Abstract
During a previously reported program-wide Corynebacterium bovis outbreak, both immunocompetent depilated (dep/dep) mutant mice and transgenic mice that express the papillomavirus E6 oncoprotein became persistently infected with C. bovis. An orthokeratotic, hyperkeratotic, acanthotic dermatitis developed in the C. bovis–infected dep/dep mice, which remained C. bovis PCR-positive for >45 days prior to euthanasia as part of the program-wide C. bovis eradication effort. Since both affected strains of mice have altered skin homeostasis, immune status or the presence of hair may not alone be sufficient to explain strain susceptibility to C. bovis–related cutaneous disease. In order to avoid invalidation of preclinical studies due to C. bovis infection, it may be necessary to isolate immunodeficient mouse strains, implement facililty-wide surveillance for C. bovis, and sterilize equipment with vaporized hydrogen peroxide.
Keywords
The spontaneous depilated (dep/dep) homozygous mutation in mice, mapped to chromosome 4 (MGI:1915518), results in a recessive phenotype, due to a defect in the epidermis, characterized by variable hair loss with thinner and shorter hairs remaining in a greasy coat. 10,15,19 The B6C3Fe a/a-Zdhhc21dep /J (dep/dep) mutant carries a 3-bp deletion resulting in the loss of a single phenylalanine residue (Phe233del) close to the C-terminus of zinc finger, DHHC domain containing 21 (ZDHHC21) and loss of its palmitoyl-acyl transferase (PAT) activity. 12 Normal PAT activity provides a posttranslational modification of specific intracellular proteins, adding the fatty acid palmitate to specific cysteine residues, which is reversible, and regulates membrane localization and trafficking of target proteins. 5,12 Postnatally in dep/dep skin, mutant ZDHHC21Phe233del is not characterized by punctate immunostaining in the post-mitotic cells of Henle’s layer or the hair shaft cuticle in anagen and catagen follicles, as is wild type ZDHHC21, and epidermal expression of the terminal differentiation markers loricrin and filaggrin is delayed. 12 Cutaneous histopathology of dep/dep mice includes hyperplasia of the interfollicular epidermis (IFE) and sebaceous glands, disoriented hair follicles, delayed hair shaft differentiation, hair shaft structural abnormalities, and clumps of pigment reflecting remains of degenerating follicles. 10,12,19
The opportunist Corynebacterium bovis, a lipophilic, gram-positive coryneform bacteria, can reside on the skin of mice, more persistently so on the skin of immunodeficient mice, including nu/nu, Prkdcscid , SCIDbeige, NOD/LtSz-scid, NOD/LtSz-scid/IL2Rγnull (NSG), and NOD-scid IL2Rgnull-3/GM/SF (NSGS) strains. 2,11 C. bovis–infected immunodeficient mice can develop an orthokeratotic, hyperkeratotic, acanthotic dermatitis, may shed C. bovis–infected keratin flakes that are spread by airborne and fomite transmission, and may transmit C. bovis in resected patient-derived xenografts (PDX) or allograft tumors, resulting in broad contamination of the preclinical research facility. 2,3,9,11,16,20 Cutaneous C. bovis infection diminishes patient-derived chronic myelomonocytic leukemia (CMML) engraftment of NSGS mice, 21 and anecdotally reduces confidence in preclinical data validity derived from infected murine inventories. 3,9,16 We have previously reported the eradication of C. bovis from a preclinical setting achieved using facility-wide C. bovis polymerase chain reaction (PCR) murine and environmental surveillance, culling of C. bovis PCR positive mice, and sterilization of equipment and rooms with vaporized hydrogen peroxide (VHP) to prevent further study invalidations. 11,13,14
The mechanism by which C. bovis colonizes murine skin and induces acanthosis and hyperkeratosis has not been described, although immunodeficiency and “hairlessness” have been suggested as predisposing factors of infection. 2,7,20 Although both immunodeficient and immunocompetent mice can become at least transiently infected, including immunocompetent “haired” heterozygous nu/+ mice, 2,7,20 reportedly only immunodeficient strains permit “persistent” infection that develops into clinically apparent hyperkeratosis, although a subpopulation of a single “hairless” immunocompetent strain, the outbred SKH1-Hrhr mice (MGI: 1856057), do develop hyperkeratosis when experimentally infected with C. bovis. 2 That an immunocompetent murine strain may become naturally, persistently C. bovis infected during an outbreak, and develop clinically apparent and microscopically confirmed hyperkeratosis has not been previously reported, although suggested in a letter anecdotally describing symptomatic C. bovis infection in waltzing pet mice. 6
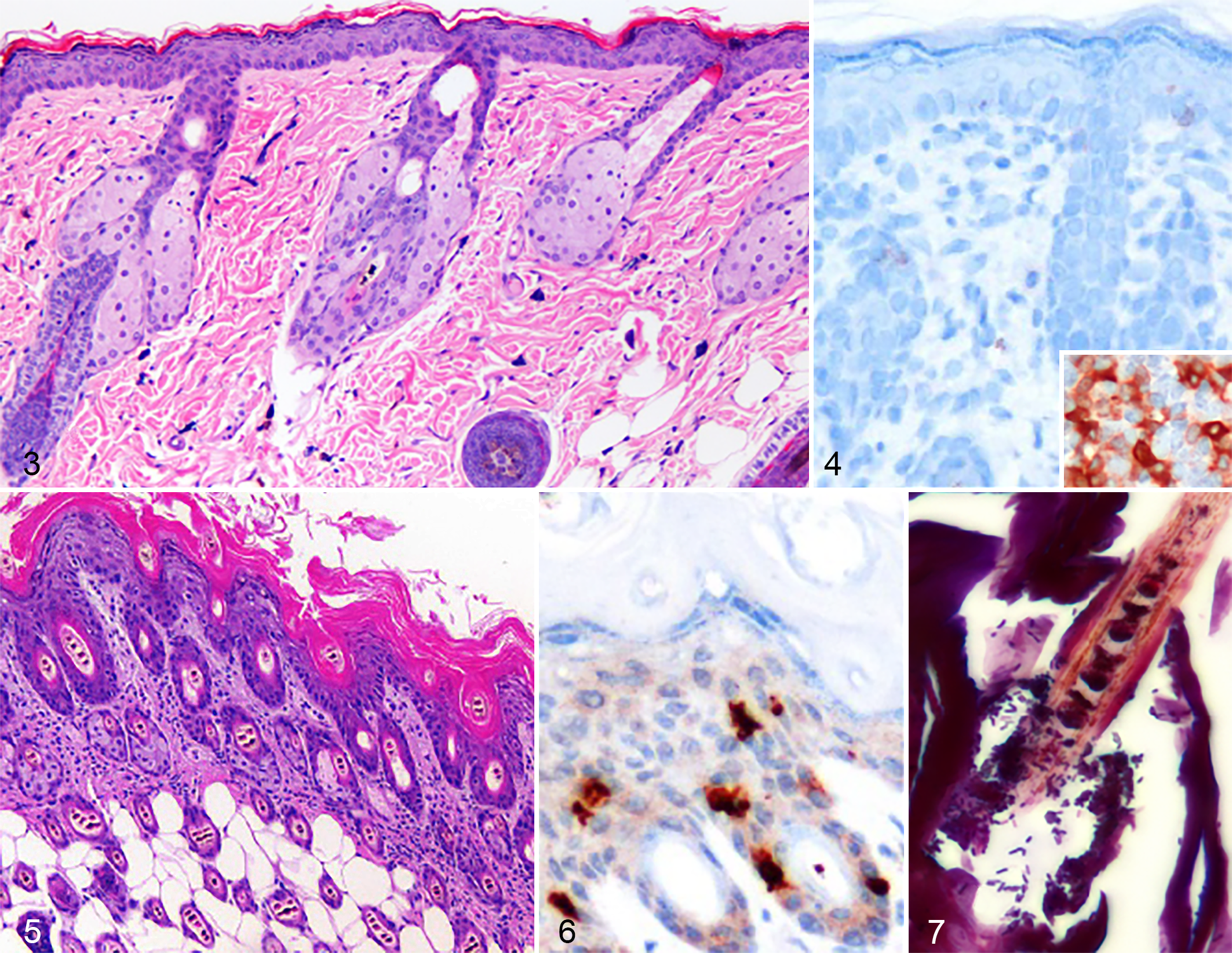
While responding to the previously reported natural outbreak of C. bovis, wherein a wide range of immunodeficient strains became persistently infected and C. bovis PCR-positive, confirmed by culture and identification, 11 we recognized that some immunocompetent strains had also become persistently infected, including transgenic mice that express the human papillomavirus (HPV) E6 oncoprotein (FVB-Tg(KRT14-HPV16E6)5737Plam/Nci, MGI:3521744), 18 and epidermal mutant dep/dep mice. Epidermal mutant dep/dep mice remained persistently infected and C. bovis PCR-positive for >45 days, and developed clinically apparent hyperkeratosis. Unfortunately, tissues of the C. bovis–infected HPV-E6 oncoprotein expressing transgenic mice were not preserved during the eradication effort. C. bovis–infected dep/dep mice were euthanized as part of the program-wide C. bovis eradication effort, and skin specimens collected and processed for microscopic analysis. Deparaffinized thin sections were stained with either hematoxylin and eosin (HE), Gram stain (Brown & Brenn, Poly Scientific, Bay Shore, NY), or rabbit anti-human CD3-immunostain (Clone SP7, Roche Molecular Systems, Pleasanton, CA) that also reacts with murine T-lymphocytes, confirmed by using murine spleen as a positive control tissue. CD3 immunostaining used antigen retrieval (Cell Conditioning 1, Catalog #950-124, Ventana Medical, Tucson, AZ), a 1:100 dilution of the primary anti-CD3 monoclonal antibody, a secondary anti-rabbit antibody (OmniMap anti-Rb HRP, Catalog #760-4311, Ventana Medical), 3,3′-diaminobenzidine (DAB) as chromogen, an automated system (Ventana Medical), and a hematoxylin counterstain. During the previously reported eradication, C. bovis PCR environmental surveillance supported the notion that a contributing source of C. bovis may have been human skin or upper respiratory mucous membranes, since C. bovis remained PCR detectable only on vivarium office computer keyboards in the final months of eradication efforts, after being otherwise eradicated from all murine inventories and all other rooms and equipment. 11 To date, C. bovis has remained undetected program-wide by monthly PCR environmental monitoring for >3 years, likely due in part to regular program-wide VHP sterilization efforts. 11,13,14
The dep/dep strain was established by a cross-intercross mating to C3HeB/FeJ × C57BL/6J (B6C3F1) mice, 10 hence herein, adult B6C3F1 skin (n = 2 mice) was used as wild type background strain control, and compared to adult dep/dep skin (n = 4 mice). Uninfected nursling dep/dep skin (n = 2 mice) was compared to C. bovis–infected nursling dep/dep skin (n = 4 mice). Skin from uninfected B6C3F1 and uninfected dep/dep mice were obtained from C. bovis PCR-negative cohorts, after the eradication of C. bovis program-wide. All such specimens were derived from the dorsal or ventral truncal skin of euthanized mice, and comparisons made to like regions. Clinically, pelage truncal hairs of dep/dep colony mice were thin, short, matted, greasy, and variably lost beginning at 1 to 3 weeks of postnatal age, so that dep/dep adults developed zonal or complete alopecia. In some adult dep/dep mice, cilia (eyelashes) were also lost, so that affected eyes required frequent cleaning. Compared to the thin epidermis and associated sebaceous glands of adult background strain B6C3F1 mice, the IFE and sebaceous glands of adult dep/dep mice were hyperplastic, and the acanthotic epidermis had mild orthokeratotic hyperkeratosis, with an occasional small epidermal cyst containing keratinized material, present. Scattered follicles of dep/dep mice contained deformed hair shafts, were occasionally disoriented and misshapen, and follicle remnants were represented by scattered clumps of pigment.
In contrast to C. bovis PCR-negative and clinically unaffected dep/dep nurslings (Fig. 1), C. bovis–infected dep/dep nurslings were covered with variable-sized adherent yellow-white flakes (Fig. 2). Microscopically, compared to uninfected dep/dep nursling epidermis (Figs. 3, 4), C. bovis–infected dep/dep nursling epidermis had marked orthokeratotic hyperkeratosis, and the dermal-epidermal junction was infiltrated with scant CD3-positive T lymphocytes (Figs. 5,6). Numerous gram-positive pleomorphic rods were present in the stratum corneum, surrounding hair shafts (Fig. 7), and in hair follicles, often arranged in palisading or irregularly branching angular arrays. The hyperkeratotic skin lesions, dermal-epidermal infiltration by CD3-positive T-lymphocytes, and gram-positive bacteria only in C. bovis–infected dep/dep mice supported the impression of an opportunistic infectious dermatitis.

Skin homeostasis relies on the differentiated progeny of lineage-restricted stem cells, habitat niche-specific microbiome, cutaneous innate and adaptive immunity, and on interactions between these major components. 1,4 Progeny of lineage-restricted stem cells differentiate to form either sebaceous glands, hair follicles, or the IFE. Disintegrating sebocytes release lipid-rich sebum that lubricates the skin and hair, and are replaced by progeny from the periphery of the gland. Hair shafts are shed and replaced in a cycle of catagen regression, telogen rest, and anagen regeneration. Shed cornified squames are replenished by progeny of mitotically active epidermal basal cells that differentiate to become spinous cells, which become enucleated granular cells with intercellular tight junctions, which become terminally differentiated squames. In addition, the composition of cutaneous microbiota is influenced by distinct pH, temperature, moisture, sebum content, and topography of diverse skin microenvironments. 20 Resident cutaneous microorganisms control the expression of innate antimicrobial peptides (AMP), resident oxygen species, and components of the complement system. 1,8 Skin homeostasis, the balance of self-renewal and differentiation, and the evolving dialog between the skin microbiome and immunity, is upset by the dep/dep mutation, and the HPV-E6 oncoprotein.
Corynebacterium species cannot produce their own lipids, making them well suited to reside among the lipid-rich sebum and stratum corneum. 17 The excessive sebum of the hyperplastic sebaceous glands, and abundant lipids of the acanthotic IFE of dep/dep mice may provide an ideal lipid-rich niche for C. bovis colonization and growth. Cutaneous dysbiosis with overrepresentation of Corynebacterium species may contribute to an undetermined mechanism of epidermal hyperplasia, hyperkeratosis, and skin inflammation in the opportunistically infected host. Disturbances in skin homeostasis may contribute to C. bovis infection, not simply immunodeficiency or “hairlessness.” C. bovis infection of the immunocompetent transgenic mice that express the HPV-E6 oncoprotein (MGI:3521744) observed during the C. bovis outbreak 11 may also be referable to altered skin homeostasis due to human keratin 14 promoter directed E6 oncogene expression in the hair follicle and epidermal basal layer, and the resultant epidermal hyperplasia of that strain. Altered skin homeostasis, follicle and hair shaft development, or modulated immunity may contribute to the experimental C. bovis infection and hyperkeratosis of the “immunocompetent” and “hairless” SKH1-Hrhr mice, 2 which have modestly reduced numbers of splenic T helper cells, but normal cell immune function.
Murine immunity has been progressively disabled, either spontaneously and retained by breeding, or also intentionally in severe combined immunodeficient strains by targeted gene disruptions, to improve human specimen engraftment, or unexpectedly during the creation of other genetically engineered mutants. The functional immune status of mutants or genetically engineered strains is not always fully defined. As larger and more diverse inventories of mutant, genetically engineered, and immunodeficient strains are maintained, each potentially prone to C. bovis colonization and the development of clinically apparent hyperkeratosis, the risk that preclinical research data may be invalidated due to C. bovis infection may increase. 21 Preventing C. bovis colonization of murine skin may require more than the isolation of immunodeficient murine strains, since even immunocompetent epidermal mutant dep/dep mice may become C. bovis–infected and may shed infectious keratin flakes that contaminate core resources, including imaging and intravital microscopy, thereby limiting movements of personnel and mice in the contaminated facility. Preventing a C. bovis outbreak may require a comprehensive, program-wide approach involving PCR environmental and murine surveillance, and frequent VHP sterilization of equipment and rooms. 11,13,14
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
