Abstract
The genus Neorickettsia includes obligate, intracellular bacteria responsible for diseases including Potomac horse fever caused by Neorickettsia risticii and salmon poisoning disease (SPD) caused by Neorickettsia helminthoeca. The Stellanchasmus falcatus (SF) agent is a member of this genus previously associated only with mild clinical signs in dogs. Between 2013 and 2016, 3 dogs in Washington State (USA) presented with disease suggestive of SPD, but N. helminthoeca was not detected by molecular techniques. Clinical signs included depression, anorexia, and diarrhea. Cytologic examination of aspirates supported a diagnosis of granulomatous lymphadenitis with organisms suggestive of Neorickettsia. Dogs either died or were humanely euthanized due to poor response to therapy. Necropsy findings included lymphadenomegaly and hepatomegaly. Histopathology identified granulomatous and lymphoplasmacytic splenitis, lymphadenitis, enteritis, and hepatitis with extensive necrosis. Neorickettsia DNA was detected using genus-specific primers and direct sequencing showed 100% sequence identity to the SF agent in all 3 dogs. This is the first clinicopathologic description of severe disease in dogs attributed to the SF agent. These findings may suggest the emergence of a novel neorickettsial disease in the Pacific Northwest.
Keywords
In the Pacific Northwest (USA), infection with Neorickettsia helminthoeca is a primary consideration in dogs with severe vomiting and diarrhea, with correspondent lymphadenomegaly and splenomegaly. 3 This infectious disease, also known as salmon poisoning disease (SPD), is especially considered if the dog has a history of fresh fish consumption. 3 Between 2013 and 2016, postmortem tissue samples or whole bodies were submitted to the Washington Animal Disease Diagnostic Laboratory (WADDL) for diagnostic investigation from 3 dogs with clinical signs suggestive of SPD. N. helminthoeca was not detected in any of the dogs, but the Stellanchasmus falcatus (SF) agent, a rarely diagnosed member of the Neorickettsia genus that has previously only been associated with mild clinical signs in dogs, was detected in tissues by polymerase chain reaction (PCR) and genomic sequencing.
In 2013, a 9-year-old spayed female Australian cattle dog (dog No. 1) was found dead at home 1 day after the owner noticed that the dog was not doing well. One day prior, a 3-year-old, castrated, male Labrador Retriever (dog No. 2) from the same household presented to a primary care clinic extremely depressed with projectile diarrhea and injected sclerae. Dog No. 2 was afebrile and all lymph nodes were palpably within normal limits, but the dog was azotemic, hyperphosphatemic, hyperbilirubenimic, and hyponatremic, with a leukocytosis due to neutrophilia and an increased hematocrit. The animal was treated with intravenous lactated Ringer’s solution and showed an initial positive clinical response but then continued to decline, becoming hypersthenuric with orange-brown urine, and died in hospital 4 days after presentation. Both dog Nos. 1 and 2 had access to a large acreage, including a free-running creek, and were reported to have recently consumed portions of a gut pile from a deer found dead of an unknown cause.
Dog No. 3, in 2016, was a 10-year-old spayed female Australian cattle dog that presented with a 1-week history of anorexia, regurgitation, and profuse hemorrhagic diarrhea. On physical exam, the dog was dehydrated, weak, and febrile (39.6°C); had generalized lymphadenomegaly; and had diarrheic staining of the perineum. An abdominal ultrasound examination revealed generalized lymphadenomegaly with the medial iliac node 3.7 cm in diameter, mild hepatomegaly, dilated and fluid filled intestines, and a heterogenous and swollen right limb of the pancreas. The dog was hospitalized on supportive care and was treated with doxycycline (5 mg/kg intravenously every 12 hours) due to a presumptive diagnosis of SPD. The dog demonstrated only minimal clinical improvement over a 2-day period and was humanely euthanized.
Antemortem fine-needle aspirates of enlarged mesenteric and submandibular lymph nodes from dog No. 3 were stained with Wright’s Giemsa. These aspirates contained a high percentage of macrophages (7%–9%, granulomatous lymphadenitis) with numerous individual and clumped basophilic cytoplasmic granules suggestive of Neorickettsia organisms (Fig. 1). The dog was negative for Anaplasma phagocytophilum, Ehrlichia canis, Borrelia burgdorferi, Dirofilaria immitis, Anaplasma platys, and Ehrlichia ewingii by Snap 6DX test (IDEXX, Westbrook, ME).
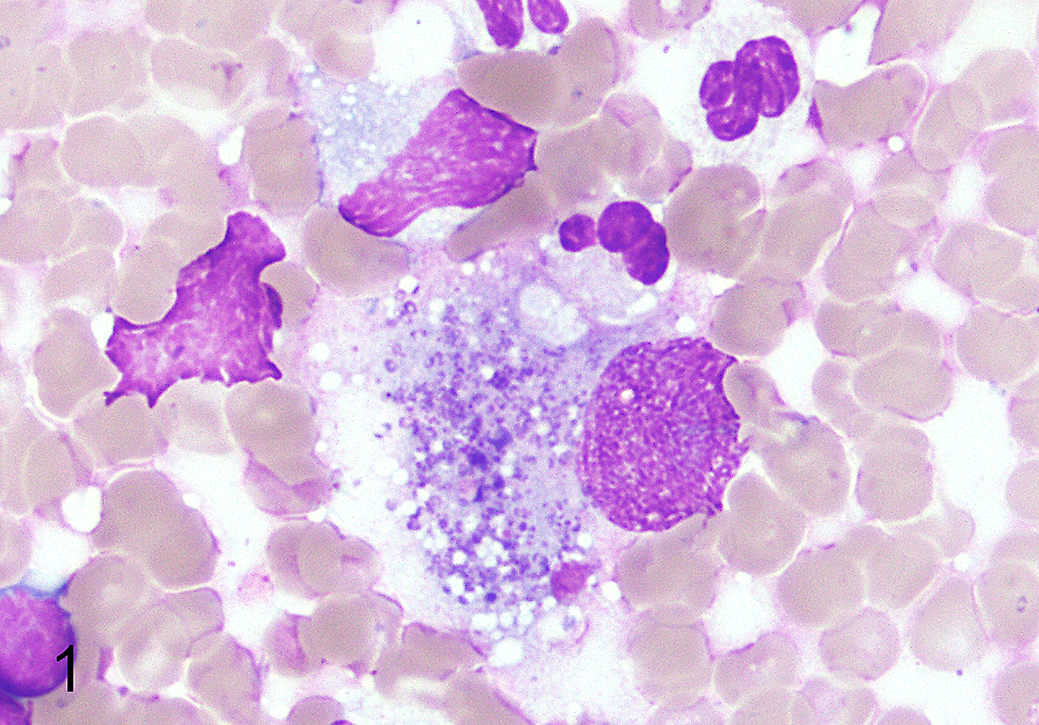
Stellanchasmus falcatus–associated disease, mesenteric lymph node, dog No. 3. Fine-needle aspirate cytology. There is an increased proportion of large macrophages with numerous intracytoplasmic Giemsa-positive granules consistent with neorickettsial organisms. Wright’s Giemsa stain.
At necropsy, dog No. 1 had large amounts of bile-stained fluid in the stomach, two 0.2- and 0.5-cm diameter splenic masses, red-brown pasty fluid in the colon, pale renal cortices, a mottled tan to brown liver, and enlarged tonsils. Gross findings were not reported for dog No. 2. Dog No. 3 had moderate red discoloration of the intestinal mucosa, most predominant in the jejunum; enlarged and reddened mesenteric lymph nodes; hepatomegaly (4.5% of body weight); and mottled, pale tan to red hepatic parenchyma. Tissue samples from all cases were fixed in neutral-buffered 10% formalin.
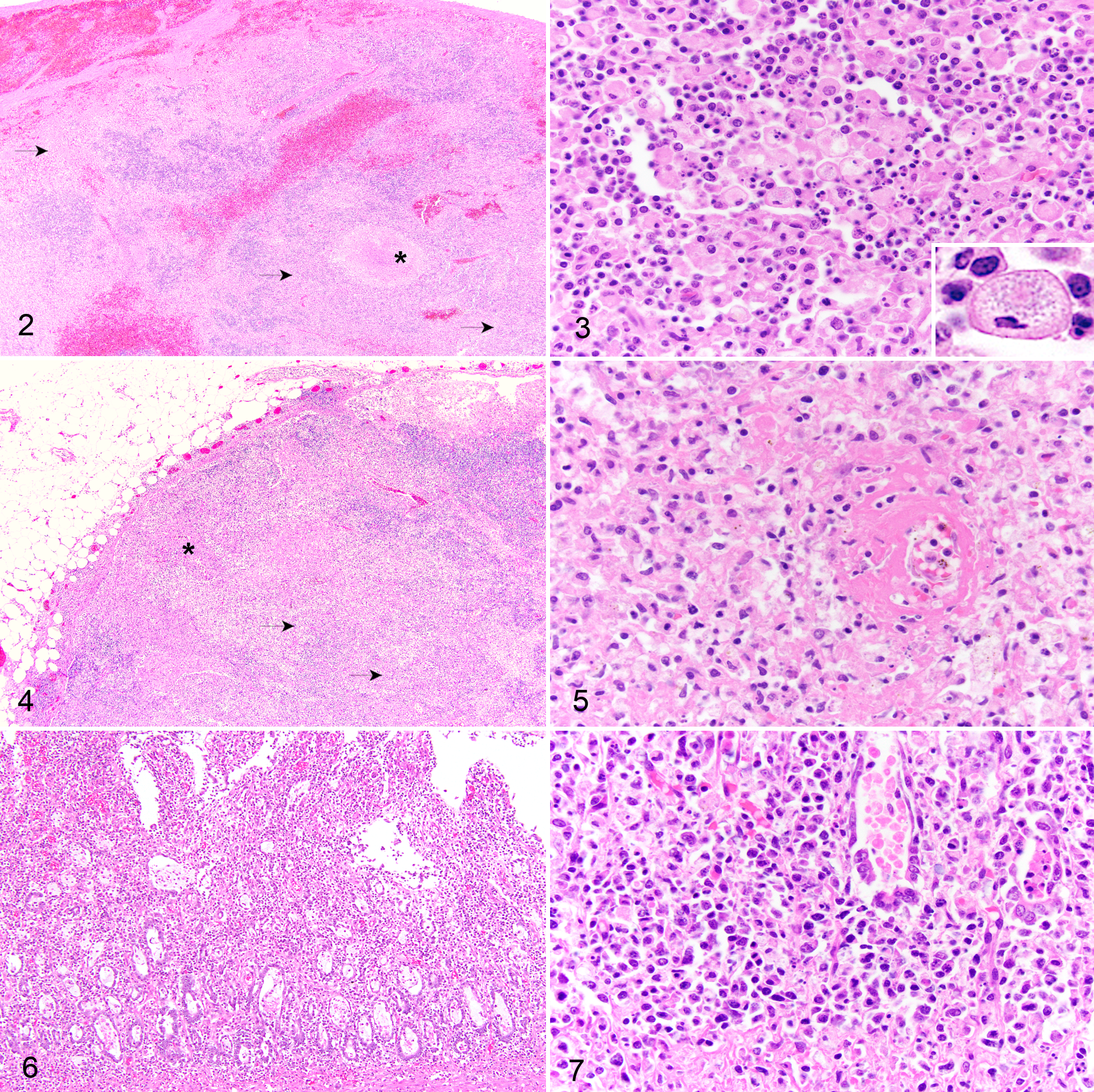
For histologic evaluation, samples were embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin, Giemsa, Macchiavello, and Brown and Hopp’s histochemical stains. The histopathological findings in the spleen, lymph nodes, and gastrointestinal tract were similar for the 3 cases. In all 3 cases, the architecture of the spleen and multiple lymph nodes, particularly the mesenteric lymph nodes, was expanded and effaced by granulomatous and necrotizing splenitis and lymphadenitis (Figs. 2 –4). The cytoplasm of macrophages commonly contained fine amphophilic cytoplasmic granules consistent with neorickettsial organisms (Fig. 3, inset). Neorickettsial organisms were not observed in macrophages stained with Giemsa, Macchiavello, or Brown and Hopp’s in any of the 3 cases. In less severely affected lymph nodes, the granulomatous inflammation was predominantly cortical, while in more severely affected lymph nodes, the inflammation expanded both the cortex and medulla regions. Large irregular areas of necrosis were present in the spleen and lymph nodes and comprised fibrin admixed with necrotic cellular debris. Fibrinoid necrosis of vessels was particularly prominent in the mesenteric lymph node of dog No. 1 (Fig. 5). All 3 dogs had significant inflammation in the gastrointestinal tracts consisting of severe histiocytic and lymphoplasmacytic enteritis with crypt necrosis and attenuation or erosion of villous epithelium (Figs. 6, 7). Other histopathologic findings included mild to severe multifocal random granulomatous hepatitis with occasional piecemeal hepatocellular degeneration/necrosis (dog Nos. 1–3). Additional findings included granulomatous tonsillitis (dog No. 1), acute renal infarction (dog No. 2), necrohemorrhagic pancreatitis, myocardial necrosis, chronic interstitial nephritis, and amyloidosis of renal glomeruli and coronary arteries (dog No. 3).
Frozen lung, spleen, kidney, and liver from dog No. 2 were plated on Columbia blood agar and EMB-Levine agar, quadrant streaked for isolation, and incubated at 37°C for 24 hours. Feces from dog No. 2 were placed in tetrathionate broth and iodine, incubated at 37°C for 24 hours, streaked to XLT4/BGN agar, and incubated for an additional 24 hours. Aerobic culture of lung, liver, kidney, spleen, and feces from dog No. 2 did not yield significant pathogen growth. Feces from dog No. 2 were suspended in sodium nitrate solution and microscopically evaluated after flotation on a coverslip. No Nanophyetus salmincola were observed, although the sample volume was considered insufficient for quantification (0.1 g total).
DNA was extracted from formalin-fixed, paraffin-embedded samples containing spleen (dog No. 1), intestine (dog No. 2), and lymph node and intestine (dog No. 3) (Qiagen, Valencia, CA), 7 and then pan-Neorickettsia primers ER2 and ER3 were used to amplify a portion of the 16S ribosomal RNA gene by PCR. 5 A band of approximately 510 base pairs corresponding to the expected amplicon size for Neorickettsia was visualized and directly sequenced in duplicate in both directions. 5 The sequencing data for dog Nos. 1 to 3 had 100% sequence identity (508/508 bp) with Neorickettsia sp. SF Agent, GenBank #U34280, isolated from metacercariae in mullet from Japan.
The SF agent was first isolated from metacercariae of SF flukes in gray mullet fish in Japan in 1962. 1 A dog was inoculated with the isolate and developed a mild fever after a 10-day incubation period, but no other clinical signs or macroscopic changes were detected at necropsy, and the pathogenicity of the bacteria was considered low. 2 Recently, it has been reported that the trematode N. salmincola, the vector of N. helminthoeca, also harbors the SF agent. 4 In this publication by Greiman et al., 4 they report that 2 dogs from Oregon were treated multiple times for SPD, suggesting either a lack of protective immunity after primary infection in these dogs or lack of cross-protection between the N. helminthoeca and the SF agent. In addition, in unpublished data, an earlier SF agent isolate from an Oregon trout was experimentally inoculated into a dog and resulted in severe clinical signs similar to those of SPD and what we report here (Yasuko Rikihisa lab, personal communication, 2013). It is of interest that dog No. 3 was treated with doxycycline, which is the antibiotic of choice for SPD, but did not show a positive response, and variability in responses to standard antibiotic therapies is critical information for clinicians assessing treatment responses. It remains undetermined whether the poor response after the initiation of doxycycline therapy was due to antimicrobial resistance or the severe nature of the lesions.
These 3 cases show that the clinical signs and lesions are similar for disease associated with the SF agent as for that caused by N. helminthoeca. 3,6 These 2 organisms can be differentiated by PCR and sequencing. Because only 3 dogs were investigated, it remains undetermined whether those infected with the SF agent have a different prognosis than those infected with N. helminthoeca, especially given the short duration of antimicrobial treatment reported in this case. To the authors’ knowledge, this is the first report of severe disease caused by the SF agent in dogs and the first report of naturally acquired SF disease in dogs in the United States.
Footnotes
Acknowledgements
We thank the Washington Animal Disease Diagnostic Laboratory personnel; histopathology, bacteriology, parasitology, and molecular diagnostics divisions; and Dr. Yasuko Rikhisa of the Ohio State University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
