Abstract
In the small intestine, localized innate mucosal immunity is critical for intestinal homeostasis. Porcine epidemic diarrhea virus (PEDV) infection induces villus injury and impairs digestive function. Moreover, the infection might comprise localized innate mucosal immunity. This study investigated specific enterocyte subtypes and innate immune components of weaned pigs during PEDV infection. Four-week-old pigs were orally inoculated with PEDV IN19338 strain (n = 40) or sham-inoculated (n = 24). At day post inoculation (DPI) 2, 4, and 6, lysozyme expression in Paneth cells, cellular density of villous and Peyer’s patch microfold (M) cells, and the expression of polymeric immunoglobulin receptor (pIgR) were assessed in the jejunum and ileum by immunohistochemistry, and interleukin (IL)-1β and tumor necrosis factor (TNF)-α were measured in the jejunum by ELISA. PEDV infection led to a decrease in the ratios of villus height to crypt depth (VH–CD) in jejunum at DPI 2, 4, and 6 and in ileum at DPI 4. The number of villous M cells was reduced in jejunum at DPI 4 and 6 and in ileum at DPI 6, while the number of Peyer’s patch M cells in ileum increased at DPI 2 and then decreased at DPI 6. PEDV-infected pigs also had reduced lysozyme expression in ileal Paneth cells at DPI 2 and increased ileal pIgR expression at DPI 4. There were no significant changes in IL-1β and TNF-α expression in PEDV-infected pigs compared to controls. In conclusion, PEDV infection affected innate mucosal immunity of weaned pigs through alterations in Paneth cells, villous and Peyer’s patch M cells, and pIgR expression.
Small intestinal mucosal immunity is critical in defending the body from pathogen exposure while at the same time coordinates nutrient and water intake. Absorptive epithelial cells and specialized enterocytes including goblet cells, Paneth cells, and microfold (M) cells contribute to immune responses for maintenance of gut homeostasis. 32 Intestinal epithelial cells form a complete physical barrier through intercellular junctional complexes and produce cytokines and chemokines for the elimination of pathogens. 39 A thin or discontinuous mucus layer containing lysozyme and antimicrobial peptides covers on the epithelium and forms a chemical defense. Mucin is secreted by goblet cells, while lysozyme and antimicrobial peptides are mainly produced by Paneth cells at the base of crypts. Hence, the physical and chemical barriers spatially segregate the intestinal epithelium from bacteria, viruses, protozoa, and toxins in the lumen. In addition to innate immunity, different enterocytes contribute to induction of the adaptive immune response. 36 M cells on the intestinal epithelium capture antigens from the lumen and deliver them to dendritic cells in the lamina propria. Dendritic cells act as antigen-presenting cells and stimulate lymphocyte activation. Activated lymphocytes and plasma cells secrete dimeric immunoglobulin A (IgA). Free dimeric IgA binds to polymeric immunoglobulin receptor (pIgR) expressed in crypts and is subsequently transported into the intestinal lumen through enterocytes. Thus, enterocytes have not only digestive functions but also critical immunological capabilities against pathogens entering the intestinal mucosa.
Porcine epidemic diarrhea virus (PEDV) is an enveloped single-stranded RNA virus of the Coronaviridae family that infects pigs at all ages via fecal-oral transmission. 11 The virus replicates in small intestinal epithelial cells, mainly in the jejunum and ileum, and induces apoptosis and necrosis of infected cells with accompanying retraction of villus stromal cores. 11,20 As a result, PEDV-infected pigs have decreased villus height to crypt depth (VH–CD) ratio, watery malabsorptive diarrhea, and anorexia. Weaned pigs with PEDV infection often have milder clinical signs and lesions and lower mortality rates compared with nursing piglets. 24,25 Nevertheless, lower average daily gain (ADG), average daily feed intake (ADFI), and feed efficiency (G–F) have been described in PEDV-infected post-weaned pigs. 1,7,8,40 Poor growth performance in post-weaned pigs is certainly unfavorable to the swine industry, and therefore investigating PEDV pathogenesis in weaned pigs is warranted.
Since PEDV infection causes a predominately intestinal disease, it is necessary to understand the influences of PEDV infection on small intestinal mucosal immunity. PEDV is able to block type III interferon (IFN-λ) production by enterocytes. 41,52 Furthermore, PEDV infection affects intestinal barrier function by decreasing the expression of cell junction–associated molecules in epithelial cells, reducing the number of goblet cells, and disturbing the composition of the microbiota. 9,16,17,22 These studies indicate that PEDV infection impairs localized immune responses. However, changes in innate mucosal immunity are not examined thoroughly and warrant further investigation. The aim of this study was to characterize alterations of specific enterocyte subtypes and innate immune components in post-weaned pigs during PEDV infection. The a priori hypothesis was that PEDV infection might disrupt innate mucosal immunity in the small intestine including Paneth cells, M cells, and pIgR expression.
Materials and Methods
Animals and Experimental Design
All experimental protocols were approved by the Institutional Animal Care and Use Committee at Iowa State University (IACUC# 8-17-8580-S). A total of 64 four-week-old, commercial cross-bred gilts were selected from a PEDV-negative herd. Animals were randomly allotted to different treatment groups with 6 to 10 pigs per pen and 4 pens per treatment. The treatments were PEDV-infected (n = 40) and sham-inoculated (control; n = 24). Each treatment group was housed in one room separately with strict biosecurity and biocontainment. Animals were given free access to water and ad libitum fed a diet that met or exceeded NRC2012 requirements for this size pig throughout the study period. Pigs were allowed to acclimatize to the facility for 4 days prior to inoculation. On day post inoculation 0 (DPI 0), PEDV-infected pigs were inoculated orally with 10 ml of 104 TCID50/ml PEDV isolate (USA/IN19338/2013) as previously described, while control pigs received 10 ml of sterile media. 6,46 At DPI 2, 4, 6, and 10, a subset of pigs was euthanized from each group (ie, 6 control pigs and 10 PEDV-infected pigs at each time point) for tissue collection and analysis as described below. Pigs were randomly chosen within health status at each DPI and were euthanized via captive bolt and exsanguination.
Clinical Evaluation
Animals were evaluated daily throughout the study period for the presence of clinical signs typical of PEDV infection. These signs included lethargy and changes in fecal consistency. Fecal consistency scores were assigned using a 4-point scale, as follows: 0 = normal feces; 1 = soft feces; 2 = loose and shapeless diarrhea; 3 = watery diarrhea.
Analysis of PEDV RNA Titer in Fecal Samples
Rectal swabs were collected before euthanasia and submitted to Iowa State University Veterinary Diagnostic Laboratory (Ames, IA) for quantitative real-time polymerase chain reaction (RT-qPCR) of PEDV in feces. 7,8 A cycle threshold (Ct) of ≥36 was considered negative and <36 considered positive for PEDV.
Gross and Histopathology
The necropsy was performed immediately following euthanasia and gross lesions were recorded. The following tissues were collected for each pig: tonsil, jejunum (2 sections from the aboral third), ileum (2 sections), and mesenteric lymph nodes. Tissues were fixed in 10% neutral buffered formalin for no more than 48 hours, then transferred into 70% ethanol for short-term storage. Fixed tissues were routinely processed, embedded in paraffin, and sections of 4-µm thickness were stained with hematoxylin and eosin (HE).
Villus length and crypt depth in jejunum and ileum were measured blindly without knowledge of infection status or time point. The average measurements of 10 normally oriented villus and crypt units were presented as VH–CD ratios for each intestinal segment per pig.
Immunohistochemistry
Immunohistochemistry (IHC) for PEDV antigen was performed at the Iowa State University Veterinary Diagnostic Laboratory (Ames, IA) using a PEDV-specific mAb (BioNote). 6,25 PEDV antigen-positive cells were estimated in the jejunum, ileum, and lymphoid tissues, including tonsils, ileal Peyer’s patches, and mesenteric lymph nodes. Assigned scores for each tissue were as follows: 0 = negative; 1 = 1% to 10% of cells immunopositive; 2 = 11% to 50% of cells immunopositive; 3 = more than 50% of cells immunopositive.
The following primary antibodies were used for evaluating small intestinal mucosal immune cells: polyclonal rabbit anti-lysozyme EC 3.2.1.17 (Dako, Glostrup, Denmark), monoclonal mouse anti-cytokeratin peptide 18 (Sigma-Aldrich, St Louis, MO), and polyclonal rabbit anti-pIgR (Sigma-Aldrich, St Louis, MO). Specificity of these antibodies has been documented, indicating that lysozyme is a Paneth cell marker in Peyer’s patches, and cytokeratin 18 is an M cell marker 13,19,47,48
Based on the results of histological examination and PEDV immunolabeling, pigs at DPI 2, 4, and 6 were selected to study mucosal immune cells. Sections of jejunum (2 locations) and ileum (2 locations) were cut and placed onto positively charged slides, deparaffinized, and rehydrated in a series of ethanol dilutions. Antigens were retrieved in Tris/EDTA (pH 9.0), followed by 20 minutes of steam treatment in an autoclave. After 15 minutes of cooling, slides were incubated in 3% hydrogen peroxide in methanol for 20 minutes to suppress endogenous peroxide. Slides were blocked with 10% normal goat serum for 1 hour and then incubated with primary antibody (anti-lysozyme, 1:1000; anti-cytokeratin 18, 1:1000; anti-PIGR, 1:600) for 14 hours in a humidity chamber at 4 °C. Secondary antibody, peroxidase-conjugated goat anti-rabbit IgG or goat anti-mouse IgG at 1:200 dilution (ThermoFisher Scientific, Carlsbad, CA), was applied for 1 hour. Slides were bathed in 3% hydrogen peroxide in methanol for 25 minutes and then incubated in metal-enhanced 3,3′-diaminobenzidine (DAB) substrate (ThermoFisher Scientific, Carlsbad, CA) for 30 minutes. Slides were then bathed in NovaRED chromagen (Vector, Burlingame, CA) for 10 minutes followed by a 5-minute Scott’s tap water bath.
For evaluating activity of Paneth cells, lysozyme expression in Paneth cells was semiquantitatively estimated using HALO image analysis software (Indica Labs, Corrales, NM). The average measurements of four 200× fields were evaluated for each intestinal segment per pig. Lysozyme immunopositive area (μm2) in Paneth cells was presented as one number per µm basement membrane of epithelium in jejunum and ileum, respectively. For quantifying the number of M cells, CK18-immunopositive cells were classified as villous M cells on villous epithelium in the jejunum and ileum and Peyer’s patch M cells on dome epithelium in the ileum and counted throughout sections. Cellular density of villous M cells and Peyer’s patch M cells was presented as a number per µm villus epithelium and follicle-associated epithelium, respectively. For evaluating the presence of pIgR, pIgR immunopositive area was semiquantitatively estimated via HALO image analysis software. The average measurement of four 200× fields was evaluated for each intestinal segment per pig. The pIgR immunopositive area (μm2) was presented as one number per µm of basement membrane of epithelium in jejunum and ileum, respectively.
Measurement of Cytokines
For evaluating cytokine concentrations in the small intestine, tissue samples from the jejunum were collected from pigs at DPI 2, 4, and 6 and were frozen in liquid nitrogen for storage at −80 °C until protein extraction and analysis. Tissues were homogenized in HEPES buffer (50 mM HEPES, 150 mM NaCl, 50 mM NaF, 2 mM EDTA, 5% glycerol, 1% Triton X, 0.1% protease inhibitor cocktail) and centrifuged at 1000 × g for 10 minutes at 4 °C. The supernatant was collected, and protein concentrations were determined via a Pierce bicinchoninic acid (BCA) assay (Thermofisher Scientific, Carlsbad, CA). Interleukin (IL)-1β and tumor necrosis factor-α (TNF-α) were assayed using porcine IL-1 beta/IL-1F2 DuoSet ELISA and porcine TNF-alpha DuoSet ELISA kits (R&D Systems, Minneapolis, MN) according to the manufacturer’s instructions.
Statistical Analysis
All data were analyzed using Statistical Analysis Software (SAS) 9.4 (SAS Institute Inc, Cary, NC) and pig was considered the experimental unit. The MIXED procedure was used to analyze cytokines protein abundance to determine the main effects of treatment, DPI, and their interaction. The GLIMMIX procedure was used to analyze VH–CD ratio, IHC staining of PEDV, Paneth cells, M cells, and pIgR expression. The CORR procedure was used for investigating the relationship between variables. A P value ≤.05 was considered significant.
Availability of Data and Materials
The data analyzed in this study are available as Supplemental Materials.
Results
Clinical Assessment
During the entire experiment, control pigs did not develop clinical signs or lesions associated with porcine epidemic diarrhea. From DPI 1 to 10, PEDV-infected pigs presented variable degrees of diarrhea (Fig. 1). Watery diarrhea was recorded from DPI 4 to 9. Diarrhea peaked at DPI 5 when 7/20 (35%) PEDV-infected pigs exhibited watery diarrhea (Supplemental Table S1).
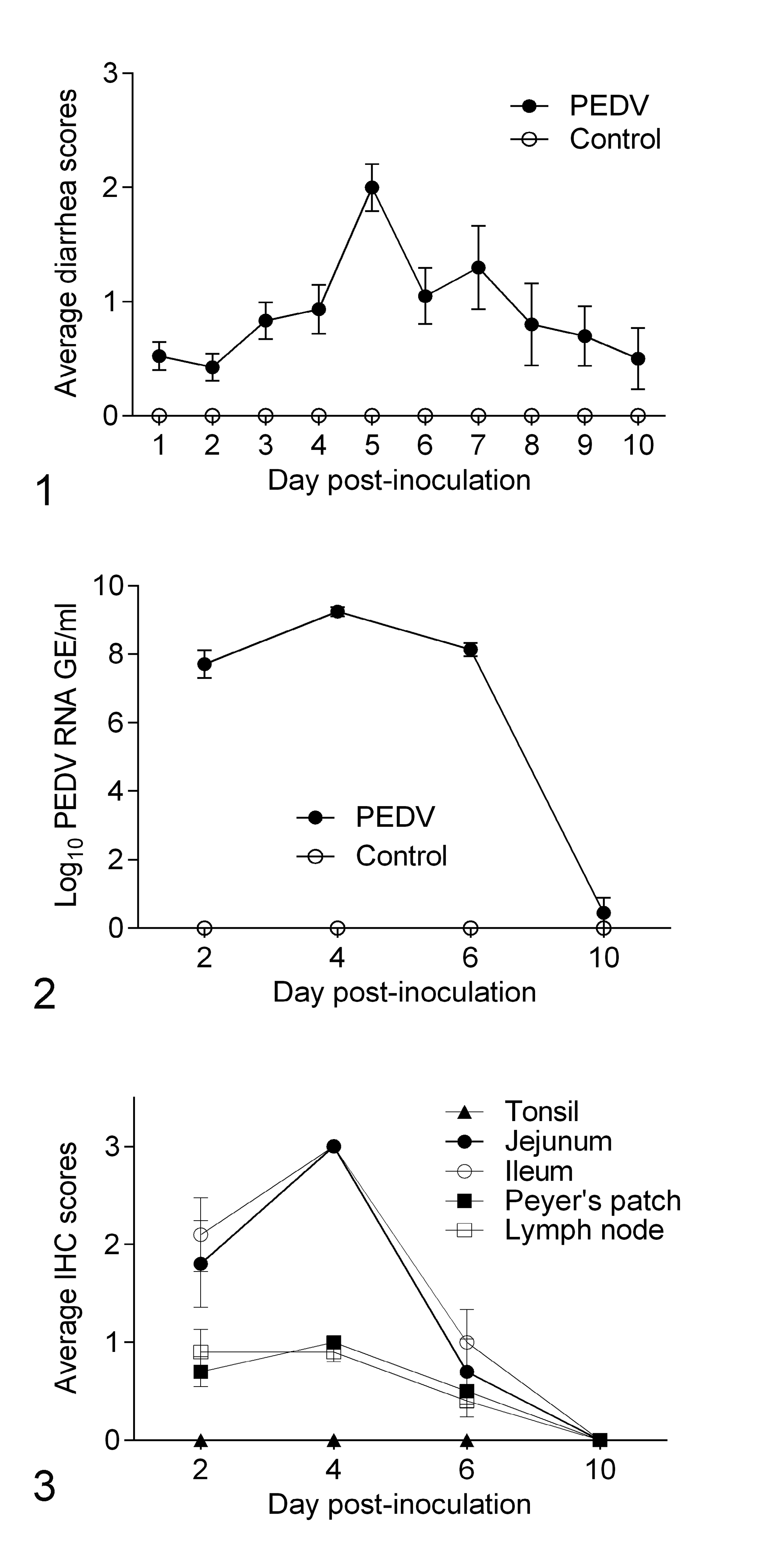
Fecal PEDV RNA Shedding and PEDV IHC Staining
By RT-qPCR, the controls had no detectable PEDV RNA in feces. All PEDV-infected pigs at DPI 2, 4, and 6 had moderate to high viral RNA shedding ranging from 5.1 to 9.8 log10 GE/ml (Supplemental Table S2). At DPI 10, only 1 of 10 pigs had detectable fecal viral RNA shedding (4.4 log10 GE/ml). Mean fecal PEDV RNA shedding at DPI 4 was significantly higher (P < .001) compared to other time points (Fig. 2).
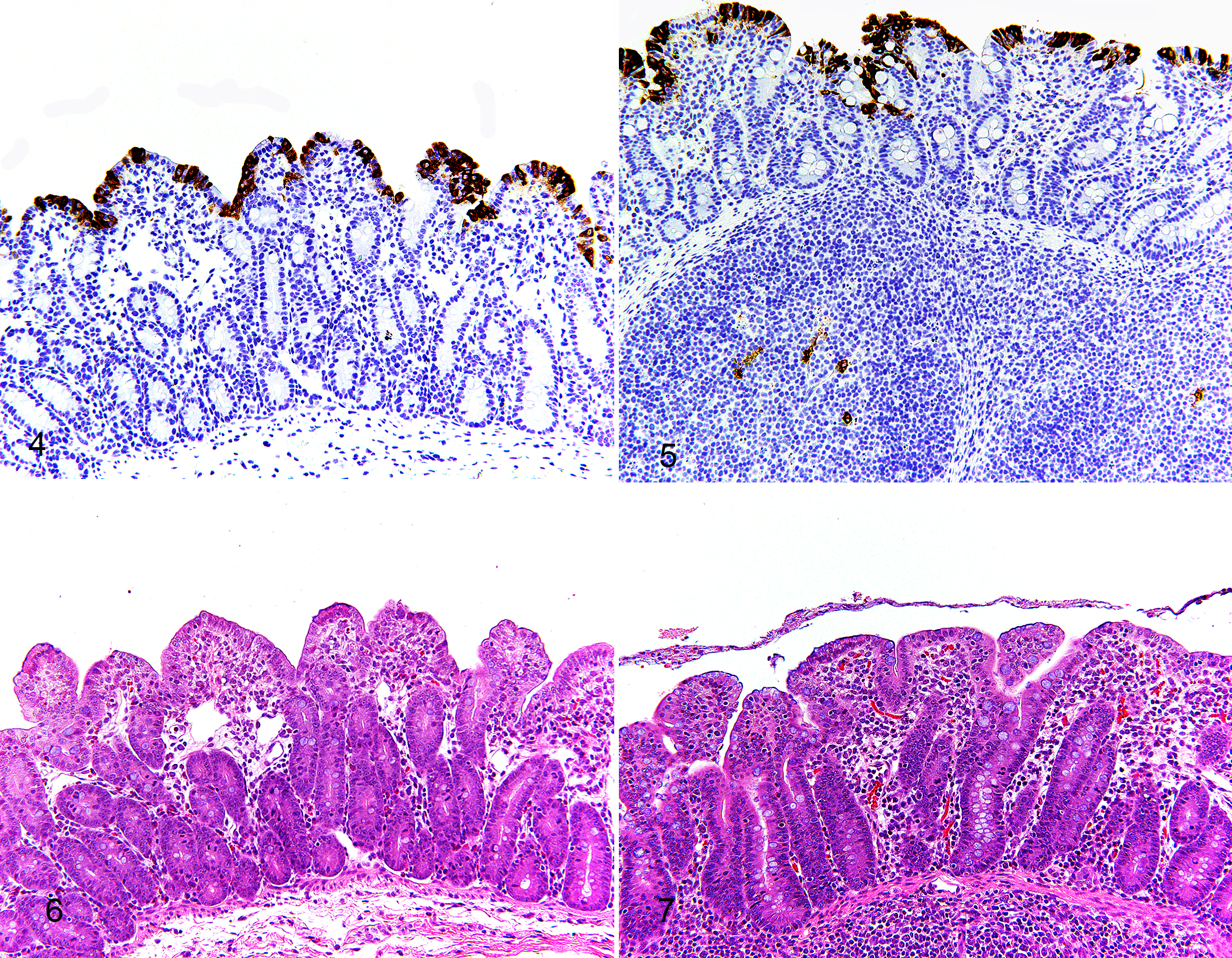
All controls were negative for PEDV antigen via IHC. PEDV antigen was detected in PEDV-infected pigs from DPI 2 to 6. These signals were located in villus enterocytes of jejunum and ileum, and in mononuclear cells in ileal Peyer’s patches and mesenteric lymph node. No PEDV-antigen signal was found in tonsils at any of the time points assessed (Figs. 3–5). At DPI 4, 10/10 pigs were positive for PEDV antigen in jejunum, ileum, and ileal Peyer’s patches, and 9/10 pigs were PEDV-immunopositive in mesenteric lymph nodes (Supplemental Table S2). At DPI 10, none of the pigs were PEDV-immunopositive.
Gross Pathology and Histopathology
At necropsy, PEDV-infected pigs had variable degrees of fecal staining in the perineal region, flaccid and thin-walled intestines, and watery content in the small intestine and colon. Histologically, jejunum and ileum showed villous atrophy, villous fusion, and superficial enterocyte attenuation (Figs. 6, 7). In comparison to controls, PEDV-infected pigs had lower jejunal VH–CD ratio at DPI 2, 4, and 6 (P < .0001) and lower ileal VH–CD ratio at DPI 4 (P = .001; Table 1; Supplemental Table S2). Within the PEDV-infected group, pigs at DPI 4 had lower VH–CD ratio in jejunum than pigs at DPI 2 and 10 (P < .01, all analyses), and pigs at DPI 4 and 6 had lower VH–CD ratio in ileum than pigs at DPI 2 and 10 (P < 0.0001, all analyses). Additionally, scores of PEDV IHC in the jejunum, ileum, ileal Peyer’s patches, and mesenteric lymph nodes positively correlated with PEDV RNA fecal shedding and negatively correlated with the VH–CD ratios of jejunum and ileum (P < .05, all analyses; Table 2).
Villus Height: Crypt Depth Ratios Over Time in Jejunum and Ileum of Pigs Infected With Porcine Epidemic Diarrhea Virus Compared to Controls.*
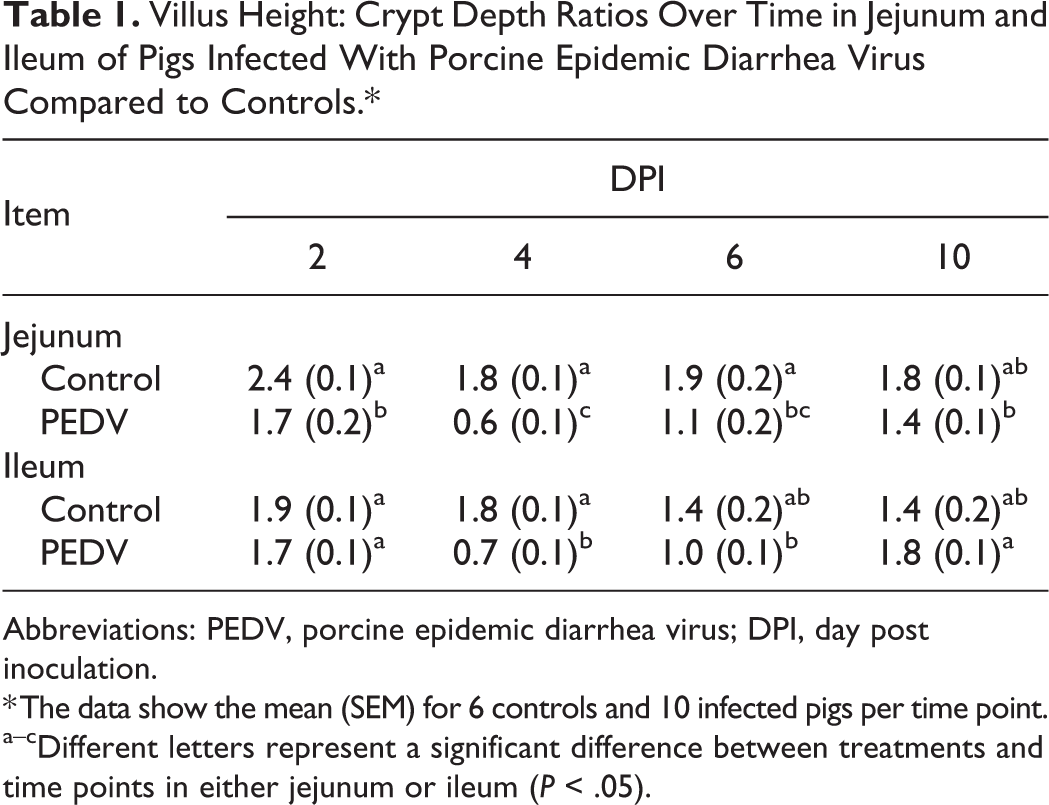
Abbreviations: PEDV, porcine epidemic diarrhea virus; DPI, day post inoculation.
* The data show the mean (SEM) for 6 controls and 10 infected pigs per time point.
a–cDifferent letters represent a significant difference between treatments and time points in either jejunum or ileum (P < .05).
Correlations Between PEDV RNA Titer and Other Study Variables.
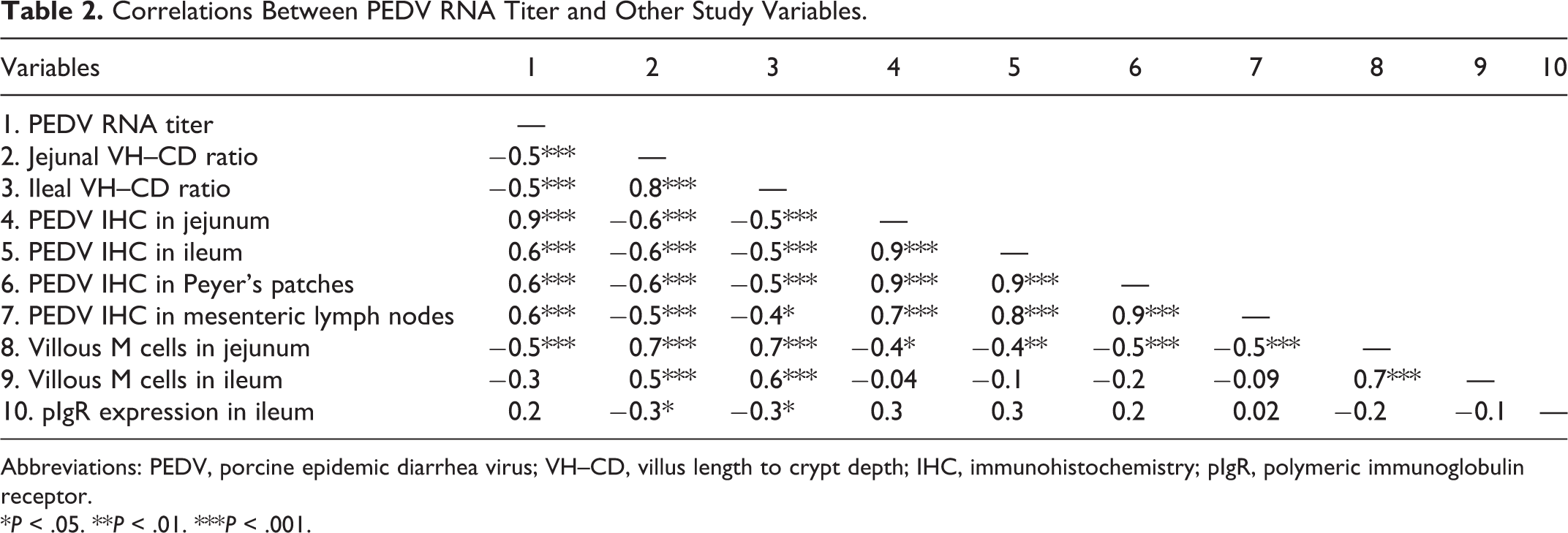
Abbreviations: PEDV, porcine epidemic diarrhea virus; VH–CD, villus length to crypt depth; IHC, immunohistochemistry; pIgR, polymeric immunoglobulin receptor.
*P < .05. **P < .01. ***P < .001.
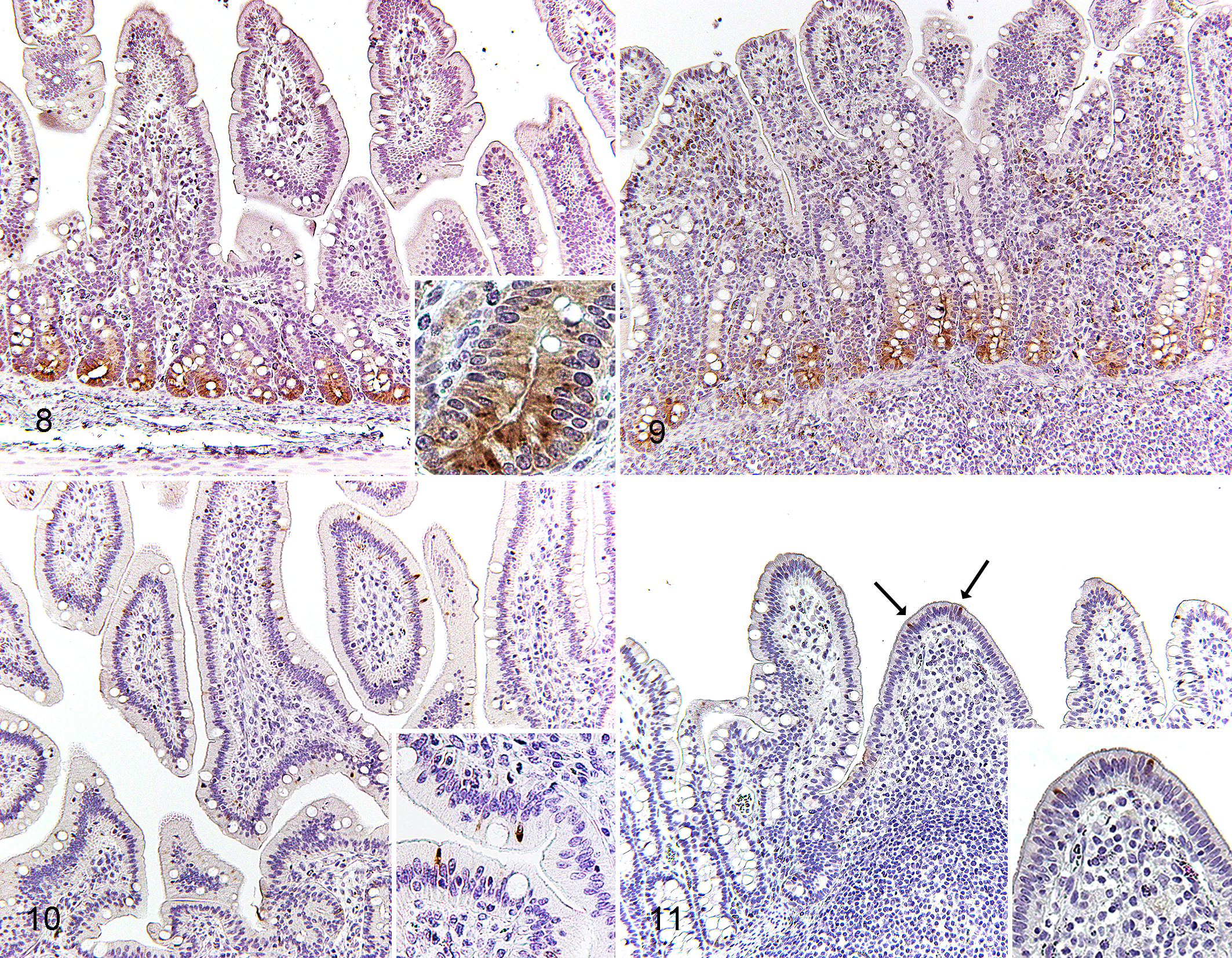
Paneth Cells
In controls and PEDV-infected pigs, Paneth cells were present at the base of crypts in jejunum and ileum, and lysozyme immunolabeling was confined to apically located granules in the cytoplasm of Paneth cells (Figs. 8, 9). In comparison to controls, PEDV-infected pigs had decreased expression of lysozyme within ileal Paneth cells at DPI 2 (P = .0026; Table 3). In the control group, pigs at DPI 2 had higher lysozyme expression in ileal Paneth cells in comparison to DPI 6 (P = .0011).
Mean Lysozyme Expression (SEM) in Paneth Cells in Jejunum and Ileum of Control (n = 6) and PEDV-Infected (n = 10) Pigs at Each Time Point.
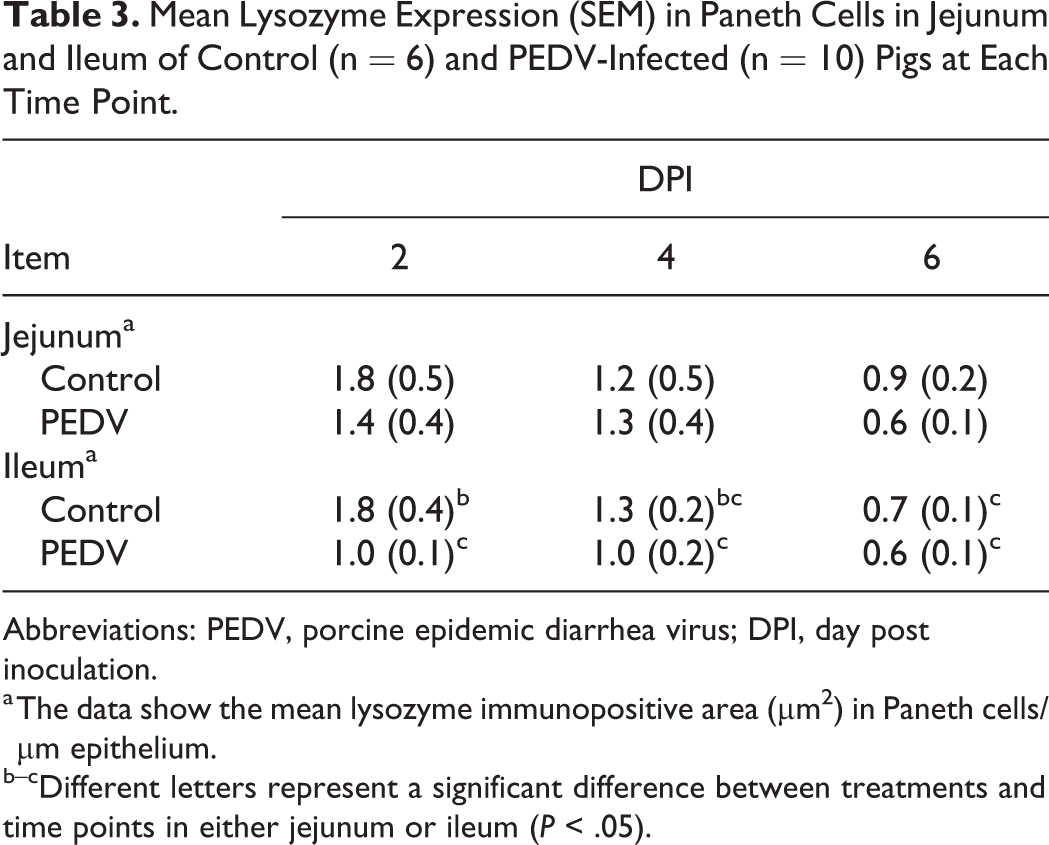
Abbreviations: PEDV, porcine epidemic diarrhea virus; DPI, day post inoculation.
a The data show the mean lysozyme immunopositive area (μm2) in Paneth cells/µm epithelium.
b–cDifferent letters represent a significant difference between treatments and time points in either jejunum or ileum (P < .05).
M Cells
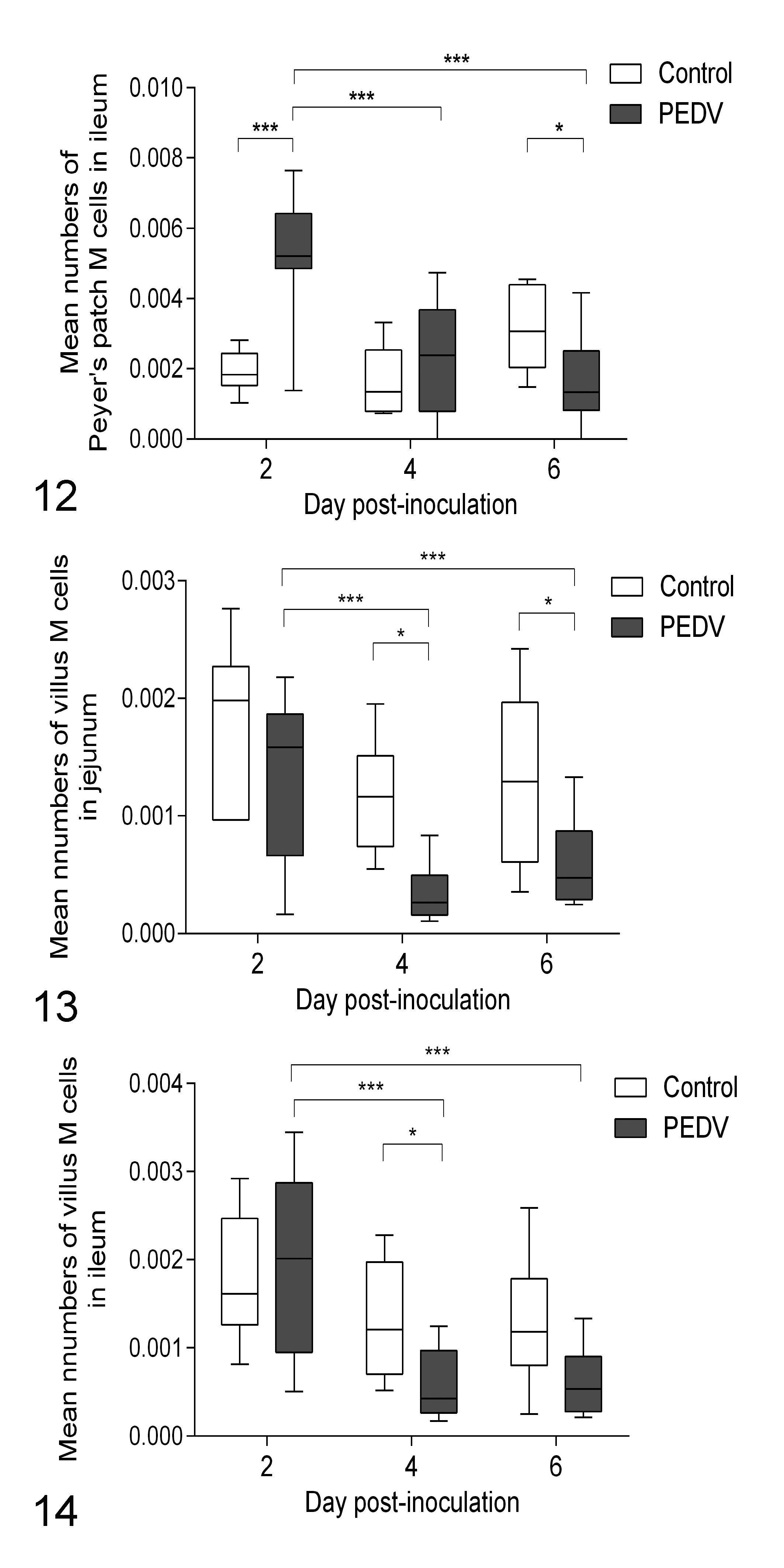
In controls and PEDV-infected pigs, the cytoplasm of M cells in both ileum and jejunum was intensely immunolabeled for cytokeratin 18. Villous M cells were scattered throughout the villous epithelium of jejunum and ileum (Fig. 10), whereas Peyer’s patch M cells were located on the dome epithelium of ileum (Fig. 11). There was no morphological difference between villous M cells and Peyer’s patch M cells. In comparison to controls, the number of Peyer’s patch M cells in PEDV-infected pigs increased at DPI 2 (P < .0001) then reduced at DPI 6 (P = .0390; Fig. 12). Villous M cells in PEDV-infected pigs also had reduced in jejunum at DPI 4 and 6 and in ileum at DPI 4 (P < .05, all analyses; Figs. 13, 14). Within the PEDV-infected group, relative to pigs at DPI 2, pigs at DPI 4 and 6 had reduced villous and Peyer’s patch M cells in jejunum and ileum (P < .05, all analyses). Furthermore, the number of villous M cells in jejunum and ileum correlated positively with their VH–CD ratios (P < .0001, all analyses; Table 2).
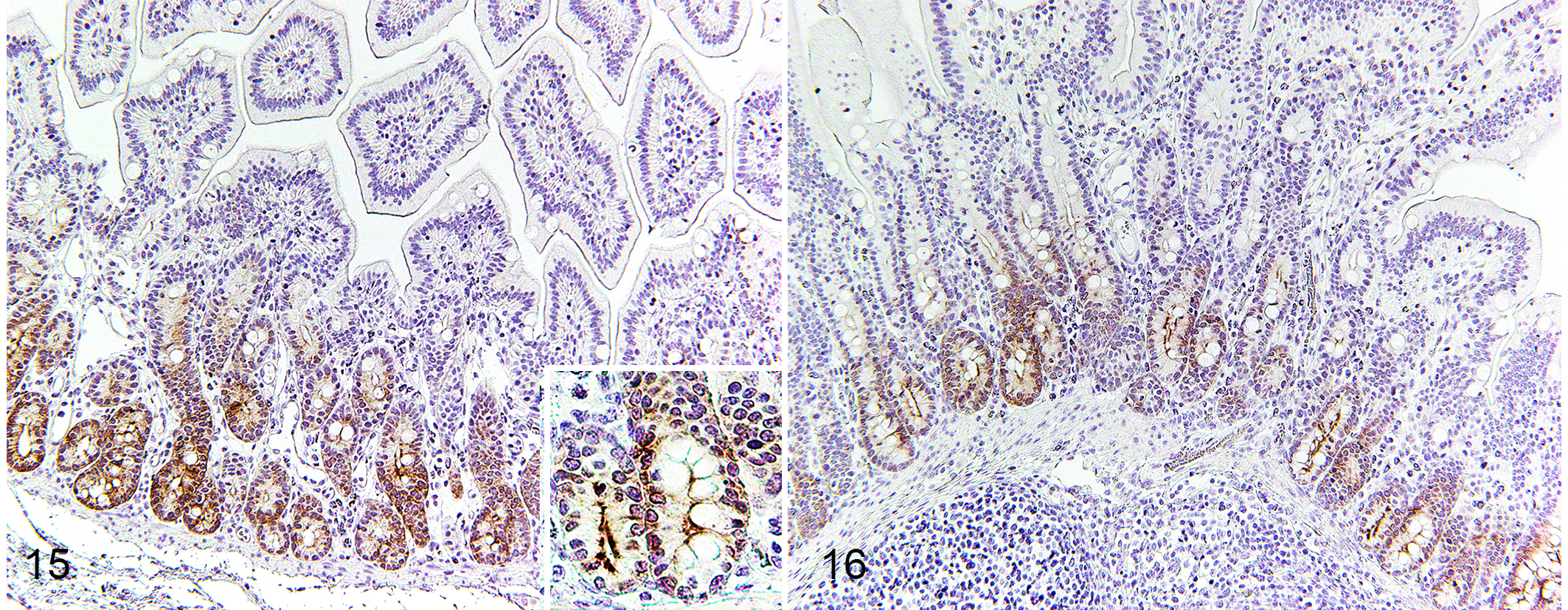
Expression of Polymeric Immunoglobulin Receptor (pIgR)
In controls and PEDV-infected pigs, pIgR protein immunolabeling was identified in the crypts of jejunum and ileum and appeared as a network of fibrils surrounding the nuclei and as a dense plaque in the most apical cytoplasm (Figs. 15, 16). In comparison to controls, PEDV-infected pigs increased pIgR expression in the ileum at DPI 4 (P = .042; Table 4). In the PEDV-infected group, pigs at DPI 4 had higher pIgR expression in the jejunum than pigs at DPI 2 (P = .0003) and had higher pIgR expression in the ileum than pigs at DPI 2 and 6 (P < .05, all analyses). Also, pIgR expression in ileum negatively correlated with ileal VH–CD ratio (P = .036; Table 2).
Mean pIgR Expression (SEM) in Jejunum and Ileum in Control (n = 6) and PEDV-Infected (n = 10) Pigs at Each Time Point.

Abbreviations: pIgR, polymeric immunoglobulin receptor; PEDV, porcine epidemic diarrhea virus; DPI, day post inoculation.
a The data show the mean pIgR-immunopositive areas (μm2)/µm epithelium.
b–cDifferent letters represent a significant difference between treatments and time points in either jejunum or ileum (P < .05).
Jejunum Cytokine Concentration
As PEDV primarily impacts the jejunum of pigs, jejunum proinflammatory cytokine markers were assessed and summarized in Table 5. Overall, there was no effect of PEDV infection on IL-1β or TNF-α protein abundance in the jejunum. In both PEDV-infected and control pigs, IL-1β decreased as the DPI increased (P < .001), and TNF-α increased as the DPI increased (P = .002).
Mean Jejunum Cytokine Concentration (SEM) in Control (n = 6) and PEDV-Infected (n = 10) Pigs at Each Time Point.
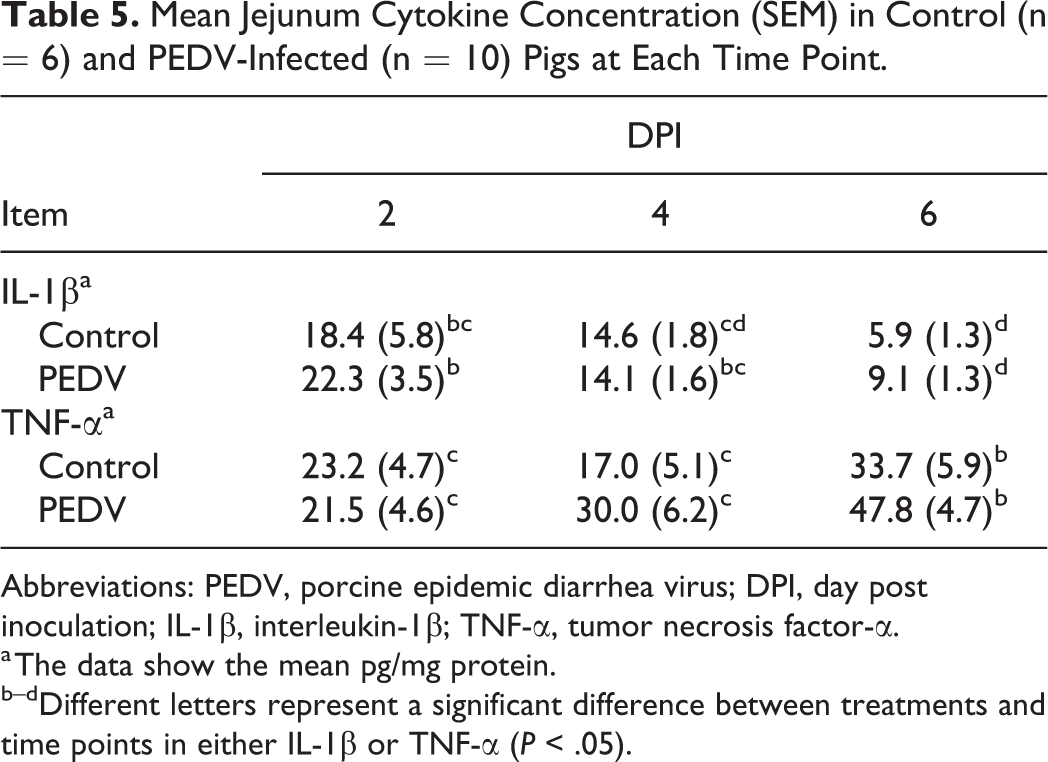
Abbreviations: PEDV, porcine epidemic diarrhea virus; DPI, day post inoculation; IL-1β, interleukin-1β; TNF-α, tumor necrosis factor-α.
a The data show the mean pg/mg protein.
b–dDifferent letters represent a significant difference between treatments and time points in either IL-1β or TNF-α (P < .05).
Discussion
In the present study, all PEDV-infected pigs from DPI 2 to 6 were positive for PEDV RNA and antigen in jejunum and ileum. PEDV antigen was no longer detected in jejunum and ileum by DPI 10. Consequently, we chose pigs at DPI 2, 4, and 6 for investigating small intestinal mucosal immunity. PEDV infection coincided with severity of diarrhea and intestinal villus injury and was consistent with previous studies performed in weaned pigs. 7,15,25,31 Additionally, the jejunal VH–CD ratio decreased from DPI 2 to 6, while ileal VH–CD ratio decreased at DPI 4. These results indicate that jejunum had more severe villus injury than ileum.
As part of mucosa-associated lymphoid tissues (MALT) of the digestive tract, tonsil is the first line of defense against ingested pathogens. A low level of PEDV nucleic acid has been detected in the tonsil as well as other non-enteric organs such as lung, thymus, and muscle by PCR during the viremic phase of infection. 6,32 However, PEDV antigen was not detectable in the tonsils by IHC in our study. This suggests that significant virus accumulation does not occur in this tissue. Variance between previous findings and this study may be due to virus strain differences, challenge doses used among experiments, or a lack of PEDV receptor in tonsils. In contrast to the negative result in tonsil. PEDV-antigen positive cells were in ileal Peyer’s patches and mesenteric lymph nodes from DPI 2 to 6. Based on these findings, we suggested that Peyer’s patches could be a main location for delivering PEDV antigen to lymphoid tissues.
Paneth cells, a specialized type of enterocyte, synthesize lysozyme and antimicrobial peptides that are important for innate immune defense and for controlling the microbiota composition. The expression of lysozyme and antimicrobial peptides in Paneth cells is regulated by both cell-intrinsic and cell-extrinsic mechanisms. In mice, invasive bacteria such as Salmonella Typhimurium are able to enter into Paneth cells and induce endoplasmic reticulum (ER) stress, leading to decrease lysozyme via the intrinsic signaling pathway. 3 Paneth cells also receive cell-extrinsic signaling from lymphocytes, including type 3 innate lymphoid cells (ILC3) and Th17 cells, which upregulate the production of lysozyme by secreting IL-17 and IL-22. 12,33 In macaques, lymphocyte depletion results in reduced lysozyme in Paneth cells. 21 Few studies have addressed the location of Paneth cells in pigs, mainly in ileal enteroid models. 43,48 Herein, we identified that Paneth cells are located at the base of the intestinal crypts in both jejunum and ileum in vivo.
The impact of PEDV infection on Paneth cells has been poorly elucidated. In the present study, PEDV-infected pigs had reduced lysozyme production in ileal Paneth cells at DPI 2, which occurred prior to the significant villus injury in ileum at DPI 4. For evading immune responses, PEDV is able to reduce cytokine expression, including type I and III interferons, IL-6, and TNF-α, and to reduce receptors on intestinal epithelial cells. 41,42 It is suspected that, in the early phase of PEDV infection, extrinsic signaling for Paneth cells might be impeded due to low levels of pro-inflammatory and pro-immune cytokines. While significant changes in pro-inflammatory cytokines were not observed in this study, correlation between lysozyme in ileal Paneth cells and inflammatory cytokines cannot be ruled out. Interestingly, decreased lysozyme expression in Paneth cells was not observed in the jejunum. It is hypothesized that, owing to interaction with immune cells in the Peyer’s patch, Paneth cells in the ileum are subjected to a greater cytokine imbalance than Paneth cells in jejunum during the acute phase of PEDV. Additionally, ileal Paneth cells in the control group had a reduction in lysozyme expression from DPI 2 to 6. This suggests that ileal Paneth cells decrease pIgR expression over time. The mechanism is still unclear and needs further investigation.
On the intestinal epithelium, M cells act as antigen delivery cells and are responsible for capturing and transporting antigens to the lamina propria and stimulating M cell-dependent lymphocyte activation. M cells also serve as a unique portal for invading pathogens. For instance, Salmonella Typhimurium is capable of invading small intestine through M cells. 27 Based on location, M cells are classified as either villous M cells or Peyer’s patch M cells. Moreover, these subtypes of M cells have different origins that villous M cells are inducible and transdifferentiated from enterocytes, while Peyer’s patch M cells are constitutive and derived from crypt stem cells. 23,49
To study the impact of PEDV infection on M cells in pigs, abundance of M cells was measured over the infection period. Herein, Peyer’s patch M cells were induced in PEDV-infected pigs at DPI 2. To our knowledge, this is the first study demonstrating that under viral infection, pigs show rapid induction of M cells. In mice and rabbits, bacteria and cholera toxin activate the generation of Peyer’s patch M cells from hours to days after infection. 4,5,28,45 Accordingly, the induction of Peyer’s patch M cell in the acute phase of PEDV infection may enhance M cell–dependent lymphocyte activation but also increase the risk of invading bacterial infection. Later, Peyer’s patch M cells in PEDV-infected pigs were reduced at DPI 6. At this time point, ileal VH–CD ratio in the PEDV-infected pigs was not different from controls, indicating that Peyer’s patch M cells were still impaired after villus recovery. The mechanism underlying a reduction in Peyer’s patch M cells is unknown. It is likely related to alteration in the mucosal epithelium during restitution. Of note, from the perspective of vaccine efficiency, Peyer’s patch M cells are more crucial than villous M cells for inducing an immune response, owing to their increased interaction with immune cells in the Peyer’s patch. 49 Given a lack of continued maternal antibodies from milk, post-weaned pigs need assistance from vaccines to establish humoral immunity. Hence, without sufficient Peyer’s patch M cells, PEDV-infected weaned pigs may be limited in their immune recognition of other ingested pathogens or antigens from oral vaccines.
We also reported that PEDV-infected pigs had fewer villous M cells in jejunum at DPI 4 and 6 and in ileum at DPI 4 compared to controls. Furthermore, the number of villous M cells positively correlated with VH–CD ratios in the jejunum and ileum. As already mentioned, villous M cells are transdifferentiated from enterocytes on villi, indicating that the reduction of villous M cells coincides with villus injury. As a result, PEDV infection leads to the reduction of not only absorptive epithelial cells but also villous M cells. Nevertheless, the mechanism of losing villous M cells is unknown. It could be associated with direct injury from viral replication in villous M cells or secondary impairment by immune cells or by loss of communication with neighboring absorptive epithelial cells.
Small intestinal epithelial transcytosis of IgA and IgM is mediated via pIgR expressed in crypts. 37 Free dimeric IgA in lamina propria binds to pIgR at the basolateral surface of enterocytes and is transported through these cells. At the apical surface of enterocytes, the extracellular ligand-binding domain of pIgR is cleaved and released as secretory IgA (SIgA) into the lumen. Thus, pIgR has been shown to be essential for adaptive immunity in the small intestine. 37 In fact, enterocytes produce and release pIgR constitutively. 29 In the absence of IgA binding, pIgR is released in its free form, and this secretory component acts as a nonspecific microbial scavenger in the intestinal lumen. Accordingly, pIgR bridges innate and adaptive responses in mucosal immunity. 18,37 Surprisingly, few studies have addressed the locations and functions of pIgR in the swine gastrointestinal tract. 35,47 Herein, we report that pIgR is expressed in crypts of both jejunum and ileum in pigs.
We found that, at DPI 4, PEDV-infected pigs had increased ileal pIgR expression, which is negatively correlated with ileal VH–CD ratio. These findings suggest that, rather than impairing pIgR expression, PEDV infection might enhance pIgR expression in ileum. pIgR is transcriptionally upregulated by cytokines, hormones, dietary factors, and microbial products. 14,30 In mice and humans, acute inflammatory cytokines including IL-1, TNF, IFN-γ, and TGF-β upregulate transcription of pIgR gene. 38,44 Weaned pigs infected with PEDV had higher levels of serum IFN-α, IL-12, and IL-8 at DPI 3 and TNF from DPI 1 to 5. 2 Increased inflammatory cytokines were not observed in the present study. However, correlation between increased pIgR expression in ileum and increased inflammatory cytokines cannot be ruled out. Similar to changes in Paneth cells, increased pIgR expression was only observed in the ileum. Again, it could be related to the high interaction between enterocytes and immune cells of the ileal Peyer’s patches. In weaned piglets infected with PEDV, PEDV-specific IgA in feces is detected by up to DPI 14. 34 Our study suggests that pIgR expression is activated before PEDV-specific IgA production. Increased pIgR expression in enterocytes is desirable for transcytosis of IgA. 10,50
PEDV has been shown to induce a pro-inflammatory cytokine response in the small intestinal epithelium. 2 Interestingly, we reported no significant differences in the protein abundance of IL-1β and TNF-α in the jejunum between controls and PEDV-infected pigs. This observation suggests 2 potential hypotheses. First, samples used for detecting cytokine activity in this study were freeze-ground tissue homogenates containing components of both mucosal immunity and of systemic immunity such as blood. The combination of these tissues could potentially obscure subtle alterations in mucosal immunity. Second, samples for detecting cytokine activity were collected from jejunum, while changes in mucosal immunity were mainly observed in ileum. The difference suggests that small intestinal mucosal immunity could be segmental and location-specific. In future investigations, fresh ileal mucosa may be a more sensitive sample type to detect potential cytokine changes. Irrespective of treatment, a decrease in IL-1β and an increase in TNF-α were observed as DPI increased. Increasing levels of IL-1β in weaned pigs have been demonstrated to be related to weaning stress. 26 It is suspected that, in our study, weaned pigs that went through weaning stress in the week prior to the experiment may have reduced IL-1 expression consequently. Besides, post-weaned pigs also show an age-related increase of TNF-α. 51 Taken together, these results suggest that it is important to be aware of physiological changes occurring at different locations of the small intestine while studying cytokine responses in weaned pigs.
This study focused only on weaned pigs. PED is an age-dependent disease and, relative to nursing piglets, weaned pigs have milder clinical signs and lesions. Variation of PEDV pathogenesis between nursing and weaned pigs may be associated with the immune response. Additional investigation is warranted in nursing piglets to determine if they experience similar alterations of innate mucosal immunity.
Conclusions
Different subtypes of enterocytes cooperate to confer innate mucosal immunity. Here we reported that the PEDV infection could affect innate mucosal immunity by decreasing lysozyme expression in Paneth cells, destroying villous and Peyer’s patch M cells, and increasing pIgR expression in the small intestine of weaned pigs. Most of these changes were observed in ileum, although ileum had milder villus injury than jejunum. Further investigation is warranted on the intestinal restitution of different enterocyte subtypes, such as M cells, after PEDV infection to expand understanding of PEDV pathogenesis.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Chen_et_al - Alterations in Intestinal Innate Mucosal Immunity of Weaned Pigs During Porcine Epidemic Diarrhea Virus Infection
Supplemental Material, Combined_supplemental_materials-Chen_et_al for Alterations in Intestinal Innate Mucosal Immunity of Weaned Pigs During Porcine Epidemic Diarrhea Virus Infection by Ya-Mei Chen, Emma T. Helm, Nicholas Gabler, Jesse M. Hostetter and Eric R. Burrough in Veterinary Pathology
Footnotes
Acknowledgements
We thank members of the Livestock Infectious Disease Isolation Facility for their assistance. We appreciate Jennifer M. Groeltz-Thrush, Debra G. Moore, and staff from the Iowa State University Veterinary Diagnostic Laboratory for slide preparation and technical assistance. We thank Min Zhang for statistical consultation.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Agriculture and Food Research Initiative Competitive Grant AH 10072632017 from the USDA National Institute of Food and Agriculture (Blikslager, Gabler, Burrough, Odle).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
