Abstract
Infections with Trichuris vulpis, the canine whipworm, may be challenging to diagnose even though characteristic bipolar eggs are shed by mature worms and may be recovered from feces. Decreased detection sensitivities because of using flotation solutions with specific gravities <1.3 and a lengthy prepatent period can lessen the diagnostician’s ability to detect infection. Coproantigen detection in feces is becoming an accepted form of diagnosing parasitic infections and can circumvent some of the factors that affect egg recovery. The development of an enzyme-linked immunosorbent assay (ELISA) for the detection of whipworm-specific coproantigens in the feces of dogs with experimental and natural T. vulpis infections is reported herein. Whipworm-specific coproantigens were evidenced in feces from experimentally infected dogs using the newly developed ELISA starting as early as day 23 postinfection, while eggs were not detected in feces until day 69. In addition, 1,156 field fecal samples were tested using fecal flotation methods and the newly developed whipworm ELISA. Of these, 27 samples were found by flotation to be whipworm egg positive, while 35 had detectable antigen on the ELISA. Discrepant results were obtained in 12 samples; 2 egg-positive samples tested ELISA negative, and 10 ELISA-positive samples did not contain detectable egg levels. Using the fecal ELISA for the detection of whipworms in dogs should allow for earlier detection of infection, aid the identification of cases in the face of low egg shedding, and increase detection sensitivity as most commercial laboratories are using flotation solutions not optimal for T. vulpis egg detection.
Introduction
Signs of canine whipworm (Trichuris vulpis) infection in dogs may range from inapparent disease, to bloody diarrhea, anemia, and in severe cases transmural ileocolitis and perhaps death. 27 The life cycle of this parasite is direct. Noninfectious, unembryonated eggs are shed in the feces of infected animals, and in the proper soil environment, these eggs become infectious in approximately 1 month. When infectious eggs are ingested, the larvae hatch in the intestine and penetrate the wall of the gut where they mature into young adults before moving to their final location in the cecum or colon where they complete development. Adult worms embed their anterior end into the mucosa and feed on blood and tissue fluids. Diagnosis is typically by identifying the eggs in the feces of the infected host.
The principles behind the morphologic diagnosis of fecal-borne stages of internal parasites, such as whipworms, are simple and straightforward: Observation and identification of these stages in fecal specimens from the patient confirms the diagnosis of infection with the parasite. In practice, a number of factors may influence the diagnostician’s ability to correctly detect an infection, however. These include, but are not limited to: 1) the number of eggs produced by the parasites, 2) the adequacy of the sample (e.g., sample size, freshness, lack of contamination with free-living organisms), 3) the method used for parasite recovery (e.g., fecal smear, sedimentation, staining, simple versus centrifugal floatation, choice of flotation medium), and 4) the ability of the person reading the slide to correctly identify the parasite (i.e., training and experience of personnel reading samples). 5 In the case of whipworms, the number of eggs produced by a single mature female T. vulpis has been calculated to be 2,035 per day18,19; thus, a dog with a single female whipworm and producing 100 g of feces per day, would be shedding approximately 20 eggs per g of feces (EPG). These eggs are known to have a specific gravity greater than that of the other common nematode eggs in dog feces, 4 and thus require higher specific gravity solutions for optimal flotation. 5 Not all veterinary practices or laboratories utilize centrifugal flotation, which is known to detect more cases than stationary flotation,9,24,28 and as laboratories try to maximize their ability to detect both helminth eggs and protozoan cysts, specifically Giardia, solutions with specific gravities of approximately 1.18–1.2 are used rather than around 1.27, which is preferred for the maximal recovery of whipworm eggs.6,14,22,28,29 In addition, biological factors such as the relatively long prepatent period of T. vulpis (approximately 3 months before eggs will be present in feces), and the fact that these worms may stop shedding eggs or shed eggs with distorted eggshells briefly after unsuccessful treatment with certain anthelmintic products2,21,26 can also impinge upon the results of fecal testing. Thus, fecal testing for T. vulpis, using these types of methods, is less efficient than one would hope for the purpose of diagnosing an infection.
Aside from the identification of cases, a major challenge facing control of this parasite in animals exposed to contaminated environments is that reinfection is common, and any of the approved parasiticides for T. vulpis available in the United States are only labeled for the removal of adult worms, leaving behind a population of immature worms still capable of causing disease, maturing, and increasing the environmental load of eggs. For all of the aforementioned reasons, a test capable of detecting worms in the intestine (without the need for necropsy), and not simply eggs in feces, would be of great use in veterinary medicine. The development of an enzyme-linked immunosorbent assay (ELISA) for the detection of coproantigen in the feces of dogs with experimental and natural T. vulpis infections is reported herein.
Materials and methods
Experimental T. vulpis infection
The experimental infections were established under an approved animal care and use protocol in accordance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Academy of Sciences, Washington, DC). Five uninfected dogs acquired from a Class-A U.S. Department of Agriculture animal vendor were treated on day –14 with Interceptor®a per dosages prescribed on the package insert (0.5–1.0 mg milbemycin oxime per kg body weight). Fecal samples were collected prior to infection on days –14, –11, and –1 to verify no helminth infections were present. Embryonation of whipworm eggs was achieved by incubation of eggs at 25°C for approximately 40 days. Dogs were each inoculated orally with approximately 700 infective (embryonated) T. vulpis eggs. Dogs were monitored for 2 hr, at 20-min intervals, to ascertain that no vomiting had occurred. Fecal samples were collected from all 5 dogs through day 87 postinfection (days 0, 2, 9, 17, 23, 31, 38, 48, 52, 62, 69, 73, 76, 79, 84, 85, and 87). On day 89 postinfection, 3 of the 5 dogs (KXZ5, RQZ5, and SEZ5) were again treated with Interceptor at the prescribed dosages and had fecal samples collected through day 111 (days 93, 97, 100, 105, 107, and 111). Egg counts were performed on all samples using 1.25 specific gravity (spg) Sheather sugar solution centrifugal flotation, as described below. Two dogs (REY5 and TGZ5) were necropsied on day 89, and adult whipworms were confirmed, but not enumerated. Dogs KXZ5, RQZ5, and SEZ5 were returned to the colony on completion of the study. Control fecal samples for ELISA were obtained from dog hookworm (Ancylostoma caninum), dog roundworm (Toxocara canis), and uninfected dogs from experimental infection studies.
Egg recovery and quantitation procedure for experimental T. vulpis infections
One gram of feces from each experimental dog was homogenized with approximately 10 ml of water and poured through a strainer; the homogenate was pressed until no more fluid passed through. The strained filtrate was poured into a 15-ml centrifuge tube, and the tube was spun at 174 RCF for approximately 10 min. Following centrifugation, the supernatant was discarded, and the fecal pellet resuspended in approximately 12 ml of 1.25 spg of Sheather sugar solution (2,267 g of sugar, 29.5 ml of melted phenol, and 1,451.2 ml of distilled water) before centrifuging, as before. Following centrifugation, the tube was carefully filled with the sugar solution until a small positive meniscus was formed. A coverslip was placed on the meniscus and let stand for at least 10 min after which time the coverslip was removed and placed on a microscope slide. Examination for T. vulpis eggs was made at 100× power, and the number of eggs counted beneath the coverslip was considered the number of EPG.
Egg recovery from canine field fecal samples
Egg flotation data was collected from 4,240 canine fecal samples submitted to IDEXX Reference Laboratories (North Grafton, Massachusetts) from Maine, New Hampshire, Massachusetts, Connecticut, and Rhode Island in the eastern United States. The method used to process these samples was as follows: approximately 2 g of feces from each fecal sample was mixed with 10–15 ml of 1.24 spg ZnSO4 and homogenized. The homogenate was strained and the liquid collected. The strained filtrate was poured into a 15-ml centrifuge tube, filled with additional ZnSO4 to within approximately 2 mm of the rim, and spun at 500 RCF for 5 min. Following centrifugation, the tube was carefully filled with more ZnSO4 solution until a small positive meniscus was formed. A coverslip was placed on the meniscus, allowed to stand for 10 min, and then removed to a microscope slide. Examination for parasite eggs was made at 100× magnification, and the number of eggs counted beneath the coverslip was reported as rare (1–2 eggs), few (3–10 eggs), moderate (11–30 eggs), or many (>30 eggs). A subset of 1,156 samples from the 4,240 canine samples contained enough fecal material for additional analysis.
A second fecal flotation was conducted on field samples obtained from the reference laboratory when a discordant ELISA coproantigen test result was obtained. For these, the Fecalyzer®b fecal analysis system was used per manufacturer’s specifications. Briefly, the fecal sample was collected in the green insert of the device, and NaNO3 (measured at 1.28 spg) was added to the level indicated on the chamber. The sample was mixed, and the insert was securely seated in the chamber before more flotation solution was added to form a positive meniscus. A 22-mm coverslip was added, and the preparation was allowed to stand for 15–20 min prior to examination under the microscope at 100× magnification.
Fecal extract preparation
One gram of fecal material was suspended in 4 ml of sample buffer (Tris buffer [pH 7.2] supplemented with Tween-20, 5% mouse serum, c 10% rabbit serum, d and 40% fetal bovine serum e ). Sample homogenization was accomplished by the addition of 4–5, 3-mm silica beads f followed by 2 min on a vortex. The fecal slurry was clarified by centrifugation at 17,441 RCF for 30 min. The fecal extract supernatant was collected by pipette and stored at 4°C for up to a week or frozen at –20°C.
Trichuris vulpis ELISA
Monoclonal antibodies (mAbs) were developed against recombinant expressed whipworm porin protein. Both capture and detection antibodies were immunoglobulin G kappa subtypes. Capture mAb coating of well plates was performed by first diluting the antibody in 10 mM phosphate buffered saline (PBS; pH 7.3) buffer to a concentration of 3 µg/ml. A 96-well microtiter ELISA plate g was then coated with 100 μl of diluted antibody per well, covered, and allowed to incubate overnight at 4°C. Coated plates were washed 3 times with 1× PBS plus 0.05% Tween-20 (PBST) on an automated plate washer. h Tris buffer i plus 1% bovine serum albumin j was prepared, and 300 μl was added to each well and incubated for 3 hr at room temperature as a blocking step. A 2.5% sucrose solution in 0.1 M Tris buffer (pH 7.0) was prepared, and 300 μl was added to each well and incubated for 3 hr at room temperature. Sucrose overcoated plates were tapped dry and stored at 4°C in sealed bags with 4 g of desiccant. Overcoated plates are stable for up to a year. Fecal extract samples (100 μl) were added to plate wells. The plate was covered and allowed to incubate for 1 hr at room temperature followed by a wash step with PBST. The wash step was repeated 4 additional times for a total of5 washes. Detection mAb (succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SMCC k ) horseradishperoxidase (HRPO l conjugate) was prepared at 3 μg/ml in sample buffer, and 100 μl was added to each well and incubated for 30 min at room temperature followed by 5 PBST wash steps. A 3,3′,5,5′-tetramethylbenzidine (TMB m ) solution containing 0.2 g/l TMB and 0.01% hydrogen peroxide was prepared, and 50 μl of substrate was added to each well and incubated for 10 min at room temperature. The color development was stopped by the addition of 50 μl of 0.1% sodium dodecyl sulfate solution to each well. Plates were read at a wavelength of 650 nm. An ELISA cutoff of 0.100 was established by adding 6 standard deviation values to the mean value of several hundred negative samples run in the assay (data not shown).
Preparation of polyclonal antibody resin
Polyclonal antibody n (pAb) conjugates were prepared for T. vulpis target-specific pAb and for a non–target-specific pAb following manufacturer’s instructions with a few modifications. Briefly, a total of 2 mg of each pAb at 1 mg/ml in PBS was reduced with 50 mM cyanoborohydrate o solution. The reduced pAb was mixed with 2 ml of resin n overnight at 4°C. Coupled resin was blocked with 1 M Tris buffer (pH 8.0), and washed with 1 M NaCl. The conjugated resin was resuspended in an equal volume of 10 mM PBS (pH 7.3). Conjugated resin was stored at 4°C.
Immunoprecipitation assay
An immunoprecipitation assay was developed to confirm that positive ELISA signal was in response to the presence of whipworm coproantigen rather than nonspecific signal. This is particularly important to determine for field dog samples. The pAb beads were prepared by pelleting 200 μl of resin and removing the supernatant. Target-specific and unrelated pAb resin was prepared for each fecal sample. The pAb beads were mixed with 260 μl of fecal extract and incubated at room temperature for 30 min with tumbling. The pAb bead complex was removed by centrifugation at 17,000 RCF for 3 min. Pretreatment of each sample with immunoprecipitation reagents resulted in 2 samples posttreatment: A, immunoprecipitated with unrelated pAb and B, immunoprecipitated with whipworm target-specific pAb. Treated samples A and B (100 μl each) were tested in the whipworm ELISA as described above. The immunoprecipitation assay was applied to samples from days 0, preinfection; day 2, prepatent, encysted small intestinal larval whipworms; day 48, prepatent, L4 infections; and day 84, patent, adult worm infections of the experimental infection for animal SEZ and all ELISA-positive field dog fecal samples.
Data analysis
The following formula was used to calculate the percent inhibition for the immunoprecipitation assay: percent inhibition = (1 – B/A)100, where A indicates the optical density at 650 nm (OD650) value for the unrelated pAb pretreatment and B indicates the OD650 value for the target-specific pAb pretreatment. A percent inhibition ≥50% was considered an indication of whipworm-specific antigen.
Results
Trichuris vulpis eggs were observed on fecal flotation in 3 of 5 experimentally infected animals starting on day 69, and from all 5 infected dogs by day 73 of infection and until treatment or necropsy on day 89 (Fig. 1A). Eggs per gram varied within and between individual animals, but no infected animal stopped shedding eggs over the time course of fecal collection prior to treatment. One animal, RQZ, had particularly low EPG counts (<100) throughout. The other 4 animals had EPG counts consistently above 100 prior to treatment. In comparison, whipworm-specific coproantigens were evidenced as elevated (>0.1 OD) ELISA signals starting as early as day 23 of infection; these remained elevated in all dogs through day 87 and for a period thereafter even following treatment (Fig. 1B). By day 38, all infected animals had robust ELISA signals, which remained elevated throughout the study, with the exception of animal KXZ on day 79; although even in that instance the ELISA signal remained elevated.
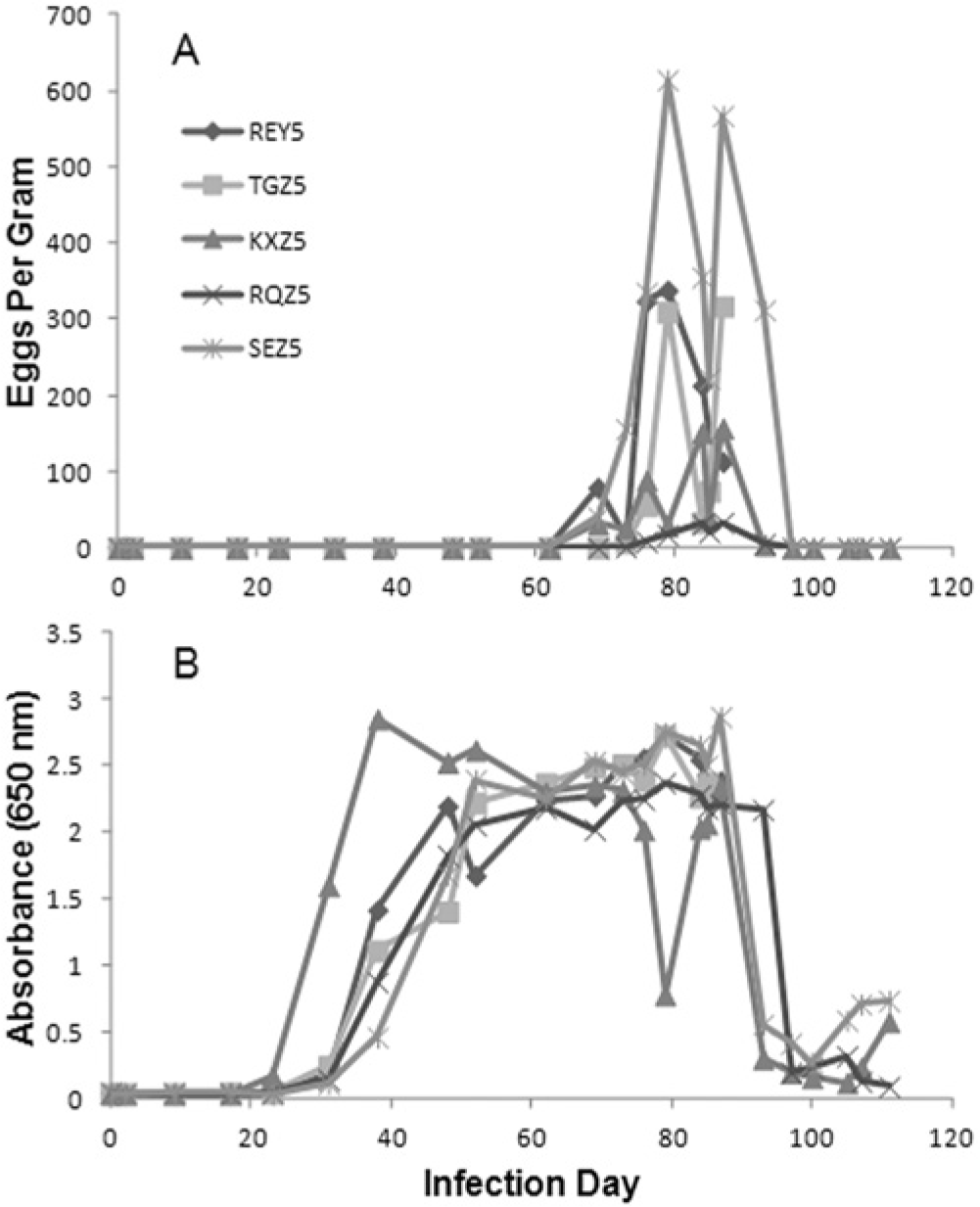
Time course results for 5 whipworm-infected dogs.
Three dogs were followed with fecal sample analysis after treatment (day 89), and only the sample collected 4 days after treatment for each dog (day 93) still contained detectable eggs. The coproantigen levels fell markedly in these 3 dogs by 8 days after treatment, and 1 dog (RQZ) ultimately dropped to negative status by the final day of collection, day 111 (Fig. 1B). Coproantigen levels from the other 2 treated dogs, KXZ and SEZ, fell to their lowest levels by 16 and 11 days after treatment, respectively, but the coproantigen levels then began to rise again.
In order to assess if the ELISA reactivity is specific to a T. vulpis–secreted antigen, an immunoprecipitation assay was conducted on selected days of the infection time course for animal SEZ. Four time points in the whipworm life cycle were assayed: day 0, uninfected; day 2, infected by no adult worms; day 48, prepatent infection; and day 84, patent infection. The elevated whipworm ELISA signals observed at days 48 and 84 were significantly reduced (>90%) by the target-specific treatment indicating the signal is not due to nonspecific binding of the antibody reagents.
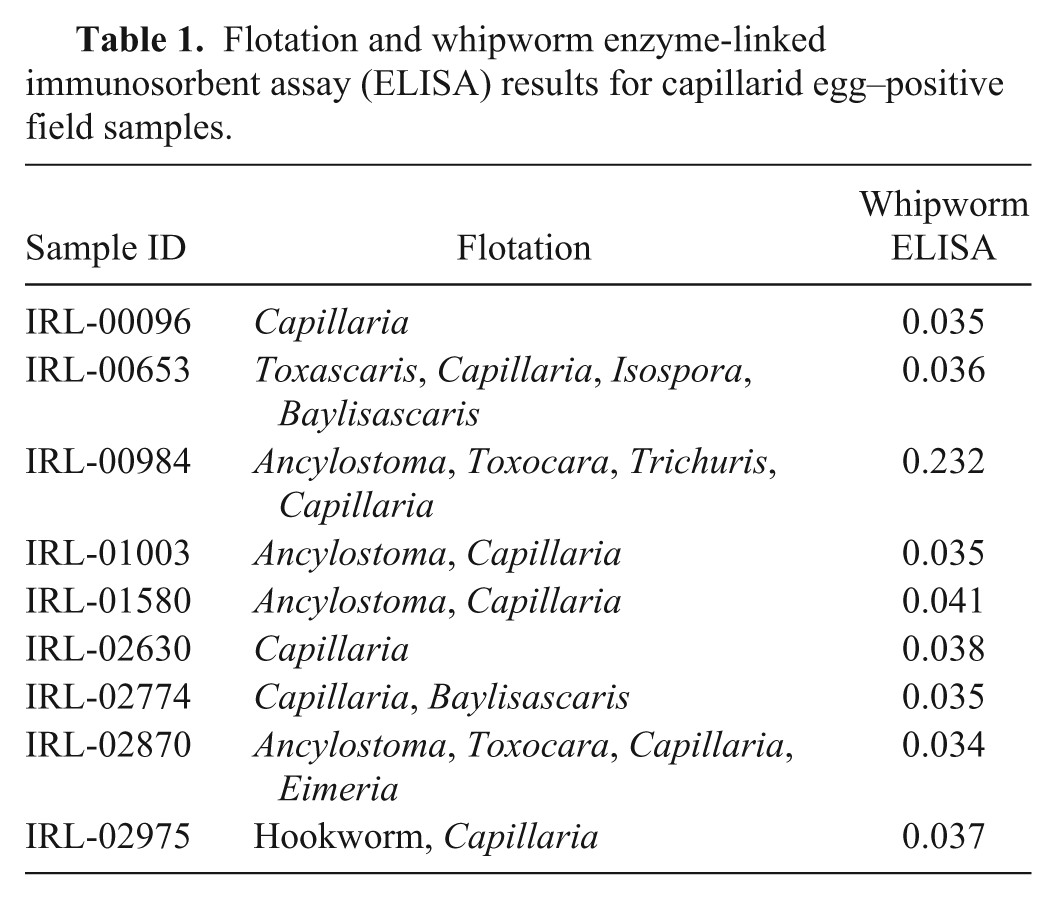
To further address the specificity of the assay, fecal samples from animals naturally infected with nematodes closely related to whipworms (i.e., capillarids) were tested and did not cross-react (i.e., did not result in elevated ODs) with this whipworm coproantigen ELISA (Table 1). One sample, IRL-00984, contained both capillarid and whipworm eggs. This sample was positive on the whipworm ELISA. Fecal samples from less closely related nematodes such as T. canis and A. caninum–infected animals also did not cross-react (data not shown).
Flotation and whipworm enzyme-linked immunosorbent assay (ELISA) results for capillarid egg–positive field samples.
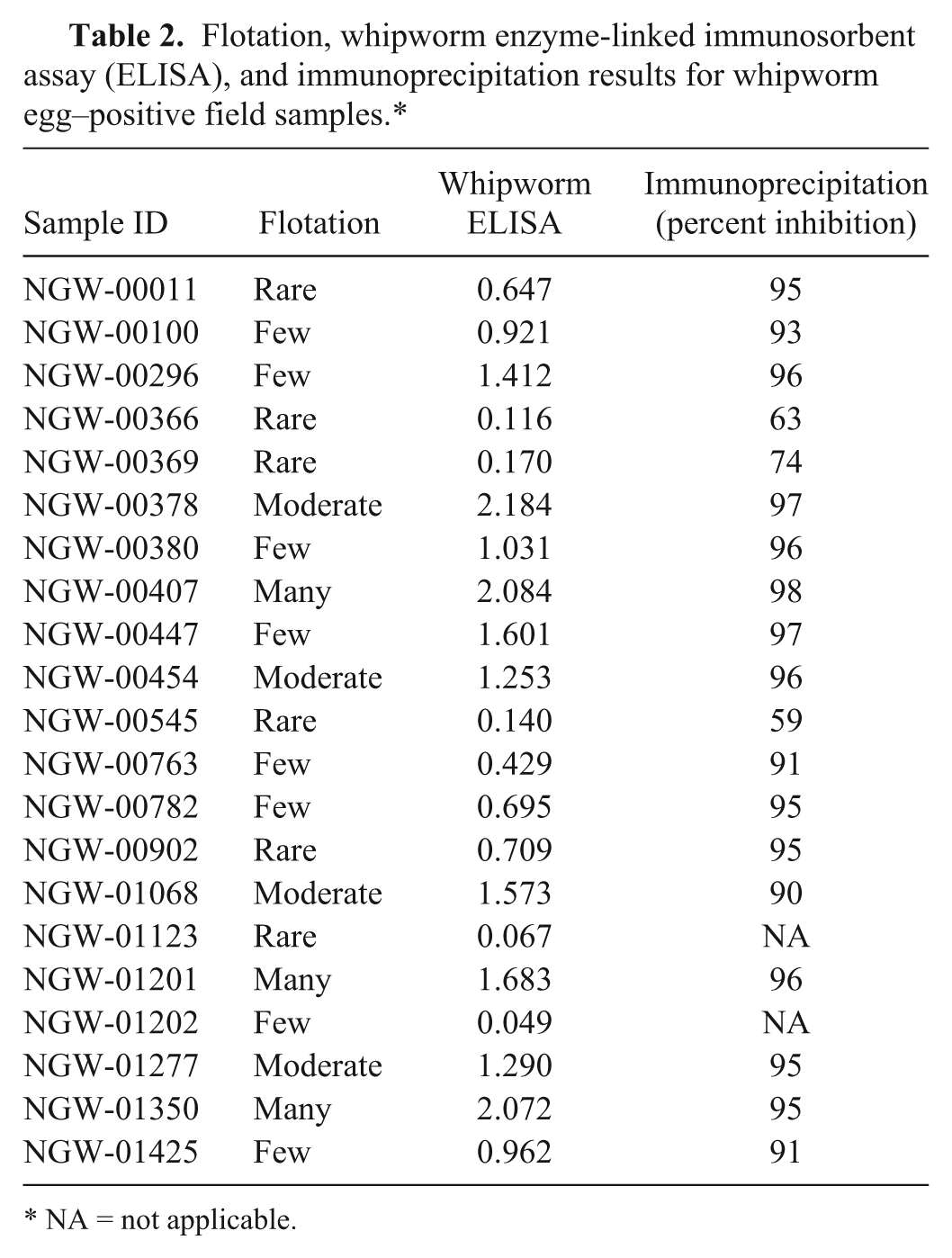
Semiquantitative egg flotation data from 4,240 canine fecal samples revealed 42 samples where whipworm eggs had been observed (0.99%). Of the 4,240 samples, 1,156 samples provided sufficient volume to allow further investigation; included therein were 21 of the original 42 reportedly whipworm egg–positive samples (Table 2). Out of these 21 samples, all but 2 were positive for antigen using the whipworm coproantigen ELISA, with signals ranging from 0.116 to greater than 2.0. On a second fecal flotation, one of these egg-positive, ELISA-negative samples, NGW-01123, was confirmed to contain T. vulpis eggs, while the other such sample, NGW-01202, was not. All egg-positive, ELISA-positive samples were confirmed in the immunoprecipitation assay, indicating detection of whipworm-specific antigen. Out of the remaining 1,135 apparently whipworm egg–negative samples, 16 were ELISA positive with signals ranging from 0.146 to greater than 1.0 (Table 3). A second fecal flotation assay on these 16 samples detected whipworm eggs in 6; all 16 samples were positive in the immunoprecipitation assay, confirming detection of whipworm-specific antigen.
Flotation, whipworm enzyme-linked immunosorbent assay (ELISA), and immunoprecipitation results for whipworm egg–positive field samples.*
NA = not applicable.
Flotation, whipworm enzyme-linked immunosorbent assay (ELISA), and immunoprecipitation results for whipworm egg–negative field samples that were positive for antigen detection using the whipworm ELISA.
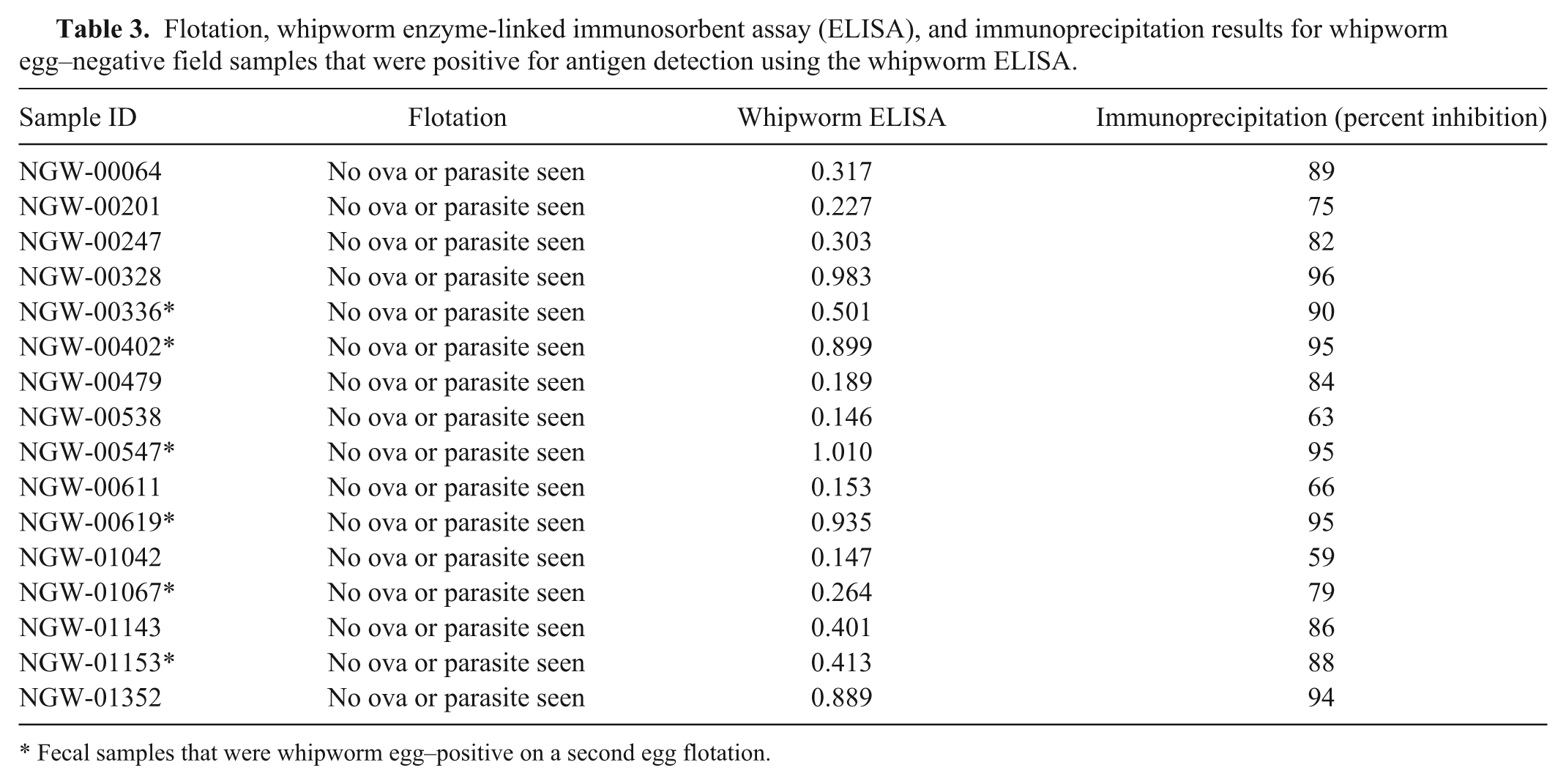
Fecal samples that were whipworm egg–positive on a second egg flotation.
Discussion
The ELISA described and utilized herein showed a very high correlation with submitted samples containing whipworm eggs diagnosed microscopically using the routine procedures of the IDEXX Reference Laboratories (i.e., 19/21 fecal samples containing T. vulpis eggs were considered positive by ELISA; Table 2). Also, of 1,135 samples apparently negative for whipworm eggs, 16 were ELISA-whipworm positive, and on a second flotation performed on these 16 samples, 6 were found to contain whipworm eggs on recheck. Out of 9 reference samples that were microscopically found to contain capillarid eggs, only the sample that also contained whipworm eggs tested whipworm positive using the ELISA (Table 1). As the test is utilized on larger numbers of field samples, increased data on sensitivity and specificity can be determined.
An immunoprecipitation assay was developed to confirm the ELISA was truly detecting whipworm-specific coproantigen. This is important to establish confidence in assay performance in field samples, where true whipworm infection status is not known, and particularly, when apparently egg-negative samples (as determined by fecal flotation procedures) produce elevated ELISA signals (i.e., samples with discordant egg detection and ELISA results). Immunoprecipitation is a common technique of precipitating a protein antigen with an antibody and has been widely used to investigate protein–protein interactions. In the present application, immunoprecipitation of the whipworm-specific coproantigen with a target-specific pAb depletes the antigen from the sample, whereas treatment with a non–target-specific pAb does not remove the whipworm coproantigen. Subsequent testing of a previously ELISA-positive sample results in a negative ELISA result only with the whipworm target-specific immunoprecipitation pretreatment. Fecal samples from the experimental whipworm infection established the validity of the immunoprecipitation assay confirming elevated ELISA signals are not the result of nonspecific binding.
Initially, a total of 18 discordant results were obtained from the 1,156 canine field samples acquired from the reference laboratory; 2 cases where eggs were seen initially, yet the ELISA provided a negative result (Table 2), and 16 cases where there were no eggs seen initially and antigen was detected by the ELISA (Table 3). Of the 2 ELISA-negative, egg-positive samples, in 1 case whipworm eggs were found when the sample was examined a second time (NGW-01123). This instance could reflect either a false-negative test or contamination with eggs from the environment (e.g., ingestion of an unembryonated, canine or other source, whipworm egg passing through the gastrointestinal tract, or fecal contamination of eggs from the environment into which the sample was deposited). In the second sample where an egg was initially detected but where no antigen was detected in the ELISA (NGW-01202), there were no eggs detected on a second flotation. In this case, again, the result could be a false negative, but it might also represent either a mistaken diagnosis during the original examination or again the presence of a few environmentally acquired eggs. In the case of the 16 antigen-positive, reportedly egg-negative samples, a second flotation recovered eggs from 6. It would seem highly likely that the remaining 10 represent either prepatent infections that are being diagnosed by the ELISA or samples with egg levels so low that 2 independent subsamples did not result in egg-positive flotation results. Immunoprecipitation of these 16 samples (Table 3) confirmed that the elevated signal was due to the presence of the whipworm coproantigen.
Although the microscopic examination of a fecal specimen for whipworm detection has the advantage that it allows the discovery of the perhaps unexpected coinfection, it has 2 potential downsides as well. One downside is caused by the not uncommon practices of coprophagia and ingestion of prey by dogs, and the other is by prepatent infections. People who routinely perform diagnostics on fecal specimens from dogs become aware that these samples can contain parasite eggs and cysts from exogenous sources. These ingested parasite stages simply pass through the dog’s intestine when ingested (e.g., unembryonated Toxocara cati eggs in a fairly fresh feline fecal or Trichuris discolor eggs in a fairly fresh bovine fecal). This pass-through phenomenon has been shown to be a quite common event with canine fecal samples where one third of apparent Toxocara infections in dogs, diagnosed through detection of eggs on fecal flotation, were established to be actually the ingested eggs of T. cati. 7 Dogs may also pass whipworm eggs of sheep, rabbits, mice, rats, and other mammals in their feces because, as carnivores, dogs ingest the bowels of these animals and any adult worms found therein. Antigen detection for helminths has the ability to decrease the period of prepatency by allowing an earlier detection of an infection, as is well known in the detection of the antigen of the heartworm, Dirofilaria immitis, in experimentally infected dogs. 3 In the case of whipworms, prepatency based on the recovery of eggs is 2.5–4 months, but the results from the work herein show that detection of infection with this method can occur as early as 3 weeks postinfection. The prepatent period for antigen detection is shorter than that for egg detection. Other advantages of fecal antigen detection are that laboratories may be using fecal diagnostic methods and solutions that are substandard for the recovery of T. vulpis eggs, and there may be instances where not all adult worms are removed by treatment, with the remaining worms shutting down egg production for some period before they recover and begin to again produce eggs.
The experimental infection of dogs with T. vulpis in the current study also allowed an evaluation of the various life-stages of whipworms in the host versus egg and worm-specific antigen detection in feces. The life cycle of T. vulpis in the dog is poorly described when compared with those of the dog roundworm and hookworm, T. canis and A. caninum, respectively. Whipworm first-stage larvae hatch from ingested infective eggs in the small intestine where they enter the crypts of Lieberkühn for 2–10 days before returning to the lumen and passing down the intestine to take up residence in the cecum and colon.11,13,15,20,25 In the case of T. vulpis, the timing of the 4 molts separating the various larval and adult stages has not been well established. It does appear that by 15 days postinfection, 70% of the worms are in the mucosa of the cecum and colon, and 5 days later, the infection is completely confined to this site. 12 At that time, the larvae are approximately 1 mm long; by 20–32 days, the larvae reach lengths of 1.2–5.4 mm; by 42 days, the larvae are 2.8–7.2 mm long and beginning to have the whip-like appearance with the wide posterior and thin anterior ends; and by 50 days after infection, male larvae are 26 mm long and female larvae 22 mm long.12,25 A 54-day-old female worm did not yet have a patent vulva, indicating this was still a fourth-stage larva, 25 an observation supported by later work reporting that fourth-stage larvae were present in dogs between 45 and 68 days postinfection (Opitz HM: 1963, Untersuchungen uber die Entwicklung des Peitschenwurmes des Hundes, Trichuris vulpis Frolich (1789) [Studies on the development of the dog whipworm, Trichuris vulpis Frolich (1789)]. Thesis. Berlin, Germany. In German). In experimental infections, the time to patency has been reported as occurring between 70 and 114 days after oral inoculation with eggs12,19,25; this prepatent period fits well with the work reported herein where eggs were observed in the feces of 3 of the 5 dogs 69 days after infection and in all dogs 73 days after infection. The coproantigen ELISA used in the current study became positive for the first time in the experimentally infected dogs using the samples collected 23 days after infection, whereas eggs were not evidenced in feces until day 69. Thus, this ELISA is capable of detecting an infection with pre-adult stages. This earliest detection point appears to correlate with the movement of larval worms from the small to large intestine. It is unfortunate that more is not known about the timing of the different molts within the life cycle of this parasite as the secretion of the antigen by the worms might be stage specific in its release. On the other hand, it may be such that coproantigen detection is totally based on the size of the worms, with larger worms shedding more antigen. In either case, in the current study, it appears that antigen detection correlates with the presence of worms, both pre-adults and adults, in the cecum and colon of infected dogs, and that these can be evidenced as little as 23 days postinfection.
The 3 dogs that were treated in the present study had no eggs in the feces 4 days after treatment, but there was reappearance of coproantigen in the fecal samples. These dogs were not necropsied, thus, it is unknown whether there may have been some worms left behind after treatment. In this case, it seems that the detected antigen was likely due to re-release by adults that were not killed, similar perhaps to how whipworms slowly begin to produce eggs that are normal again in morphology after anthelmintic treatment.2,21,26 Perhaps, if fecal samples had been collected over a longer period, the antigen would have continued to rise and ultimately, eggs would have reappeared in the feces.
The detection of coproantigens is accepted as a routine diagnostic procedure for many intestinal parasitic infections. The ELISA for Giardia coproantigen detection 23 is a test routinely used in veterinary medicine. Such coproantigen tests are also being developed and proving to be useful for the diagnosis of infections with the deadly zoonotic tapeworm Echinococcus multilocularis in dogs and foxes1,10 and the zoonotic tapeworm Echinococcus granulosus in dogs. 16 Also, although not approved for small animal use, there are Cryptosporidium assays approved for human medicine that are routinely applied to veterinary medicine. 17 Therefore, there is reason to believe that the newly developed ELISA whipworm test would be accepted by veterinarians as a useful adjunct or ultimately a replacement of microscopy for the diagnosis of whipworm infections in dogs.
Footnotes
Acknowledgements
The authors would like to thank Todd Bezold, Jen Cote, and Rita Hanna for assay expertise and discussion.
a.
Interceptor, Novartis Animal Health, Greensboro, NC.
b.
Fecalyzer, EVSCO Pharmaceuticals, Fort Worth, TX.
c.
Mouse serum, Equitech, Kerrville, TX.
d.
Rabbit serum, Pel-Freez, Rogers, AR.
e.
Fetal bovine serum, Life Technologies, Grand Island, NY.
f.
Silica beads (product 11-312A), Fisher Scientific, Pittsburg, PA.
g.
Immulon IB 96 well plates (product 6310), Thermo Electron Corporation, Milford, MA.
h.
ELx405 MicroPlate, BioTek Instruments Inc., Winooski, VT.
i.
Trizma hydrochloride buffer solution (pH 8.0, product T2694-1L), Sigma-Aldrich, St. Louis, MO.
j.
Bovine serum albumin (product 68020), Proliant Inc., Ames, IA.
k.
Succinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (product P122322), ThermoFisher Scientific, Waltham, MA.
l.
Horse radish peroxidase (product 10815462103), Roche Molecular Biochemicals, Mannheim, Germany.
m.
3,3′,5,5′-tetramethylbenzidine (product TM102-91000-128), SeraCare Live Science, Milford, MA.
n.
AminoLink plus coupling resin (product 20501), ThermoFisher Scientific, Waltham, MA.
o.
AminoLink reductant (product 44892), ThermoFisher Scientific, Waltham, MA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
L. Cruthers performed experimental studies as a fee for service funded by IDEXX. D.A. Elsemore, J. Geng, and L. Flynn were funded by IDEXX.
