Abstract
A 9-y-old male Boxer dog developed a mandibular skin tumor, which histologically had a locally invasive growth pattern composed of bilayered structures of inner eosinophilic cuboidal tumor cells and outer clear polygonal tumor cells with cytoplasm containing glycogen granules. Both cell populations gradually changed from low-grade morphologic features to highly anaplastic ones. Immunohistochemically, the eosinophilic tumor cells were positive for cytokeratin 8, a useful marker for luminal epithelial cells. In contrast, the clear tumor cells expressed several myoepithelial markers, including α-smooth muscle actin, p63, and cytokeratin 14. Based on these histologic and immunohistochemical characteristics, we diagnosed this apocrine sweat gland tumor as a carcinoma-and-malignant myoepithelioma with high-grade transformation of both luminal and myoepithelial cells. Our case may be a helpful reference for the histogenesis of carcinoma-and-malignant myoepithelioma, in which both the luminal epithelial and myoepithelial components are malignant.
Apocrine sweat glands, mammary glands, and salivary glands share the common characteristics of tubuloacinar structures consisting of luminal epithelial cells and basal myoepithelial cells. In particular, because the mammary gland is a modified gland that evolved from the epidermal apocrine gland, apocrine sweat glands and mammary glands develop biphasic neoplasms composed of both luminal epithelial and myoepithelial cells common to these glands.1,2,15 These glands may have bipotential stem or progenitor cells that give rise to both luminal epithelial and myoepithelial cells, 11 which are the putative origins of biphasic neoplasms.3,8–10
The classification of canine mammary tumors has advanced with the introduction of immunohistochemistry (IHC). 19 Carcinoma-and-malignant myoepithelioma, a diagnostic entity consisting of both malignant luminal epithelial and malignant myoepithelial cells, has been introduced. This classification was based on immunohistochemical evidence of malignant myoepithelial cells that are morphologically similar to malignant luminal epithelial cells.18,19 In canine apocrine neoplasms, myoepithelial cells of complex carcinomas have been de-scribed traditionally as having a low-grade histologic appearance. 5 Rare cases have been reported of malignant changes in the myoepithelial cells4,14; however, the details of this tumor entity remain unclear. Therefore, we present here a case arising in the canine skin of an apocrine carcinoma-and-malignant myoepithelioma with gradual progression to high-grade morphology.
A 9-y-old male Boxer dog developed a 3.0 × 2.0 × 2.0-cm ulcerative mass in the mandibular skin. The mass had enlarged gradually since its detection one month earlier; no other clinical signs had been observed. During surgical removal, no connection was noted between the mass and the underlying tissue. Based on the preoperative clinical examination, we did not suspect metastasis to the lungs or lymph nodes. Moreover, we did not observe any significant abnormalities during suture removal on postoperative day 16.
The mass was fixed in 10% neutral-buffered formalin, processed routinely, and serial sections stained with H&E and periodic acid–Schiff (PAS) reaction with or without diastase digestion, or used for IHC. We performed single IHC using the labeled streptavidin–biotin method and 3,3′-diaminobenzidine (DAB) substrate (Table 1). Subsequently, we also performed double IHC using monoclonal antibodies against α–smooth muscle actin (SMA) and cytokeratin 8 (CK8; Histofine simple stain MAX-PO, Simple stain AP, Nichirei), along with DAB and new fuchsin substrates, using methods described previously. 18
Primary antibodies, sources, clones, dilutions, and positive controls used in immunohistochemistry of an apocrine carcinoma-and-malignant myoepithelioma in a dog.
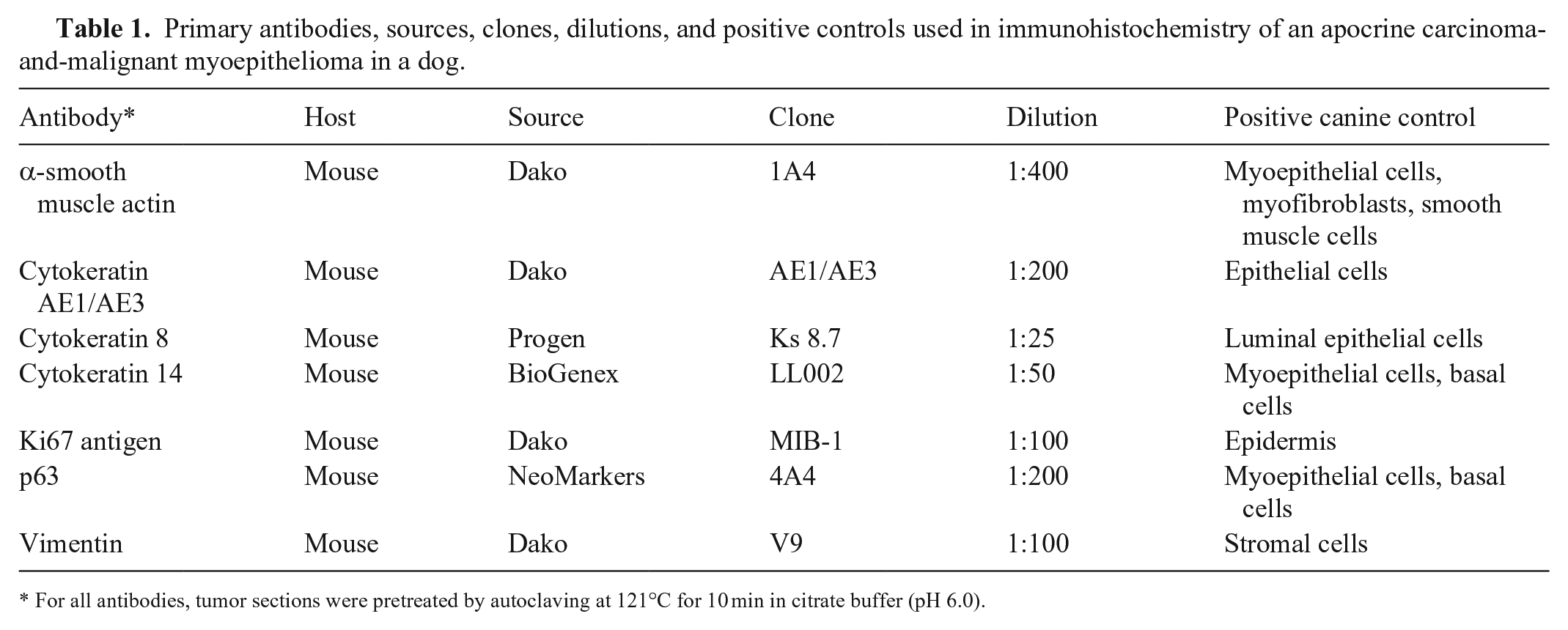
For all antibodies, tumor sections were pretreated by autoclaving at 121°C for 10 min in citrate buffer (pH 6.0).
Microscopically, the mass was lobulated with highly cellular neoplastic tissue present from the epidermis to the subcutaneous muscle (Fig. 1). The tumor was highly invasive, particularly in the superficial area of the mass. The overlying skin was ulcerated, with neutrophilic inflammation. The central area of each tumor lobe was necrotic. Cellular and structural atypia increased gradually from deep to superficial parts of the tumor.
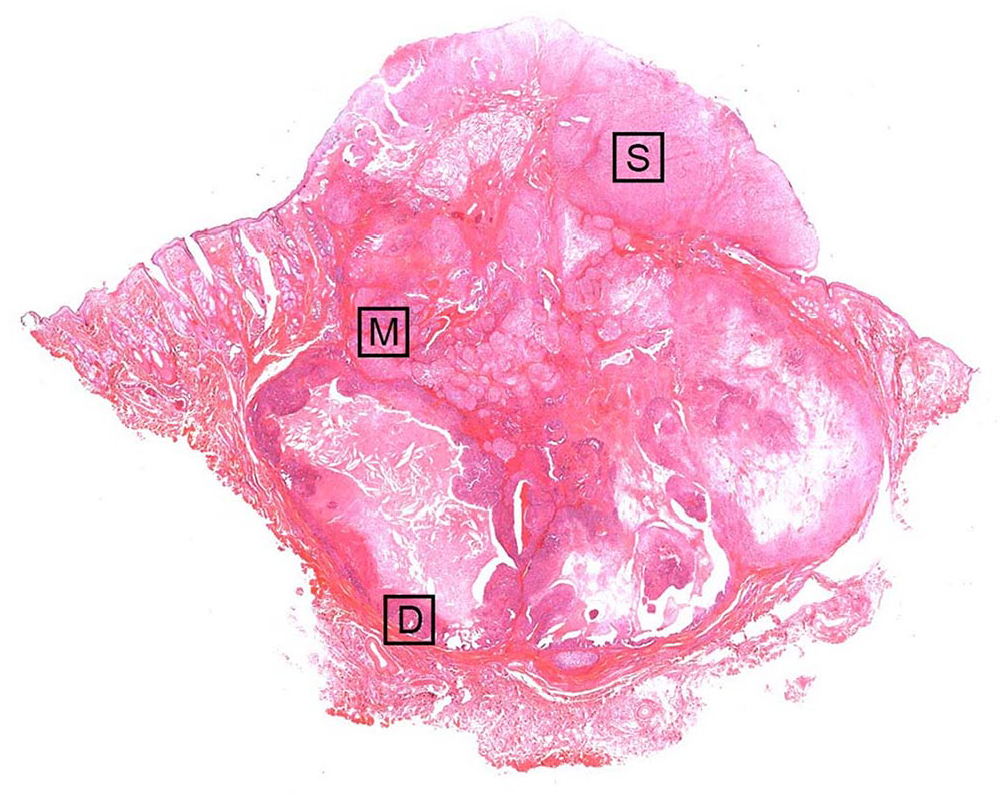
Cutaneous mass on the mandible of a 9-y-old male Boxer dog. The tumor extends from the subcutis through the epidermis. Squares (D, M, S) correspond to the sites (deep, middle, and superficial areas) in Figures 2 and 3, and Suppl. Figures 1–3. H&E.
Deep tumor lobes were composed of well-formed tubular structures lined by a monolayer of eosinophilic columnar tumor cells with 1–3 surrounding layers of clear polygonal myoepithelial cells (Fig. 2A; Suppl. Fig. 1). The eosinophilic tumor cells had a moderate amount of cytoplasm and uniform, round nuclei with small nucleoli. The clear myoepithelial tumor cells were smaller and had a moderate amount of cytoplasm with small vacuoles, including PAS-positive (diastase-digestible) glycogen granules (Fig. 2B). Their nuclei were smaller and round without apparent nucleoli. The mitotic count of eosinophilic cells and clear myoepithelial cells per 10 hpfs (2.37 mm2) was 8 and 3, respectively, in the deep area. In a count of 700 cells, we observed Ki67-positive nuclei in 5.4% of eosinophilic cells and 4.6% of clear myoepithelial cells.
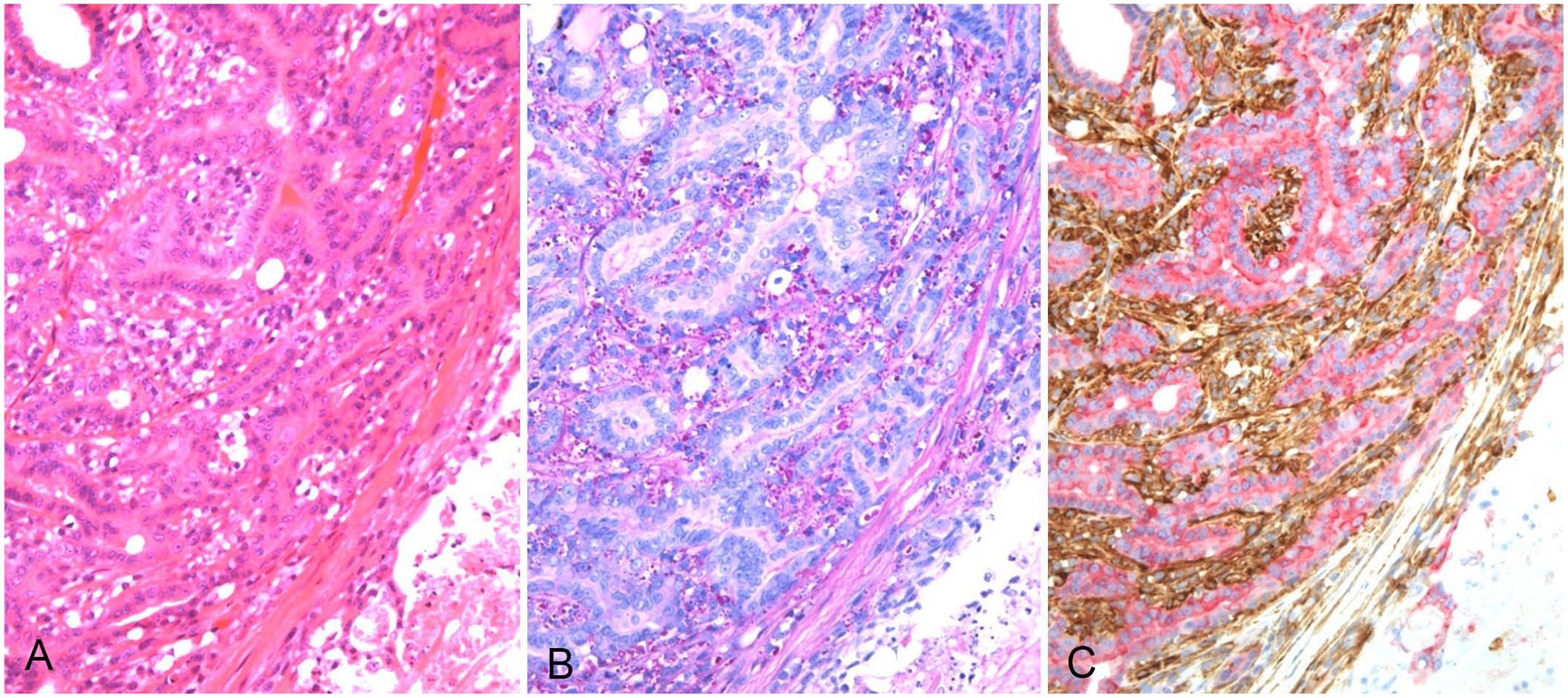
Deep area of a cutaneous mass on the mandible of a 9-y-old male Boxer dog.
In the middle level of the tumor, the tubular structures became more irregular and partially pluristratified, composed of eosinophilic columnar cells with an increased nuclear:cytoplasmic (N:C) ratio and disturbed polarity (Suppl. Fig. 2). In the clear cell population with abundant PAS-positive intracytoplasmic granules, nuclei were larger, and nucleoli were larger and more prominent. In the middle area, the mitotic counts per 2.37 mm2 for eosinophilic cells and clear myoepithelial cells were 7 and 5, and the Ki67 indices were 5.9 and 11.9, respectively.
In the superficial area, both cell populations had anaplastic features with prominent anisokaryosis and anisocytosis, and were so similar as to be indistinguishable (Fig. 3A; Suppl. Fig. 3). The eosinophilic cells did not form tubules, but rather bizarre clusters buried in sheets or nests of atypical clear cells. The eosinophilic cells were large and polygonal, with large nuclei containing prominent eosinophilic nucleoli. The clear myoepithelial cells were large, with nuclei containing multiple large nucleoli; their cytoplasm contained vacuoles, which were PAS-positive (Fig. 3B). Our mitotic count for eosinophilic cells and clear myoepithelial cells per 2.37 mm2 in the superficial area were 23 and 21, and the Ki67 indices were 13.4 and 17.7, respectively.
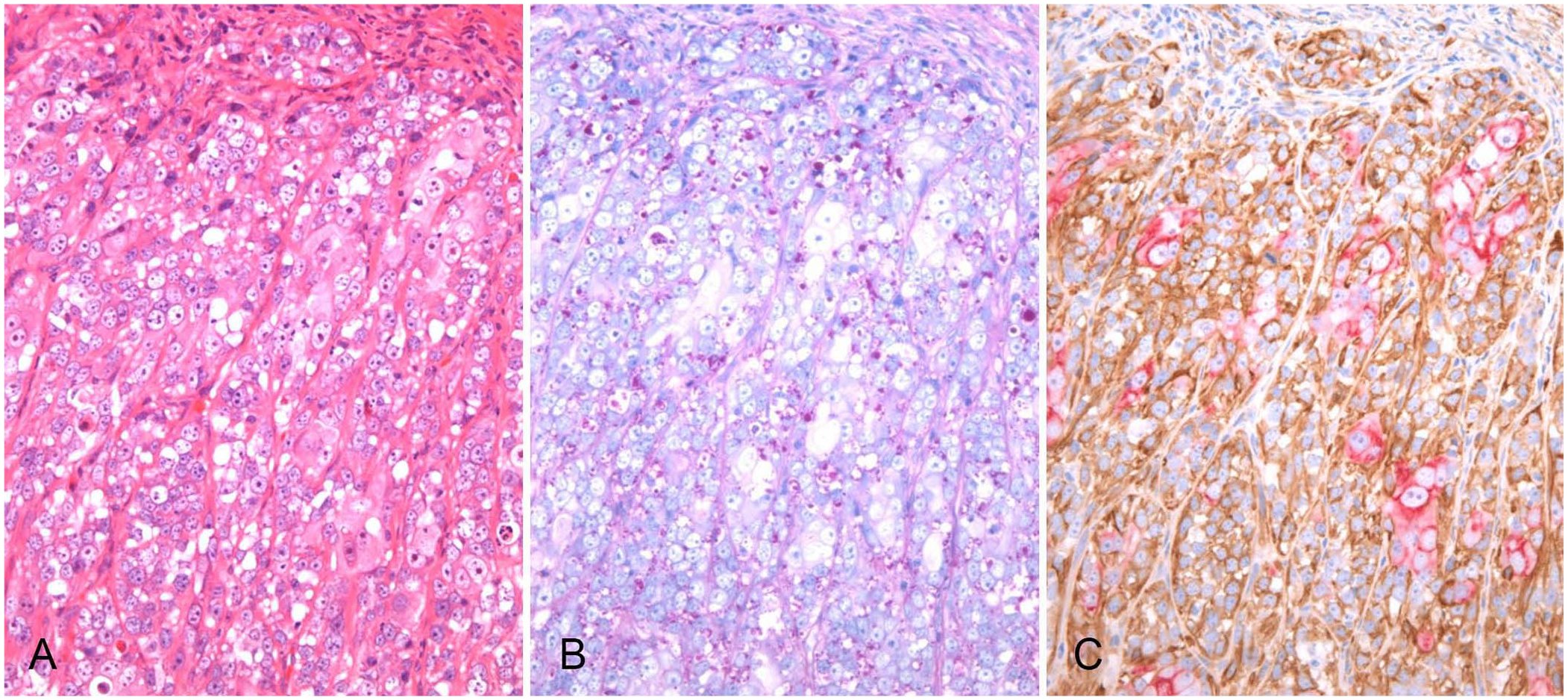
The superficial area of a cutaneous mass on the mandible of a 9-y-old male Boxer dog.
Immunohistochemically, both cell populations had a consistent immunophenotype with slight differences in staining intensity by area (Figs. 2, 3; Suppl. Figs. 1–3). Eosinophilic cells had positive CK AE1/AE3 and CK8 immunolabeling; however, labeling intensities were weaker in the superficial area than in the deep area. Eosinophilic cells were consistently negative for SMA, p63, and CK14 immunolabeling. Vimentin immunolabeling was negative in the eosinophilic cells of the deep area but sporadically positive in the superficial area. In contrast, clear myoepithelial cells were positive for SMA, p63, CK14, and vimentin immunolabeling. However, the immunolabeling intensities of SMA, p63, and CK14 were weaker in the clear myoepithelial cells in the superficial area than in the deep area. CK AE1/AE3 labeling was focal; CK8 labeling was consistently negative in the clear myoepithelial cells. The bilayered structure of CK8-positive eosinophilic cells and SMA-positive clear cells was observed not only in the deep and middle areas (Fig. 2C; Suppl. Fig. 2) but also in the superficial area, despite its anaplastic features (Fig. 3C).
The eosinophilic columnar tumor cells found lining lumens expressed CK8, a useful marker for luminal epithelial cells; clear polygonal tumor cells expressed several myoepithelial markers, including SMA, p63, and CK14.2,12,15,16 Traditionally, the myoepithelial portion of canine complex apocrine carcinoma has been thought to have a spindle-shape and low-grade histologic appearance with myxoid or chondroid features. 5 The clear cell type of neoplastic myoepithelial cells is rarely seen in canine cutaneous neoplasms but is well-known in human salivary, breast, and cutaneous neoplasms, and in canine mammary neoplasms.1,2,7,18 In human salivary neoplasms, the clear cytoplasm of myoepithelial cells results from glycogen accumulation. 1 In our case, glycogen was demonstrated in the cytoplasm of the clear myoepithelial cells by the PAS reaction.
The differential diagnosis for our case included clear cell adnexal carcinoma (CCAC), which has been reported in the canine skin,4,5,13,16 and consists of clear tumor cells containing glycogen granules. However, SMA is not expressed in the clear tumor cells of CCAC, indicating the absence of myoepithelial differentiation.4,13,16 In addition, CCACs usually have areas of hair follicle differentiation and a limited number of well-formed tubules lined by mature luminal epithelial tumor cells. 13
The characteristics of the tumor in our case are the bilayered structure of inner luminal epithelial cells and outer clear myoepithelial cells, without myxoid or chondroid features, which corresponds to epithelial-myoepithelial carcinoma (EMC) in the tumor classification of human salivary glands. 1 Human EMC rarely has a high-grade component, characterized by a greater degree of nuclear atypia and frequent mitoses. Human EMC has 2 known progressive forms: “EMC with myoepithelial anaplasia” and “EMC with high-grade transformation (or dedifferentiated EMC),” which lacks evidence of myoepithelial differentiation. 1 According to the classification of mammary tumors in dogs, tumors corresponding to human EMC can be diagnosed as “carcinoma-and-malignant myoepithelioma.” 17 Therefore, we diagnosed our case as carcinoma-and-malignant myoepithelioma of the canine apocrine gland. Transition was gradual from the typical histologic area to more aggressive and anaplastic areas. In the anaplastic superficial area in our case, IHC demonstrated that the biphasic structure of luminal epithelial and myoepithelial cells was maintained, unlike the high-grade component that is observed rarely in human EMC. However, it should be noted that the comparisons to high-grade components in human salivary gland tumors may be limited given the absence of a validated grading scheme for apocrine tumors.
Although the histogenesis of carcinoma-and-malignant myoepithelioma is poorly understood, our case (in which luminal epithelial and myoepithelial cell populations gradually increase the degree of atypia in parallel) may lead to 2 hypotheses (Fig. 4). Luminal epithelial and myoepithelial cell populations may have undergone simultaneous and independent phenotypic alterations, and come together in a unitary structure. Tumor heterogeneity is considered to be induced not only by the clonal outgrowth of tumor cells with genetic mutations but also by epigenetic alterations of tumor cells resulting from the interaction with the tumor microenvironment. 6 Specialized stromal cells and the extracellular matrix have been observed also in canine malignancies, including mammary tumors. 17 Because the superficial layer of our tumor mass was ulcerated, inflammatory cells, angiogenesis, and cytokines may have contributed to the increased atypia of both luminal epithelial and myoepithelial tumor cells. However, even in the superficial area of the tumor in which both tumor cell types had significant atypia, the bilayered architecture was preserved, with luminal epithelial tumor cells on the inner side and myoepithelial tumor cells on the outer side in the tumor nests. This suggests that the observed changes may not be solely attributed to the effects of the tumor microenvironment. A second possibility based on the cancer stem cell theory is therefore possible. A small population of tumor-initiating cells, or cancer stem cells, may be involved in the initiation and maintenance of cancer in human and canine neoplasms.8,9,11 A small population of bipotential tumor-initiating cells gradually accumulating genetic mutations might give rise to both atypical luminal epithelial and myoepithelial cells that form bilayered structures. Our case may provide a helpful reference for the histogenesis of carcinoma-and-malignant myoepithelioma of the mammary glands as well as apocrine sweat glands. Further collection of such cases in the canine mammary and apocrine glands is needed to elucidate the histogenesis of carcinoma-and-malignant myoepithelioma.
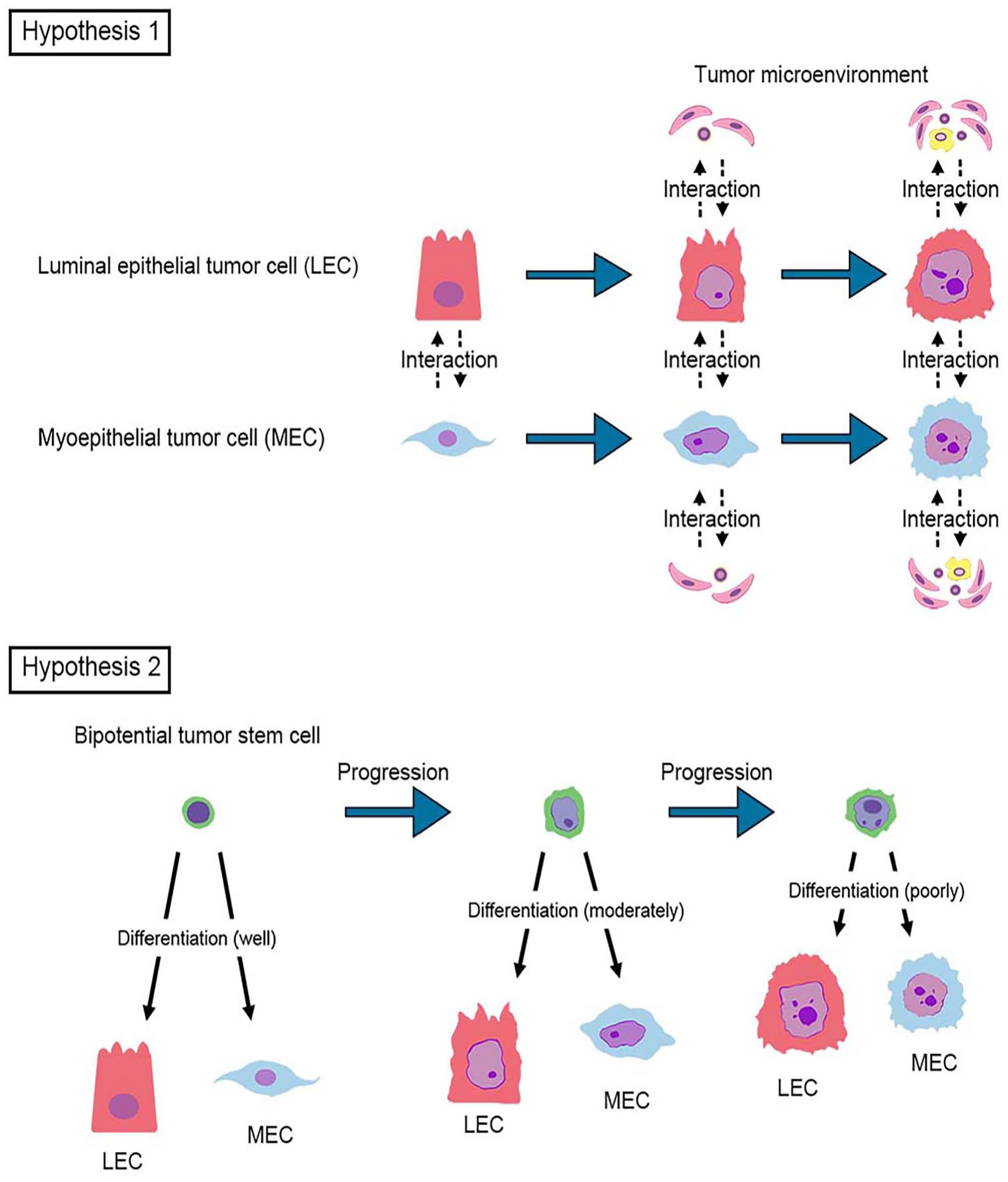
Two hypotheses for the histogenesis of carcinoma-and-malignant myoepithelioma.
Human salivary EMCs have low-grade malignancy, with recurrence in 30–40% of cases and regional lymph node metastasis in 10–20% of cases. 1 EMC with a high-grade component has a poor prognosis. 1 Canine mammary carcinoma-and-malignant myoepitheliomas have a higher frequency of metastasis compared with that of complex carcinomas (carcinomas arising in complex adenomas) but possibly lower than that of simple solid carcinomas.18,19 The biological behavior of the apocrine gland carcinoma-and-malignant myoepithelioma remains unknown. In our case, although we confirmed the absence of metastasis during the preoperative clinical examination and managed the tumor with complete excision, we were unable to confirm the outcome because the dog was lost to follow-up after postoperative day 16. Further studies are required to elucidate the metastatic potential and prognosis of apocrine carcinoma-and-malignant myoepithelioma.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231202529 – Supplemental material for Apocrine carcinoma-and-malignant myoepithelioma in a dog: a case of simultaneous malignant progression of both luminal epithelium and myoepithelium
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231202529 for Apocrine carcinoma-and-malignant myoepithelioma in a dog: a case of simultaneous malignant progression of both luminal epithelium and myoepithelium by Kana Matsumoto, Takuya Evan Kishimoto, Masami Yamamoto, Masaki Michishita, Kimimasa Takahashi and Hisashi Yoshimura in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Akihiko Yamada at Nishikyougoku Animal Hospital for providing us with clinical information.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared that they received no financial support for their research and/or authorship of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
