Abstract
Mammary cancer is a common neoplasm in women, dogs, and cats that still represents a therapeutic challenge. Wnt/β-catenin and Hippo pathways are involved in tumor progression, cell differentiation, and metastasis. The aim of this study was to evaluate mRNA and protein expression of molecules involved in these pathways in human (HBC), canine (CMT), and feline mammary tumors (FMT). Real-time quantitative polymerase chain reaction (qPCR) for β-catenin, CCND1, YAP, TAZ, CTGF, and ANKRD1, western blotting for YAP, TAZ, and β-catenin, and immunohistochemistry for estrogen receptor (ER), progesterone receptor (PR), ERBB2, β-catenin, and YAP/TAZ were performed on mammary tumor tissues. The protein expression of active β-catenin was higher in tumors than in healthy tissues in all 3 species. The mRNA expression of the downstream gene CCND1 was increased in HBC ER+ and CMTs compared to healthy tissues. Membranous and cytoplasmic protein expression of β-catenin were strongly negatively correlated in all 3 species. Tumors showed an increased protein expression of YAP/TAZ when compared to healthy tissues. Notably, YAP/TAZ expression was higher in triple negative breast cancers when compared to HBC ER+ and in FMTs when compared to CMTs. The mRNA expression of β-catenin, YAP, TAZ, CTGF, and ANKRD1 was not different between tumors and healthy mammary gland in the 3 species. This study demonstrates deregulation of Wnt/β-catenin and Hippo pathways in mammary tumors, which was more evident at the protein rather than the mRNA level. Wnt/β-catenin and Hippo pathways seem to be involved in mammary carcinogenesis and therefore represent interesting therapeutic targets that should be further investigated.
Mammary gland tumors are the most common neoplasm in women 17 and in bitches 66 and are the third most common tumor in queens. 80 The need for appropriate animal models in cancer research has led, over the past 20 years, to the use of dogs and cats with spontaneous tumors as valuable, but underutilized, resources. 1,8,74 Human breast cancer (HBC) is usually classified into 3 main subtypes according to the expression of estrogen receptor (ER), progesterone receptor (PR), and epidermal growth factor receptor ERBB2: (1) ER-positive (ER+) tumors, (2) ERBB2-overexpressing tumors, and (3) triple-negative (ER-, PR-, ERBB2-negative) breast cancer (TNBC). 56 TNBC is the most aggressive subtype and typically has the poorest prognosis with high risk of distant metastasis and death. 65 The majority (80% to 90%) of feline mammary tumors (FMT) are characterized by an aggressive biological behavior with rapid progression and metastasis. 46,81 FMT are often ER-/PR-/ERBB2-negative, and have been proposed as a potential spontaneous model for the TNBC subtype. 8,59,74
Several pathways have been implicated in regulating tumor growth, invasion, and metastasis, such as Wnt, Notch, Hedgehog, and Hippo. 10,37,53 Wnt/β-catenin pathway activation is triggered by the binding of Wnt ligand to the LRP-5/6 and Frizzled receptors, with the subsequent recruitment of the Axin, GSK-3β, CK1, and APC complex to the receptor. 7,76 The Wnt-Frizzled-Axin-LRP5/6 complex sequesters GSK-3β, inhibiting its capacity to phosphorylate β-catenin, which allows β-catenin to accumulate in the cytoplasm and eventually translocate into the nucleus. In the nucleus, β-catenin enhances the expression of several downstream genes such as c-MYC, cyclin D1, and CDKN1A. 23 Wnt/β-catenin pathway deregulation has been shown to be a key feature in many human cancers, such as breast, colorectal, melanoma, prostate, lung, and others. 38,70,73
The Hippo pathway is a tumor-suppressor signaling pathway that is involved in the regulation of organ size 47 as well as self-renewal and differentiation of embryonic stem cells, 69 mesenchymal stem cells, 30 induced pluripotent stem cells, 58 and cancer stem cells. 14 When the Hippo pathway is activated, phosphorylated MST1/2 and WW45 form a complex that activates LATS1/2 and MOBKL1A/B, which form a kinase complex. This cascade of events leads to the phosphorylation of transcription coactivators YAP and TAZ with their subsequent degradation. 40 Unphosphorylated YAP and TAZ accumulate within the cell and translocate into the nucleus, where they interact with transcription factors regulating the expression of downstream genes, such as CTGF, CYR61, and ANKRD1. 53,64 Deregulation of this pathway is thought to be one of the key events in cancer development. 9 YAP and TAZ have been found to be overexpressed in many human cancers, promoting cell proliferation and epithelial-to-mesenchymal transition (EMT). 14,28,53 The aim of this study was to investigate the mRNA as well as the protein expression of some molecules directly involved in the Wnt/β-catenin and Hippo pathways in human, canine, and feline mammary gland tumors.
Materials and Methods
Tissue Collection
Human samples were collected from Istituto Oncologico Veneto (IOV; Padua, Italy), whereas canine and feline samples were collected from local veterinary clinics and preserved in RNALater (Ambion) within 2 hours after surgery, according to the manufacturer’s instructions. Human sample collection was approved by the ethical committee of the IOV. All patients or patients’ owners provided informed, written consent to use their samples for this study.
Specifically, 5 healthy human mammary gland tissues, 5 ER-positive (ER+) HBC tissues, 5 triple-negative HBC (TNBC) tissues, 4 healthy canine mammary gland tissues, 10 canine mammary tumors (CMTs; 5 grade I and 5 grade II), 6 healthy feline mammary gland tissues, and 6 FMTs grade III were collected. To ensure uniformity of cell type, only simple carcinomas, composed of only one tumor cell type (luminal epithelial cells), 22 were included. Healthy mammary tissues were collected from tumor-bearing patients.
Before RNA extraction, a small portion of each sample preserved in RNALater was fixed in 4% formaldehyde and paraffin embedded. Four-micrometer-thick tissue sections were stained with hematoxylin and eosin, and slides were visualized under the microscope to further confirm the presence of healthy mammary tissue in the samples labelled as “healthy” and of tumor tissue in the samples labelled as “tumor.”
RNA Extraction and Real-Time Polymerase Chain Reaction (PCR)
For gene expression analyses, 50 mg of each tissue sample preserved in RNALater was used for RNA extraction using Trizol Reagent (Invitrogen), following the manufacturer’s protocol. The extracted RNA was treated with RNAse-free DNAse I (New England Biolabs). Five-hundred nanograms of total RNA from each sample was reverse transcribed using RevertAid First Strand cDNA Synthesis Kit (Thermo Scientific), according to manufacturer’s protocol. The cDNA was then used as a template for real-time PCR using the ABI 7500 Real-Time PCR system (Applied Biosystems) to evaluate expression of β-catenin (CTNNB1), cyclin D1 (CCND1), YAP, TAZ, CTGF, and ANKRD1. All samples were tested in triplicate. ACTB was used as house-keeping gene. The relative gene expression was calculated applying the 2−▵▵Ct formula, according to Schmittgen and Livak. 63 qPCR efficiency was calculated performing a dilution series experiment and applying the following formula to the standard curve: efficiency = 10(−1/slope) − 1. 21 qPCR efficiency was between 90% and 100% for all the samples. Primer sequences are reported in Table 1. The primers used in this study were designed de novo using NCBI Primer-BLAST. To assess primer specificity, the dissociation curves of all real-time qPCR products were evaluated to confirm a single amplification peak. The qPCR reactions were then purified using the ExoSAP-IT PCR Product Cleanup (Applied Biosystems) and sequenced at the BMR Genomics (Padua, Italy). Sequences were verified using NCBI BLAST database.
Real-Time Qualitative Polymerase Chain Reaction Primer Sequences Used in This Study.

Protein Extraction and Western Blotting
A subset of the above-mentioned tissues was used for protein extraction and western blotting. Specifically, proteins were extracted from 2 healthy human mammary tissues, 2 ER+ HBCs, 2 TNBCs, 3 healthy canine mammary tissues, 2 CMTs grade I, 2 CMTs grade II, 2 healthy feline mammary tissues, and 3 FMTs grade III. The isolation of soluble and nuclear fractions was carried out as described with slight modifications. 45 The protocol was standardized in our laboratory confirming the purity of soluble/cytosolic and nuclear fractions using alpha-tubulin and histone H3 markers, respectively (data not shown). Briefly, tissues (20 μg) were minced with scissor, suspended in a hypotonic solution (10 mM Tris–HCl, pH 7.4, 0.5 mM MgCl2, containing protease inhibitors 100 μM phenylmethylsulfonyl fluoride [PMSF], 1 μg/ml leupeptin) and homogenized on ice with 30 strokes in a precooled glass homogenizer. The homogenates were diluted with an equal volume of a solution of 10 mM Tris–HCl, pH 7.5, 0.5 M sucrose, 300 mM KCl. The suspensions were centrifuged for 10 minutes at 10 000g to pellet nuclei and the supernatant containing soluble fractions were collected in a fresh tube. The pellet containing nuclear fractions were resuspended in a lysis buffer containing 20 mM Hepes pH 7.5, 1.5 mM MgCl2, 0.4 M NaCl, 0.2 mM EDTA, 1% Triton and protease inhibitors 100 μM PMSF, 1 μg/mL leupeptin. Protein concentration was determined using the BCA assay (Thermo Scientific), according to manufacturer’s instructions. Proteins were stored at −80 °C, until used.
Proteins were resolved by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, using 7.5% or 10% polyacrylamide gels. After electrophoresis, proteins were transferred onto nitrocellulose membrane, which was incubated with Ponceau S (Sigma Aldrich) for 5 minutes to confirm successful transfer, and then blocked with 5% milk. Afterwards, the membrane was probed with the following antibodies: anti-WWTR1 (Sigma Aldrich, 1:1000) recognizing both the YAP and TAZ human proteins; anti-β-catenin (BD Transduction Laboratories, 1:1000); anti-non-phospho (active) β-catenin (Cell Signaling, 1:1000). Anti-β-actin (Sigma Aldrich, 1:4000) was used as a loading control for the soluble fractions in the 3 species, whereas Ponceau S was used as a loading control for the nuclear fractions in the 3 species.
Membranes were blocked 1 hour at room temperature in 10 mM Tris/HCl, pH 7.5, 150 mM NaCl, 0.05% Tween20 containing 1% bovine serum albumin (BSA) for anti-WWTR1 and anti-β-catenin antibodies and in 20 mM Tris/HCl, pH 7.6, 150 mM NaCl, 0.1% Tween20 containing 5% BSA for anti-non-phosphorylated (active) β-catenin. Incubation with primary antibodies was carried out at 4 °C overnight in the buffers used as blocking solution, without BSA. After 1-hour incubation at room temperature with the appropriate secondary antibody, proteins were detected with the staining solution (5 mg BCIP [5-bromo-4-chloro-3-indolyl phosphate] and 1 mg NBT [nitro blue tetrazolium] in 30 ml of 0.1 M TRIS, 0.1 M NaCl, 5 mM MgCl2, pH 9.5 buffer).
Immunohistochemistry
The above-mentioned samples as well as additional HB tissues selected from the Division of Anatomic Pathology archive of the Padua University Hospital and CMTs and FMTs selected from our archive (Anatomic Pathology, Dept. BCA, University of Padua) were used to perform immunohistochemistry (IHC). Specifically, IHC was performed on the following tissue samples: 10 ER+ HBC, 11 TNBCs, 11 CMTs grade I, 11 CMTs grade II, and 12 FMTs grade III.
Sections (4 μm) were processed with an automatic immunostainer (BenchMark XT, Ventana Medical Systems), as previously described. 4,8 Briefly, the automated protocol included the following steps: a high temperature antigen unmasking (CC1 reagent, 60 minutes), primary antibody incubation (see details below), an ultrablock (antibody diluent, 4 minutes), and hematoxylin counterstaining (8 minutes). Negative controls omitted the primary antibody, whereas adnexa, epidermis, and non-tumor breast/mammary gland, when present, were used as positive controls for β-catenin and YAP/TAZ. For ERBB2 an additional technical external positive control was adopted (ERBB2 3+ HBC), whereas the species-specific cross-reactivity was previously described both in the cat and in the dog. 59,72
For ER and PR, feline and canine uterus and ovary sections were also stained. The following antibodies were tested: anti-estrogen receptor alpha (ERα; NCL-ER-6F11 1:40, Novocastra, in human and feline species—NCL-ER-LH2 1:25, Novocastra, in canine species), anti-progesterone receptor (PR; 1:80; NCL-PGR-312, Novocastra in human and feline species), anti-ERBB2 (1:250; A0485, Dako in canine and feline species), anti-β-catenin (1:100; 610154, BD Biosciences), and anti-WWTR1 (YAP/TAZ; 1:100; HPA007415, Sigma-Aldrich). In the human samples, ERBB2 immunolabeling was performed with Bond Oracle HER2 IHC System for BOND-MAX (Leica Biosystems), containing the anti-ERBB2 antibody (clone CB11, ready-to-use). A semiquantitative evaluation of IHC positivity was performed. Specifically, cytoplasmic and nuclear positivity were recorded as percentage of positive cells. ERBB2 was scored as 0, 1+, 2+, and 3+ according to the American Society of Clinical Oncology (ASCO) 2018 recommendations 75 (10% cutoff) with 2+ and 3+ cases considered weakly and strongly positive for complete membrane immunolabeling, respectively. The expression of the studied markers was evaluated in the epithelial/luminal component. Immunolabeling was also observed in healthy/hyperplastic adjacent mammary tissue and in this case also normal basal/myoepithelial cells were evaluated.
The specificity of all antibodies had been confirmed either by our group (data not shown) or by other authors by western blot. 5,44,49,50,59,72
Statistical Analysis
Statistical analysis was performed using Prism 7.0 (GraphPad Software). To verify mean differences among groups, the Student’s t test in case of 2 samples and the 1-way ANOVA with Tukey’s multiple comparison test in case of more than 2 samples groups were used when values were normally distributed. Mann-Whitney test in case of 2 samples and Kruskal-Wallis test in case of more than 2 samples were used when values were not normally distributed. The Spearman’s rank correlation analysis was used to analyze associations between variables. Level of significance was fixed as P < .05.
Results
Tumor Phenotype
In order to phenotypically characterize the selected tumor samples, the expression of ER, PR, and ERBB2 was evaluated by immunohistochemistry, following the American Society of Clinical Oncology/College of American Pathologists guideline recommendations. 25,75 Anti-ER, anti-PR, and anti-ERBB2 antibodies immunolabeled all the positive controls as expected. Specifically, there was nuclear immunolabeling for ER and PR in the endometrium of canine and feline uteruses. There was membranous immunolabeling for ERBB2 in more than 10% cells in the HBC used as technical positive control (3+). The expression of ER was higher than 1% in all HBC ER+ samples, whereas in TNBC there was no labeling for ER. All TNBCs were also negative for PR (<1%) and ERBB2 (scoring 0 or 1; Table 2). All CMTs were positive (>1%) for ER. ER immunolabeling was lower in grade II CMTs than grade I CMTs (P < .01; Table 2). Progesterone expression was not evaluated in CMTs. All FMTs were negative for ER (<1%), PR (<1%), and ERBB2 (scoring 0 or 1), and therefore classified as triple negative (Table 2).
Immunohistochemistry (IHC) Data in Human (HBC), Canine (CMT), and Feline (FMT) Mammary Tumorsa.
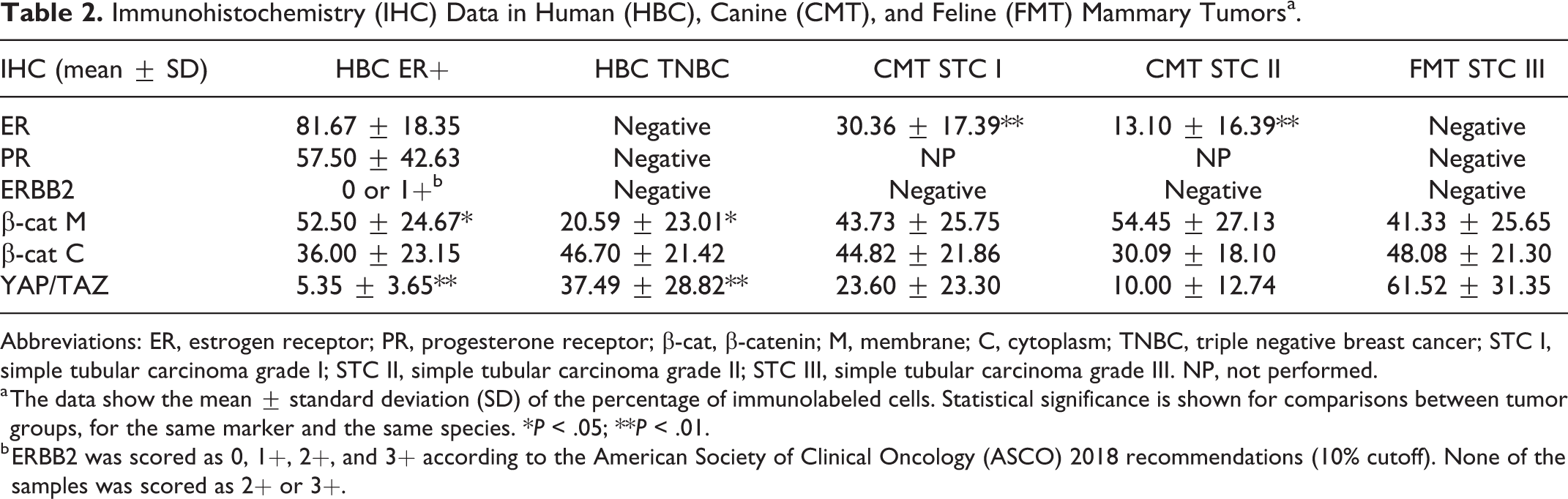
Abbreviations: ER, estrogen receptor; PR, progesterone receptor; β-cat, β-catenin; M, membrane; C, cytoplasm; TNBC, triple negative breast cancer; STC I, simple tubular carcinoma grade I; STC II, simple tubular carcinoma grade II; STC III, simple tubular carcinoma grade III. NP, not performed.
a The data show the mean ± standard deviation (SD) of the percentage of immunolabeled cells. Statistical significance is shown for comparisons between tumor groups, for the same marker and the same species. *P < .05; **P < .01.
b ERBB2 was scored as 0, 1+, 2+, and 3+ according to the American Society of Clinical Oncology (ASCO) 2018 recommendations (10% cutoff). None of the samples was scored as 2+ or 3+.
Wnt/β-Catenin Pathway
In this study, we focused on the Wnt/β-catenin pathway, investigating the mRNA and protein expression of the key molecule β-catenin/β-catenin as well as the mRNA expression of one of the downstream genes, namely CCND1. Specifically, in order to investigate whether the Wnt/β-catenin pathway was deregulated in tumor samples, the protein expression of β-catenin was evaluated by western blot using the anti-β-catenin and the anti-non-phosphorylated β-catenin antibodies, the latter recognizing the active form. Immunoblots showed bands of the expected sizes at 95 kDa (both β-catenin and non-phosphorylated β-catenin), confirming the specificity of the antibodies against the human, canine, and feline proteins. Also, we investigated the expression of β-catenin by immunohistochemistry, separately evaluating membranous and cytoplasmic immunolabeling. The anti-β-catenin antibody immunolabeled the positive control tissues as expected. Specifically, complete membranous labeling of all epithelial tissues (adnexa, epidermis, and non-tumor breast/mammary gland) was evident. Overall, in all the 3 species, β-catenin immunolabeling (Figs. 1–4) was quite heterogeneous across the samples.
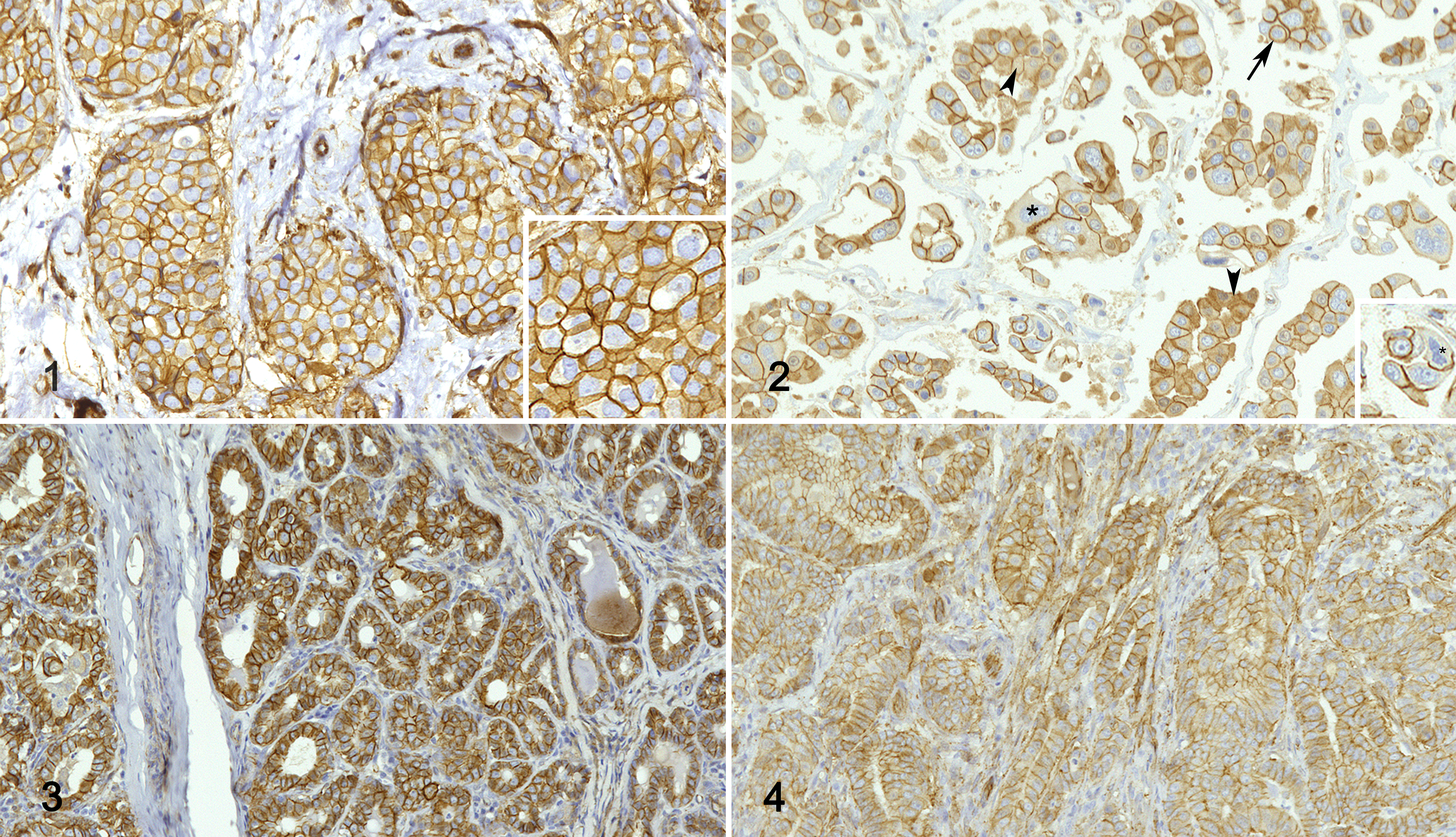
Carcinoma, mammary gland. Immunohistochemistry for β-catenin.
In women, the mRNA expression of β-catenin was not different between healthy and tumor tissues (Fig. 5), whereas the mRNA expression of CCND1 was higher in HBC ER+ tumors when compared to healthy tissues (P < .05) and to TNBCs (P < .01; Fig. 6). β-Catenin and non-phosphorylated β-catenin were present in both soluble (cytoplasmic; Fig. 7) and nuclear fractions (Fig. 8) in human samples, by western blot. Non-phosphorylated β-catenin was not evident in the soluble fraction, whereas it was present in the nuclear fraction, as expected. The expression of β-catenin in the soluble fraction (cytoplasm) was higher in tumors (ER+ tumors and TNBCs) than in healthy tissues. In the nuclear fraction, β-catenin was particularly higher in samples 4 (ER+) and 6 (TNBC) than in the other samples. In the soluble fraction (cytoplasm), non-phosphorylated β-catenin was extremely low in all the samples, whereas the nuclear fraction of tumor samples, especially samples 4 and 6, showed a higher expression than healthy tissues, suggesting an activation of the Wnt/β-catenin pathway in tumors.
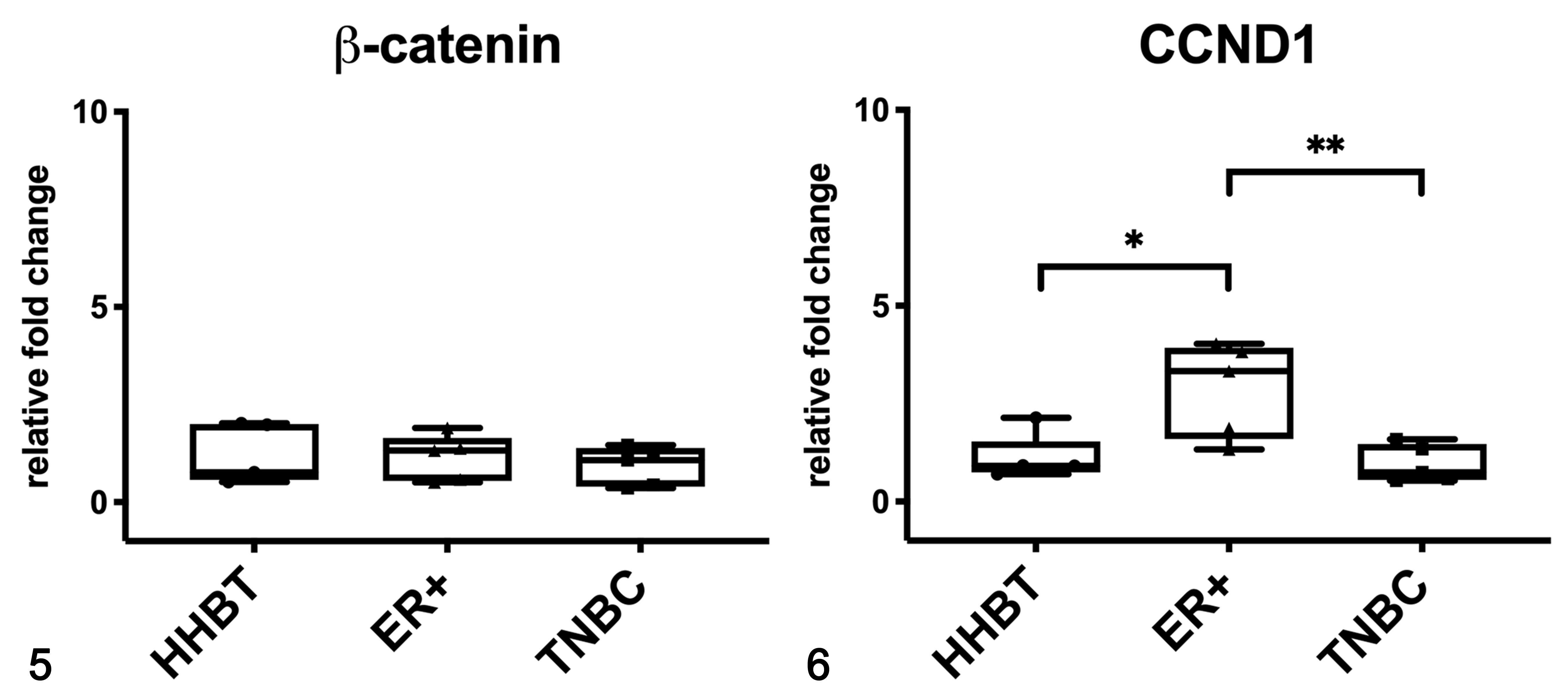
Gene expression of β-catenin (Fig. 5) and CCND1 (Fig. 6) in human breast tissues based on qPCR. Estrogen receptor-positive (ER+) tumors had a higher mRNA expression of CCND1 when compared to healthy breast tissues (HHBT) and triple negative breast cancer (TNBC). *P < .05, **P < .01. The boxes extend from the 25th to the 75th percentiles and the horizontal line represents the median. The whiskers include the minimum and maximum values. Individual values are plotted as a point/square/triangle superimposed on the graph.
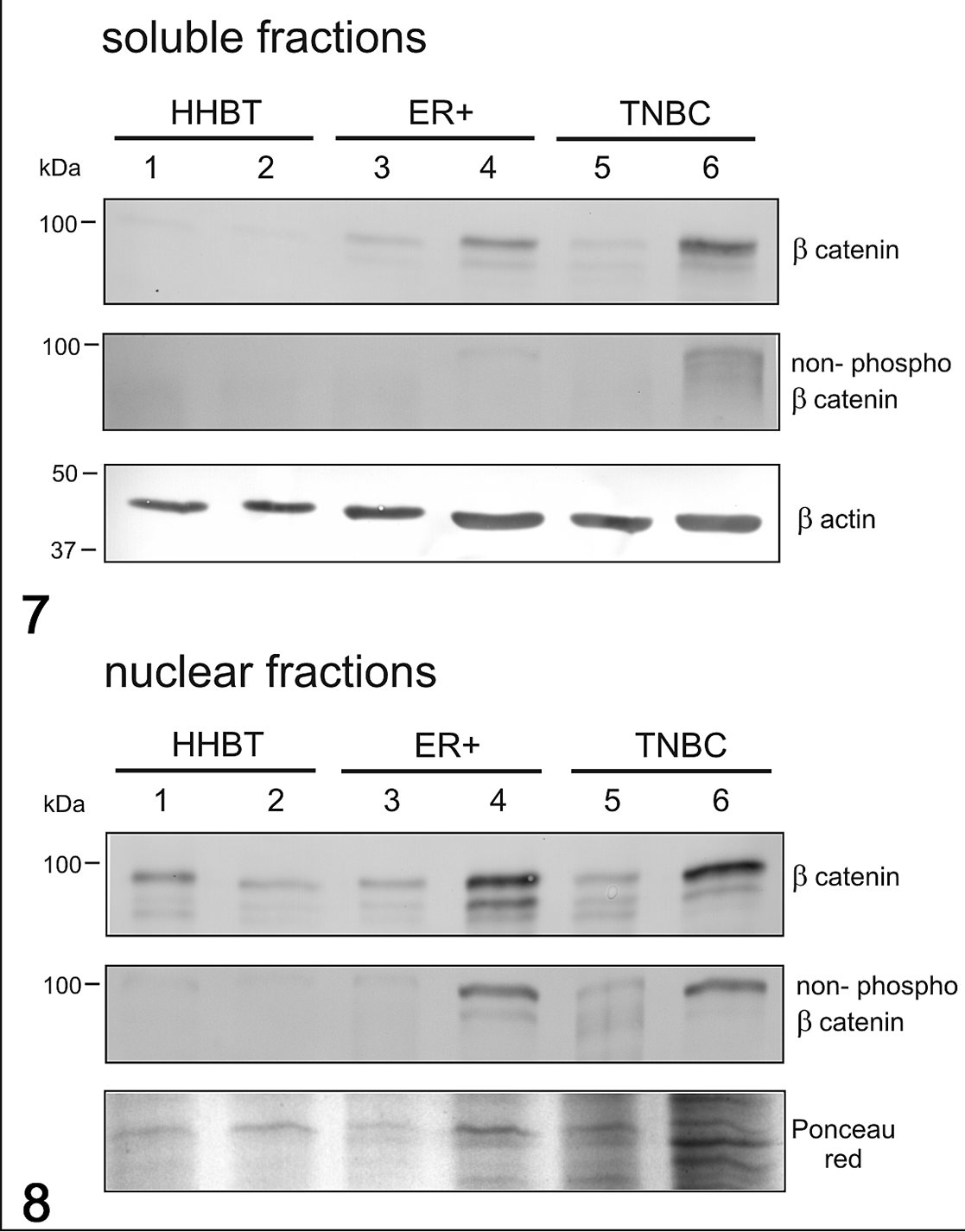
Western Blot for β-catenin and non-phosphorylated β-catenin in soluble (cytoplasmic; Fig. 7) and nuclear (Fig. 8) fractions in human breast cancer. The expression of β-catenin in the soluble fraction was higher in tumors than in healthy tissues. In the soluble fraction, non-phosphorylated β-catenin was extremely low in all samples, whereas in the nuclear fraction, the tumor samples (especially samples 4 [ER+] and 6 [TNBC]) showed higher expression than healthy tissues. β-Actin and Ponceau red were used as loading controls for soluble and nuclear fractions, respectively. HHBT, healthy human breast tissues; TNBC, triple negative breast cancer; ER+, estrogen receptor-positive breast cancer.
Immunohistochemistry results are summarized in Table 2 and graphically represented in Figure 9. In HBC ER+ tumors, β-catenin immunolabeling was predominantly membranous (Fig. 1). Conversely, TNBCs were often characterized by partial or complete loss of membranous β-catenin and by cytoplasmic β-catenin immunolabeling, suggesting an activation of the Wnt/β-catenin pathway (Fig. 2). Interestingly, the expression of membranous β-catenin in HBC ER+ tumors was higher than in TNBCs (P < .05; Table 2, Fig. 9). A negative correlation between membranous and cytoplasmic β-catenin immunolabeling was detected in HBC ER+ tumors (r = −0.888, P < .05; Fig. 10).
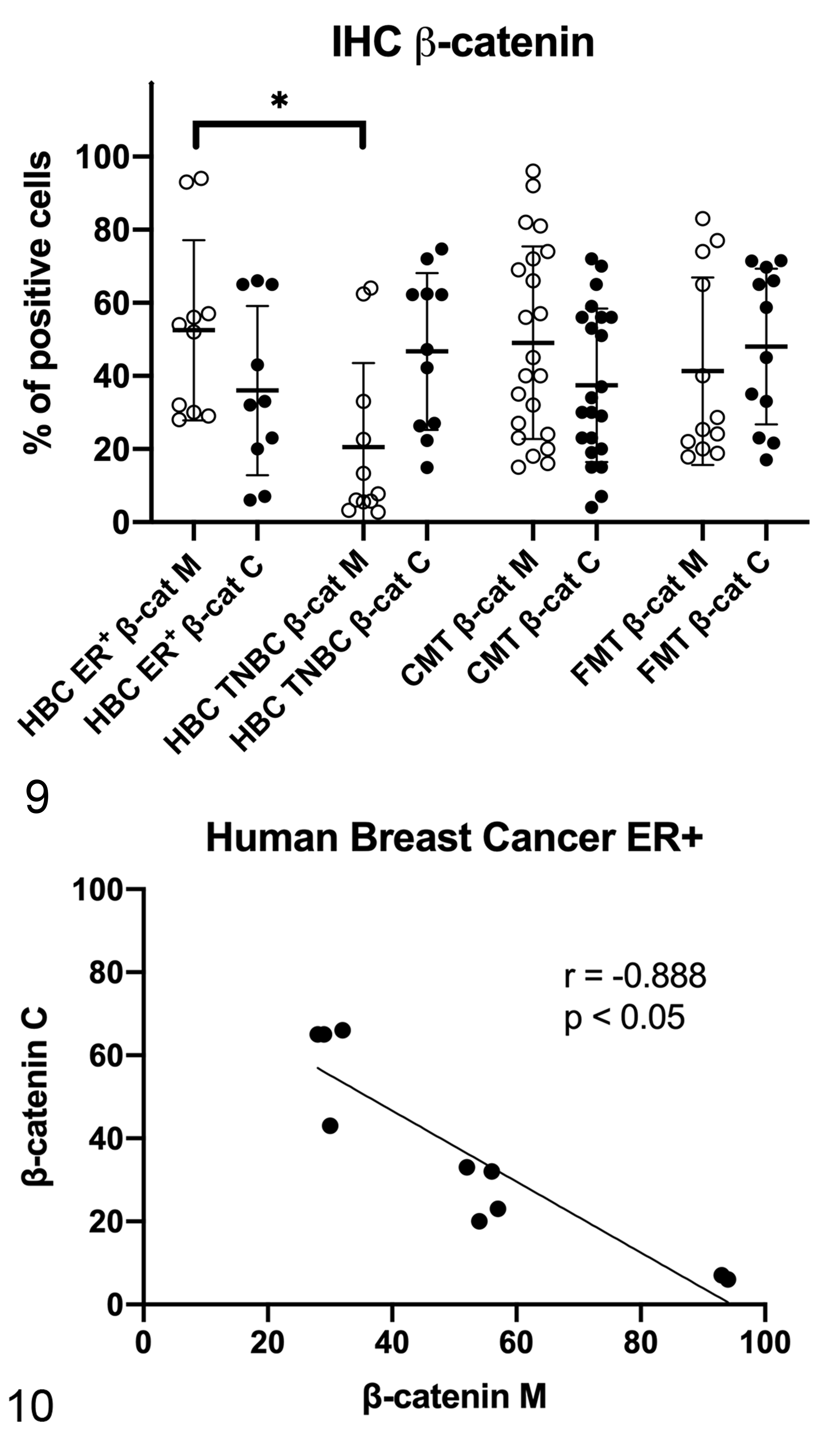
Immunohistochemistry results for β-catenin in human breast cancer (HBC), canine mammary tumors (CMTs), and feline mammary tumors (FMTs). Membranous β-catenin expression was higher in HBC estrogen receptor-positive (ER+) tumors when compared to triple negative breast cancer (TNBC). β-cat M, membranous β-catenin; β-cat C, cytoplasmic β-catenin. *P < .05.
In dogs, the mRNA expression of β-catenin was not different between healthy and tumor tissues (Fig. 11), whereas the mRNA expression of CCND1 was higher in STC grade II when compared to healthy tissues (P < .05; Fig. 12). In canine samples, western blot analysis showed β-catenin and non-phosphorylated β-catenin in both soluble (cytoplasmic; Fig. 13) and nuclear fractions (Fig. 14). β-Catenin in the soluble fraction (cytoplasm) showed a clearly higher expression in samples 5 (grade I CMT) and 6 (grade II CMT) than in healthy tissues, sample 4 (grade I CMT), and sample 7 (grade II CMT). β-Catenin in the nuclear fraction showed a higher expression in tumors than in healthy tissues. In the soluble fraction (cytoplasm), non-phosphorylated β-catenin was nearly absent in all samples, whereas in the nuclear fraction, it was higher in tumors than in healthy tissues, suggesting an activation of the Wnt/β-catenin pathway in tumors. By IHC of canine samples, a negative correlation between membranous and cytoplasmic β-catenin immunolabeling was detected (r = −0.814, P < .001; Fig. 15).
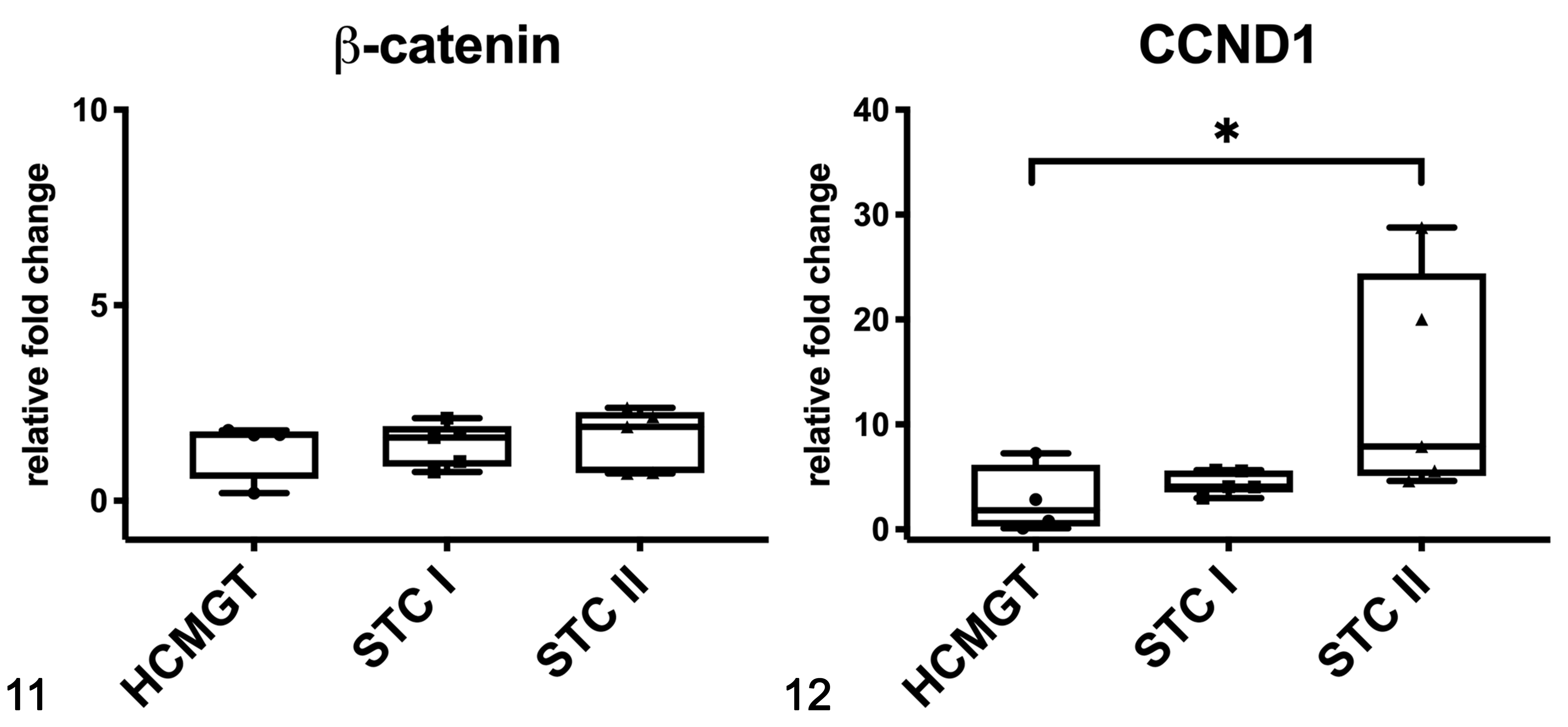
Gene expression of β-catenin (Fig. 11) and CCND1 (Fig. 12) in canine mammary tissues based on qPCR. CCND1 expression in grade II tumors was higher than in healthy tissues. HCMGT, healthy canine mammary gland tissues; STC I, simple tubular carcinoma grade I; STC II, simple tubular carcinoma grade II. *P < .05. The boxes extend from the 25th to the 75th percentiles and the horizontal line represents the median. The whiskers include the minimum and maximum values. Individual values are plotted as a point/square/triangle superimposed on the graph.
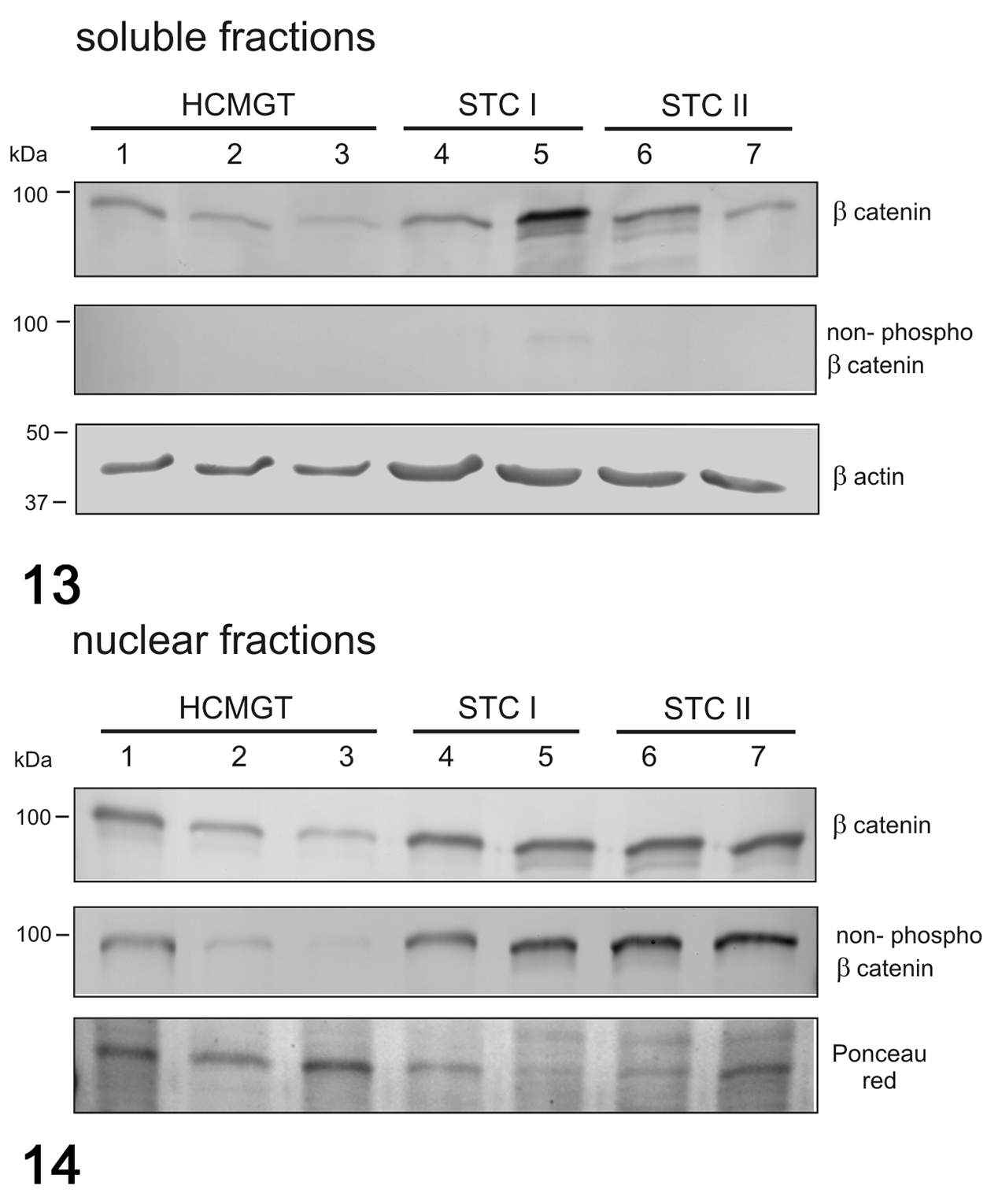
Western blot for β-catenin and non-phosphorylated β-catenin in soluble (cytoplasmic; Fig. 13) and nuclear (Fig. 14) fractions in canine mammary tumors. β-Catenin in the nuclear fraction showed a higher expression in tumors than in healthy tissues. Non-phosphorylated β-catenin was nearly absent in all the samples in the soluble fraction, whereas in the nuclear fraction, it was higher in tumors than in healthy tissues. β-Actin and Ponceau red were used as loading controls for soluble and nuclear fractions, respectively. HCMGT, healthy canine mammary gland tissues; STC I, simple tubular carcinoma grade I; STC II, simple tubular carcinoma grade II.
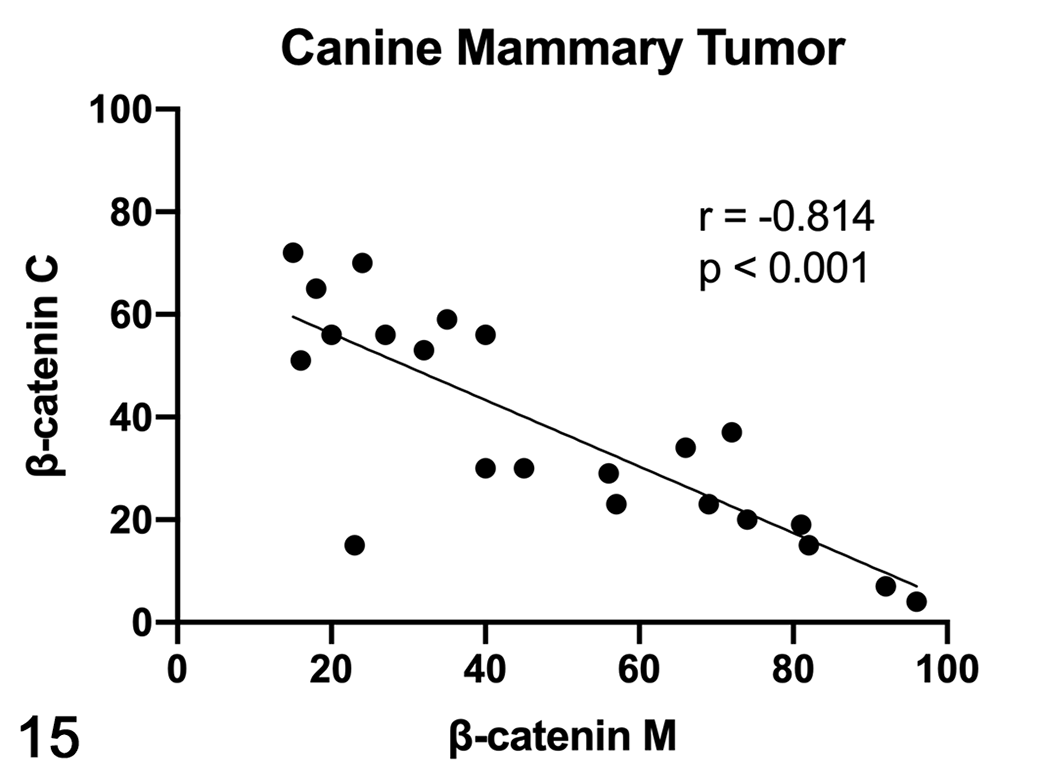
Scatterplot of membranous (M) and cytoplasmic (C) β-catenin expression in canine mammary tumors (CMTs), measured by immunohistochemistry. A strong negative correlation was found between membranous and cytoplasmic β-catenin expression in CMTs.
In cats, the mRNA expression of β-catenin (Fig. 16) and CCND1 (Fig. 17) were not different between healthy and tumor tissues. In feline samples, western blot analysis showed β-catenin and non-phosphorylated β-catenin in both soluble (cytoplasmic; Fig. 18) and nuclear fractions (Fig. 19). β-Catenin in the soluble fraction (cytoplasm) was higher in tumors than in healthy tissues, whereas in the nuclear fraction it was not clearly different across the samples. In the soluble fraction (cytoplasm), non-phosphorylated β-catenin was nearly absent in all samples, whereas in the nuclear fraction, the expression was higher in tumors than in healthy tissues, suggesting an activation of the Wnt/β-catenin pathway in tumors. By IHC of feline samples, a negative correlation between membranous and cytoplasmic β-catenin immunolabeling was detected (r = −0.909, P < .001; Fig. 20).
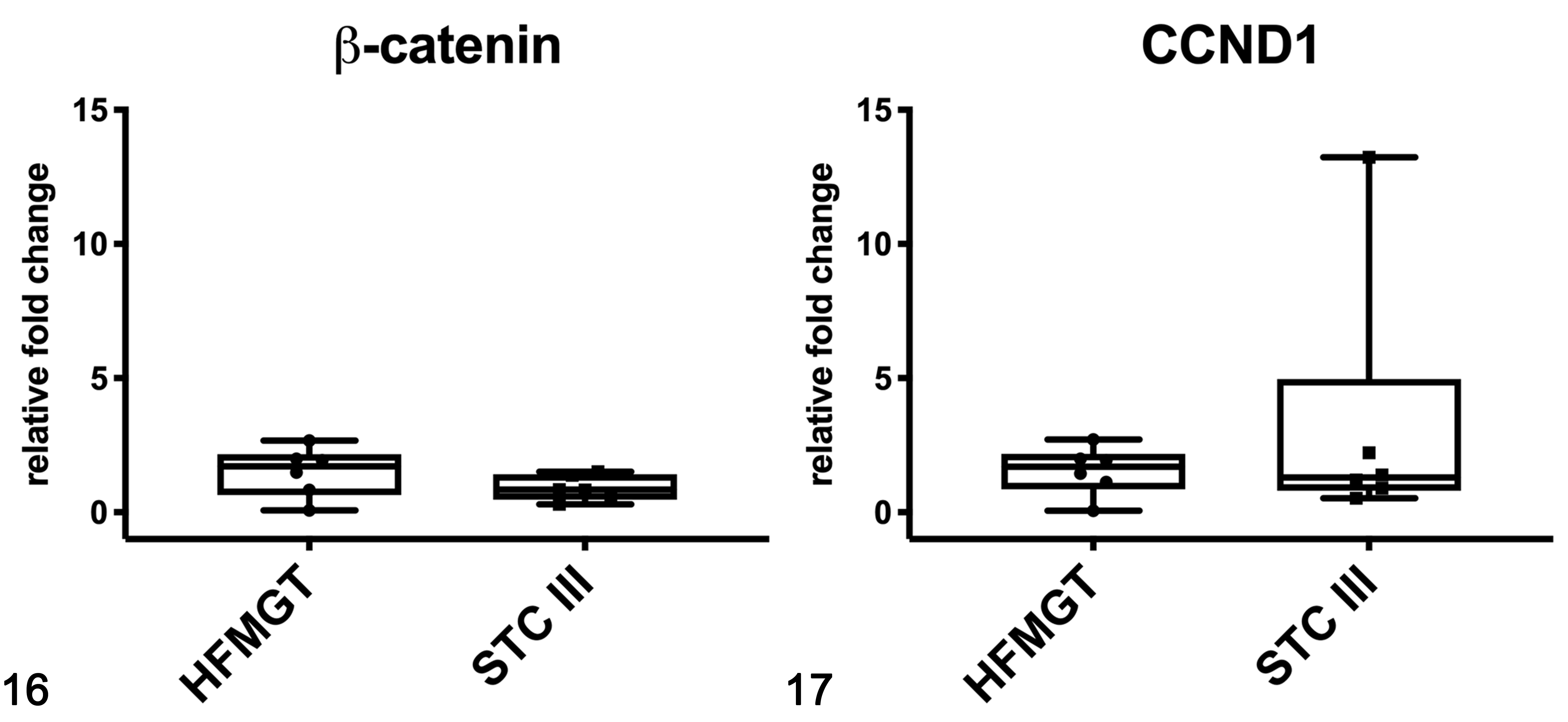
Gene expression of β-catenin (Fig. 16) and CCND1 (Fig. 17) in feline mammary tissues based on qPCR. HFMGT, healthy feline mammary gland tissues; STC III, simple tubular carcinoma grade III. The boxes extend from the 25th to the 75th percentiles and the horizontal line represents the median. The whiskers include the minimum and maximum values. Individual values are plotted as a point/square superimposed on the graph.
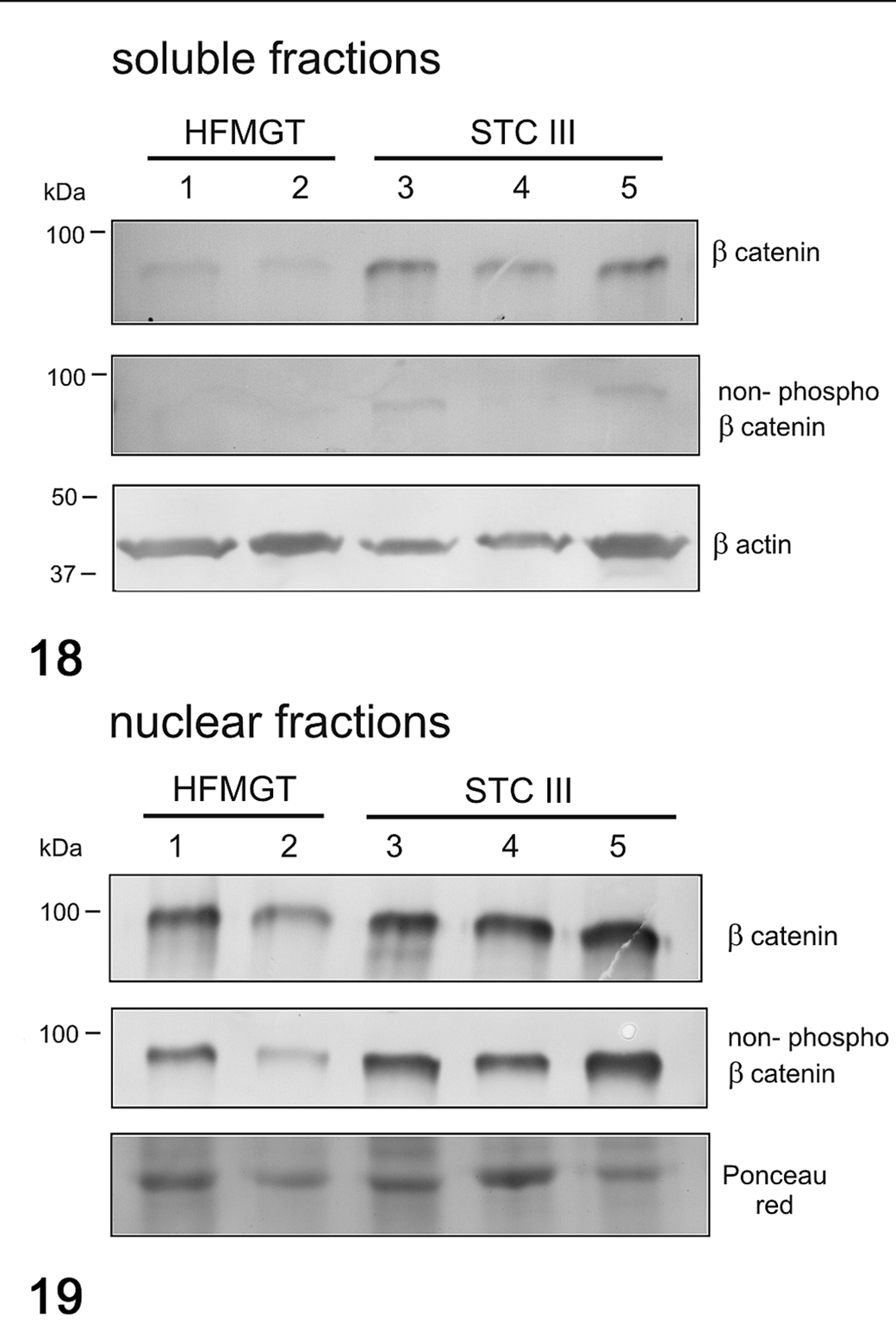
Western blot for β-catenin and non-phosphorylated β-catenin in soluble (cytoplasmic; Fig. 18) and nuclear (Fig. 19) fractions in feline mammary tumors. β-Catenin in the soluble fraction was higher in tumors that in healthy tissues. Non-phosphorylated β-catenin was nearly absent in all samples in the soluble fraction, whereas in the nuclear fraction, it was higher in tumors than in healthy tissues. β-Actin and Ponceau red were used as loading controls for soluble and nuclear fractions, respectively. HFMGT, healthy feline mammary gland tissues; STC III, simple tubular carcinoma grade III.
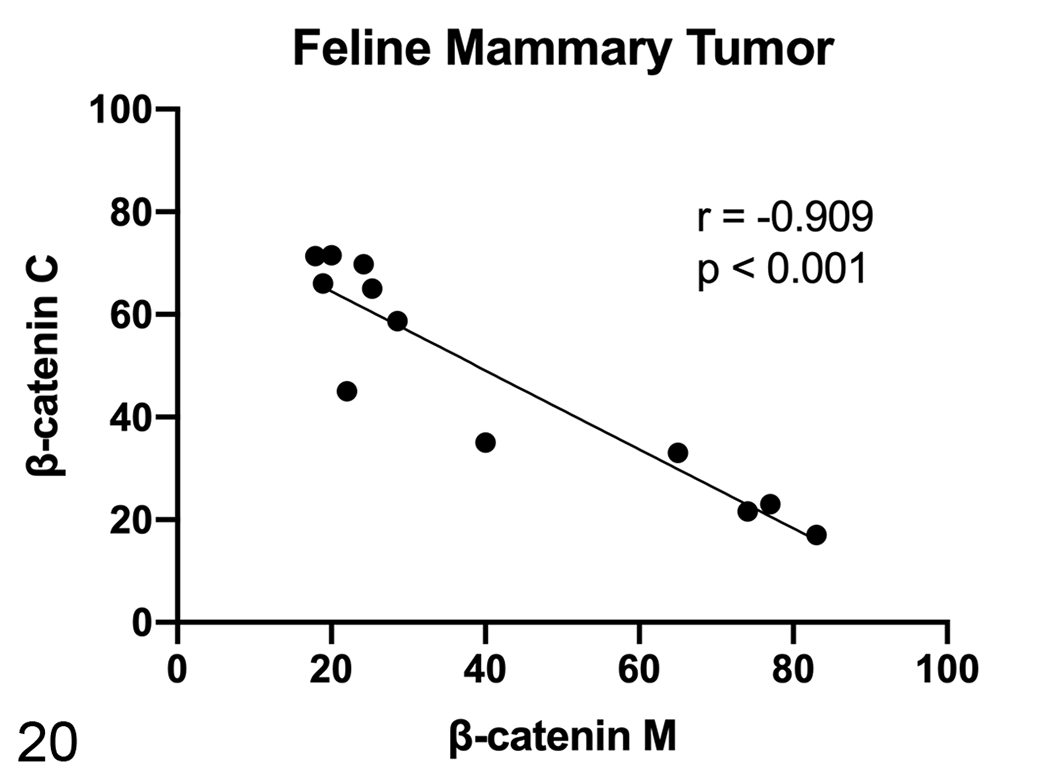
Scatterplot of membranous (M) and cytoplasmic (C) β-catenin expression in feline mammary tumors (FMTs), measured by immunohistochemistry. A strong negative correlation was found between membranous and cytoplasmic β-catenin expression in FMTs.
Hippo Pathway
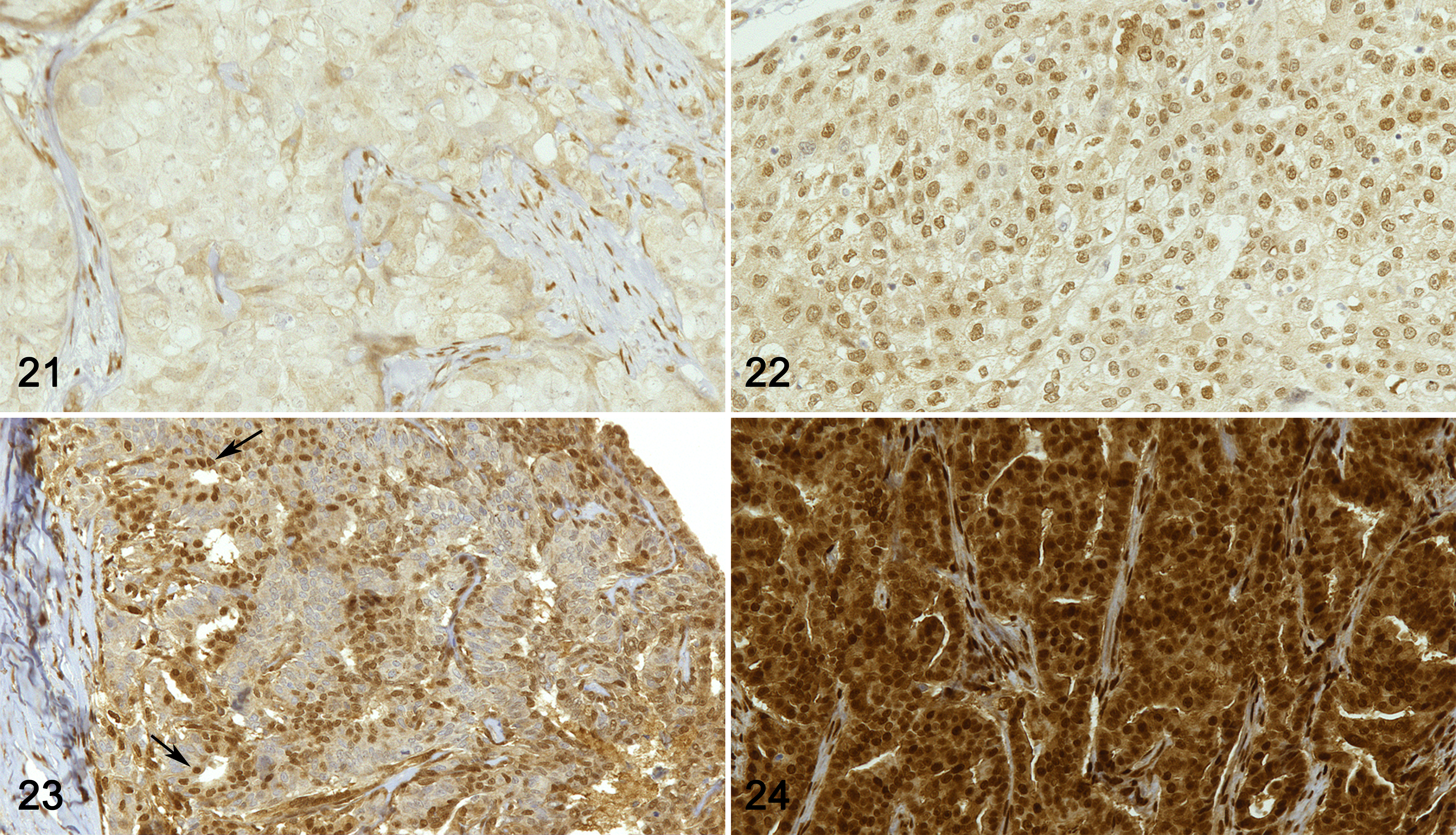
In addition to the Wnt/β-catenin pathway, we investigated the Hippo pathway, studying the mRNA and the protein expression of 2 transcriptional coactivators that play a crucial role in the signaling cascade, namely, YAP and TAZ, as well as the mRNA expression of downstream genes, namely, CTGF and ANKRD1. The protein expression of YAP and TAZ was evaluated by immunohistochemistry as well as by western blotting, the latter recognizing the 2 molecules separately. Immunoblots showed bands of the expected sizes at 70 kDa (YAP) and 55 kDa (TAZ), confirming the specificity of the antibodies against human, canine, and feline proteins, as previously described. 4 Anti-WWTR1 antibody immunolabeled all of the positive control tissues, as expected. Specifically, adnexa and epidermis showed nuclear positivity in basal cells in all species. Additionally, the dermis, subcutaneous tissue, and tumor stroma showed nuclear positivity in fibroblasts and endothelial cells. Feline and canine hyperplastic mammary gland, adjacent to the lesions, showed intense nuclear labeling of the basal/myoepithelial cells, whereas luminal cells were negative. Positive cells were based on nuclear labeling. Immunohistochemistry results for YAP/TAZ are shown in Table 2 and Figures 21 to 25.
Carcinoma, mammary gland. Immunohistochemistry for YAP/TAZ.
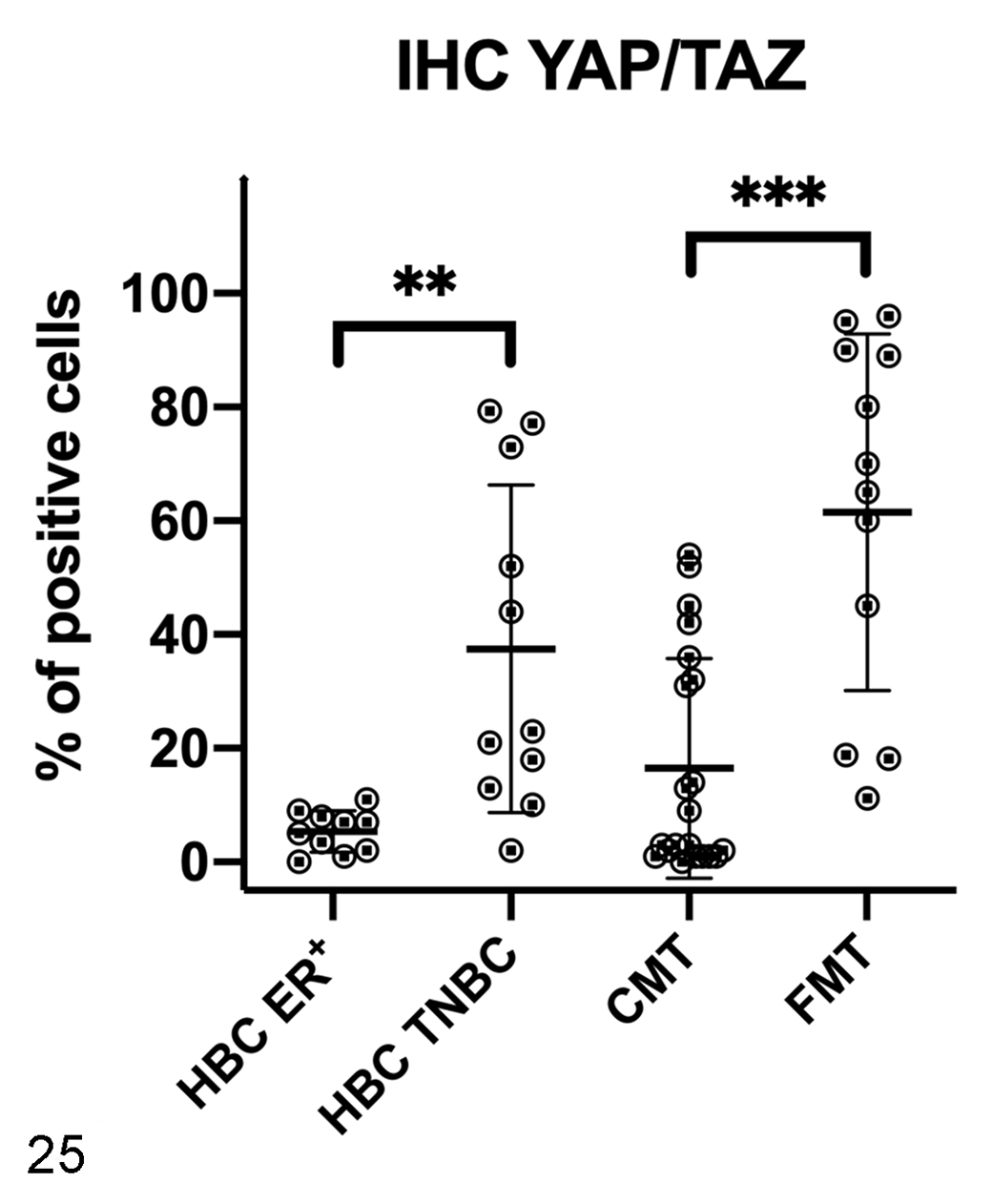
Immunohistochemistry results for YAP/TAZ in human breast cancer (HBC), canine mammary tumors (CMTs), and feline mammary tumors (FMTs). YAP/TAZ expression was higher in triple negative breast cancer (TNBC) and FMTs when compared to HBC estrogen receptor-positive (ER+) and CMTs, respectively. **P < .01, ***P < .001.
In women, the mRNA expression of YAP was surprisingly lower in HBC ER+ and in TNBCs when compared to healthy tissues (P < .05; Fig. 26), whereas no significant differences between healthy and tumor tissues were found for the mRNA expression of TAZ (Fig. 27), CTGF (Fig. 28), and ANKRD1 (Fig. 29). In human samples, western blot analysis showed YAP and TAZ in both soluble (cytoplasmic; Fig. 30) and nuclear fractions (Fig. 31). The expression of YAP and TAZ was higher in both soluble (cytoplasm) and nuclear fractions in tumors (ER+ tumors and TNBCs) than in healthy tissues. By IHC, YAP/TAZ immunolabeling, although heterogeneous, was higher in TNBCs when compared to HBC ER+ (P < .01; Figs. 21, 22, 25).
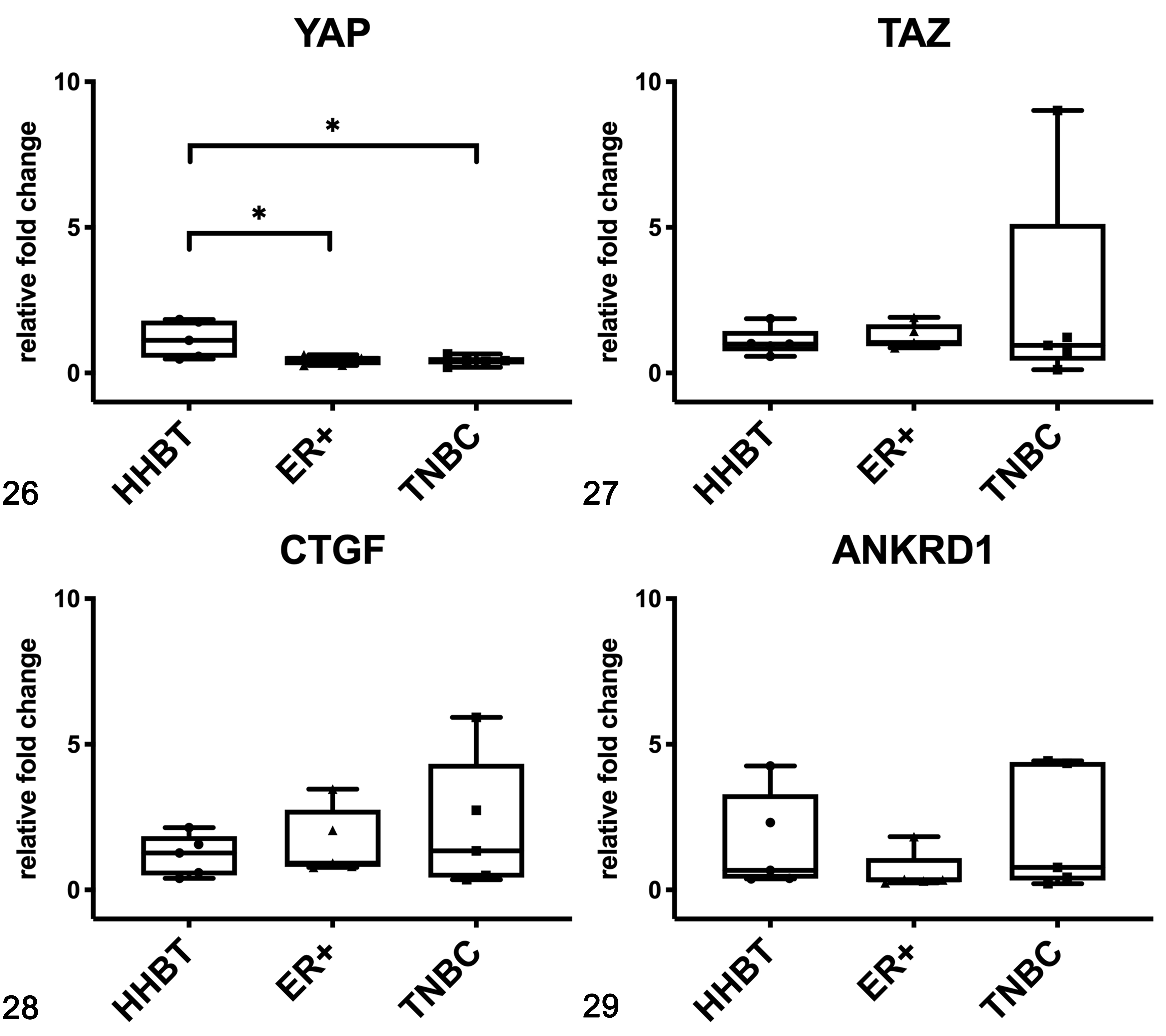
Gene expression of YAP (Fig. 26), TAZ (Fig. 27), CTGF (Fig. 28), and ANKRD1 (Fig. 29) in human breast tissues based on qPCR. Estrogen receptor-positive (ER+) tumors and triple negative breast cancer (TNBC) had a lower expression of YAP than healthy breast tissues (HHBT). *P < .05. The boxes extend from the 25th to the 75th percentiles and the horizontal line represents the median. The whiskers include the minimum and maximum values. Individual values are plotted as a point/square/triangle superimposed on the graph.
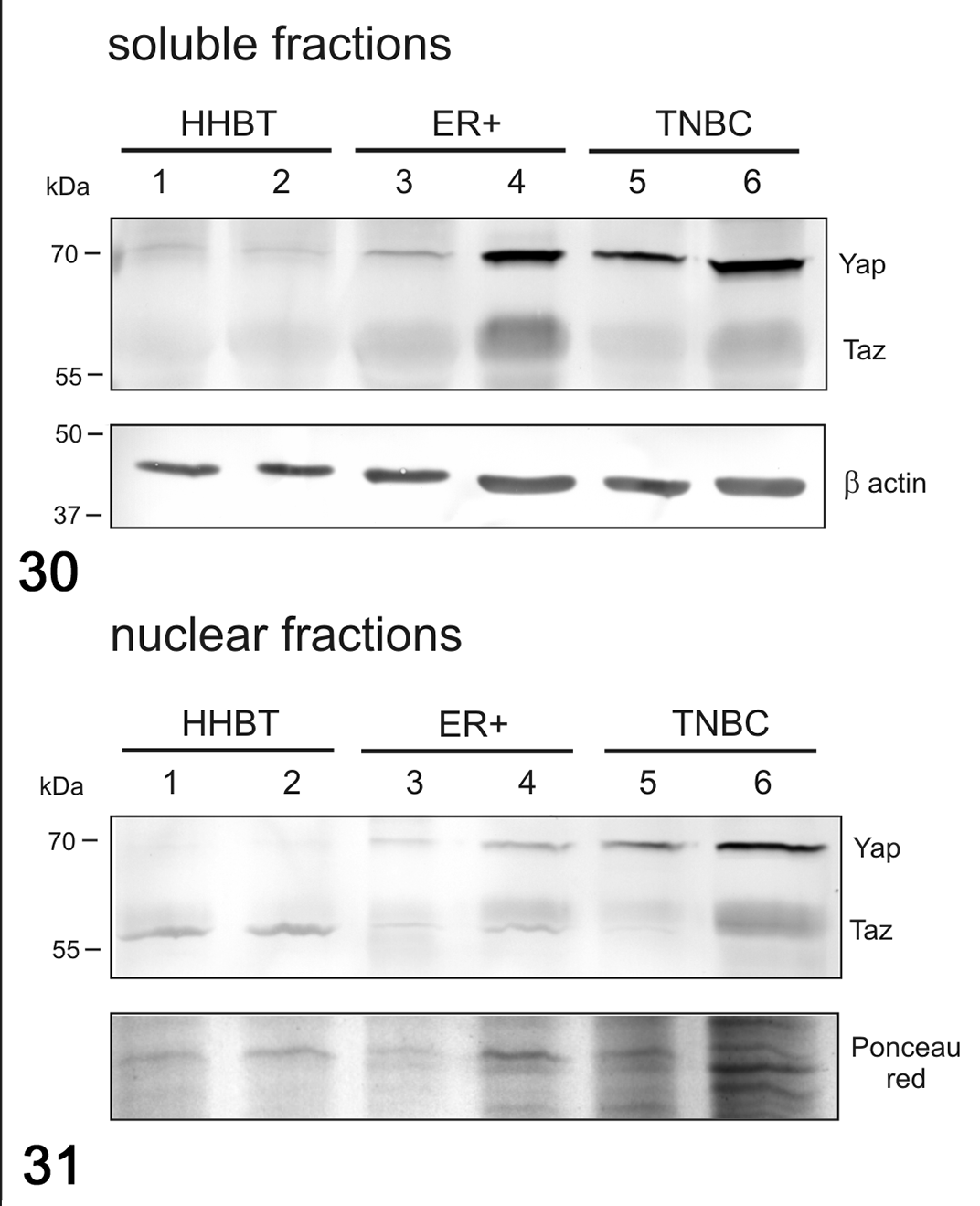
Western Blot for YAP and TAZ in soluble (cytoplasmic; Fig. 30) and nuclear (Fig. 31) fractions in human breast cancer. The expression of YAP and TAZ was higher in tumors than in healthy tissues for both the soluble and nuclear fractions. β-Actin and Ponceau red were used as loading controls for soluble and nuclear fractions, respectively. HHBT, healthy human breast tissues; ER+, estrogen receptor positive breast cancer; TNBC, triple negative breast cancer.
In dogs, no differences were found in mRNA expression of YAP (Fig. 32), and TAZ (Fig. 33) between healthy and tumor tissues, whereas the mRNA expression of CTGF (Fig. 34) and ANKRD1 (Fig. 35) tended to be higher in STC grade II than in STC grade I and healthy tissues, although not statistically significant. In canine samples, western blot analysis showed YAP and TAZ in both soluble (cytoplasmic; Fig. 36) and nuclear fractions (Fig. 37). The expression of YAP and TAZ in the soluble fraction (cytoplasm) was higher in tumors when compared to healthy tissues. In the nuclear fraction, YAP and TAZ were higher in grade II CMTs than healthy tissues and grade I CMTs.
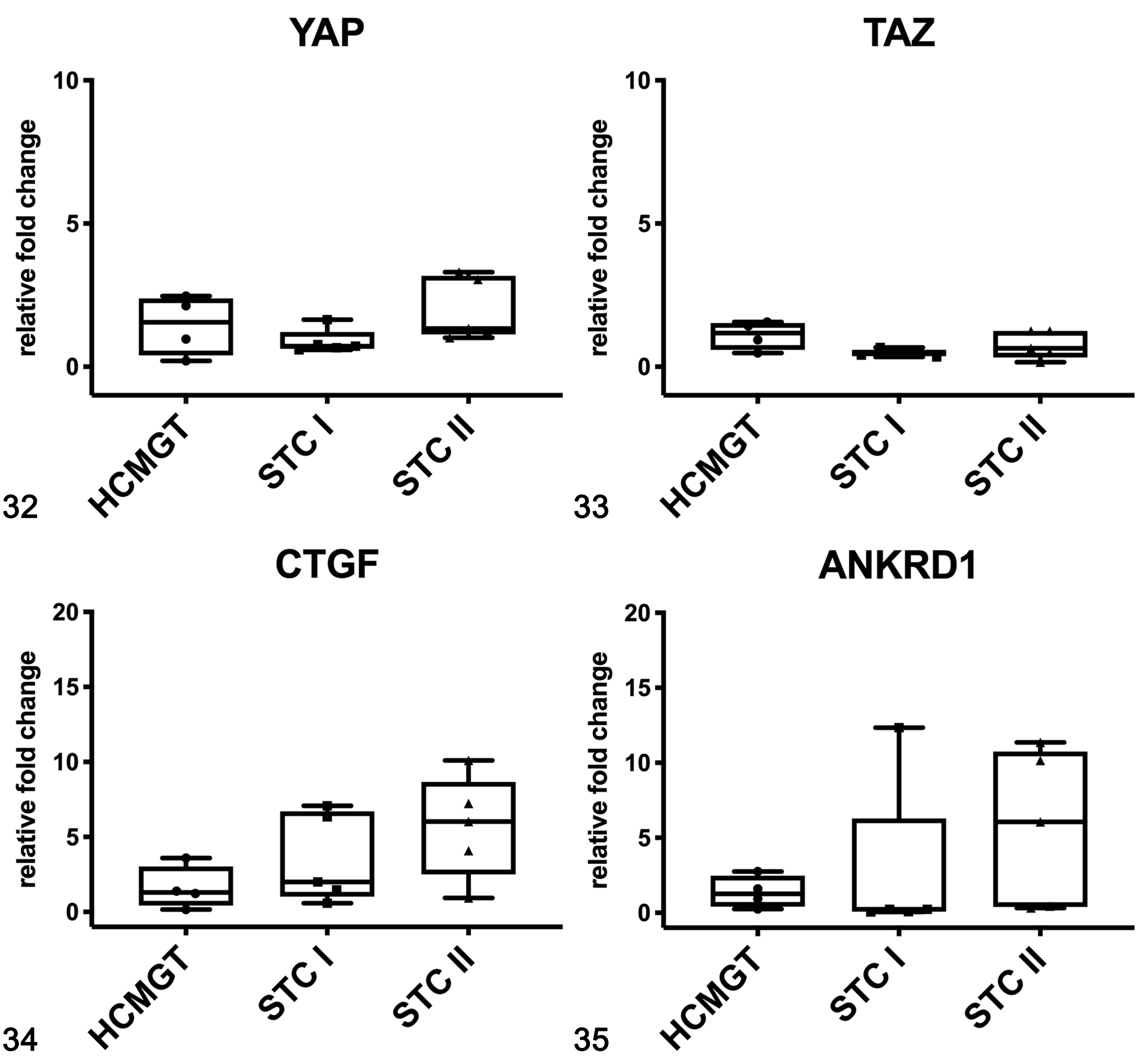
Gene expression of YAP (Fig. 32), TAZ (Fig. 33), CTGF (Fig. 34), and ANKRD1 (Fig. 35) in canine mammary gland tissues based on qPCR. CTGF and ANKRD1 mRNA expression in grade II tumors was higher than in healthy tissues. HCMGT, healthy canine mammary gland tissues; STC I, simple tubular carcinoma grade I; STC II, simple tubular carcinoma grade II. The boxes extend from the 25th to the 75th percentiles and the horizontal line represents the median. The whiskers include the minimum and maximum values. Individual values are plotted as a point/square/triangle superimposed on the graph.
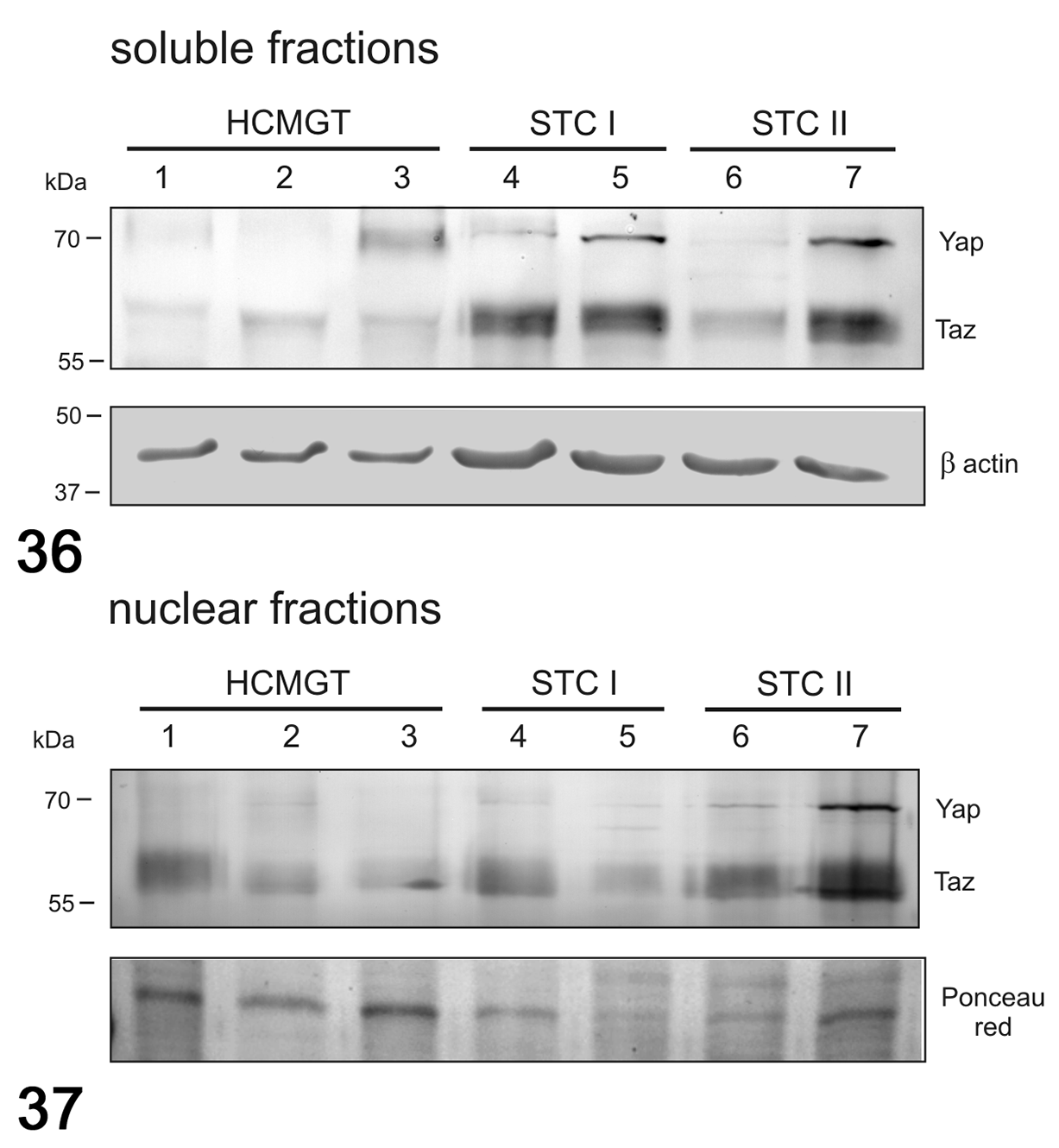
Western blot for YAP and TAZ in soluble (cytoplasmic; Fig. 36) and nuclear (Fig. 37) fractions in canine mammary tumors. The expression of YAP and TAZ was higher in the soluble fraction in tumors compared to healthy tissues. In the nuclear fraction, YAP and TAZ were higher in grade II tumors than healthy tissues and grade I tumors. β-Actin and Ponceau red were used as loading controls for soluble and nuclear fractions, respectively. HCMGT, healthy canine mammary gland tissues; STC I, simple tubular carcinoma grade I; STC II, simple tubular carcinoma grade II.
In cats, no differences were found in the mRNA expression of YAP (Fig. 38), TAZ (Fig. 39), CTGF (Fig. 40), and ANKRD1 (Fig. 41) between healthy and tumor tissues. In feline samples, western blot analysis showed YAP and TAZ in both soluble (cytoplasmic; Fig. 42) and nuclear fractions (Fig. 43). The expression of YAP in the soluble fraction (cytoplasm) was higher in FMTs samples 4 and 5 than in healthy tissues and FMT sample 3, whereas in the nuclear fraction the expression was higher in tumors than in healthy tissues. TAZ was higher in both soluble (cytoplasm) and nuclear fractions in tumors when compared to healthy tissues. By IHC, YAP/TAZ immunolabeling, although heterogeneous, was higher in FMTs when compared to CMTs (P < .001; Figs. 23–25).
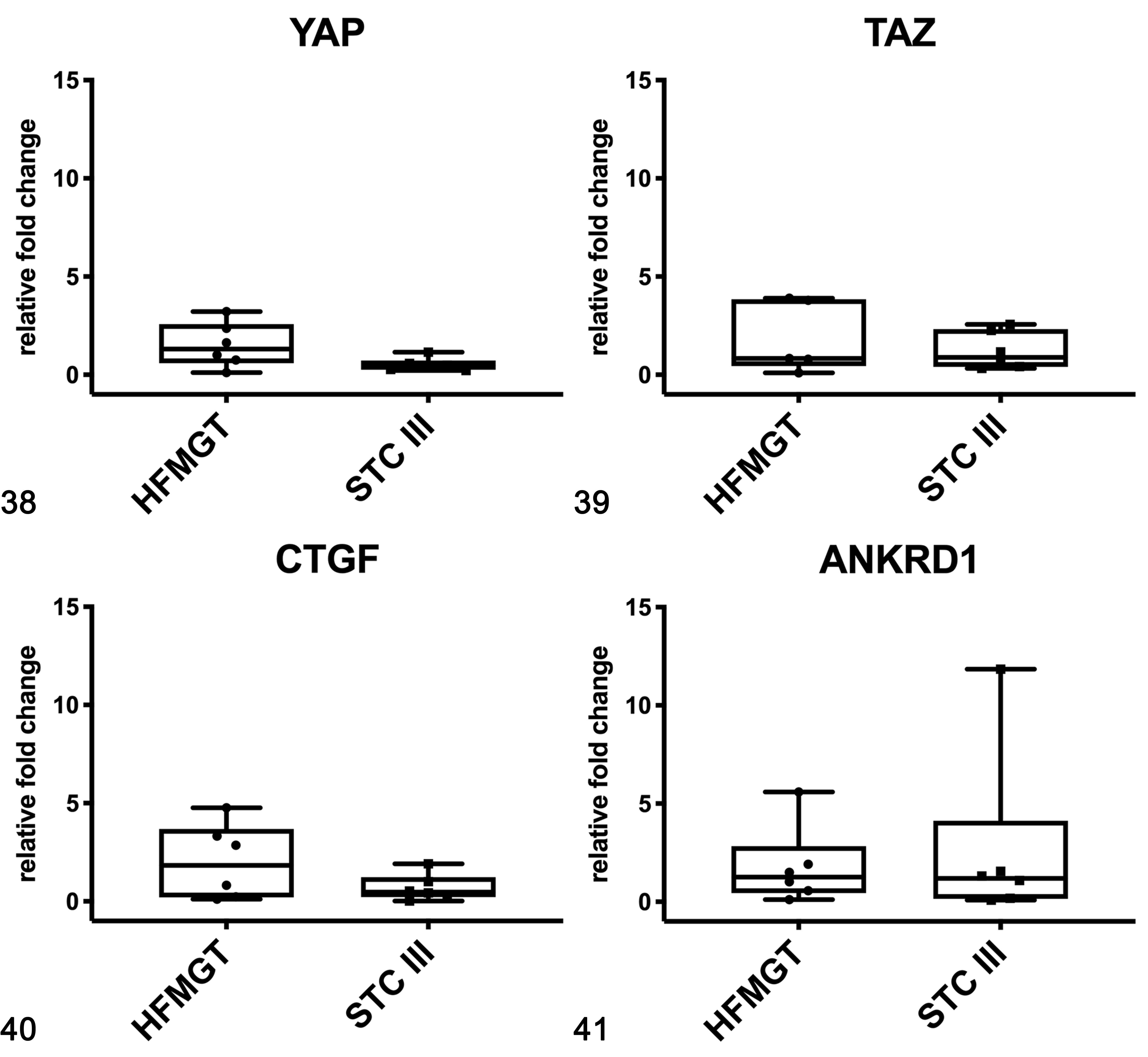
Gene expression of YAP (Fig. 38), TAZ (Fig. 39), CTGF (Fig. 40), and ANKRD1 (Fig. 41) in feline mammary gland tissues based on qPCR. HFMGT, healthy feline mammary gland tissues; STC III, simple tubular carcinoma grade III. The boxes extend from the 25th to the 75th percentiles and the horizontal line represents the median. The whiskers include the minimum and maximum values. Individual values are plotted as a point/square superimposed on the graph.
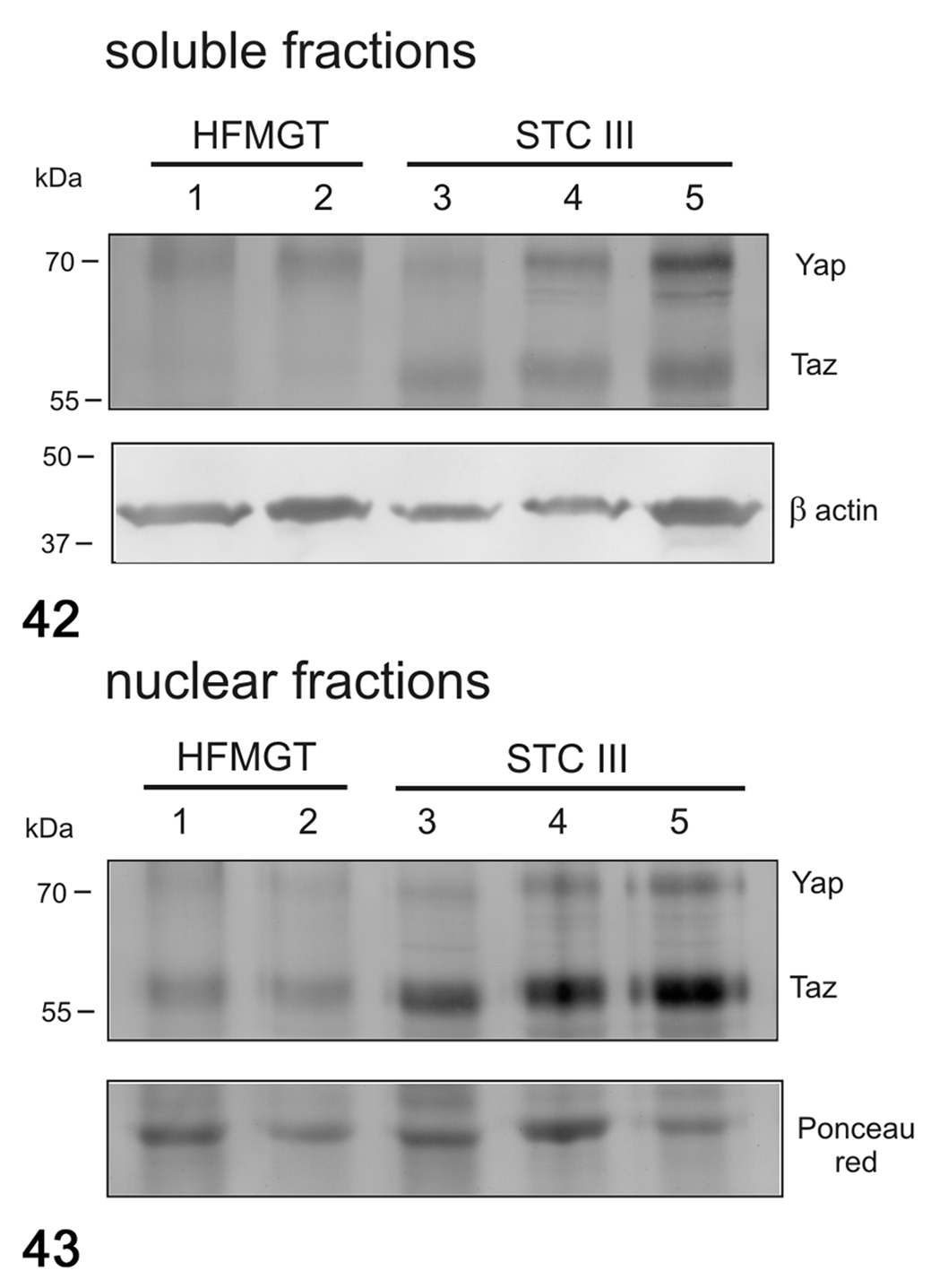
Western blot for YAP and TAZ in soluble (cytoplasmic; Fig. 42) and nuclear (Fig. 43) fractions in feline mammary tumors. The expression of YAP and TAZ in both soluble and nuclear fractions was higher in tumors than in healthy tissues. β-Actin and Ponceau red were used as loading controls for soluble and nuclear fractions, respectively. HFMGT, healthy feline mammary gland tissues; STC III, simple tubular carcinoma grade III.
Discussion
In this study, we focused on two of the main pathways that are thought to be deregulated in cancer: the Wnt/β-catenin and Hippo pathways. 19,53
The Wnt family is a group of proteins implicated in several cellular functions, such as organ formation, stem cell renewal, and cell survival. 15 Accumulating evidence indicates that the Wnt/β-catenin pathway contributes to the neoplastic process, of which β-catenin is one of the key downstream effectors. 48 An inappropriate activation of this pathway leads to the development of human cancer, including breast cancer. 33
More than half of HBC are characterized by an activation of Wnt, which is associated with lower overall survival. 38 High levels of Wnt receptor and co-receptor expression, as well as aberrant activation of β-catenin, have been detected in HBC. Downregulation of the Wnt/β-catenin pathway can reduce the spontaneous invasion of breast cancer cells. 2,77 Several studies have reported increased cytoplasmic and nuclear β-catenin in primary breast cancers, especially in basal-like breast cancers, and this has been correlated with poor prognosis and decreased survival. 38,42,55
In our study, we did not find any difference in terms of mRNA expression of β-catenin between healthy and neoplastic tissues in the 3 species. CCND1, one of the downstream genes regulated by the Wnt/β-catenin pathway, was higher in ER+ tumors, but not in TNBCs, when compared to healthy tissues. Similarly, CCND1 mRNA level was higher in CMTs grade II when compared to healthy tissues. These results are quite different from what other authors found in human breast, ovarian, colorectal, and squamous cell carcinomas. 11 –13,32 Cheng and collaborators found a higher mRNA level of β-catenin and CCND1 in HBC tissues when compared to the adjacent non-cancerous cells. 12 However, they did not distinguish between ER+ tumors and TNBCs. Also, they measured mRNA expression on laser capture microdissected tissues, thereby eliminating any non-mammary tissue, that was instead present in our samples and that could significantly lead to unexpected results. In dogs, Han and collaborators found a higher mRNA expression of β-catenin in canine oral melanomas when compared to normal melanocytes. 27 Moreover, Yu and collaborators found a higher mRNA expression of β-catenin in CMTs compared to healthy tissues. However, their samples were not restricted to simple carcinomas, but also included other tumor subtypes, making the 2 studies difficult to compare. 78 To the best of our knowledge, β-catenin and CCND1 mRNA expression have never been investigated in FMTs.
In all the 3 species, a slightly higher protein expression of β-catenin in tumor than healthy tissues was found by western blot in both cytoplasm and nucleus. Interestingly, increased non-phosphorylated β-catenin (active form) was found in the nuclear fraction of tumors compared to healthy tissues. These results suggest activation of the Wnt/β-catenin pathway in tumors, which is apparently not regulated at the mRNA level but at the protein level. In other words, our data could indicate a posttranscriptional regulation of β-catenin accumulation into the cytoplasm, as already reported by other studies. 39 Indeed, increased β-catenin within the cytoplasm and increased non-phosphorylated protein within the nucleus indicates the translocation of β-catenin from the membrane to the cytoplasm and subsequently into the nucleus where it activates downstream gene expression (oncogenic function). As mentioned, one example of these downstream genes is CCND1, which was increased in human ER+ tumors and in grade II CMTs, although the mRNA expression of β-catenin was similar between healthy and tumor tissues. Conversely, the mRNA expression of CCND1 was not increased in TNBCs and FMTs compared to healthy tissues. This could be due to the fact that CCND1 expression is regulated not only by the Wnt/β-catenin pathway, but also by the PI3K-Akt and NF-kB pathways, as well as by Ras, Rac, Rho, and growth factors such as the epidermal growth factor (EGF). 57 Also, it has been shown that CCND1 overexpression correlated with ER positivity. 35,67 This could be one of the reasons why mRNA expression of CCND1 was increased in HBC ER+ tumors and CMTs (positive for ER) and was not increased in TNBCs and FMTs (negative for ER). Other downstream genes should be further investigated to analyze Wnt/β-catenin pathway. To our knowledge, protein expression of β-catenin and phosphorylated β-catenin in cytoplasmic and nuclear fractions using western blot has never been previously investigated in dogs and cats.
By IHC, β-catenin protein expression in the 3 species was highly heterogeneous. In human ER+ tumors, its protein expression was predominantly membranous, whereas in TNBCs it was predominantly cytoplasmic, indicating a possible activation of the Wnt/β-catenin pathway in the latter. 20 Interestingly, a negative correlation between membranous and cytoplasmic β-catenin protein expression was found in all 3 species as confirmed by other authors in HBC, 20 CMTs, 60 and FMTs. 79 No nuclear positivity to β-catenin was found in any of the 3 species, possibly due to a low sensitivity of IHC to detect the protein.
The Hippo pathway regulates organ size and maintains tissue stability by controlling cell proliferation and apoptosis. 18 Its deregulation results in tissue overgrowth and tumorigenesis in humans. 26,31,34,41,52,62,73 YAP and TAZ transcriptional coactivators are the key effectors of this pathway, and both proteins are overexpressed in a wide spectrum of cancers. 29,36,51,68 Bartucci and collaborators suggested that YAP and TAZ play key roles in the aggressive behavior of HBCs and may represent a major determinant of fatal clinical outcome in advanced HBC patients. 3 Very little is known about the potential involvement of the Hippo pathway in canine and feline mammary tumors. In a preliminary study performed on CMTs and FMTs, we found by IHC that YAP/TAZ protein expression was higher in grade III carcinomas compared to grade I carcinomas in both species, suggesting a potential role in disease progression. 4 More recently, Guillemette and collaborators investigated the protein expression and function of YAP/TAZ in 6 CMT cell lines. 24 They found that pharmacologic inhibition of YAP-mediated transcriptional co-activation induces apoptosis in CMT cells, and attenuates key behaviors of malignant cells including migration, invasion, and anchorage-independent growth. Rico and collaborators evaluated the protein expression of YAP and TAZ in normal canine mammary gland tissue, hyperplastic tissue, and in benign and malignant CMT across all stages of disease progression. 61 They found that, by IHC, the protein expression of TAZ was increased in both the cytoplasm and nucleus of cells within tissues with lobular hyperplasia when compared to normal tissues. Moreover, by IHC, nuclear YAP and TAZ protein expression was significantly increased in malignant versus benign tumors, whereas cytoplasmic levels of both proteins were comparable in both groups. Immunoblots showed that TAZ increased in malignant tumors relative to normal mammary gland, suggesting a correlation between increased TAZ expression and the acquisition of a malignant phenotype. Conversely, YAP protein expression did not vary between normal and neoplastic tissues. 61
In this study, we analyzed the mRNA expression of YAP, TAZ, CTGF, and ANKRD1, as well as the protein expression of YAP/TAZ in a subset of normal and tumor tissues of human, canine, and feline mammary gland. In all the 3 species, TAZ mRNA expression was not different between normal and tumor tissues and between different tumor subgroups. In dogs and cats, YAP mRNA expression was not different between normal and tumor tissues or between different tumor subgroups. Surprisingly, the mRNA expression of YAP in ER+ tumors and TNBCs was lower than in the healthy mammary gland.
Conversely, in the 3 species, the protein expression of YAP and TAZ as analyzed by western blot was overall higher in tumors than in healthy tissues in both cytoplasm and nucleus, which reflects what we previously found by IHC. 4 This discrepancy between mRNA and protein expression data could be due to a regulation of YAP/TAZ expression at a posttranscriptional level, rather than at the mRNA level, as proposed by others. 82 In other words, the greater presence of the proteins was not due to a higher mRNA expression but to an accumulation of the unphosphorylated proteins presumably caused by a deregulation of the Hippo pathway. Notably, Luu and collaborators found an association between YAP, metastasis, and survival in canine osteosarcoma, which was limited to the protein level and was not observed at the mRNA level, further corroborating our hypothesis based on the posttranscriptional/posttranslational regulation of their expression. 43
Although in CMTs there was a trend in the mRNA expression of CTGF that was higher in grade II than in grade I tumors and healthy tissues, the mRNA expression of Hippo pathway downstream genes CTGF and ANKRD1 was not significantly different between normal and tumor tissues in any of the 3 species. This could be due to the high variability among different samples in our study as well as to the low statistical power due to the small number of cases. Additionally, other downstream genes related to the Hippo pathway could be included to better analyze this system.
Another possible explanation is that healthy tissues were not only composed of the mammary gland per se, but also of other tissues such as connective tissue, adipose tissue, and blood and lymph vessels. Indeed, some authors showed that YAP/TAZ expression in adipose tissue could be deregulated by several conditions, such as obesity. 71 Different patients could also have a slightly different composition of the healthy mammary gland; for instance, the amount of adipose tissue compared to connective tissue could be higher in some patients than others, thereby explaining the high variability found between samples. It is also important to mention that healthy MGTs were collected from tumor-bearing patients. Therefore, what was considered “healthy mammary gland” could have been stimulated by the same microenvironment changes as the tumor tissues. 6
A high variability in mRNA expression among samples was also found in tumor tissues. This reflects the tremendous cellular heterogeneity that is characteristic of mammary cancer. 54 In our study, we found a remarkable heterogeneity in YAP/TAZ protein expression by IHC across all samples, especially in TNBCs, CMTs, and FMTs. Human ER+ tumors showed a more homogeneous distribution. Nevertheless, TNBCs showed a higher protein expression of YAP/TAZ when compared to ER+ tumors, as found by others. 16 This suggests that, in TNBCs, which are highly aggressive, the Hippo pathway is deregulated. Also, although interspecies differences are difficult to interpret, FMTs showed a higher protein expression of YAP/TAZ than CMTs. Though this result may reflect that all FMTs analyzed in this study were grade III, whereas the CMTs were grade I and II, it is well known that FMTs are more aggressive than CMTs. 66,81
These data suggest a Hippo pathway deregulation in HBCs, CMTs, and FMTs. The higher level of YAP/TAZ in highly aggressive tumors (TNBCs and FMTs) suggests a highly tumorigenic role of these proteins, as we previously found in mammary tumors, 4 and as others also found in canine osteosarcoma. 43
In conclusion, in this study we found a deregulation of the Wnt/β-catenin and Hippo pathways in mammary tumors when compared to healthy tissues, which was more evident at the protein level. Gene expression analysis showed controversial results, which could be due to either a posttranscriptional regulation of the analyzed genes or more likely to the abundant presence of non-mammary tissues in the healthy samples. Finally, we found similarities across the 3 species, suggesting that dogs and cats could be considered as spontaneous models of human breast cancer.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
