Abstract
Accumulating data highlight the role of neurotrophins and their receptors in human breast cancer. This family includes nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF), both synthetized as proneurotrophins (proNGF and proBDNF). (pro)NGF and (pro)BDNF initiate their biological effects by binding to both their specific receptors TrkA and TrkB, respectively, and the common receptor p75NTR. Currently, no data are available about their expression and potential role in canine mammary tumors. The aim of this study was to investigate expression of proNGF and BDNF as well as their receptors TrkA, TrkB, and p75NTR in canine mammary carcinomas, and to correlate them with clinicopathological parameters (grade, histological type, lymph node status, recurrence, and distant metastasis) and survival. Immunohistochemistry was performed on serial sections of 96 canine mammary carcinomas with antibodies against proNGF, BDNF, TrkA, TrkB, and p75NTR. Of the 96 carcinomas, proNGF expression was detected in 71 (74%), BDNF in 79 (82%), TrkA in 94 (98%), TrkB in 35 (37%), and p75NTR in 44 (46%). No association was observed between proNGF, BDNF, or TrkA expression and either clinicopathological parameters or survival. TrkB and p75NTR expression were associated with favorable clinicopathological parameters as well as better overall survival.
Over the last decade, accumulating data have suggested that canine mammary tumors could be a valuable model of human breast cancer in comparative oncology. Mammary neoplasms are a major health issue in both species: breast cancer is the most prevalent cancer and the first cause of cancer-related death in women worldwide, and 1 in 10 women will develop breast cancer during their lifetime. 20,27 In parallel, mammary tumors are the most common cancer in intact bitches, and one third of unspayed bitches will develop a mammary tumor by the age of 10. 45,67 Canine mammary tumors share numerous clinical (age of onset, predominance of carcinomas, type of metastases), genetic (role of BRCA1/2, overlapping of gene signature), and molecular similarities with their human counterpart. 2,22,28,30,34,35,49,52,54,66,68,69 Finally, their spontaneous occurrence allows for the study of mammary neoplasm development and the influence of the microenvironment. 53
Accumulating data highlight the role of neurotrophins in a wide range of human tumors, including breast cancer. 8,13,26,74 Neurotrophins are a family of structurally conserved growth factors composed of nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin 3 (NT-3), neurotrophin 4/5 (NT-4/5), and their associated precursors named proneurotrophins (proNGF, proBDNF, proNT-3, and proNT-4/5), which can have a biological action by themselves. The action of (pro)neurotrophins is mediated by a low-affinity p75 neurotrophin receptor (p75NTR), which can bind all (pro)neurotrophins, and high-affinity Trk tyrosine kinases receptors, which bind specifically one (pro)neurotrophin: (pro)NGF binds to TrkA, (pro)BDNF and NT-4/5 bind to TrkB, and NT-3 binds specifically to TrkC. 57
p75NTR contributes to resistance to chemotherapy in triple negative breast cancers, a group with poor outcome that cannot be treated with endocrine or HER-2 targeting therapies. 7,21 An autocrine stimulation loop of BDNF mediated by p75NTR and TrkB.T1, the truncated TrkB isoform that lacks the intracellular tyrosine kinase domain, results in a stimulation of breast cancer cell survival and resistance to apoptosis. 75 More important, BDNF-TrkB signaling was reported to affect the outcome of triple-negative breast cancer by modulating cancer-endothelial cells interaction. TrkB has also been reported to promote breast cancer metastasis, and TrkB+ cancer stem cells were found to be responsible for post-chemotherapy recurrences in triple negative breast cancers. 33,73,81
P75NTR and TrkA are involved in breast cancer cell survival and proliferation. 1,12,36,76 Recent data suggest that proNGF, even more than NGF, would have a negative impact on breast cancer outcome. ProNGF stimulates breast cancer cell metastasis via TrkA and sortilin, and is able to enrich the cancer stem cell pool in breast cancer cell lines. 11,72
Within this context, neurotrophin Trk receptors have emerged as novel therapeutic targets in breast and other cancers, including colorectal and lung cancer. 10,44,61,62 Several Trk-targeting drugs are under clinical development. Two of the leading Trk inhibitors, entrectinib and LOXO-101, are currently ongoing phase 2 trials, and other Trk inhibitors like MGCD516, PLX7486, DS-6051b, and TSR-011 are ongoing phase 1 trials. 32,39 However, application of tyrosine kinase inhibitors in veterinary medicine is relatively recent. Only 2 tyrosine kinase inhibitors have been approved by the US Food and Drug Administration for the treatment of canine mast cell tumors: toceranib (Palladia; Zoetis, Madison, NJ) and masitinib (Kinavet; Catalent Pharma Solutions, Somerset, NJ). 25,41 Toceranib phosphate is very closely related to sunitinib (Sutent) as both inhibit a variety of tyrosine kinase receptors including PDGFRα/β, VEGFR2/3, CSF1R, FLT-3, RET, andKIT. 42,47,79,80 The second small molecule inhibitor approved for use in dogs is masitinib mesylate (Kinavet), which inhibits KIT and PDGFRα/β along with transduction pathway Lyn/FAK. 25 A third molecule, Gleevec (imatinib), is less employed but has also been successfully used in dogs. 6
So far, neurotrophins and their receptors have been poorly studied in dog, and no study has investigated their expression in canine mammary tumors. The aim of our study was to characterize the expression of proNGF, BDNF, and their receptors TrkA, TrkB, and p75NTR, in a cohort of canine mammary carcinomas and to correlate their expression with clinicopathological parameters (histological type, grade, emboli, lymph node status, recurrence, and distant metastasis) and survival.
Material and Methods
Case Selection and Tumor Samples
The cohort of canine malignant mammary carcinomas evaluated in this study has been previously described. 60 The present study is based on a series of 96 cases of canine malignant mammary tumors and corresponding lymph nodes selected from the biobank of OCR (Oncovet Clinical Research, Parc Eurasanté Lille Métropole, France). Specimens were obtained from bitches from all ages and breeds, which underwent surgery between July 2011 and October 2015. All tissues were fixed in 10% neutral buffered formalin and embedded in paraffin wax. The study followed as far as possible the recommended guidelines for the conduct and evaluation of prognostic studies in veterinary oncology and more specifically on mammary neoplasms, with the inherent limitations of a retrospective study. 43,77 Dogs included in the study had either a single malignant mammary tumor or multiple mammary tumors with only one of them being malignant. Forty-eight other cases were excluded from the study. Specifically, 12 cases of recurrences, and 8 animals presenting more than one malignant tumor whatever the subtype, were excluded because of the difficulty to determine which tumor would affect the follow-up. Thirteen carcinomas in situ (which may not be fully malignant) and 15 cases with insufficient clinical data were also excluded.
Follow-up Data
The follow-up period was defined as 24 months between surgery and data collection. Clinicopathological parameters gathered for each tumor included histological type, grade, nodal status, tumor recurrence or distant metastasis, type of surgery (lumpectomy, regional mastectomy, unilateral mastectomy, or bilateral mastectomy), treatment modalities (no treatment after surgery, nonsteroidal anti-inflammatory drugs, nonsteroidal anti-inflammatory drugs plus chemotherapy,
Immunohistochemistry
Expression of proNGF, BDNF, and their receptors TrkA, TrkB, and p75NTR was assessed using immunohistochemistry (IHC) on 5-µm sections of the tumor, following the recommendations for IHC studies. 16 When several blocks were available for a single tumor, all archival hematoxylin and eosin–immunolabeled slides were reviewed by a veterinary anatomic pathologist (QP) and one representative slide of each tumor was selected. All antibodies (Table 1) are directed against human epitopes. However, the antibody against p75NTR has already been validated in canine tissues. 31 The human and canine BDNF and TrkB epitopes have 100% sequence identity. Human and canine epitopes of TrkA and proNGF share 87% sequence identity (data not shown). As neurotrophins are well known to play important role in brain development, 23,40,57 and are also found to be expressed in canine brain, 5,17,78 we incubated sections of canine cerebellum either with antibodies against neurotrophins and their receptors (positive control; Supplemental Figs. S1–S5), or with isotype-matched immunoglobulins (Table 1) at the same concentrations as the specific antibodies (negative control; Supplemental Figs. S7–S11). ProNGF, BDNF, TrkB, and p75NTR immunolabeling was observed in the cytoplasm and membrane of canine cerebellar cells, while TrkA immunolabeling was also observed in the nucleus. P75NTR immunolabeling could be observed as well on nerve fibers, which was used as an internal control (Supplemental Fig. S6). The negative control in which primary antibody was replaced by isotype-matched Ig was systematically included in samples of canine mammary carcinomas from all grades and histological types (data not shown). Immunostainings for proNGF, BDNF, TrkA, TrkB, and p75NTR were performed using a Discovery XT automated immunostaining device (Ventana Medical Systems). Slides were deparaffinized and a short antigen retrieval using Cell Conditioning solution (CC1, Ventana Medical Systems) was performed (95°C, 28 minutes, CC1 short). After incubation with primary antibodies or isotype-matched immunoglobulins, slides were incubated with biotin-free HRP multimer detection kit (DISCOVERY Ultramap anti-Rt/Rb/Ms HRP) and antibodies were revealed using an HRP driven chromogen (DISCOVERY ChromoMap DAB Kit). Reaction with 3,3-diaminobenzidine (DAB) led to a brown color for proNGF, BDNF, TrkA, and p75NTR. For TrkB, a hapten-based amplification system was used (DISCOVERY HQ-HRP detection kit), which led to a purple color. A second negative control in which primary antibody was omitted and replaced by reaction buffer was also included in each immunolabeling run. Slides were counterstained with a modified Mayer’s hematoxylin (Hematoxylin II, Roche) for 4 minutes and an aqueous solution of buffered lithium carbonate (Bluing Reagent, Roche) for 4 minutes. For TrkB, slide without primary antibody was systematically used to exclude nonspecific immunolabeling due to the amplification system. An example of nonspecific immunolabeling as intense as immunolabeling in presence of the antibody is shown in Supplemental Figure S12. In the case where negative control was as intense as the slide immunolabeled with the primary antibody, immunolabeling was considered as negative.
Antibodies and Reaction Conditions for Immunohistochemical Labelling of Canine Mammary Tumors.
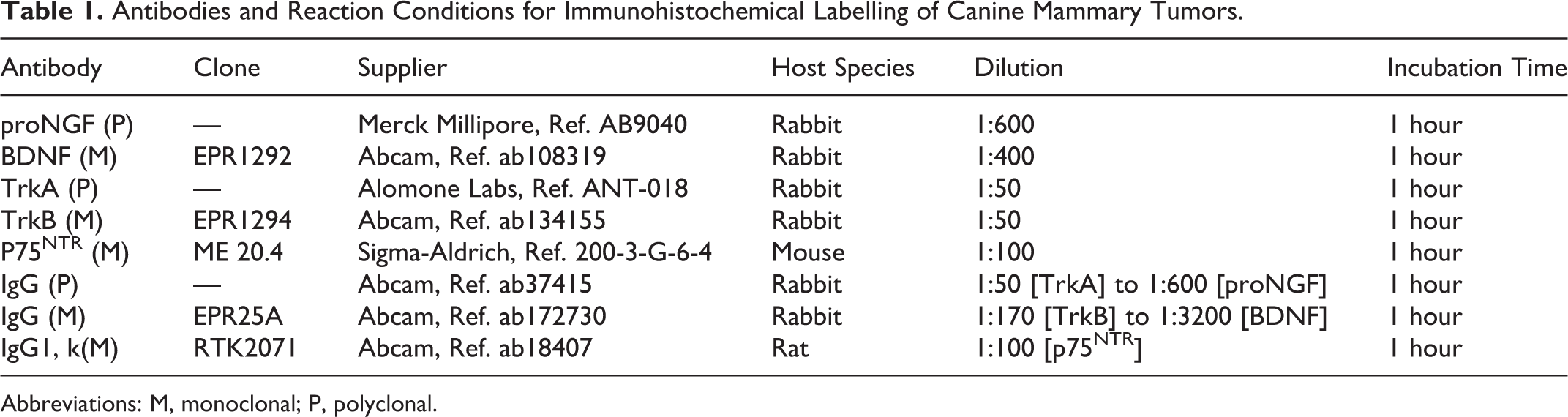
Abbreviations: M, monoclonal; P, polyclonal.
Immunohistochemical Evaluation
For each tumor, images of 10 medium-power cellular fields (20× objective, corresponding to 477 µm × 717 μm) were captured using a Nikon Eclipse Ni microscope. A consistent subset of proNGF-, BDNF-, TrkA-, TrkB-, and p75NTR-labeled slides were scored by 2 observers (BR, QP), including confirmation by an anatomic pathologist (QP) in order to reach a scoring assessment consensus. Immunolabeling for all markers were then scored by one observer (BR). All cases were scored without prior knowledge of the tumor pathology or patient outcome. On each field and for each marker, semiquantitative estimates of intensity of immunolabeling and percentage of positive cells were evaluated. Proportion of cells immunolabeled was expressed by a score ranging from 0 to 3 (0 = none or <5%; 1 = 5% to 25%; 2 = 25% to 50%; 3 = >50%). Immunolabeling intensity was also expressed by a score ranging from 0 to 3 (0 = Negative; 1 = Weak; 2 = Moderate; 3 = Strong), as presented in Supplemental Figures S13–S27. For TrkB, due to the use of an amplification system, intensity of immunolabeling was constant. Strength of the immunolabeling was assessed by evaluating the quantity of signal in the cells and was therefore measured as a vesicular immunolabeling (equivalent to weak), both vesicular and cytoplasmic immunolabeling (equivalent of moderate) or cytoplasmic immunolabeling (equivalent of strong). For each field, immunolabeling intensity (0–3) was multiplied by proportion of immunolabeled cells (0–3) to obtain an immunolabeling score ranging from 0 to 9. Final scores for the tumor were obtained by adding the scores of the 10 fields, which led to an immunolabeling score ranging from 0 to 90. The threshold used to define immune-positive or negative tumors was a proportion of immunolabeled cells ≥1/90. Tumors were considered as negative when the proportion of immunolabeled was 0/90, which corresponded to a complete lack of labeling.
Classification by Components
In addition to the frequently used classifications of canine mammary tumors according to grade and histological type, we also identified different cellular types and classified tumors into 3 components. The luminal component corresponded to all structures related to a lumen (ductal, tubular, or papillary). The myoepithelial component corresponded to myoepithelial or myoepithelial-like (spindle-shaped) cells. The diffuse component corresponded to cells closely packed in solid areas or anaplastic areas. Every tumor could present with 1, 2, or 3 components.
Statistical Analysis
The quantitative variables were expressed by mean and standard deviation or by median and interquartile range. The normality of the distributions was checked graphically and using the Shapiro-Wilk test. The qualitative variables were described using frequencies and percentages.
The relationship between the expression of a marker and the types of component (luminal, diffuse, and myoepithelial) was estimated using generalized linear mixed models. Post hoc pairwise comparisons were performed in case of global significant difference, and a Bonferroni correction for multiple comparisons was applied.
In case of sufficient sample size, proportion of expression of marker between groups of clinicopathological parameters were compared using χ2 tests. In case of expected counts lower than 5, Fisher’s exact tests were used.
Staining scores of markers were compared between grade and histological type using nonparametric Kruskal-Wallis tests; and between presence and absence of emboli, lymph node infiltration, tumor recurrences, and distant metastases using nonparametric Mann-Whitney
Survival curves were estimated using Kaplan-Meier method. Survival curves were compared between tumor groups expressing or not a marker using a log-rank test. Comparisons were further adjusted on age using a Cox proportional regression model; hazard ratios were estimated as effect size.
Finally, we assessed the relationship between the staining score of the different markers and overall survival in Cox proportional hazard regression model treating scores as continuous variables. Proportionality hazards and log-linearity assumptions were examined using Schoenfeld and martingale residual plots.
Data were analyzed using the SAS software version 9.4 (SAS Institute Inc, Cary, NC), and all statistical tests were performed with a 2-tailed α risk of 0.05.
Results
Animal Data and Histopathological Characteristics of Tumors
The study included 96 female dogs. Of those with known neuter status, at the time of diagnosis, most were intact: 67/88 (76%). Of 38 breeds represented, the most frequent were Yorkshire Terrier (13/96), Brittany Spaniels (7/96), and Shih Tzu (6/96). The mean age of dogs at the time of surgical removal of tumors was 10.1 ± 2.1 years (mean ± SD; range 3.3–15.5 years of age). According to the Peña grading method, tumors were classified as grade I (30/96; 31%), grade II (21/96; 22%), or grade III (32/96; 33%). 48 Inflammatory mammary carcinomas represented 13/96 cases (14%). Tumors were histologically classified into subtypes according to the Goldschmidt classification of canine mammary tumors. 24 Our cohort was further classified into 4 major histological types: solid, tubular simple, tubular complex, and anaplastic (Table 2). Tubular simple and tubular complex tumors are recognized to have the best prognosis, while solid and even more anaplastic tumors have a poor prognosis. 46,55 When tumors were heterogeneous (ie, solid and tubular or adenosquamous and tubular, etc) the solid component was thus privileged over the tubular one as the first is commonly associated with worse prognosis. Histological evaluation yielded 31/96 tubular simple carcinomas (32%), 17/96 tubular complex carcinomas (18%), 34/96 solid carcinomas (35%), and 14/96 anaplastic carcinomas (15%). Concordance between grades and histological types is presented in Table 3.
Relationship Between Morphological Diagnosis and Histological Type of Malignant Canine Mammary Tumors.

Histological Malignancy Grades and Histological Diagnosis in the Cohort.a
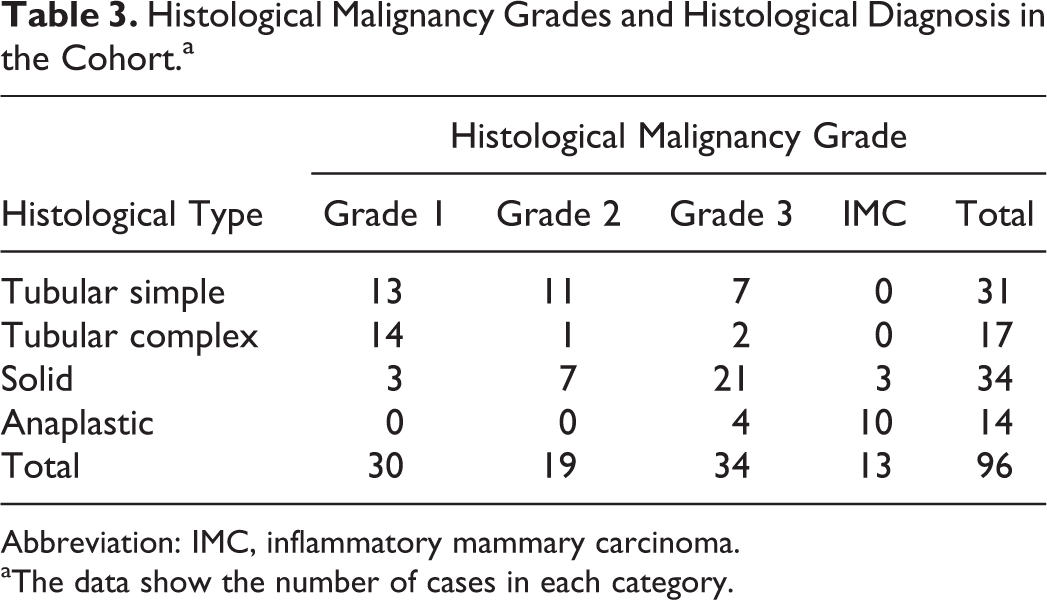
Abbreviation: IMC, inflammatory mammary carcinoma.
aThe data show the number of cases in each category.
Tumors were also classified according to criteria of aggressiveness. Emboli were present in 29/96 cases (30%) including 16 grade III tumors and 13 inflammatory mammary carcinomas. Lymph nodes were available in 49/96 cases (51%), with confirmed infiltration by tumor cells in 17/49 cases (35%).
Tumors were finally classified in 3 groups according to the components: 75/96 tumors (78%) contained a luminal component, 37/96 (39%) had a myoepithelial component, and 68/96 (71%) had a diffuse component.
The individual-animal data are available in Supplemental Table S1.
proNGF and BDNF Expression
Both proNGF and BDNF immunolabelings were predominantly cytoplasmic. proNGF (Figs. 1–3) and BDNF (Figs. 4–6) immunolabeling patterns were similar, with labeling being either locally extensive or diffuse. The intensity of immunolabeling varied from weak to strong within the same tumor. proNGF expression (Table 4) was significantly associated with the component type (
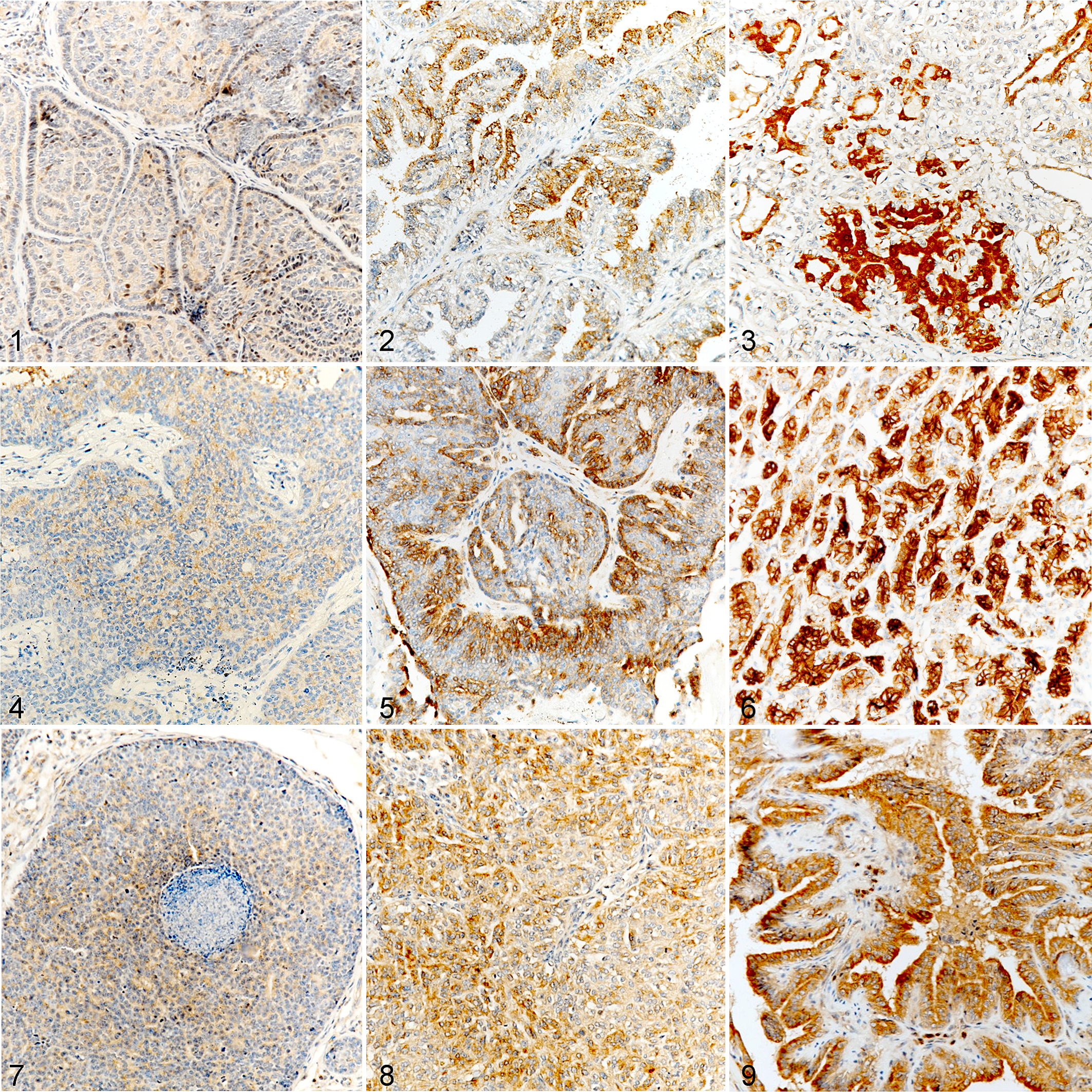
Malignant mammary carcinomas, dog. Immunohistochemistry (brown labeling) for proNGF, BDNF, and TrkA.
Expression of proNGF, BDNF, TrkA, TrkB, and p75NTR in Relation to the Type of Componenta.

Abbreviation: NA, not applicable.
a Values are presented as frequency (percentage). LGroup significantly different from Luminal; DGroup significantly different from Diffuse; MGroup significantly different from Myoepithelial.
Expression of proNGF, BDNF, TrkA, TrkB, and p75NTR in Relation to Clinicopathological Parametersa.
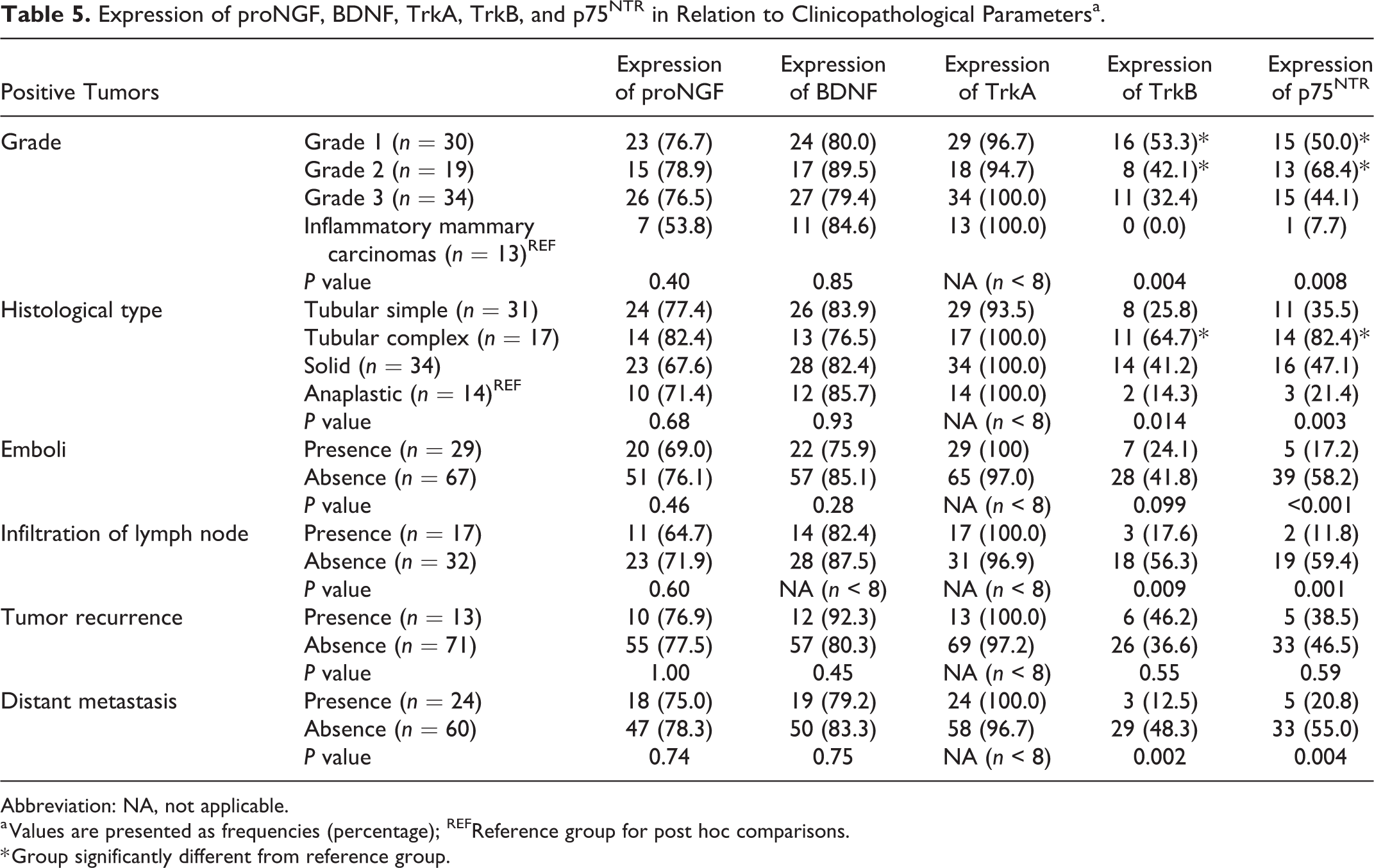
Abbreviation: NA, not applicable.
a Values are presented as frequencies (percentage); REFReference group for post hoc comparisons.
* Group significantly different from reference group.
Staining Score of proNGF, BDNF, TrkA, TrkB, and p75NTR in Relation to Clinicopathological Parametersa.
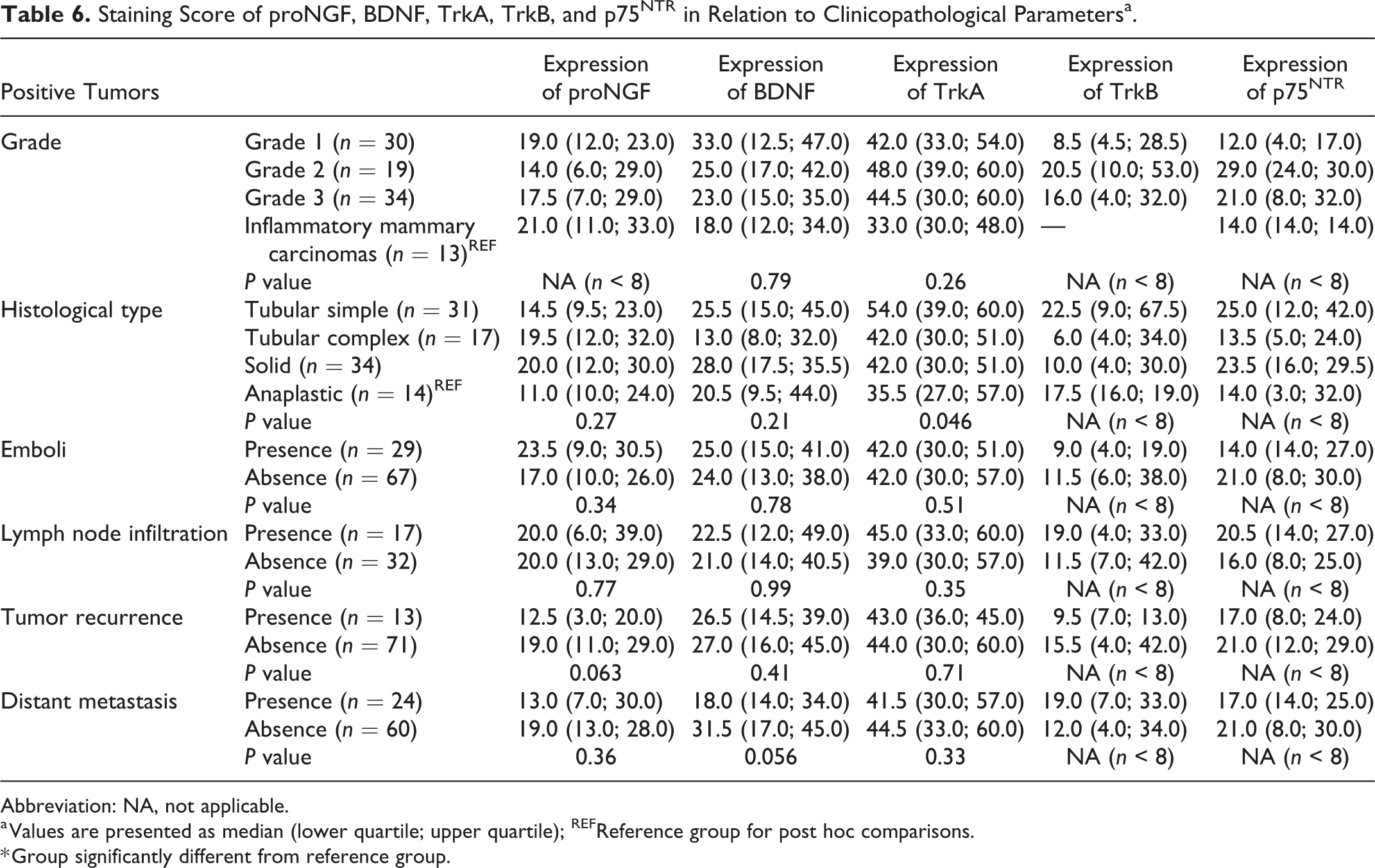
Abbreviation: NA, not applicable.
a Values are presented as median (lower quartile; upper quartile); REFReference group for post hoc comparisons.
* Group significantly different from reference group.
TrkA Expression
TrkA immunolabeling was predominantly cytoplasmic. TrkA immunolabeling pattern was predominantly diffuse, with a constant intensity throughout tumor (Figs. 7–9). TrkA was expressed in 98% of tumors (94/96). No statistical analysis could be performed on its association with components (Table 4) or clinicopathological parameters (Table 5) as a consequence of its very high prevalence. Immunolabeling scores for TrkA (Table 6) ranged from 0 to 90. Positive tumors presented a median score of 42 (Q1: 30.0; Q3: 57.0). Levels of expression of TrkA were not associated with grade (
TrkB Expression
TrkB immunolabeling was almost entirely cytoplasmic. TrkB immunolabeling pattern was the most variable. Different patterns were observed like the immunolabeling of a single small area of cells, a multifocal immunolabeling or a strong diffuse, immunolabeling (Figs. 10–13). Nonspecific immunolabeling was identified in 9 tumors: 2 of them presented with extensive nonspecific signal (1 grade 3 and 1 inflammatory mammary carcinoma), 2 tumors presented with moderate non-specific signal (1 grade 1 and 1 inflammatory mammary carcinoma), and 5 had minimal nonspecific immunolabeling that did not affect the interpretation. Non-specific staining was more frequent in emboli.
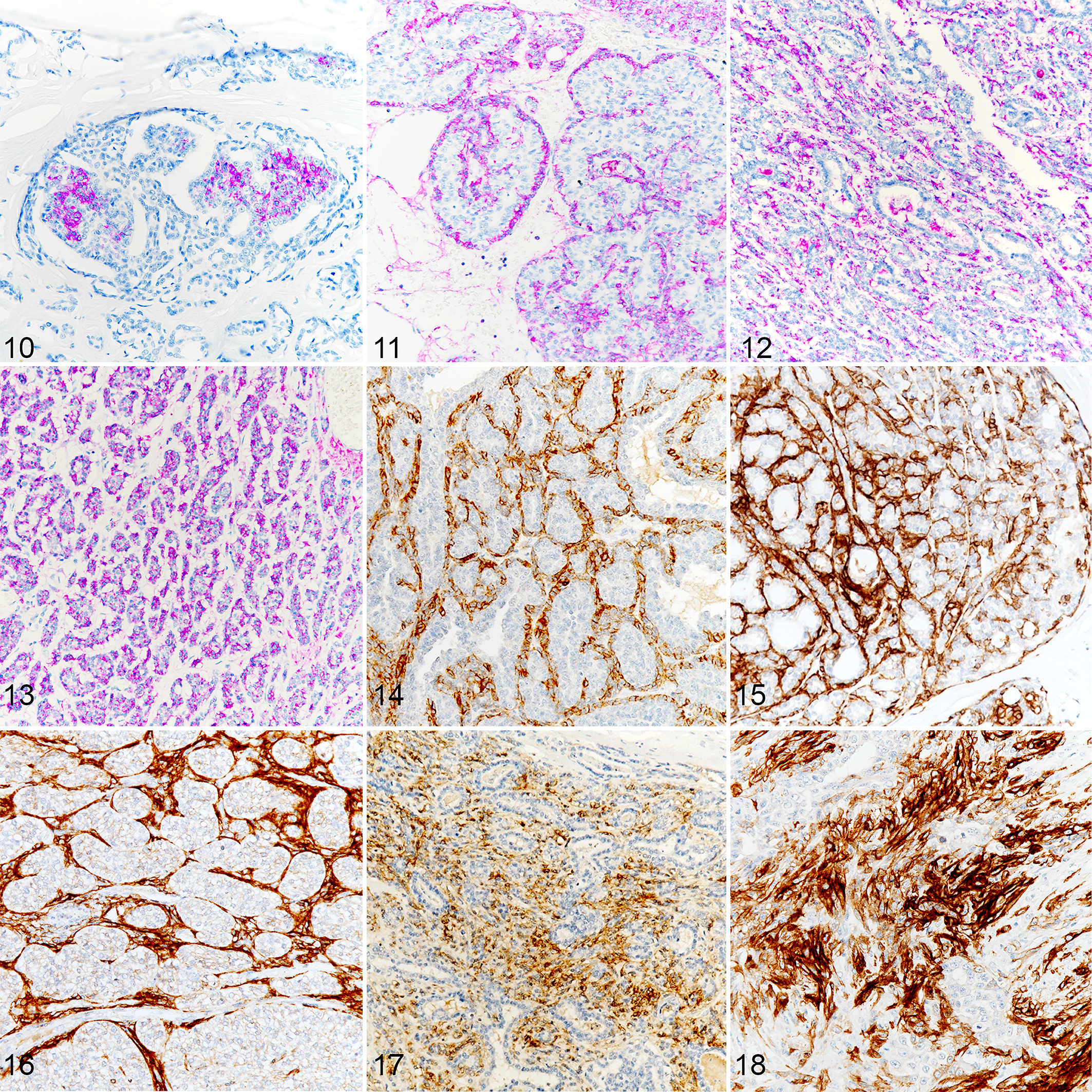
Malignant mammary carcinomas, dog. Immunohistochemistry for TrkB (purple labeling) and p75NTR (brown labeling).
TrkB expression was not significantly associated with the tumor component type (
Collectively, our data indicated that TrkB expression was associated with favorable clinicopathological parameters.
P75NTR Expression
P75NTR was expressed in the cytoplasm and on the plasma membrane (Figs. 14–18). Within the same tumor, all areas of myoepithelial components immunolabeled with similar intensity. P75NTR expression was strongly associated with the component (
P75NTR was expressed in 44/96 tumors (46%). P75NTR expression (Table 5) was significantly associated with lower grades (
Globally, p75NTR was associated with favorable clinicopathological parameters.
Survival Study
Follow-up data were available for 84/96 cases (88%). These 84 cases included 26 grade I (31%), 19 grade II (23%), 31 grade III (37%), and 8 inflammatory mammary carcinomas (10%). During the follow-up period, 13 animals relapsed and 24 animals died or were euthanized due to metastatic disease and/or local recurrence. Animals that died for reasons unrelated to cancer or that were still alive at the end of the 2-year follow-up period were censored. The median DFS time was 380 days and the median OS time was 417 days.
Survival was compared between subgroups regarding expression or not of a particular marker. No survival analysis could be performed regarding TrkA due to its ubiquitous expression. No association was found between expression of proNGF or BDNF and survival in the cohort. On the other side, in the whole cohort, expression of both TrkB and p75NTR was significantly associated with a better survival (
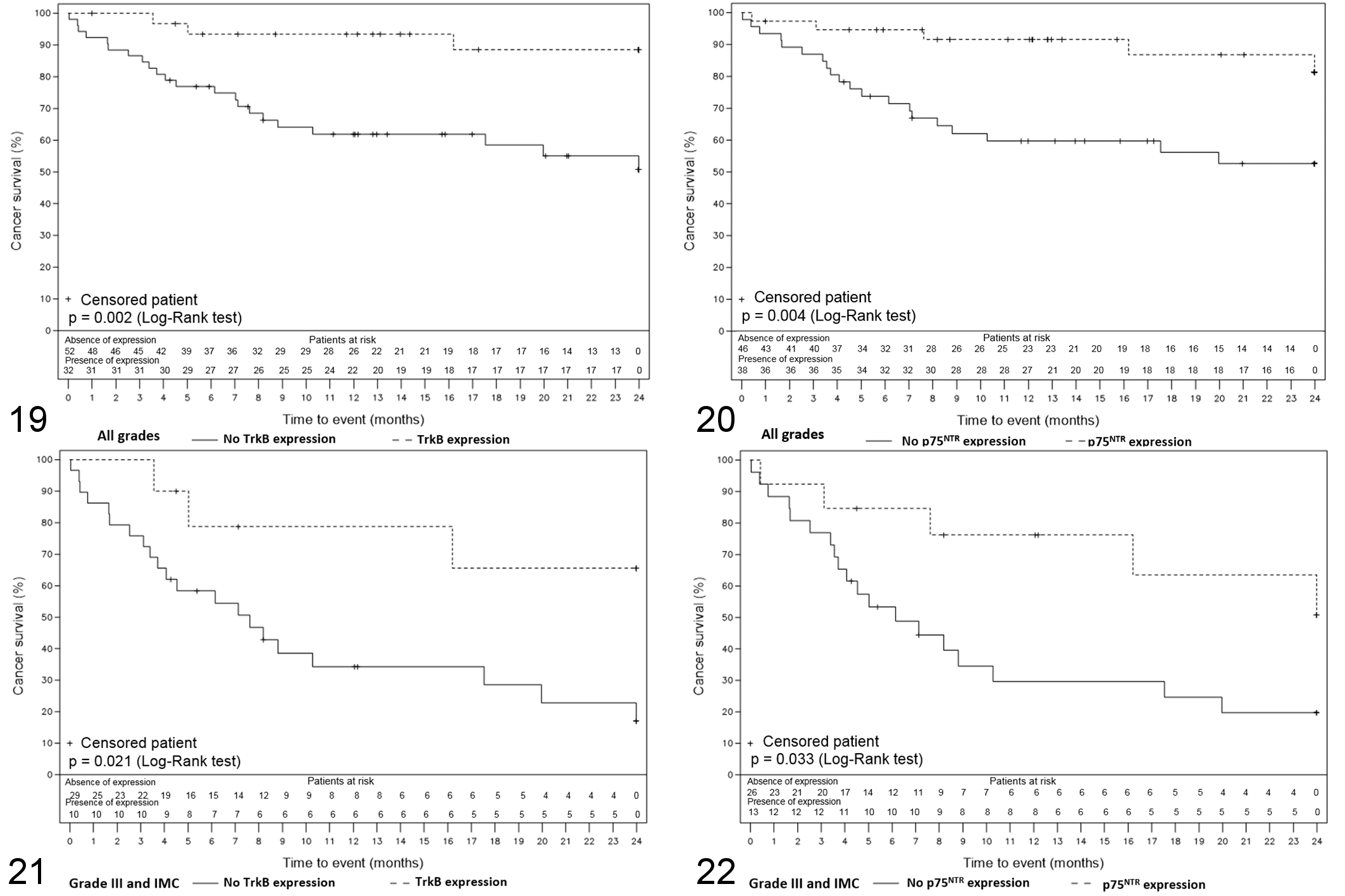
Kaplan-Meier overall survival curves representing the survival of the dogs included in the survival study according to time. The numbers under the
Discussion
The aim of our study was to characterize the expression of proNGF and BDNF as well as their corresponding receptors TrkA, TrkB, and the common receptor p75NTR in a well-defined cohort of canine mammary carcinomas and to correlate them with tumor grade, histological type, emboli and lymph node status, and survival. We found that proNGF and BDNF were largely expressed in canine mammary carcinomas and their expression did not seem to be associated with clinicopathological parameters. TrkA was expressed nearly ubiquitously in canine mammary carcinomas, with a globally high level of expression. TrkB and p75NTR seem to be good prognostic factors as their expression is associated with criteria of low aggressiveness and a more favorable outcome both in the whole cohort and in the poor prognosis subgroup.
Previous literature described an association between TrkA overexpression and tumor aggressiveness
BDNF/TrkB signaling plays important roles in tumor metastasis and is considered as a potential therapeutic target in the treatment of human breast cancer. 14,59,71 Overexpression of BDNF and TrkB is associated with a poor clinical outcome in patients suffering from human bladder cancer, neuroblastoma, and breast carcinoma. 29,37,71,75 More specifically, upregulation of the BDNF/TrkB signaling pathway seems to be involved in early recurrence in triple negative breast cancers. 73 In our study, BDNF was widely expressed in canine mammary carcinomas but its expression could not be associated with adverse clinicopathological parameters or to survival. Contrary to what is known in human breast cancer, TrkB was associated with favorable clinicopathological parameters and a more favorable outcome. Even though nonspecific immunolabeling was observed due to the amplification system, it remained marginal, and was observed equally in low-grade and high-grade tumors. Thus, this nonspecific immunolabeling may have hidden a true TrkB immunolabeling in a small proportion of our tumors, but it is not suspect to significantly impact our results or interpretation.
Finally, in our study, p75NTR is associated with favorable clinicopathological parameters and good prognosis. Studies on breast cancer cell lines have shown that p75NTR signaling could induce breast cancer cell survival. 12,19,76 On the other side, p75NTR expression has been associated with favorable outcome in primary breast cancers. 3,58,65 Several studies have demonstrated that p75NTR is a marker of breast myoepithelial cells in women. 51,58 These cells are poorly studied in human breast cancer because of their scarcity, but could be associated with a good prognosis. 15,38 Myoepithelial cells would be natural paracrine suppressors of invasion and metastasis and may specifically regulate the in situ to invasive carcinoma transition. 70 Based on these functions, normal mammary myoepithelial cells have been called “natural tumor suppressors.” 50 Our results suggest that p75NTR could be a marker of mammary myoepithelial cells in dogs as well. It was in most cases expressed in the myoepithelial component of tumor samples and could also be observed in physiological myoepithelial cells in areas of healthy tissues entrapped in the tumors (data not shown). These observations are consistent with the predominance of p75NTR in tubular complex tumors, a specific canine tumor type in which the tumor exhibits both epithelial and myoepithelial proliferation. 46 The identification of myoepithelial cells was based on a visual estimation performed by 2 persons including a veterinary anatomic pathologist (QP). Further studies are needed to confirm p75NTR expression in myoepithelial cells, by using specific markers such as smooth muscle actin, calponin, p63, CD10/CALLA, cytokeratins CK 5/6 or CK14 or CK17, or epidermal growth factor EGFR. 56,64,65
In conclusion, our study was the first one to assess the expression and prognostic significance of neurotrophins and their receptors in canine mammary tumors. Results still have to be sharpened but first tendencies are being drawn up. Especially, TrkB and p75NTR could be linked to myoepithelial cells, which highlights the potential interest of the canine model to study the role of myoepithelial cells in breast tumors development and invasion. Of additional importance, although Trk inhibitors are currently under investigation as possible therapeutic options in human cancers as well as several canine tumors, good prognosis values of TrkB and p75NTR together with the absence of any clinical value of TrkA expression suggest that canine mammary tumors might not be a suitable model to assess a potential therapeutic employment of Trk inhibitors. Furthermore, it will be also crucial to evaluate the expression of neurotrophins and their receptors in normal mammary tissues to determine if expression of these proteins is uniquely increased in mammary cancer or is this just a reflection of their normal mammary expression.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Rogez_et_al - Expression and Prognostic Significance of Neurotrophins and Their Receptors in Canine Mammary Tumors
Supplemental Material, Combined_supplemental_materials-Rogez_et_al for Expression and Prognostic Significance of Neurotrophins and Their Receptors in Canine Mammary Tumors by Bernadette Rogez, Quentin Pascal, Audrey Bobillier, François Machuron, Robert-Alain Toillon, Dominique Tierny, Valérie Chopin and Xuefen Le Bourhis in Veterinary Pathology
Supplemental Material
Supplemental Material, Supplemental_table_10.03.19 - Expression and Prognostic Significance of Neurotrophins and Their Receptors in Canine Mammary Tumors
Supplemental Material, Supplemental_table_10.03.19 for Expression and Prognostic Significance of Neurotrophins and Their Receptors in Canine Mammary Tumors by Bernadette Rogez, Quentin Pascal, Audrey Bobillier, François Machuron, Robert-Alain Toillon, Dominique Tierny, Valérie Chopin and Xuefen Le Bourhis in Veterinary Pathology
Footnotes
Acknowledgements
The authors are grateful to the staff of OCR (Oncovet Clinical Research) for their support and to the biostatisticians of Lille University Hospital, particularly Hélène Behal and Julien Labreuche, for their time and implication. Special thanks are addressed to Tiffany Cheval and Emmanuel Bouchaert for their excellent technical assistance, and to Agata Rybicka, Virginie Coste, and Thomas Bucher for their help in building and improving this study. Finally, we would like to thank the referring veterinarians who gave us the clinical and follow-up data and the owners of the dogs included in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by FEDER FSE Nord-Pas de Calais, GEFLUC Flandres Artois, SIRIC OncoLille and OCR (Oncovet Clinical Research).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
