Abstract
Polyneuropathy is defined as the simultaneous dysfunction of several peripheral nerves. In dogs, a number of breeds are predisposed to a variety of immune-mediated and/or degenerative inherited forms of polyneuropathy, with laryngeal paralysis and/or megaesophagus as important clinical features of many of these conditions. This case series describes degenerative and inflammatory polyneuropathies in 7 young Siberian huskies that were categorized based on clinicopathological characteristics as follows: (1) slowly progressive laryngeal paralysis and megaesophagus caused by primary axonal degeneration with large fiber loss (n = 2); (2) slowly progressive polyneuropathy without megaesophagus or laryngeal paralysis caused by primary axonal degeneration with large fiber loss (n = 2); (3) acute inflammatory demyelinating neuropathy causing sensory, motor and autonomic nerve deficits (n = 2); and (4) ganglioradiculitis (sensory neuronopathy; n = 1). Based on the predominantly young age at onset, slow progression, relatedness of affected dogs, and clinical and pathological similarities with inherited neuropathies reported in other dog breeds, a hereditary basis for the degenerative polyneuropathies in Siberian huskies is suspected. However, 5 different mutations in 3 genes known to cause polyneuropathy in other dog breeds (NDRG1, ARHGEF10, or RAB3GAP1) were not detected in the affected Siberian huskies suggesting that more genetic variants remain to be identified. This study highlights the varied underlying lesions of polyneuropathies in young Siberian huskies.
Keywords
Polyneuropathy is defined as the simultaneous dysfunction of several peripheral nerves. In dogs, a number of breeds are predisposed to a variety of immune-mediated and/or degenerative inherited forms of polyneuropathy, with laryngeal paralysis and/or megaesophagus as important clinical features of many of these conditions. This case series describes degenerative and inflammatory polyneuropathies in 7 young Siberian huskies that were categorized based on clinicopathological characteristics as follows: (1) slowly progressive laryngeal paralysis and megaesophagus caused by primary axonal degeneration with large fiber loss (n = 2); (2) slowly progressive polyneuropathy without megaesophagus or laryngeal paralysis caused by primary axonal degeneration with large fiber loss (n = 2); (3) acute inflammatory demyelinating neuropathy causing sensory, motor, and autonomic nerve deficits (n = 2); and (4) ganglioradiculitis (sensory neuronopathy; n = 1). Based on the predominantly young age at onset, slow progression, relatedness of affected dogs, and clinical and pathological similarities with canine-inherited neuropathies reported in other breeds, a hereditary basis for the degenerative polyneuropathies in Siberian huskies is suspected. However, 5 different mutations in 3 genes known to cause polyneuropathy in other dog breeds (NDRG1, ARHGEF10, or RAB3GAP1) were not detected in the affected Siberian huskies suggesting that more genetic variants remain to be identified. This study highlights the varied underlying lesions of polyneuropathies in young Siberian huskies.
Peripheral neuropathy refers to disease or damage affecting single or multiple peripheral nerves and may result in paresis, paralysis, change in sensation, or autonomic dysfunction. 11 While the clinical signs vary considerably depending on the group of nerves affected, megaesophagus and laryngeal paralysis occur commonly in dogs with peripheral neuropathies. 23 The most commonly identified causes of acquired megaesophagus are myasthenia gravis and generalized myopathies, followed by a variety of immune-mediated, endocrine, gastrointestinal, paraneoplastic, and toxic neuromuscular diseases. 26 Less commonly, acquired megaesophagus in dogs has been associated with a variety of polyneuropathies such as giant cell axonal neuropathy, 11 ganglioradiculitis (sensory neuropathy), 12 polyradiculoneuritis, 21 cranial polyneuritis and ganglionitis, 19 and dysautonomia. 34 The simultaneous presentation of megaesophagus with gait abnormalities and laryngeal paralysis is suggestive of laryngeal paralysis–polyneuropathy complex, 23 an inherited polyneuropathy that has been reported in Dalmatians, 5 juvenile Rottweilers, 27 Pyrenean mountain dogs, 20 Alaskan malamutes, 31 white-coated German shepherds, 40 and miniature schnauzers. 42
Inherited motor and sensory neuropathies have been suspected in about 23 dog breeds, 14,23 but the genetic variants responsible have been described in only a few breeds. 2,9,14,17,18 There have been reports of inherited laryngeal paralysis in Siberian huskies and husky crossbreds in the literature, 35,37 but these cases cannot be classified as peripheral neuropathies because causative lesions were observed in the brain. Siberian huskies were overrepresented (3 out of 8 cases) in 2 small case series of dogs with sensory neuropathy, 12,45 suggesting that this breed may be predisposed to polyneuropathy. The present study reports the clinicopathological features of degenerative and inflammatory polyneuropathies with and without the presence of megaesophagus and laryngeal paralysis in 7 Siberian husky dogs.
Material and Methods
Seven Siberian huskies (3 female and 4 male) between the ages of 3 months and 4.5 years with peripheral nerve deficits and generalized weakness were investigated. Two of these dogs (cases 3 and 4) were related, bred in the United States, and evaluated at the William R. Pritchard Veterinary Teaching Hospital, University of California Davis. The other 5 dogs presented to the University Veterinary Hospital, University College Dublin, Ireland.
Each dog received physical and neurological examinations. Diagnostic testing performed included hematology and biochemistry analysis, laryngoscopy, and thoracic radiographs. Two dogs (cases 3 and 4) had a lumbar CSF (cerebrospinal fluid) tap and an electrophysiological evaluation including electromyography and measurement of motor and sensory nerve conduction velocity. Nerve and skeletal muscle biopsies including teased nerve fibers were available from these 2 dogs (cases 3 and 4).
Postmortem examinations were performed on 5 dogs (cases 1, 2, 5, 6, and 7) and samples of brain, spinal cord, peripheral nerves, and different muscle groups were fixed in 10% neutral buffered formalin. Additional tissues including autonomic ganglia, laryngeal muscles, and esophagus were taken from 4 cases (cases 1, 2, 5, and 6). Following fixation, tissues were embedded in paraffin wax, sectioned at 4 µm and stained with Gill-2 hematoxylin and eosin (H&E). Additional sections of brain, ganglia, peripheral nerves, and esophagus were stained with Luxol fast blue and Masson’s trichrome. 1
Frozen muscle specimens of 6 dogs (cases 1–6; cranial tibial muscle and/or esophagus, dorsal cricoarytenoid muscle) were shipped on dry ice to the Comparative Neuromuscular Laboratory at the University of California, San Diego, where sections (8-μm-thick) were examined using a standard panel of histochemical stains and enzyme reactions. 15 In addition, fixed peripheral nerve tissue was processed for resin sections, cut (1 μm) and stained with toluidine blue acid fuchsin or paraphenylenediamine (PPD) for light microscopy. Additional nerve sections of cases 3 and 4 were processed for teased nerve fibers. 42
For identification of the inflammatory infiltrate, formalin-fixed paraffin-embedded sections were investigated by immunohistochemistry (IHC) using the Dako Link automated stainer with primary antibodies specific for CD3 as a T-cell marker (Dako, Gostrup, Denmark; polyclonal rabbit antibody; antigen retrieval by incubation in Citrate buffer at pH 6; dilution 1 in 400), CD79a as a B-cell marker (AbD Serotec, Raleigh, NC; monoclonal mouse antibody; antigen retrieval by incubation in citrate buffer at pH 6; dilution 1 in 500), and CD18 as a marker for activated macrophages and polymorphonuclear cells (Peter Moore, Leukocyte Antigen Biol Lab, Davis, CA, monoclonal mouse antibody, protease digestion for antigen retrieval, dilution 1 in 10). For detection of infectious agents, formalin-fixed paraffin-embedded sections from the cranial cervical ganglion and the cranial tibial muscle of case 6 were investigated by IHC using primary antibodies specific for Neospora caninum and Toxoplasma gondii (Veterinary Pathology, Institute of Veterinary Science, University of Liverpool, UK).
Genetic Analysis
In 3 cases (cases 1, 5, and 6), genomic DNA was isolated from frozen tissue using a traditional phenol-chloroform extraction method. A saliva sample (case 2) was obtained using the Oragene Animal saliva collection kit (OA-400, DNA Genotek, Ottawa, Canada) and genomic DNA was isolated according to the manufacturer’s instructions.
PCR was performed on genomic DNA (30 ng/μl) using Platinum Taq DNA polymerase (ThermoFisher Scientific, Dublin, Ireland) and the following protocol: 3 minutes at 94°C; 35 cycles of 94°C, 30 seconds; 65°C, 30 seconds; 72°C, 1 minute. Five different primer pairs, capable of amplifying mutation-containing regions, were used (Supplemental Table S1). PCR (polymerase chain reaction) products were purified using the JETQUICK PCR product purification Kit (Genomed, FL) or the QIAquick PCR purification kit (Qiagen, Manchester, UK) and sequenced commercially (Eurofins MWG, Ebersberg, Germany) with the primers used in the PCR amplifications. Two cases (cases 3 and 4) had been tested for NDRG1 (Gly98Val; Exon 4) previously. 9
Results
Based on key shared clinicopathological features (Supplemental Table S2), the 7 Siberian huskies were categorized into 2 major groups: degenerative and inflammatory polyneuropathy.
Degenerative Polyneuropathy
Two unrelated female dogs (cases 1 and 2) presented with regurgitation and inspiratory stridor at 3 and 11 months of age. Laryngoscopy and thoracic radiography confirmed bilateral laryngeal paralysis and megaesophagus in both cases. Hematology and serum biochemistry showed no significant abnormalities. Hypothyroidism and myasthenia gravis were ruled out in both dogs based on T4 and cTSH levels within normal range and a low titer of acetylcholine receptor antibody, respectively. Case 1 had a female litter mate that presented at the age of 4 months with the same clinical signs and results of diagnostic imaging and blood work and was euthanized at the age of 13 months due to severe aspiration pneumonia, but no necropsy was performed. Cases 1 and 2 were euthanized; one dog (case 1) due to a perceived poor prognosis 1 month after clinical onset, and one dog (case 2) due to severe aspiration pneumonia after 7.9 years. Case 2 had been treated conservatively using postural feeding. It developed mild ataxia at the age of 4 years and had 5 episodes of aspiration pneumonia all successfully treated with antimicrobial therapy and anti-inflammatories prior to the terminal bout.
Two male related dogs (half-brothers; cases 3 and 4) were presented at 9 months and 4.5 years of age for pelvic limb ataxia and weakness. Neither dog had evidence of laryngeal dysfunction or megaesophagus. In one dog (case 4), there was generalized muscle atrophy present with reduced segmental reflexes. In the other dog (case 3), there were subtle deficits of mild generalized ataxia and proprioceptive positioning present in the pelvic limbs. Both dogs had spontaneous activity on EMG examination, as well as reduced motor and sensory nerve conduction velocities. The more clinically severely affected dog (case 4) had reduced M-wave amplitude, abnormal M-wave configuration, and dispersion. This dog further had a mildly elevated protein concentration in the lumbar CSF (45 mg/dl, normal is <35 mg/dl).
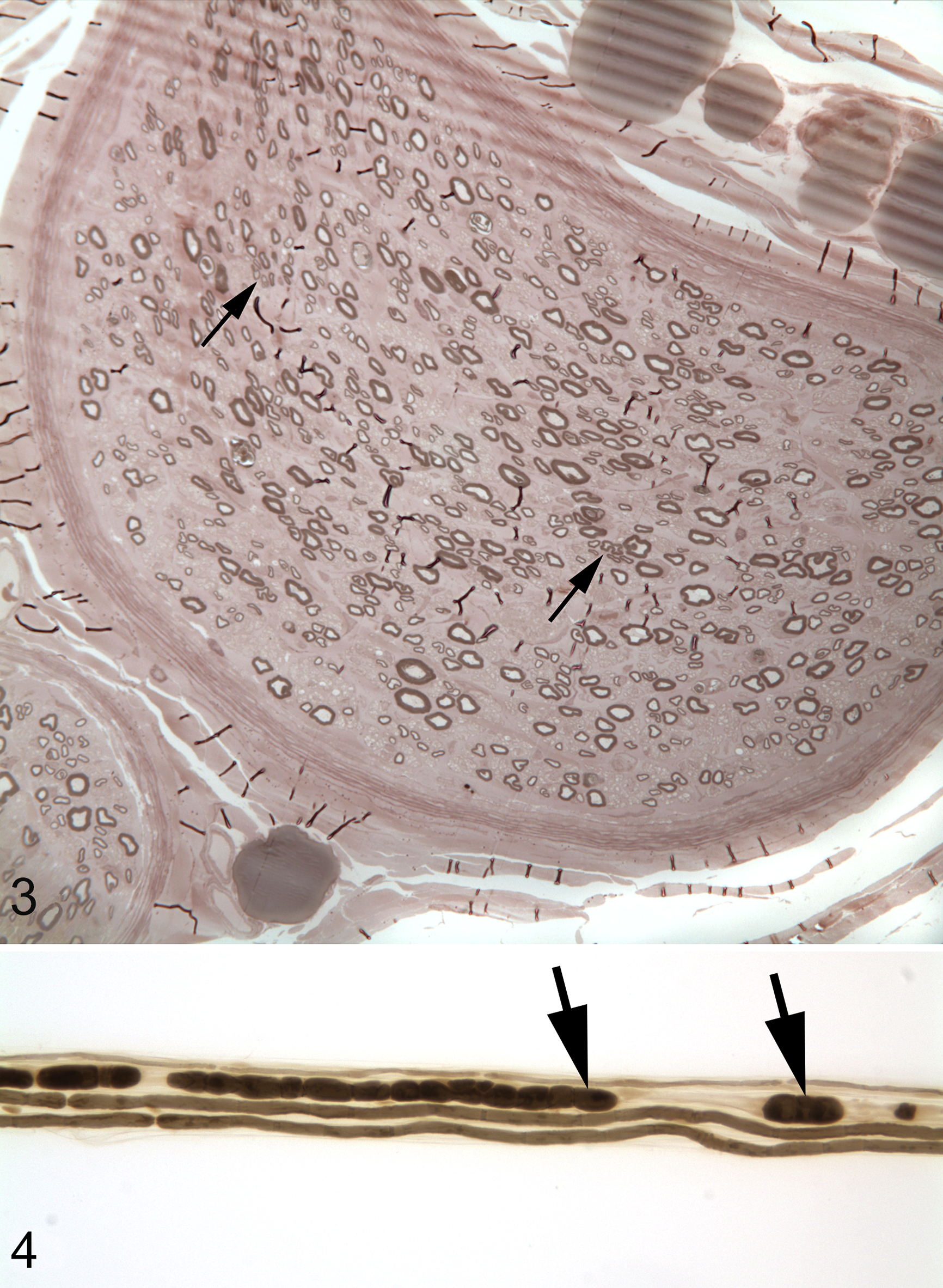
The consistent finding in all 4 dogs were the changes in the peripheral nerves, based on evaluation of nerve root and laryngeal nerves from cases 1 and 2, and of peroneal nerves from cases 3 and 4. On semi-thin sections of the nerves, changes were chronic and ranged from moderate to severe nerve fiber loss resulting from axonal degeneration (Fig. 1). Variable endoneurial fibrosis was present. Rare myelin ovoids were observed in the endoneurium but were prominent along the length of teased nerve fibers (Figs. 2–4).
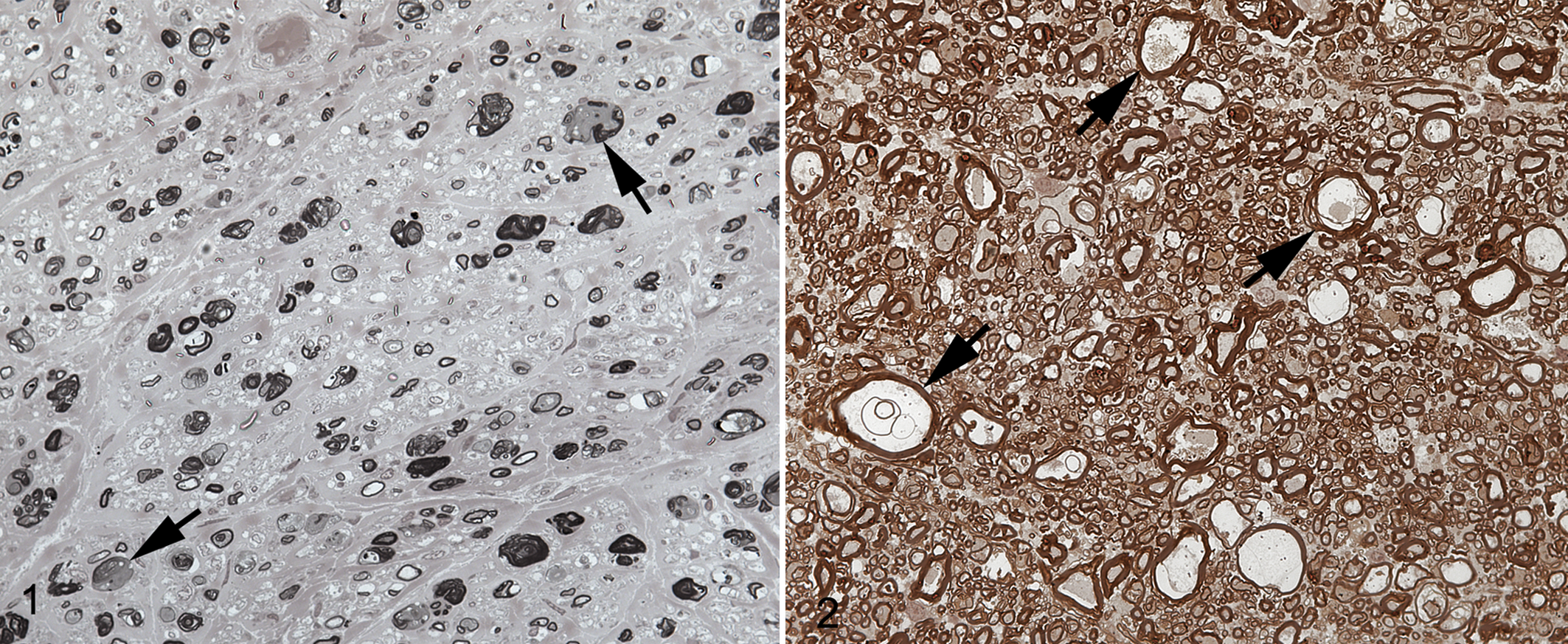
Degenerative polyneuropathy, Siberian husky dog, case 2.
Degenerative polyneuropathy, peroneal nerve, Siberian husky dog, case 3.
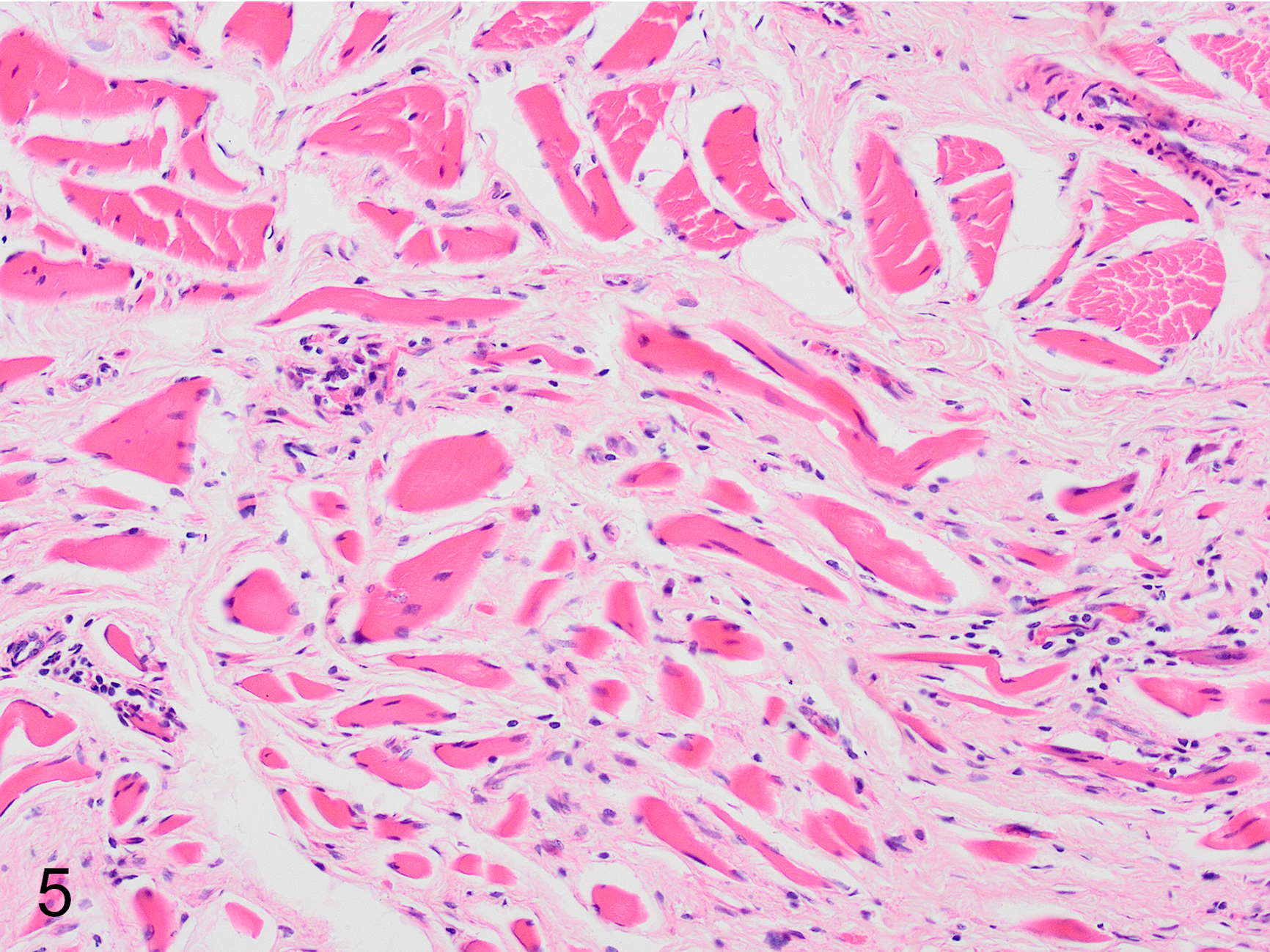
Microscopically, there was neurogenic muscle atrophy in the muscles of the larynx and esophagus (cases 1 and 2) and marked muscle atrophy in the cranial tibial muscle (cases 2 and 4) and the brachialis muscle (case 2). This was characterized by excessive variability in myofiber size, scattered atrophic fibers having an angular shape, swelling of other fibers with hypereosinophilic amorphous cytoplasm, and occasional infiltration of myofibers by neutrophils and macrophages (Fig. 5). Rare regenerating myofibers with rows of large vesicular nuclei in the center or periphery of myocytes were present. Fiber-type grouping was not observed. Some intramuscular nerve branches within the affected muscle groups were depleted of myelinated fibers and contained myelin ovoids.
Degenerative polyneuropathy, esophagus, Siberian husky dog, case 2. Variably sized muscle fibers are separated by collagen (neurogenic muscle atrophy). Hematoxylin and eosin (HE).
The main postmortem findings for cases 1 and 2 were poor body condition, and a diffusely dilated flaccid esophagus (up to 7 cm in diameter). There was marked bilateral atrophy of the dorsal cricoarytenoid muscles in both dogs and marked generalized muscle atrophy in case 2. This dog had a severe purulent bronchopneumonia and microscopic examination of various organs further revealed systemic amyloidosis affecting the liver, kidney, thyroid, and spleen. No changes were observed in the autonomic and sensory ganglia, brain and spinal cord in either dog.
These findings were present in all 4 cases but targeted different peripheral nerves, and are characteristic of a degenerative polyneuropathy due to primary peripheral axonal loss of large fibers and associated neurogenic muscle atrophy.
Acute Inflammatory Demyelinating Polyneuropathy
Case 5 (male) presented at 2.5 years of age with a 3-day history of regurgitation, vomiting, and anorexia. Lameness had been noted the previous week. Six months prior to this presentation for regurgitation, the dog had left-sided facial paralysis (suspected to be due to otitis media) that completely resolved. Over the next 5 days, the dog became lethargic and developed aspiration pneumonia. Left thoracic limb proprioceptive deficits progressed to reluctance to walk and a stiff gait with collapse, weakness, and hyperesthesia (touch). Routine hematology and serum biochemistry findings were unremarkable.
Case 6 (male) was 16 months old when presented with an acute onset of weight loss, masticatory muscle atrophy, failure of jaw closure, and proprioceptive deficits in the thoracic limbs. About 6 weeks prior to presentation, the dog had fractured the right radius and ulna requiring fixation with a dynamic compression plate but with no apparent complications. Hematology and biochemistry revealed mature neutrophilia (19.5 × 109 cells/l; reference interval 6–15 × 109 cells/l) and markedly elevated CK activity (3920 U/L; reference interval 50–230 U/L), suggestive of an acute inflammatory process and possible myositis. The clinical signs progressed rapidly despite symptomatic management with antibiotic, anti-inflammatory, and intravenous fluid therapy. Both dogs were euthanized having become painful, inappetent, recumbent, and nonresponsive.
The main gross finding in cases 5 and 6 was a markedly dilated and flaccid esophagus (up to 8 cm). There was distension of the urinary bladder, colon, and rectum, and severe suppurative bronchopneumonia. In addition, case 6 had bilateral atrophy of the masseter muscles. The repaired fracture of the right radius and ulna was stable, and the implant was firmly in position with no evidence of inflammation.
Microscopically, within the esophagus, the majority of myenteric plexi were markedly infiltrated by macrophages and lymphocytes (Fig. 6a–c), whereas neurons appeared to be spared. Multifocally, single to small groups of myofibers were variably sized with loss of striations and infrequent calcified or hyalinized cytoplasm. Overall, myofibers were markedly separated by clear spaces (edema) with some muscle fibers disrupted, necrotic, and infiltrated by macrophages. Occasionally myofibers had multiple central large vesicular nuclei suggestive of regeneration. Similar changes were observed within other skeletal muscle groups, including the cricoarytenoid, masseter, temporal, and cranial tibial muscles. The pattern of myofiber atrophy with mild regeneration and no myofiber grouping was consistent with denervation atrophy. In addition, in case 6, skeletal muscle showed mild multifocal infiltration of macrophages and fewer plasma cells into necrotic myofibers (mild myositis). Mild to marked inflammation of the autonomic and spinal ganglia was observed in both dogs. Severe diffuse infiltration of neutrophils into the cranial cervical and cervicothoracic ganglion was evident in case 6 (Fig. 7). Mild to moderate infiltration of mainly mononuclear cells, macrophages (CD18 positive), and about 20% T-lymphocytes (CD3 positive) was present in autonomic and spinal ganglia in case 5 and in the vagal, caudal mesenteric, and spinal ganglia in case 6. This inflammation was associated with severe loss of myelin seen as pale vacuolated thin eosinophilic fibers located between neurons, which were also decreased in number. Within the peripheral nerves, there was marked myelin loss and axonal swelling along with infiltration of neutrophils, macrophages, and lesser numbers of lymphocytes around blood vessels and within myelin sheaths (Fig. 8a, b). Semi-thin sections of a nerve root revealed scattered inappropriately thin myelinated fibers (Fig. 9) consistent with mild demyelinating polyneuropathy. Changes in the brain and spinal cord were not observed. The above-mentioned features are consistent with a ganglionitis, including myenteric plexi, with demyelination and secondary axonal degeneration. The lesions were mainly lymphohistiocytic in case 5 and predominantly suppurative in case 6. Neurogenic muscle atrophy was observed in various muscle groups suggesting a generalized neuritis with involvement of cranial nerves and other peripheral nerves. Neospora and Toxoplasma antigens were not detected in the ganglion or skeletal muscle of case 6.
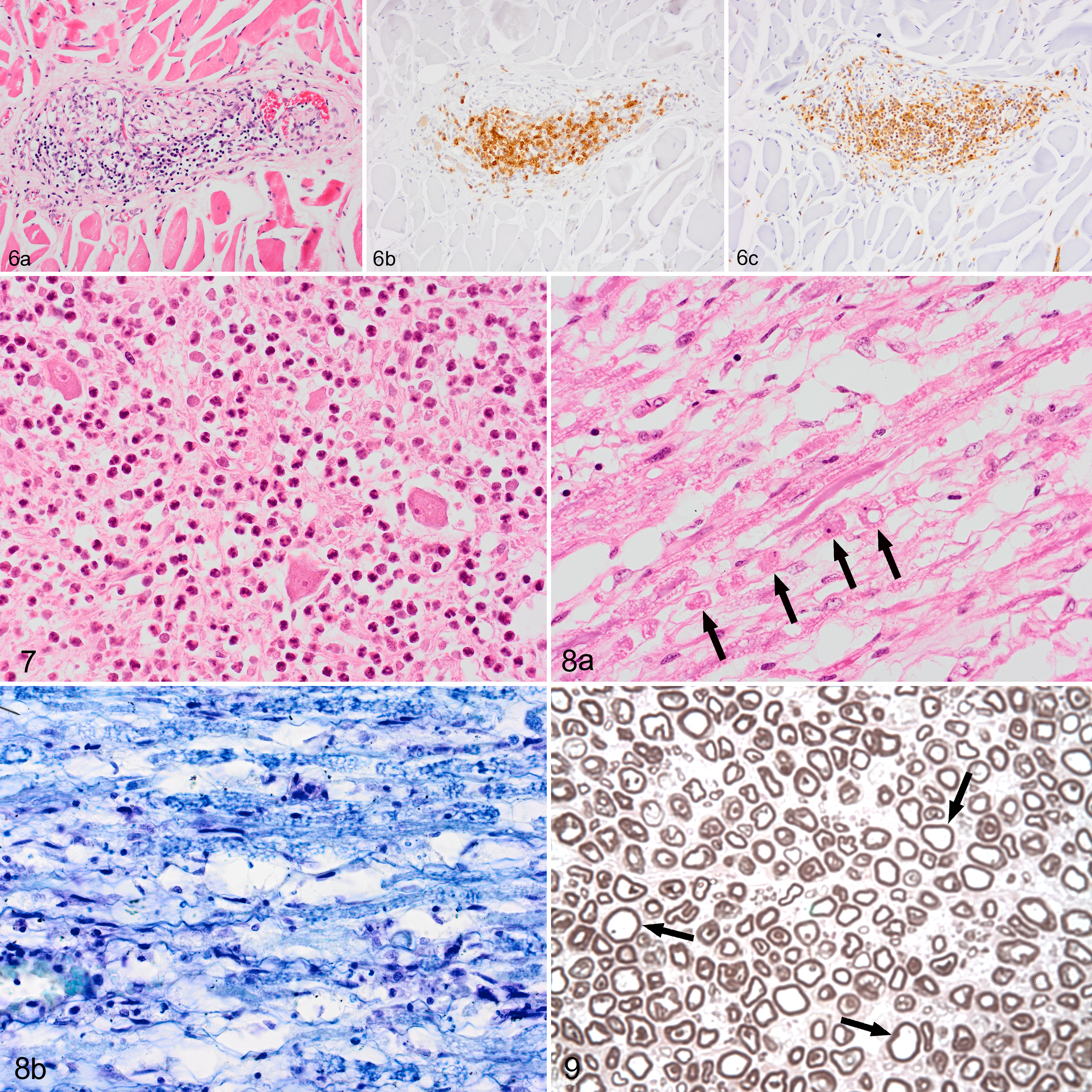
Acute inflammatory demyelinating neuropathy, Siberian husky dog, case 6.
Ganglioradiculitis (Sensory Polyneuronopathy)
Case 7, a female, was diagnosed with megaesophagus and mild ataxia at the age of 18 months. No further details on the clinical history were available. The dog was euthanized due to poor prognosis and submitted for postmortem examination.
The dog was in poor body condition and the only macroscopic finding was a diffusely dilated esophagus.
Microscopically the dorsal root ganglia were infiltrated by low to moderate numbers of lymphocytes, which formed multifocal nodules with a perivascular distribution and targeting neurons (Fig. 10a and b). There was mild neuronal loss. The sensory nerves had marked vacuolation of myelin sheaths, fragmentation of myelin, infiltration of macrophages, swollen axons, and axonal loss. Marked Wallerian degeneration in the dorsal funiculi along the spinal cord was characterized by dilation of myelin sheaths, marked myelin loss, loss or degeneration of axons, and infiltration of microglia with foamy cytoplasm. Wallerian degeneration was also observed in the descending tract of the trigeminal nerve. Mild infiltration of CD3-positive cells was also noted in the peripheral nerves. Skeletal muscle (triceps and gastrocnemius) samples were normal. The above features were characteristic of ganglioradiculitis (sensory neuronopathy).
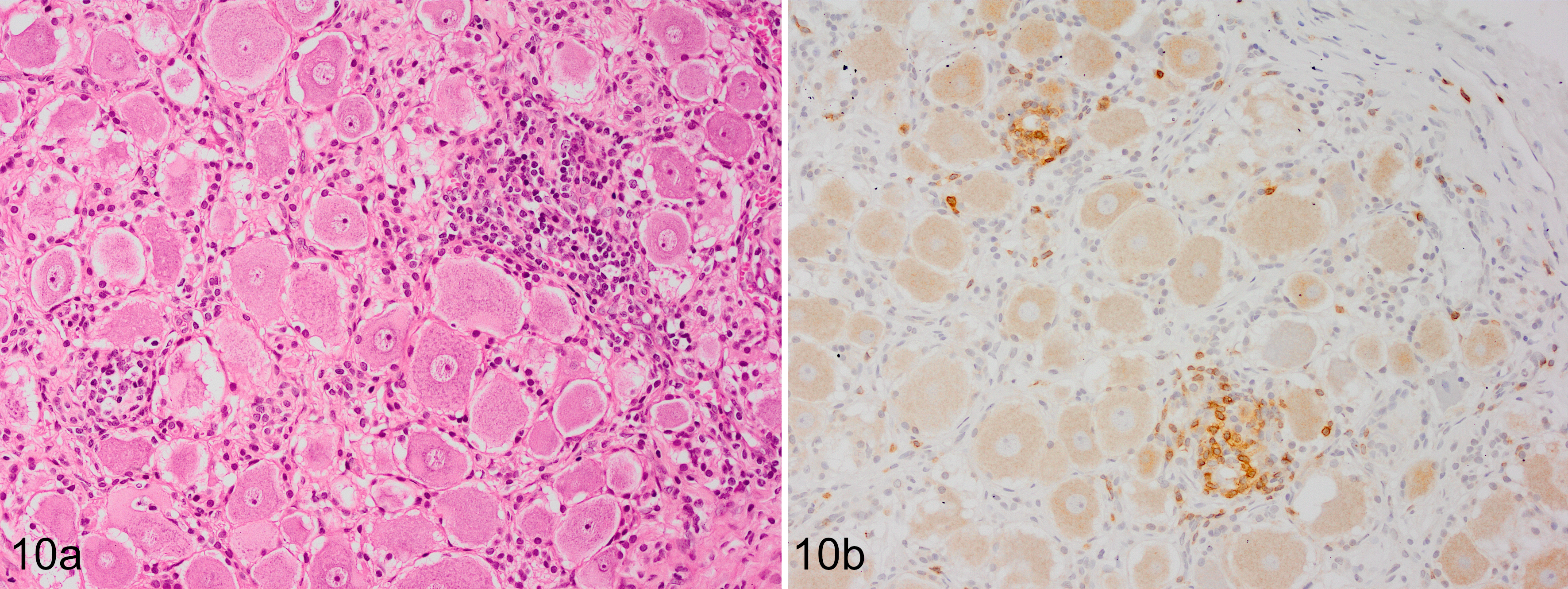
Sensory neuronopathy, dorsal root ganglion, Siberian Husky dog, case 7. Multifocal aggregates of inflammatory cells centered on neurons (a; HE) and composed of T lymphocytes (b; IHC for CD3).
Genotyping
None of the 4 dogs tested (cases 1 and 2 with primary axonal degeneration, cases 5 and 6 with acute inflammatory demyelinating neuropathy) carried any of the 5 known mutations.
Discussion
The Siberian husky dogs in our study were classified into 2 groups distinguished by the underlying pathological process. The degenerative polyneuropathy group had mainly an early age of onset, a relatively slow disease progression, and primarily loss of large-caliber nerve fibers resulting from axonal degeneration. In addition, there was no evidence of myasthenia gravis, endocrinopathy, neoplasia, infection, or immune-mediated disease in any of the dogs in this group and there was no history of toxin or drug ingestion which might have triggered the axonal loss. 11 All these features and the fact that related dogs were affected would be consistent with canine inherited neuropathy, which has not been previously described in the Siberian husky.
To date, inherited polyneuropathy has been reported in 22 dog breeds. 23 Specifically, inherited axonal neuropathies with laryngeal paralysis and megaesophagus as observed here have only been described in Dalmatians, 5 white German Shepherd dogs, 40 and Pyrenean mountain dogs. 20 While a hereditary form of laryngeal paralysis has been previously reported in Siberian huskies and husky cross bred dogs, 35,37 this is a different disease to the phenotype reported here and has pathological changes in the CNS.
Two of the Siberian huskies described here (cases 1 and 2) were bred in Ireland and had laryngeal paralysis and megaesophagus as the predominant clinical sign. One of these dogs (case 2) survived more than 7 years, suggesting a reasonably good prognosis with management of the megaesophagus and regurgitation. Case 2 developed systemic amyloidosis, which was interpreted as the reactive type caused by systemic AA amyloid rather than directly linked to the polyneuropathy. This was based on the absence of amyloid depositions in endo- and epineurium, early onset, slow progression, and long survival of the polyneuropathy and the similarities with case 1, where amyloid was not detected. Furthermore, in domestic animals the reactive type of amyloidosis, that is, secondary to chronic suppurative or granulomatous lesions in other organs is most commonly seen. 10 In the present case, it may have been associated with chronic suppurative aspiration pneumonia subsequent to regurgitation. This dog showed mild proprioceptive deficits in the pelvic limbs at the age of 4 years, which has been reported in other dog breeds with laryngeal paralysis–polyneuropathy complex. 5,18,21,28,44 Since laryngeal paralysis and megaesophagus occurred simultaneously in these cases, it is likely that initial focal damage occurred to both the recurrent laryngeal and vagal nerves, 26,33 the 2 longest peripheral nerves in the dog. 33
One of the Siberian huskies (case 1) had a littermate that exhibited the same clinical signs and there were anecdotal reports of additional littermates being affected, suggesting that the disease might have a genetic basis. Interestingly, during the 18 months when these cases presented, 2 additional young Siberian huskies were diagnosed with laryngeal paralysis and megaesophagus but were lost to follow-up and the pedigree could not be established. No further cases have been reported prior to this time or since.
The other 2 young related Siberian huskies (case 3 and 4) were bred in the United States and had initial presenting clinical signs of pelvic limb paresis that later progressed to generalized polyneuropathy without laryngeal paralysis or megaesophagus. This presentation would be more similar to polyneuropathy in breeds such as the Alaskan Malamute 6 and Rottweiler dogs. 7
Several dog breeds, for example, Alaskan Malamute, Rottweiler, Leonberger, German Shepherd, and Golden Retriever, may present with more than one type of inherited polyneuropathy, 23 which appears to be the case in Siberian huskies. These polyneuropathies may be associated with one single mutation causing all disease manifestations as seen in Alaskan malamutes, 9 or with different single mutations causing a percentage of cases as reported in Leonberger. 2,17 The Siberian huskies in the current study were bred in 2 different countries, and although the pedigree is not known, it is unlikely that they had common ancestors. Furthermore, although the underlying nerve lesions were similar, the clinical presentations differed.
In dogs, similarities have been recognized with inherited neuropathies in humans collectively known as Charcot-Marie-Tooth disease (CMT). 8 In the last few years, specific genetic mutations that are known to cause certain types of CMT have been identified in the greyhound, 14 Alaskan malamute, 9 Leonberger, and St Bernard. 2,17 In addition, mutations in the RAB3GAP1 gene (Russian Terrier, Rottweiler and Alaskan husky), which has been associated with the Warburg micro syndrome 1 in humans, were reported. 29,30,43 None of these 5 mutations were found in 2 Siberian huskies with degenerative polyneuropathy. The 2 related dogs from the United States tested negative for the Alaskan Malamute mutation. 9 This is not necessarily surprising, since in humans there are more than 80 different genetic mutations that have been implicated in the development of the various forms of CMT. 8 In addition to this, a variety of different mutations have been identified in affected dog breeds. 2,9,14,17 While polyneuropathy in Siberian huskies appears to be uncommon, it is a variably severe disease in individual animals and future cases should be investigated for underlying genetic mutations to aid in breeding programs.
Previously it has been suggested that the Siberian husky may be predisposed to polyneuropathy, as it was overrepresented in 2 small case studies on sensory neuropathy in dogs. 12,45 Therefore, 3 cases of inflammatory polyneuropathy in Siberian huskies were included in this case series especially as similar clinical signs of multiple peripheral nerve deficits and megaesophagus were observed. The cause of megaesophagus, which can result from disruption of peripheral nerve pathways at the level of the myenteric plexus, the vagal nerve, and/or the sensory and autonomic ganglia in the cervicothoracic region, 26 could be established in all 3 cases, by the marked inflammation seen in myenteric plexi, autonomic or sensory ganglia. These dogs with inflammatory polyneuropathy had later disease onset (16–30 months) and rapid disease progression.
Two cases were diagnosed as acute polyganglioneuritis with marked demyelination and secondary axonal loss. The severe acute ganglioneuritis affected mainly the cranial nerves, cervical ganglia (and mild changes in mesenteric ganglia), and esophageal myenteric plexi, resulting in clinical signs indicative of sensory (painful to touch), motor (muscle atrophy) and autonomic (megaesophagus, distended colon and urinary bladder) dysfunction. These findings resemble acute inflammatory demyelinating polyneuropathy a form of Guillain-Barré syndrome in humans, which often involves cranial nerves and autonomic ganglia. 44 Guillain-Barré syndrome is an aberrant autoimmune response targeting peripheral nerves and their spinal roots. 44 Complement-fixing antibodies, macrophages, and T cells are the main effector mechanisms in Guillain-Barré syndrome 13 and are thought mainly to be triggered by recent infections, but also potentially by recent trauma or surgery. 22,46 It is of interest that in the present cases, one dog had recently had surgery for a fractured radius and ulna prior to onset of disease.
While similar areas of the peripheral nervous system were affected in the 2 cases of acute inflammatory demyelinating polyneuropathy reported here, they differed in the severity and nature of the inflammatory infiltrate. A severe neutrophil infiltrate was observed in one of the cases which would be unusual even in the acute phase of the disease and has rarely been reported in Guillain-Barré syndrome in humans. 3,13,39 It has been suggested in a peracute course of disease in acute polyradiculoneuritis in dogs. 11 However, no infectious agent was found and given the specific location and the targeting of myelin an underlying autoimmune disease is likely. In autoimmune diseases, neutrophils can be activated by immune complexes and complement and may play a role in the release of autoantigens and the effector phase. 32 A mild myositis was observed which may be a comorbidity with the fulminant neuritis. Clinical signs progressed rapidly in both dogs.
The acute inflammatory demyelinating polyneuropathy in the Siberian huskies reported here differs markedly from acute polyradiculoneuritis, which is characterized by progressive tetraparesis 11 with inflammatory changes mainly in the dorsal and ventral root ganglia; 41 it is the most common acute inflammatory neuropathy recognized in dogs 12,24,44 and is an animal model for Guillain-Barré syndrome. 24 Guillain-Barré syndrome can present as several different syndromes or variants, relating to the main focus of the immune attack in a given patient, for example, motor or sensory nerve fibers, and whether it is predominantly affecting myelin or the axon itself. 13 This may also be the case in dogs, as cases of acute inflammatory neuropathies are reported that primarily target locations other than ventral nerve roots, for example, the cranial nerves. 19,28,36
Sensory neuropathy 11,16 or sensory neuronopathy 38,45 was another inflammatory polyneuropathy diagnosed in one of the Siberian huskies in the present study, as only sensory nerves were affected and no muscle atrophy was observed. 11,38 The inflammatory infiltrate mainly consisted of T-lymphocytes and macrophages targeting neurons in the dorsal root ganglia and forming perivascular aggregates 16,41 as in the current case. Neuronophagia in the dorsal root ganglia leads to axonal loss and secondary myelin degeneration 11,38 with retrograde extension along the dorsal funiculi of the spinal cord and into the spinal tract of the trigeminal nerve. 16,41 This is thought to be an immune-mediated destruction of neurons due to type IV cell-mediated cytotoxicity mediated by CD8-positive T cells or antibody. 38
A breed predisposition for inflammatory polyneuropathy in Siberian huskies was perceived, as during the same study period only 2 dogs of different breeds were diagnosed postmortem with sensory polyneuronopathy and ganglionitis at the University Veterinary Hospital, Dublin. However, this is unconfirmed because of the small number of cases and absence of pedigree information. In humans, no distinct underlying genetic predisposition for autoimmune neuroinflammatory disease has been found. 4,25 Nevertheless, 2 Siberian huskies with acute inflammatory demyelinating polyneuropathy were tested for the 5 different genetic mutations causing degenerative forms of polyneuropathy and none were found.
In this case series the clinicopathological features were used to categorize polyneuropathies in young Siberian husky dogs and confirmed a degenerative and possibly inherited polyneuropathy in this breed. Furthermore, inflammatory polyneuropathies with unique lesions were identified in Siberian huskies, highlighting the importance of histopathological characterization to provide clinical and prognostic information. The sampling of autonomic ganglia and esophageal muscle at postmortem examination in these dogs proved essential in diagnosing some of the inflammatory conditions.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Jahns_et_al - Polyneuropathy in Young Siberian Huskies Caused by Degenerative and Inflammatory Diseases
Supplemental Material, Combined_supplemental_materials-Jahns_et_al for Polyneuropathy in Young Siberian Huskies Caused by Degenerative and Inflammatory Diseases by Hanne Jahns, Karen M. Vernau, Catherine M. Nolan, Emma J. O’Neill, Robert E. Shiel and G. Diane Shelton in Veterinary Pathology
Footnotes
Acknowledgements
Technical assistance was given by Brian Cloak, University College Dublin, School of Veterinary Medicine. We would like to thank Sarah McCormack, Caitlin Dalla Pria, and Jasmine Mengers, University College Dublin, School of Biology and Environmental Science, for running the PCRs. IHC labelling was conducted by Veterinary Pathology, Institute of Veterinary Science, University of Liverpool, UK. Thanks to the veterinary practitioners for referring these cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant (SF976) from the Seed Funding Scheme of University College Dublin, Ireland.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
