Abstract
Peripheral neuropathies are common sequelae to human immunodeficiency virus (HIV) infection in humans and are due to a variety of mechanisms, including direct antiretroviral toxicity, HIV-mediated damage, immune-mediated disorders, and opportunistic viral infections. Rhesus macaques (
Keywords
Retroviral-associated neuropathies in humans, most notably those due to infection with human immunodeficiency virus (HIV) or human T-lymphotropic virus type 1 (HTLV-1), are significant causes of morbidity and mortality in immunosuppressed patients. Although there has been significant improvement in the quality of life and longevity of patients infected with HIV, neruologic complications remain one of the most important chronic concerns in infected individuals. 14 HIV-associated neuropathy may be mediated by (a) antiretroviral drug toxicity, (b) direct infection with the HIV virus, (c) immune-mediated mechanisms, or (d) opportunistic infections such as cytomegalovirus (CMV). 5,12 The pathogenesis of direct HIV-induced neuropathy involves both virally infected and uninfected macrophages that, through a variety of mechanisms, cause damage to the nerve, nerve roots, and ganglia, whereas the pathogenesis associated with recrudescent CMV infection is less understood.
Human cytomegalovirus (HCMV) is a ubiquitous betaherpesvirus with adult seroprevalence rates of 50% to >90%. 6 Although primary infection is mostly subclinical in the immunocompetent host, HCMV reactivation is the most important opportunistic viral infection in immunocompromised HIV-infected patients with AIDS, and it accounts for significant morbidity and mortality. Almost 100% of patients seropositive for HIV are coinfected with HCMV. 9 HCMV causes multiorgan pathology in the infected individuals, including in the central nervous system (CNS) and peripheral nervous system (PNS). Different patterns of HCMV-related lesions of the nervous system have been recognized and include (a) polyradiculopathy of the lower spinal roots and cauda equina; (b) multifocal neuropathy involving spinal roots, nerve trunks, and occasionally the cranial nerves; (c) encephalitis; and (d) cerebrospinal fluid abnormalities, including high protein content and pleocytosis with reactive polymorphonuclear leukocytes. 13 HCMV can typically be localized to Schwann cells, endoneurial fibroblasts, and macrophage-lineage cells within and around the affected region. 1,9 Among the most common manifestations of HCMV disease in HIV-infected patients is retinitis, which accounts for a high percentage of all cases of HCMV clinical syndromes. 10
Rhesus cytomegalovirus (RhCMV; macacine herpesvirus 3) is a similarly common opportunistic infection in simian immunodeficiency virus (SIV)–infected rhesus macaques with a disease spectrum that simulates its HCMV counterpart. 3,15 The RhCMV animal model of HCMV disease has aided the understanding of viral biology, disease pathogenesis, and vaccine development. 2 RhCMV infection in adult rhesus macaque colonies nears 100%, closely paralleling the situation in humans. 15 In immunosuppressed macaques, common RhCMV-associated disease manifestations include interstitial pneumonia, encephalitis, gastroenteritis, and lymphadenitis. 2,4 The degree to which RhCMV causes neuritis and neuropathies in immunosuppressed macaques has not been explored; however, there is brief mention of RhCMV-associated retinitis in the literature. 8
Cytomegalovirus remains one of the most important viral opportunistic infections in HIV-infected individuals and continues to be a common cause of morbidity and mortality. Although there are several animal models of cytomegalovirus infection, the SIV-infected macaque offers a reproducible model that illustrates the pathologic consequences of RhCMV recrudescence. Herein we characterize the prevalence and histopathologic and immunohistochemical findings of RhCMV-associated facial neuritis and demonstrate that peripheral nerve damage and myelin loss associated with RhCMV recrudescence are secondary to inflammation rather than a direct infection of Schwann cells.
Methods
Necropsy records at the New England Primate Research Center (NEPRC) were searched from 1997 to 2012 for cases of RhCMV reactivation in animals coinfected with RhCMV and SIV. This search yielded 1206 SIV-infected rhesus macaques, of which there were 115 cases of recognized RhCMV infection. All animals included in this study were cared for in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals (eighth edition, 2011) and the standards of the Harvard Medical School Standing Committee on Animals and the Association for the Assessment and Accreditation of Laboratory Animal Care. All animals were necropsied within 24 hours of death, and representative sections of all major organs were collected and fixed in 10% neutral buffered formalin for 1 week prior to paraffin embedding, with 5-μm sections of representative samples routinely prepared and stained with hematoxylin and eosin (HE) and used for microscopic examination. Additional sections of the affected tissues were stained with the Luxol fast blue method and Bielschowsky silver stain.
To characterize the facial neuritis using immunohistochemical methods, we used standard immunoperoxidase staining for RhCMV, CD68 (macrophage marker), vimentin (mesenchymal marker), 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase; Schwann cell marker), and myelin basic protein (MBP; myelin marker). To rule out other potential etiologies of neuritis and oral ulcer in rhesus macaques, we stained additional sections for simian immunodeficiency virus nef protein (SIVnef) and herpes simplex virus, type 1 (HSV). Formalin-fixed, paraffin-embedded sections were deparaffinized, rehydrated, and subsequently blocked with hydrogen peroxide. Pretreatment for all RhCMV, SIVnef, CNPase, MBP, and CD68 involved microwaving for 20 minutes in 0.01M citrate buffer followed by 20 minutes of cooling. Pretreatment for vimentin involved 5 minutes of proteinase K digestion. There was no pretreatment for HSV. All steps were followed by a Tris-buffered saline (TBS) wash. Prior to application of primary antibodies, all slides were treated with Dako (Carpinteria, CA) protein block for 10 minutes. Sections were incubated with anti–human CNPase (Thermo Scientific [Billerica, MA], polyclonal, 1:200, 60 minutes at room temperature), anti–human CD68 (Dako, monoclonal, 1:410, overnight in refrigerator), anti–human myelin basic protein (Dako, polyclonal, 1:1047, 30 minutes at room temperature), anti–human vimentin (Dako, monoclonal, 1:162, overnight in refrigerator), anti–rhesus SIVnef (clone KK75, gift from Dr K. Kent, NIBSC Centralized Facility for AIDS Reagents, monoclonal, 1:200, overnight in refrigerator), anti–human HSV (Dako, polyclonal, 1:870, 30 minutes at room temperature), and anti–RhCMV IE1 (gift from Peter A. Barry, UC Davis, polyclonal, 1:1600, 30 minutes at room temperature). Slides were then incubated with secondary antibody biotinylated goat anti–rabbit (Vector Laboratories [Burlingame, CA], 1:200, 30 minutes at room temperature) for RhCMV, HSV, and MBP and biotinylated horse anti–mouse (Vector Laboratories, 1:200, 30 minutes at room temperature) for vimentin, SIVnef, CNPase, and CD68. This was followed by a 30-minute incubation at room temperature with Vectastain ABC Elite (Vector Laboratories) (MBP, SIVnef, HSV, and CNPase) or Vectastain ABC standard (Vector Laboratories) (RhCMV, vimentin, CD68). All slides were developed with DAB chromagen (Dako) and counterstained with Mayer’s hematoxylin. In all cases, step sections were incubated with isotype-specific irrelevant antibodies for negative controls. Positive controls consisted of sections of spleen (CD68), brain (MBP, CNPase), small intestine (vimentin), SIV encephalitis (SIVnef), B virus cheilitis (HSV), and RhCMV enteritis (RhCMV IE1).
Colocalization and cellular tropism of RhCMV in facial neuritis was achieved by double immunohistochemistry experiments for CNPase/RhCMV, CD68/RhCMV, and vimentin/RhCMV. All sections were treated with the first antibody in the same fashion as described above. Following treatment with the first antibody, sections were washed in TBS and received a second biotin block. Sections were then incubated with Universal Block (KPL, Gaithersburg, MD) for 30 minutes at room temperature. Prior to application of the second antibody, all sections were treated with Dako protein block for 10 minutes. Sections were incubated with anti-RhCMV IE1 (1:1600, 30 minutes at room temperature). Slides were then incubated with secondary antibody biotinylated goat anti–rabbit (1:200, 30 minutes at room temperature), followed by a 30-minute room temperature incubation with Vectastain ABC Alkaline Phosphatase (Vector Laboratories). All sections were developed with Vector Red (Vector Laboratories) and counterstained with Mayer’s hematoxylin. In all cases, step sections were incubated with isotype-specific irrelevant antibodies for negative controls. Positive controls were the same as noted above. Double-labeled experimental slides were examined using a Leica microsystems DMR light microscope (Leica Microsystems CMS GmbH, Wetzlar, Germany) equipped with a spectral imaging camera and software (Nuance Multispectral Imaging System, PerkinElmer, Waltham, MA). A colocalization threshold algorithm was subsequently determined to account for cytoplasmic expression of vimentin, CD68, and CNPase and cytoplasmic and nuclear expression of RhCMV. The presence/absence of colocalization was determined via a colorimetric analysis of images from each case and further corroborated by visual assessment. Individual cells were highlighted in the colocalization software, and colocalization was only considered accurate if the cells showed greater than 20% colocalization via image analysis on a specific cell using percent area of colocalization.
Results
Clinical and Gross Pathology Findings
RhCMV recrudescence was identified in 115 cases (9.5% prevalence) in archived tissues of 1206 SIV-infected rhesus macaques from 1997 to 2012. Confirmed RhCMV cases were further analyzed for the presence of facial neuritis, of which 10 were found (8.7% prevalence) (Suppl. Table S1). Of these 10 animals, 7 had been inoculated with SIVmac239 and 3 had been inoculated with SIVmac251. All animals presented with cachexia and weight loss with varying degrees of gastroenteritis and respiratory disease consistent with prolonged immunosuppression and progression to simian AIDS. Gross lesions were present in 1 animal and consisted of a large labial ulcer (Fig. 1).
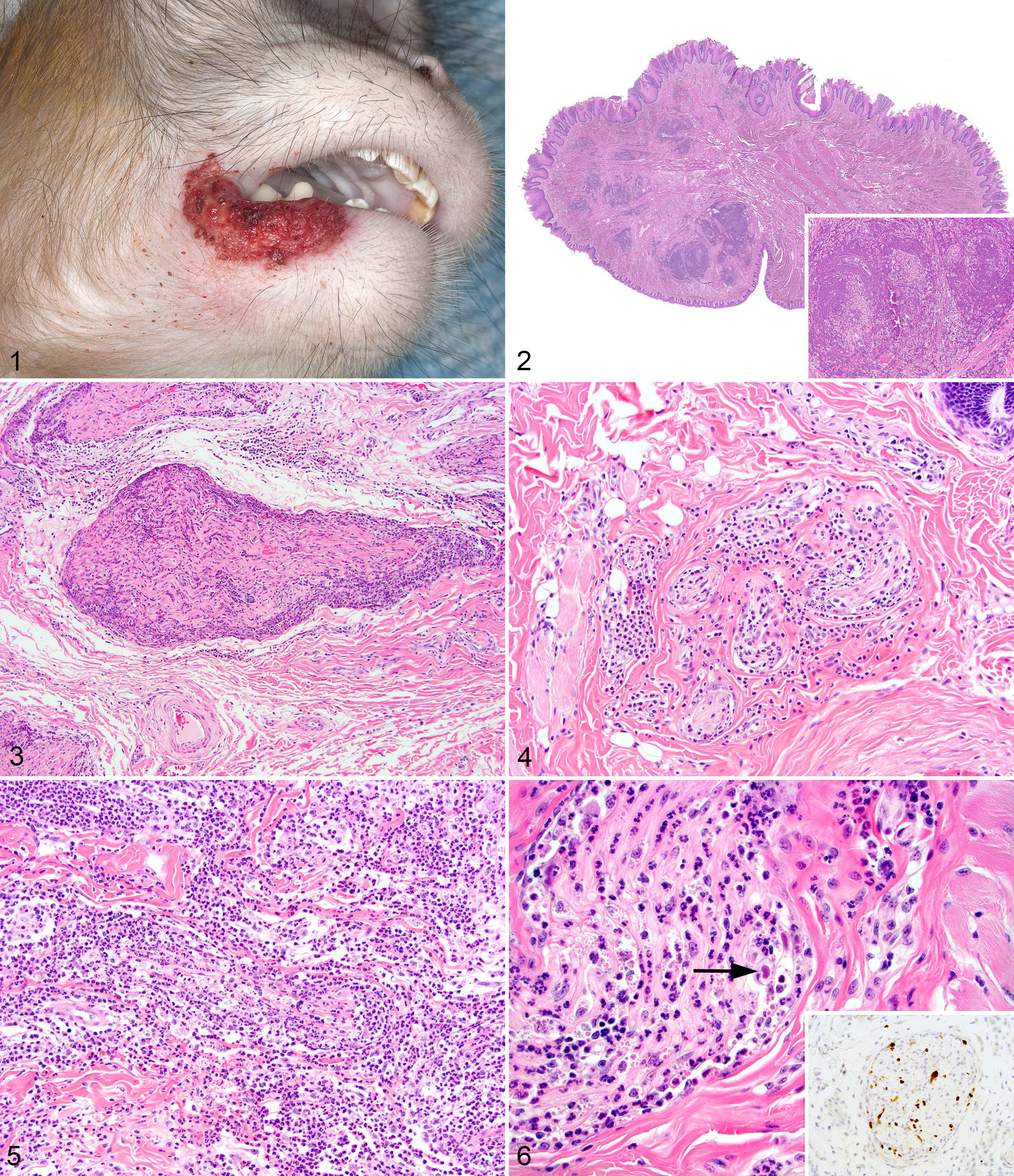
Histologic Findings
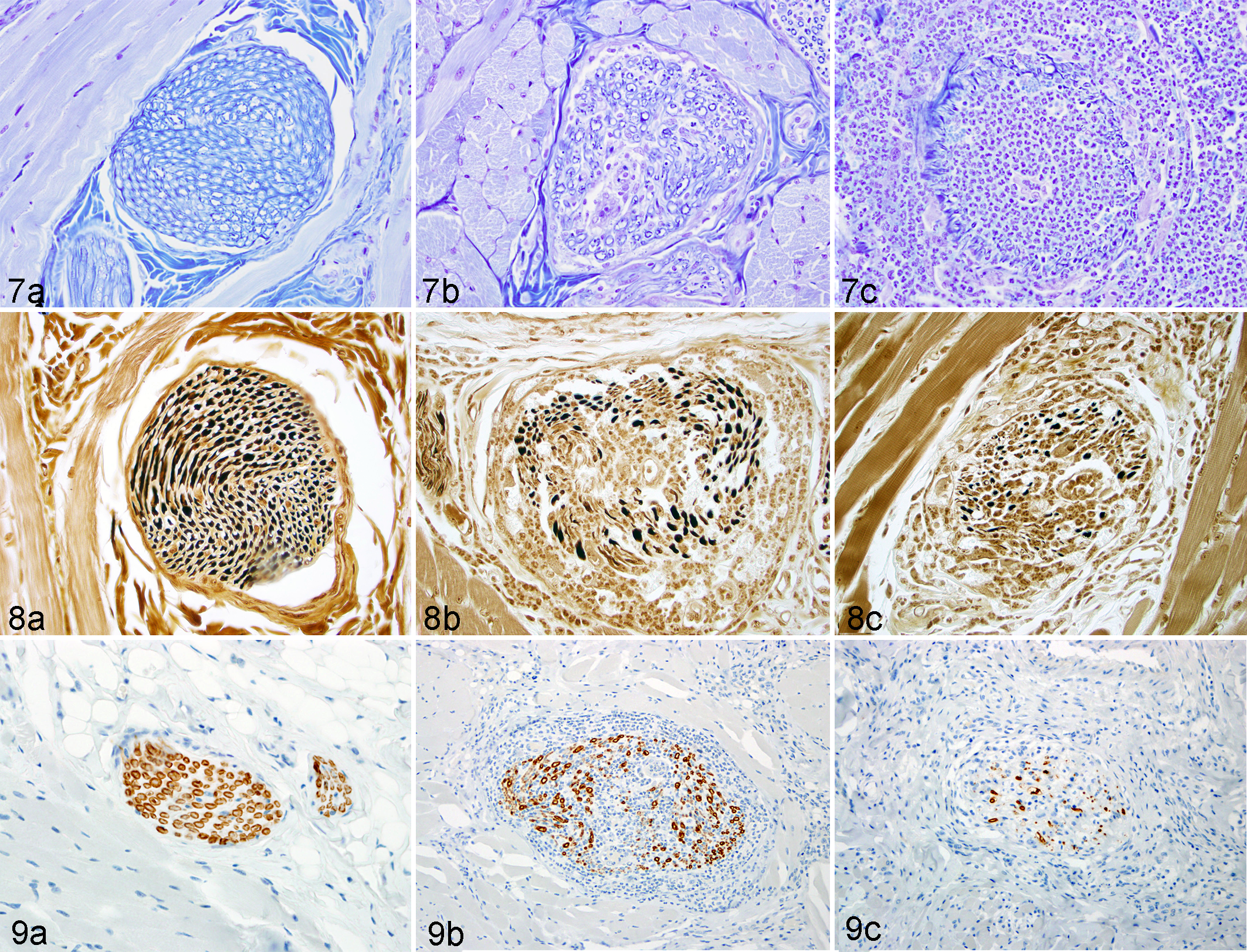
The distribution of histologic lesions is listed in Supplemental Table S1. Based on the location of histologic lesions, portions of the trigeminal and facial nerves were predominantly affected. The histologic findings were similar in all cases and were characterized by varying degrees of perineurial and intraneurial neutrophil and macrophage-rich inflammation that variably replaced and obscured normal nerve architecture (Figs. 2–6). The inflammation typically involved multiple nerves of varying sizes. There were frequent cytomegalic cells that occasionally contained a large, intranuclear, round to ovoid 10- to 15-μm diameter inclusion body (Fig. 6). The inflammation variably spread into the adjacent connective tissue and deep musculature of the tongue, lip, and face. The most severe cases had concurrent myodegeneration and necrosis with variable fibrosis and edema. The perineurium was either absent or partially breached by the inflammatory cells, and axons were often difficult to discern. Severely affected nerves often had only small remnants of myelin within the nerve. Case No. 9 had a large ulcer on the lip that was covered by a sheet of hemorrhage, fibrin, and bacterial cocci. Multiple animals had systemic RhCMV infection (Suppl. Table S1), including 2 animals that had involvement of other nerves (cauda equina and sciatic nerve). Luxol fast blue staining of the affected sections revealed loss and fragmentation of myelin and disruption of axons with infiltration by neutrophils and macrophages (Fig. 7a–c). Silver staining of the affected sections revealed loss of axons that correlated directly with the severity of inflammation and the loss of myelin (Fig. 8a–c).
All animals in this report were enrolled in SIV pathogenesis studies and had progressed to simian AIDS. Consistent with simian AIDS, all had concurrent opportunistic infections that contributed to morbidity:
Immunohistochemical Findings
Cytomegalovirus staining revealed numerous immunoreactive cytomegalic cells admixed within the damaged nerves and scattered throughout the adjacent tissue (Fig. 6, inset). Similarly large numbers of CMV-immunopositive neutrophils with nuclear staining were noted in these regions. Staining for HSV in the animal with the lip ulcer failed to reveal any antigen and ruled out HSV as the cause of the neuritis or lip ulcer. None of the animals had SIV-immunoreactive cells within the lesions. Myelin basic protein (MBP) staining paralleled the Luxol fast blue staining with varying severity of nerve damage and loss (Fig. 9a–c). Nerves that were deemed to have only minor inflammation on the histologic examination typically had more myelin loss and fragmentation than expected.
All affected sections were studied using double immunohistochemical staining to determine the cellular tropism of RhCMV-infected nerves. Double staining for RhCMV and vimentin confirmed the presence of numerous RhCMV-infected cells that coexpressed vimentin, indicating infection of cells with a mesenchymal origin. To further define the predominant RhCMV-infected mesenchymal cell population, double immunostaining for CMV and CD68 was performed. Using both visual assessment and colocalization image analysis, RhCMV predominantly colocalized within macrophages both within the affected nerve and in the adjacent tissue (Fig. 10a,b). Double immunostaining for RhCMV and CNPase failed to reveal colocalization of cytomegalovirus within Schwann cells or myelinated nerves both via visual assessment and use of colocalization imaging software. Although rare scattered double-labeled areas were noted in the RhCMV/CNPase staining, the percentage of colocalization was less than 5% and considered incidental (Fig. 11a,b).
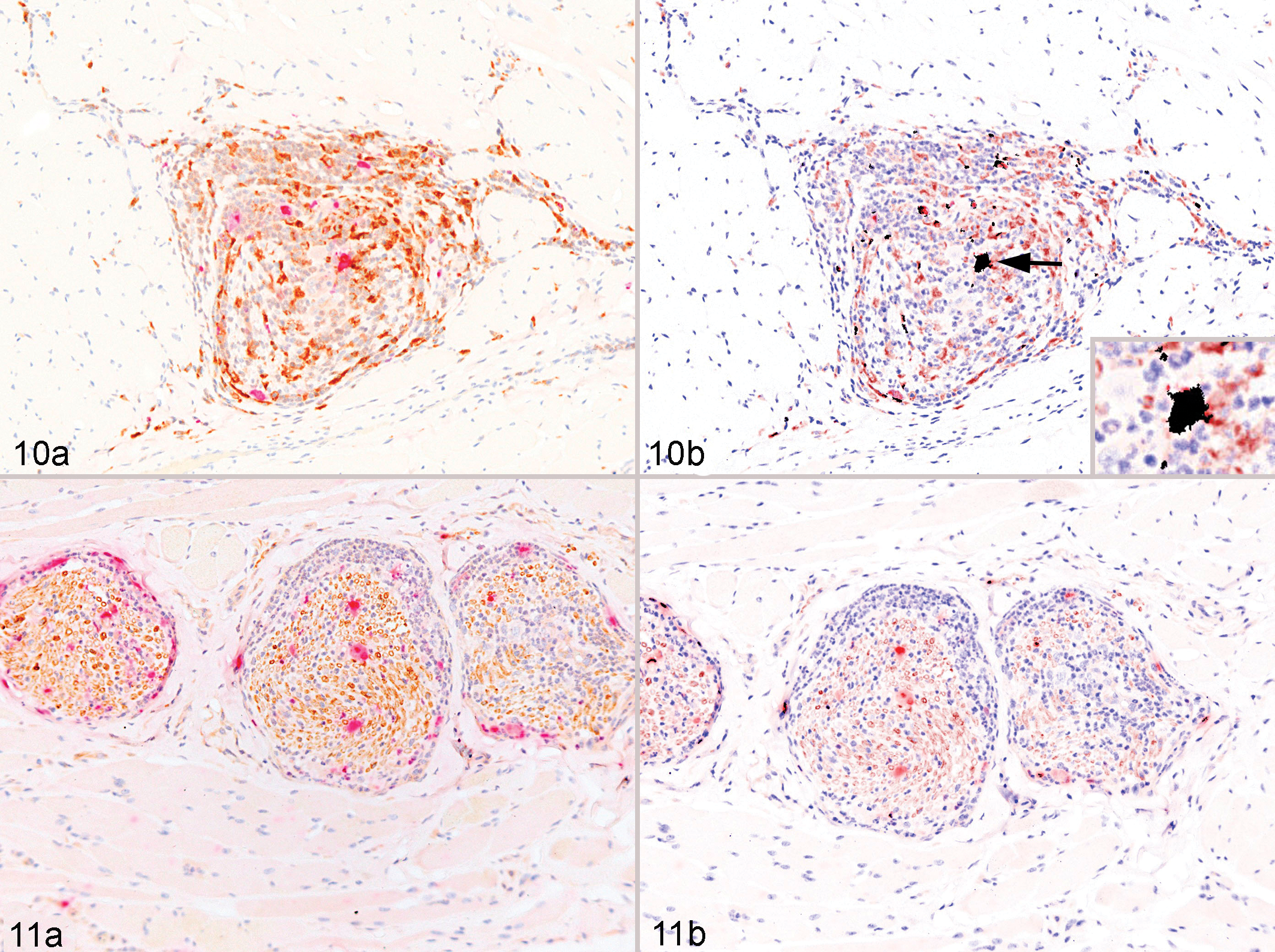
Discussion
In humans, polyradiculoneuritis is a common sequela to HIV infection. HIV, HSV, and HCMV have been implicated in the pathogenesis of the nerve lesions. 7,9 HCMV and RhCMV are ubiquitous viral infections of humans and nonhuman primates, respectively, that target a broad range of host cells and cause extensive lesions. In the absence of immunosuppression, infection is limited by cellular immune response that controls viral replication and results in viral latency within the infected host. Upon immunosuppression, as in the case of AIDS and transplantation, the virus reactivates in the absence of adequate cellular immunity and manifests in a systemic and often fatal infection. HCMV has tropism to the majority of organs within the body, including the central and peripheral nervous systems. Features of HCMV-associated polyradiculoneuritis include inflammatory lesions within peripheral nerves that are most common in facial nerves and consist of obliterative lesions composed of neutrophil and macrophage-rich inflammation admixed with numerous inclusion bodies. 11,12 In human patients with AIDS, HCMV retinitis and subsequent blindness are considered the most common histopathologic and clinical sequelae of HCMV reactivation; however, the propensity of RhCMV to cause lesions in the peripheral nervous system is not known. 10
In this report, we demonstrate the prevalence of RhCMV-associated facial neuritis in 8.7% of animals that had systemic RhCMV infection. Lesions affected multiple nerves within facial tissues, including the tongue, lacrimal gland, salivary gland, and deep soft tissue of the face. Histologically, typical RhCMV-associated lesions were observed and consisted of infiltration of nerves by viable neutrophils and macrophages admixed with variable amounts of necrotic cellular debris and multiple cells bearing intranuclear inclusion bodies. 2 It is noteworthy that nerves within the tongue and the lacrimal glands were overrepresented and may indicate a route for virus exit through lacrimal and salivary glands, potentially aiding in virus shedding; however, systemic collection of facial nerves is required to confirm the significance of such observation. Although severe lesions effaced the entire architecture of the nerve, milder lesions were often localized to the periphery and were accompanied by loss of myelin that was best appreciated on the Luxol fast blue stain and myelin basic protein immunohistochemistry. Parallel lesions were noted in the axons in which there was axonal damage and loss that was most significant in the nerves with the most robust inflammation. These lesions were indicative of a secondary demyelinating process in which the inflammation was the cause of the progressive myelin damage and loss of axons. The neutrophilic nature of RhCMV and HCMV is unique in that the virus encodes its own CXC chemokine within the genomic UL/b′ region responsible for neutrophil chemoattraction. 2
Unlike the case for HCMV, we were unable to conclusively identify RhCMV-infected Schwann cells by IHC colocalization studies. Therefore, the myelin loss observed in the examined sections is likely a bystander effect secondary to the inflammation that disrupted and damaged the myelin sheaths and subsequently the axons. Whether the discrepancy between HCMV and RhCMV tropism to Schwann cells represents a deviation in the pathogenesis of the 2 related viruses requires further investigation. 9 Although it is not known whether RhCMV in facial nerves arises from local reservoirs of latent virus or is spread to the facial nerves from distant tissues, the pattern of histologic damage in the cases presented in this study suggests that RhCMV-infected macrophages and infiltrating neutrophils initially invade into the nerves from the adjacent interstitial tissues and results in inflammation-driven nerve damage to the axons, myelin, and Schwann cells. This sequential mode of infection explains the progressive loss of nerve architecture.
While SIV-induced polyneuropathy is well characterized, the lesions are fundamentally different, with ganglioneuritis and infiltration of activated macrophages into dorsal root ganglia being key features. 5 In addition, there was no evidence of SIV immunoreactivity in the cases presented herein, and the presence of RhCMV-infected cells directly within the lesions implicates RhCMV as the etiologic agent. Seven of 10 animals in the current study had disseminated RhCMV disease, and 2 animals had lesions in other nerves (cauda equina and sciatic nerve). Lesions within the nerves of the cauda equina are also quite common in humans infected with HCMV and may indicate a broader neurotropism of the virus than previously recognized; however, systemic collection of cranial nerves that supply structures in the face and their ganglia is required to determine if this is the case in rhesus macaques. Indeed, it is known that RhCMV often causes meningitis and can track along the nerve roots as they exit the spinal cord. 15
Clinical neurologic manifestations of RhCMV are not fully characterized in the rhesus macaque and are usually restricted to lesions attributed to either brain or spinal cord meningitis. 15 One animal presented with lip and buccal ulceration, which was negative for HSV by immunohistochemistry and was most likely related to the pain and/or desensitization associated with the neuritis and secondary self-trauma. The identification of RhCMV within the ulcer and nerves supplying salivary and lacrimal glands may suggest a significant role of facial neuritis in the natural history of RhCMV shedding and transmission. There is striking similarity between RhCMV and HCMV in clinical manifestations and pathologic features, supporting this as an animal model for CMV-associated neuropathies in immunosuppressed patients.
Footnotes
Acknowledgment
We thank Kristen Toohey for assistance with photography and Elizabeth Curran for tissue procurement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded, in part, by NIH grants NEPRC Base grant OD0111103 and T32 OD011064.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
