Abstract
An 11-day-old little blue penguin (Eudyptula minor) died unexpectedly. Prior to hatching, the egg experienced trauma and resultant defects were repaired. The chick hatched without complication and was clinically normal prior to death. Necropsy revealed congested lungs. Histologic examination showed moderate nonsuppurative encephalitis with focally extensive neuronal necrosis and intranuclear inclusions in neurons within necrotic foci. Herpesvirus DNA was detected in brain tissue with a generic herpesvirus polymerase chain reaction. Sanger sequencing demonstrated 100% and 98% sequence homology to sphenicid alphaherpesvirus 1 and penguin herpesvirus 2, respectively. In situ hybridization demonstrated large amounts of herpesvirus nucleic acid in intranuclear inclusions and neuronal nuclei. Combined histology, polymerase chain reaction, Sanger sequencing, and in situ hybridization results were most consistent with herpesviral encephalitis, most likely caused by sphenicid alphaherpesvirus 1. To our knowledge, this is the first report of a herpesvirus infection causing encephalitis in a penguin and the first report of herpesvirus in this species.
Herpesviruses are double-stranded DNA viruses known to infect a wide range of vertebrates including birds, mammals, amphibians, reptiles, and fish along with a few invertebrate species, and are categorized as alpha, beta, and gamma subtypes. 1 –3,6,19 Each herpesvirus species has evolved with a specific host species and the virus generally shows low pathogenicity in its natural host. 1,2 Disease severity can range from latent infections with no clinical signs to severe and sometimes fatal disease depending on host immune status, host-virus relationship, and the presence of secondary infections. 7,9,14,20
Different herpesvirus species have been documented in several bird families, including psittacines, poultry, waterfowl, and seabirds. 4,7,11,16 In penguins, herpesvirus and herpes-like viruses have been isolated from the African (Spheniscus demersus), Humboldt (Spheniscus humboldti), and Magellanic (Spheniscus magellanicus) penguin species from the genus Spheniscus. 8,11,13,14 Herpesviruses have been identified in the respiratory and gastrointestinal tract in these species, associated with severe lesions in these organ systems in some individuals. These infections have been documented mainly in immunocompromised and often young individuals, both captive penguins and wild penguins in rehabilitation settings. 8,13,14 Herpesviruses have been found in apparently healthy individuals in a wild population of Magellanic penguins in South America. 11 This report describes a case of herpesvirus encephalitis in a captive little blue penguin chick (Eudyptula minor) diagnosed via histology, polymerase chain reaction (PCR), and in situ hybridization (ISH).
In February 2018, a little blue penguin embryo presented with a defect in the eggshell from trauma. The egg was being incubated by surrogate parents as the original parents were not properly incubating the egg. There was a 1 × 5 mm depression in the egg, not in the air cell, with several radiating hairline fissures approximately 2 cm in length. The egg surface was cleaned with iodine and cyanoacrylate (GLUture, Zoetis Inc, Kalamazoo, MI) was applied to the fissures followed by 1% silver sulfadiazine (Ascend Laboratories LLC, Parsippany, NJ), then covered with a transparent dressing (Tegaderm, 3M, St. Paul, MN). The chick successfully hatched 8 days later and had normal weight gain. However, the chick was unexpectedly found dead 11 days post hatching.
A necropsy was performed within 16 hours of death. Grossly, the lungs were bilaterally congested. All other tissues were grossly unremarkable. Aerobic cultures of the left abdominal air sac and left kidney were submitted to IDEXX laboratories (Grafton, MA). Samples were plated on sheep blood agar and MacConkey’s agar and incubated aerobically at 35 °C for up to 48 hours. When positive growth was observed, the isolate was identified by matrix-assisted laser desorption/ionization-time of flight (MALDI-TOF) mass spectrometry (Bruker Scientific LLC, Billerica, MA). Samples of all organs were collected and fixed in 10% neutral-buffered formalin (Thermo Scientific, Richard Allan Scientific, Kalamazoo, MI) for 5 days, processed routinely to paraffin blocks, sectioned at 5-µm thickness, mounted on frosted glass slides, and stained with hematoxylin and eosin (HE).
Corynebacterium species was isolated from the abdominal air sac culture and the renal culture was negative. Histologic examination showed marked changes in the brain. Moderate neuronal necrosis was present at all levels especially in the optic lobes and cerebrum (Fig. 1). Numerous neurons in the most severely affected parts of the brain had intranuclear amphophilic inclusions (Fig. 2). Some neurons had intranuclear inclusions filling almost the entire nucleus resulting in nuclear swelling; fewer neurons had intranuclear inclusions surrounded by halos (Fig. 2). Moderate gliosis and mild multifocal perivascular lymphoid cuffing were also seen. There was mild infiltration of lymphocytes in the meninges, primarily around blood vessels. Additional changes in other organs were considered to be unrelated processes including mild interstitial pneumonia with congestion and mild parabronchial hemorrhage, mild Kupffer cell activation and hepatic portal lymphocytic inflammation, a few activated macrophages in the red pulp of the spleen, and mild myofiber necrosis in the heart. All remaining tissues were histologically normal.
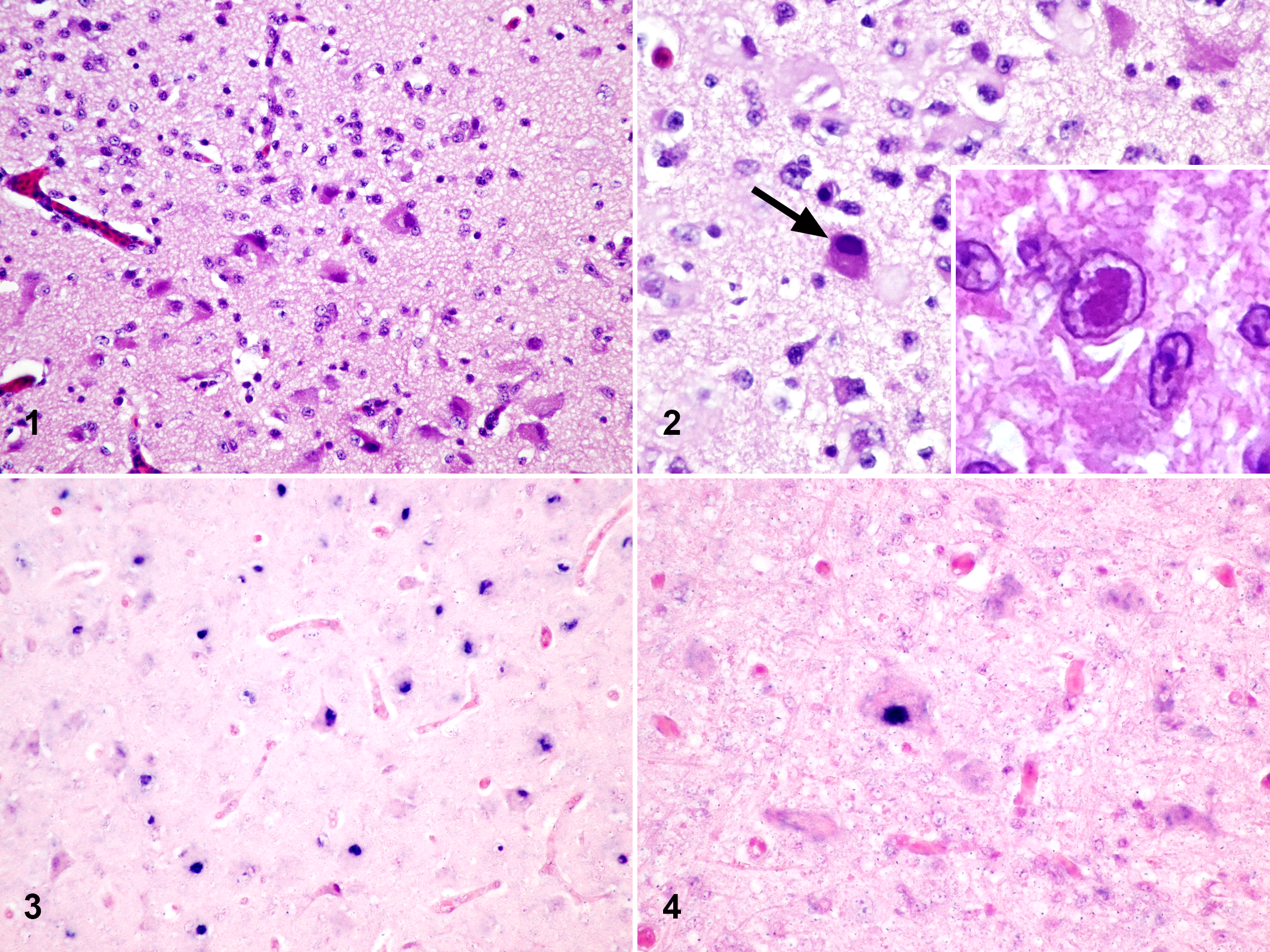
Herpesvirus encephalitis, brain, little blue penguin.
Based on the nonsuppurative encephalitis with intranuclear inclusions, viral testing for paramyxovirus and herpesvirus was performed on paraffin blocks of the brain. Generic paramyxovirus PCR was negative. 15 For generic herpesvirus PCR, DNA was extracted from paraffin-embedded tissues with the described lesions using the DNA extraction kit from Qiagen (QIAamp DNA FFPE Tissue Kit, Qiagen, Germantown, MD). Generic herpesvirus PCR targeting the polymerase gene was positive, amplifying an approximately 215-bp fragment from the penguin DNA sample. 18 The purified product was sequenced using bidirectional Sanger sequencing. A 181-bp sequence that excluded primer sequences was obtained and analyzed by BLAST against the GenBank (GB) database (Supplemental Materials 1). The case sequence was found to have 100% and 98% nucleotide sequence similarities to Sphenicid alphaherpesvirus 1 (GB Accession No. NC_033464.1) and Penguin herpesvirus 2 (GB Accession No. KR 338839.1), respectively.
While the sequence data are most supportive of an infection with sphenicid alphaherpesvirus 1, there are only limited sequence data available for Penguin herpesvirus 2 and the sequenced PCR product was only 215 bp. A newly designed oligoprobe with 100% homology to both viruses was developed and used for ISH. ISH using the digoxigenin-labeled oligonucleotide probe (DIG-5′-TCCATGTCTTCCTGTAGCTGCCACCGTAACTACGATAGGGAGGA-3′) was performed on formalin-fixed, paraffin-embedded brain tissue on an automated system (Discovery XT; Ventana Medical Systems, Tucson, Arizona) following a previously published approach. 17 The ISH labeled the intranuclear inclusions and neuronal nuclei within necrotic areas (Figs. 3, 4). A probe against canine herpesvirus used as the negative control had no labeling. No positive control was available; however, the positive signal was specific to cells that had intranuclear inclusions or were in necrotic areas. The histologic lesions, PCR, sequencing, and ISH results are consistent with herpesviral encephalitis, most likely caused by Sphenicid alphaherpesvirus 1.
The virus source and mode of transmission in this case are unknown. Exposure as an embryo when the egg was compromised prior to hatching and/or soon after hatching is possible. Also, the virus source could be fecal material and/or sputum from the original parents, the surrogate brooding individuals, or another source. The little blue penguins were housed in the same exhibit and holding space as 2 other penguin species—African penguins (Spheniscus demersus) and southern rockhopper penguins (Eudyptes chrysocome). There have been no cases of diptheritic stomatitis or necrotizing enteritis in any of the penguins in the collection, lesions thought to be associated with spheniscid alphaherpesvirus 1 in banded penguins. 14
A variety of diseases have been reported in wild little blue penguins with the exception of herpesvirus infection. 5,10,12 Herpesviruses have only rarely been documented in wild seabird populations, and our findings broaden the current understanding of viral infections in penguins. 4,11 ISH confirmed the presence of herpesvirus DNA in the nuclei and intranuclear inclusions of neurons within necrotic areas of the brain, while limited sequencing data were most consistent with sphenicid alphaherpesvirus 1. There have been no other cases of encephalitis in the little blue penguin colony or the other housed penguin species, African and southern rockhopper penguins.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Tuxbury_et_al - Herpesvirus Encephalitis in a Little Blue Penguin (Eudyptula minor)
Supplemental Material, Combined_supplemental_materials-Tuxbury_et_al for Herpesvirus Encephalitis in a Little Blue Penguin (Eudyptula minor) by Kathryn A. Tuxbury, Charles J. Innis, Tuddow Thaiwong, Annabel G. Wise, Roger Maes, Michael M. Garner and Matti Kiupel in Veterinary Pathology
Footnotes
Acknowledgements
We thank the Penguins Department staff at New England Aquarium for their diligent care of the penguin colony.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
