Abstract
Impaired keratinocyte differentiation has recently been suggested as a key event in equine hoof canker development. Koilocytotic appearance of keratinocytes, one of the most characteristic morphological alterations in hoof canker tissue, is also a common marker for papillomavirus (PV) infection, and bovine PV-1 and/or -2 (BPV-1/2) has previously been detected in equine canker patients. Therefore, the present study aimed to correlate the frequency and severity of koilocytotic keratinocytes with BPV detection in hoof canker samples. Hoof tissue of 5/18 canker-affected horses and 2/6 control horses tested positive for BPV-1/2 DNA using polymerase chain reaction. Thus, no association between the presence of BPV-1/2 papillomaviral DNA and koilocytotic appearance was found. Proteins associated with but not specific for PV infection were also investigated. Using immunohistochemistry, specific adhesion molecules (E-cadherin and β-catenin) and intermediate filaments (keratins 6 and 14) important for intact epidermal barrier function and keratinocyte differentiation were documented in control samples (n = 6) and in hoof canker tissue samples (n = 19). Altered expression patterns of intermediate filaments and adhesion molecules were demonstrated in canker tissue, confirming the importance of incomplete keratinocyte differentiation, as well as the crucial role of keratinocyte differentiation in hoof canker.
Keywords
Equine hoof canker (pododermatitis chronica verrucosa sive migrans) has been described as progressive pododermatitis of the equine hoof with extensive proliferation of the dermal papillary body and absent epidermal cornification. 18,36 Morphological and protein-based analyses are infrequent in hoof canker research. In a recent study, 3 we showed that the increase in tissue volume occurring in hoof canker is not based on increased proliferation of dermal or epidermal cells, but is instead associated with hypertrophy of keratinocytes. 3 These were morphologically characterized as large keratinocytes featuring a perinuclear vacuole, clear cytoplasm, and pyknotic nuclei with inconspicuous nucleoli, therefore resembling koilocytotic keratinocytes 3 according to earlier reports, where koilocytes were defined as epithelial cells containing an eccentric, hyperchromatic, moderately enlarged nucleus that is displaced by a large perinuclear vacuole. 20,26 This abnormality was first described in vaginal cytology to characterize intraepithelial lesions of the cervix 38 associated with long-standing inflammation or infection (viral or other). 20,38 Nowadays, koilocytosis is considered as the major histopathological finding when determining HPV infection in humans. 26,42 Koilocyte-like cells have also been described in bovine papillomavirus (BPV)-infected keratinocytes in early-stage equine sarcoids. 5 As consistent detection of BPV DNA in canker lesions, intact skin, and mononuclear blood cells of 25 canker-affected horses has been reported, 9 we hypothesized that koilocyte-like ballooning degeneration of the keratinocytes in equine canker samples is associated with the presence of BPV-1/2 DNA in canker tissue. Therefore, the first aim of the present study was to assess a possible association of koilocyte-like keratinocytes with the presence of BPV-1/2 DNA.
Furthermore, laser capture microdissection of keratinocytes and fibroblasts in equine sarcoids have shown that in some sarcoids the epidermis was infected with BPV-2, whereas the dermis was infected with BPV-1. 5 Therefore, a second objective was to ascertain the presence of epidermal and dermal portions in canker and control tissue samples, as well as of koilocyte-like keratinocytes in canker tissue samples, which were used for BPV-1/2 screening.
As PVs interfere with the intermediate filaments, they cause disruption of the intracellular network 4,21,50 and can reduce membrane expression of the cell-adhesion molecule E-cadherin. 28 In hoof canker patients, the disruption of tissue integrity is a relevant clinical factor, as maceration and disintegration of the hoof facilitates invasion of pathogens and secondary infection, for example, Treponema sp. and Spirochetes. 27,44 In a previous study, we identified increased PCNA levels in poorly differentiated keratinocytes in hoof canker samples, as well as a lack of keratinization and desquamation. 3 Keratinocyte differentiation is functionally associated with the expression of specific cytokeratin intermediate filaments, 14 which structure the cytoplasm in order to resist external stresses and provide tissue integrity and stability. 17 For the present study, keratin 6 (K6) was selected for evaluation as it is essential for the maintenance of keratinocyte integrity in inflamed epidermis. 31 Although K6 is reported to indicate mainly proliferative keratinocytes, 16 the pro-inflammatory cytokine interleukin-1 also induces the synthesis of K6 in epidermal keratinocytes. 25 As inflammation is a common finding in hoof canker tissue, 3 differences between canker-affected regions versus healthy control tissue regarding K6 expression were expected. Additionally, keratin 14 (K14) was investigated, as its expression is downregulated by increasing keratinocyte differentiation, 1 which is lacking in canker, 3 where increased levels of K14 were expected.
Dysregulation of epithelial differentiation, associated with changes in K6 and K14 expression, is also accompanied by the loss or reduction of the adhesion molecules E-cadherin and β-catenin. 46,55 The E-cadherin/β-catenin complex is the major adhesive component of adherence junctions in the epidermis and crucial for intact epidermal barrier function. 34,55 E-cadherin acts as the major partner of β-catenin at the plasma membrane, whereby β-catenin has both adhesion and signaling function in keratinocytes. 34 Therefore, it was hypothesized that keratinocyte expression patterns of the adhesion molecules E-cadherin and β-catenin differ between canker and healthy tissue.
In summary, the aim of the present study was to investigate expression patterns of K6, K14, E-cadherin, β-catenin, and to perform molecular analyses to compare the frequency of BPV-1/2 DNA in canker and control tissue.
Materials and Methods
Sample Collection
Tissues were assessed from 19 horses treated for hoof canker at the Equine University Clinic of the University of Veterinary Medicine, Vienna, Austria. 3 Hoof canker was diagnosed based on its typical clinical appearance. 2,40,41 Tissue, including the macroscopically changed areas, as well as a small margin, were removed based solely on clinical necessity during routine hoof surgery under regional anesthesia. Samples were then taken from the deep regions of the removed tissue of the frog, as the most commonly canker-affected region, including the dermo-epidermal junction. Samples of 7 horses euthanized for reasons other than hoof or skin disease, including dermal neoplasia, were used as control tissue. Prior to inclusion as controls, samples of all hooves and areas of skin were examined macroscopically and histologically for absence of pathological alterations. Thus, hoof tissue of corresponding regions of the frog of 6/7 control horses were sampled post mortem, and skin of 1/7 control horse was used for the establishment of immunohistochemical (IHC) protocols. Tissue sampling and evaluation, as well as anonymized publication of the data obtained, were carried out with the written consent of the horse owners, given on admission of the horse to the clinic.
Screening for BPV-1/2 E5 and L1 DNA
Tissue samples of the 19 canker-affected horses were taken as described above and, additionally, tissue samples of the 6 control horses were used to document the BPV-1/2 presence in comparison to previously published results for normal skin. 6,52 For histological, immunohistochemical, and molecular analyses, samples were formalin-fixed and paraffin-embedded. Native samples for molecular analyses were stored at −196°C or were directly used for DNA extraction immediately after surgery and euthanasia, respectively.
DNA from canker and control tissue was purified by using DNeasy Blood & Tissue Kit (Qiagen, Hilden, Germany) for native tissue and QIAmp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany) for formalin-fixed paraffin-embedded (FFPE) tissue according to manufacturer’s instructions. Following DNA extraction, amplification-compatible purity of isolates was confirmed by routine equine β-actin PCR (polymerase chain reaction). 7 One canker case was excluded from PCR analyses, because no suitable quality DNA was extracted. Afterwards, 2 µl DNA aliquots of the DNA isolates of native tissue were analyzed using a PCR protocol allowing detection of ≤10 copies of BPV-1/2 E5 or L1 DNA, as published previously. 7 Prior to analyses of DNA isolates of FFPE canker and control tissue, a PCR protocol was established, using previously tested BPV-1-positive FFPE sarcoid tissue. 7,8 A minimum of one negative control, one positive control, and one no template control (NTC) were included in each reaction. Previously analyzed BPV-1-positive tumor tissue (nucleic acids stored at −20°C) derived from sarcoid-affected horses, 7,8 as well as a BPV-1 plasmid served as positive controls. Amplification products (16 μl) were visualized on 1.5% or 2% Tris/acetate agarose gels by ethidium bromide staining.
Histology and Immunohistochemistry
Histological sections from FFPE tissue were prepared for morphological evaluation (hematoxylin and eosin staining) and IHC assays as published earlier. 3 Proteins of interest were marked using a polyclonal rabbit anti-cytokeratin 6 antibody (Biorbyt, Cat. No.: orb10412, Cambridge, UK; dilution 1:100; without pretreatment), a monoclonal mouse anti-cytokeratin 14 antibody (clone LL02; Leica Biosystems, Cat. No.: NCL-L-LL02, Newcastle, UK, dilution 1:100, pretreatment for 2 hours in a water bath at 65°C with Tris-EDTA pH 9.0), a monoclonal mouse anti-β-catenin antibody (clone 9G2; Acris, Cat. No.: AM00020PU-N, Herford, Germany, dilution 1:100, pretreatment for 2 hours in a water bath at 80°C with Tris-EDTA pH 9.0), and a polyclonal rabbit anti-E-cadherin antibody (Santa Cruz Biotechnologies, Cat. No.: sc-7870, Santa Cruz, CA, dilution 1:150, pretreatment for 2 hours in a water bath at 65°C in Tris-EDTA pH 9.0).
These antibodies were then detected with a poly-HRP-anti-rabbit and anti-mouse (respectively) secondary antibody (BrightVision Immunologic, Duiven, The Netherlands) using 3,3′-diaminobenzidine-tetrahydrochloride substrate (Sigma Aldrich) in Tris-HCl buffer, pH 7.4, and 0.03% H2O2 as chromogen.
Negative controls were performed by substitution of the primary antibodies with phosphate-buffered saline (PBS) to demonstrate the specificity of the secondary system. Evaluation of the histological sections was performed using light microscopy (Polyvar; Reichert-Jung, Vienna, Austria) and a digital camera (Nikon, Austria). Sections of healthy equine skin were used as positive controls for the establishment of the IHC staining protocols.
Morphological and Immunohistochemical Analyses
Morphological analysis focused on intensity and frequency of koilocytotic appearance of keratinocytes in hoof canker tissue; the scoring was performed as previously documented. 3 Briefly, blinded analysis was performed by 1 person (CG). Koilocytotic appearance was graded for intensity and frequency as mild (+), moderate (++), or severe (+++) in 3 sections per sample per horse with a minimum of 2 samples per horse examined, resulting in a minimum of 6 sections evaluated per horse.
In the stratum basale and the stratum spinosum of surgically removed hoof canker tissue, and there in regions with typical canker-associated morphological alterations and in adjacent regions without these typical morphological alterations (Supplemental Fig. S1), as well as in healthy control tissue, the presence (+) or absence (−) of each of the proteins was documented (Tables 2, 3). Intracellular protein distribution patterns in canker and control tissue were compared for all investigated proteins.
Results
BPV-1/2 E5 and L1 DNA Detection in Canker and Control Hoof Tissue
From 36 canker isolates (18 native tissues, 18 FFPE tissues) derived from the 18 canker-affected horses with positive β-actin PCR, 4/18 native tissues tested positive for a 266 bp segment of the major capsid gene L1, and 1/18 FFPE tissue tested positive for a 497–499 bp segment of the major oncogene E5. In total, viral DNA was found in hoof horn of 5/18 canker-affected horses. No association between the presence of BPV-1/2 DNA and the presence or grade of koilocytotic keratinocytes was found (Table 1).
Summary of Morphological and Molecular Analyses of Hoof Tissues of 19 Canker-Affected Horses.a
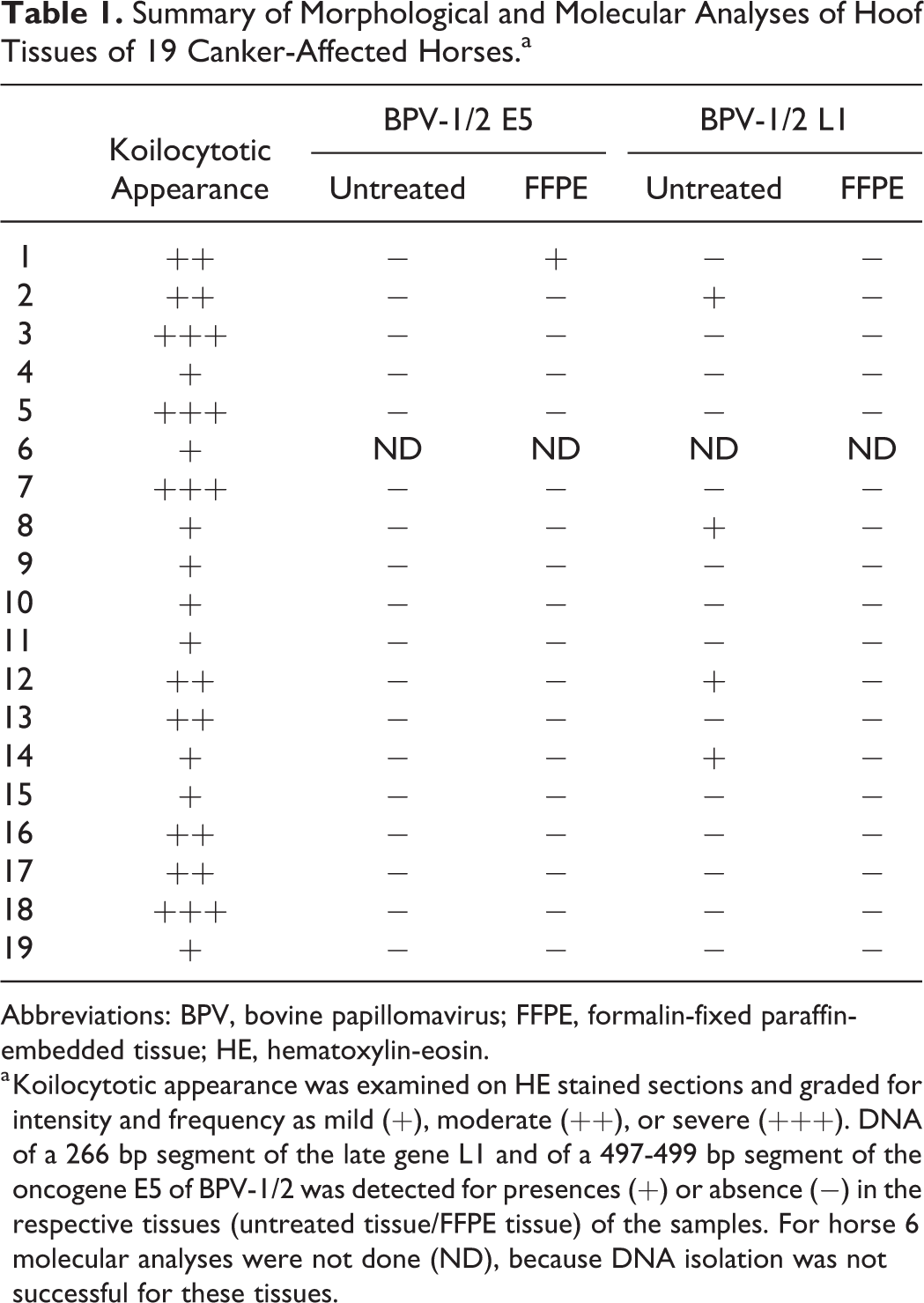
Abbreviations: BPV, bovine papillomavirus; FFPE, formalin-fixed paraffin-embedded tissue; HE, hematoxylin-eosin.
a Koilocytotic appearance was examined on HE stained sections and graded for intensity and frequency as mild (+), moderate (++), or severe (+++). DNA of a 266 bp segment of the late gene L1 and of a 497-499 bp segment of the oncogene E5 of BPV-1/2 was detected for presences (+) or absence (−) in the respective tissues (untreated tissue/FFPE tissue) of the samples. For horse 6 molecular analyses were not done (ND), because DNA isolation was not successful for these tissues.
A total of 12 DNA isolates (6 native tissues, 6 FFPE tissues) derived from the 6 control horses were run in parallel. One native tissue and 1 FFPE tissue tested positive for a 266 bp segment of the major capsid gene L1, showing the presence of papillomaviral DNA in the hoof horn of 2/6 healthy horses.
Morphological Analyses
The stratum germinativum was defined as the entire proliferative zone of the hoof epidermis, combining the single-layered stratum basale and the proliferative cells of the stratum spinosum. The koilocytotic appearance was graded for intensity and frequency as mild (+), moderate (++), or severe (+++) in hoof canker tissue (Figs. 1–4). In mildly affected samples, koilocyte-like keratinocytes were observed as islands between healthy keratinocytes. The affected keratinocytes were characterized by an irregular, hyperchromatic nucleus with a perinuclear halo (Fig. 1). Most of the cells showed an intact cytoplasmic matrix around this halo. Moderate alterations featured additional cellular enlargement, whereby nuclei of these koilocyte-like cells (ballooning keratinocytes) were displaced to the periphery of the cell and the perinuclear halo was enlarged over the whole cell (Fig. 2). Severe alterations were accompanied by tissue disintegration, either by eosinophilic fluid filled lacunae (Fig. 3), or by enlargement of cells showing disintegrated, empty cytoplasm (Fig. 4).
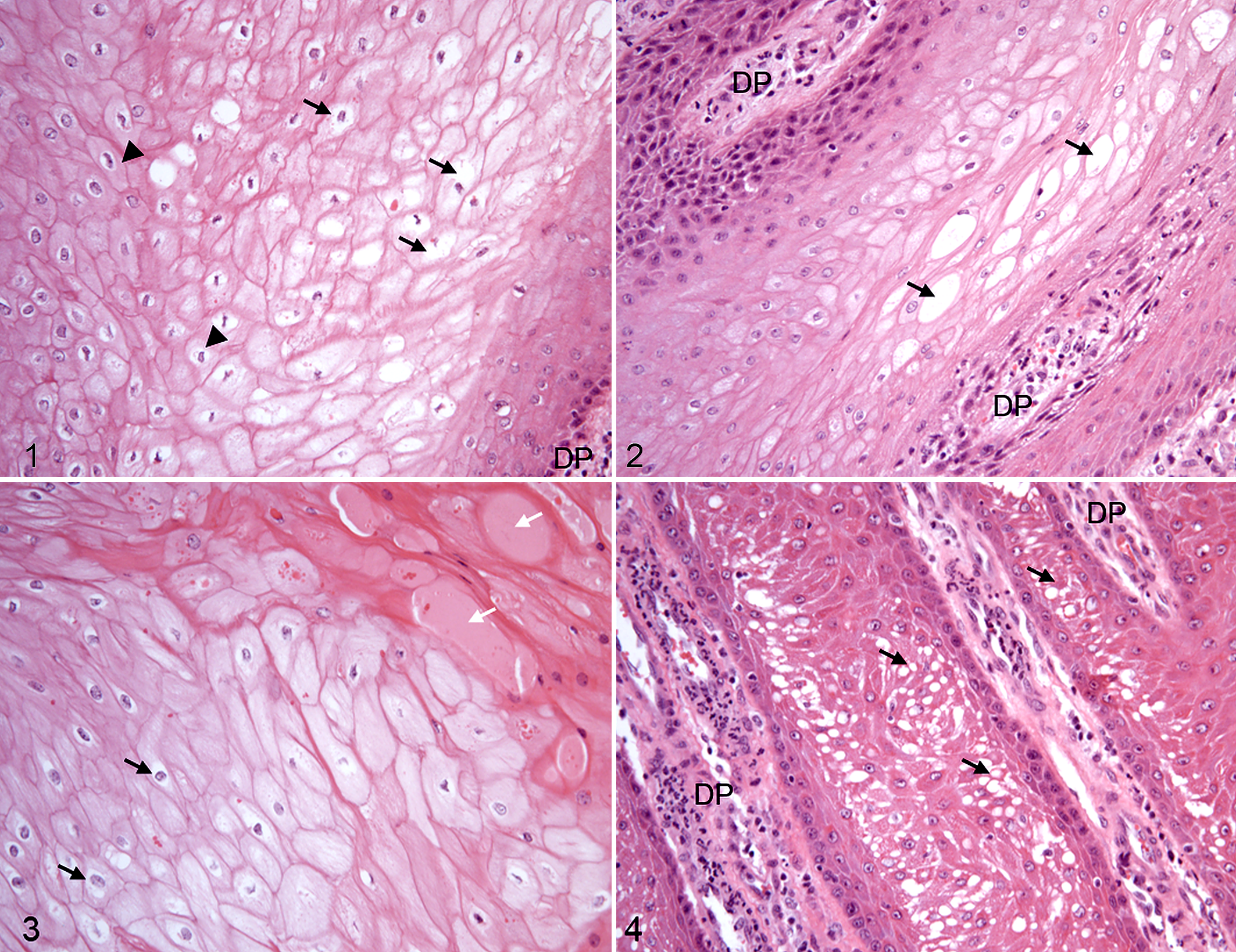
Koilocytotic appearance was graded as mild in canker tissue of 9/19 horses, as moderate in 6/19, and as severe in 4/19 horses (Table 2). Koilocyte-like cells were not observed in any tissues from the 6 control horses.
Summary of Morphological and Immunohistochemical Analyses of Hoof Tissues of 19 Canker-Affected Horses.a

a Koilocytosis was examined on HE (hematoxylin-eosin) stained sections and graded for intensity and frequency as mild (+), moderate (++), or severe (+++). The proteins of interest (E-cadherin, β-catenin, keratin 6, and keratin 14) were detected for their presences (+) or absence (−) in the respective regions (Stratum basale and Stratum spinosum) of the samples. Keratin 6 was detected partially in heterogenic, mosaic-like patterns (m) with positive and negative cells lying closely together within the same area of the respective region.
Immunohistochemical Analyses
The staining protocols for the 4 proteins investigated were established on equine skin (Supplemental Fig. S2) as expression patterns for these proteins are well known in this tissue type. The primary antibodies used in the present study were not suitable for western blot analysis; thus, the correct anatomical/intracellular localization of the targeted proteins was used for evaluation of primary antibodies. Expression patterns in healthy equine hoof (control group) are presented in Supplemental Figure S2, summarized in Table 3 and described in the respective sections below. Negative controls for IHC using PBS were performed for the respective secondary antibodies applied in the present study, which documented that there was no nonspecific staining (Supplemental Figs. S3–S6).
Summary of Immunohistochemical Analyses of Hoof Tissue of 6 Control Horses.a

a The proteins of interest (E-cadherin, β-catenin, keratin 6, and keratin 14) were detected for their presences (+) or absence (−) in the respective regions (Stratum basale and Stratum spinosum) of the samples.
The expression patterns of E-cadherin, β-catenin, and keratins 6 and 14 were described as follows in closely adjacent regions with and without typical hoof canker-associated morphological alterations, within the same surgically removed hoof canker tissue. The intracellular localization of the proteins was of interest as it is closely related to their function.
E-Cadherin
In healthy equine hoof tissue membranous E-cadherin expression was present in the keratinocytes of the stratum spinosum and in vital keratinocytes of the stratum granulosum, but it was missing in cells without nuclei (superficial layers of the stratum granulosum and all cells of the stratum corneum; Supplemental Fig. S2). In the dermo-epidermal transition zone most keratinocytes of the stratum basale exhibited membranous E-cadherin expression; this was only absent in the most basal layer of keratinocytes, which were in direct contact to the dermal tissue via the basement membrane. In deeper regions keratinocytes of the stratum basale surrounding the dermal papillae lacked the specific membranous staining and featured strong perinuclear cytoplasmic E-cadherin expression (Supplemental Fig. S2).
In hoof canker tissue membranous E-cadherin expression was rare in keratinocytes of the stratum basale and the basal layers of the stratum spinosum of the dermo-epidermal transition zone (Fig. 5), whereas strong cytoplasmic staining was observed in this region. Also, some keratinocytes of the superficial layers of the stratum spinosum and the stratum granulosum featured strong cytoplasmic E-cadherin expression in the perinuclear regions (Fig. 5). In regions with typical mild to moderate canker-associated morphological alterations (ie, koilocyte-like keratinocytes) membranous E-cadherin expression was visible in the stratum basale and the stratum spinosum, whereas neighboring regions of the stratum spinosum surrounding the dermal papillae lacked membranous E-cadherin expression and featured only slight perinuclear staining (Fig. 6). Strong cytoplasmic E-cadherin expression was observed in the stratum basale of severely affected regions, whereas distinct membranous staining patterns were demonstrated in affected keratinocytes of the stratum spinosum (Fig. 7).
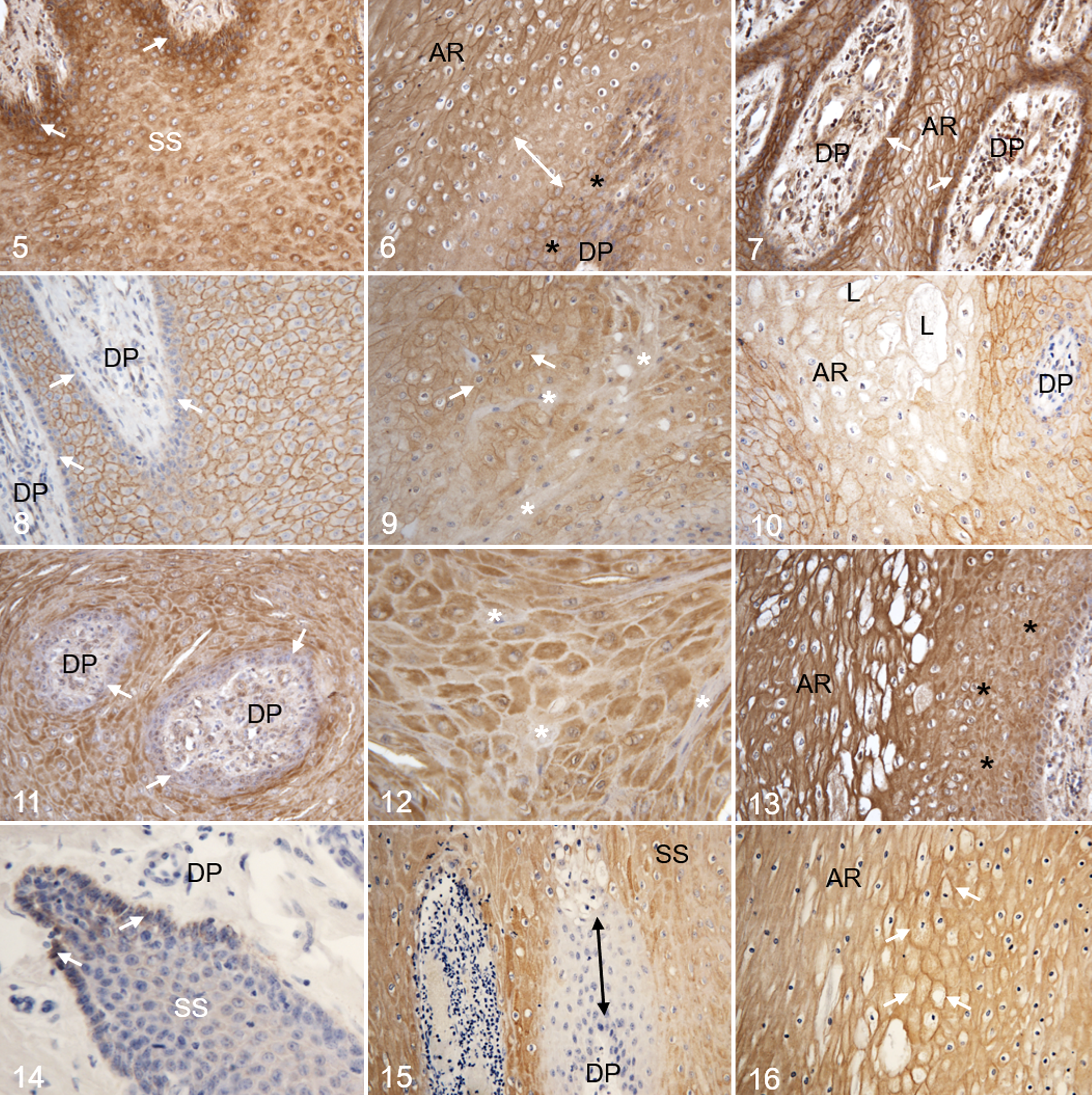
β-Catenin
Control hoof tissue featured prominent membranous β-catenin expression in the keratinocytes of the stratum spinosum and in vital keratinocytes of the stratum granulosum (Supplemental Fig. S2). There was no β-catenin staining in the majority of keratinocytes of the stratum basale or the basal layers of the stratum spinosum, and β-catenin staining was absent in all keratinocytes of the stratum corneum (Supplemental Fig. S2).
In regions adjacent to the morphologically canker-associated cellular alterations the expression pattern of β-catenin was comparable to the control tissue (Fig. 8). In mild to moderately altered regions cells partially lacked β-catenin expression or featured only cytoplasmic or a combination of cytoplasmic and membranous expression of the protein (Fig. 9). There was no β-catenin expression in keratinocytes of severely affected regions, and particularly in the enlarged keratinocytes of the stratum spinosum and the lacunae (Fig. 10).
Keratin 6
In control hoof tissue the intermediate filament K6 was expressed homogenously in the cytoplasm of the keratinocytes of the whole stratum germinativum (Supplemental Fig. S2). Keratinocytes of the stratum corneum lacked K6 expression (Supplemental Fig. S2).
Hoof canker tissue featured markedly heterogeneous expression patterns of K6. The majority of keratinocytes of the stratum basale lacked any signs of K6 expression, whereas the majority of cells of the stratum spinosum and stratum granulosum partially showed strong K6 expression, but only in the perinuclear zone (Fig. 11). A mosaic-like pattern was characteristic for the stratum spinosum in regions with typical morphological canker-associated alterations; K6 positive cells were in direct contact to K6 negative cells in these areas (Fig. 12). In these severely altered regions K6 expression of the keratinocytes was very strong and seemed increased compared to adjacent regions without the typical canker-associated alterations (Fig. 13).
Keratin 14
In healthy equine hoof tissue membranous and cytoplasmic K14 expression was present in the keratinocytes of the stratum basale and in basal layers of the stratum spinosum (Supplemental Fig. S1). In deeper regions around the dermal papillae, keratinocytes of the stratum basale and adjacent layers of the stratum spinosum showed strong staining reactions for K14, whereas keratinocytes of the remaining stratum spinosum, the stratum granulosum, and the stratum corneum lacked K14 expression (Supplemental Fig. S2).
In regions adjacent to the canker-associated cell morphology alterations, most of the keratinocytes of the stratum germinativum lacked K14 expression (Fig. 14). In regions with canker-associated alterations keratinocytes of the stratum basale and adjacent layers of the stratum spinosum surrounding the dermal papillae were negative for K14 expression, whereas the more superficial layers of the stratum spinosum featured heterogeneous cytoplasmic expression patterns for this intermediate filament (Fig. 15). In severely altered regions, cells of the stratum spinosum were positive for K14 and showed cytoplasmic expression, as well as intense membranous staining (Fig. 16).
Discussion
Koilocyte-like keratinocytes have been previously described in equine canker tissue, 3 and the first aim of the present study was to assess a possible association of koilocytosis with the presence of BPV-1/2 DNA. This was based on an earlier study, where consistent detection of BPV-1/2 DNA was found in lesions, intact skin, and mononuclear cells of 25 canker-affected horses. 9 Contrary to this, the present study found BPV DNA in hoof tissue of less than a third of the canker-affected horses and also in a third of the control horses using PCR. As BPVs, in contrast to HPVs, express their oncogenes not only in the basal layers of the equine epidermis, but also in dermal fibroblasts, 10,32,37 we used FFPE tissue sections for DNA extraction in addition to native tissue samples to guarantee the presence of dermis and epidermis in samples. The present study detected BPV-1/2 E5 DNA in only one FFPE canker sample, whereas DNA of the major capsid gene L1 was found in 4 native canker tissues, where the presence of dermal parts was not histologically ascertained. In the control samples, we found L1 DNA in one FFPE sample and in one native hoof tissue. Our results are consistent with other studies, also showing BPV-1/2 DNA presence in normal and inflamed skin samples with comparable prevalences. 5,35,52 Thus, the findings of the present study do not support a relationship between BPV-1/2 infection and equine hoof canker. However, we did not investigate PVs other than BPV-1/2, even though the presence of koilocytosis makes this a promising area of research for the future, as in humans, koilocytosis is considered a reliable marker of HPV infection. 26,42 Nevertheless, koilocytes can also be present without PV infection, 33 and koilocytosis seems less specific in horses, where koilocyte-like cells are described in predominantly inflammatory skin diseases, such as equine verrucous pastern dermatitis. In such cases, koilocytes are found mainly in the parakeratotic stratum corneum, 39 whereas in canker they are localized mainly in the stratum germinativum. 3
The second aim of the study was to investigate adhesive components of the epidermal barrier in canker-affected tissue by means of immunohistochemistry. The E-cadherin/β-catenin complex is the major adhesive component of adherence junctions in the epidermis and crucial for intact epidermal barrier function. 34,55 E-cadherin acts as the major partner of β-catenin at the plasma membrane, whereby β-catenin has both, adhesion and signaling function in keratinocytes. 34 The abundant expression of E-cadherin and β-catenin does not accurately reflect their action and function, but instead the membranous localization of these proteins (in contact to the cytoskeleton) is crucial for their function. 54 Therefore, the description of the intracellular localization of these proteins was important for the present study.
As none of these molecules have previously been described in the equine hoof, equine skin samples served as positive control for the establishment of the immunohistochemical assays. Similar to other species, E-cadherin and β-catenin were expressed in specific patterns in equine skin keratinocytes; 30,45 also expression patterns of K6 and K14 were in accordance with observations in skin samples of other species. 12,49,51 Therefore, detection of these 4 proteins using immunohistochemistry was supported by localization analysis; expression patterns were comparable to those of other species. In hoof canker tissue, E-cadherin expression was detected mainly in cytoplasmic and perinuclear regions of the keratinocytes of the stratum spinosum and stratum basale. Keratinocytes of control hoof samples featured distinct membranous staining patterns of this adhesion molecule. E-cadherin is the major intramembranous linking protein for β-catenin, 23 and therefore, the lack of distinct membranous E-cadherin expression suggests interruption of cell adhesion in canker-affected keratinocytes, which is consistent with the clinical evidence of disintegrated epidermis in hoof canker. However, the background of the characteristic translocation from membranous to cytoplasmic expression of E-cadherin in hoof canker tissue has to be investigated in more detail to clarify its role in the degenerative process. In hoof canker tissue β-catenin expression patterns were distinct from those of E-cadherin. Only in regions with hoof canker–associated morphological alterations, keratinocytes lacked β-catenin expression or showed altered cytoplasmic expression patterns compared to the membranous expression patterns in control keratinocytes. In healthy epithelial cells the cytoplasmic E-cadherin/β-catenin complex is degraded physiologically during cell cycle events, to prevent the translocation of unbound β-catenin to the nucleus. 11 The translocation of these 2 adhesion molecules from membranous to cytoplasmic expression is suggested to play a pivotal role in disruption of tissue integrity and loss of cellular barrier function, 45 as is present in hoof canker tissue. Hypoxia, reduced blood supply, or metabolic imbalances might also cause the observed changes in adhesion molecule expressions associated with keratinocyte swelling in canker tissue. Oxygen deprivation is shown to inhibit basal keratinocyte proliferation, thus inducing changes in the distribution of the epidermal adherence junction proteins E-cadherin and β-catenin. 43 Zinc deficiency, as a metabolic factor, has been reported to induce acanthosis and parakeratosis in different species including the horse. 15,22
Intact cell-cell adhesions are closely related to the cytoskeleton and specific expression patterns of keratins. 48 Also, keratinocyte differentiation is closely correlated to specific tempo-spatial expression patterns of keratins. 14 Therefore, the third aim of the study was to analyze expression patterns and intracellular distribution of specific keratins (K6 and K14) in canker-affected tissue in comparison to control tissue. Based on the results of the present study, the reduction and/or loss of K6 and K14 proteins in hoof canker–affected keratinocytes is strongly associated with the described incomplete differentiation and lack of keratinization seen in canker. 3 Keratin 6 expression is essential for the maintenance of keratinocyte integrity in inflamed epidermis and during wound healing. 31 Control hoof samples featured homogenous cytoplasmic expression of K6 within the whole stratum germinativum, whereas in hoof canker–affected tissue, only a mosaic-like pattern of keratinocytes was positive for K6. Keratinization potential of keratinocytes is highly sensitive to the loss of K6. 53 In wound healing experiments with K6-null mice, swelling and lysis of keratinocytes were observed at locations normally showing prominent K6 expression. 53 Comparable lysis was never documented in wild-type and hemizygous animals. 53 Also in hoof canker, swelling of keratinocytes (ballooning) is a major morphological alteration in canker-affected regions and the described lacunae formation might be the result of keratinocyte lysis and degradation. As demonstrated in murine keratinocytes, intracellular edema in hoof canker–associated keratinocytes might also be a result of the loss of K6 expression. In some regions, K6-negative keratinocytes featured an elongated shape similar to fibroblasts (Fig. 12) and such cells were negative for β-catenin (Fig. 9). Keratin 6 was identified to be involved in maintaining keratinocyte integrity in challenged skin tissue, whereby K6-null keratinocytes exhibit an enhanced migratory potential. 53 The switch from an epithelial to a more spindle-cell phenotype might also reflect the lack of differentiation of the equine canker–associated keratinocytes. This might be an explanation for the localization of K14 expression in hoof canker–affected tissue, where keratinocytes of the superficial layers of the stratum spinosum were positive for K14 while in control samples keratinocytes of this region lacked K14. As K14 is a marker for undifferentiated, proliferating keratinocytes, 1 its presence in superficial regions of the stratum spinosum in hoof canker–affected tissue is an indicator for this dysregulation of keratinocyte differentiation. In healthy epidermal tissue, K6 expression is related to K14 expression; 29 however, differentiation potential of keratinocytes is more sensitive to the loss of K6 than of K14 proteins. 53 Therefore, changes in K14 expression might be the result of dysregulated K6 expression and as such independent of potential other effects. Similar alterations in K6 and K14 expression have been described in chronic pastern dermatitis of heavy draught horses in association with hyperplasia, 19 which is lacking in hoof canker tissue. 3 As seen in a diverse spectrum of human cutaneous diseases, 24,47 as well as in mild recessive epidermolytic hyperkeratosis in Norfolk terriers, 13 genetic disorders, especially mutations of keratins, may also be a possible reason for the disturbed intermediate filament expression, and as a consequence also for koilocytotic appearance of canker-associated keratinocytes and impaired cell-cell adhesion. 24,47 As the observations of the present study were made on clinical samples, the reason for the altered distribution patterns of these intermediate filaments in hoof canker–associated keratinocytes is still unclear. Whether the changes documented here are the cause for hoof canker development or the result of any factor(s) associated with the presence of the disease is as yet unknown.
In summary, the results of the present study confirm that impaired keratinocyte differentiation plays a crucial role in hoof canker. No association between BPV-1/2 DNA and presence or degree of koilocyte-like keratinocytes or other morphological alterations in hoof canker tissue was detected. At this stage, a virus-induced pathogenesis remains only one of several possible causative pathways for canker.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Apprich_et_al - Equine Hoof Canker: Bovine Papillomavirus Infection Is Not Associated With Impaired Keratinocyte Differentiation
Supplemental Material, Combined_supplemental_materials-Apprich_et_al for Equine Hoof Canker: Bovine Papillomavirus Infection Is Not Associated With Impaired Keratinocyte Differentiation by Veronika Apprich, Theresia Licka, Sabrina Freiler and Cordula Gabriel in Veterinary Pathology
Footnotes
Acknowledgements
Dipl.Ing (FH) Claudia Höchsmann for IHC staining, and Brigitte Machac and Anne Flemming for histological preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the support offered by the inner-university research funding program “Profillinien,” Profile Line 2—Infection and Prevention.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
