Abstract
An outbreak of canine distemper virus in a private zoo in eastern Tennessee in July 2016 led to fatal clinical disease in 5 adult, wild-caught Linnaeus’s 2-toed sloths (Choloepus didactylus). Clinical signs included hyporexia, lethargy, mucopurulent nasal discharge, and oral and facial ulcers. At necropsy, affected animals had crusts and ulcers on the lips, nose, tongue, and oral cavity. Microscopically, all sloths had widespread, random, hepatic necrosis; lymphoid depletion; and bronchointerstitial pneumonia. The central nervous system did not contain gross or histopathologic lesions in any of the 5 sloths, although immunoreactivity for viral antigen was present within vessel walls. Epithelial cells and histiocytes within numerous organs contained intranuclear and intracytoplasmic inclusions and occasional syncytial cells. Canine distemper virus was confirmed with immunohistochemistry and virus isolation. Viral sequencing identified the novel American-4 strain prevalent in eastern Tennessee wildlife. This is the first pathologic characterization of canine distemper virus infection in sloths (family Choloepodidae, order Pilosa) and emphasizes the significant morbidity and mortality in this species.
Canine distemper virus (CDV) is a single-stranded RNA morbillivirus in the family Paramyxoviridae. CDV is a cause of morbidity and mortality in domestic dogs, as well as in numerous nondomestic species. Natural infection has been documented within the orders Carnivora, Rodentia, Artiodactyla, Primates, and Proboscidea. 7,12
Beginning in 2012, outbreaks of canine distemper virus have occurred in fully vaccinated adult dogs. A novel strain of CDV is prevalent in the southeastern United States and causes outbreaks in vaccinated domestic dogs and in eastern Tennessee wildlife. 14,15 Two reports of CDV in superorder Xenarthra (anteaters, armadillos, and sloths) have been published. Both reports are in anteaters (order Pilosa, family Myrmecophagidae). 4,9 To our knowledge, the outbreak reported here in 5 Linnaeus’s 2-toed sloths (Choloepus didactylus) represents the first pathologic description of canine distemper virus in sloths and expands the distribution of CDV to the family Choloepodidae.
Five adult, wild-caught, Linnaeus’s 2-toed sloths (C. didactylus) from a private zoo were submitted to the University of Tennessee in July 2016 for gross and histologic examination. They were in quarantine for 15 months with 3 conspecifics, who survived the outbreak. Necropsies were performed using a standardized protocol and gross lesions were recorded. Representative sections of tissues were fixed in neutral-buffered 10% formalin for at least 24 hours, trimmed, embedded in paraffin, sectioned at 5-µm thickness, routinely processed, and stained with hematoxylin and eosin.
Signalment, gross, and histopathologic findings are summarized in Table 1. All adult sloths died after a 2- to 3-week history of hyporexia, lethargy, and oronasal discharge. Multifocal crusts and ulcers were the most common gross finding (Fig. 1). Severe, multifocal ulcers of the tongue with overlying white to tan plaques and multifocal ulcers of the soft palate, gingiva, and pharynx were present in case Nos. 1 to 3 (Fig. 2). Case Nos. 4 and 5 had no apparent gross lesions, but the carcasses had been frozen and thawed prior to necropsy.
Signalment and Pathologic Findings in 2-Toed Sloths With Canine Distemper.

Abbreviation: GI, gastrointestinal.

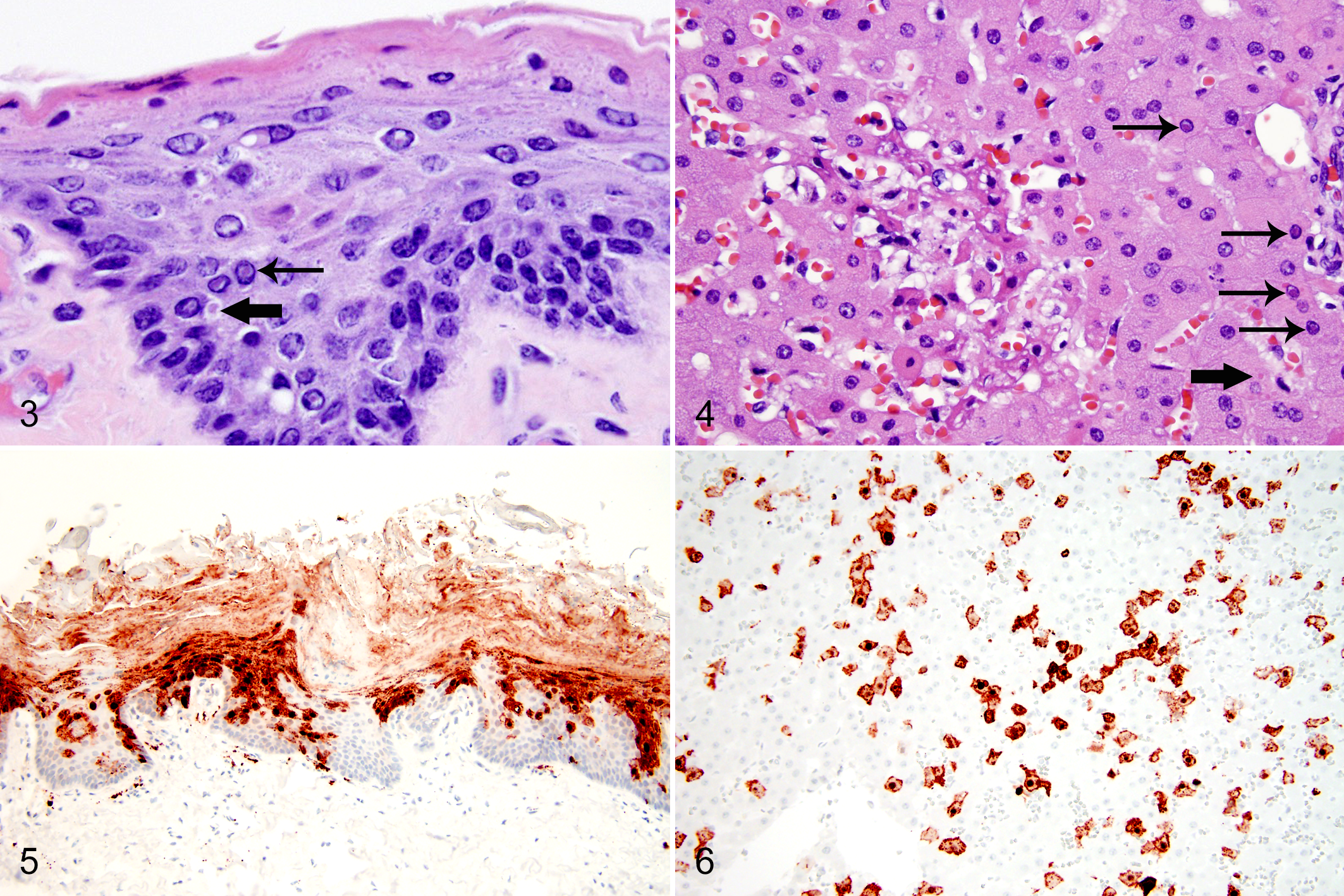
Microscopic lesions were present in each case. In all cases, the tongue had multifocal extensive erosions and ulcers, keratinized crusts with neutrophils, and numerous discrete eosinophilic intranuclear and intracytoplasmic inclusions within epithelial cells. Erosions or ulcers and eosinophilic intranuclear and intracytoplasmic inclusions were within the epithelium of the nares, lips (Fig. 3), trachea, esophagus, glandular stomach, duodenum, and urinary bladder. Syncytial cells were within the epithelium of the tongue and lip.
Neutrophilic, necrotizing, bronchointerstitial pneumonia was also present in all cases. Alveoli and terminal bronchioles often contained inflammatory cells, and alveolar septa were moderately thickened by mononuclear cells and fibrin. Numerous eosinophilic intranuclear and fewer eosinophilic intracytoplasmic inclusions were within alveolar macrophages and type II pneumocytes. Few syncytial cells were within bronchiolar epithelium.
Multifocal splenic necrosis, lymphoid depletion, and intracytoplasmic and intranuclear inclusions within histiocytes occurred in all cases. All sloths also had widespread, random, hepatic necrosis with numerous hepatocytes containing intranuclear and intracytoplasmic inclusions (Fig. 4). Inclusions were not present within biliary epithelium.
Immunohistochemistry for canine distemper virus was performed on paraffin-embedded sections of liver and tongue from case No. 1, tongue and lip from case No. 5, and brain from case Nos. 1, 4, and 5. Brain was not available for case Nos. 2 and 3. Antigen was retrieved using a pressure cooker and Target Retrieval Solution (DAKO Corp., Carpinteria, CA). Endogenous peroxidase activity was blocked with 3% hydrogen peroxide, and nonspecific binding sites were blocked by 10% goat serum. The slides were incubated with mouse monoclonal anti-CDV nucleoprotein antibody (VMRD, Pullman, WA) at a dilution of 1:900. A secondary horseradish peroxidase (HRP)–conjugated goat anti-mouse IgG antibody (DAKO Corp.) was applied, followed by 2% canine serum. A positive antigen-antibody reaction was observed by labeling with 3-amino-9-ethylcarbazole (DAKO Corp.). Approximately 90% of epithelial cells in the tongue (2/2 tested cases) (Fig. 5) and lip (1/1 tested cases) and 80% of hepatocytes (1/1 tested cases) (Fig. 6) had strong positive labeling for mouse monoclonal anti-CDV nucleoprotein antibody (VMRD). CDV antigen was also present within meningeal vessel walls, choroid plexus, and ependymal cells of the brain in case Nos. 1, 4, and 5. CDV antigen was not present within the neuroparenchyma. Sections of tissue without the primary antibody had no positive labeling.
Canine distemper virus was isolated from an ulcerated area of soft palate in case No. 1. A tissue suspension was inoculated onto a monolayer of Vero cells expressing the canine CD150 receptor. 16 Cells were cultured in Eagle’s minimum essential medium with 10% fetal bovine serum at 37°C. Cells were monitored at 24-hour intervals for the development of a cytopathic effect.
Isolated virus was shipped to the molecular laboratory at the Veterinary Diagnostic and Investigational Lab, University of Georgia. The viral nucleic acid was extracted using a commercial kit (DNeasy Blood and tissue kit; Qiagen, Valencia, CA) according to the manufacturer’s instructions. The viral RNA was converted to complementary DNA (cDNA) and sequenced by targeted whole-genome sequencing as described elsewhere. 1
Multinucleated cells typical of CDV were noted in the cell culture and the agent identified as CDV by fluorescent antibody staining (VMRD).
Sequences from genomic regions used for phylogenetic study, including the M-F region and H gene, 14,15 were compared with known sequences in GenBank. The H gene sequence was 98% homologous to the America-4 lineage previously sequenced from dogs (GenBank-KJ747372) and foxes (GenBank-KJ747372) in Tennessee. 14,15 The H gene sequence was deposited in GenBank (MK577461). The M-F region has been described elsewhere and was not deposited in GenBank. 19
Three of 4 adult kinkajous (Potos flavus) housed in the same building died after developing similar clinical signs as the affected sloths; they had similar gross and histopathologic lesions and canine distemper virus was confirmed via polymerase chain reaction (PCR) in 1 animal.
To our knowledge, this outbreak represents the first gross and histopathologic description of canine distemper virus in Linnaeus’s 2-toed sloths (C. didactylus). Canine distemper virus in this species was characterized clinically by high morbidity and high mortality; 5 of 8 within the collection died.
Marked hepatic tropism was a striking feature in this outbreak. Hepatic necrosis and numerous viral inclusions were within hepatocytes in all cases. Hepatocellular necrosis has not been associated with canine distemper virus infection in domestic dogs or nondomestic carnivores. 2,3,5,11 Rare reports of hepatic lesions exist in the literature, which may not reflect the actual prevalence of CDV-associated liver changes. 10 Intranuclear inclusions within hepatocytes have also been described in a captive adult Hoffman’s 2-toed sloth (Choloepus hoffmanni), although a virus could not be isolated. 7
Clinical neurologic disease and central nervous system lesions, specifically demyelinating leukoencephalomyelitis, are common in domestic dogs. 3,8,11 Central nervous system disease is also common in nondomestic carnivores but may be predominately inflammatory or may have a more patchy distribution compared to domestic dogs. 5,18 There were no gross or histologic lesions in the central nervous system of any of the sloths in this outbreak. However, CDV antigen was present within meningeal vessels, choroid plexus, and ependyma. Systemic disease was severe in this group of sloths, and it is possible that the sloths died before developing neurologic disease.
To the authors’ knowledge, there have been only 2 previous reports of CDV infection in other members of the superorder Xenarthra, both in anteater species within order Pilosa, family Myrmecophagidae. A family group of 3 giant anteaters (Myrmecophaga tridactyla) under professional care developed fatal CDV infection. 4 The 2 adults had interstitial pneumonia with syncytia and intranuclear and intracytoplasmic viral inclusions, and the juvenile had ulceration of the tongue and “hard pad” disease. Intranuclear and intracytoplasmic inclusions were in epithelial cells of multiple organs. Similar to this group of sloths, encephalitis was not present in any of the 3 cases. CDV was confirmed via immunohistochemistry, PCR, and fluorescent antibody testing. An approximately 1-year-old, wild southern tamandua (Tamandua tetradactyla) in Brazil developed progressive neurologic disease. 9 Neuronal necrosis with intranuclear inclusions and Purkinje cell loss in the cerebellum were described, and CDV was confirmed via immunohistochemistry and viral sequencing. Hepatic tropism was not described as a feature in any of the affected animals; however, liver was not examined histologically in the southern tamandua, and it is unclear if any lesions were present in the giant anteaters.
Wildlife species are frequently implicated as the source of outbreaks of canine distemper virus in domestic and nondomestic animals, and they often serve as intermediate hosts of CDV. Alternatively, domestic dogs can be a source of exposure of CDV to wildlife species. Phylogenetic analysis has determined that domestic species are important reservoirs of CDV throughout the world and have contributed to worldwide spread. 13 Increased contact between domestic dogs and wildlife occurs as a result of urbanization, as well as the increase in human and domestic dog populations. 6 Canine distemper virus is prevalent in wild raccoons (Procyon lotor) and gray foxes (Urocyon cinereoargenteus) in eastern Tennessee. 14 Increased numbers of CDV cases, including cases in vaccinated, adult domestic dogs at the University of Tennessee, led to the characterization of a novel CDV strain, named American-4. 15 American-4 was found in 77% of CDV-positive raccoons and gray foxes in Tennessee. 14,15
Virus isolation and sequencing of tissue from case No. 1 determined that this sloth was infected with the novel American-4 strain. Although housed in individual indoor enclosures, transmission from organic material or other fomites may have occurred following wildlife exposure. The CDV strain was not sequenced from the affected kinkajous. The 3 kinkajous died soon after the sloths developed clinical signs and were housed in the same building; infection with the American-4 strain is likely. Like the sloths, none of the kinkajous had central nervous system lesions, and 1 of 3 had hepatic necrosis and intranuclear inclusions in hepatocytes.
The clinical details of this outbreak have been published previously. 17
Natural canine distemper virus infection in this group of zoo-housed sloths expands the known host range of this promiscuous virus. In this outbreak, canine distemper virus caused high morbidity and mortality in Linnaeus’s 2-toed sloths, and the gross and microscopic lesions differ from those previously described in other species. The cause of the hepatic tropism in sloths is unclear but may be related to either host species or CDV strain variability.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
