Abstract
Contagious bovine pleuropneumonia (CBPP) is a respiratory disease of cattle that is listed as notifiable by the World Organization for Animal Health. It is endemic in sub-Saharan Africa and causes important productivity losses due to the high mortality and morbidity rates. CBPP is caused by Mycoplasma mycoides subsp. mycoides (Mmm) and is characterized by severe fibrinous bronchopneumonia and pleural effusion during the acute to subacute stages and by pulmonary sequestra in chronic cases. Additional lesions can be detected in the kidneys and in the carpal and tarsal joints of calves. Mmm infection occurs through the inhalation of infected aerosol droplets. After the colonization of bronchioles and alveoli, Mmm invades blood and lymphatic vessels and causes vasculitis. Moreover, Mmm can be occasionally demonstrated in blood and in a variety of other tissues. In the lung, Mmm antigen is commonly detected on bronchiolar and alveolar epithelial cells, in lung phagocytic cells, within the wall of blood and lymphatic vessels, inside necrotic areas, and within tertiary lymphoid follicles. Mmm antigen can also be present in the cytoplasm of macrophages within lymph node sinuses, in the germinal center of lymphoid follicles, in glomerular endothelial cells, and in renal tubules. A complete pathological examination is of great value for a rapid presumptive diagnosis, but laboratory investigations are mandatory for definitive diagnosis. The purpose of this review is to describe the main features of CBPP including the causative agent, history, geographic distribution, epidemiology, clinical course, diagnosis, and control. A special focus is placed on gross and microscopic lesions in order to familiarize veterinarians with the pathology and pathogenesis of CBPP.
Keywords
Contagious bovine pleuropneumonia (CBPP) is a severe OIE-notifiable respiratory disease of cattle caused by Mycoplasma mycoides subsp. mycoides (Mmm), characterized by severe fibrinous bronchopneumonia and pleural effusion during the acute to subacute stages and by pulmonary sequestra in chronic cases. CBPP can cause important productivity losses for the bovine industry due to the high mortality and morbidity rates. 6,104
CBPP has been present in Central and Northern Europe since the second half of the 16th century. Due to the increased trade in livestock—mainly from Europe—CBPP has since spread worldwide. 30,42 To date, it has been largely eradicated through the implementation of stamping-out policies. However, CBPP is still endemic in sub-Saharan Africa, where it represents a serious concern for livestock production. 6
The purpose of this review is to describe the main features of CBPP and to provide an overview of the causative agent, history, geographic distribution, epidemiology, clinical course, diagnosis, and control strategies. Special emphasis is placed on gross and microscopic lesions, in order to familiarize veterinarians with the pathology and pathogenesis of CBPP. In fact, a detailed pathological examination, along with ancillary diagnostic procedures, are very helpful to guarantee an accurate diagnosis.
The Causative Agent
Like all mycoplasmas, Mmm is a “mollicute,” that is, a small, self-replicating, pleomorphic bacterium lacking a cell wall. This latter feature explains their natural resistance to β-lactam antibiotics. Culture requirements of mycoplasmas can vary widely. Mmm is not among the most fastidious of these; therefore, different culture media can be successfully used to isolate and grow it. These usually consist of a meat extract (eg, heart-infusion broth) as the basic medium, supplemented with a yeast extract, inactivated horse serum, glucose, tryptose, sterol, sodium pyruvate, and DNA. Selective culture media, supplemented with penicillin G and thallium acetate, are used to isolate Mmm from contaminated samples. Mmm is able to grow under aerobic and anaerobic conditions, the optimum being at 37°C with 5% CO2. 6,72 The growth is quite slow and can take several days for primary isolation. In liquid media, growth is visible within 3 to 10 days as a homogeneous cloudiness (Fig. 1) with whirls when shaken. On solid culture media, Mmm colonies are small (diameter <1 mm) with a typical “fried-egg” shape (Fig. 2). 6 Mmm is easily inactivated by most common disinfectants, by acid and alkaline pH and by heat, and it survives for a short time (up to 2 weeks) in the environment, and such features strongly affect the epidemiology of CBPP. 5
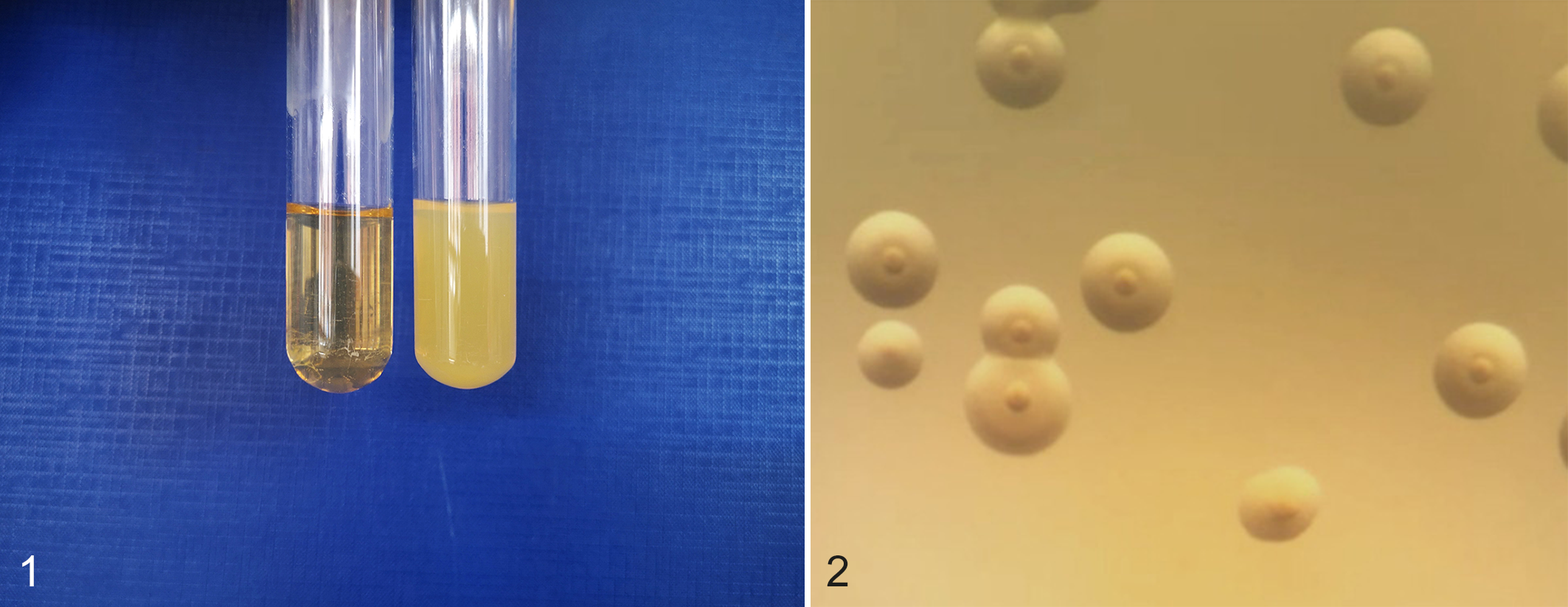
In broth culture, Mmm growth is visible within 3 days of cultivation as a homogeneous cloudiness (right tube), whereas no growth is visible in the control broth (left tube).
Mmm belongs to the so-called “mycoides cluster” consisting of 5 pathogenic mycoplasmas (namely, Mycoplasma mycoides subsp. mycoides, Mycoplasma mycoides subsp. capri, Mycoplasma capricolum subsp. capricolum, Mycoplasma capricolum subsp. capripneumoniae, Mycoplasma leachii), able to cause disease in ruminants and sharing many genotypic and phenotypic traits. 31,58 The causative agent of CBPP was previously specified as the small colony (SC) type of Mmm. However, the recent taxonomic revision has dropped this designation, after the new classification of Mmm “large colony” (LC) as Mycoplasma mycoides subsp. capri. At present, the availability of specific biomolecular tools allows a clear-cut differentiation within the mycoides cluster and among Mmm strains, providing useful genetic data for epidemiological studies, as well as deeper knowledge of the biological features and virulence factors of this pathogen. 30,55 Several Mmm strains have been fully sequenced, showing very low diversity among genomes. 30,35,77,114,116 As an example, the genome of the Italian strain 57/13 showed an overall identity of 99% as compared with the type strain PG1T. 77 The genome of the 57/13 strain consisted of a single chromosome of 1 192 498 bp, and 1077 protein-encoding genes were identified of which 332 encode for hypothetical proteins conserved among other Mmm isolates. 77
History and Geographic Distribution
CBPP is considered an “old” disease, although little is known about its exact origin. It may have been documented for the first time in 1550 by the Italian surgeon Gallo, who named it “polmonera.” However, Dupuy et al 30 consider that CBPP was actually described for the first time by de Haller in Switzerland in 1773. The infectious and contagious nature of CBPP was suspected at the end of the 18th century, but it was later confirmed in 1898 by Nocard and Roux, who were the first to isolate the causative agent. 80
For a long time, the disease remained confined to the central-eastern Alps, which represents the cradle of CBPP. Due to the increased trade of livestock, it then spread like wildfire worldwide (Europe, USA, Australia, Africa, Asia) with the only exceptions of South America and Madagascar. 42 During the 20th century, the implementation of mass vaccination and stamping-out strategies allowed the eradication of CBPP from most countries. In Europe, the disease reappeared in the Iberic peninsula, probably around 1950 although the exact date is unknown. Afterwards, CBPP outbreaks occurred from 1984 onwards in some Mediterranean countries (Portugal, France, Italy, and Spain) until it was finally eradicated in 1999, when the last case was recorded in Portugal. 72,81,104
At present, CBPP is endemic in many countries of sub-Saharan Africa where it greatly reduces the profitability of cattle farming. 2,76,102 Little is known about its presence in Asia. Currently, India and China are officially free from CBPP, while the disease has not been reported in many other Asian countries. 6,10,117 Along with foot-and-mouth disease, CBPP is currently considered the most serious transboundary threat for livestock in the African continent causing severe economic losses exceeding those sustained by any other infectious disease. 4 It is estimated that the cost of CBPP attributed to mortality, reduced livestock production, and disease control efforts reaches €44.8 million/year (€3.7 million per country) in endemic African countries. 68,102
Epidemiology
A wide range of domestic and wild ruminants are demonstrated to be susceptible to Mmm infection, but only cattle (Bos taurus) and zebuine cattle (Bos indicus) play a relevant role in the epidemiology of CBPP, 64 representing its natural hosts and main reservoirs. 6,17,51,90,98
Under field conditions, the efficient transmission of Mmm infection depends on prolonged and direct contact between susceptible and infected animals (clinically affected individuals or subclinical carriers), which actively excrete the pathogen through aerosolized infected droplets. CBPP transmission over distances (a few hundred meters) is exceptional; instead, Mmm infection is fostered when animals are closely stabled or trucked. 63,72 Although Mmm can survive in sequestra for months, the epidemiological role played by cattle with pulmonary sequestra is questionable and never clearly demonstrated. 104
Experimentally, Mmm infection can be also transmitted vertically (transplacental infection), and the agent can be detected in urine or semen; however, the real importance of these routes of transmission is negligible. Likewise, indirect transmission through contaminated pastures, water, fodder, and fomites appears irrelevant. 6,36,96,101,115 Therefore, the major risk of introducing Mmm infection in a CBPP-free region mainly relies on the trade and movement of infected animals. 4
Clinical Features
The CBPP incubation period is variable, often longer than 3 weeks and up to 6 months; however, it can be considerably shorter with experimental infection and/or overcrowded rearing conditions. In endemic territories, the long incubation time and the subclinical appearance of the disease facilitate the establishment of infection in a herd before it is noticed. In this situation, tracing back to the origin of infection is cumbersome and the frequent use of antibiotics makes CBPP diagnosis challenging. 63,72
Clinical signs are highly influenced by the virulence of the Mmm strain and the susceptibility of the host (ie, breed, age, immune status). As an example, European outbreaks showed lower morbidity and mortality rates when compared with the African ones, due to the lesser virulence of the European strains and the higher health status of the European cattle. 38,73,79,82
The clinical presentation ranges from hyperacute to subclinical forms. Hyperacute CBPP is rare and cattle may suddenly die due to the abundant pleural and pericardial effusion, with no significant clinical signs other than fever. Acute CBPP commonly occurs at the beginning of an epidemic and is characterized by high fever (up to 42°C) and severe respiratory distress (Fig. 3, Supplemental Videos S1 and S2) that can be further exacerbated by exercise. As the disease progresses, cattle can show tachypnea, chest pain with grunting, coughing, subcutaneous edema (Fig. 4), catarrhal to purulent nasal discharge, and ptyalism (Fig. 5).

Acute contagious bovine pleuropneumonia (CBPP), zebuine cattle.
Subacute-to-chronic form of CBPP predominate in endemic areas. These usually develop in animals that previously showed the acute form of disease, but some animals may initially present with the subacute to chronic form. Clinical signs in the subacute to chronic form are less severe and less specific, mainly consisting of coughing and progressive cachexia. The clinical outcome depends on the severity of the disease; during an epidemic outbreak, mortality rates can be very high (up to 90%) and the recovery period is often long, whereas in endemic zones mortality is low (<10%). 63,72,81
In calves up to 6 months, Mmm shows a distinct tropism toward the joints, although severe respiratory disease can be also observed in young animals. 81,104 Speculatively, it has been hypothesized that the different age-related tropism could be due to a partial colostral-derived passive immunity in CBPP-endemic areas. 104
Pathogenesis
Knowledge of CBPP pathogenesis is still poor, fragmented, and greatly challenging, due to the lack of suitable models in laboratory animals and the difficulty of experimentally reproducing the disease in its natural host. 45,75,91 In this respect, the assessment of in vitro models (cell cultures, ex vivo tissue cultures) is contributing to solve some crucial issues. 7,16,28,29,112,120 The pathogenesis of CBPP can be usefully dissected into the following inter-linked steps.
Adhesion to and Invasion of the Host’s Cells and Tissues
Under natural conditions, Mmm infection occurs through the inhalation of infected aerosol droplets. Therefore, Mmm adhesion to the airway cells represents the first pathogenetic step, which is crucial for the successful colonization of the host. 72,78 The adhesion is considered both host- and tissue-specific. Immunohistochemical investigations carried out on CBPP-affected cattle and on in vitro models confirm that Mmm has a selective tropism for the non-ciliated cells lining the bronchioles and the alveoli, which should be regarded as the earliest targets of infection. 28 Several adhesins and/or a specialized “terminal organelle” have been suggested in various mycoplasma species, but have never been identified in Mmm. Recent evidence indicates that Mmm adhesion molecules are immunogenic, as the organism attachment is inhibited by immune sera. 8,28 Unlike other mycoplasmas, the capsular galactan (a polysaccharide made of 6-O-β-D-galactofuranosyl-D-galactose) seems to reduce the attachment of Mmm to host cells, probably masking the adhesion molecules that become less exposed to the immune system. 93 A protein (“P19”) has also been suggested to contribute to Mmm adhesion in vitro. 120 However, current data indicate that the adhesion of respiratory mycoplasmas is a more complex and multistep process, which reasonably involve both specific (ie, ligand-receptor interactions) and nonspecific (hydrophobic interactions) mechanisms. 8
Mycoplasmas have long been considered extracellular parasites, strictly adhering to the host cell membrane to acquire nutrients. However, this dogma has been largely disavowed, thanks to the development of suitable in vitro models of study and more sophisticated microscopic technologies. In fact, a growing list of mycoplasmas, including Mmm, proved to enter nonphagocytic cells as a defensive strategy, in order to escape the immune response and some antimicrobial drugs. 18,28,43,74,85
After the colonization of bronchioles and alveoli, Mmm invades blood and lymphatic vessels, demonstrating a distinct tropism toward the endothelial cells; 28,36,81 this probably contributes to stimulate thrombosis, a dominant feature of CBPP pathology. Moreover, during the early febrile stage of CBPP, the presence of Mmm can be occasionally demonstrated in the bloodstream (“mycoplasmaemia”), as well as in several other tissues: lymph nodes, 92 kidneys, 36,96 joints, 108 liver, 81 uterus, 115 brain, 81 fetus, 100 and fetal membranes. 115
Host Cell Damage
Galactan induces vascular and cytopathic effects after intravenous injection, thus being considered among the major Mmm virulence factors. 19 A recent study in goats experimentally infected with Mycoplasma mycoides subsp. capri further supports the relevance of this capsular polysaccharide as a virulence factor in mycoplasmas of the “mycoides cluster.” 47
Many reports suggest that Mmm, as well as other respiratory mycoplasmas, can damage host cells by producing reactive oxygen species (ROS) via the metabolism of glycerol. 79 The latter is driven by 2 principal factors: (1) the glycerol transport ABC (GtsABC), a carrier protein providing the uptake of glycerol; and (2) the enzyme L-alpha-glycerophosphateoxydase (GlpO), which generates dihydroxyacetone phosphate and hydrogen peroxide (H2O2), one of the most important ROS. 40,109 –111 It is well known that African strains of Mmm have a more efficient GtsABC and produce larger amounts of ROS when compared with the European ones, this capacity being related to their presumed higher virulence. 1,79,109
ROS could damage the host cells through different mechanisms both directly, affecting the integrity of cellular membranes, and indirectly, activating the nuclear factor-kB molecular pathway. 22,49 It should be noted that Mmm is able to produce low quantities of H2O2, which can be rapidly destroyed by the host catalase activity. Therefore, the close contact between Mmm and the host cell is a prerequisite for ROS to cause their cytotoxic effect, if any. 16,79 In this respect, we remark that Mmm is not able to induce any pathological change in bovine respiratory explants, thus raising further questions about the real cytotoxic activity of ROS produced by Mmm. 28
In addition, mycoplasmas are known to induce the production of ROS by phagocytic cells, which could amplify the cell and tissue damage. 50,78 In particular, the generation of ROS by Mmm could prime the respiratory burst of neutrophils in a “dose-dependent” manner. The amount of ROS produced by neutrophils is enormously greater when compared with Mmm, thus likely playing a more important role in tissue damage. 29
Mmm and the Inflammatory Response
Alveolar macrophages are among the first and most important line of defence against microorganisms of the lower airways. Apart from their phagocytic and microbicidal activities, alveolar macrophages contribute to modulate the inflammatory response by producing a wide range of chemical mediators. 37 Mycoplasmas stimulate monocytes and macrophages to synthesize a number of cytokines, after the interaction between membrane lipoproteins and Toll-like receptors (TLRs). 62,86 Concerning Mmm, available data are currently poor and conflicting. In fact, macrophages can produce pro-inflammatory (TNF-α) or anti-inflammatory (interleukin [IL]-10) cytokines after in vitro stimulation with Mmm or galactan, respectively. 48,105 TNF-α, IL-1β, and IL-17A contribute to the inflammatory response in CBPP-affected lungs, 99 plasma levels of TNF-α being positively correlated with the severity of the disease. 88 In this respect, Mmm demonstrated the ability to induce the apoptosis of bovine leukocytes (including monocytes and polymorphonuclear cells). Similarly to other mycoplasmas, Mmm could have developed the ability to trigger apoptosis in leukocytes as an efficient strategy to escape the host inflammatory and immune response. 25
Immune-Mediated Mechanisms in the Pathogenesis of CBPP
Lymphoid proliferation, often arranged as follicles (so-called “tertiary lymphoid follicles”), is among the main features of mycoplasma infections. It has been observed that mycoplasmas (eg, Mycoplasma gallisepticum and Mycoplasma agalactiae) induce the mucosal infiltration of lymphocytes in a well-ordered way: (1) CD8+/TCR− natural killer (NK) lymphocytes prevail during the first week postinfection (pi); (2) CD4+/TCR+ helper T cells and CB8+/TCR+ cytotoxic T lymphocytes arrive at 2 to 3 weeks pi; (3) B lymphocyte infiltration occurs and becomes dominant later, the antigenic variation of mycoplasma lipoproteins likely inducing a long-term stimulation of the immune response. 21,32,33
Although plausible and long suggested, the role of the immune response in the pathogenesis of respiratory mycoplasmosis—including CBPP—is still controversial. 39, 72, 78 Cytotoxic T lymphocytes have been shown to control the immune-mediated mechanisms of disease after Mycoplasma pulmonis infection, while the depletion of helper T cells apparently reduces the severity of the disease. 44 As far as CBPP is concerned, helper T cells seem to play a primary role during the early stages of Mmm infection, greatly contributing to the production of interferon-γ (IFN-γ), the latter being considered protective during CBPP. 23,24,46,106,107 In contrast, depletion of CD4+ lymphocytes did not affect the clinical outcome, the pathology, the humoral immune response, or the serum level of IFN-γ after Mmm experimental infection, 89 thus raising further questions about the real significance of helper T cells in the pathogenesis of CBPP.
Likewise, data about the humoral immune response after Mmm infection are few and somewhat conflicting. There was no positive correlation between the antibody response to Mmm (immunoglobulin isotypes and titres, both in serum and in bronchoalveolar lavage fluid) and either the clinical severity or the lesions. 71 More recently, it was observed that high levels of serum anti-Mmm antibodies did not prevent the occurrence of CBPP; in fact, recovering cattle showed lower amounts of anti-Mmm immunoglobulins when compared to cattle with more severe and worsening lesions. 94 On the contrary, the mucosal immune response seems more effective, with higher levels of anti-Mmm secretory IgA fostering a positive outcome of the disease. Other data suggest that antibodies targeting the N terminus of lipoprotein Q could trigger the deposition of immune complexes and the development of glomerulonephritis, thus supporting the involvement of immune-mediated mechanisms in the pathogenesis of CBPP. 67
Pathology of CBPP
Most relevant lesions of CBPP are confined to the thoracic cavity. As suggested by its name, CBPP is characterized by a fibrinous bronchopneumonia, with the constant involvement of the pleural surfaces. During the acute stage of the disease, many litres of effusion can be present in the pleural cavity (Fig. 6), along with an abundant deposition of fibrin on both the visceral and parietal pleura that is often very thick, yellowish in colour, and typically resembling an “omelette” (Fig. 7). In chronic cases, fibrinous pleuritis progresses to strong, fibrotic adhesions between the lung and the chest wall.
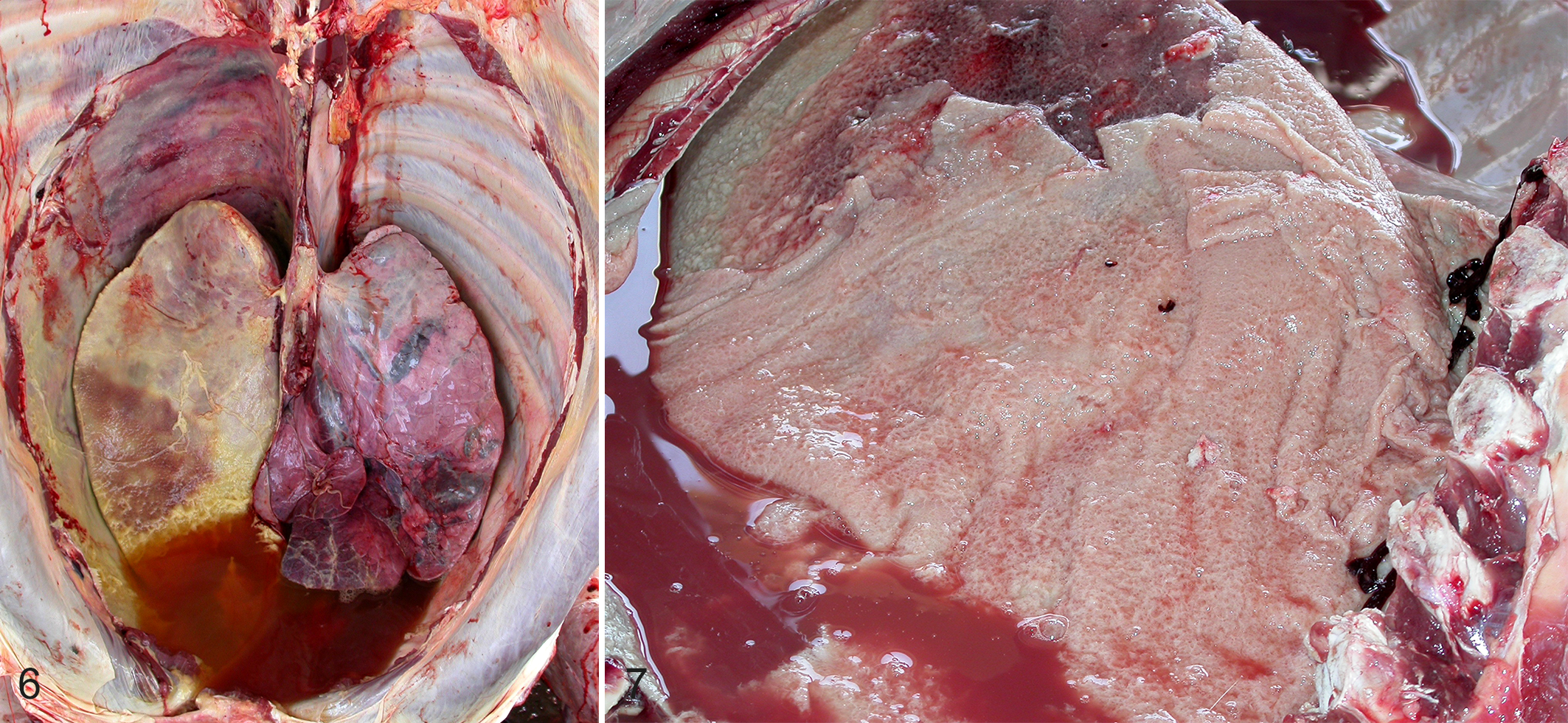
Acute contagious bovine pleuropneumonia (CBPP), thoracic cavity, cattle.
In most cases, pneumonic areas are unilateral and affect one or more lung lobes, with a marked predilection for the caudal lobes. During the early stages, the lung lesions typically show a “marbled” appearance, due to intermingled healthy and diseased (“hepatized”) lobules that are separated by thickened interlobular septa (Figs. 8, 9). Large, well-demarcated foci of necrosis are also frequent (Fig. 10), likely resulting from thrombosis of blood vessels. Afterwards, such necrotic masses can develop as variably sized sequestra or as scars (Figs. 11–13). Typically, the necrotic parenchyma is odorless and its lobular structure is well recognizable. Sequestra can persist for months in clinically healthy recovered animals and can harbor live Mmm for long periods of time. 6,72,81
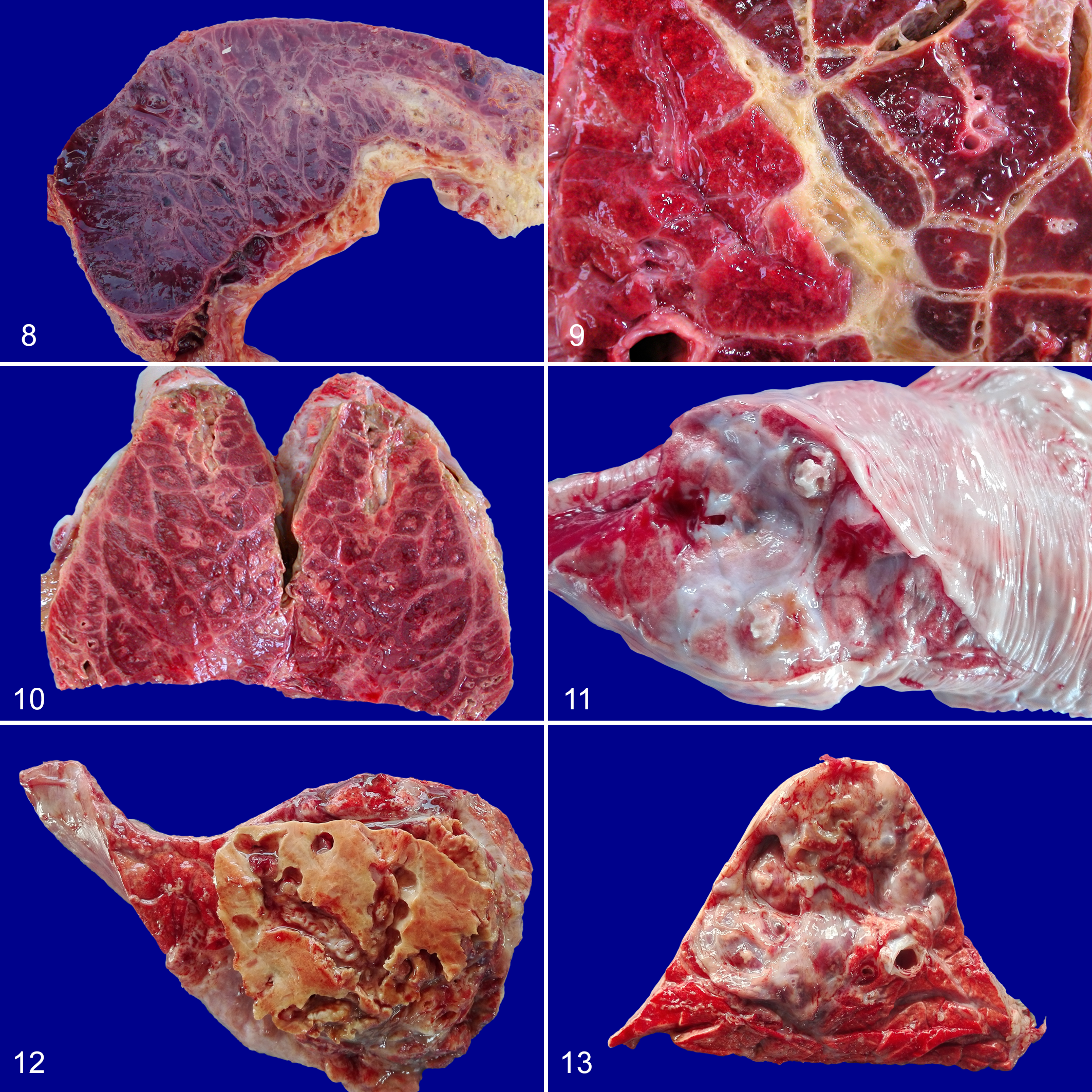
Acute contagious bovine pleuropneumonia (CBPP), lung, cattle. Diffuse red hepatization of the lung parenchyma; the lobules and interlobular septa both appear hemorrhagic. The pleura is covered by fibrin.
The major microscopic features of acute CBPP are sero-fibrinous and purulent exudate within bronchioles, alveoli, and interlobular septa, as well as vasculitis and thrombosis of blood and lymphatic vessels. In sequestra, areas of coagulative necrosis are circumscribed by a fibrotic reaction, often associated with the development of newly formed (“tertiary”) lymphoid follicles (Figs. 14–19).
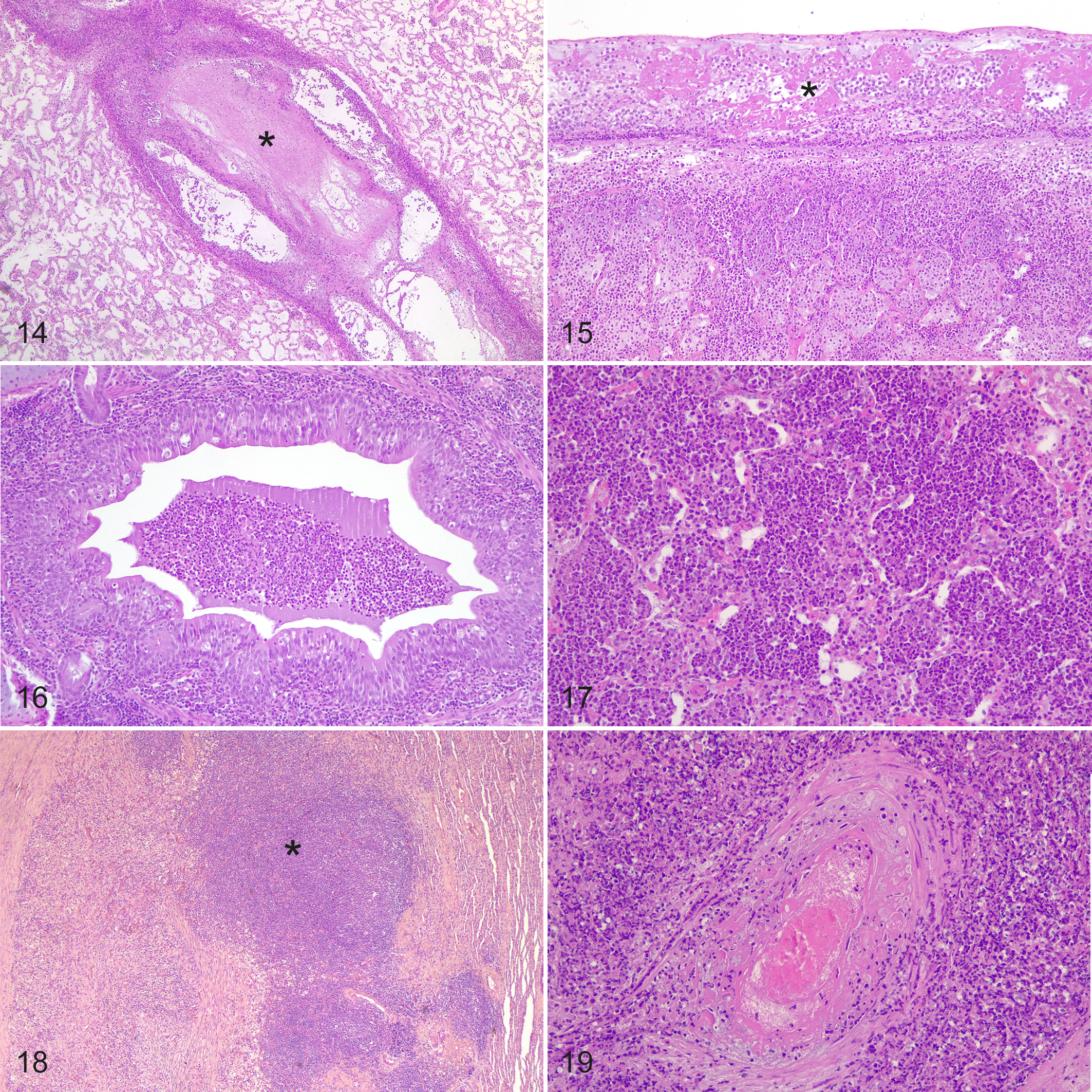
Contagious bovine pleuropneumonia, lung, cattle, hematoxylin and eosin.
The lymph nodes that drain the lungs and thoracic cavity (ie, mediastinal and tracheobronchial) are often enlarged and edematous, mostly during the acute stage of the disease (Fig. 20), and can occasionally develop foci of necrosis and small sequestra (Fig. 21). Additional lesions can be detected in the kidneys as multiple infarcts in the renal cortex (Fig. 22) and serofibrinous tenosynovitis in the carpal and tarsal joint of young calves. 36,104,108
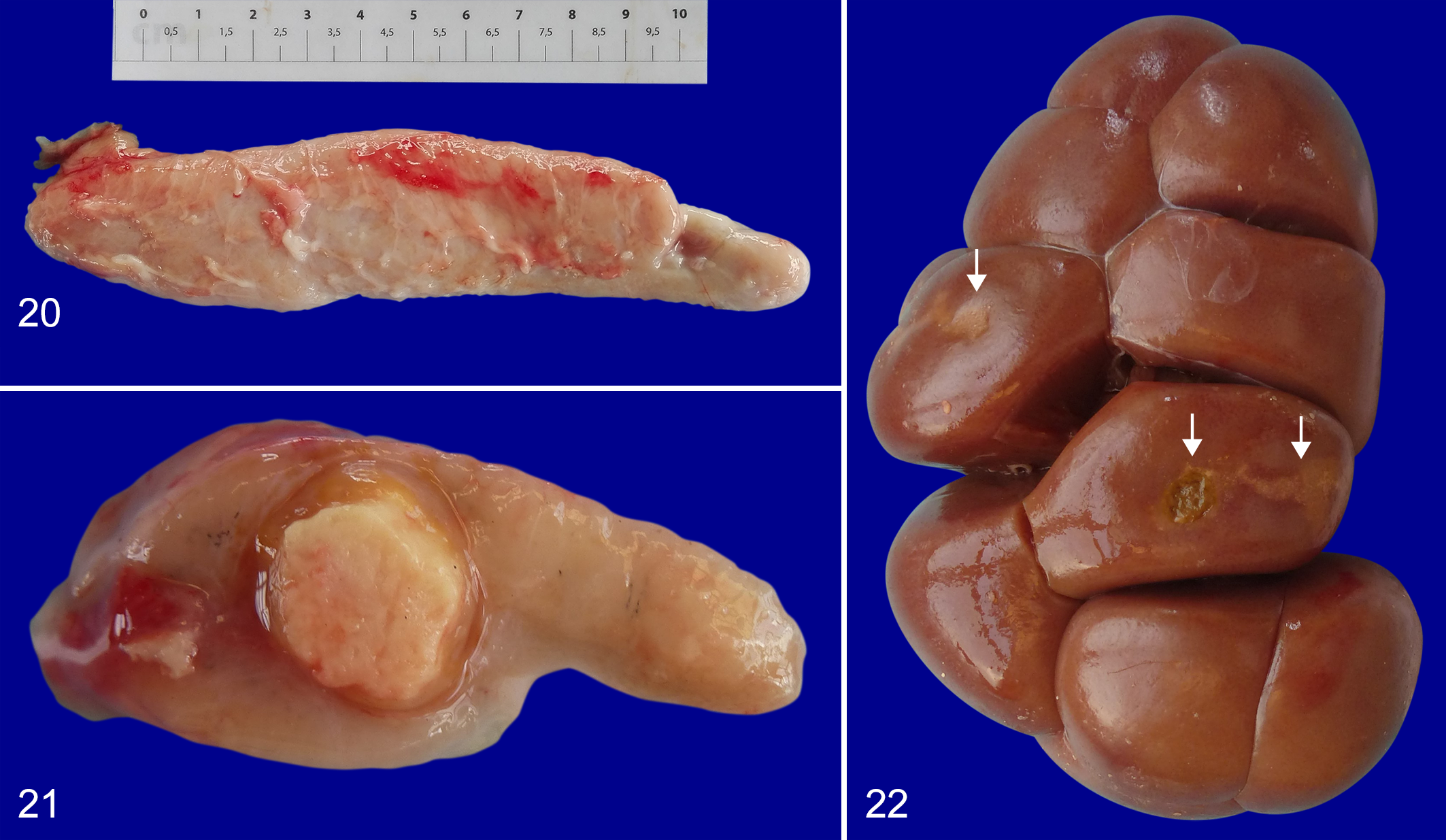
Acute contagious bovine pleuropneumonia (CBPP), mediastinal lymph node, cow. Massive enlargement of the caudal mediastinal lymph node.
The distribution pattern of Mmm has been investigated by immunohistochemistry in different tissues. 28,36,57,92 In lungs, large amounts of Mmm antigens can be commonly detected on the bronchiolar and alveolar epithelial cells, in phagocytic cells (neutrophils and macrophages), within the wall of blood and lymphatic vessels, inside the necrotic areas, and within lymphoid follicles (Figs. 23–28). 14,28,57 In lymph nodes, 2 different distribution patterns of Mmm can be observed: (1) in the acute stage of the disease, antigen is present in the cytoplasm of macrophages within the sinuses; (2) in chronic cases, the Mmm antigen is restricted to the germinal center of lymphoid follicles (Figs. 29, 30). 92 Likewise, different immunohistochemical patterns can be observed in renal parenchyma: (1) abundant Mmm antigen is commonly detected in glomerular endothelial cells during the acute stage of CBPP; (2) prominent Mmm-immunoreactivity is mainly in renal tubules of cattle with chronic CBPP (Figs. 31, 32). 36
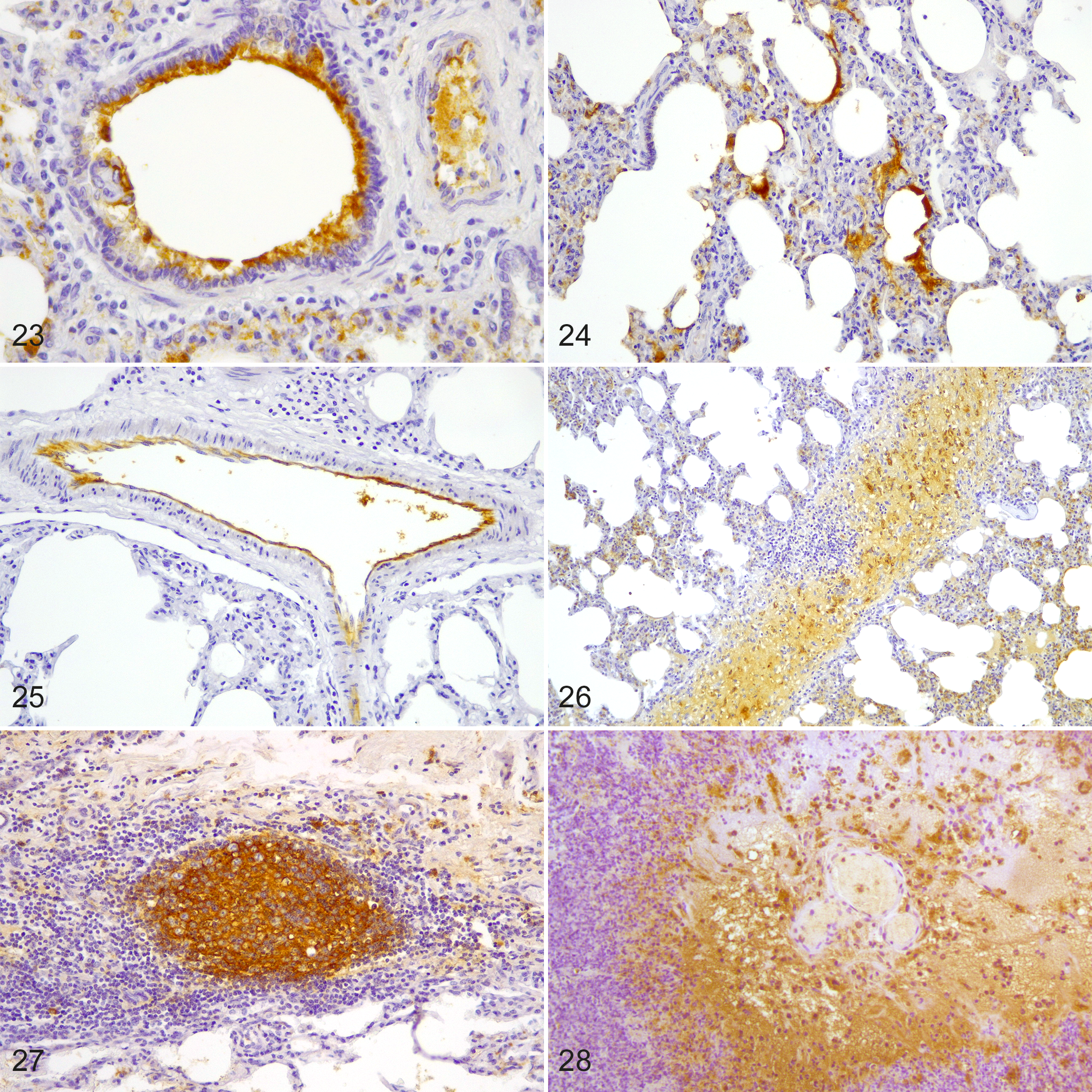
Contagious bovine pleuropneumonia, lung, cattle. Mycoplasma mycoides subsp. mycoides (Mmm) immunolabeling is present (brown staining) on the bronchiolar epithelium (Fig. 23), the alveolar epithelium (Fig. 24), within the endothelium of a blood vessel (Fig. 25), the interlobular septum (Fig. 26), the tertiary lymphoid follicles (Fig. 27), and surrounding the blood vessels affected by vasculitis (Fig. 28). Immunohistochemistry for Mmm.
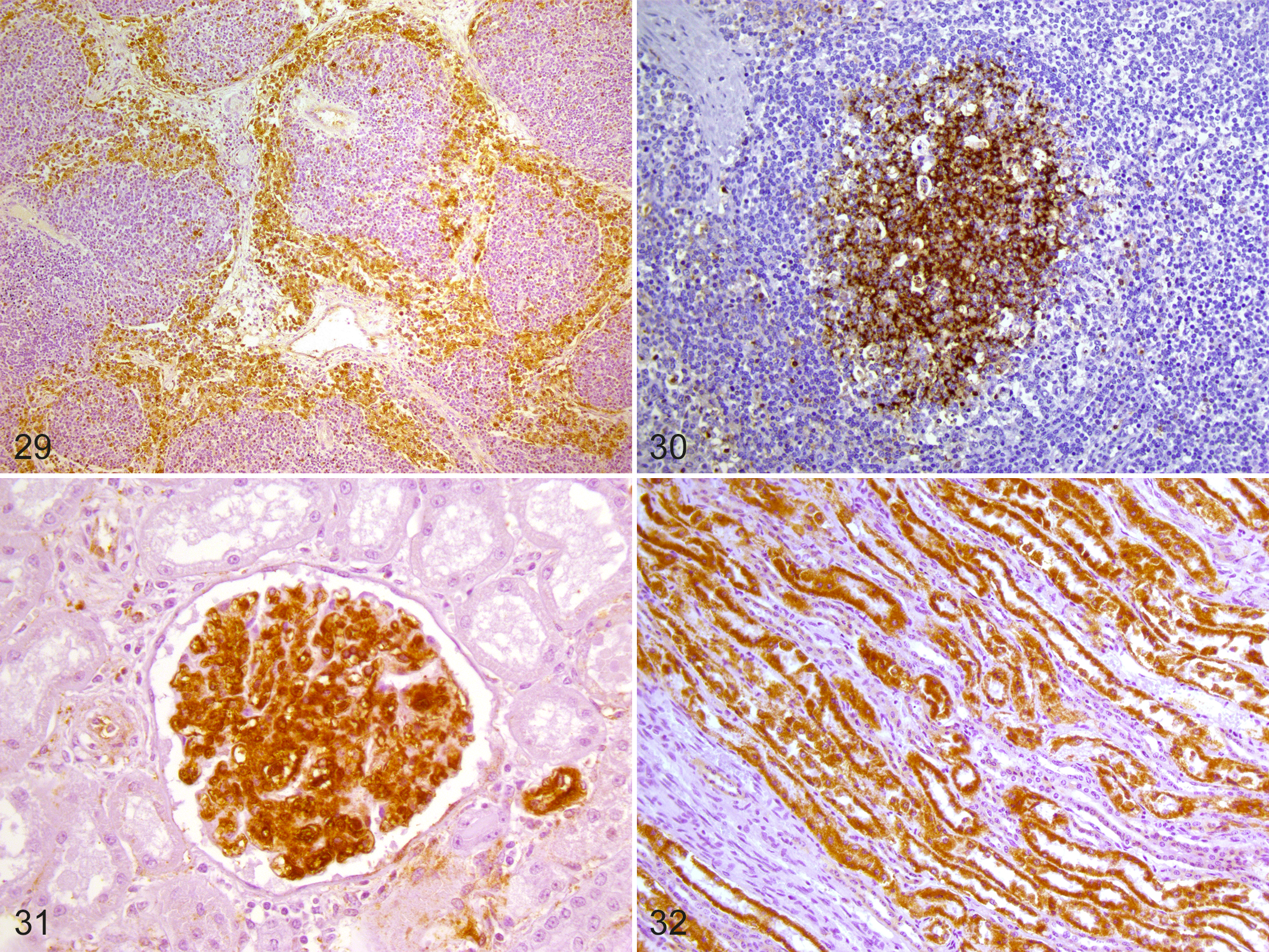
Contagious bovine pleuropneumonia (CBPP), tracheobronchial lymph node, cattle. In the lymph node, there is immunoreactivity (brown staining) in macrophages within the sinuses (Fig. 29) and in lymphoid follicles (Fig. 30).
Diagnosis
Pathological findings are of great value for a rapid presumptive diagnosis and for disease surveillance and management. 27 However, laboratory investigations are essential to confirm or to rule out CBPP, especially when it is suspected in disease-free areas. Mmm can be isolated from nasal swabs or discharges of live animals; or at necropsy, pleural fluid represents the first-choice sample, followed by diseased lung and lymph nodes draining the respiratory tract.
Polymerase chain reaction (PCR) tests have proven extremely useful to rapidly identify Mmm. 11,15,53 More recently, real-time PCR methods have become available for the diagnosis of CBPP, improving the rapidity of conventional PCR. 34,56,95 Although immunohistochemistry is only supplementary to CBPP diagnosis it can be very useful in chronic cases, in cases with suspected antibiotic treatment that would interfere with isolation of Mmm, and especially on kidney tissues where it is difficult to isolate Mmm. 14,36,92,104
The serological tests recommended by OIE (complement fixation test, cELISA, immunoblotting test) represent suitable tools for herd diagnosis and for the implementation of control plans. 6,20,52,61,83,104 However, under natural field conditions, no single serological test is capable of detecting all infected animals, 61 either during the early stage of disease or in its chronic phases. A number of acute and chronic respiratory diseases must be considered as differential diagnoses such as pneumonic pasteurellosis and Mycoplasma bovis infection. However, in acute cases, the marbled appearance of the lungs, the constant involvement of the pleural surfaces, and the histological presence of vasculitis can aid the diagnosis of CBPP. In chronic cases, necrotic sequestra should be differentiated from pulmonary abscesses and tuberculosis; however, CBPP induced-seuquestra are odorless and caseous necrosis and calcification are usually absent. 3,13,80 Therefore, diagnosis is based on both the presence of CBPP lesions and the demonstration of Mmm from infected tissues. In contrast, serological tests are valid at the herd level only. 6,12
Prevention and Control
As previously stated, Mmm is poorly resistant in the environment; thus, its survival strictly depends on the infection of susceptible animals, 6 justifying the assertion: “CBPP walks on the feet of cattle.” Considering that, the control of the disease in CBPP-free areas is based on the following points: (1) ban on importing cattle from infected countries; (2) control measures at frontiers; (3) quarantine; and (4) early detection of outbreaks with application of strict stamping-out policies. 4,5,60
In most CBPP-endemic areas, managing the disease is really challenging, mostly due to the absence of animal identification systems and to the difficulty in restricting and controlling animal movement. The OIE recommend 2 attenuated vaccines, both derived from the Tanzanian T1 strain: namely, T1/sr and T1/44. The latter was attenuated after 44 passages on chicken embryos and demonstrated to be more efficacious, although it provides a short-term immune protection (<1 year) and it can induce severe adverse reactions. 6,45,69,84,87,97,113 Moreover, the attenuated strain Ben-1 proved useful to eradicate CBPP in China. 118
Numerous efforts have been made to try to develop new inactivated, mucosal, subunits, and recombinant vaccines for CBPP. However, a better understanding of CBPP immunology and protective response and further studies on CBPP vaccine formulation are needed to achieve more effective, safer, cheaper vaccines that could possibly allow for the discrimination of infected and vaccinated animals (DIVA vaccines). 26,45
The use of antimicrobials is banned during official eradication programs, 4 but it is a common practice to treat CBPP cases in sub-Saharan Africa. 59,66 Several studies demonstrated the therapeutic efficacy of tetracyclines, macrolides, and fluoroquinolones. 9,41,65,70,119 However, the growing concern about antimicrobial resistance and the possibility to increase the number of cattle with persistently infected sequestra make such a control strategy questionable. 45,54,103
Conclusions
More than 5 centuries after its first description, CBPP still represents a fascinating, poorly understood, and very relevant transboundary disease of cattle that can cause severe economic losses in livestock production. Nowadays, CBPP is a serious concern for many endemic countries of sub-Saharan Africa, constraining their development and economic improvement and restricting the trade of food and livestock between nations and continents. We hope this review will be of value to efficiently guide the diagnostic approach of CBPP, as well as to stimulate further interest in this intriguing field of research.
Footnotes
Acknowledgements
The authors gratefully thank Dr Angie Colston (The Global Alliance for Livestock Veterinary Medicines, GALVmed, UK), Dr Hezron Wezonga (Kenyan Agricultural and Livestock Research Organization, KALRO, Nairobi, Kenya), and Dr Geoffrey Muuka (Central Veterinary Research Institute, CVRI, Lusaka, Zambia) for their kind and outstanding collaboration during CBPP-experimental investigations. The authors gratefully thank Dr Abigail Rose Trachtman for kindly revising the English language.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
