Abstract
Cervidpoxvirus is one of the more recently designated genera within the subfamily Chordopoxvirinae, with Deerpox virus (DPV) as the only recognized species to date. In this study, the authors describe spontaneous disease and infection in the North American moose (Alces americanus) by a novel Cervidpoxvirus, here named Moosepox virus (MPV). Three 4-month-old moose calves developed a multifocal subacute-to-chronic, necrotizing, suppurative-to-granulomatous dermatitis that affected the face and the extremities. Ultrastructurally, all stages of MPV morphogenesis—that is, crescents, spherical immature particles, mature particles, and enveloped mature virus—were observed in skin tissue. In vitro infection with MPV confirmed that its morphogenesis was similar to that of the prototype vaccinia virus. The entire coding region, including 170 putative genes of this MPV, was sequenced and annotated. The sequence length was 164,258 bp with 98.5% nucleotide identity with DPV (strain W-1170-84) based on the whole genome. The genome of the study virus was distinct from that of the reference strain (W-1170-84) in certain genes, including the CD30-like protein (83.9% nucleotide, 81.6% amino acid), the endothelin precursor (73.2% nucleotide including some indels, 51.4% amino acid), and major histocompatibility class (MHC) class I–like protein (81.0% nucleotide, 68.2% amino acid). This study provides biological characterization of a new Cervidpoxvirus attained through in vivo and in vitro ultrastructural analyses. It also demonstrates the importance of whole-genome sequencing in the molecular characterization of poxviruses identified in taxonomically related hosts.
Keywords
Cervidpoxvirus is a recently designated genus within the subfamily Chordopoxvirinae of the family Poxviridae. 8 The only member of this new genus is Deerpox virus (DPV). Disease caused by DPV is variable in distribution and severity, but it is generally characterized by keratoconjunctivitis and/or mucocutaneous lesions consisting of ulcers, plaques, or crust-covered areas of alopecia, often around the eyes, lips, muzzle, and/or extremities. 5,7,17,30 DPV was first isolated from 2 diseased wild mule deer (Odocoileus hemionus hemionus) fawns in Wyoming in 1983 and 1984. Sequencing of the isolates resulted in the identification and classification of the new poxvirus. 1,30
For some time, the host range of DPV appeared to have been limited, as it was described only in mule deer, black-tailed deer (O. h. columbianus), white-tailed deer (O. virginianus), and reindeer (Rangifer tarandus tarandus). The latter was originally considered an orthopox-like viral infection when first recognized in 1980. 4,5,17 A serosurvey of 5 cervid species in Oregon substantiated these observations, revealing high serological prevalence of DPV in Odocoileus spp. and no detectable antibody in elk (Cervus elaphus). 13
There are 2 pathogenic DPV strains, W-848-83 (W83) and W-1170-84 (W84), whose genomes are 166 and 170 kbp, respectively. The DPV genomes encode several genes that may be involved in regulation of immune responses. These genes are thought to modulate pathogenesis and are predicted to encode proteins similar to cellular endothelins (ET), interleukin 1 receptor antagonists (IL-1Ra), C-type lectin-like receptors (CTLR), and major histocompatibility complex–I (MHC-I). One of the DPV strains (strain W-848-83) encodes an open reading frame (ORF) similar to transforming growth factor β1 (TGF-β1). Homologs of TGF-β1 are not found in other mammalian Chordopoxvirinae viruses, although distantly related homologs are encoded by avipoxviruses. 1 Despite significant progress in the understanding of the DPV genome, the morphogenesis of Cervidpoxvirus has not been described. In fact, this is true for a majority of the members of Chordopoxvirinae.
The Chordopoxvirinae subfamily contains 11 genera, which collectively infect a wide range of vertebrate hosts: Orthopoxvirus, Parapoxvirus, Molluscipoxvirus, Crocodylipoxvirus, Avipoxvirus, Yatapoxvirus, Capripoxvirus, Centapoxvirus, Cervidpoxvirus, Suipoxvirus, and Leporipoxvirus. 12,25 With morbidity and mortality rates approaching 100% in naïve animals, capripoxviruses are the most serious poxvirus diseases of production animals and the most important differential diagnoses. Outbreaks in free regions are immediately notifiable diseases according to the World Organization for Animal Health guidelines. 3
There is increasing evidence on the emergence of novel poxviruses and the identification of animal hosts in the epidemiology of these viruses. 7 With some exceptions, a significant feature of poxviruses is their ability to enter a variety of animal host cells by mechanisms that do not require species-specific receptors. 19 However, permissive infection within a host and successful replication are a function of the virus’ ability to evade the host immune response. 12 As a result, poxviruses demonstrate great variation in their host range, even among closely related species. 16 Recently, our team described DPV infection in a captive-managed goitered gazelle (Gazella subgutturosa), representing the first report of spontaneous infection and clinical disease in a species outside of the Cervidae family. 7
The wild moose population in Minnesota is rapidly declining. In a recent morbidity and mortality study, diseases caused by environmental changes, parasitism, and anthropogenic trauma were responsible for losses in adult moose herds of northern Minnesota. 31 The aforementioned study identified the potential role of P. tenuis as a significant mortality factor. Thus, efforts have been intensified to detect disease that may represent a risk to the current population. In this study, we describe spontaneous infection and disease by a novel Cervidpoxvirus in the North American moose (Alces americanus), its full genome sequence, as well as the in vivo and in vitro morphogenesis of this new Cervidpoxvirus.
Materials and Methods
Clinical History and Diagnostic Sampling
The Minnesota Zoological Garden is a 485-acre facility located in Apple Valley (44° 46’ 4.19” N –93° 11’ 33.60” W), a suburb of Minneapolis and St. Paul, Minnesota. It is bordered by residential neighborhoods, as well as certain county parks. From 5 to 20 May 2014, six wild moose calves, aged less than 1 week, were acquired by the zoo following abandonment by their mothers in northern Minnesota. The calves were quarantined for 8 weeks prior to moving to their permanent holding areas, during which time they were isolated in pairs, observed for disease, and managed separately from the rest of the zoo collection with biosecurity protocols in place to prevent disease introduction. After observing no evidence of disease in quarantine, pairs of calves were kept together and transferred to the zoo’s moose holding and exhibit on 6 July 2014 at approximately 7–9 weeks of age. Calf pairs rotated between outdoor holding pens and a public exhibit they shared with a 5-year-old, captive-born, female moose. Calf pairs were housed in adjacent pens but did not have direct contact with counterpart pairs or the adult female at any time, although space and facility equipment were shared resources in this area. Personnel providing care for the calves managed other zoo species, including Bactrian camels (Camelus bactrianus), woodland caribou (Rangifer tarandus caribou), Sichuan takin (Budorcas taxicolor tibetana), pronghorn antelope (Antilocapra americana), and goitered gazelle (Gazella subgutturosa). All of these species were housed and managed in separate areas within the zoo. Personnel routinely observed animals daily and reported any abnormal observations to the veterinary staff for follow-up.
In September 2014, when the moose calves were approximately 4 months old, 2 female calves and 1 male calf developed proliferative skin lesions on their extremities and face. An initial sample of crust material was collected from the leg of 1 affected calf (calf 1) for initial screening by transmission electron microscopy (TEM) for negative contrast (negative staining) of viral particles. Following this, 2 additional animals (calves 2 and 5) were sampled by biopsy of lesions of the periocular region, as well as from an extremity. Given the need to anesthetize the animals for sample acquisition, the calves with the most severe and numerous lesions were selected for biopsy and diagnostic workup. Diagnostic workup included a complete physical examination, as well as blood sampling for hematology and serum biochemistry profiles by Marshfield Laboratory (Marshfield, WI, USA). Hematology and biochemical values were compared with species-specific reference ranges of clinically healthy moose aggregated through the Zoological Information Management Systems database. 27 A single 6-mm-diameter skin biopsy was obtained from eyelid and fetlock from both animals (calves 2 and 5); each biopsy was further divided into fresh and formalin-fixed specimens, which were submitted for routine diagnostic workup at the Minnesota Veterinary Diagnostic Laboratory. Further, the fresh skin tissue was divided, and a sample of eyelid of calf 2 and front fetlock skin of calf 5 were submitted to the Plum Island Foreign Animal Disease Diagnostic Laboratory to rule out reportable animal diseases including vesicular stomatitis virus, Capripoxvirus, and Parapoxvirus. 7 Virus isolation in cell culture, TEM, and polymerase chain reaction (PCR) tests were performed as described by Bracht et al 7 (2013). Aside from the diagnostic testing described herein of the biopsy material, no other samples were collected for pathogen detection or serological survey. Medical management included broad-spectrum parenteral (calf 5) or oral and topical ophthalmic (calf 2) antibiotics, and biosecurity protocols to reduce potential transmission were put in place around moose holding, including foot baths and elimination of equipment sharing. Other animals did not show similar lesions and were not sampled as part of this investigation.
Histopathology
Half of the 6-mm-diameter formalin-fixed skin biopsies from the eyelid and from the rear and front fetlocks of calves 2 and 5 were embedded in paraffin, cut in series, and stained with hematoxylin and eosin. Periodic acid-Schiff and Grocott’s methenamine silver (Ventana Medical System, Tucson, AZ, USA) were performed for detection of fungi. Twort’s Gram stain (Newcomer Supply, Middleton, WI, USA) for detection of bacteria and acid fast stain (American MasterTech, Lodi, CA, USA) were performed for detection of mycobacteria.
Virus Culture
Skin tissue (∼1 g) with crusty lesion of the eyelid of calf 2 and front fetlock skin of calf 5 were homogenized in 5 mL Eagle’s minimum essential medium supplemented with 2% donor horse serum (Life Technologies Corp, Grand Island, NY, USA), 50 μg/mL gentamicin (Mediatech, Corning, NY, USA), 150 μg/mL neomycin sulfate (Sigma, Inc, St Louis, MO, USA), 1.5 μg/mL fungizone (Sigma, Inc), and 455 μg/mL streptomycin. After adding media, skin tissue was subjected to 3 freeze-thaw cycles (−80oC/25 oC), followed by homogenization and centrifugation at 2500 × g for 15 minutes at 4 oC. The clear suspension (200 µL) was inoculated in duplicate wells of a 24-well plate containing monolayers of Vero CCL-81 cells (American Type Culture Collection, Manassas, VA, USA). Inoculated cells were incubated at 37 oC for 45 minutes for adsorption. Maintenance media contained 4% DHS (Gibco, Life Technologies Corp) and the above-mentioned antibiotics. Cells were incubated at 37 oC under 5% CO2 for 7 days. On the seventh day, cells were subjected to 3 freeze-thaw cycles (−80oC/25 oC), followed by inoculation for second passage. Cytopathic effects (CPE) were observed in Vero cells in the second passage of the calf 2 sample. Cultures presenting CPE were examined by TEM. Negative contrast was performed on the culture supernatant. For ultrastructure examination, infected cells were fixed by immersion scraped and pelletized. Complete genome sequencing was performed to characterize the isolated poxvirus.
Transmission Electron Microscopy
To study the morphology and morphogenesis of this poxvirus, TEM was performed. Negative contrast was done on fresh samples from calves 1, 2, and 5, as well as cell culture isolate from calf 5. Ultrastructure on fixed tissue was performed on eyelid and fetlock skin from both calves 2 and 5, as well as infected cell culture from calf 2 . For negative contrast, 0.15 x 0.3 x 0.8-cm frozen skin and/or scab was thawed, ground, and sonicated. This sample was placed in a 20-mL tube, and double-distilled water was added to 15 mL final volume. After 10 minutes at room temperature, 1 mL of the tissue suspension or the infected cell culture supernatant was filtered with 0.45 micron syringe filters (Fisherbrand, Fisher Scientific, Dublin, Ireland). Fifty µL filtered suspension or supernatant was transferred to airfuge tubes (Beckman Coulter, Brea, CA, USA) with formvar-coated 200 mesh copper grids (Electron Microscopy Sciences, Hatfield, PA, USA) and centrifuged at 30 PSI (Beckman Coulter) for 10 minutes. Grids were washed and stained with 1% phosphotungstic acid (Electron Microscopy Sciences) for 1 minute. Ultrastructure was also performed on infected cells and formalin-fixed skin. Pellets of infected cells were fixed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer and postfixed with 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. (All reagents were from Electron Microscopy Sciences.) Formalin-fixed skin tissues (1-mm3 fragments) were postfixed with 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer followed by a second postfixation with 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. After 3 washes in distilled water, samples were dehydrated using a 25%–100% ethyl alcohol gradient. Samples were then infiltrated with 2:1 ethanol: EMbed 812 resin (Electron Microscopy Sciences) for 1 hour and subsequently transferred to a 1:2 ethanol: EMbed 812 resin mixture for 1 hour. Cells and tissues were further infiltrated with 100% resin and were embedded and incubated at 58°C for 24 hours to polymerize the resin. Embedded samples were trimmed and sectioned on a Leica UC6 ultramicrotome (Leica Microsystems, Vienna, Austria). Thin sections (60–70 nm) were obtained and collected on a 200 mesh copper grid (Electron Microscopy Sciences) using a perfect loop. Grids were contrasted with 5% uranyl acetate for 20 minutes and Santos’ lead citrate for 6 minutes. These samples were visualized using a JEOL 1400 transmission electron microscope (JEOL LTD, Tokyo, Japan). Images were obtained using an AMT Capture Engine Version 7.00 camera and software (Advanced Microscopy Techniques Corp, Woburn, MA, USA). Image analysis was carried out using ImageJ (NIHR public domain).
Polymerase Chain Reaction
Total DNA was extracted from 30 μL of tissue homogenate from calves 2 and 5, as well as CPE-positive cell culture supernatant from calf 2 using the MagMAX-96 Total DNA Isolation Kit (Invitrogen by ThermoFisher, Waltham, MA, USA). The DNA was eluted in 50 μL of elution buffer. Real-time PCR was performed on original tissue homogenate samples for the detection of the poly(A) polymerase gene of capripox viruses and the major envelope protein (B2 L) gene of parapoxviruses, as previously described. 7,22 Conventional PCR was used to test for multiple poxvirus genera, including capripox, suipox, and cervidpox viruses, by targeting sequences within the DNA polymerase (DNApol) and DNA topoisomerase (DNAtopo) genes. 7 Amplified DNA fragments were visualized by agarose gel electrophoresis of 8 μL of the PCR product in 2.0% agarose E-Gels (Invitrogen by ThermoFisher), viewed under ultraviolet light, and photographed using a gel documentation system (Bio-Rad Laboratories, Hercules, CA, USA). 7 In addition, DNA was extracted from 80 µm of formalin-fixed paraffin-embedded sections (8 sections of 10 μm) of skin lesions of calf 5 using the modified formalin-fixed paraffin-embedded DNA extraction protocol provided with the Qiagen DNeasy blood and tissue kit (Qiagen, Hilden, Germany). The extracted DNA was diluted with 50 µL elution buffer and stored at –80 oC until used for PCR. Previously published primers A21LF2 5’- TCC TCG AAC ACG TAA GTG GAT-3’and A21LR2 5’- TGC AGT TGC CCA ATC AAT TA-3’ targeting the conserved region of the A21 gene were used to detect the presence of MPV. 17 PCR was performed in a Mastercycler (Eppendorf, Hamburg, Germany G) using HotStarTaq Master Mix kit (Qiagen). The reaction mix and PCR cycling conditions were the same as described previously. 17 The extracted DNA from formalin-fixed paraffin-embedded tissue of unrelated species was used as a negative control. The PCR-amplified products were visualized on 1.2% agarose gel to recognize the MPV amplicons of the expected size (349 bp).
Next-Generation Sequencing
To obtain the complete viral genome, clarified supernatant of the infected cell culture suspension from moose calf 2 was processed for next-generation sequencing (NGS). Briefly, semipurified viral DNA was extracted using the NucleoSpin Virus kit (Clonetech, Mountain View, CA, USA). Extracted DNA was used for library preparation and sequencing on an Illumina MiSeq machine at the University of Minnesota Genomic Center. The DNA was quantified using fluorimetry (PicoGreen assay) and further processed after passing quality control. The library was prepared using the Nextera kit, followed by sequencing using 500-cycle (2 x 250 bp paired-end) MiSeq Reagent Kits v2 (Illumina, San Diego, CA, USA).
Bioinformatic and Genomic Analysis
Bioinformatic analysis on NGS data and genomic analysis on full viral sequence were performed according to previous studies. 10 Sequence data was analyzed by a customized NGS pipeline as previously described. 17,20,21 Specifically, human- and bacterial-subtracted reads were trimmed for quality and de novo assembled into contigs using SPAdes. In addition, trimmed reads were also mapped using the DPV genome as reference. An NGS coverage map was created using previously described parameters. 10 Once a draft genome was obtained, manual inspection and gene annotation were performed using Geneious R8. For comparative genomics, full MPV genome and other DPV genomes were aligned using MAFFT. 14 Maximum likelihood (ML) trees were generated from full genome alignment using RAxML. 28 Maximum likelihood trees were run with 100 bootstrap replications with a gamma distribution for rates over sites. Bayesian inference trees and neighbor-joining trees were also performed for validation (data not shown). Midpoint rooting was conducted using FigTree.
Results
Clinical Presentation, Gross and Histological Lesions
Two female moose calves (calves 2 and 5) and 1 male moose calf (calf 1), age 4 months, developed proliferative skin lesions on their extremities and face, which persisted for several weeks before spontaneously resolving (Supplemental Table S1). An additional female calf, age 4 months, had a small, circular surface abrasion near the left nares, which did not develop into a proliferative lesion. At the time the skin lesions occurred, one calf (calf 2) had lower weight gain compared to other calves, but no significant abnormalities in behavior or feeding were reported by animal care staff. Preliminary screening of sample crust material from calf 1 by TEM revealed numerous orthopox-like viral particles; however, this sample was insufficient for further pathogen identification. Thus, 2 other calves (calves 2 and 5) demonstrating similar lesions were immobilized for biopsy collection, physical examination, and further diagnostic workup. On physical examination, lesions were well-demarcated areas of raised plaques to nodules (approximately 0.5 to 1.5 cm) covered by thick crusts, some of which were associated with purulent discharge. Lesions of varying number and severity affected the face, primarily around the medial and lateral canthus of the eyes (Fig. 1), and fetlocks of all 4 legs in the 3 calves. All other physical examination findings were unremarkable. The serum biochemistry profile of calf 2 revealed a mild hypernatremia (152 mmol/L; reference range for moose, 132–148 mmol/L) and a mild hypoglycemia (37 mg/dL; reference range for moose, 43–200 mg/dL); otherwise, all hematologic and serum biochemistry values were within the normal range for clinically healthy moose. 27 Overall, the clinical disease process was determined by veterinary staff to be mild and limited to the skin; therefore, further sampling and disease screening were not pursued. Although regular examinations for disease monitoring did not continue following the sampling event, lesions were no longer observed during a visual examination of the calves at 1 month after the sampling event.
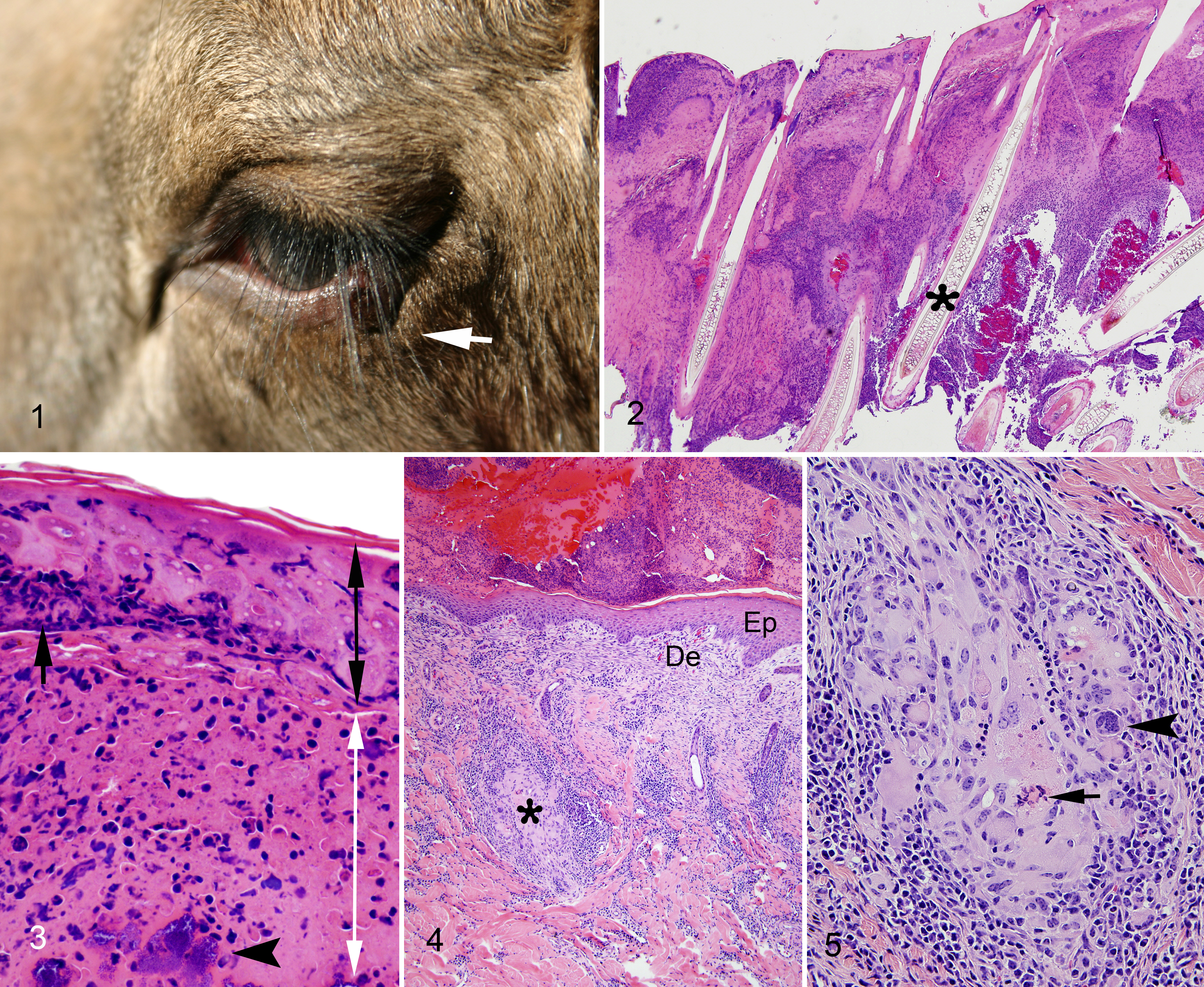
The skin of the canthus of the eyes and the legs included subacute to chronic lesions. The subacute lesion obtained from the eyelid of calf 5 was characterized by necrosis of the epidermal hair shaft and adnexal glands. Intracytoplasmic inclusions in epithelial cells were not distinct. The dermis was severely infiltrated by intact and degenerated neutrophils and eosinophils (Figs. 2, 3). There were multifocal hemorrhages. Numerous gram-negative bacilli and gram-positive cocci and bacilli were present in the crusts, epidermis, and dermis. Periodic acid-Schiff and Grocott’s methenamine silver stains did not demonstrate any fungi or yeast.
In the chronic lesions obtained from the eyelid of calf 2, left front limb of calf 2, and rear fetlock of calf 5, there was formation of a thick crust composed of proteinaceous exudate, epidermal, dermal, and leukocytic cellular debris mixed with bacteria. There was regeneration of the underlying epidermis with orthokeratotic hyperkeratosis, hyperplasia of the stratum spinosum, and fibrosis of the papillary dermis. The subjacent dermis was devoid of hair follicles and adnexal glands. An inflammatory infiltrate composed of a large number of lymphocytes and plasma cells and a moderate number of macrophages with occasional multinucleated giant cells replaced hair follicles and adnexal glands, and extended into the hypodermis (Figs. 4, 5). Numerous gram-negative bacilli and gram-positive cocci and bacilli were present in the crusts, epidermis, and dermis. Periodic acid-Schiff, Grocott’s methenamine silver, and acid fast stains did not demonstrate any fungi, yeast, or mycobacteria.
Virus Culture
Vero CCL-81 cells inoculated with tissues homogenate obtained from eyelid skin of calf 2 presented CPE in the second passage (Supplemental Table S1). CPE included mild, multifocal cytolysis, cell detachment, and diffuse degeneration. Virus isolation, TEM, and PCR on clarified cell culture suspension confirmed the presence of poxvirus both at the Minnesota Veterinary Diagnostic Laboratory and at the Plum Island Foreign Animal Disease Diagnostic Laboratory. No CPE was observed from skin of left fetlock from calf 5.
Transmission Electron Microscopy
A moderate number of virus particles were detected in crusty skin lesions on negative contrasted preparations of the initial skin crust sample, as well as subacute and chronic lesions from calves 1, 2, and 5. Virus particles were approximately 336.9 nm (SD, 22.09) in length by 254.9 nm (SD, 17.89) in width, characteristic of Poxviridae (250–450 nm by 140–260 nm). 25 This virus consistently presented brick-shape morphology across all lesions, with a surface membrane displaying surface tubular elements (STEs). The virus was enveloped and particles displayed both mulberry (M) and capsular (C) forms (Fig. 6).
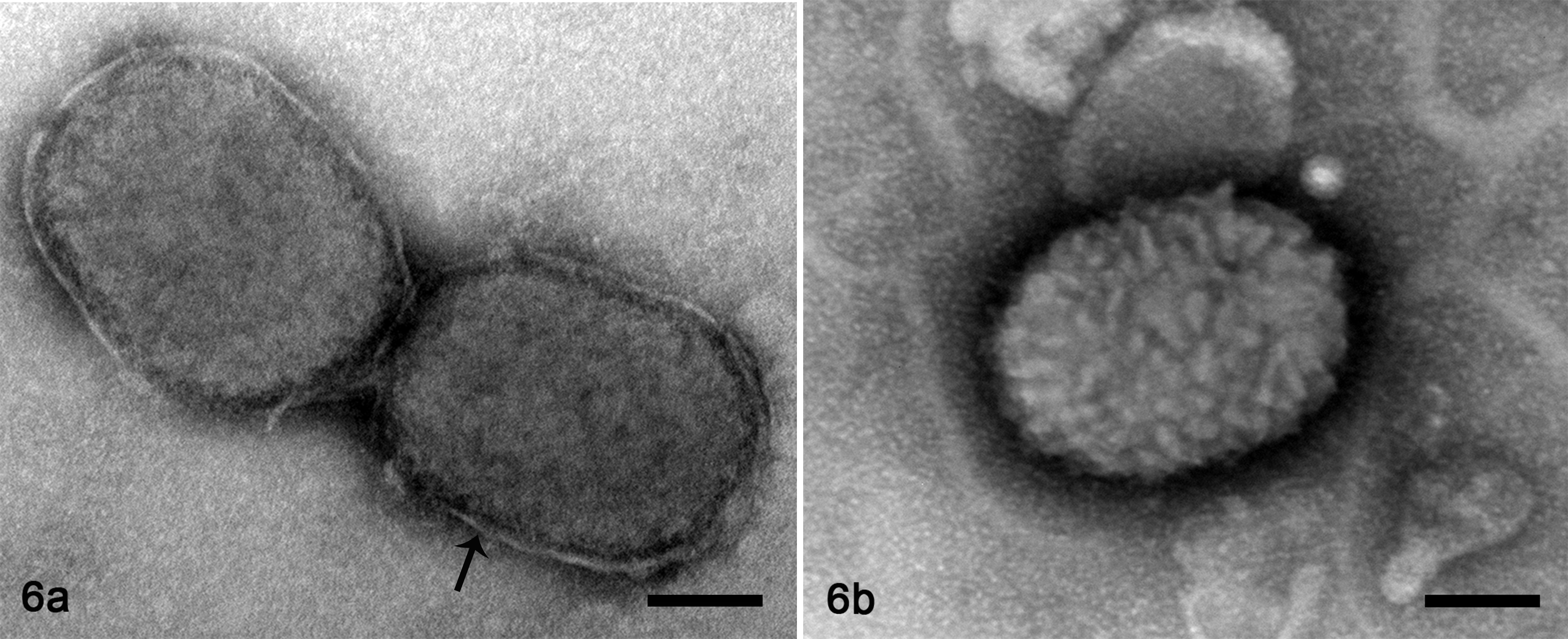
Poxvirus particles, crusty subacute skin lesion, eye canthus, moose, calf 2 . Transmission electron microscopy. Bar = 50 nm. Figure 6a. Poxvirus particles present a “capsular form,” characterized by attenuation of the surface tubular elements, and the envelope is evident (arrow). Figure 6b. A poxvirus particle displays a “mulberry form,” characterized by obvious surface tubular elements; the envelope is not distinct. Bar = 50 nm.
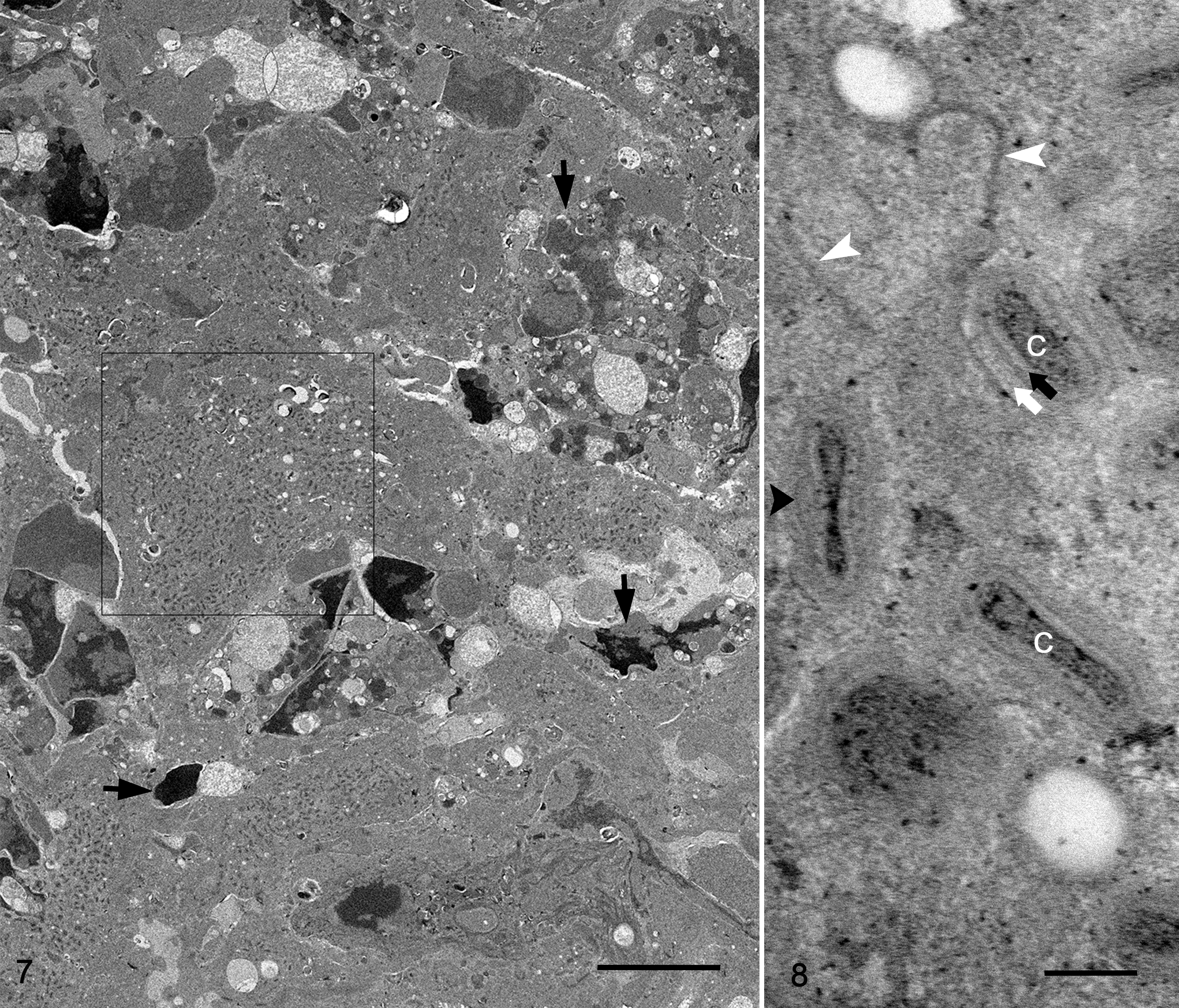
On a plastic-embedded preparation of the subacute eyelid skin lesion, large numbers of virus particles or virions were present in crusty, necrotic epidermis and hair root sheath (Figs. 7, 8). Cells of the stratum spinosum and stratum granulosum contained in the crusty and necrotic epidermis presented mildly electron-dense cytoplasm and exhibited diffuse, fine, electron-dense stippling, which represented large numbers of mature virions (Fig. 7). Cytoplasmic structures and organelles in these necrotic cells were obscured. Occasionally, aggregations of mature virions were found entrapped in a dense meshwork of keratin intermediary filaments within necrotic dried epithelial cells (crust). Intracellular mature virions displayed multiple membrane layers covering a concave shape of the core (Fig. 8). The particles were surrounded by a small amount of fine, granular, moderate-electron-dense matrix in which distinct crescent-like membranes were sometimes found (Fig. 8). Immature virions were rarely seen.
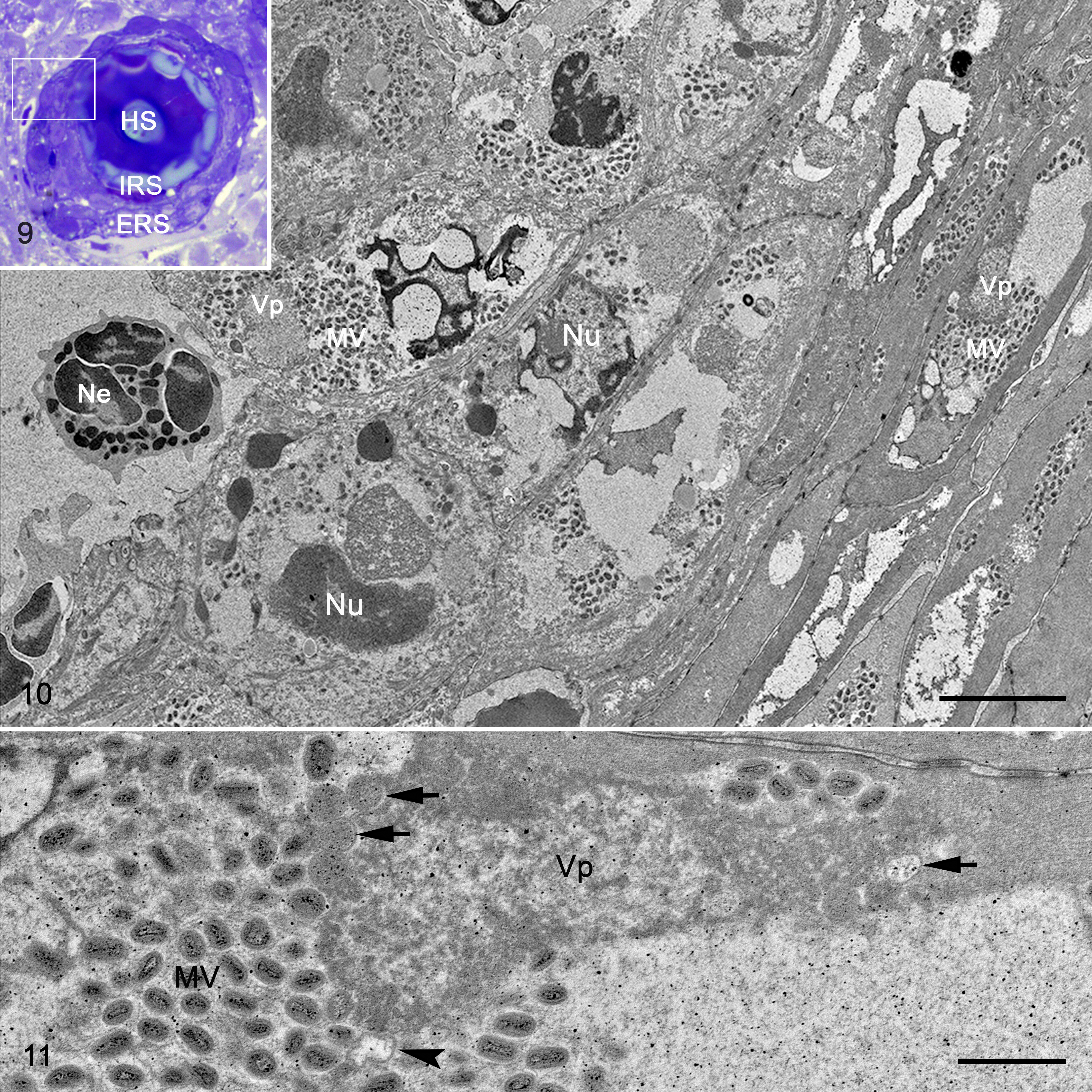
A few hair follicles were partially preserved (Figs. 9, 10). Epithelial cells of the inner hair sheath showed hydropic (ballooning) degeneration. The cytoplasm of the infected epithelial cells of the inner and outer hair root sheath was devoid of organelles and largely replaced by viral factories characterized by random aggregations of mature virions and aggregations of fine, granular, moderate-electron-dense matrix (presumably virosome) with few crescent membranes and immature virions (Figs. 10, 11). Epithelial cells exhibited large empty spaces in their cytoplasm, and their nuclei displayed either condensation or fragmentation of the chromatin (Fig. 11).
The epidermis and dermis were severely infiltrated by inflammatory cells composed of intact and degenerated granulocytes (Fig. 12). Based on their granules, these cells were identified as neutrophils and eosinophils. A number of these granulocytes were actively phagocytizing cellular debris with a myriad of mature virions or free mature virions (Figs. 13, 14). In contrast to subacute lesions, large numbers of virions were present only in necrotic dry epidermal cells within the crust of chronic lesions. Virions were not found in macrophages or multinucleated giant cells.
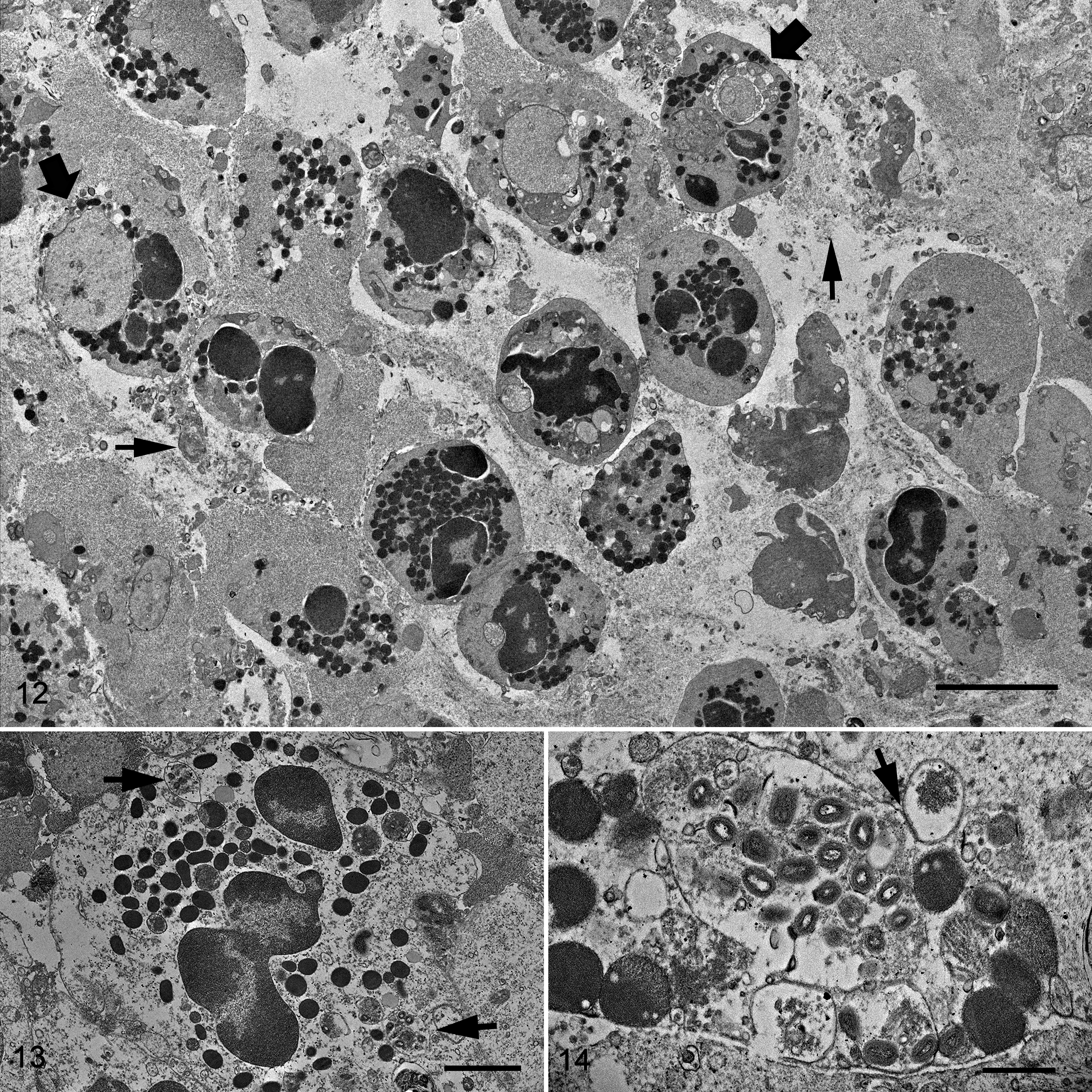
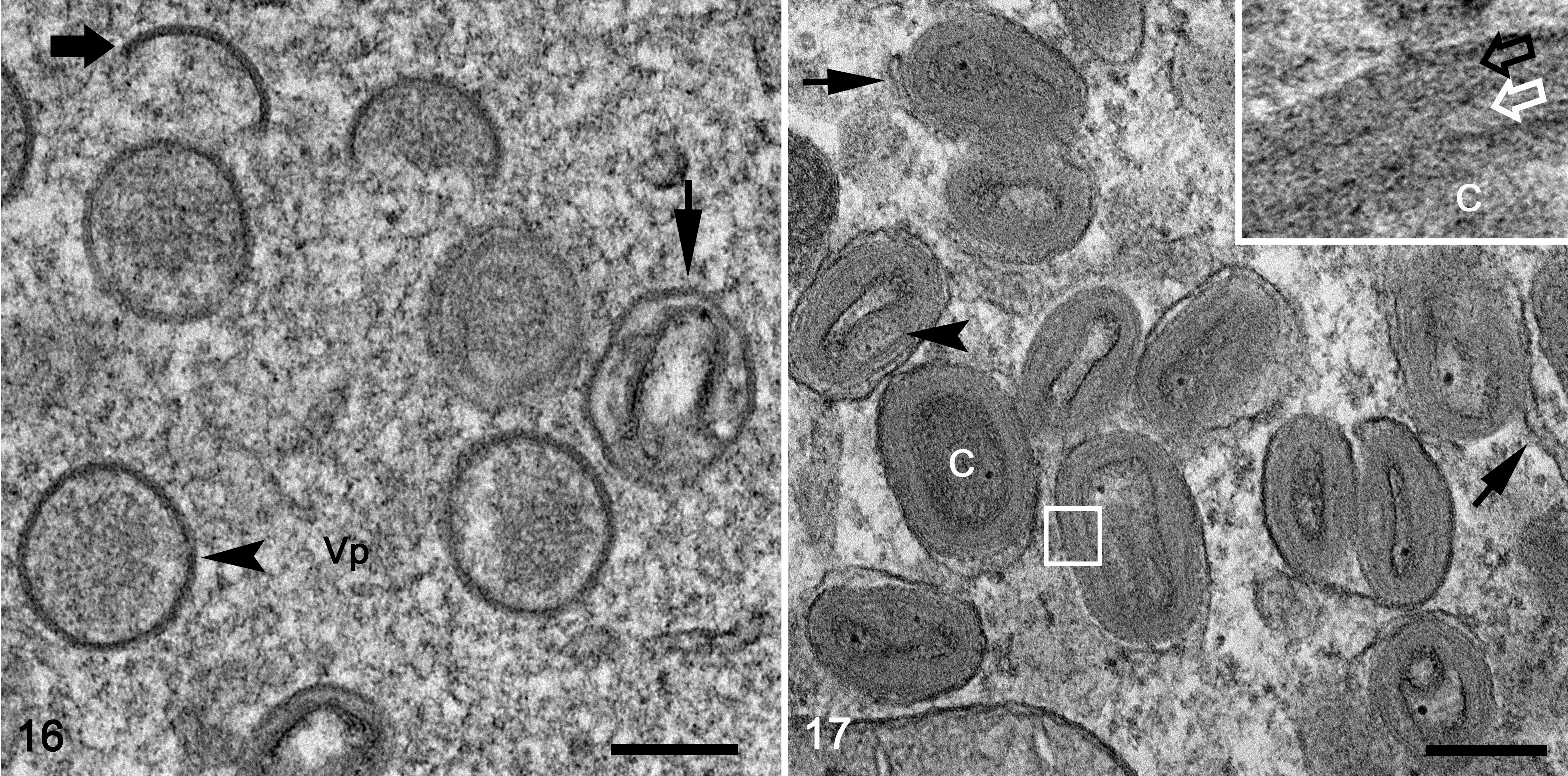
The morphological features of the virion, assembly, and cellular structural changes observed in vivo were confirmed and were further described in detail from virus replicating in cell culture. Under light microscopy, passage 1 virus stock presented a cytopathic effect in less than 20% of the cells. On the Toluidine blue–stained preparations, affected cells were enlarged with diffusely distributed basophilic stippling of the cytoplasm and rarely had intracytoplasmic inclusions. Ultrastructurally, fine intracytoplasmic stippling represented cytoplasmic clustering of virus particles, cell organelles and virosome, or viral factories (Fig. 15). The initial cellular changes in the early stage of the infection consisted of markedly distended cisternae, presumably originating from endoplasmic reticulum, which contained large amounts of amorphous, granular-fibrillary, moderate-electron-dense material. The early virosomes consisted of an aggregation of amorphous, granular-fibrillary material (viroplasm) free in the cytoplasm surrounded by few cellular organelles and endoplasmic reticulum cisternae. In infected cells, virosomes varied in size and in degree of development. Most developed virosomes occupied large extensions of the cytoplasm and consisted of large amounts of amorphous, granular-fibrillary material, which contained crescent membranes, immature virions, and immature virion nucleoids (Fig. 15). Cell organelles were normally absent in the center of virosomes. In general, immature virions, mature virions, and cell organelles were clustered and displaced to the margin of the virus factories. The first indication of viral assembly was the formation of crescents within the viroplasm. Crescents consisted of concave single or bilayer membranes of approximately 24.9 nm (SD, 2.6) in thickness and presented periodic radiations of the outermost layer (Figs. 15, 16). Crescent membranes were of different lengths and maintained a slight curvature until becoming spherules (ie, immature virions). Immature virions were observed as a single bilayer of membrane-bound spheres of about 380.3 nm (SD, 14.9) diameter and contained a variably dense material (Figs. 15, 16). Nucleoids (a form of immature virions) were characterized by an amorphous, electron-dense core (Fig. 15). Immature virions developed to intracellular mature virions that were characterized by the presence of an electron-dense core with a distinct concave shape and lateral body covered by a layered membrane (Figs. 15, 17). Mature virions measured approximately 310.3 nm (SD, 10.2) by 184.8 nm (SD, 33.4) and were present outside of the virosome in large aggregations (ie, type A inclusions). The mature virion core measured approximately 222 (SD, 25.7) by 106.6 (SD, 19.5) by 49.3 nm (SD, 7.8). The core wall was approximately 22.8 nm (SD, 4.7) in thickness. The mature virion membrane, which surrounded the lateral body and core wall, was approximately 23.6 nm (SD, 4.1) in thickness (Fig. 17). Mature virion acquired its envelope in the vicinity of the rough endoplasmic reticulum, Golgi system, and aggregate of small vesicles to form intracellular enveloped virus particles (Fig. 15). Occasionally, enveloped virions were observed outside of the cell, free, and/or associated with the cell plasma membrane. Infected cells at the early stages of infection showed rearrangement of Golgi cisterns and vesicles, mitochondria, and distended rough endoplasmic reticulum.
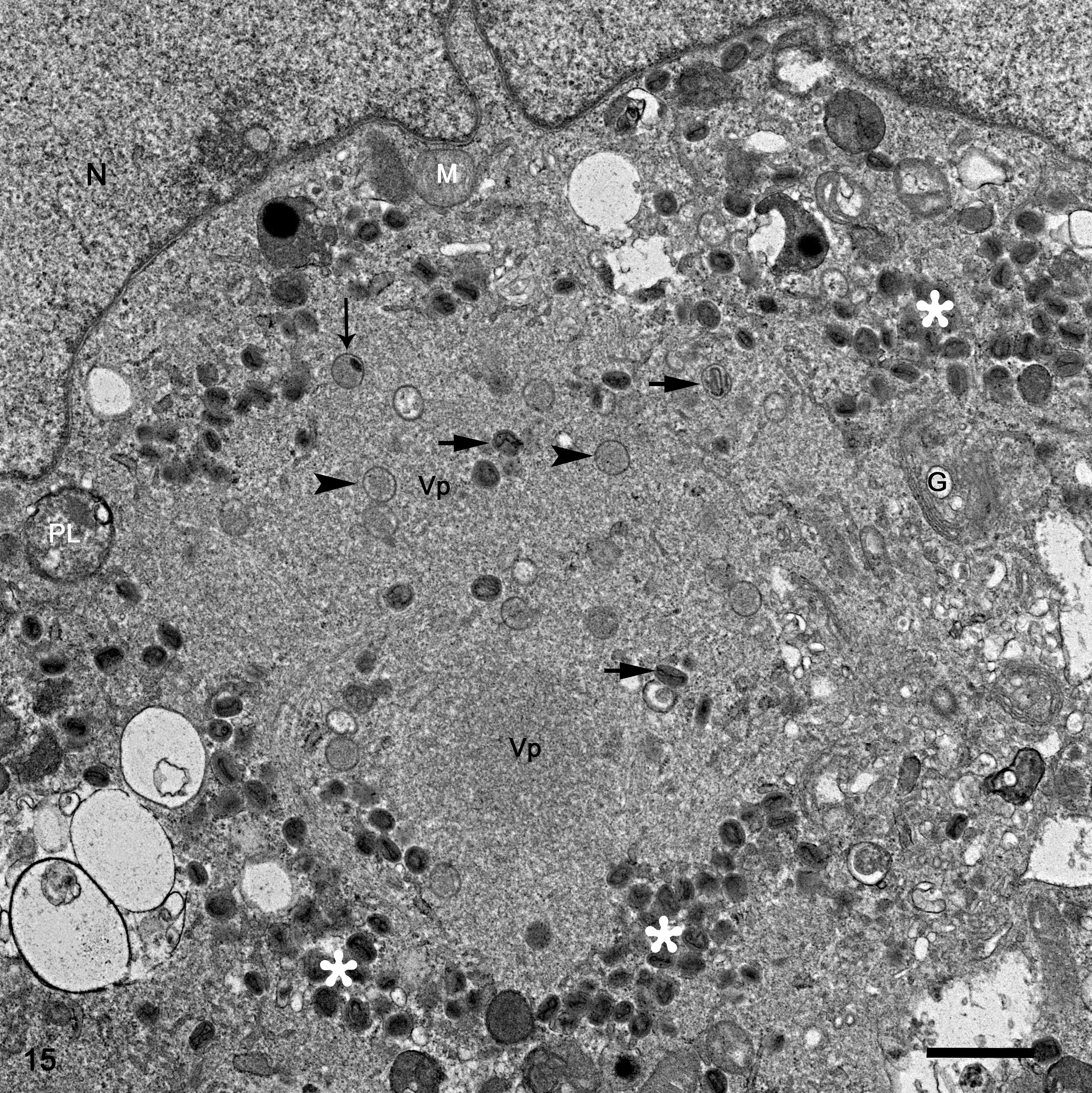
Moosepox virus isolate, Vero CCL-81 cells. Transmission electron microscopy. The cell cytoplasm is occupied by a large viral factory. N, nucleus. Viral factories are composed of areas of viroplasm (Vp) that are devoid of cell organelles and contain areas of maturing and mature virions (white asterisks). Intermingled within the viroplasm are immature virions (arrowheads), immature virion nucleoid (thin arrow), and some particles intermediate between immature and mature virions (thick arrows). Around the viral factory zone, there are altered cell organelles such as phagolysosome (PL), Golgi system (G), and mitochondria (M). Bar = 1 μm.
Polymerase Chain Reaction
The tissue homogenate from both calves 2 and 5 was negative for the presence of capripox viruses using a real-time PCR targeting the poly (A) polymerase gene and negative for parapoxviruses using real-time PCRs targeting the major envelope protein (B2 L) (Supplemental Table S1). However, amplification of poxvirus using consensus primers for both the DNApol and DNAtopo genes, designed to detect species of the Suipoxvirus, Cervidpoxvirus, and Capripoxvirus genera, was positive by conventional PCR for the fresh-tissue homogenate of calf 2 but not calf 5. The amplified product of 349 bp confirmed the presence of poxvirus from DNA extracted from formalin-fixed, paraffin-embedded sections of skin lesions of calf 5 (Supplemental Table S1).
Complete Genome and Genomic Characterization
The genome of MPV was obtained from an isolate from the eyelid of calf 2 (Supplemental Table S1); the entire coding region, including its 170 putative genes, was sequenced, annotated, and compared with existing genomes. There were only 2 other cervidpoxvirus genomes publicly available, namely DPV isolate W-848-83 and W-1170-84. 1 Full genome phylogeny showed that MPV was related to both DPV isolates (Fig. 18) but more closely to DPV isolate W-1170-84. The genome-wide nucleotide (NT) identity between the 2 isolates was 98.5% (Fig. 18). The sequence length of the MPV (GenBank MG751778) was 164,258 bp, compared to 162,891 bp of the DPV isolate W-1170-84 (AY689437); the repeat region at the terminus of MPV was not resolved by short NGS reads. Both MPV and the DPV isolate W-1170-84 encoded the CD30-like protein and a truncated IFN-α/βBP protein, whereas isolate W-848-83 encoded a single IFN-α/βBP protein gene, lacked the CD30-like protein, but encoded a TGF-β1 protein that the other 2 viruses did not contain. Concordantly, these differences were reflected in the whole-genome pairwise identity comparison and phylogeny (Fig. 18). The MPV and DPV isolate W-1170-84 were distinct in certain genes, sharing lower nucleotide and amino acid identities in the CD30-like protein (83.9% nucleotide, 81.6% amino acid), endothelin precursor (73.2% nucleotide including some indels, 51.4% amino acid), and MHC I–like protein (81.0% nucleotide, 68.2% amino acid) (Fig.18, Supplemental Fig. 1). Though characterized in different host species, all 3 viruses were closely related in the same clade, under the species DPV. The nucleotide identities of the DNApol (543 nucleotides) and DNAtopo (344 nucleotides) gene sequences of MPV and gazelle DPV were 97.79% and 98.84%, respectively.
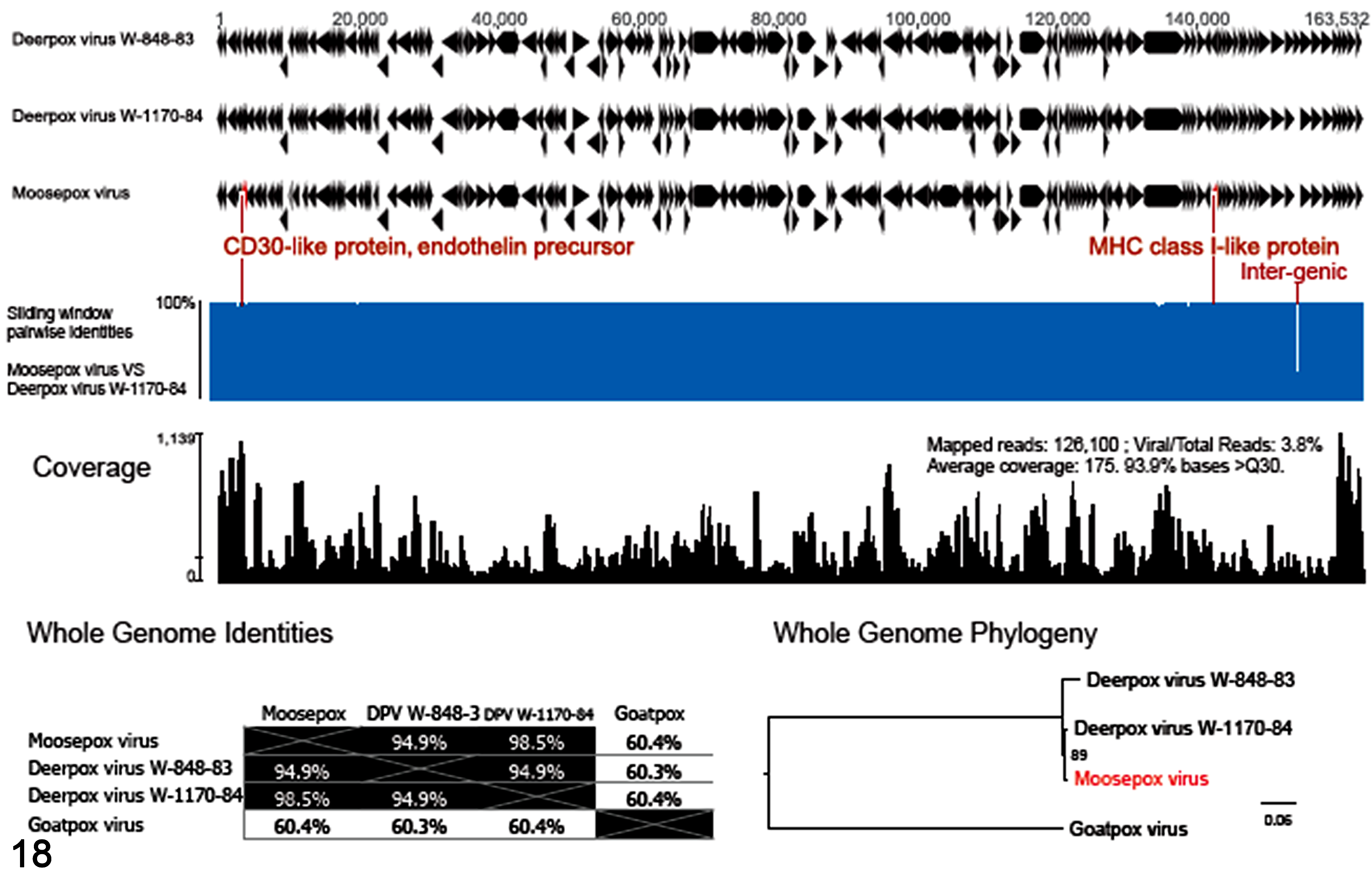
Genomic characteristics of the Moosepox virus. Genome organization was compared between Moosepox virus and the 2 closest relatives of Deerpox viruses. Sliding window analysis compared the nucleotide identities between Moosepox and Deerpox virus W-1170-84, with the difference annotated in red font. Read coverage of the Moosepox virus showed the construction of genome by next-generation sequencing. The whole-genome pairwise identity table (whole-genome identities) and maximum likelihood phylogeny analysis (whole-genome phylogeny) demonstrate the relatedness of viruses within the genus Cervidpoxvirus. The Goatpox virus of the genus Capripoxvirus was used as the outgroup. The phylogenetic relationship of Cervidpoxvirus genus in Poxviridae can be referred in published taxonomic classification. 25
Discussion
We report an outbreak of poxvirus in 3 North American moose calves in a zoological collection in Minnesota. Genomic characterization shows that this North American moose isolate is a new virus of the genus Cervidpoxvirus. Afonso et al. (2005) reported complete genomes of the 2 closely related DPV, describing the difference in CD30-like, TGF-β-like, and IFN-α/βBP genes between strain W-848-83 and W-1170-84. That study concluded that these differences could relate to virus-specific host range and virulence functions for each DPV. Similar to DPV W-1170-84, the MPV in this study contained a CD30-like gene, lacked a TGF-β-like gene, and encoded truncated IFN-α/βBP genes. This suggests the MPV may have shared a common ancestral lineage with DPV W-1170-84, before the divergence that gave rise to W-1170-84. The lower nucleotide and amino acid identity in the CD30-like protein of DPV further confirmed the finding of Afonso et al 1 (2005) that this gene may have a role in virus-specific host range and virulence. Afonso et al 1 (2005) suggested that DPV endothelins contribute to the marked proliferative and necrotizing character of DPV-induced lesions. The lower nucleotide and amino acid identity in the endothelin-precursor gene from DPV and different clinical lesions supports that this gene may influence the MPV-induced lesions as well. These distinguishing features would have been missed by standard molecular tests that covered only the core genes. The evolutionary history of the genus Cervidpoxvirus is far from clear, hindered by the low number of complete genomes for detailed evolutionary analysis. This warrants future effort in monitoring poxvirus in cervids and related animals and observing the genetic diversity through NGS. Fortunately, as demonstrated here, full genomes can be readily obtained by deep sequencing of the cell culture supernatant. Recently, Günther et al 11 (2017) reported the first full-length poxvirus genome, including the terminal hairpin region, directly from skin lesions of a grey seal (Halichoerus grypus) using NGS (MiSeq and Oxford Nanopore MinION). Thus, in addition to isolate sequencing, direct sequencing from clinical samples could provide further benefit, as some poxviruses are challenging to isolate, and culture-adapted genomes might not faithfully represent those of virulent field strains. To advance the field of molecular detection and whole-genome sequencing, NGS is a tool that can advance molecular surveillance in animal health, as it did for surveillance in human health. 18
The clinical presentation of the cervidpoxvirus infection was characterized by focal proliferative lesions in the skin that were limited to the face and extremities. Comparable distribution of lesions has been observed in experimental infection in black-tailed deer. 6 Similar to natural and experimental infection of some of the deer species and a case of DPV infection in gazelle, the moose cervidpoxvirus also seemed to produce a mild and self-limiting disease. 5 –7 Nevertheless, it has been speculated that various pathotypes of DPV circulating within the population are responsible for different clinical presentations and variations in genome sequences. 1 In addition to widespread cutaneous lesions, DPV infection can induce severe buccal, esophageal, and gastric manifestations in black-tailed deer. 5,6 Interestingly, in the few lethal spontaneous cases reported in confined black-tailed deer fawns, a coinfection with adenovirus was documented. 5 Thus, the question of whether the MPV can induce severe or even lethal disease in moose under analogous circumstances remains to be answered.
Examination of the skin submitted for histopathology from moose calves demonstrated subacute necrotic and chronic crusty lesions. In experimental cutaneous inoculation of black-tailed deer with DPV, papules were detectable on days 6 to 8 postinoculation, pustules on days 8 to 12 postinoculation, and formation and sloughing of crusts on days 11 to 13 and thereafter. Subacute lesions in moose calves were histologically characterized by serocellular crusts, epidermal necrosis, and a large number of neutrophils and eosinophils infiltrating the dermis, follicle, adnexal glands, and dermis. These are similar to lesions observed in black-tailed deer 6 to 8 days after cutaneous inoculation with DPV. 6 Chronic lesions of moose calves were histologically characterized by serocellular crust, epidermal regeneration, large numbers of lymphocytes and plasma cells, and a moderate number of macrophages with occasional multinucleated giant cells infiltrating the dermis, follicle, and adnexal glands. As with subacute lesions, these features were similarly observed 11 to 13 days after experimental cutaneous inoculation of DPV in black-tailed deer. 6 Due to the advanced character of the lesions, marked vacuolation and ballooning degeneration with eosinophilic inclusion bodies of epidermal cells 5,6 were not observed in HE-stained sections of skin of any of the affected moose calves. Experimental DPV infection demonstrated that lesions can rapidly evolve to a crusty stage in which histological features of the lesion are dermatitis, folliculitis, and furunculosis with multinucleated giant cells; those lesions were completely healed after 27 days even with the presence of secondary bacterial infections. 6 Among the current moose calf cases, some of the lesions rapidly evolved to crusty lesions as with the aforementioned DPV experimental infection of black-tailed deer. Yet experimental studies with MPV have to be performed to properly characterize the histological lesions in moose.
Studies show that the immune response to poxvirus varies significantly depending on host and poxvirus species; both humoral and cell-mediated immunity are crucial for virus clearance and the prevention of reinfection. 26 With MPV infection in moose, our observations demonstrate a strong local leukocyte response, which in other species plays a key role in controlling poxvirus infection through a number of different complex mechanisms. 26 Phagocytosis, especially by neutrophils, contributes to poxvirus depletion initially and, through the release of oxygen- dependent and -independent antimicrobial products, is able to limit poxvirus infection. Also, leukocytes release interferon and other cytokines, activating an antiviral state in local cells and recruiting new leukocytes into the area. However, it is well-accepted that while an inflammatory response is required to slow the progression of the poxvirus infection, it is not sufficient to eliminate poxvirus infection without additional host humoral and cell-mediated defense mechanisms. 26 In our study, subacute lesions were characterized by a dominant neutrophilic infiltration, which in the more chronic lesions was replaced by macrophages and lymphoplasmacytic infiltrates. On TEM, while the presence of numerous poxvirus particles in phagolysosomes of neutrophils was a prominent feature of subacute lesions, poxvirus particles within phagolysosomes of macrophages were not observed in the chronic lesion.
Although significant limitations impeded precise characterization of the histological lesion, the ultrastructural analysis of the calves’ skin and infected Vero CCL-81 cells offered a robust insight into cytopathology and virus morphogenesis. On TEM, with application of both negative and positive contrast on subacute lesions, the virus was found in cellular debris, necrotic epidermal cells, and large numbers of reactive neutrophils. Like other members of the Chordopoxvirinae subfamily, on which fresh crust and skin negatively contrasted with phosphotungstic acid, this MPV presented brick-shape morphology with a surface membrane displaying the STEs, which are important diagnostic characteristics. 25 STEs are randomly arranged ridges on the membrane surface and obvious on M forms of virus particles. On the contrary, the C form of the virus observed most likely resulted from penetration of phosphotungstic acid solution into damaged virions, which distorts the contrast that ordinarily highlights STEs. 25 Interestingly, the MPV was significantly larger (336.9 x 254.9 nm) than the DPV in white-tailed deer (230 x 200 nm). 5,21 In necrotic epidermal cells, particles of MPV completely replaced the normal cytoplasmic cell components; in preserved epithelial cells of hair follicles, virus factories and aggregation of mature virions (presumably Type A inclusion) were conspicuously present. 25 All stages of poxvirus morphogenesis, including crescents, spherical immature virion, mature virion, and enveloped virions, were observed in these epithelial cells of hair follicles. Nonetheless, crescent and spherical immature virions, which were scarce in epidermal cells of calves, were prominent in Vero CCL-81 cells, which was probably related to the advanced stage of infection of the particular cell: older infections will have more mature virions, whereas earlier infections will have more viroplasm, crescents, and immature virions at the replication site. Furthermore, the amorphous, loose appearance of the viroplasm in epidermal cells of calves was probably due to degradation of structural components of the infected epidermis as a consequence of the cell death or autolysis.
The examination of Vero CCL-81 cells infected with MPV likewise confirmed that all stages of Cervidpoxvirus morphogenesis are, in general, similar to those described in the poxvirus proposed model for the prototype vaccinia virus 2,23,25 and summarized as follows. Vaccinia virus binds and enters the host cell by losing the membranes and releasing the viral core and lateral bodies into the cytoplasm. The core is then transported near the cell nucleus. Transcription of the early mRNAs leads to core uncoating and subsequent DNA replication. Several integral viral membrane proteins are made in the endoplasmic reticulum and transported to viral factories along with endoplasmic reticulum–derived lipid to be assembled into crescents that contain a lipid bilayer and the membrane proteins, scaffolded on a honeycomb structure. Crescents encapsidate genome material, subsequently evolving to immature virions and immature virion nucleoids. Conversion to mature virion or mature virus particle is accompanied by the addition of membrane proteins and movement of particle outside of factories. The majority of intracellular mature virions are released from the cell by cell lysis. Some intracellular mature virions are transported to sites of wrapping by endosomes and the trans-Golgi network. Intracellular mature virions are wrapped by a double membrane to form intracellular enveloped virions that are then transported to the cell surface on microtubules. The outer intracellular enveloped virions’ membrane fuses with the plasma membrane to expose cellular enveloped virus at the cell surface. Polymerization of an actin tail beneath the cellular enveloped virion can occur to drive the virion into a neighboring cell or release the virion as an extracellular enveloped virus particle. 9,23,25 In current work, preliminary observations in Vero CCL-81 cells provide evidence of active involvement of the rough endoplasmic reticulum in the formation of the MPV virion membranes, as has been observed by Weisberg et al 29 (2017) in vaccinia virus cultures. Moreover, TEM tomography contributes to understanding the role of the virion membrane in the complex assembly of poxvirus. 29 This technology may identify similarities and differences between various poxviruses.
It is unknown how, when, and where these moose calves were infected. An experimental study in black-tailed deer fawns (Odocoileus hemionus) demonstrated transmission of Cervidpoxvirus by cutaneous inoculation and direct contact. 6 There is also evidence that Orthopoxvirus and Cervidpoxvirus can be transmitted to a sensitive novel host when exposed to infected carriers of other species housed in the same facility. 7,15,24 While the moose calves were never in direct contact with any other animals managed by the zoo, they regularly rotated through the same pens and exhibit space with the single adult moose housed at the zoo at the time of this outbreak. As these pens and exhibit space are outdoors, we cannot rule out that local wildlife (eg, rodents) played a role in transmission. In addition, husbandry staff worked with other nondomestic ruminants. However, since the time the calves were acquired by the zoo, none of the nondomestic ruminants or mammals housed in the zoo developed lesions suggestive of poxvirus infection. Further, direct contact with native deer within the zoological facility was considered highly unlikely.
Our epidemiological investigation did not confirm a source of infection at the zoological facility. All 3 calves presented poxvirus lesions approximately 4 months after being acquired by the zoo, which coincided with weaning to an adult diet. It is conceivable that the stress these calves experienced during weaning may have predisposed to clinical disease resulting from latent infection; the latter has been proposed in DPV infection in black-tailed deer. 5,6 This scenario would suggest that the poxvirus was acquired by one or more of the calves in the wild, although we are not aware of any reports of cutaneous proliferative disease suggestive of poxvirus in wild moose in northern Minnesota. It is important to note that DPV infection was diagnosed in a goitered gazelle at the same facility 4 years prior to the current outbreak. 7 Although genetic sequencing of only 2 genes, DNApol and DNAtopo, was performed to identify the virus, the genome was not fully sequenced. 7 Given the genetic similarity of the known DPV strains and the MPV strain, it is clear that MPV is divergent from gazelle DPV. We are not sure if MPV is the result of continuous evolution of previously reported DPV or new introduction in the facility. It is also possible the virus has been subclinically circulating within the zoo population, and further investigation of the possible involvement of a vector or reservoir of Cervidpoxvirus (eg, rodents or invertebrates) should be conducted. 12
This study provides biological characterization of a new Cervidpoxvirus attained through in vivo and in vitro ultrastructural analyses. It also demonstrates the importance of whole-genome sequencing in the molecular characterization of poxviruses identified in taxonomically related hosts. This outbreak of poxvirus in moose calves expands the host spectrum of Cervidpoxvirus. Although we were unable to identify the source of the MPV, this event highlights the importance of careful surveillance for infectious diseases in facilities with dense populations of potentially susceptible host species. Taken together, our findings provide new insights into the clinical-pathological presentation and pathogenesis of infection with the genus Cervidpoxvirus in moose.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Armien_et_al - Molecular and Biological Characterization of a Cervidpoxvirus Isolated From Moose with Necrotizing Dermatitis
Supplemental Material, Combined_supplemental_materials-Armien_et_al for Molecular and Biological Characterization of a Cervidpoxvirus Isolated From Moose with Necrotizing Dermatitis by Anibal G. Armién, Tiffany M. Wolf, Sunil Kumar Mor, Terry Fei Fan Ng, Alexa J. Bracht, Sagar M. Goyal and James M. Rasmussen in Veterinary Pathology
Footnotes
Acknowledgements
The authors are thankful to Dean Muldoon for his outstanding electron microscopy preparation. We are grateful to the histology and virology team at Veterinary Diagnostic Laboratory of University of Minnesota for the outstanding support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
