Abstract
Equus caballus papillomavirus type 2 (EcPV-2) has been recognized as a potential cause of a subset of genital squamous cell carcinomas (SCCs) in horses. In the current study, we measured EcPV-2 seropositivity in 50 healthy horses from Western Canada, and these were compared to a herd of horses with known EcPV-2 exposure. Second, the presence of EcPV-2 DNA was measured using EcPV-2-specific PCR (polymerase chain reaction), performed on a variety of tissues collected at necropsy from 70 horses that lacked any history, gross, or histologic evidence of neoplasia or papillomavirus-associated disease. EcPV-2-specific RNA in situ hybridization (R-ISH) was performed on PCR-positive samples to identify the specific tissues infected. The prevalence of asymptomatic infection with EcPV-2 in Western Canadian horses was 20/70 (29%). Exposure to EcPV-2 as measured by seropositivity was 18/50 (36%). EcPV-2 positivity by anatomic location, as measured by R-ISH, was as follows: penis 10/29 (35%), vulva 5/34 (15%), eyelid 8/68 (12%), oral mucosa 7/65 (11%), skin from muzzle 7/68 (10%), and retropharyngeal lymph node 2/64 (3%). The youngest horses with EcPV-2 infection, based on PCR, were fetuses, suggesting for the first time that vertical transmission of EcPV-2 occurs in horses. The current study observed an increased prevalence of EcPV-2 as compared to previous studies. We suggest that this difference is due to our use of biopsies in place of superficial swabs. We propose that EcPV-2 infection in asymptomatic horses is more common than previously reported and that the virus’ role in equine genital SCCs may be more complex than originally thought.
Keywords
Papillomaviruses (PVs) are nonenveloped, double-stranded DNA viruses with a circular genome of approximately 8000 bp. 6 These viruses are ancient and generally highly host-specific and even tissue-specific. 23 Papillomaviruses are responsible for a subset of cancers in a variety of species. 23 Of these, human papillomaviruses (HPVs) are the best studied. Over 200 types of HPVs have been identified, of which only a small subset have known oncogenic abilities, the most notable of which are HPV types 16 and 18, associated with virtually all cases of human cervical cancers. 4,14,15,41 HPVs have also been implicated in a subset of ano-genital and head and neck cancers and possibly some melanocytic and breast cancers. 1,8,9,15,27,38,39 It is important to recognize that subclinical HPV infection is more common than clinical disease, with many infections being cleared and fewer becoming persistent. 6 Understanding how HPVs are transmitted, the molecular interactions between host and viral proteins, the identification of at-risk populations and development of screening protocols have all contributed to early detection and use of preventive measures such as vaccination. 41
In contrast to HPV, our understanding of PVs infecting horses is limited. Recently, a subset of equine genital SCCs and precancerous lesions (papillomas and plaques) have been associated with the presence of Equus caballus papillomavirus type 2 (EcPV-2) DNA, 3,11,20,29,35,42 and it is now generally accepted that this virus plays an active role in the development of these tumors. However, little is definitively known regarding the mechanism of oncogenicity, route(s) of transmission, existence of a carrier state, or impact of PV status on prognosis or therapeutic protocols. Given the anatomic distribution of EcPV-2-positive SCCs, it has been proposed that this virus is transmitted by sexual contact, although other possible routes such as direct contact and insects have been contemplated. 3,17,18,29
To date, 5 studies 3,11,16,17,35 have measured the prevalence of exposure or infection of EcPV-2 within apparently healthy horse populations. The purpose of the current study was 2-fold. First, to evaluate the prevalence of EcPV-2 seropositivity among clinically normal horses from across Western Canada, using the same methods as previously described by Fischer et al. 11 These findings were compared to those of a herd of horses with known exposure to a gelding with an EcPV-2-positive penile SCC. The second objective was to determine the frequency of EcPV-2 infection in asymptomatic horses by sampling several strategically selected sites from necropsied horses that lacked any known history or gross evidence of neoplasia or PV-associated disease (ie, lacking papillomas, aural plaques, sarcoids, SCCs).
The prevalence of EcPV-2 infection in normal horse tissues is crucial information when discussing the potential role of EcPV-2 as a risk factor in SCC development. We discussed and compared our findings to previously published reports of the prevalence of EcPV-2 positivity of equine SCCs and samples collected from healthy horses from similar anatomical sites.
Materials and Methods
Selection of Horses for EcPV-2 Polymerase Chain Reaction (PCR)
Seventy horses submitted to Prairie Diagnostic Services (Saskatoon, Saskatchewan) for necropsy or disposal during the period of October 2016 to June 2017 were selected for collection of tissues. Inclusion criteria allowed for any horse lacking a history of, or lesions at necropsy, indicative of any kind of neoplasia or PV-associated disease, such as papillomas, aural plaques, sarcoids, or SCCs. For the purposes of this study, these horses were termed “normal,” although many had unrelated diseases. No restrictions were placed on age, breed, or sex. Tissues were collected from the following sites: haired skin from the middle of the upper lip (muzzle), lower eyelid, buccal mucosa at the level of the second premolar, retropharyngeal lymph node, and either vulvar labia or the glans of the penis. In addition to these tissues, placenta was collected, whenever available, from fetal submissions. For each sample, a portion was retained frozen at −20 °C for future studies and a second portion was fixed with 10% buffered formalin. For each animal sampled, a single formalin-fixed, paraffin-embedded (FFPE) block was produced which included tissues from all the sampled sites. These FFPE blocks constituted the material for both EcPV-2-specific conventional PCR and chromogenic RNA in situ hybridization (R-ISH).
EcPV-2 PCR
In brief, 4 sequential 25 µm shavings were taken from each of the FFPE blocks and deparaffinized using sequential xylene washes. DNA extraction was performed as described previously. 26 Overnight incubation in lysis buffer and proteinase K (5 μL) was followed by extraction of nucleic acids using phenol-chloroform-isoamyl alcohol. Nucleic acids were precipitated out using 2.0 volumes of 100% ethanol and 1/10 volume of 3 M sodium acetate. Samples were resuspended in 50 µL of TE buffer and nucleic acid concentrations were measured using Nano Drop spectrophotometer. The presence of amplifiable DNA was confirmed by performing PCR using primers for the equine housekeeping gene beta-actin as previously described. 40
Consensus primers were designed for amplification of a 306 bp segment of the L1 gene of EcPV-2 based on alignment of the EcPV-2 isolates (HM461973.1 and EU503122.1). Sequences were as follows: EcPV-2For 5′-TCCTCCACCAATTTTAAA-ACCTAT-3′ and EcPV-2Rev 5′-ATCCAAGTCAAGGGAAAG-3′. The DNA was amplified in a standard PCR reaction using the Eppendorf Mastercycler using a 50 µL PCR reaction. The PCR assay was optimized using 2 µL of DNA template and end concentrations of 4 mM MgCl2, 0.25 dNTPs, 1.25U of Taq polymerase, 0.8 μM of each reverse and forward EcPV-2 L1 primers. The following cycling conditions were used: 3 minutes denaturation step at 94 °C, 40 cycles of amplification employing 94 °C for 30 seconds, annealing at 58.4 °C for 30 seconds and extension at 72 °C for 1 minute. A final extension step was used at 72 °C for 10 minutes, then held at 10 °C. The amplification products were subjected to 1.2% TE-gel electrophoresis and visualized by ethidium bromide staining. A sample was considered positive for EcPV-2 if a band was visible at the anticipated 300 bp size.
To confirm that the newly designed EcPV-2 L1 primers were amplifying EcPV-2 DNA, known positive EcPV-2 samples were amplified using the new EcPV-2-specific primers and the amplicons were sent to Macrogen (Seoul, South Korea) for sequencing. The known positive EcPV-2 samples were FFPE biopsies of SCCs used in a separate study whereby broad spectrum MY09/11 PCR and sequencing had identified them as EcPV-2-positive. These samples were assembled using the Staden Package software program and had 99% to 100% sequence identity to HM461973.1 and EU503122.1 based on the alignment over 424 bp in National Center for Biotechnology Information database.
Internal controls included block blanks, extraction blanks, and nontemplate controls. A previously sequenced EcPV-2 sample from an equine penile SCC was used as positive control. A known EcPV-2-negative equine tissue was used as a negative control. A sample was considered EcPV-2-positive if a band was visible under UV light on the gel at the expected size and position, consistent with the positive control.
EcPV-2 RNA In Situ Hybridization (R-ISH)
All FFPE blocks that were EcPV-2 PCR-positive were submitted for EcPV-2 R-ISH to both identify the specific tissues responsible for positivity and confirm PCR-positive results. The methods used were similar to that previously described by Zhu et al in 2015. 42 Reagents used for the RNAscope assay included the V-EcPV-2 probe that targets the E6 and E7 genes, the 2.5 HD Detection Reagent Red, H2O2, Protease Plus Reagents, Target Retrieval Reagents, and Wash Buffer Reagents (RNAscope, Advanced Cell Diagnostics, Hayward, CA). In brief, tissues were placed in the target retrieval solution for 15 minutes at 98 °C and apart from reducing the hematoxylin counterstain from 1:2 to 1:5 and allowing air drying of slides for 18 hours prior to cover-slipping, manufacturer instructions were followed. Control tissues (see Suppl. Figs. S1–S3) included equine SCCs that were (1) known PV-positive but EcPV-2-negative (EcPV-1), (2) PV-negative, and (3) EcPV-2-positive. Control tissues had been subjected to previous broad-spectrum PV PCR (MY09/11 and FAP 59/64) or EcPV-2 L1 PCR, followed by sequencing.
EcPV-2 E6/E7 R-ISH probe hybridization (hereafter called “signal”) was visualized by routine light microscopy. Slides were examined at 40× magnification, and the presence and location (cell type) of signal was recorded for each of the 5 tissue types on the slide. Based on previous work (unpublished) and similar publications, 10,12,42 signal distribution pattern was categorized as being either granular or diffuse nuclear. A granular signal pattern was typically seen within the nucleus and cytoplasm of epithelial cells and was characterized by fine, granular, or punctate signal. A diffuse nuclear signal pattern was characterized by intense, complete filling of the nucleus by signal, which obscured nuclear detail and was visible at low (4× or 10× objective) magnification.
Selection of Horses for Serology
Fifty equine serum samples were selected over a 2-week period from submissions to Prairie Diagnostic Services for equine infectious anemia (EIA) testing. Only horses designated as healthy and requiring testing for export, sale, or showing purposes and whose EIA results were negative were included. This group of samples was referred to as the “general population.” From these, sera were randomly selected from stratified groups such that a broad range of ages and geographical distribution was represented. Groups were stratified geographically by the Western Canadian province where the horse resided at the time of sampling: British Columbia, Alberta, Saskatchewan, Manitoba. Age was stratified as either “young” (10 years and younger) or “old” (11 years and older).
In addition, serum was collected from a herd of 17 horses which included one 18-year-old gelding with a known EcPV-2-positive penile SCC, and these samples constituted what is referred to as the “known exposure population.” The affected gelding had been born into the herd, the penile tumor had been noticed 1 year prior to the study, and had been treated surgically. Full-thickness biopsies and swabs from this horse were subjected to 2 different EcPV-2 L1 PCR reactions that were part of a separate, unpublished, study. EcPV-2 PCR amplicons were sent for sequencing and identification (University Core DNA Services at the University of Calgary, Alberta, Canada). Additionally, EcPV-2 (E6/E7) R-ISH was performed, as described in the current study, and were also positive.
EcPV-2 Serology
EcPV-2 serology was performed as previously described. 11 To test horse sera for the presence of virus-specific antibodies, a glutathione-S-transferase-capture ELISA with EcPV-2 L1 and CPV1 protein was performed. The EcPV-2 plasmid produced from Fischer et al 11 and the CPV1 plasmid from Lange et al 19 were used to heterologously express the proteins in Escherichia coli strain BL21 (DE3) as described previously 31 with minor modifications including cell lysis by sonification instead of using a high-pressure homogenizer.
Polysorb 96-well plates were coated at 4 °C overnight with 50 mM sodium carbonate buffer, pH 9.6, containing 0.1% glutathione casein. Plates were washed 3 times with phosphate-buffered saline (PBS) buffer supplemented with 0.3% Tween 20 (PBS-T). These washing steps were applied between every incubation step. Plates were blocked at 37 °C for 1 hour with casein buffer (PBS-T containing 0.2% casein). The GST-tagged antigens (EcPV-2 and CPV1), diluted 1:10 in casein buffer, were applied to the plates and incubated at 37 °C for 1 hour.
The horse sera were diluted 1:500 in casein buffer, mixed with an equivalent of lysed untransformed E coli strain BL21 (DE3) and incubated at 4 °C for 1 hour to block reactions with contaminating bacterial proteins. 31 The coated ELISA plates were then incubated with the diluted preincubated sera or, for evaluation purposes as plate control, with goat anti-GST antibody diluted 1:1000 in casein buffer. As a secondary antibody, goat anti-equine IgG conjugated to horseradish peroxidase (HRP) or HRP-conjugated anti-goat antibody was used for the plate controls; both were diluted 1:1000 in casein buffer and applied for 1 hour at 37 °C. Following 6 final washes, substrate (78 mM CH3COOH, 24 mM CH3COONa, 50 mM NaH2PO4, 2 mM ABTS with 1.25 mM H2O2 applied shortly before use) was added. The absorbance was measured after 45 minutes at 405 nm in a Sunrise microplate reader.
The horse sera were tested in triplicates against the antigen EcPV-2 L1-GST and, as a negative control, against CPV1 L1-GST. On each plate, one triplicate per antigen was tested with GST antibodies as a plate antigen coating control. In order to normalize the results of the different plates, the same positive and negative control sera were included on every plate.
Serum samples were tested in triplicates in ELISA. Plate to plate variability was compensated by dividing every value by the mean of the control sera values from the corresponding plate and multiplying the result by the mean of all control sera from all plates.
Bootstrapping with thousand replicas was applied on the plate-normalized values of the negative controls to calculate the mean and the 97.5% confidence interval (CI). The cutoff value was set to the upper CI limit. Means of the plate-normalized values including the cutoff value were plotted using R (Free Software Foundation; Boston, MA).
Demographic Categorization
Horses were categorized by age as either “young” (10 years and younger) or “old” (11 years and older), using submission information. Breed information was extracted from the histopathology submission forms and used as a surrogate marker to categorize horses by expected patchiness of hair and skin pigmentation. Breeds typically displaying depigmented skin areas included Appaloosa, American Paint, and Pinto. Breeds lacking this feature included Quarter Horse, Draft, Thoroughbred, Arabian, and Warmblood.
Statistical Analysis
Microsoft Excel (2016) software was used for descriptive statistical analytical measurements such as mean and median ages and proportion of positivity. Openepi online software was used to calculate χ2 values (95% CI, mid-p exact, 2 tail) when determining significance of differences in EcPV-2 positivity by age and breed categories in the current study and for comparison by anatomic location to previous publications. Significance was set as P < .05.
Results
EcPV-2 PCR (Necropsy)
Seventy equids were sampled postmortem, including 35 mares, 21 geldings, 9 stallions, and 5 horses where sex was not recorded. Ages ranged from 0 (fetus) to 30 years. The average age was 9 years and 11 months, and the median age was 7 years. A variety of breeds were represented, the most common of which were Quarter Horses and their crosses (32/70; 46%). Other breeds included Warmbloods (Hanoverian, Canadian, Foreign; 5/70; 7%), Appaloosa (4/70; 6%), Thoroughbred (4/70; 6%), Draft breeds (ex. Percheron, Clydesdale, Belgian; 4/70; 6%), Miniature Horse (4/70; 6%), American Paint (3/70; 4%), Arabian (3/70; 4%), Pony (2/70; 3%), and one each (1/70; 1%) of Peruvian Paso, Saddlebred, and Mule. Five horses were of unknown breeds (5/70; 7%).
Reasons for euthanasia or death included lameness, advanced age, trauma, colic, dystocia, developmental heart defect, chronic airway disease, sepsis, suppurative arthritis, ionophore toxicity, and research (including studies on septic joints and intra-abdominal adhesions).
All 70 of the FFPE blocks prepared for these horses had amplifiable DNA as determined by a positive PCR reaction using primers against the housekeeping gene, beta-actin. Twenty of the 70 horses (29%) yielded a positive result on EcPV-2-specific PCR performed on FFPE blocks of combined tissues (see Suppl. Fig. S4). The youngest horses for which an EcPV-2-positive PCR result was obtained included 2 near-term fetuses and 2 one-day-old foals. One of the fetuses was still in utero and belonged to a dam who was also necropsied (euthanized due to dystocia); the dam’s tissues were negative for EcPV-2.
When comparing EcPV-2 positivity by coat pigmentation (as indicated by breed information where available), there was no difference between breeds with patchy (2/9; 2%) versus solid (17/56; 30%) colored skin (P = .67). Similarly, there was no significant difference in EcPV-2 positivity when comparing younger horses (10 years old and younger; 8/38; 21%) versus older horses (11 years and older; 10/27; 37%; P = .173).
EcPV-2 R-ISH (Necropsy)
All of the FFPE blocks that were positive on EcPV-2 PCR were submitted for chromogenic EcPV-2-specific RNA in situ hybridization to identify the tissue source of the PCR positivity (Figs. 1–21). Of the horses deemed EcPV-2-positive by PCR, 17/20 (85%) showed EcPV-2 positivity by R-ISH in at least one tissue. The remaining 3/10 (15%) samples identified as EcPV-2-positive on PCR were negative on EcPV-2 R-ISH.
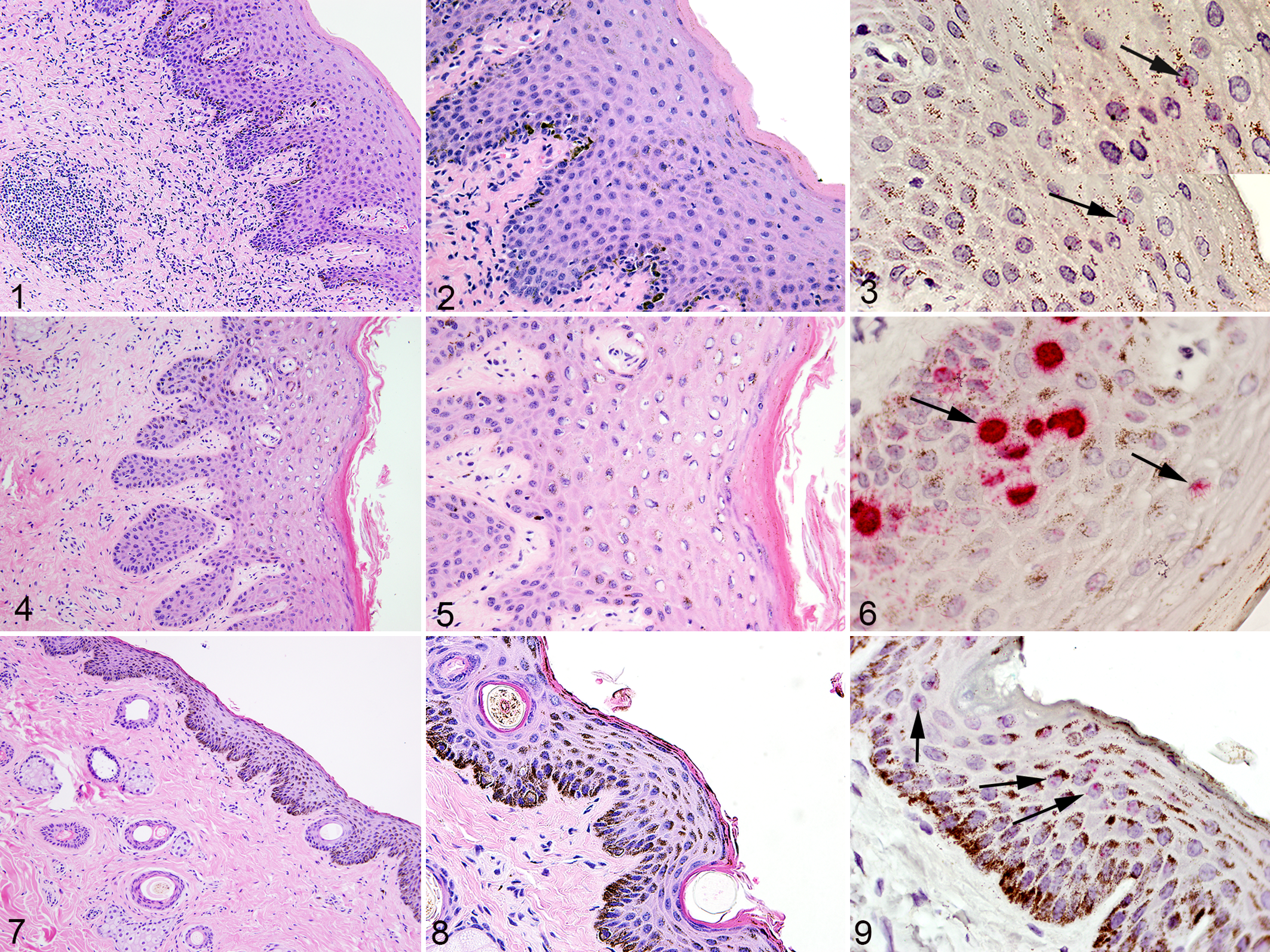
Normal tissues with Equus caballus papillomavirus type-2 (EcPV-2) infection (positive by L1 PCR), horse. Figs. 1, 2, 4, 5, 7, 8: Hematoxylin-eosin. Figs. 3, 6, 9: RNA in situ hybridization (R-ISH) for EcPV-2 E6/E7 (red labeling, arrows).
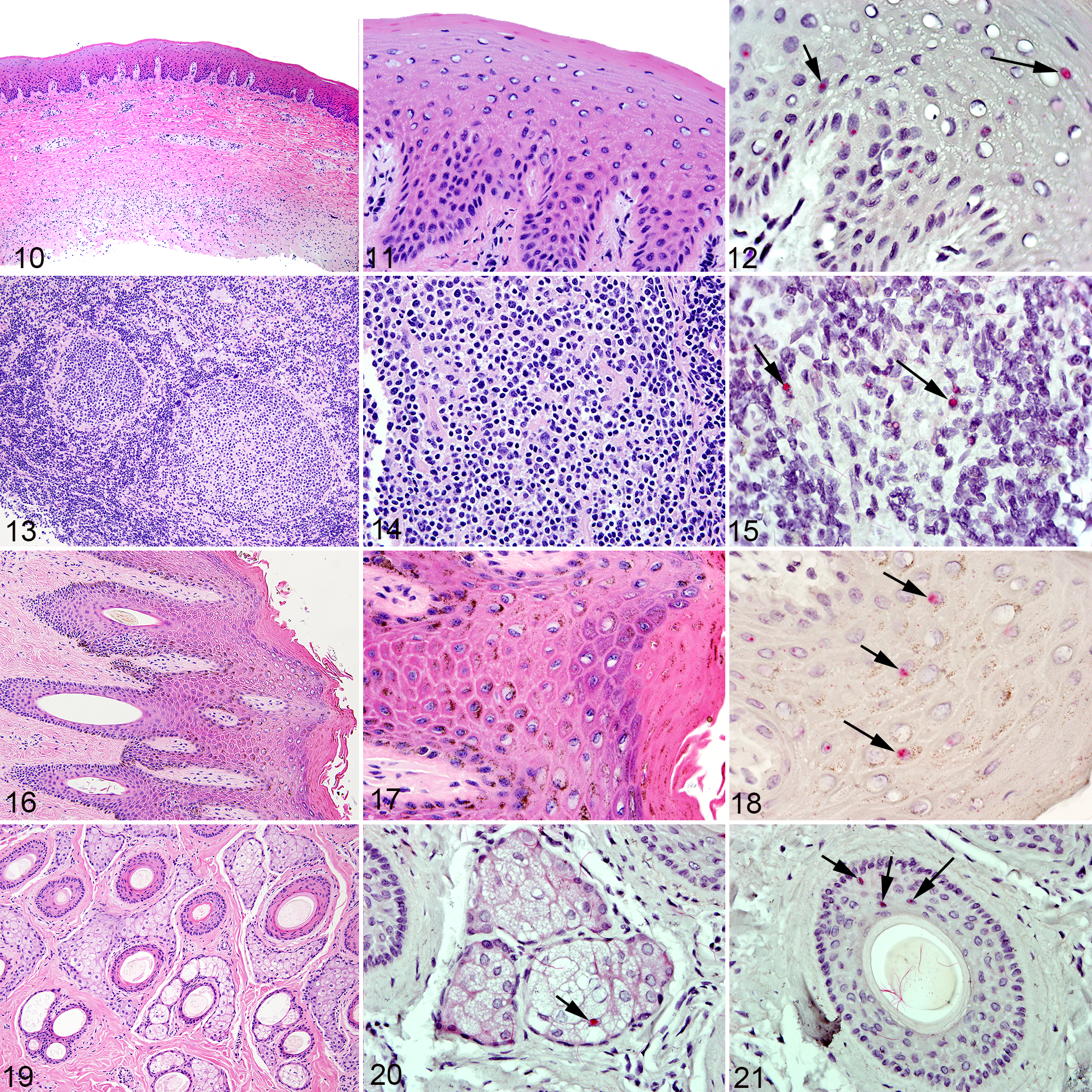
Normal tissues with Equus caballus papillomavirus type-2 (EcPV-2) infection (positive by L1 PCR), horse. Figs. 10, 11, 13, 14, 16, 17, and 19: Hematoxylin-eosin. Figs. 12, 15, 18, 20, and 21: EcPV-2(E6/E7) RNA in situ hybridization (R-ISH).
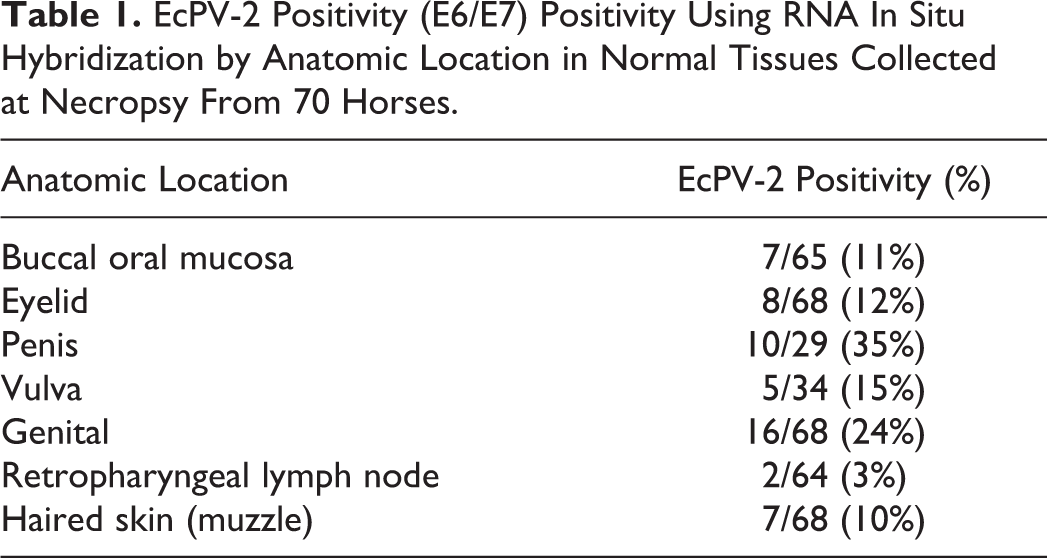
EcPV-2 R-ISH results were as follows (Table 1
EcPV-2 Positivity (E6/E7) Positivity Using RNA In Situ Hybridization by Anatomic Location in Normal Tissues Collected at Necropsy From 70 Horses.
Signal in all sections deemed positive for EcPV-2 by R-ISH was typically present within the nucleus of epithelial cells and was characterized by fine, granular, or punctate signal (Figs. 3, 9, 12, 18). A subset of sections also had intense, diffuse nuclear signal that obscured detail of the nucleus (Fig. 6). In an unrelated penile section with both types of signal (granular and diffuse nuclear; unpublished data), pretreatment with RNAse greatly attenuated granular signal but only mildly attenuated diffuse nuclear signals. Pretreatment with DNAse had no effect on granular signals but partially attenuated diffuse nuclear signals.
EcPV-2 Serology (General Population)
Serum samples were collected from 50 horses (general population) including 21 mares, 27 geldings, and 2 stallions, with a range in age from 1 to 33 years, an average age of 10 years and 10 months, and a median of 10 years. Represented breeds included Quarter Horse (22/50; 44%), Warmblood (7/50; 14%), Thoroughbred (6/50; 12%), American Paint (3/50; 6%), Draft and crosses (3/50; 6%), Arabian (2/50; 4%), and one each (1/50; 2%) of Grade, Miniature Horse, Morgan, Mule, and Welsh Cobb. Two submissions lacked breed information. Of the sampled horses, 27 were from the province of Alberta, 16 from Saskatchewan, and 7 from Manitoba. These 50 horses represented 40 different geographic locations at the time of sampling as indicated on the EIA submission (9 submissions each included 2 horses from the same farm, and 1 submission included 3 horses from the same farm).
Eighteen of the 50 (36.0%) horses had evidence of EcPV-2 seropositivity. The youngest horse showing seropositivity was 3 years old and the oldest was 16 years old.
When EcPV-2 seropositivity of the general population group was compared by age category, young horses (10 years or younger; 7/25; 28%) were similar (P = .26) to their older counterparts (11 years or older; 11/25; 44%). When EcPV-2 seropositivity of the general population group was compared by breed category there was no statistical difference (P = .16) between horses with patchy and solid skin pigmentation.
EcPV-2 Serology (Known Exposure Population)
Serum samples were collected from 16 horses in the “known exposure population,” including 6 mares and 10 geldings. Fourteen of the 16 horses had known ages and ranged from 5.5 to 24 years, with a mean of 14 years and 11 months, and a median of 15 years 6 months. Breeds included Quarter Horse (4/16; 25%), American Paint (3/16; 19%), Appaloosa (2/16; 13%), one of each (1/16; 6%) Thoroughbred, Pony, Arabian, Morgan-Welsh cross, and unknown (3/16; 19%). Six of the 16 (38%) asymptomatic horses known to be in contact with the horse having the EcPV-2-positive penile SCC showed seropositivity to EcPV-2 L1 capsid protein. The horse with the EcPV-2-positive SCC also showed seropositivity. When comparing EcPV-2 seropositivity between the “general” (18/50; 36%) and “known exposure” (6/16; 38%) populations, there was no statistically significant difference (P = .91).
Discussion
The current study shows that EcPV-2 infection occurs among grossly normal horses in Western Canada (20/70; 29%) when assessed by EcPV-2 L1-specific PCR performed on multiple tissue samples taken from each horse. The prevalence of EcPV-2 infection (as measured by PCR) and exposure (as measured by seropositivity) was similar in horses of different age categories and skin pigmentation patterns.
Seventeen of 20 samples (85%) that were positive for EcPV-2 DNA using PCR were also found to be positive when using R-ISH. Similar findings of EcPV-2 PCR positivity but R-ISH negativity have been previously reported in equine genital SCCs. 42
When using the same EcPV-2 R-ISH procedure on FFPE samples of equine SCCs, from unpublished studies, we found no false positives (0/5) by EcPV-2 R-ISH on samples deemed PV-negative by PCR. Similarly, no false positives were identified when EcPV-2 R-ISH was performed on non-EcPV-2, PV-positive samples (0/8). However, we did find disagreement whereby EcPV-2 nucleic acids detected by PCR were not detected by R-ISH and this was mostly limited to periocular SCCs (7/10; 70%). Of the remaining 3/10 (30%) of periocular SCCs that were positive on both EcPV-2 PCR and R-ISH, the R-ISH signal was significantly weaker and less frequent as compared to other tumors evaluated, such as EcPV-2-positive penile SCCs.
Given the above, we propose that the discrepancy is best explained by R-ISH being less sensitive at detecting EcPV-2 nucleic acids as compared to PCR given that only the latter amplifies the targeted nucleic acid. The presence of multiple PCR controls in the current study makes contamination a less likely explanation for why 3 of the 20 samples in the current sample were positive on EcPV-2 PCR but not R-ISH. Such differences in sensitivity should be considered particularly when evaluating tissues which may have low viral loads, as is suspected in asymptomatic infections as seen in the current study. Additionally, the starting material for PCR required four 25-μm-thick shavings be collected, whereas for an R-ISH slide, only 4-µm thickness is analyzed. It is possible that EcPV-2 nucleic acids are not uniformly distributed within tissues sampled; differences in tissue volumes sampled between the 2 methods could result in disagreement between PCR and ISH results from the same tissue sample.
In the current study, 3/7 fetuses (43%) tested positive for EcPV-2 DNA by PCR. EcPV-2 RNA-ISH identified the positive tissues as follows: genital (3/7; 43%), oral mucosa (1/7; 14%), eyelid (3/7; 43%) and skin from muzzle (1/7; 14%), placenta (0/5). Equine fetal infection with EcPV-2, although not previously documented to the authors’ knowledge, is not surprising given that vertical transmission of HPV and BPV has been documented. 32,33 Interestingly, in one submission, both the fetus and corresponding mare were available for sampling as euthanasia was due to dystocia; the fetus was positive for EcPV-2, while the mare was negative. This finding suggests that vertical transmission of EcPV-2 might not result from simple passage through an infected birth canal or through nursing. Instead, the finding of an EcPV-2-positive fetus from an EcPV-2-negative mare might suggest either that the mare had been infected and cleared the virus, that the mare was infected but not in the tissues sampled, or that EcPV-2 came from the stallion. Although a single previous study evaluating the presence of EcPV-2 nucleic acids in healthy equine semen did not identify the virus, 35 PV nucleic acids have been identified in peripheral blood and semen of both healthy cattle and those with bladder tumors. 22,28,33 Identification of in utero infection with EcPV-2 also probes the possibility of immunotolerance in some animals and might explain the findings in a previous study of a subset of horses that were infected but lacked evidence of seropositivity. 11
Our finding of 20/70 (29%) overall EcPV-2 PCR positivity among “normal” horses is significantly higher (P < .001) than that reported in previous primary studies 3,11,16,35 and in a review of published studies (20/315; 6.3%; Table 1). 34 However, when we examined EcPV-2 positivity by anatomic location, using EcPV-2 R-ISH performed on the EcPV-2 PCR-positive FFPE blocks, statistically significant differences between the current study and previous ones were limited to penile and ocular sites, as summarized below and in Table 2.
Prevalence of Positive Tests for EcPV-2 in Tissues From Horses in the Current Study and Previous Studies.
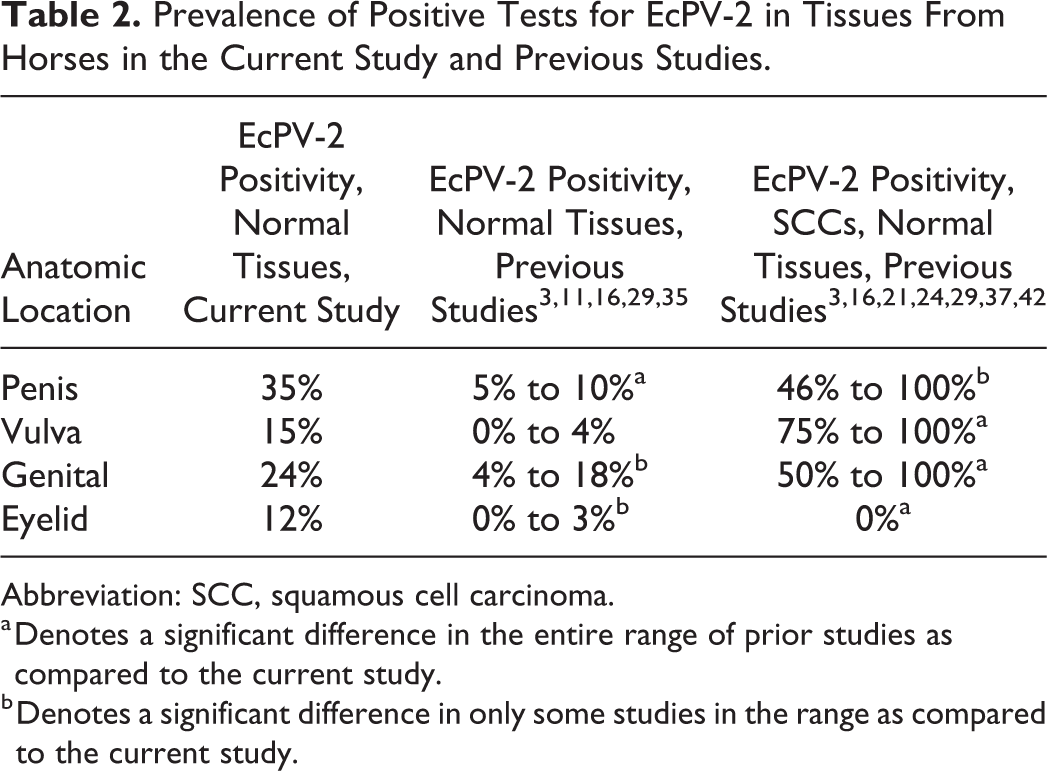
Abbreviation: SCC, squamous cell carcinoma.
a Denotes a significant difference in the entire range of prior studies as compared to the current study.
b Denotes a significant difference in only some studies in the range as compared to the current study.
EcPV-2 positivity in the current study of normal penile tissue samples was 10/29 (35%), which is significantly higher compared to 3 prior reports 3,16,35 using penile urethral fossa swabs in which the prevalence ranged from 5% 35 (P < .001) to 10% 3 (P = .01). EcPV-2 positivity in the current study of vulvar tissue samples was 5/34 (15%) and was not statistically different than the 3 prior reports 3,16,35 using vulvar swabs. EcPV-2 positivity in the current study of genital tissue samples was 16/68 (24%), which was higher than one report (4.17%; P ≤ .01), 16 which used swabs, but similar (18.0%; P = .48) 11 to another report which used cytobrush to obtain DNA.
EcPV-2 positivity in the current study of eyelid tissue samples was 8/68 (12%), which is significantly higher than a study performed on sampled nictitans membrane (0.0%; P < .01) 16 but similar to another study using ocular swabs (3.3%; P = .2). 35 EcPV-2 positivity in the current study of tissue samples taken from the oral buccal mucosa was 7/65 (11%), which was similar to the 2.7% 16 (P = .06) identified in a prior study that used oropharyngeal tissue samples.
A notable difference between the current study and earlier publications is that we compared EcPV-2 positivity by PCR and R-ISH between tissue samples from “normal” horses whereas other studies collected DNA using swabs, cytobrush, or body fluids of live horses free of neoplasia. The exception was one study from New Zealand which harvested the third eyelid from abattoir horses. 16 We propose that in asymptomatic EcPV-2 infection in horses, viral copy numbers might be very low and restricted to the deeper layers of the stratified epithelium, consistent with what is known of HPV infection, 6,13 and therefore unlikely to be detected by superficial swabbing. Conversely, swabs may be effective for detecting horses with productive infections (shedders).
Second, the methodology of PCR varies somewhat between studies, both in the types and numbers of primers used and whether conventional versus quantitative PCR was employed. Finally, prior studies have varied in the number of sites sampled per horse. In the current study, each necropsied horse had 5 tissues examined whereas the prior studies referenced above sampled only genital sites with or without periocular or oral sites. Increasing the number of mucocutaneous sites sampled per animal will increase the likelihood of attaining a positive result in at least one site, thus increasing the estimated prevalence of EcPV-2 infection.
EcPV-2 has been proposed to play a causal role in a subset of genital SCCs based on detecting the virus within SCCs but rarely in swabs from healthy horses. As discussed above, testing superficial swabs may not adequately detect low copy number infections present within deeper layers of epithelium, which is likely what is occurring in asymptomatic EcPV-2 infection in horses. When comparing the prevalence of EcPV-2 infection of normal horse tissues in the current study to that of SCCs from similar anatomic locations in prior publications (limited to genital, periocular, and oral-pharyngeal sites), significant differences were detected in some cases but not all, as discussed below and summarized in Table 2.
Prior studies of EcPV-2 positivity of penile SCCs (and in one study, both penile and preputial SCC) reported a range from 46% to 100%. 3,16,20,24,29,30,36,42 In the current study, EcPV-2 positivity of normal penile skin was 10/29 (35%), which was similar to the lower range of previous reports of 43.0% 24 (P = .45) but significantly lower than the upper end of the range (P < .001). EcPV-2 positivity of vulvar SCCs in previous studies ranged from 75.0% to 100%, 3,29,35 which is significantly greater than the current study of normal vulvar tissue (5/34; 15%; P = .02 and .03, respectively). However, sample sizes in the previous vulvar SCC studies were small (n < 5) and further studies on vulvar SCC EcPV-2 positivity are warranted to confirm the significance of our comparison. When comparing EcPV-2 positivity of the current study on normal genital tissues to prior publications (which evaluated both vulvar and penile SCCs), the current study reported 16/68 (24%), which was significantly lower than the previously reported range of 50% to 100% 3,29,35 (P = .04, <.001, and .0014, respectively).
Interestingly, EcPV-2 positivity of ocular and periocular SCCs has previously been reported as 0%, 24,29 whereas normal eyelid tissue samples from horses in the current study were significantly higher at 8/68 (12%; P = .018 and .03). Possible explanations for the higher prevalence of EcPV-2 infection in normal eyelid tissues from the current study as compared to those with SCCs could be (1) periocular sites are commonly infected and readily cleared by the immune system (decreased tropism); (2) other carcinogens, such as UV radiation, play a more important role in SCC development at this site; or (3) excessive UV exposure to EcPV-2 infected periocular skin may result in clearance of the virus through mechanisms yet to be described.
EcPV-2 positivity of oral-pharyngeal SCCs has been reported as 15.0%, 16 which was similar (P = .6) to the current study on normal oral mucosa (7/65; 11%).
The use of RNA-ISH for microscopic localization of E6/E7 nucleic acids highlighted the presence of EcPV-2 mostly within epithelial cells but also in occasional lymphocytes within the retropharyngeal lymph nodes. This is perhaps not surprising as prior research on BPVs and HPVs have shown lymphocyte infection and it has been proposed that this may even represent a potential source of latency for the virus. 2,5,7,22,25, 28,33 EcPV-2 nucleic acids have been identified within lymph nodes of horses with regional metastasis of SCC 36,42 but to the authors’ knowledge, not in any cases of lymphosarcoma.
To the authors’ knowledge, there are no published studies pertaining to EcPV-2 prevalence in equine body sites other than genital, oral-pharyngeal or periocular, although in the current study, we demonstrated that 7/68 (10%) of haired skin from the muzzle contained EcPV-2 nucleic acid. This site was selected for analysis because of equids’ use of their upper lip to contact other horses (nuzzling, grooming), agitate surfaces, and explore interesting textures or smells.
The current study shows that EcPV-2 exposure, as measured by seropositivity, is not uncommon (18/50; 36%) in this Western Canadian horse population. The youngest age of horse that had seroconverted was 3 years old. When EcPV-2 seropositivity of the general population group was compared by age category, there was no significant difference (P = .26) between young and old horses, also suggesting that EcPV-2 exposure likely occurs early in life.
Conclusion
The current study suggests that infection with EcPV-2 is not uncommon in asymptomatic Western Canadian horses and can occur as early as in utero. The presence of EcPV-2 infection in a variety of tissues suggests that transmission may not be solely via sexual contact and that other routes should be considered. We reported a similar level of seropositivity as compared to previous reports 11,30 but observed an increased proportion of positivity of EcPV-2 DNA in normal horse tissues as compared to some previous studies. We believe that this is the result of differences in sampling methodologies and that tissue biopsy is superior, as compared to surface swabbing or cytobrush, for detecting asymptomatic infection. Based on our observation that EcPV-2 DNA can be detected frequently in normal tissues, we propose that a subset of equine mucocutaneous SCCs may be influenced by EcPV-2 infection, but additional host and environmental factors are also likely necessary. Further research seems warranted, particularly if preventative measures such as vaccination are to be applied in a meaningful manner.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Greenwood_et_al - Prevalence of Equus caballus Papillomavirus Type-2 Infection and Seropositivity in Asymptomatic Western Canadian Horses
Supplemental Material, Combined_supplemental_materials-Greenwood_et_al for Prevalence of Equus caballus Papillomavirus Type-2 Infection and Seropositivity in Asymptomatic Western Canadian Horses by Sarah Greenwood, Betty Chow-Lockerbie, Sophie Ramsauer, Garrett Wachoski-Dark, Cameron Knight and Bruce Wobeser in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank the following people and organizations: Prairie Diagnostic Services Inc for providing serum samples, in particular Victoria Maye and the postmortem room technicians for their assistance in collecting biopsies and veterinary students Jane Westendorf, Peter Watson, and Christina Mackessey for their assistance with PCR.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the Townsend Equine Research Health Fund, Western College of Veterinary Medicine, University of Saskatchewan.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
