Abstract
Fifty years ago, inoculation with bovine papillomavirus (BPV) was found to cause mesenchymal tumors of the skin in cattle and horses, as well as tumors of the bladder in cattle. Subsequent to these studies of BPVs, human papillomaviruses (HPVs) were found to cause cervical cancer resulting in intense research into papillomaviruses. During the past 50 years, the ways that HPVs and BPVs cause disease have been investigated, and both HPVs and BPVs have been associated with an increasingly diverse range of diseases. Herein, the biology, oncogenic mechanisms, and diseases associated with BPVs are compared with those of HPVs. As reviewed, there are currently significant differences between BPVs and HPVs. However, research 50 years ago into BPVs formed a prologue for the recognition that papillomaviruses have a significant role in human disease, and it is possible that future research may similarly reveal that BPVs are less different from HPVs than is currently recognized.
Keywords
In 1964, Veterinary Pathology published the observations of Cheville and Olson 30 that inoculation of cattle with bovine wart tissue resulted in epithelial hyperplasia when administered by scarification and fibroblastic proliferation when administered by subcutaneous administration. This timely study added to earlier observations that inoculation with bovine wart tissue also induced regressing bladder tumors in cattle 86 and sarcoma-like lesions in the dermis of horses (Table 1). 83 While an association between papillomaviruses (PVs) and skin cancer in rabbits was reported in 1935, 104 at the time that bovine papillomaviruses (BPVs) were being shown to cause a diverse range of hyperplastic and neoplastic diseases in animals, human papillomaviruses (HPVs) were generally thought to only cause regressing papillomas (warts) of the skin or mucous membranes.
Observations on Bovine Papillomaviruses (BPVs) Made Prior to or in the Paper by Cheville and Olson, Published in Veterinary Pathology in 1964, and the Subsequently Determined Relevance to Human Papillomavirus (HPV) Biology.

In the 50 years since Cheville and Olson’s study, interest in PVs has exploded following the demonstration by zur Hausen et al 129 in 1981 that most human cervical cancers are caused by HPVs. In addition to cervical cancer, PVs are now thought to cause a significant proportion of oropharyngeal, anal, and penile neoplasms, and PV infection is considered responsible for 5% of all human cancers. 91 During the past 50 years, studies of BPV-associated lesions have been useful in helping to understand the complex relationship between HPVs and human cancer. In addition, unlike HPVs, BPVs were found to readily transform cells in vitro, and BPV-transformed cells have been valuable in determining how PVs cause cancer. 69 Research using BPVs has made a significant contribution to our understanding of PV-induced diseases and to the subsequent development of protective HPV vaccines.
Herein the biology and the diseases that are associated with HPVs and BPVs are reviewed and compared. The major oncogenic mechanisms of PVs are also briefly reviewed. Current research indicates that significant differences exist between HPVs and BPVs (summarized in Table 2). However, it is interesting to speculate that, as with the initial BPV studies 50 years ago, it may subsequently be discovered that HPVs are less different from BPVs than is currently recognized.
Comparison of Human and Bovine Papillomaviruses.
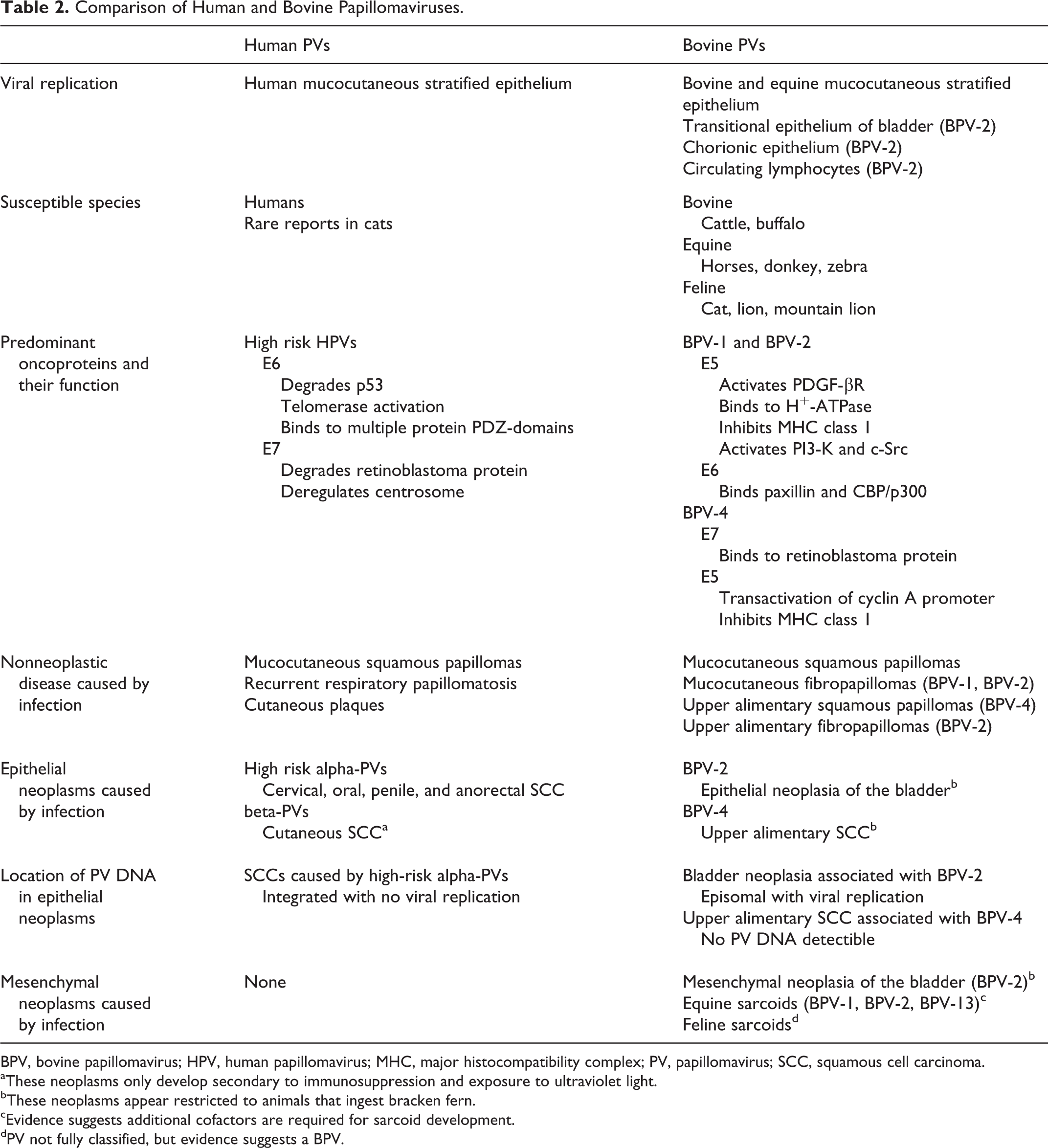
BPV, bovine papillomavirus; HPV, human papillomavirus; MHC, major histocompatibility complex; PV, papillomavirus; SCC, squamous cell carcinoma.
aThese neoplasms only develop secondary to immunosuppression and exposure to ultraviolet light.
bThese neoplasms appear restricted to animals that ingest bracken fern.
cEvidence suggests additional cofactors are required for sarcoid development.
dPV not fully classified, but evidence suggests a BPV.
Classification of Papillomaviruses
Papillomaviruses are double-stranded circular DNA viruses. Their genome consists of around 8000 base pairs that includes 5 or 6 early (E) open reading frames (ORFs) and 2 late (L) ORFs. 9 The highly conserved L1 ORF is used to classify PVs, and a PV is considered a different type if its L1 ORF contains less than 90% similarity with another PV. 9 PV types are grouped into genera with members of the genus containing greater than 60% L1 ORF similarity. 9 PVs within a genus often demonstrate similar host, location, and behavioral characteristics. Currently, over 280 PV types have been defined in 35 genera. 33,98
Around 170 HPV types have been classified within 5 genera: alpha-, beta-, gamma-, mu-, and nu-PV. In contrast, only 13 BPVs have been classified into 4 genera: xi- (n = 7), delta- (n = 3), epsilon- (n = 2), or dyoxi- (n = 1) PV. 98 This most likely reflects the fact that human PVs have been extensively studied compared with animal PVs, and it appears likely that many animal PV types have not yet been detected. Most PVs are highly host specific, and reports of cross-species infections by HPVs are rare. 71 However, the bovine delta-PVs are an exception and are well recognized to infect multiple species.
Biology of Papillomaviruses
Human PV Biology
It is currently generally believed that HPV infections are limited to mucocutaneous stratified epithelium with the viral life cycle dependent on, and intimately coordinated with, epithelial cell replication and differentiation. 38,39 Infection starts when the PV gains access to a basal cell, probably due to microtrauma. 108 The virus is transported to the nucleus, and expression of E1 and E2 results in the production of 10 to 200 episomal PV copies. 66 Basal cell replication maintains the infection; however, terminal differentiation and keratinization of an infected cell is required for a productive infection. 37 Differentiation triggers the production of the PV early proteins, which prevent the suprabasal cell from leaving the cell cycle (which would result in nuclear degeneration) and instead causes the cell to reenter the S-phase of the cell cycle and produce numerous viral copies. This continuation of the cell cycle is predominantly driven by the PV E6 and E7 proteins. 39 As the infected cell reaches the upper epithelium, the PV late genes are expressed and viral assembly occurs close to the cell surface. PVs do not cause cell lysis, and viral particles are only released after the epithelial cell has been sloughed from the epithelial surface and degraded. 37
Bovine PV Biology
The basic biology of most BPV infections is probably quite similar to HPVs with microtrauma, resulting in a productive infection of cutaneous or mucosal epithelium. An important difference in the delta-BPVs is the predominance of the E5 protein and the lesser roles of E6 and E7 proteins in disrupting the cell cycle during a productive infection. 12,111 Another significant difference is the recent observation that BPV-2 is able to infect and, as evidenced by L1 protein production, replicate in a wider range of tissues, including the transitional epithelium of the bladder 103 and the chorionic epithelium of the placenta. 100 BPV-2 L1 protein production has also been reported in circulating lymphocytes, suggesting viral replication in nonepithelial cells. 102 Confirmation that lymphocytes allow BPV-2 replication would indicate that the coordinated interplay between epithelial maturation and PV gene expression is not essential for PV replication and therefore fundamentally change the understanding of PV biology. Whether BPV-2 causes lysis of infected lymphocytes or has developed other methods to release the viral particles from the infected lymphocytes remains unknown. Possibly due to the presence of BPV-2 in lymphocytes, BPV-2 DNA can also be detected in a wide range of samples, including peripheral blood, 24,101 urine, semen, and milk. 62 In cattle, infection by BPV-2 via blood transfusion has been reported, and there is evidence that infection can be passed vertically to calves. 115 Current evidence suggests that most cattle are infected by BPV-2, and lymphocytes may provide a reservoir for infection. 102,115 DNA of BPV-1 and BPV-4 has also been detected in peripheral blood samples from clinically normal cattle or cattle with cutaneous papillomas. 5 In humans, while some evidence of blood-borne 29 and vertical 106 HPV transmission has been reported, neither has been definitively demonstrated.
Papillomaviruses are thought to be one of the oldest viral families and have co-evolved with their hosts over millions of years. 98 Therefore, it is not surprising that most PV infections in all species occur without the development of visible disease. Some PVs cause papillomas. This is thought to be due to greater promotion of epithelial cell cycling. In addition, due to their ability to interfere with normal cell cycle regulation, infection by some PVs predisposes to the development of neoplasia. 68
Papillomaviruses and Hyperplastic Lesions
Hyperplastic Human Diseases Caused by PVs
Hyperplastic lesions that are caused by PV infection in people include papillomas (warts), cutaneous plaques, and recurrent respiratory papillomatosis. Cutaneous papillomas can be caused by many different PV types, including low-risk alpha-, mu-, or nu-PVs while anogenital papillomas are almost exclusively caused by low-risk alpha-PVs, especially HPV-6 and HPV-11. 39 Papillomas appear histologically as foci of thickened folded epithelium with enlarged keratinocytes that contain shrunken dark nuclei surrounded by a clear halo (koilocytes) typically visible. The underlying tissue can contain small numbers of inflammatory cells but is often histologically normal. 47 In situ hybridization reveals that viral DNA is present in cells within all layers of the epithelium while immunohistochemistry demonstrates that L1 protein is restricted to the upper epithelium. Papillomas are self-resolving but persist for over 2 years in 22% to 35% of patients. 114 Anogenital papillomas have been reported to undergo neoplastic transformation; 58 however, many anogenital papillomas also contain a high-risk PV type, 124 suggesting the possibility of de novo development of a carcinoma within the epithelium of the papilloma. Neoplastic transformation of cutaneous papillomas appears to be extremely rare.
Immunosuppression predisposes to PV infection and can result in extensive and persistent papilloma formation. This is dramatically illustrated in people with epidermodysplasia verriciformis, a rare inherited condition in which defects in the transmembrane channel-like 6 (TMC6, EVER1) or TMC8 (EVER2) genes prevent an immune response against cutaneous PV infection. 88 Numerous other causes of immunosuppression, most notably secondary to organ transplantation, also predispose to PV infection. 39 Immunosuppression can also result in the development of cutaneous plaques. 39 These are caused by beta-PVs, which are commonly detectable on the skin of healthy humans but only result in visible lesions in immunosuppressed people. 39 These plaques are important as, in contrast to cutaneous papillomas caused by other PV genera, they can progress to squamous cell carcinomas (SCCs) if exposed to UV light. 2
Recurrent respiratory papillomatosis (RRP) is caused by HPV-6 or HPV-11 and occurs in both adults and children. 35,55 In children, the infection is thought to occur as the baby passes through the birth canal, and the presence of maternal genital warts at parturition is the strongest risk factor for the disease. 112 The presence of numerous large papillomas in the larynx can interfere with airflow, and papillomas can recur following excision. However, neoplastic transformation is rare. 35
Hyperplastic Bovine Diseases Caused by PVs
Papillomavirus infection of cattle causes mucocutaneous papillomas that develop on the haired skin, tongue, teats, penis, and vulva and upper alimentary papillomas that develop in the oral cavity, esophagus, and rumen. Virally induced papillomas typically spontaneously resolve and so are considered hyperplastic rather than neoplastic.
Mucocutaneous papillomas
Mucocutaneous papillomas (warts) are common in cattle with papillomas detected in around one-third of cattle at slaughter. 61 They are more common in younger animals, and most cattle probably develop warts during their lives. Mucocutaneous papillomas have been reported to be caused by 12 BPV types (all except BPV-4), including members of the delta, xi, dyoxi, and epsilon-PV genera. 98 However, a recent study revealed 83% of bovine skin papillomas contain 3 or more different BPV types. 109 In addition, BPV DNA can be detected in samples of clinically normal bovine skin 82 and within circulating lymphocytes. 5 Therefore, it is currently uncertain which PV types actually cause papilloma development and which PV types are detectable in papillomas as “innocent bystanders.”
Bovine mucocutaneous papillomas include squamous papillomas and fibropapillomas. Squamous papillomas have the same histological appearance as human papillomas. In contrast, fibropapillomas consist of a proliferation of mesenchymal cells covered by hyperplastic epithelium (Fig. 1). The mesenchymal cells are most densely packed close to the surface of the fibropapilloma. 52 The cells are plump and lack an orderly arrangement. The overlying epithelium may contain koilocytosis as well as large cells with blue-gray cytoplasm (Fig. 2). While PV DNA is detectable in both the epithelial and mesenchymal cells, viral L1 protein is present only within the superficial layers of the epidermis. This suggests that both cell populations are infected by PVs, but productive PV replication is limited to the epithelium. 52 Fibropapillomas are generally considered to be caused by the delta-PVs BPV-1 and BPV-2.
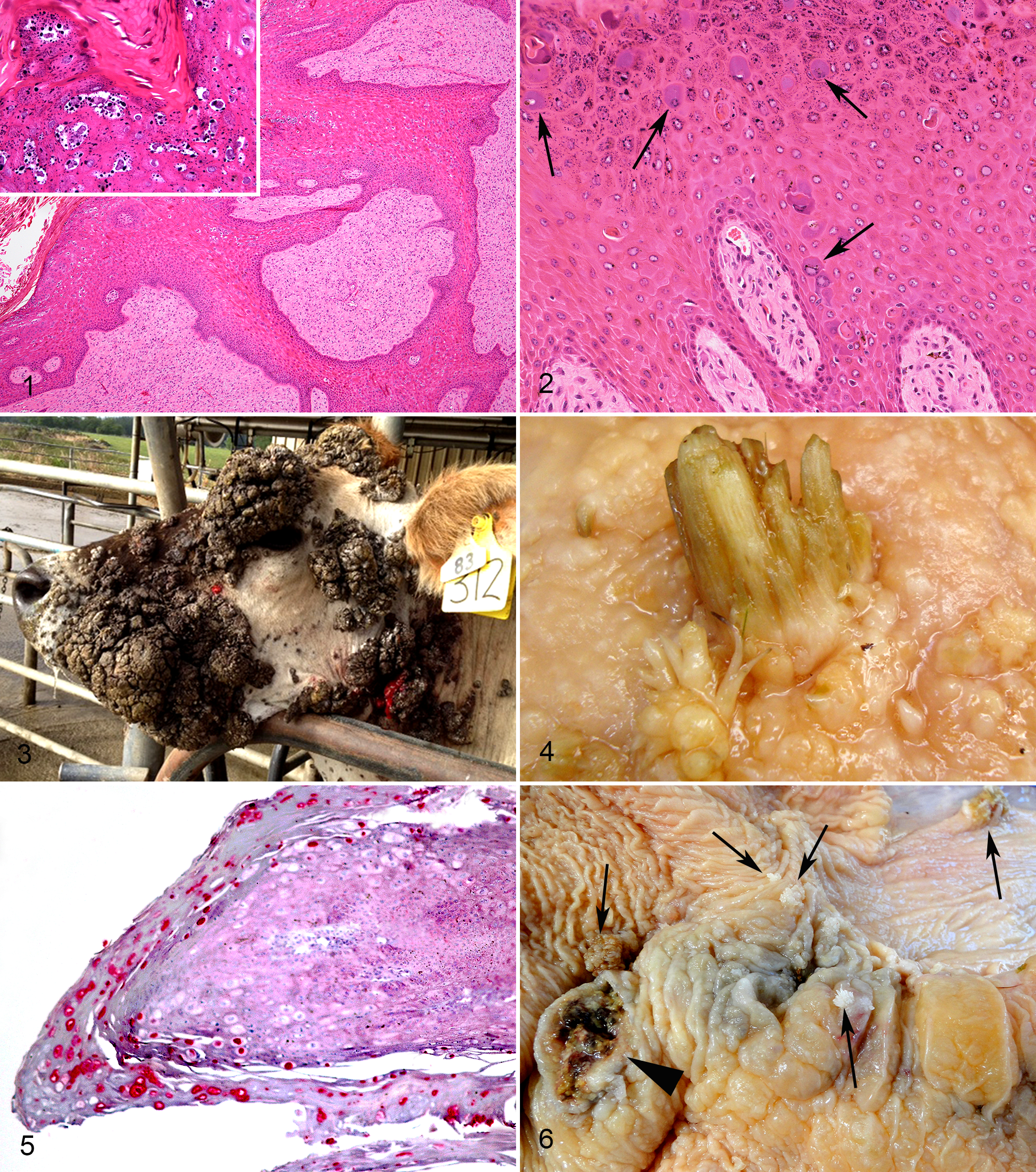
Cutaneous fibropapilloma; cow. Hyperplastic epithelium covers an orderly arranged proliferation of mesenchymal cells. Thin rete pegs of hyperplastic epithelium extend into the mesenchymal tissue. Even at low magnification, a papillomaviral etiology is suggested by the zone of cells with cytoplasmic clearing in the superficial epidermis. High magnification reveals large keratinocytes containing shrunken dark nuclei surrounded by a clear halo (koilocytes) often with clumped keratohyalin granules (inset). Sections of DNA from bovine papillomavirus 2 and from the feline sarcoid-associated papillomavirus were amplified from this lesion using polymerase chain reaction. Hematoxylin and eosin (HE).
Papillomas can be experimentally induced by inoculating scarified skin with either BPV-1 or BPV-2. 50,84 Papillomas develop only at the site of inoculation, suggesting that any BPV DNA circulating within lymphocytes does not produce additional cutaneous lesions. The overwhelming majority of bovine mucocutaneous papillomas spontaneously regress, although this can take up to 3 years. 84 Occasionally, cattle develop widespread persistent papillomas that can interfere with vision and predispose to myiasis or bacterial infection (Fig. 3). Why a small proportion of cattle develop extensive disease is unknown, although a defective immune response rather than exposure to a different BPV type is suspected. Neoplastic transformation of virally induced mucocutaneous papillomas appears to be extremely rare in cattle.
Vaccination using inactivated virus-containing tissue or purified PV structural proteins stimulates the production of virus-neutralizing antibodies and prevents the development of experimentally induced papillomas. 50 It is uncertain whether vaccination influences the regression of cutaneous papillomas in cattle. Injecting wart tissue harvested from the cow (autologous vaccination) has been widely used, although there is little published evidence of efficacy. Injection of BPV-2 L2 proteins was reported to accelerate lesion resolution in 3 calves with experimentally induced cutaneous papillomas, although no statistical analysis was reported. 51 In humans, most studies have not detected any therapeutic effect of vaccination after papilloma development. 124
Upper alimentary papillomas
Upper alimentary tract papillomas are common in cattle. Rates of disease are dependent on bracken fern exposure, and 20% of cattle from geographical areas with bracken fern will have papillomas compared with just 4.4% of cattle from areas without this plant. 49,53 The immunosuppressive properties of bracken fern are thought to predispose to papilloma development. Papillomas can develop from the caudal oral cavity to the rumen (Fig. 4). They often occur in linear arrangements or clusters, suggesting that PV infection could develop due to epithelial damage caused by swallowing rough herbage. Upper alimentary tract papillomas are histologically classified as squamous papillomas or fibropapillomas. Squamous papillomas are caused by a productive BPV-4 (a xi-PV) infection and appear histologically similar to cutaneous squamous papillomas (Fig. 5). 21 Fibropapillomas are caused by BPV-2 (a delta-PV). They appear similar to mucocutaneous fibropapillomas, although they do not contain viral cytopathology in the overlying epithelium. 21
Upper alimentary papillomas do not usually result in clinical disease and most spontaneously resolve within a year of development. Immunosuppression can result in extensive squamous papilloma formation and, rarely, ruminal tympany and weight loss. 26 As discussed later, the primary importance of upper alimentary squamous papillomas is their association with SCCs. There are sporadic reports of large fibropapillomas causing obstruction of the ruminal groove and subsequent bloat. 43
Squamous papillomas can be experimentally induced by inoculation of the mouth with BPV-4. Prior vaccination with BPV-4 structural proteins prevents papilloma development. 41 Similarly to cutaneous papillomas, it is currently uncertain whether vaccination influences regression of established upper alimentary papillomas. Although no statistics were reported, vaccination using BPV-4 proteins was reported to accelerate the regression of experimentally induced papillomas in calves. 22 In contrast, administration of BPV-4 virus-like particles in a separate experiment did not significantly influence papilloma resolution. 56
Papillomaviruses and Epithelial Neoplasia
Human PV-Associated Epithelial Neoplasia
The neoplasms that are most well established to be caused by PVs in people include the high-risk (especially HPV-16 and HPV-18) alpha-HPV–induced SCCs of the cervix, oral cavity, penis, and anorectum. In addition, beta-PVs have been associated with SCCs of the skin, predominantly in immunosuppressed people. PV infection has also been suggested as a possible cause of other cancers in people, most notably those of the lung and breast, but also including cancer of the bladder 120 and esophagus, 65 although the role of PVs in these cancers has not been defined.
The high-risk alpha-HPVs are spread by sexual contact. High-risk HPV infections can cause flat inconspicuous lesions but do not cause papillomas. In most infections, the PV DNA remains episomal. In these infections, the production of viral proteins is well regulated and, after a period of viral replication, the infection spontaneously resolves. However, in a small proportion of infections, the PV DNA becomes integrated into the cell’s genome. This is a critical event in carcinogenesis as integration can result in a marked increase in viral oncoproteins, promoting malignant transformation. 54 DNA integration is detrimental to the PV as this prevents viral replication, and PV late proteins are not present in most HPV-induced neoplasms.
The predominant viral oncoproteins of the HPVs are the E6 and E7 proteins. 39 The high-risk HPV E6 protein prevents apoptosis by degrading p53, prolongs cell life by telomerase activation, and binds to PDZ domains within several proteins involved in cell polarity, proliferation, and signaling. 57,80,107 The E7 protein of the high-risk HPVs binds and degrades key regulators of cell cycling, including retinoblastoma protein (pRb), p107, and p130. 68 In addition, E7 protein deregulates the centrosome cycle, predisposing to mistakes in chromosome segregation and subsequent DNA mutation during cell division. 68 The rapidly dividing immortal cells quickly accumulate additional mutations, resulting in malignant progression. 94
Most high-risk alpha-HPV–induced SCCs develop in the absence of any identifiable cofactors. Of the suggested cofactors for alpha-HPV-induced cancers, immunosuppression is the best established. 28 Immunosuppression probably increases the risk of neoplasia by delaying the resolution of the HPV infection and subsequently increasing the risk of PV DNA integration.
In contrast to the high-risk alpha-HPVs, immunosuppression is essential for the development of beta-PV–induced cutaneous viral plaques. 88 These plaques are predisposed to neoplastic transformation; as a result, immunosuppressed organ transplant recipients have a 100-fold higher risk of cutaneous SCCs than immunocompetent people. 60 While viral plaques are considered preneoplastic, exposure to UV light is required for progression to SCC. 88 This apparent necessity of UV light for malignant transformation is probably because the E6 and E7 proteins of the beta-PVs do not have many of the oncogenic actions of the high-risk alpha-HPV oncoproteins. 123 beta-PVs have also been suggested to promote cutaneous SCC development in immunocompetent people. 1 However, as beta-PV DNA is not consistently detectable in cutaneous SCCs and is often detectable in normal skin, it is uncertain whether any causal association exists. 2
Bovine PV-Associated Epithelial Neoplasia
In cattle, BPVs have been most strongly associated with neoplasia of the bladder and of the upper alimentary tract. The first evidence that BPVs cause bladder neoplasia was reported in 1959 when it was shown that regressing tumors of the bladder could be induced by wart extracts. 86 It was subsequently observed that injecting suspensions of bladder tumors into the skin of calves resulted in cutaneous papillomas, leading the authors to speculate that BPVs may be an etiological agent or a “passenger virus” within the bladder neoplasms. 85 In 1992, it was reported that cattle fed bracken fern consistently develop bladder cancers and that 69% of the cancers contained BPV-2 DNA. 23 An evaluation of naturally occurring bladder tumors revealed that 49% contained BPV-2 DNA, but BPV-2 DNA was detectable in only 10% of nonneoplastic bladder samples. 23 A later study reported that BPV-2 DNA was present in 77% of bladder neoplasms and 50% of nonneoplastic samples. However, BPV-2 E5 protein was present only in the bladder neoplasms. 14 Subsequently, diffuse immunostaining for BPV-2 E5 and E7 protein has been demonstrated in both epithelial and mesenchymal tumors of the bovine bladder. 15,18
A significant difference between human PV-induced cervical SCCs and bovine bladder tumors is the uncertainty of the precise role of BPV-2 in tumor development. The high-risk HPVs almost always cause neoplasia without any identifiable cofactor. In contrast, environmental carcinogens, such as ptaquiloside in bracken fern, are important cofactors with BPV-2 in the development of bovine bladder neoplasia. This is illustrated by the rarity of bladder neoplasia in cattle from geographical areas without bracken fern, despite the worldwide distribution of the PV. The necessity of bracken fern as a cofactor with BPV-2 makes it difficult to determine the contribution of each factor in the process of carcinogenesis. Ingestion of bracken fern has been shown to cause bladder cancer in rats, 90 presumably in the absence of PV infection.
Another difference between HPV-induced SCCs and bovine bladder neoplasia is the presence of a productive infection within the bovine cancers. In HPV-induced cancers, PV DNA integration prevents viral replication but causes overexpression of PV oncoproteins. In bovine bladder neoplasms, BPV-2 remains episomal and viral replication is present. 103 As E5 is expressed during PV replication, 10,103 the presence of BPV-2 E5 protein within bladder neoplasms must be interpreted with care. However, recent evidence of E5 expression diffusely within neoplasms 15 and E5 protein immunostaining within neoplastic endothelial cells 18 (which are not known to permit replication) is evidence that E5 expression in at least a proportion of neoplasms is not simply due to the presence of viral replication.
BPV-2 DNA can be detected in up to half of nonneoplastic bovine bladder samples. 14 The source of this infection is uncertain; however, an ascending infection from the skin or a hematogenous route is possible. As bracken fern causes immunosuppression, hematuria, and cystitis, this could promote both hematogenous spread of BPV-2 to the bladder and allow prolonged infection of the bladder by the PV. Therefore, it is possible that one of the oncogenic mechanisms of bracken fern is by increasing infection of the bladder by BPV-2. However, it is also possible that some bladder cancers could contain BPV-2 DNA as an “innocent bystander” due to the more permissive environment created by ingestion of bracken fern.
The mechanisms by which BPV-2 alters cell regulation are different from those seen in HPV-induced cervical SCCs. BPV-2 E7 protein cannot bind pRb, 76 and the E5 protein is thought to be the primary cause of altered cell regulation. Few studies have specifically examined BPV-2 E5 protein, but this protein has been shown to bind and activate platelet-derived growth factor–β receptor (PDGF-βR). 17 In addition, the amino acid sequence of BPV-2 E5 protein is identical to that of BPV-1 E5 protein, suggesting they are likely to have similar biological functions. 17 The BPV-1 E5 protein has recently been reviewed 125 and so will be described only briefly. The predominant interaction of the protein is with PDGF-βR, resulting in numerous downstream signaling effects. 36,95 In addition, BPV-1 E5 protein binds to vacuolar H+-ATPase, possibly altering the intraorganelle acidity, 42 although a recent study did not find altered enzyme activity due to this interaction. 6 BPV-1 E5 protein also inhibits expression of major histocompatibility complex (MHC) class 1 on the cell surface, interferes with normal gap junctions, disturbs the actin cytoskeleton, and increases motility of transformed cells. 7,122 The BPV-1 E5 protein can also activate phosphoinositide 3-kinase (PI3-K) 118 and the tyrosine kinase c-Src. 116 In contrast, HPV-16 E5 protein has not been shown to activate PDGFβ-R, PI3-K, or c-Src. 117 In vitro, BPV-1 has been shown to transform a variety of cells, including bovine palate cells, multiple mouse cell lines, and equine fibroblasts. 75,122,127 While BPV-2 has been less studied, it has been reported to transform murine cells. 40
The observation that injection of a wart extract into the bladder causes tumors, 86 the presence of BPV-2 in a high proportion of bovine bladder cancers, 23 the more frequent detection of BPV-2 in bladder cancers than in normal tissues, 23 the diffuse BPV-2 E5 and E7 protein immunostaining present in some bladder cancers, 15,18 and the ability of BPV-2 to cause cell transformation in vitro 40 provide convincing evidence that BPV-2 contributes to the development of bovine bladder cancer. Current evidence suggests that the development of cancer is probably the result of interactions between carcinogens in bracken fern such as ptaquiloside, BPV-2, and host factors. However, defining the significance of BPV-2 in carcinogenesis is critical to determine if preventing infection could prevent cancer. If BPV-2 infection is the predominant factor driving the development of bladder cancer, then preventing BPV-2 infection should reduce cancer rates. Alternatively, if environmental carcinogens or host factors are the primary factors, then preventing BPV-2 infection may not protect against bladder cancer in cattle. In 1967, a formalized wart vaccine did not prevent subsequent bracken fern–induced hematuria or bladder neoplasia when administered to calves. 89 However, this study was limited by the development of papillomas at the site of vaccine administration (suggesting the virus had not been completely inactivated), the possibility that the vaccine did not contain BPV-2, and the possibility that some calves were infected by BPV-2 prior to vaccine administration. Determining whether preventing BPV-2 infection alters rates of neoplasia in bracken fern–exposed cattle may be necessary to definitively determine the extent to which BPV-2 promotes the development of bovine bladder cancer.
BPV infection is also associated with the development of bovine upper alimentary SCCs (Fig. 6). An association between papillomas and neoplasia was recognized in African and Brazilian cattle as early as 1955. 96 In 1978, it was observed that papillomas are frequently present in cattle with upper alimentary SCCs, that the papillomas and SCCs had a similar distribution, and that some SCCs developed from preexisting papillomas that contained histological and electron microscopic evidence of PVs. 48 These observations suggested a role of BPV-4 in the development of upper alimentary SCCs. However, in contrast to HPV-induced SCCs, no BPV-4 DNA can be detected in the neoplasms. 25 In addition, although BPV-4 E5 and E7 proteins are present within upper alimentary papillomas, no PV proteins can be detected in upper alimentary SCCs. 4 As suggested by the “hit-and-run” hypothesis, this could be because BPV-4 infection causes malignant transformation and alters the microenvironment, making the virus undetectable. 113
As with bovine bladder neoplasms, the rates of upper alimentary SCCs in cattle are dependent on exposure to bracken fern. In parts of Brazil where this plant is common, upper alimentary tract SCCs are the most common bovine neoplasm. 63 In contrast, upper alimentary SCCs are extremely rare in cattle from geographical locations in which this plant is not found. Experimental feeding of bracken fern to cattle consistently resulted both in numerous large papillomas and in SCCs of the upper alimentary tract. The development of numerous papillomas is thought to be due to immunosuppression caused by bracken fern; immunosuppression induced by azathioprine also results in numerous papillomas. 26 However, azathioprine did not cause SCCs, suggesting that the presence of papillomas does not necessarily result in SCC development.
Dysregulation of the normal epithelial cell cycle by BPV-4 is primarily mediated by the E7 protein, which, in contrast to BPV-1 and BPV-2 E7 protein, can bind pRb. 76 The BPV-4 E5 protein does not activate PDGF-βR, PI3-K, or c-Src 125 but does promote cell cycling by upregulation of cyclin A through transactivation of a cyclin A promoter 44 and reduced p27, possibly due to increased cyclin D. 81 BPV-4 E5 protein also prevents surface expression of MHC class 1. 67 However, transformed cells expressing BPV-4 E5 and E7 are not immortal and do not cause tumors in nude mice. 93 This is thought to be due to an absence of an E6 ORF and therefore the transformed cells do not lose p53 function. 8 Exposure of BPV-4–transformed cells to quercetin, a mutagenic component of bracken fern, results in cells that are tumorigenic, possibly due to the development of p53 mutations within the cell and upregulation of viral protein expression. 92 This suggests that, compared with a noninfected upper alimentary epithelial cell, a BPV-4–infected cell is more likely to continue cycling after quercetin-induced DNA damage. 8,32 Therefore, a BPV-4–infected cell may be more likely than a noninfected cell to become neoplastic subsequent to bracken fern exposure.
While there is good evidence from in vitro studies that BPV-4 infection predisposes to neoplastic transformation, the absence of BPV-4 DNA in bovine upper alimentary SCCs and the requirement of bracken fern for neoplasia makes it is hard to determine the precise role of BPV-4 in cancer development. While BPV-4 appears likely to be an important cofactor in neoplasia development, it cannot be excluded that papillomas and SCCs are both independently induced by bracken fern. As with bladder cancer, observing if preventing BPV-4 infection decreases rates of upper alimentary tract cancer in cattle may be necessary to definitively determine the extent to which BPV-4 promotes cancer development.
BPVs have also been suggested to cause bovine ocular SCCs. However, while BPVs were detected in a proportion of samples from the eye, there is no evidence that BPVs act as cofactors with UV exposure in the development of these neoplasms. 105 In addition, while PVs have been associated with skin cancer in other domestic species, 72 there is little evidence that BPVs influence the development of cutaneous SCCs in cattle. The lack of association between BPVs and skin neoplasia in cattle is especially interesting considering their ability to cause mesenchymal skin neoplasia in horses and cats.
Papillomaviruses and Mesenchymal Neoplasia
There is currently no evidence that HPVs are able to infect, or cause malignant transformation of, human mesenchymal cells. In contrast, BPVs have been recognized to cause proliferation of equine dermal fibroblasts for over 60 years. 83 Since these initial reports, multiple additional species have been shown to develop cutaneous mesenchymal lesions due to natural BPV infection, and mesenchymal neoplasia has been induced in mice and hamsters after experimental inoculation of BPVs. 13,99
The association between BPVs and mesenchymal neoplasia is best studied in horses, and as recently reviewed, 77 evidence suggests that delta-BPVs contribute to equine sarcoid development. This evidence includes the almost universal presence of BPV-1, BPV-2, or, as more recently reported, BPV-13, in equine sarcoids. 11,27,64 Rare reports of epizootics of equine sarcoids also support an infectious cause. 79 Immunohistochemistry can be used to demonstrate BPV-1 E5 and E7 proteins within the proliferating fibroblasts of the sarcoids, 16 and BPV E2, E5, E6, and E7 messenger RNA (mRNA) can be consistently amplified from sarcoids. 11 BPV-1 has been shown to transform equine fibroblasts in vitro, resulting in cells that proliferate more rapidly, show anchorage-independent growth, have an expanded life span, and have abnormal p53 location within the cell. 127 Interestingly, like the BPV DNA in bovine bladder neoplasms, but in contrast to the HPV DNA in human cervical cancers, the BPV DNA in the mesenchymal cells of a sarcoid remains episomal. 3,59 However, no studies to date have examined sarcoids specifically to examine integration events within the tumors.
While BPVs are involved in the etiology of sarcoids, the precise pathogenesis remains uncertain. Inoculation of BPVs into the skin of horses results in “pseudosarcoids” that, unlike naturally occurring sarcoids, remain small and spontaneously resolve. 46 In addition, there is evidence that a proportion of horses may be asymptomatically infected by BPVs, 10,126 and a genetic susceptibility to sarcoid development has been identified in some horse breeds. 31 These observations suggest that sarcoids are not simply due to BPV infection, and additional factors, such as proliferation of mesenchymal cells (eg, due to trauma) as well as the immune reaction of the host, may be necessary. 31 BPV-1 virus-like particle vaccines have recently been developed 45 and may allow a more precise definition of the role of BPVs in sarcoid development.
The natural history of BPV infection of horses is also uncertain. Horses have historically been thought to be infected after exposure to cattle and to be dead-end hosts in which no viral replication occurs. However, more recent evidence suggests that productive BPV replication may occur in the overlying epidermis. 19,77,78 If BPVs can replicate in both cattle and horses, this is in contrast to the HPVs that are thought to be highly species specific. Productive BPV infections in horses would suggest that direct contact with cattle is not required for sarcoid development and would explain why being in contact with a horse with a sarcoid appears to predispose to sarcoid development. 78,97 Recently, BPVs have been detected in the blood of horses both with 19 and without 20 sarcoids, suggesting the possibility of blood-borne and horizontal transmission.
As discussed with bladder cancer, E5 protein appears to be the major oncoprotein protein of BPV-1 and BPV-2. Other BPV-1 proteins that may also be important in sarcoid development include the E6 protein, which has been found to interact with paxillin, disrupting the actin cytoskeleton and possibly influencing the morphology, motility, adhesion, and division of the cell. 121 Interestingly, high-risk, but not low-risk, HPVs have also been found to bind to paxillin. 121 BPV-1 E6 protein may also promote neoplastic transformation by interacting with CBP/p300, possibly influencing p53 function. 128 While playing a secondary role, BPV-1 E7 protein may also contribute to carcinogenesis by binding to cellular p600 protein and subsequently inhibiting detachment-induced apoptosis (anoikis). 34
Cats, mountain lions, and lions are all reported to develop lesions that are histologically similar to equine sarcoids. 70 The use of in situ hybridization reveals PV DNA within the proliferating mesenchymal cells of feline sarcoids but not within the overlying hyperplastic epidermis, and no evidence of viral replication is present. 119 The same PV, designated feline sarcoid-associated (FeSar) PV, has been consistently amplified from feline sarcoids. 74 Evidence that FeSarPV is a BPV includes the detection of the virus within bovine papillomas and samples of normal bovine skin, 73 the failure to detect FeSarPV on nonsarcoid skin samples from cats, 74 the association between sarcoid development in lions and feeding unskinned bovine carcasses, 87 and the higher prevalence of feline sarcoids in cats from rural areas. 110 Interestingly, the PV that causes feline sarcoids is different from BPV-1, BPV-2, or BPV-13. In addition, FeSarPV has not been detected in a sarcoid from an equine species. This suggests that BPV-1, BPV-2, and BPV-13 are restricted to bovids and equids, and FeSarPV is restricted to bovids and felids.
Summary
Over the past 50 years, the spectrum of diseases associated with HPVs and BPVs has greatly expanded, along with our understanding of the biology and carcinogenic mechanisms of these viruses. As discussed in this review, there are significant differences in the biology and oncogenic actions of BPVs and HPVs. In humans, sexually transmitted HPVs are well established to cause SCCs, and vaccines are commonly used to prevent PV infection and neoplasia. In cattle, vaccination has been shown to effectively prevent papilloma formation. However, the extent to which vaccinating cattle against BPV-2 and BPV-4 would reduce neoplasia in animals exposed to bracken fern is currently unknown. Unlike HPVs, some BPVs are well recognized to be able to infect multiple species, and in horses and cats, BPVs appear to cause mesenchymal neoplasia. Current evidence suggests that BPVs infect a wider range of tissues and may cause a more diverse range of neoplasms than HPVs. However, it is interesting to note that some of the observations of BPVs 50 years ago were subsequently found to be applicable to HPVs, and it is possible that as HPVs are further studied, additional similarities between BPVs and HPVs may be identified.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
