Abstract
This report presents a novel canine condition in 32 dogs in which aberrant migration of Spirocerca lupi larvae through mesenteric arteries, instead of gastric arteries, led to small or large intestinal infarction. This form of spirocercosis was first recognized in Israel in 2013 and is currently ongoing. Typical clinical signs were anorexia and weakness of 3 to 4 days and, less frequently, vomiting and diarrhea, followed by collapse, bloody diarrhea, and severe vomiting. Exploratory laparotomy showed 1 or more infarcted and often perforated intestinal segments in all cases. Microscopically, there was intestinal mucosal to transmural coagulative necrosis and mesenteric multifocal necrotizing eosinophilic arteritis, thrombosis, hemorrhage, and early fibroplasia. Third-stage S. lupi larvae were identified by morphologic features in 9 of 32 (28%) cases, and the species was confirmed by polymerase chain reaction in 4 cases. Nearly 50% of the dogs had been receiving prophylactic therapy, which did not prevent this form of spirocercosis.
The parasitic nematode Spirocerca lupi is found worldwide in warm countries, including Israel, and causes significant morbidity and mortality in dogs. 1,7 Following ingestion of intermediate (dung beetles) or paratenic (eg, reptiles and birds) hosts, infective third-stage (L3) larvae excyst in the host’s stomach and penetrate the gastric wall, where most of them are found near or in small arteries at 48 hours postinfection. 5,8 The larvae penetrate gastric arterial walls and migrate within them to the wall of the caudal thoracic aorta, where they are found from approximately 14 days after hatching 5,8 and where they mature to L4 larvae. Following a final molt and approximately 3 months postinfection, they leave the aorta as immature adults and settle in the wall of the caudal esophagus, where they induce the formation of fibrous nodules (so-called granulomas). 7,8,12 The typical and most common lesions of spirocercosis are esophageal granulomas, which frequently undergo neoplastic transformation to sarcoma (most commonly osteosarcoma 1,12 ), aortic aneurysms and scars, and caudal thoracic spondylosis. 7,12 Aberrant larval migration is fairly common and can manifest as adult-containing fibrous nodules in a wide variety of organs 7,12 or lead to diverse atypical presentations. 1
The prevalence of spirocercosis in Israel has increased dramatically since the early 1990s. 1,7 In this report, we describe the gross and microscopic pathologic findings of a novel presentation of spirocercosis in which aberrant larval migration in mesenteric arteries causes arterial necrosis with eosinophilic arteritis and thrombosis, leading to intestinal infarction.
The 32 dogs included in this report (Table 1) presented to the Hebrew University Veterinary Teaching Hospital (n = 20) and to private veterinary practices (n = 12) from October 2013 to June 2019 (2013: 1 case, 2014: 2 cases, 2015: 5 cases, 2016: 2 cases, 2017: 7 cases, 2018: 12 cases, and 2019: 3 cases), mostly (27/32) during the dry season (April to October). All dogs were large (body weight ≥25 kg) of pure or mixed breeds. The age range was wide (0.5 to 14.5 years old). There were 19 males and 13 females, and most dogs were from central Israel (data not shown). Clinical data were obtained from medical records and, in selected cases, by telephone interviews with veterinarians and owners. Routine prophylaxis (doramectin 200 μg/kg subcutaneously every 3 months 1 ) had been administered to 15 of 26 dogs (59%) for which information was available. Clinical data were available for 22 of 32 cases. The usual history was 3 to 4 days of progressive anorexia, weakness, and, in some cases, vomiting and/or diarrhea followed by acute deterioration characterized by collapse, continuous vomiting, and bloody diarrhea.
Clinical and Pathologic Findings in Dogs With Intestinal Infarction Due to Infection with L3 Larvae of Spirocerca lupi. a
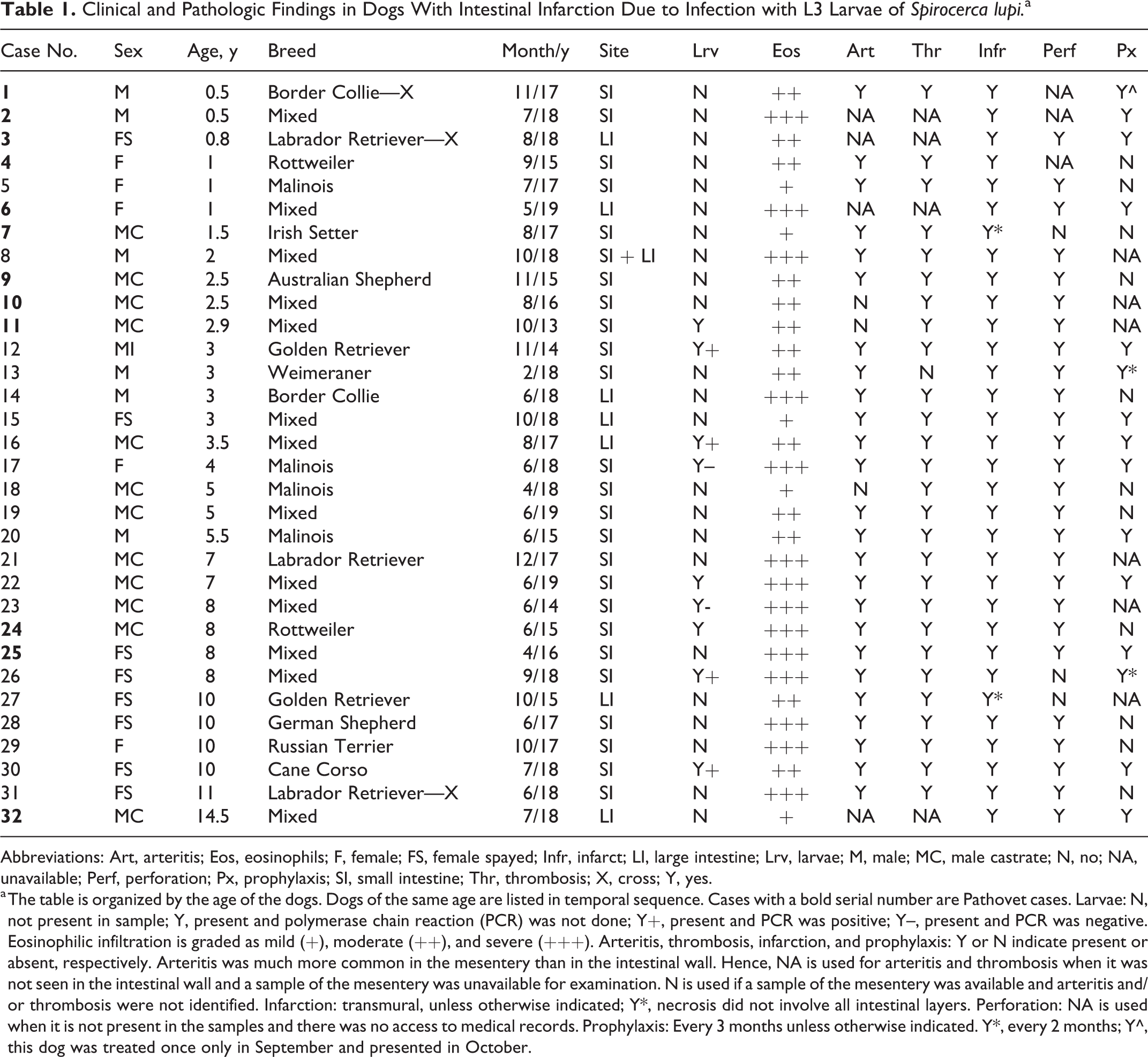
Abbreviations: Art, arteritis; Eos, eosinophils; F, female; FS, female spayed; Infr, infarct; LI, large intestine; Lrv, larvae; M, male; MC, male castrate; N, no; NA, unavailable; Perf, perforation; Px, prophylaxis; SI, small intestine; Thr, thrombosis; X, cross; Y, yes.
a The table is organized by the age of the dogs. Dogs of the same age are listed in temporal sequence. Cases with a bold serial number are Pathovet cases. Larvae: N, not present in sample; Y, present and polymerase chain reaction (PCR) was not done; Y+, present and PCR was positive; Y–, present and PCR was negative. Eosinophilic infiltration is graded as mild (+), moderate (++), and severe (+++). Arteritis, thrombosis, infarction, and prophylaxis: Y or N indicate present or absent, respectively. Arteritis was much more common in the mesentery than in the intestinal wall. Hence, NA is used for arteritis and thrombosis when it was not seen in the intestinal wall and a sample of the mesentery was unavailable for examination. N is used if a sample of the mesentery was available and arteritis and/or thrombosis were not identified. Infarction: transmural, unless otherwise indicated; Y*, necrosis did not involve all intestinal layers. Perforation: NA is used when it is not present in the samples and there was no access to medical records. Prophylaxis: Every 3 months unless otherwise indicated. Y*, every 2 months; Y^, this dog was treated once only in September and presented in October.
Emergency exploratory laparotomy, performed in all cases, invariably showed 1 or more well-demarcated segments of intestinal necrosis and hemorrhage with severe congestion and hemorrhage of the adjacent mesentery. The small intestine was affected in 24 cases (75%), the large intestine in 7 (22%), and in 1 case, both were affected. Perforation and septic peritonitis were common (26 of the 29 dogs for which these data were available, 90%). Resection and anastomosis were performed, and intestinal segments containing the necrotic regions with associated mesentery were collected and fixed in 10% neutral buffered formalin. Specimens that were received in their entirety were up to 80 cm long and included 1 or more approximately 3- to 20-cm-long well-demarcated areas of locally extensive hemorrhage and necrosis. Affected segments were mildly dilated, the wall was often mildly thickened, and some contained intraluminal blood. In perforated sites, the intestinal wall was friable and often green-tinged (Fig. 1). Congestion and multifocal hemorrhage were usually present in the flanking viable tissue. Representative samples (1–8 slides) were collected from each case, processed routinely, embedded in paraffin, cut into 4- to 5-µm sections, and stained with hematoxylin and eosin.
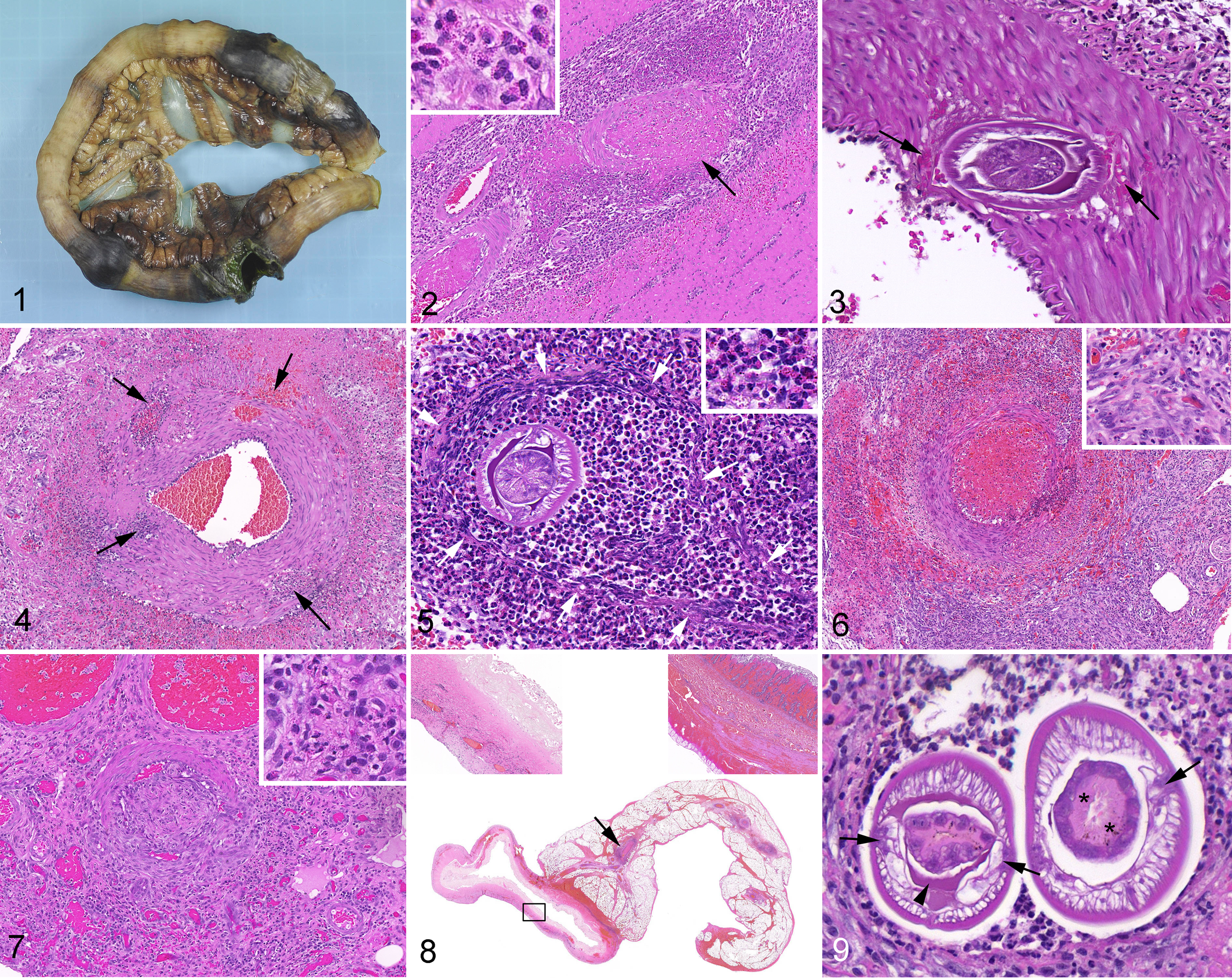
The microscopic changes in all dogs included eosinophil infiltration and intestinal infarction. The hallmark of this condition was multifocal to diffuse necrosis of small- and medium-sized (0.2–1.2 mm in diameter) mesenteric arteries, accompanied by perivascular eosinophil infiltration, fibrin deposition, hemorrhage, thrombosis, and, in longer-standing cases, fibroplasia. Veins were conspicuously spared. Arteritis of this nature was identified in 25 of the 28 dogs from which mesentery samples were available. In the 3 cases in which samples of the mesentery were available but arteritis or arterial necrosis were not identified, arterial thrombosis was seen, suggesting the presence of arterial lesions that were not included in the samples. Similar lesions were occasionally identified in serosal and rarely in intestinal arteries.
Arterial damage took several morphologic forms. In the most prevalent form, randomly distributed and variably sized areas of the arterial wall were replaced by variable proportions of fibrin, nuclear debris, erythrocytes, and eosinophils (Fig. 2). Second, in some cases, sharply defined holes in the arterial wall were noted. The presence of larvae, surrounded by hemorrhage, within such holes was rare but diagnostically significant (Fig. 3). More commonly, these holes were filled with fibrin, erythrocytes, and cellular debris in various amounts, but their diameter suggested that they were produced by larval burrowing (Fig. 4). A third, relatively uncommon form of arterial damage was diffuse necrosis of the arterial wall, leading to its disintegration. Affected arteries were always surrounded by numerous eosinophils (Fig. 5).
To estimate the age of lesions, the most advanced degree of fibroplasia, the organization stage of thrombi, and the presence of a mononuclear infiltrate were evaluated. In the vast majority of cases, there was significant fibroblast hypertrophy and hyperplasia or early granulation tissue formation, which was mainly located around blood vessels (Fig. 6) and in interlobular septa, becoming more widespread with chronicity. In lesions interpreted as being older, necrotic foci in arteries were replaced by granulation tissue and the number of eosinophils decreased (Fig. 7). Acute and subacute arterial lesions coexisted in some cases. Intestinal infarction was identified by locally extensive transmural coagulative necrosis, with or without hemorrhage (Fig. 8). The fully necrotic segment was often flanked by areas with mucosal or mucosal and submucosal coagulative necrosis. Neutrophilic infiltration at the edges of necrotic areas was uncommon.
Sections of nematode larvae were identified in 9 of 32 (28%) cases in the wall or lumen of mesenteric arteries or free in the mesentery or in the intestinal wall. In 7 of 9 cases, they were identified focally, and in 2 cases, they were detected in 2 different areas. They occurred as a single or a group of sections (maximum 4) near each other and in nearly all cases (9/11 sites) were surrounded by inflammatory cells. Their morphologic features were similar in all cases. In transverse sections, their diameter was 75 to 166 μm. They had a smooth cuticle, polymyarian-coelomyarian musculature, and lateral chords of variable size. The lateral chords were large and projected into the pseudocoelom. Some larvae contained homogeneous eosinophilic material in the pseudocoelom, a characteristic feature of spirurid nematodes. 3 The intestine was composed of uninucleated cuboidal cells with a prominent brush border. The absence of gonads indicated a larval stage, and the small diameter identified them as L3 (Fig. 9). 5,11
DNA was extracted from formalin-fixed, paraffin-embedded (FFPE) sections of <20 µm thickness from 6 different cases using the Zymo Research Quick-DNA FFPE kit (Zymo Research, Irvine, CA) according to the manufacturer’s instructions with the following modifications. Briefly, excess paraffin was removed and samples transferred into a tube. Then, the volumes of the deparaffinization solution, digestion buffer, and proteinase K were doubled, and the digestion was performed overnight at 55°C. Finally, DNA was eluted in 50 μl DNA elution buffer. After this, a high-resolution melt (HRM) real-time polymerase chain reaction (PCR) targeting a fragment of the 18S gene of S. lupi was run using primers and conditions described previously. 10 S. lupi adult DNA and PCR-grade water were used as positive and negative nontemplate controls in each reaction. DNA amplicons from positive reactions were purified (Exo-SAP New England Bio-Labs, Ipswich, MA) and sequenced using the Big-Dye Terminator cycle sequencing chemistry from the Applied Biosystems ABI3700 DNA Analyzer and the ABI’s Data Collection and Sequence Analysis software (ABI, Carlsbad, CA). The obtained DNA sequences were compared to the S. lupi sequences available in GenBank using the BLAST 2.2.28 program. Samples were considered positive for S. lupi when the obtained DNA sequence was at least 98% identical to S. lupi sequences in GenBank. Based on this protocol, S. lupi was confirmed in 4 of 6 cases.
To ensure the quality of the DNA in the 2 of 6 cases that were negative, a PCR that amplifies a 600-bp fragment of the cytochrome b oxidase of dogs was performed using the primers and conditions described previously. 6 All extracted DNA samples were positive in this reaction, indicating that the quality of the extracted DNA was good, and thus, the FFPE samples were truly negative for S. lupi. The negative results may be due to technical reasons (eg, loss of parasite tissue from material submitted to PCR despite its presence in the paraffin blocks or concentration of the parasite-associated DNA, which was lower than the detection limit of the assay) or due to infection by larvae of a different helminth species within the order Spiruridae, although the authors are not aware of such a parasite infecting dogs in Israel.
With few exceptions, due to sample limitations, the cases included in this report had all the above tissue changes (ie, mesenteric arteritis, thrombosis, intestinal infarction, and eosinophil infiltration) (see Table 1). These microscopic features were found both in cases in which L3 spirurid larvae were identified (9/32, 28%) and in cases in which an etiologic agent was not detected (23/32, 72%). Based on this similarity, all 32 were considered as a single condition. In our region, this combination of lesions is virtually pathognomonic and permits a presumptive diagnosis in the absence of the parasite.
S. lupi–induced intestinal infarction was first recognized in Israel in October 2013, and cases have continued to occur. The highest number of cases was recorded in 2018 (12/32, 38%). The reasons for the emergence and increased incidence are unknown. The signalment of aberrant S. lupi–induced intestinal infarction was similar to that of esophageal spirocercosis in Israel, except for the relatively large number of young (<5-year-old) dogs affected (19/32, 59%). The seasonal distribution, with 27 of 32 (84%) dogs presenting in the dry season (April to October), differed from both the predominance of cases presented during the cold season (December to April) described for 1991 to 1999 and the relatively even distribution described for 2004 to 2009. 1,7 The geographic distribution (not shown) was similar to the more recent report. 1 The microscopic anatomic features of the nematode identified it as a spirurid. 3 Speciation using HRM-PCR identified it as S. lupi. Its small size and anatomic location in the host were consistent with L3, the infective stage. 11 This suggests that the condition occurred in recently infected dogs. Since spirocercosis-associated esophageal nodules develop approximately 3 to 7.5 months postinfection, 11 a high rate of infection in the dry season could account for a predominance of such clinical cases in the cold season. 7 The relatively low number of cases in which larvae were identified (9/32, 28%) suggests that not many larvae were present in the tissue of each case, but sampling error may also play a role.
There was a good correlation between the clinical signs and the pathologic findings. The microscopic findings indicate that the larvae migrated in mesenteric arteries for several days before the intestine undergoes infarction. The initial nonspecific clinical signs appear to correspond to this period. The acute clinical deterioration was most likely due to intestinal infarction attributable to arterial thrombosis of an increasing number of small arteries and/or occlusion of larger arteries reached by the larvae. This supposition is supported by data from human patients with acute mesenteric ischemia. The nonspecific clinical presentation of this condition makes its diagnosis before bowel infarction occurs difficult. 9 Infarcted intestine undergoes perforation rapidly, which explains why perforation occurred in the vast majority of dogs (26/29, 90%). An experimental study of small intestinal infarction in dogs showed that all animals subjected to 8 hours or more of ischemia died with gangrene with or without perforation. 4
The arterial lesions are considered to be due to a combination of physical microtrauma inflicted by the migrating larvae and the eosinophilic response they evoke. They are very similar to those described in an old experimental study. 5 But whereas the lesions that develop in the course of normal migration through gastric arteries heal rapidly without significant sequelae, 5 aberrant migration in mesenteric arteries leads to devastating consequences. We suggest that the reason is the collateral circulation, which is well developed in the stomach and, in contrast, poor in the intestine. 2
The majority of dogs (59%) received injectable doramectin as prophylaxis, according to the regimen that is currently used in Israel, proving that it does not confer protection against this early stage of S. lupi infection. This protocol has also been shown to be nonprotective in some cases of esophageal spirocercosis 1 and in some cases of aberrant S. lupi migration in the spinal cord (Merav Shamir, personal communication, 2019), raising concern about the dosing interval and possibly the emergence of resistance against macrocytic lactones in the S. lupi population in Israel. 1
Footnotes
Acknowledgements
We thank Prof. Itamar Aroch, Prof. Merav Shamir, and Prof. Sigal Yudelevich for their kind assistance and helpful discussions; Marina Cohen for translating the article by N. S. Nazarova from Russian; and Omer Lerman for his assistance to Dr. Shelly Hahn. We are grateful to Marina Cohen and Lena Prichisliv for their expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
