Abstract
Infection of small ruminants with peste des petits ruminants virus (PPRV) and goatpox virus (GTPV) are endemic and can have devastating economic consequences in Asia and Africa. Co-infection with these viruses have recently been reported in goats and sheep in Nigeria. In this study, we evaluated samples from the lips of a red Sokoto goat, and describe co-infection of keratinocytes with PPRV and GTPV using histopathology and transmission electron microscopy. Eosinophilic cytoplasmic inclusion bodies were identified histologically, and ultrastructural analysis revealed numerous large cytoplasmic viral factories containing poxvirus particles and varying sizes of smaller cytoplasmic inclusions composed of PPRV nucleocapsids. These histopathological and ultrastructural findings show concurrent infection with the 2 viruses for the first time as well as the detection of PPRV particles in epithelial cells of the mucocutaneous junction of the lip.
Keywords
Peste des petits ruminants (PPR) and goatpox (GTP) are economically important transboundary animal diseases of small ruminants in Asia and Africa that show devastating endemics in these regions. 3,4,15 Furthermore, co-infection of these viral infections have recently been reported in Nigeria. 2
PPR is a highly contagious viral disease of sheep and goats 14 caused by the PPR virus (PPRV), a single stranded nonsegmented RNA virus (negative sense) in the genus morbillivirus within the virus family Paramyxoviridae. 13,16 The GTP virus (GTPV) is a large enveloped and double-stranded DNA virus belonging to the genus capripoxvirus within the family Poxviridae. 19 Both PPRV and GTPV are endemic in the Middle East, Asia, and Africa and are highly contagious among small ruminants causing severe economic losses due to high morbidity and mortality rates. 5,16 Although GTPV has not been associated with increased mortality, its occurrence reduces the profitability of the farmers. 15 Mixed natural infections involving PPRV, GTPV, or PPRV and other poxviruses have been reported in Asia and Africa. 7,11,15
A mixed infection of PPRV and GTPV has been recently reported in a mixed flock of sheep and goats in Nigeria. 2 In that investigation, PPRV was identified by c-ELISA (complement-enzyme linked immunosorbent assay) on pulmonary and splenic tissues, and GTPV was identified by PCR (polymerase chain reaction) testing of the skin scab. 2 In the current study, we demonstrate co-infection of PPRV and GTPV in a biopsy from the mucocutaneous junction of the lower lip of one of the red Sokoto goats from the study mentioned above, using histopathology and transmission electron microscopy.
Clinical signs in the goats included mucopurulent oculo-nasal discharge, scab, crusts, and vesicles around the mouth (Fig. 1), and generalized cutaneous pox lesions with a high mortality rate. 2
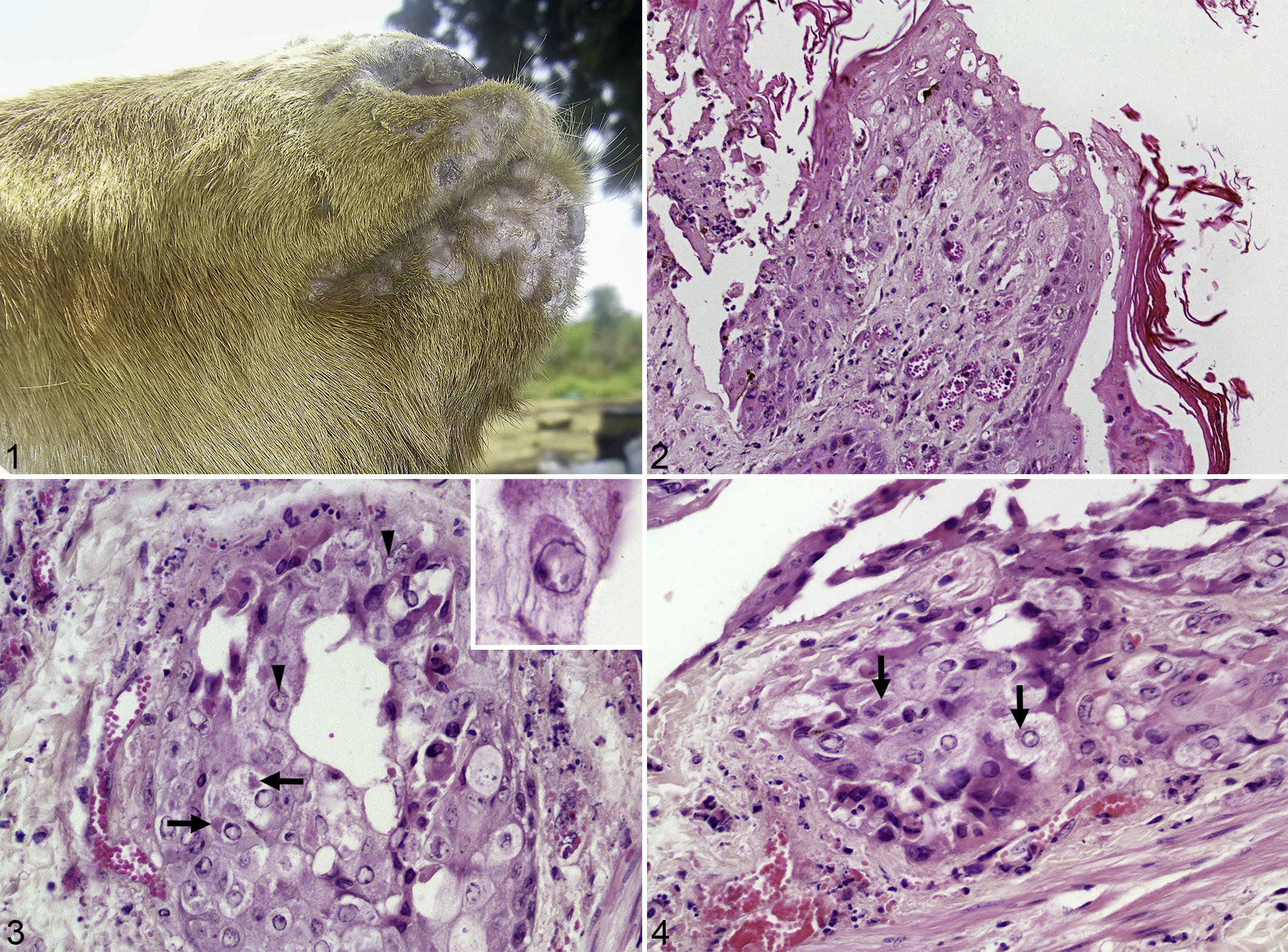
Co-infection of peste des petits ruminants and goatpox viruses, lip, goat.
For histopathology, 2 × 2 cm samples from the mucocutaneous junction of the lower lip of one affected goat were immersed in 10% phosphate-buffered formalin for up to 72 hours. The tissue was cut in 0.5-cm-diameter blocks and 5-μm-thick histologic sections were routinely prepared and stained with hematoxylin and eosin (HE). 1
Microscopic lesions consistent with GTPV and PPRV co-infection were focally extensive and deep proliferative dermatitis with orthokeratotic hyperkeratosis, pustules, and serocellular crusts. Focally, there was epidermal hyperplasia leading to an undulated surface, moderate congestion, and hemorrhages. In areas with deep dermatitis an infiltration with neutrophils and occasional macrophages and lymphocytes was present. Keratinocytes were acanthotic and showed ballooning degeneration. Some keratinocytes and cells within sebaceous glands had medium to large eosinophilic intracytoplasmic inclusion bodies (ICIB). In very few epithelial cells, small eosinophilic intranuclear inclusions were also detected (Figs. 2–4) as previously described for PPRV-infected pneumocytes in goats. 6
For transmission electron microscopy, 0.1-cm diameter areas of the paraffin-embedded tissue that contained ICIB in the corresponding HE-stained section were used. The paraffin wax was removed by heating the samples to 60 °C for 15 minutes followed by incubation in xylene. Tissue was rehydrated in ethanol and placed in 2.5% glutaraldehyde solution buffered in 0.1 M sodium cacodylate at pH 7.4 overnight, post-fixed in osmium tetroxide, and stained with uranyl acetate. The samples were dehydrated, transferred to propylene oxide, and infiltrated with epoxy resin (glycidyl ether). The polymerization in gelatin capsules was carried out at 60 °C for 3 days. With a toluidine blue–stained semithin section, the point of interest (keratinocytes) was identified and trimmed for ultrathin sectioning. The ultrathin sections were placed on 300 mesh nickel grids and stained with uranyl acetate and lead citrate. For analyses a Tecnai G2 Spirit (FEI, Netherlands) transmission electron microscope was used at an acceleration voltage of 80 kV. 10,12
The histopathological findings of ICIB corresponded to the ultrastructural presence of numerous intracytoplasmic capripoxvirus particles, condensation of chromatin, protein aggregation and varying sizes of intracytoplasmic PPRV particles. Both structures were found co-localized in one degenerating keratinocyte (Fig. 5).
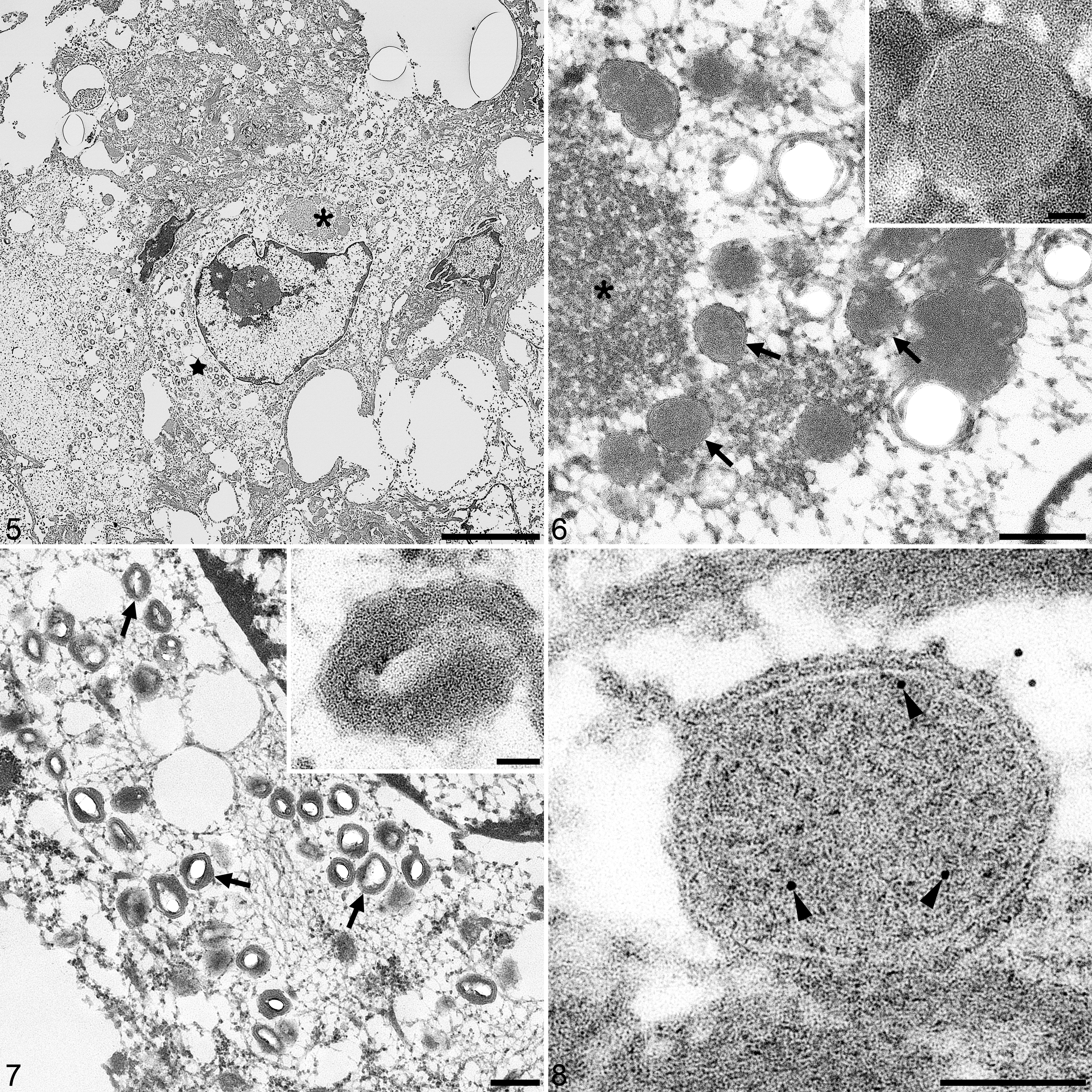
Co-infection of peste des petits ruminants virus (PPRV) and goatpox virus (GTPV) in keratinocytes, lip, goat. Transmission electron microscopy.
The GTPV particles had a typical brick-shaped structure and were about 250 to 300 nm diameter (Fig. 7). For PPRV, we observed aggregation of ribonucleocapsids in the cytoplasm and intracellular PPRV particles of about 300 nm diameter lacking the outer protein shell (Fig. 6). For confirmation of the PPRV findings, epoxy-embedded material was stained with α-PPRV monoclonal antibody that was characterized to bind specifically to the PPRV nucleocapsid (Fig. 8). 18 The monoclonal antibody was used undiluted and the sections were incubated overnight at 4 °C directly on the grid. For detection, we used a α-mouse immunogold conjugate (GMHL10, BBI Solutions, UK) at an 1:50 dilution and incubated for 45 minutes.
The ultrastructural properties of PPRV particles were consistent with earlier descriptions of morbilliviruses structures, 6,8,9 while that of GTPV were in line with established dimensions. 17 Overall, the process of formalin fixation, deparaffinization and post-fixation in glutaraldehyde for transmission electron microscopy preserved the ultrastructure of these viral particles and allowed the morphological differentiation. This study demonstrates for the first time the co-infection of PPRV and GTPV in haired skin biopsies of goats and the detection of PPRV particles in keratinocytes.
Footnotes
Editor’s Note
Dr Teifke, an Associate Editor with the journal, was not involved in the editorial review of or decision to publish this article.
Acknowledgements
We thank Dr C. Fast for the α-PPRV monoclonal antibody, P. Meyer for technical assistance in EM preparation, G. Czerwinski and G. Afolabi for histological slides preparation, and M. Jörn for graphical design of the images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Olatunde B. Akanbi was funded by German Academic Exchange Service DAAD, Re-invitation Grant 2018, for this work.
