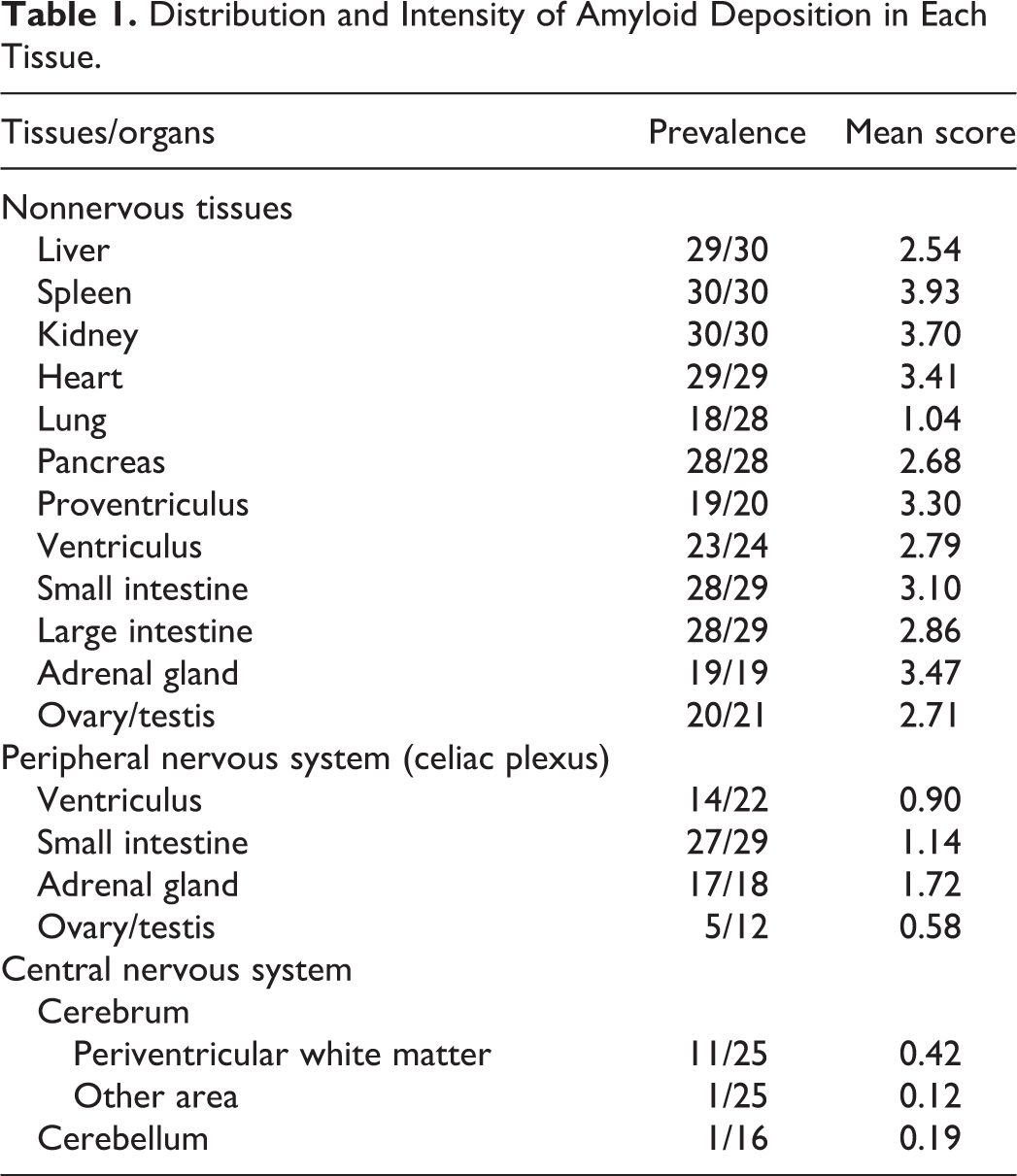
Abstract
AA amyloidosis is characterized by amyloid deposition in systemic organs, but amyloid deposition in the central nervous system (CNS) or peripheral nervous system (PNS) is rare. In this study, AA amyloidosis was observed in 31 of 48 flamingos that died at a Japanese zoo. Almost all cases developed AA amyloidosis secondary to inflammatory diseases such as enteritis. Affected flamingos had AA amyloid deposition around blood vessels in periventricular white matter of the brain and in peripheral nerves. In addition, cerebral Aβ amyloidosis was observed in one of the 31 cases with AA amyloidosis. In conclusion, flamingos in the zoo commonly developed systemic amyloidosis with frequent amyloid deposition in the CNS and PNS, which seems to be a unique distribution in this avian species. Comparative pathological analyses in flamingos may help elucidate the pathogenesis of amyloid neuropathy.
Amyloid is an insoluble fibrillar protein that is generated by misfolding of precursor proteins that forms a cross-β sheet structure and stains positively with Congo red with green birefringence under polarized light. In humans, 36 types of amyloidogenic proteins have been identified and the associated diseases are categorized as systemic or localized. 3
Systemic amyloidosis is caused by overproduction and saturation of amyloid precursor protein in serum, resulting in amyloid deposition throughout the body. 4 AA amyloidosis is characterized by systemic deposition of amyloid derived from serum amyloid A (SAA), 13 an acute-phase protein synthesized in the liver during inflammation. 23 Although chronic inflammatory diseases or malignant tumors cause long and sustained excessive SAA synthesis and misfolding, leading to AA amyloidosis, 1,22 idiopathic AA amyloidosis is also frequently observed. 15 AA amyloidosis is the most frequent amyloidosis in animals and is observed in various species of mammals and birds. 10,14 Several species, such as cheetahs and island foxes, are highly susceptible to AA amyloidosis, 9,20 and Siamese and Abyssinian cats and Shar Pei dogs develop familial AA amyloidosis. 31 Amyloid deposition can be fatal, but subclinical amyloidosis is also common. 15
In birds, only 2 types of amyloidosis have been identified: AA amyloidosis and cerebral Aβ amyloidosis accompanied by cerebral amyloid angiopathy. 7,12,14,18 Avian AA amyloidosis is frequent in waterfowl, small passerine birds, and chickens as a secondary disease of chronic inflammation. 8,16,24,27 Waterfowl with AA amyloidosis develop amyloid deposition mainly in spleen, liver, and kidney. 8,27 In flamingos, one case was reported of systemic AA amyloidosis in a roseate flamingo, 5 but no reports have investigated the incidence and etiology of amyloidosis in flamingos. Aβ amyloidosis has been reported in great spotted woodpeckers and eagles 12,18 but not in flamingos.
Generally, systemic amyloidosis does not involve amyloid deposition in the central nervous system (CNS). 25 Peripheral amyloid neuropathy often occurs in certain systemic amyloidoses such as amyloid-transthyretin (ATTR) amyloidosis and amyloid-light chain (AL) amyloidosis. 26 However, in human and animal AA amyloidosis, amyloid deposition is rarely observed in the CNS and peripheral nervous system (PNS). 26
In this study, we report high prevalence of AA amyloidosis in flamingos. In those cases, vascular AA deposition was observed in the CNS and PNS. Furthermore, we also identified a flamingo case of systemic AA amyloidosis that was complicated with cerebral amyloid angiopathy.
Materials and Methods
Animals
Forty-eight flamingos including 32 roseate flamingos (
Pathological Analyses
Formalin-fixed paraffin-embedded tissues were cut into 2-μm-thick sections and stained with hematoxylin and eosin or Congo red. To evaluate the argyrophilia of amyloid deposition in the nervous system, periodic acid-methenamine-silver (PAM) stain was also performed. Amyloid deposits were detected in Congo red–stained sections as emerald green birefringence under polarized light. The degree of amyloid deposition was scored as follows: −, no deposition; +, deposition in a few vessels; ++, deposition in several vessels; +++, deposition in almost all vessels and perivascular areas; ++++, severe deposition in vessels and interstitial tissues. The amyloid index scores in nonnervous tissues and the PNS were calculated by averaging the amyloid score in each organ as follows: −, 0; +, 1; ++, 2; +++, 3; ++++, 4.
Immunohistochemistry (IHC) was performed with anti-human SAA rabbit polyclonal antibody (Cloud-Clone Corp, Katy, TX) and anti-human Aβ mouse monoclonal antibody (Lab Vision Cor., Fremont, CA) as the primary antibody. Horseradish peroxidase–labeled polymer anti-mouse or rabbit IgG antibody (Dako, Glostrup, Denmark) were used as the secondary antibody. IHC was visualized using 3,3′-diaminobenzidine tetrahydrochloride for color development. For negative controls, the primary antibody was omitted.
Statistical Analyses
Correlations were determined by calculating Spearman’s rank correlation coefficient, using GraphPad Prism8 (GraphPad Software, San Diego, CA).
Results
Prevalence and Disease Associations
The case information and the presence or absence of systemic amyloidosis are shown in Table S1. Pathological examination showed amyloidosis in 31 of 47 flamingos (64%). This was 17 of 23 males (74%) and 14 of 24 females cases (58%); no clear sex difference was noted. No clear difference between species was observed; the incidence of amyloidosis in roseate flamingos was 22/32 (69%), and that in greater flamingos was 8/15 (53%). Of the 22 amyloidosis cases in which the age was recorded, 6 cases were 1 or 2 years old, and the remaining 16 cases were over 10 years old. Most of the 31 flamingos with amyloidosis had chronic inflammatory diseases such as chronic enteritis (16 cases), bumblefoot (10 cases), and arthritis (3 cases).
Amyloid Deposition in Nonnervous Tissues
The distribution and intensity of amyloid deposition are shown in Table 1 and Table S2. Almost all cases developed severe systemic amyloid deposition which most often affected the liver, spleen, kidney, heart, proventriculus, small intestine, and gonads. In the liver, amyloid was deposited in the vessel walls of Glisson’s capsule and in the space of Disse. Splenic amyloid deposition was severe in most cases, and amyloid deposition occupied the entire red pulp. In the kidney, amyloid was deposited in the glomerulus and the interstitial vessel walls. In the heart, amyloid deposition was present in the interstitium of the myocardium. In the lung, mild amyloid deposition was present in the vessel walls. In the pancreas, amyloid was deposited in the vessel walls and in the periphery of ducts. In the alimentary tract, amyloid deposition was observed in the lamina propria or the vessel walls. In the adrenal gland, amyloid deposition was present in the vessel walls and stroma. In the testis and ovary, amyloid deposition was observed in the interstitium and vessel walls. IHC showed that these systemic amyloid deposits were positive for AA, and thus systemic AA amyloidosis was confirmed.
Distribution and Intensity of Amyloid Deposition in Each Tissue.
Amyloid Deposition in the PNS
Of the 29 cases in which systemic amyloidosis was observed and the PNS was available, amyloid deposition was observed in the PNS in 28 cases (97%). Amyloid deposits were observed in endoneurial microvessels and the perivascular interstitium of the celiac plexus in the alimentary tract, adrenal gland, and testis or ovary. These amyloid deposits were negative with PAM staining. IHC revealed that amyloid deposits in the PNS were positive for AA and negative for Aβ (Figs. 1, 2). The amyloid index of nonnervous tissues and the amyloid index of the PNS were moderately correlated (
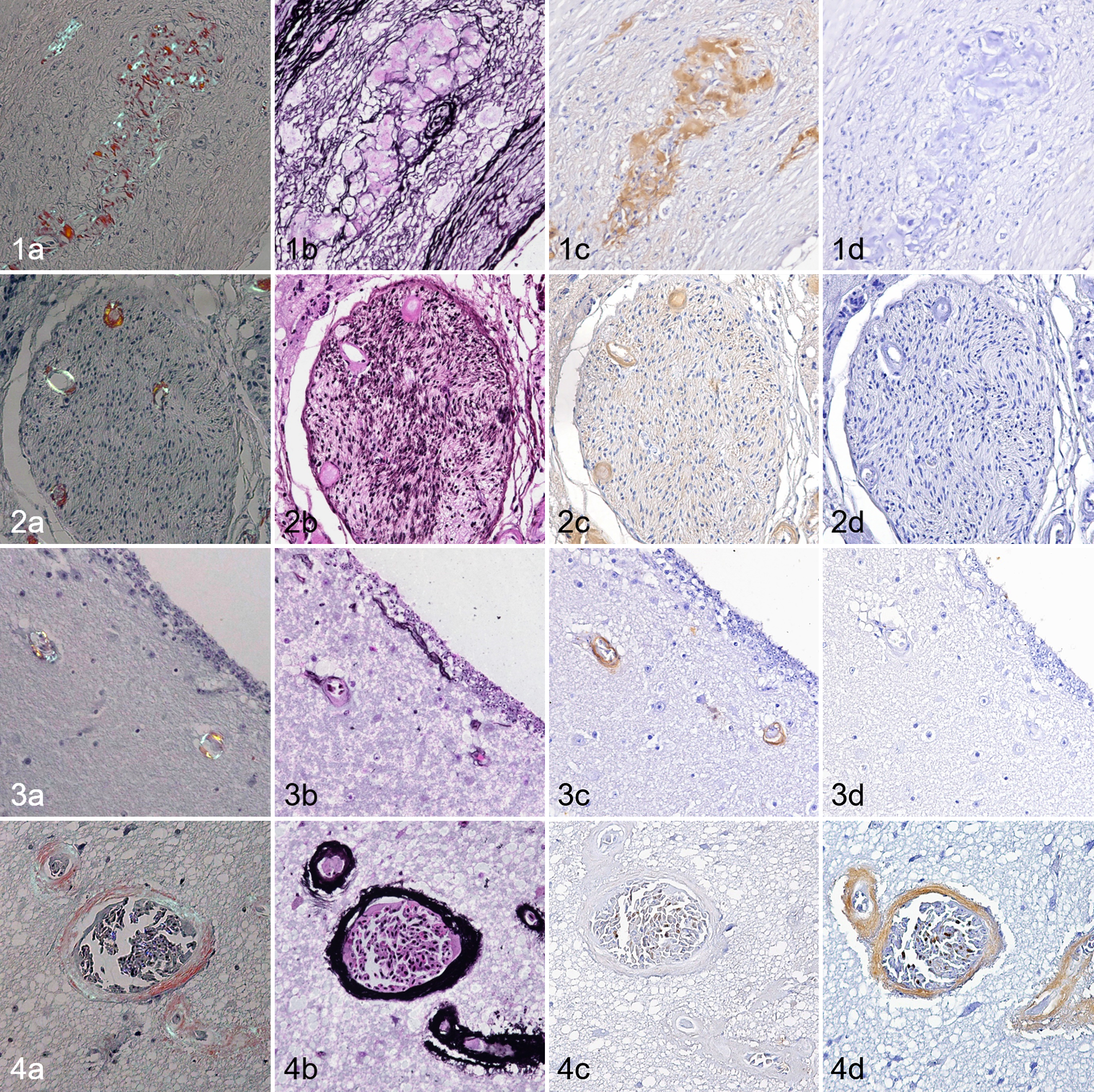
Amyloidosis, flamingo. (a) Congo red stain under polarized light. (b) Periodic acid-methenamine-silver (PAM). (c) Immunohistochemistry (IHC) for serum amyloid A (SAA). (d) IHC for β-amyloid (Aβ).
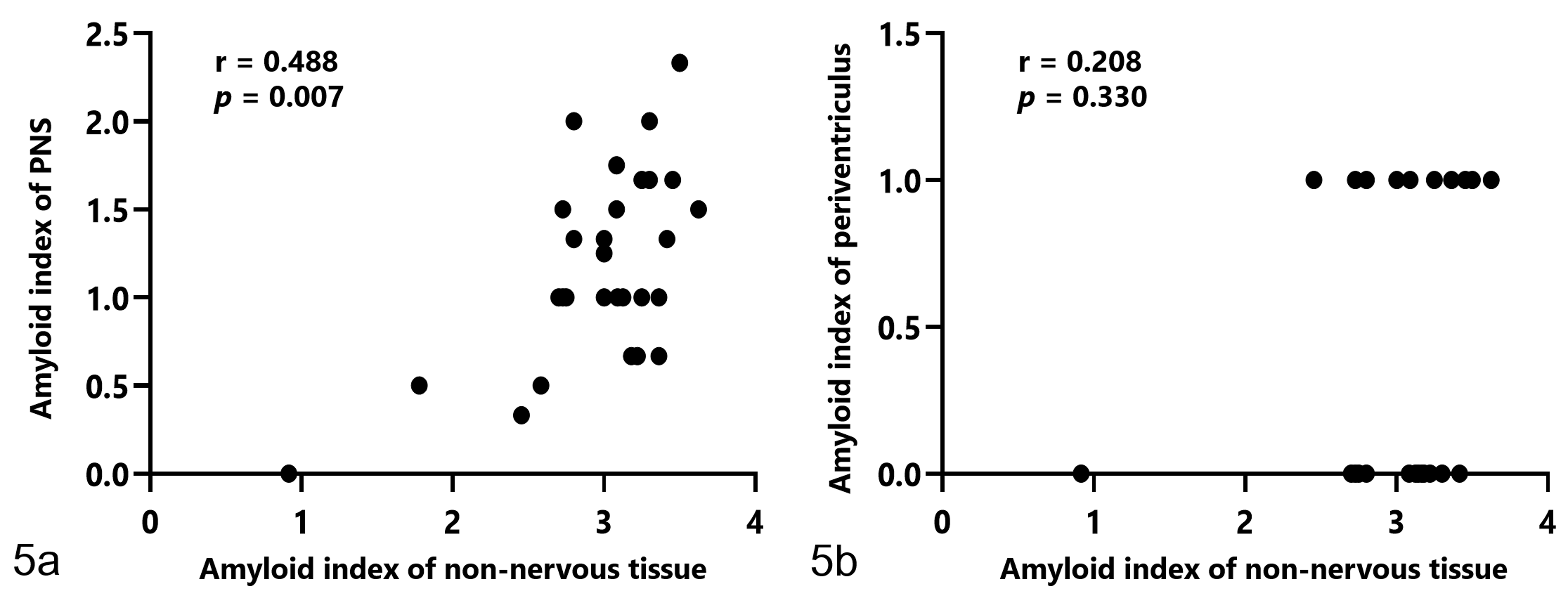
In flamingos with amyloidosis, there is a moderate correlation between the amyloid index of nonnervous tissue and the amyloid index of the peripheral nervous systems (PNS) (a). There is no correlation between the amyloid index of nonnervous tissue and the amyloid index of vessels in the periventricular white matter of the brain (b).
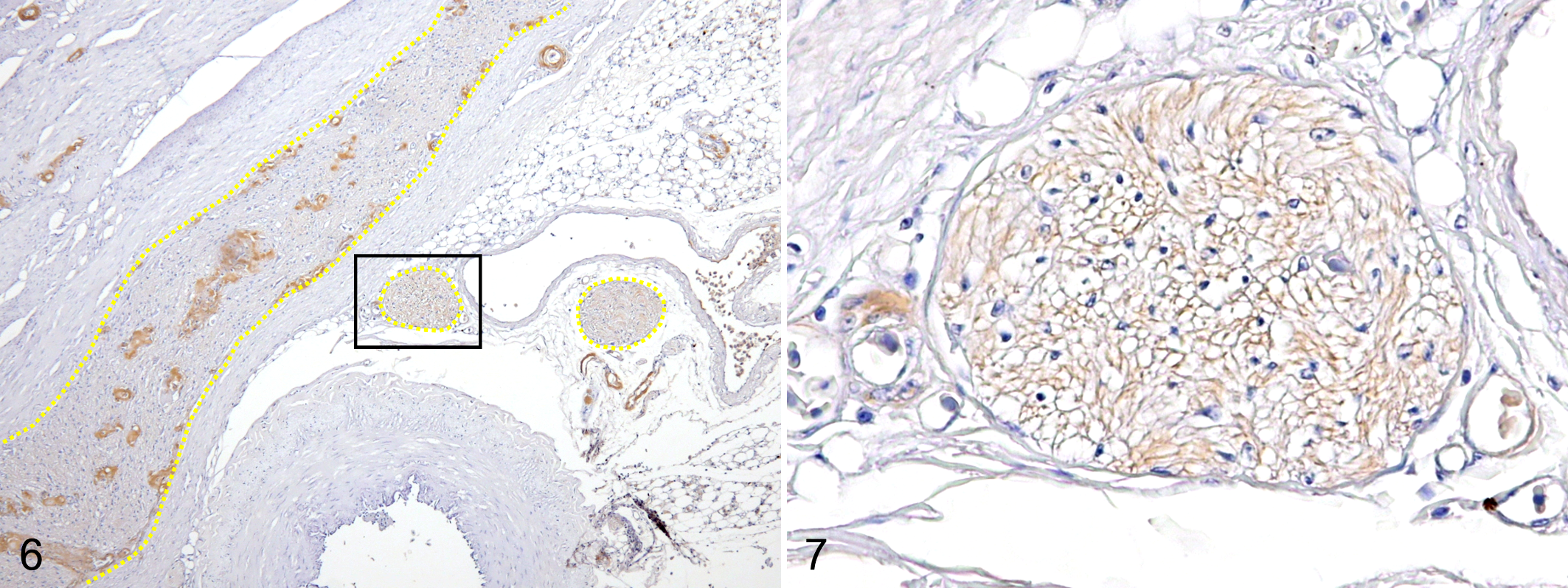
Amyloidosis, ventriculus, flamingo, case 23. Immunohistochemistry for serum amyloid A (SAA).
Amyloid Deposition in the CNS
Of the 25 cases in which systemic amyloidosis developed and the CNS was available, amyloid deposition was observed in the CNS in 11 cases (44%). Ten of 11 cases developed amyloid deposition limited to the periventricular area of the brain. Amyloid deposits were observed in the blood vessel walls (Fig. 3) or perivascular areas (Fig. S1). In these cases, amyloid deposits were negative with PAM staining. IHC revealed that the periventricular amyloid deposits were positive for AA and negative for Aβ (Fig. 3).
Except for case 14, there was no clear correlation between the amyloid index of non-nervous tissues and periventricular amyloid deposition (
Discussion
In this study, nearly one third of flamingos with systemic amyloidosis developed cerebrovascular AA amyloid deposition. In animals, cerebrovascular amyloidosis is generally due to Aβ amyloidosis 32 and AA deposition in the CNS has not been reported. AA deposition in periventricular blood vessels associated with systemic AA amyloidosis has been reported only once in humans, and the etiology is suggested to involve a leaky blood-brain barrier in circumventricular organs. 25 In this study, cerebrovascular AA deposition was observed only in periventricular vessels, suggesting that the specific structure of periventricular blood vessels may be related to this pathologic process.
In humans, amyloid deposition in the PNS has been reported in various systemic amyloidoses. ATTR amyloidosis and AL amyloidosis are frequently accompanied by amyloid deposition in the peripheral nerve endoneurium and microvessels and causes neuropathy. 2,26 Furthermore, amyloid-ApoAI amyloidosis, 28 amyloid-gelsolin amyloidosis, 6 and amyloid-β2 microglobulin amyloidosis 30 also cause autonomic neuropathy due to amyloid deposition in the PNS. On the other hand, AA deposition in PNS is extremely rare in humans, although idiopathic cases of AA deposition in the radial nerve 21 and vestibulocochlear nerve 29 have been reported. Why AA amyloidosis rarely causes peripheral nervous amyloid deposition has not been clarified.
To our knowledge, no reports of peripheral nervous amyloid deposition have been reported in animals. However, in this study, flamingos frequently showed systemic AA amyloidosis accompanied by amyloid deposition in the PNS. The degree of amyloid deposition in the PNS was positively correlated with the severity of systemic amyloidosis, suggesting that amyloid deposition in the PNS develops as one type of pathological change in systemic amyloidosis in flamingos. In other words, amyloid deposition in the peripheral nerves of flamingos seems to not require a special external factor, and species-specific intrinsic factors such as the blood vessel structure are likely involved.
In ATTR amyloidosis, amyloid deposition in the PNS is considered to be partially derived from transthyretin synthesized by Schwann cells. 17 In addition, production of SAA1 and SAA3 in Schwann cells has also been demonstrated in mice. 11 In this study, Schwann cells were positive for SAA by IHC. Whether Schwann cells of flamingos might produce SAA and thus cause amyloid deposition will require further investigation.
Avian cerebral amyloid angiopathy has been reported in a woodpecker and eagles, 12,18 and aging may be a causative factor. Case 14 in this study developed cerebral amyloid angiopathy with perivascular plaques containing Aβ amyloid as identified by IHC. Although the age was not recorded in this individual, histopathological analyses revealed notable lipofuscin deposition in cerebellar Purkinje cells compared to other elderly individuals, suggesting that this individual was considerably aged. Therefore, aging might be related to development of cerebral amyloid angiopathy in flamingos as well as other birds.
Among flamingos with amyloidosis, the age distribution was bimodal with cases involving birds around 1 year old and over 10 years old. Flamingos of intermediate ages did not show amyloidosis, indicating that the age distribution tends to be bimodal. Bear in mind, the amyloidosis-negative group also included juvenile or geriatric individuals. In general, AA amyloidosis is observed in mature animals, 19 but amyloidosis was also observed in juvenile flamingos, suggesting that flamingos may be highly susceptible to AA amyloidosis.
In conclusion, captive flamingos in this zoo developed systemic AA amyloidosis at high incidence. Furthermore, flamingos developed AA deposition in the CNS and PNS which is not previously reported in animals. In humans, the pathology of amyloid neuropathy varies with the amyloid subtype, 26 and the etiology is not fully understood. We hope our comparative pathological analyses using flamingos will contribute to elucidation of the pathogenesis of amyloid neuropathy.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Ono_et_al - AA Amyloid Deposition in the Central and Peripheral Nervous Systems in Flamingos
Supplemental Material, Combined_supplemental_materials-Ono_et_al for AA Amyloid Deposition in the Central and Peripheral Nervous Systems in Flamingos by Ayumi Ono, Yumi Nakayama, Maki Inoue, Tokuma Yanai and Tomoaki Murakami in Veterinary Pathology
Footnotes
Acknowledgements
The authors are grateful to zoo staffs for their cooperation in collecting samples and information. We also greatly indebted to Prof Hidefumi Furuoka (Obihiro University of Agriculture and Veterinary Medicine) for providing positive control for avian cerebral amyloid angiopathy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by JSPS KAKENHI (Grant Numbers 16H05027 and 17K17702) and TUAT president’s discretionary funding to support the launch of next-generation research projects.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
