in the glomeruli or the proximal convoluted tubules but instead occurred predominantly in collecting tubules.
Distribution of Viral Antigen in Different Areas of the Kidney for Specimens With Positive Immunolabeling for Rift Valley Fever Virus in Sheep.a
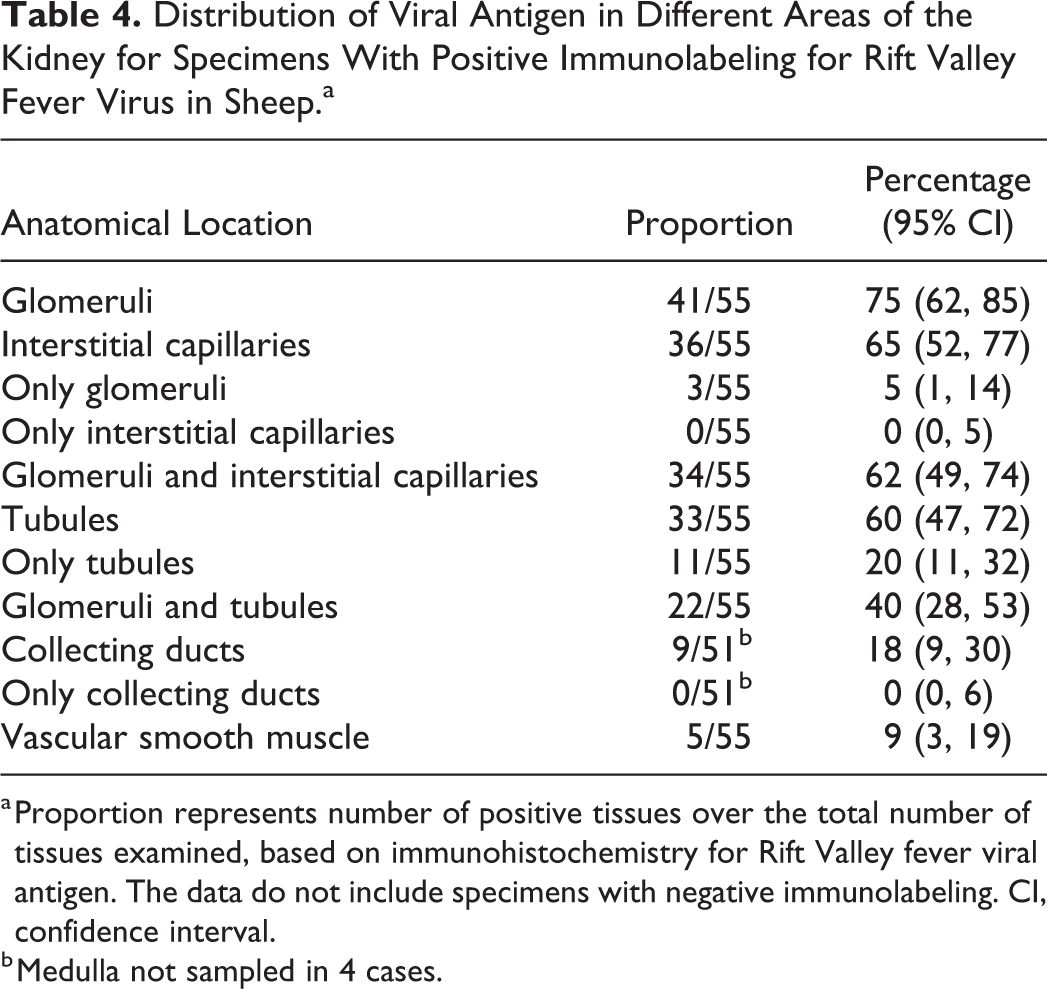
a Proportion represents number of positive tissues over the total number of tissues examined, based on immunohistochemistry for Rift Valley fever viral antigen. The data do not include specimens with negative immunolabeling. CI, confidence interval.
b Medulla not sampled in 4 cases.
Predominant Location of Viral Antigen Within Renal Tubules for Specimens With Positive Immunolabeling for Rift Valley Fever Virus in Renal Tubules.a
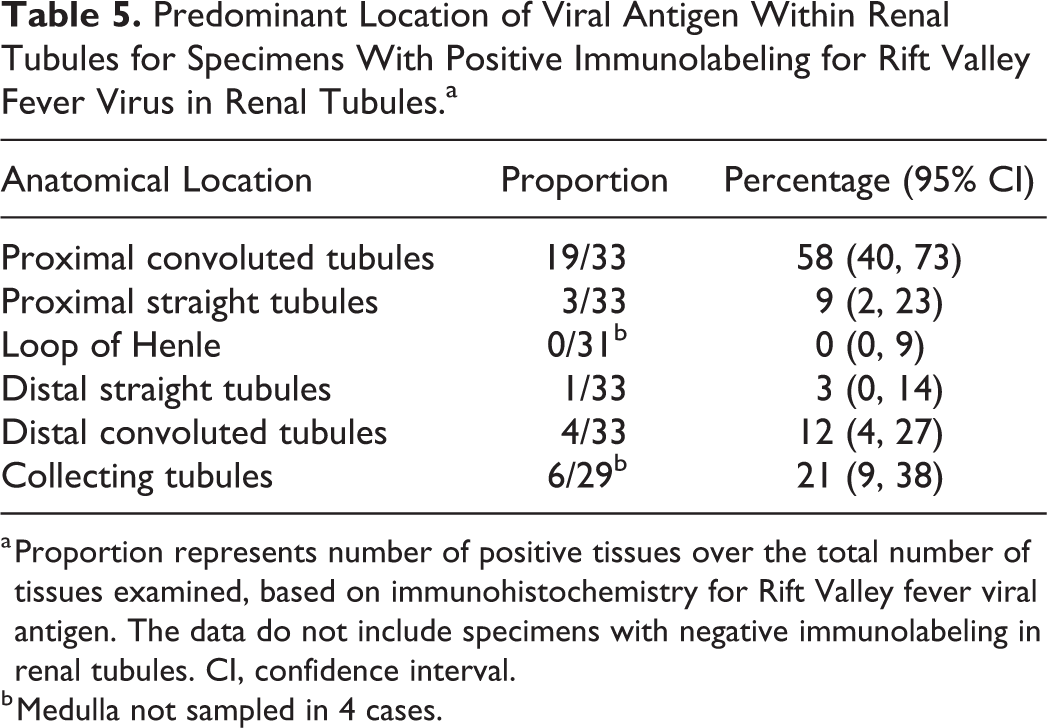
a Proportion represents number of positive tissues over the total number of tissues examined, based on immunohistochemistry for Rift Valley fever viral antigen. The data do not include specimens with negative immunolabeling in renal tubules. CI, confidence interval.
b Medulla not sampled in 4 cases.
Adrenal
Macroscopically, occasional cortical petechiae were present in the adrenal glands. Microscopically, 5 of 11 (45%) of the cases with available specimen had multifocal adrenocortical necrosis and hemorrhage. Necrosis involved individual cells to aggregates of cells predominantly in the zona fasciculata (Fig. 17). In 1 case, groups of cells in the zona glomerulosa were also involved. In 4 of the cases with necrosis, RVFV antigen was present in secretory cells of the zona fasciculata and in the cytoplasm of necrotic cells (Fig. 18). Labeling was focally disseminated in areas of necrosis or in single cells or small groups of cells that were not necessarily necrotic. One case with adrenocortical necrosis had no observable viral antigen labeling. Lesions and viral antigen were absent from the adrenal medulla.
Lung
Marked lung edema and congestion were a consistent macroscopic finding (Fig. 19). The lungs were wet and heavy with fluid oozing from the cut surfaces and copious amounts of foam filling the trachea and bronchi. This was accompanied by mild to moderate hydrothorax. Microscopic examination revealed congestion, occasional hemorrhage, emphysema, and intra-alveolar and interstitial edema in 51 of 70 (73%) of the cases (Fig. 20). Inflammation, varying from mild to severe, was present in the alveolar capillaries in all cases and consisted of predominantly mononuclear cells accompanied by fewer neutrophils. Single cell pyknosis and karyorrhexis was occasionally present in the alveolar septa and peribronchial lymphoid tissues (Fig. 20). Hemorrhage was present in 9 of 70 (13%) of the cases.
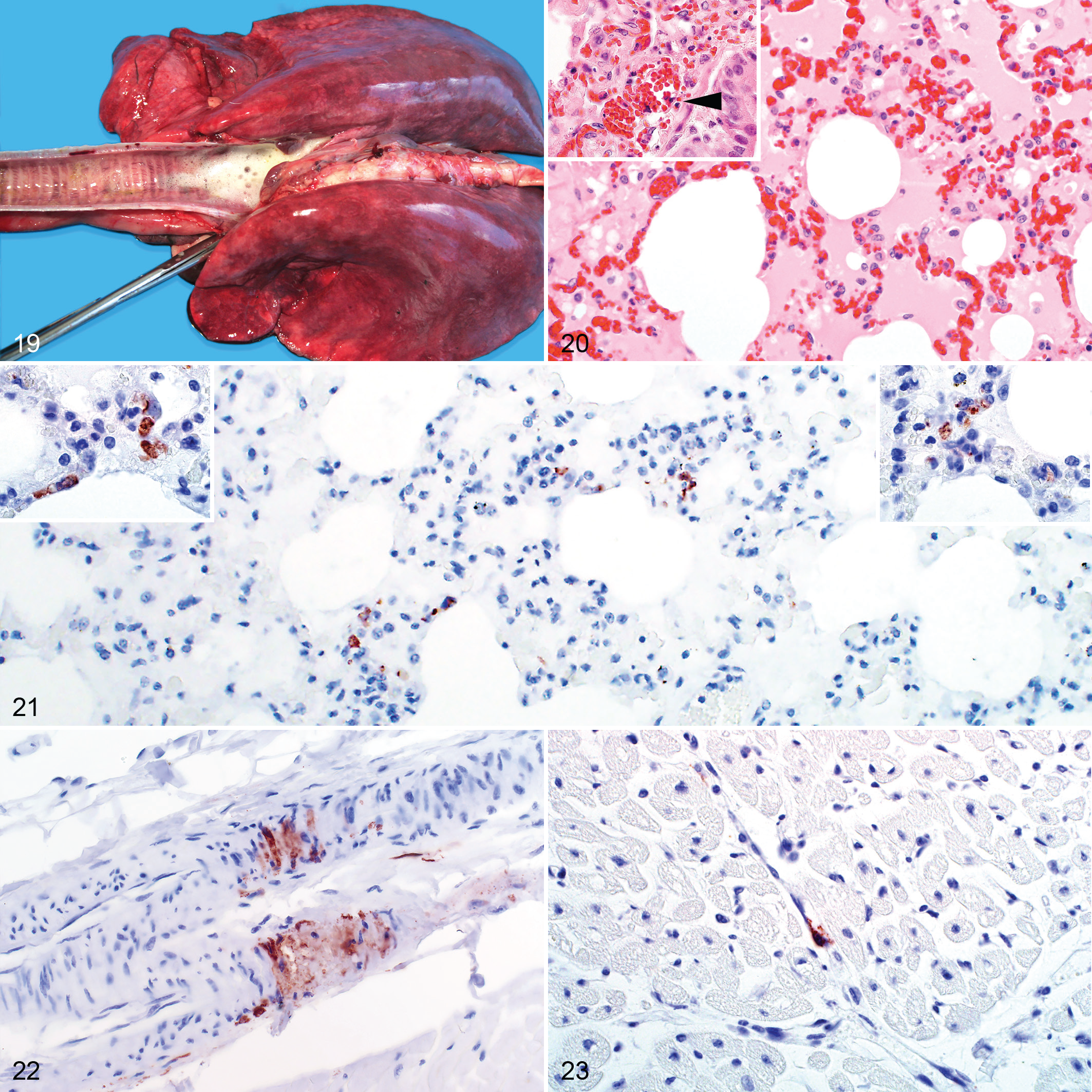
RVFV antigen was present in lung specimens of 40 of 70 (57%) cases. Labeling was generally difficult to identify and was classified as isolated in 24 of 40 (60%) of the positive cases. Scattered and widespread viral antigen was present in 10 of 40 (25%) and 6 of 40 (15%) of the positive cases, respectively. RVFV antigen was present in interstitial mononuclear cells histomorphologically consistent with pulmonary intravascular macrophages or in the capillaries associated with cellular debris (Fig. 21). Labeling was absent from alveolar macrophages and pulmonary epithelial cells.
Heart
Macroscopically, epicardial and endocardial hemorrhages and mild to moderate hydropericardium were present in most cases. Sub-epicardial ecchymoses were common in the coronary grooves and occasionally also present in paraconal and subsinuosal grooves. Sub-endocardial hemorrhages were generally in the left ventricle. Microscopically, there were focal hemorrhages in the endo- and epicardium in 13 of 57 (23%) cases. Histomorphological lesions attributable to RVFV infection were not present in the myocardium of any case.
Of the cases with available heart specimens, only 8 of 57 (14%) were positive for RVFV antigen. Immunolabeling was either isolated (n = 7) or scattered (n = 1) and in the interstitium around capillaries or small blood vessels in association with cellular debris. In 2 cases, viral antigen was present in vascular smooth muscle cells within small blood vessels (Fig. 22). Viral antigen was also present in vascular endothelial cells (Fig. 23) or in circulation in association with cellular debris. Cardiomyocytes and intravascular mononuclear cells were negative for viral antigen.
Gastrointestinal Tract
Although multifocal serosal hemorrhages could be present anywhere along the course of the gastrointestinal tract, they were often prominent on the surfaces of the rumen and abomasum. Occasionally, hemorrhages were also in the mucosa and submucosa. Peritoneal hemorrhages were sometimes present along the entire length of the major abdominal blood vessels. Mild to moderate serosanguinous ascites was also frequently present.
Necrosis was present in the lamina propria of the small intestine in 8 of 19 (42%) cases. However, autolysis was severe in 5 of 19 (26%) of these cases, and as a result, the prevalence of necrosis related to RVFV infection in the small intestine was likely underestimated. Small necrotic foci were sporadically present between the intestinal crypts and at the base of the villi. Necrotic foci were comprised of cellular debris and a mild to moderate infiltrate of degenerate neutrophils and mononuclear cells. In some of the specimens, a heavy infiltrate of eosinophils, likely unrelated to RVFV infection, was also present. Submucosal hemorrhage was present in 1 case.
Two cases had necrosis of the gut-associated lymphoid tissue, and consequently the Peyer’s patches (like other lymphoid tissues) appeared depleted of lymphocytes. In other cases, nuclear and cellular debris were present in capillaries and in the connective tissue of the lamina propria.
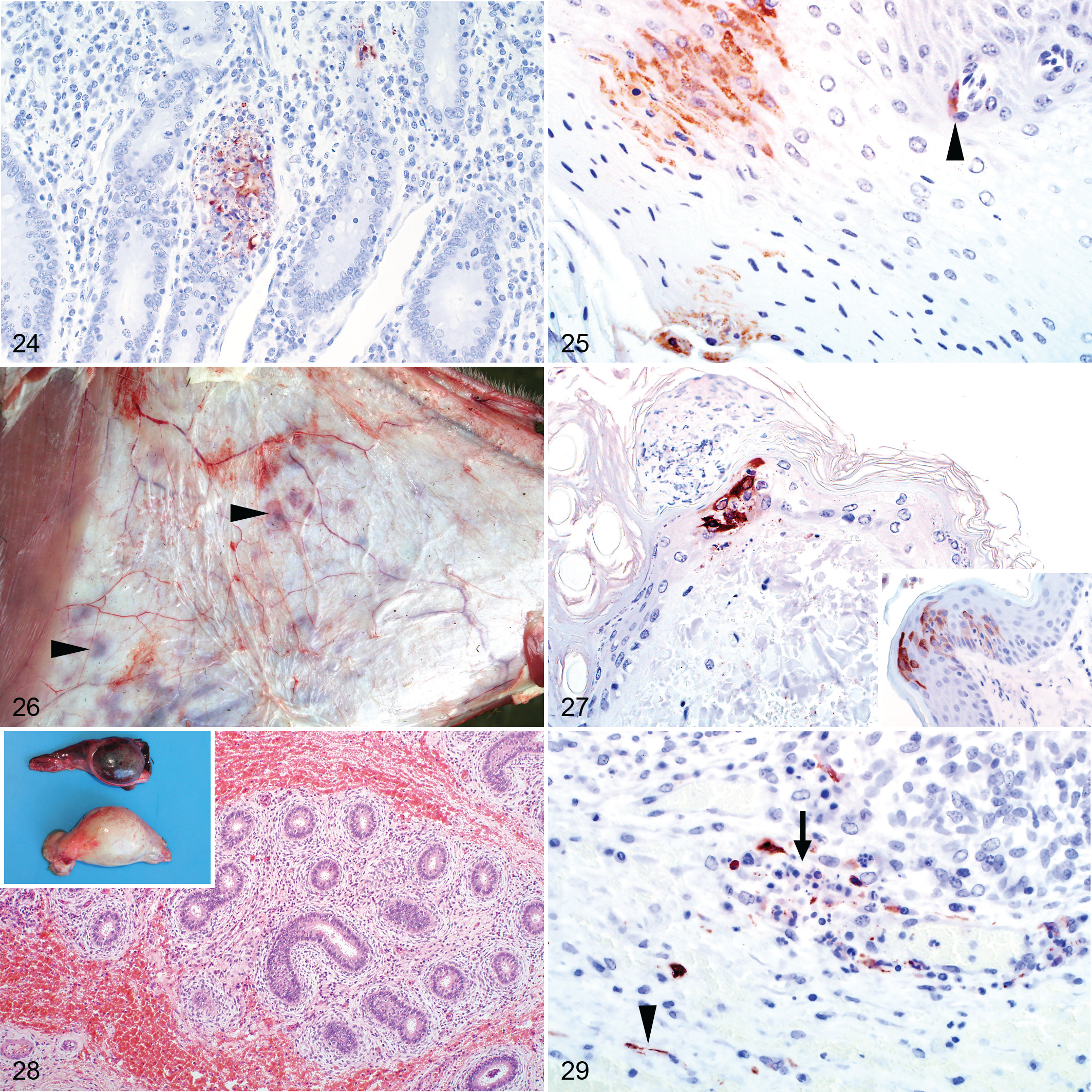
RVFV antigen was present in the small intestine of 7 of 19 (37%) cases. Most positive labeling was present in necrotic foci, either associated with cellular debris or on rare occasions in the cytoplasm of cells histomorphologically consistent with macrophages (Fig. 24). Viral antigen was not present in epithelial cells. Lesions and immunolabeling were also absent from all rumen (n = 5), abomasum (n = 24), and large intestine (n = 2) specimens. Two of 10 examined tongue specimens had scattered groups of positive epithelial cells, and 1 had positive endothelial cell labeling (Fig. 25).
Skin
Macroscopically, hemorrhages were commonly present in the subcutis and mucous membranes (Fig. 26). Crusts, presumably caused by insect bites, were often on the nose and ears. Histological lesions in the cases with available specimens (11) were minimal, with scattered foci of hemorrhage and necrosis in 2 cases. Multifocal cellular crusts overlying areas of mild eosinophilic inflammation, interpreted as insect bite hypersensitivity, were present in a further 2 cases.
RVFV antigen was present in the skin of 6 of 11 (55%) cases. Typically, it was in the epidermis in the cytoplasm of keratinocytes (Fig. 27), and in 2 cases, antigen was present below cellular crusts. Less frequently (2/11), viral antigen was present in the superficial dermis in association with cellular debris. In 1 case, viral antigen was in the epithelium of a hair bulb and in another in sebaceous gland epithelium.
Reproductive Organs
Two young rams had multifocal petechiae to suffusive hemorrhages in the testis. Microscopically, hemorrhage was in the tunica albuginea and the connective tissue surrounding the seminiferous tubules, efferent ductules, and duct of the epididymis (Fig. 28). Viral antigen was present multifocally within the connective tissue surrounding these structures in a variety of cell types, including vascular smooth muscle and endothelial cells and possibly macrophages and fibroblasts (Fig. 29). Viral antigen was also present in cell fragments and neutrophils in the blood vessels. Viral antigen was absent from the seminiferous tubules, efferent ductules, or duct of the epididymis.
Ten uterine specimens were available for evaluation, but unfortunately, most were of insufficient diagnostic quality. Hemorrhage was in the perimetrium and myometrium of 3 of 10 of the cases. Isolated viral antigen was in cellular debris or mononuclear cells in 2 of 10 cases.
Discussion
The aims of this study were to describe the tissue tropism and target cells of RVFV in a large number of naturally infected sheep and summarize gross, histopathological, and immunohistochemical findings relative to results from previous research. The 2010 RVF outbreak in South Africa provided a valuable opportunity to study the pathology and cellular tropism of RVFV in sheep and compare this with previous reports. Liver, spleen, kidney, and lungs were the specimens most often submitted. A fairly high number of gall bladder specimens (n = 23) were present, not surprisingly, since historically in South Africa much emphasis has been placed on edema and hemorrhage of the gall bladder as a typical RVF lesion. 15,28 Similarly, many abomasal specimens were included (n = 24) due to the perceived importance of edema and hemorrhages in the wall of this organ. 15,41 It should be noted that since organs were not consistently sampled in these field cases, the frequencies of lesions and immunolabeling could be an overestimate of the general population of RVF cases if there was a tendency to sample grossly abnormal organs.
Macroscopically, necrosis and hemorrhage of the liver was accompanied by lesions suggestive of vascular endothelial injury, including mild hydropericardium, hydrothorax and ascites, marked and diffuse pulmonary congestion and edema, congestion and edema of lymph nodes, and hemorrhages in many organs. The exact mechanism for hemorrhage and plasma leakage in RVFV infection in sheep has not been elucidated. However, the mechanisms of increased vascular permeability in viral hemorrhagic fever have often been attributed to direct viral infection or damage of vascular endothelial cells, endothelial activation and dysfunction, infection of macrophages causing the production of pathologic concentrations of cytokines and other mediators, and thrombocytopenia. 32 Hemorrhage and necrosis in organs other than the liver have also been ascribed to activation of the coagulation cascade accompanied by depressed production of coagulation factors due to severe hepatic necrosis, which may cause disseminated intravascular coagulation. 32 Previously, thrombocytopenia and prolonged prothrombin and clotting times have been demonstrated in sheep. 31 In the present study, RVFV antigen was present in endothelial cells, and microscopic foci of necrosis were sporadically identified in various organs like the gall bladder, small intestines, skin, heart, and testis. However, there was no convincing evidence of vascular fibrinoid necrosis or microvascular fibrin thrombi in any organ. Their absence may be attributable to hyperfibrinolysis or enhanced-fibrinolytic-type disseminated intravascular coagulation, which was described in dengue hemorrhagic fever. 3,27 Therefore, direct infection and injury of endothelial cells accompanied by enhanced activation of coagulation with consumption of platelets and coagulation factors could play a role in the pathogenesis of RVFV in sheep. However, the formation of fibrin thrombi might be inhibited by increased clot lysis causing widespread bleeding.
Microscopically, multifocal-random, necrotizing hepatitis was confirmed as the most distinctive histopathological feature of RVF cases in adult sheep. Experimental studies in adult sheep and lambs wherein animals were examined at known days post-infection have reported a sequence of events for the liver. 10,11,13,18 The earliest change affected either a single hepatocyte or groups of 2 to 5 hepatocytes randomly distributed in the lobule. Daubney et al 11 observed that hepatocytes in the central zone were more frequently infected first, whereas Findlay 18 and Easterday et al 13 reported that initial lesions were more frequently midzonal. Focal degeneration of hepatocytes was closely followed by infiltration of the lesion with chiefly neutrophils and macrophages. These cells became degenerate, and lesions evolved into foci consisting of karyorrhectic debris derived from the nuclei of both hepatocytes and infiltrating cells. 11 More advanced lesions might involve up to two-thirds or more of the entire lobule. In the present study, a single pattern of necrosis could not be discerned. Instead, necrosis was distributed irregularly throughout the lobule. The severity and pattern of necrosis could also vary considerably between different liver specimens from the same animal.
Concerning the diagnostic specificity of lesions in the liver, early apoptotic hepatocytes (also referred to as Councilman bodies or acidophilic bodies) and late apoptotic bodies with nuclear fragments and condensed cytosol in areas of hepatocellular damage are diagnostically useful. In contrast, characteristic eosinophilic intranuclear inclusions are of limited diagnostic value since they are rare in older animals and often not clearly identifiable or confused with pseudo-inclusions. They are, however, very useful in neonates and especially in fetuses where liver necrosis is often very subtle. Hepatocyte injury due to other concurrent causes such as hepatotoxic plants or hypoxia due to severe anemia might also complicate hepatic histopathological interpretation. However, one consistent finding in this study was that unlike the necrosis caused by hepatotoxic plants or hypoxia, RVFV invariably caused foci of necrosis in 1 or more of the periportal zones, specifically affecting hepatocytes of the limiting plate.
Sheep breeds might differ in their response to RVFV infection with an associated variation in lesions that could explain why previous reports concerning splenic, lung, and skin lesions are inconsistent. Daubney et al 11 reported that the spleen was not enlarged, while Schultz 38 reported splenomegaly. Daubney et al 11 reported that the lungs were normal in size and color and did not exhibit hemorrhages. Subsequent researchers described pulmonary congestion, edema, and emphysema with occasional hemorrhagic foci that are consistent with the findings of the present study. 9,16,17,38,43 Another study comparing the susceptibility of 3 breeds of sheep to experimental infection reported that the spleen of the Yankasa breed of sheep was enlarged while that of West African Dwarf and Ouda sheep were shrunken and firm. 31 All 3 breeds exhibited lung edema with hemorrhages and edema in the bronchial lymph nodes, but pulmonary hemorrhage was more severe in the Yankasa breed. Petechiae and ecchymoses were reported in the visible mucous membranes and hairless areas of the skin of the Yankasa sheep but not in the West African Dwarf and Ouda sheep. 31 The results of the present study confirmed the findings of previous reports concerning RVF in South Africa whereby subcutaneous hemorrhages were observed in the axillary region, medial aspect of the hind limbs, and lower portions of the extremities. 15,38 Although Merino and Dorper are the most common breeds of sheep in South Africa, ascribing the lesions reported here to these breeds is not possible since breed was rarely recorded on laboratory submission forms.
Differences in lesions among published studies could also be ascribed to differences in viral strains, or inoculum route. 4,34 The physiological state of the animal, diet, rearing practices, and concurrent infection with other pathogens might also account for differing responses to viral infections in general. 4,34 However, studies in rats suggest that host genetic background and host immunological response might play a more important role than viral virulence and inoculum size on the clinical outcome of RVFV infection. 5,22,34 Genetically resistant rats treated with an immunosuppressive drug and challenged with up to 5 × 106 pfu were more resistant to infection than healthy rats bearing a susceptibility gene and challenged with 5 to 50 pfu. 34 Genetic resistance might also explain why naturally occurring encephalitis, a complication of RVF in humans that has been experimentally reproduced in mice and rats, has not been described in sheep. 2,5,12 Whether genetic resistance is more important than viral virulence for the outcome of RVFV infection in sheep should be further explored.
Microscopically, lymphoid necrosis in the spleen and other lymphoid tissues was mentioned in previous studies but not described in detail. 9,16,17,31,39,43 The results of the present study confirmed that necrosis occurs in the spleen and lymph nodes of adult sheep and that splenic necrosis was significantly associated with necrosis in the lymph nodes. Prior, Coetzer 9 reported that lymphocytes in the Peyer’s patches had varying degrees of pyknosis and karyorrhexis, corresponding in degree of intensity to similar lesions in other lymphoid tissues, and that pyknosis and karyorrhexis also occasionally occurred in the bronchus-associated lymphoid tissues. Additionally, in the intestines karyorrhexis of cells in the interglandular tissues and particularly around the lymphoid follicles, has been reported. 11 In the present study, severe changes were observed in the gut-associated lymphoid tissues in the small intestine that mirrored changes in the lymph nodes. Scattered lymphocytolysis was also noted in the lamina propria of the small intestine, the periportal areas of the liver, and occasionally in the bronchus-associated lymphoid tissues. Previous reports described marked pyknosis and karyorrhexis of mononuclear cells in the distal jejunum and ileum in lambs. 9 Unfortunately, this finding was not corroborated in the current study since the different anatomical regions of the small intestine could not be reliably identified. Therefore, all lymphoid tissues and anatomical locations where lymphocytes often occur should be meticulously examined since pyknosis and karyorrhexis of lymphocytes seem to be typical for RVFV infection.
The present study also identified lymphocytolysis as being prominent in the B-cell rich areas of lymphoid organs, suggesting that B lymphocytes were preferentially targeted. The pathogenic mechanism of lymphocytic depletion in sheep is likely apoptosis, as demonstrated in mice using terminal deoxynucleotidyl transferase dUTP nick-end labeling. 40 Also, tingible-body macrophages, described in this study, are by definition cells that phagocytose fragments of apoptotic bodies. 1 However, further examination of lymphocyte subsets with cell markers would clarify which cells undergo lymphocytolysis and elucidate pathogenic mechanisms.
In the present study, multifocal acute renal tubular injury was present in most of the cases (68/83). Daubney et al 11 made the astute inference that renal tubular degeneration (nephrosis) was absent in lambs due to the peracute course of the disease in that age group. The same authors surmised that infected adult sheep might live long enough for kidney damage to develop and whether lesions are observed or not depended on the length of time that sheep survived, hence the variable lesion severity observed in the present study. 11 Daubney et al 11 also observed that renal changes in adult sheep that died acutely occured in the convoluted tubules while other tubules appeared normal. In less acute cases, the tubular injury was less advanced and affected all the tubular epithelial cells in the cortex. 11 In the present study, changes were in the convoluted, straight, and collecting tubules in the cortex but not in the loops of Henle or the medulla.
Furthermore, scattered pyknotic and karyorrhectic nuclear and cellular debris was present in the glomeruli. The origin of this debris could not be ascertained with certainty, but it often appeared to be present within capillary lumens and hence could originate from leucocytes or endothelial cells. The fragments could also be remnants of necrotic leucocytes or vascular endothelial cells from distant tissues, trapped within the capillary beds of an end-artery organ. Similar to the renal glomerular findings, cellular debris was also observed in the renal interstitial capillaries and pulmonary alveolar septa. The latter was also reported by Coetzer. 9 Comparable findings have been made in dengue hemorrhagic fever where apoptosis of leucocytes was demonstrated in the pulmonary microvasculature and apoptosis of vascular endothelial cells in pulmonary and intestinal tissues. 24 High levels of circulating endothelial cells were demonstrated in fatal human dengue hemorrhagic fever cases. 6 Therefore, in RVF cases in sheep, it seems likely that nuclear and cellular debris in the renal glomerular capillaries, renal interstitial capillaries, and pulmonary alveolar septa all have the same origin, namely, necrotic vascular endothelial cells or leucocytes. This futher supports the proposed mechanism of observed hemorrhage and plasma leakage in RVFV infection.
Immunolabeling for RVFV was most unequivocally positive in the liver, with labeling in the cytoplasm of injured hepatocytes and cytoplasmic fragments. Labeling was also readily identified in spleen, although positive labeling was observed more readily in the white pulp, particularly in the marginal zone. In the kidney, viral antigen was more often in the cortex within glomerular and interstitial capillaries, tubular epithelial cells, and vascular smooth muscle cells. Additionally, viral antigen might first appear in the glomerular and peritubular capillaries, second in proximal convoluted tubular epithelial cells, and then gradually move through the more distal tubules and the collecting ducts of individual nephrons, progressively clearing from the glomeruli and more proximal aspects until the virus completely disappears from the kidney or the animal dies. This interpretation was supported by our observation of a stronger correlation between glomerular and interstitial labeling than between glomerular and tubular labeling.
Lung specimens were often positive for RVFV antigen, but labeling was generally sparse and difficult to identify. Viral antigen in the adrenal glands was in adrenocortical cells in the zona fasciculata. RVFV antigen was also present in macrophages in lymph nodes, epithelial cells, and a vascular endothelial cell in the tongue; in foci of necrosis in the gall bladder and small intestine; and within vascular smooth muscle cells and vascular endothelial cells in heart specimens. Viral antigen has been previously detected in lymph node but not in gall bladder, small intestine, tongue, or heart. 17,43,45
Skin specimens might be useful diagnostically since a surprisingly high percentage (55%) were IHC positive. However, viral antigen was rarely present in skin specimens taken from areas with subcutaneous hemorrhages. We suspect that specimens from the nose and ears or other areas with crusting due to insect bites might be more useful diagnostically. A study to determine the competance of Culex pipiens as a vector of RVFV supports this conclusion. 44 In this study, RVFV was absent in skin specimens not exposed to mosquitoes. Instead, antigen was detected in mosquito-exposed skin in macrophages, vascular endothelial cells, smooth muscle cells, lipocytes, keratinocytes, and fibroblasts.
To our knowledge, this is the first report of lesions and RVFV antigen detection in the testis of sheep. However, the successful passaging of RVFV in lamb testis cell cultures was previously reported. 8 In the present study, RVFV antigen was not within the seminiferous tubules, efferent ductules, or duct of the epididymis and was instead only in the connective tissue surrounding these structures. However, there is still a risk that RVFV might be transmitted through semen. RVFV genomic RNA was detected by RT-PCR in the semen of a human kidney transplant recipient for up to 4 months after onset of symptoms. 21 RVFV antigen and antibodies were also detected using ELISA and AGID in the semen of bulls 1 to 3 weeks after vaccination with a live attenuated RVFV vaccine. 36 Currently, international trade rules do not require the screening of ruminant semen for RVFV. 30 Therefore, a study of RVFV-infected ruminants should be performed that attempts to detect viable virus from semen to determine concentrations and duration of shedding.
Macrophages were positive for RVFV antigen in this study, and based on morphology, microanatomical location, and comparisons with descriptions of these cells in mice and humans, we consider these to include Kupffer cells in the liver; marginal zone, metallophilic, tingible-body, and red pulp macrophages in the spleen; sinus macrophages in the lymph nodes; pulmonary intravascular macrophages; lamina proprial macrophages in the intestinal tract; intravascular macrophages in the renal interstitium and glomerular tuft; and testicular interstitial macrophages. RVFV antigen was also reported to occur in macrophages in the skin of experimentally infected sheep. 44
Vascular smooth muscle cells and endothelial cells were also positive for RVFV antigen in this study. One common feature of tissue macrophages, vascular smooth muscle cells, and endothelial cells is that they all express class AI scavenger receptors (SCARA1). 48 In mice, tissue macrophages expressing SCARA1 were detected in the red pulp and marginal zone of the spleen, subcapsular region of lymph nodes, the lamina propria of the intestines, Kupffer cells in the liver, and macrophages of the lung. 23 In human tissues, anti-scavenger receptor class A antibodies have been shown to recognize macrophages such as Kupffer cells in the liver, pulmonary macrophages, macrophages in lymphoreticular organs, macrophages in the lamina propria of the intestines, and interstitial macrophages in kidney and testis. 42 This corresponds with the distribution of RVFV immunolabeled macrophages detected in sheep in the present study. Scavenger receptors are membrane-bound pattern recognition receptors that bind to a variety of ligands, including endogenous proteins and pathogens, and have been classified into 10 eukaryote families, defined as Classes A through J. 48 It has been shown that the uptake of Adenovirus 5, Herpes simplex virus type 1, and Vaccinia virus is mediated via class A scavenger receptors. 20,25,26 Although class B scavenger receptors differ from the class A scavenger receptors in terms of structure and function, a number of them have been identified as receptors for viruses such as enterovirus 71, Coxsackie virus, and hepatitis C virus. 37,46,47 The finding that diverse viruses exploit scavenger receptors and that the cellular tropism demonstrated for RVFV in sheep might coincide with the distribution of SCARA1 in tissues should be explored in future studies.
The minimum set of specimens to be submitted for histopathology and IHC to confirm or exclude a diagnosis of RVFV should include liver, spleen, and kidney with lung and skin useful additional samples (Table 6). Characteristic microscopic lesions occur in the liver, spleen, kidney, and lungs. These lesions include necrosis of limiting plate hepatocytes with apoptosis, marked lymphocytolysis in B-lymphocyte dependent areas of the spleen, acute renal tubular epithelial injury, and nuclear pyknosis and karyorrhexis in the capillaries of the glomeruli and pulmonary interstitium. A definitive diagnosis of RVFV can be obtained by performing IHC on FFPE liver, spleen, and kidney.
Preferred Diagnostic Specimens for Investigation of Rift Valley Fever Using Histopathology and Immunohistochemistry.

However, when typical liver and spleen lesions are absent but renal tubular epithelial injury is apparent, IHC should be performed to exclude a possible renal form of RVFV. In the present study, 3 cases had acute renal tubular injury with labeling for RVFV in the kidney but no histological lesions or immunohistochemical labeling in the liver or spleen of 2 cases with available specimens. This result could simply be a reflection of inadequate sampling since often only a single specimen from the liver and/or the spleen and/or the kidney was submitted to diagnostic laboratories. Alternatively, these cases could represent an atypical form of RVF in sheep. Acute renal failure associated with RVF was described in human patients in Saudi Arabia and Sudan, where 13 and 85 patients, respectively, had signs and symptoms of renal failure without hepatic involvement. 2,14 Therefore, in RVF-endemic areas, routine screening of acute renal tubular epithelial injury for RVFV antigen presence should be considered, especially when other common causes (e.g., pulpy kidney disease and nephrotoxic plants and antimicrobials) have already been excluded.
Conclusion
This largest investigation of RVFV pathology and tropism in sheep provides a comprehensive description of lesion distribution at the tissue and cellular levels. Liver necrosis was confirmed as the most distinctive feature of RVF cases in adult sheep. Furthermore, 45 of 70 (64%) of the cases where liver, spleen, and kidney tissues were available also had foci of acute renal tubular epithelial injury in addition to necrosis in both the liver and spleen. Acute renal tubular and glomerular injury was the most frequent RVF lesion in some cases, suggesting that an atypical renal form of RVF might occur in sheep. The liver was most consistently positive for RVFV antigen followed by the spleen, kidneys, lung, and skin. Antigen-positive cells included hepatocytes, renal tubular epithelial cells, adrenocortical cells, macrophages, neutrophils, vascular smooth muscle cells, and vascular endothelial cells.
In most cases, a diagnosis of RVF can be confirmed using histopathology and IHC of the liver, kidney, and spleen. Skin with crusting due to insect bites and lung could be useful additional samples. Also, we recommend that in endemic areas, cases of acute renal tubular injury be investigated further using IHC if other more common causes have already been excluded. The finding that RVFV can cause an acute infection in the testis requires further investigation with possible revisions to international trade rules to include testing of semen for RVFV using PCR and virus isolation.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818806049 - Lesions and Cellular Tropism of Natural Rift Valley Fever Virus Infection in Adult Sheep
Supplemental Material, DS1_VET_10.1177_0300985818806049 for Lesions and Cellular Tropism of Natural Rift Valley Fever Virus Infection in Adult Sheep by Lieza Odendaal, Sarah J. Clift, Geoffrey T. Fosgate, and A. Sally Davis in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the staff of the histopathology laboratory of the Department of Paraclinical Sciences, Section of Pathology, especially Rephima Phaswane, Naomi Timmerman, and Marie Smit for technical assistance. We thank colleagues at the National Department of Agriculture, Fisheries and Forestry, Directorate of Veterinary Services for providing access to original diagnostic data. We also thank Antoinette Odendaal for preparing the panels for this manuscript with assistance from Mal Rook Hoover, CMI, KSU.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Institutional Research Theme, Biotechnology and the Management of Animal and Zoonotic Diseases, of the University of Pretoria provided funding for the research project. Publication of this study was funded by the Department of Paraclinical Sciences of the Faculty of Veterinary Science, University of Pretoria, and the Department of Diagnostic Medicine/Pathobiology, College of Veterinary Medicine, of Kansas State University.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
