Abstract
Infection due to Entamoeba spp. is known to cause serious disease in primates (Entamoeba histolytica) and snakes (Entamoeba invadens), but there are no detailed descriptions of the pathology associated with Entamoeba spp. infection in amphibians. In 2014, an outbreak of entamoebiasis associated with a novel species of Entamoeba induced clinical illness and poor body condition in free-ranging cane toads in Australia’s Northern Territory. Here, we describe the gross pathology, histology, and clinical pathology linked to the outbreak. The study compared 25 toads with invasive entamoebiasis, defined as histologically visible amoebas within tissue, and 12 toads without invasive entamoebiasis. Grossly, affected toads had mild to marked congestion of colonic serosal vasculature, with variable thickening of the intestinal wall and serosanguineous to hemorrhagic colonic content. Histologically, invasive entamoebiasis manifested primarily as moderate to severe, variably hyperplastic to ulcerative colitis. The small intestine was affected in 10 of 25 toads, and 5 of 25 toads also had gastric lesions. Amoebas consistent in morphology with Entamoeba sp. were commonly intermingled with mucosal epithelium, frequently along the basement membrane, with deeper invasion into the superficial lamina propria in only 5 toads. Toads with invasive entamoebiasis had neutrophilia, monocytosis, and lymphopenia, and thus elevated neutrophil to lymphocyte ratios, suggestive of an inflammatory and/or stress leukogram.
The genus Entamoeba contains species that inhabit a range of invertebrates and vertebrates, often as commensals in the intestinal tract and less commonly as pathogens. 13,35,46 The most notorious species, Entamoeba histolytica, the agent of human amoebic dysentery, causes significant morbidity and mortality in the Third World. 25,43 Another well-known potentially pathogenic species is Entamoeba invadens of captive reptiles. 29,41 The association of E. invadens with disease varies; for example, in snakes, it may be innocuous or cause severe necrotizing enterohepatitis but is generally a commensal in turtles. 18,21,23,32,40 In captive amphibians, infection with Entamoeba ranarum, a species closely related to E. invadens, 13,46 is rarely associated with intestinal or hepatic disease, and a detailed description of any associated pathology is lacking. 15,21,36,37,55
Cane toads (Rhinella marina) are an invasive species that were introduced into Queensland, Australia, in 1935 and have since expanded their range northwest, arriving in Darwin, Northern Territory, in 2005. In 2014, researchers noticed dead and sick cane toads at their research facility near Darwin. Sick toads had invasive entamoebiasis, which we defined as histologically evident colitis with intralesional amoebas. The amoebas were consistent in morphology with Entamoeba sp. Using both next-generation sequencing and sequencing of Entamoeba-specific primers, a novel species of Entamoeba was detected in all affected toads. The same Entamoeba was present in most clinically normal toads, but toads that were sicker and in lower body condition exhibited increased histological severity of colonic entamoebiasis. 45 Here we describe in detail the pathology of entamoebiasis associated with this outbreak.
Methods
Cane toads in this study were collected from sites 60 km southeast of Darwin in Australia’s Northern Territory. The local climate is “wet-dry tropical” characterized by high temperatures year-round (average monthly maxima above 30°C in every month), with high rainfall during a 6-month wet season (November to April) but little or no rainfall during the 6-month dry season (May to October). Toads in this study include 22 toads collected during the outbreak. The outbreak occurred in free-ranging toads in the anthropogenic landscape surrounding a research station in the dry season over September to October 2014 (group A, toad Nos. 1–22). We also examined 2 further groups for comparison. The first comparison group was 7 toads from a wetland 30 km distant from the outbreak site, sampled 1 month after the outbreak (group B, toad Nos. 23–29). This group was selected because they inhabited a more natural setting compared to the surrounds of the research station. While a few of the toads from this site were deemed to be clinically ill and/or in poor body condition (Table 1), the population at the site was not obviously undergoing a disease outbreak. This group was essentially an attempt to closely scrutinize the colons of “normal” control toads. The second comparison group was 8 toads from the outbreak site, sampled 4 months later in the wet season. During the wet season, toads are less stressed (hydrically and energetically) and at lower densities. 7 Although a few of these toads were deemed clinically ill (Table 1), we were no longer finding dead or moribund toads surrounding the research facility and judged the outbreak to be over (group C, toad Nos. 30–37) (Table 1). As previously reported, Illumina (San Diego, CA) 18S ribosomal DNA (rDNA) amplicon environmental sequencing to identify the community of eukaryotes was performed separately on colon scrapings from 18 of these toads. 45 Testing was performed on 5 toads in each of groups A and B and all 8 toads in group C (Supp. Table S1). The purpose of molecular testing of group A toads was to speciate the Entamoeba that we could see histologically. Molecular testing of a subsample of toads from groups B and C was to use a more sensitive means than histology to determine if Entamoeba was present in populations we deemed to be “normal” control toads, compared to those in the outbreak. Among the 18 samples, 3 operational taxonomic units (OTUs) demonstrated perfect or high-percentage identity with small subunit (SSU) rDNA sequences of amoebas of the genus Entamoeba. One of these OTUs, a novel Entamoeba sp., was present in all toads except toad No. 30 and was the only Entamoeba sp. detected in all toads with histologically diagnosed invasive entamoebiasis. 45
Epidemiological, Clinical, and Pathological Summary of Toads in the Study by Group.
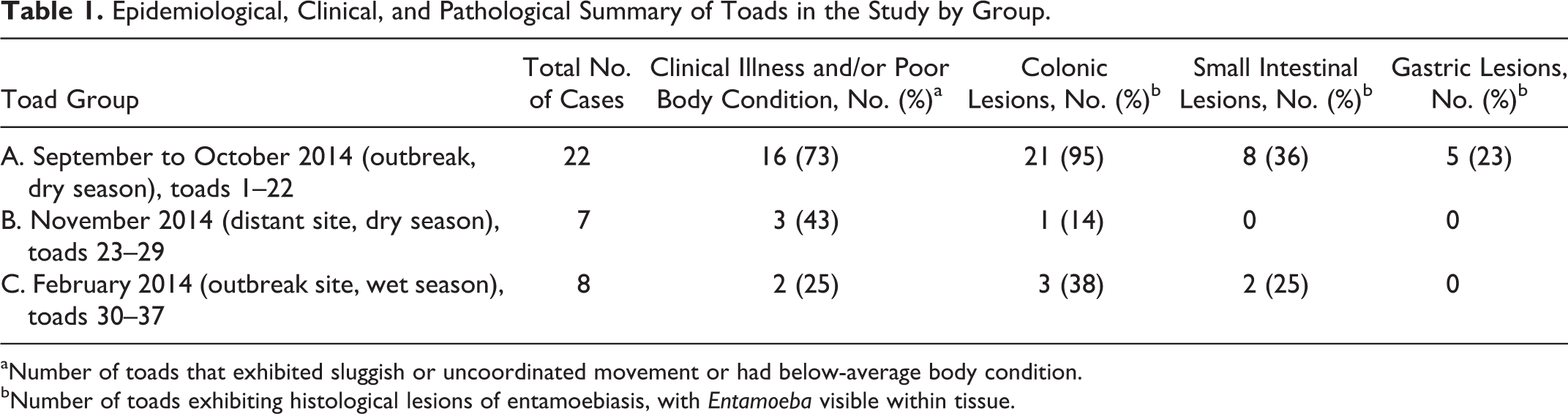
aNumber of toads that exhibited sluggish or uncoordinated movement or had below-average body condition.
bNumber of toads exhibiting histological lesions of entamoebiasis, with Entamoeba visible within tissue.
We collected all toads live. We assessed demeanor as normal or abnormal (sluggish response or uncoordinated movement), weighed them, and measured snout-urostyle length. Toads were classified as juvenile (snout-urostyle length <6 cm), subadult (snout-urostyle length 6–8 cm), or adult (snout-urostyle length >8 cm). Sex was determined by gross examination of the gonads. Toads were assessed as clinically ill if their demeanor was abnormal or body condition score was below average (see statistics section). We euthanized toads by overdose (30 mg/100 g body weight) of pentobarbital sodium diluted 50% with water injected into the dorsal lymph sac (Lethabarb, 325 mg/ml; Virbac Australia Pty. Limited, Milperra, NSW, Australia). This resulted in death after a period of 5 to 10 minutes of gradually deepening anesthesia, during which time 1 ml of blood was collected from the heart. Blood was collected using syringes rinsed with lithium heparin and was used for hematology and clinical chemistry. Toad handling, blood sampling, and euthanasia were approved by the University of Sydney Animal Ethics Committee Protocol 2013/5805.
A full necropsy including gross examination of all internal organs was performed on all 37 toads. Samples for histological examination of the stomach and small and large intestine were taken from all toads. In the first 11 cases from the outbreak, we also sampled liver, kidney, spleen, heart, lung, brain, and skin to investigate the range of tissues affected and to look for other conditions that may have been involved in the outbreak. Tissues for histology were preserved in 10% phosphate-buffered formalin, processed in standard fashion, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Although amoebas were typically readily visible with standard HE staining, periodic acid–Schiff reaction (PAS), which stains the glycogen-laden cytoplasm of Entamoeba spp., 29 was used in selected cases to corroborate the presence or absence of organisms in ulcers or disorganized epithelium (toad Nos. 1, 5, 6, 8, 16, 37). Mallory’s phosphotungstic acid hematoxylin was used to enhance nuclear morphology of amoebas 38 in toad No. 16, which had particularly abundant intraepithelial amoebas. Masson’s trichrome stain was used to demonstrate fibrosis of the lamina propria and submucosa in toad Nos. 16 and 37. 16 We defined invasive entamoebiasis based on histological presence of amoebas consistent in morphology with Entamoeba spp. within tissue. We categorized severity of gastrointestinal entamoebiasis based on presence and extent of ulceration: in mild cases, ulceration was not present; in moderate cases, there was multifocal ulceration; and in marked cases, ulceration was extensive.
Hematology was performed in all toads. Thin smears were prepared from blood drawn through cardiac puncture of anesthetized toads and stained (Diff Quik; Lab Aids Pty Ltd, Narrabeen, NSW, Australia), cover-slipped, and scanned in a zigzag pattern at 1000×. The first 100 white blood cells (WBCs) encountered were identified as neutrophils, lymphocytes, monocytes, eosinophils, or basophils. While there is variation in the literature regarding nomenclature for amphibian granulocytes, since the cytoplasmic granules in cane toad neutrophils are inconspicuous and stain pale pink, we used the term neutrophils rather than heterophils. 10,11,36 The numbers of WBCs per 1000 red blood cells (RBCs) in each blood smear were also counted to serve as an approximate measure of absolute number of WBCs. 10,42 Packed cell volume was measured in all toads except toad Nos. 3 and 13, in which the microhematocrit tubes broke during centrifugation. Lithium heparinized plasma was used to measure selected biochemical analytes. For the first 8 clinically ill toads with invasive entamoebiasis (toad Nos. 1–8), we used a benchtop analyzer with prepackaged rotors that included tests for plasma potassium, sodium, glucose, calcium, phosphorus, aspartate aminotransferase, creatine phosphokinase, total protein, albumin, globulin, bile acids, and uric acid (VetScan, Abaxis, Dunnington, UK). Many analytes were below the limit of detection for these first 8 toads using the VetScan system, and results that we did obtain did not seem obviously abnormal (see Results). Therefore, for the remainder of the cases (except toad No. 11, in which insufficient plasma was available for analysis), biochemical testing was reduced to include only total protein, albumin, and urea, measured on an automated clinical biochemistry analyzer according to the manufacturer’s directions (Konelab, Thermo Electron Corporation, Ratastie, Finland). Globulins were estimated by subtraction of albumin from total plasma protein. These 4 analytes were selected since severe inflammatory intestinal disease and dehydration in mammals are often associated with loss of protein (decreased total protein and albumin), elevated globulins, and elevated urea. 1,33,50
A body condition index was calculated using residuals from a linear regression of body mass on snout-urostyle length (after ln-transforming both variables). Toads were judged to be in below-average body condition if this score was negative. We performed Kruskal-Wallace nonparametric tests to compare clinical pathology measurements of toads with and without invasive entamoebiasis. We did not adjust significance levels for multiple comparisons. All statistics were performed using JMP11 (SAS Institute, Cary, NC).
Results
We collected a total of 37 toads, of which 21 were clinically ill and 25 had histological invasive entamoebiasis (21/22 in group A, 1/7 in group B, 2/8 in group C). Sixteen of the clinically ill toads and 21 of the toads with invasive entamoebiasis were collected during the outbreak (Table 1, Supplemental Table S1). Ten toads were subadults and the rest were adults (Suppl. Table S1). Eighteen toads were female, 17 were male, and 2 were of unknown sex (Suppl. Table S1). There was no obvious age or sex predominance of either clinical illness or presence of invasive entamoebiasis.
Gross Pathology of Entamoebiasis
The intestinal tract of cane toads is short and simple and, apart from a widening of the colon 2 to 3 cm from the rectum, does not exhibit gross features distinguishing the various segments (Fig. 1). 5,14 In toads without invasive entamoebiasis, or toads with mild invasive disease, the small intestinal and colonic walls were pale tan, with inconspicuous mesenteric and serosal vasculature. When empty, the colon contained a small amount of watery fluid. The wall was thin, moderately transparent, and flaccid when cut, with a pale tan, smooth, shiny mucosa (Fig. 1). Toads with invasive disease often did not exhibit appreciable gross lesions beyond prominence of mesenteric and serosal vasculature (Fig. 2). In some cases, the intestinal wall was entirely or segmentally thickened and stiff, with serosanguineous or hemorrhagic content (Figs. 3, 4). Lesions in the small intestine were rarely sufficiently severe to be appreciable grossly (Fig. 4), and gastric amoebiasis was not noted on gross examination.
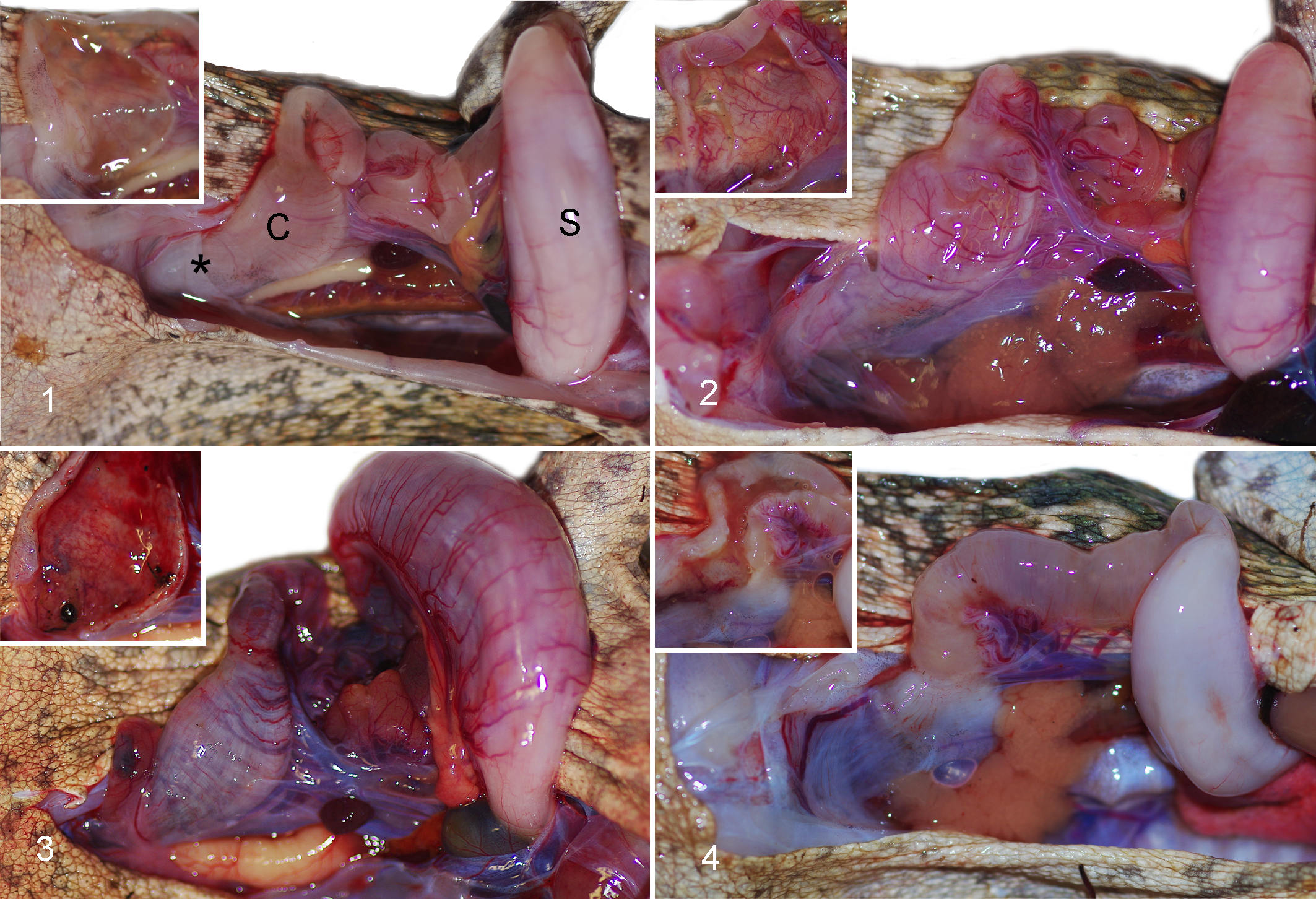
Gastrointestinal tract, cane toads.
Histopathology of Entamoebiasis
Intestinal lesions varied from mild to marked among the 25 toads with invasive entamoebiasis and frequently varied in severity among different intestinal segments within individual toads (Suppl. Table S1). Lesions were always present and most severe in colon, in many cases commencing in the region of gradual transition of distal small intestine to the colon and abruptly ending at the junction of colon with rectum. In 10 toads with moderate to severe colonic lesions, generally milder lesions extended cranially in the small intestine to the level of mid-intestine or, rarely, proximal small intestine (Suppl. Table S1). In relatively mild lesions, epithelium was multifocally disorganized and hyperplastic (Figs. 5–8). There were intraluminal, partially sloughed segments of hyperplastic epithelium at edges of eroded or ulcerated areas (Figs. 7, 8). In some lesions, presumably previously eroded or ulcerated areas, mucosal epithelium was attenuated or composed of cuboidal cells and there was marked remodeling of underlying lamina propria and submucosa (Figs. 9–12). In areas of ulceration, there was edema and fibrosis of the lamina propria that sometimes extended through the submucosa, generalized vascular congestion of the intestinal wall, and hyperplasia of serosal mesothelium (Figs. 13, 16). Ulcers extending deep into the lamina propria or submucosa were present focally in 3 toads and extensively in 1 (Suppl. Table S1). Deep ulcer beds were covered by fibrin and necrotic cell debris, and there was hemorrhage and thrombosis in the underlying lamina propria and/or submucosa (Figs. 15, 16). Throughout lesions, there was a moderate to marked lymphohistiocytic inflammatory infiltrate within the lamina propria and submucosa, with increased numbers of neutrophils in necrotic or ulcerated regions. In all cases, amoebas were present within hyperplastic or sloughing epithelium at the edges of erosions or ulcers, often aligned beneath the basal layer of epithelium (Figs. 6, 8, 10, 12, 14). In extensively ulcerated mucosa, amoebas were usually present only in association with scant remaining mucosal epithelium (Fig. 17). Amoebas were present focally in the lamina propria in 5 toads, usually underlying ulcers (Suppl. Table S1, Fig. 15). By our subjective assessment of severity based on histological presence and extent of ulceration, all of the toads with severe colon lesions were in the initial outbreak group (Suppl. Table S1). Of note is the disparity in some toads between gross appearance of the colon and histological assessment of severity. For example, toad No. 19 was judged histologically to have marked colonic lesions due to extensive ulceration, but grossly, the only notable feature of the colon was prominent vasculature (Fig. 2). By comparison, toad No. 18 was judged to have relatively moderate lesions histologically, based on multifocal rather than extensive ulceration, yet the colon wall was grossly relatively thick and reddened compared to toad No. 19 (Fig. 3).
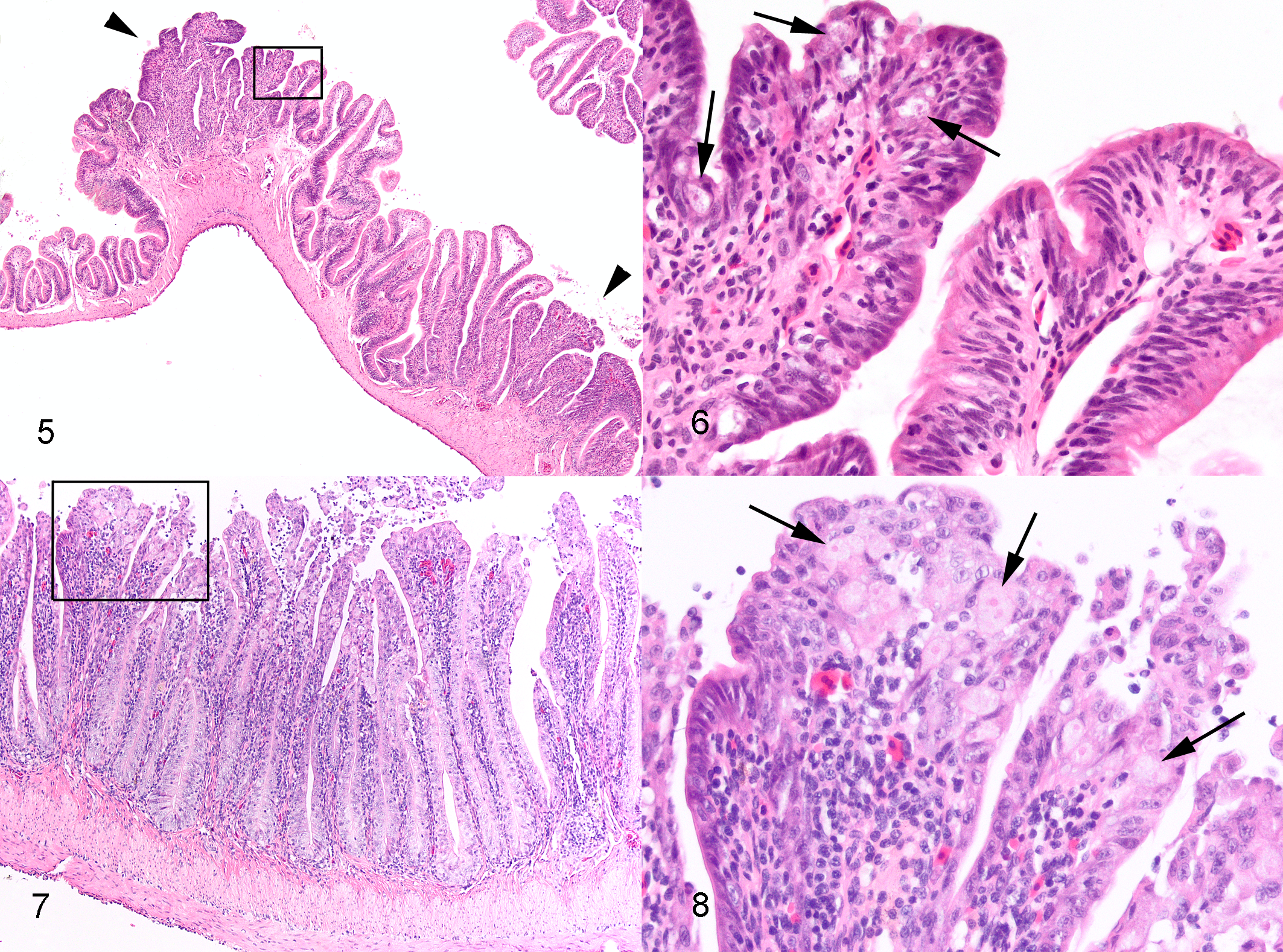
Invasive entamoebiasis, intestine, cane toads. Hematoxylin and eosin (HE).
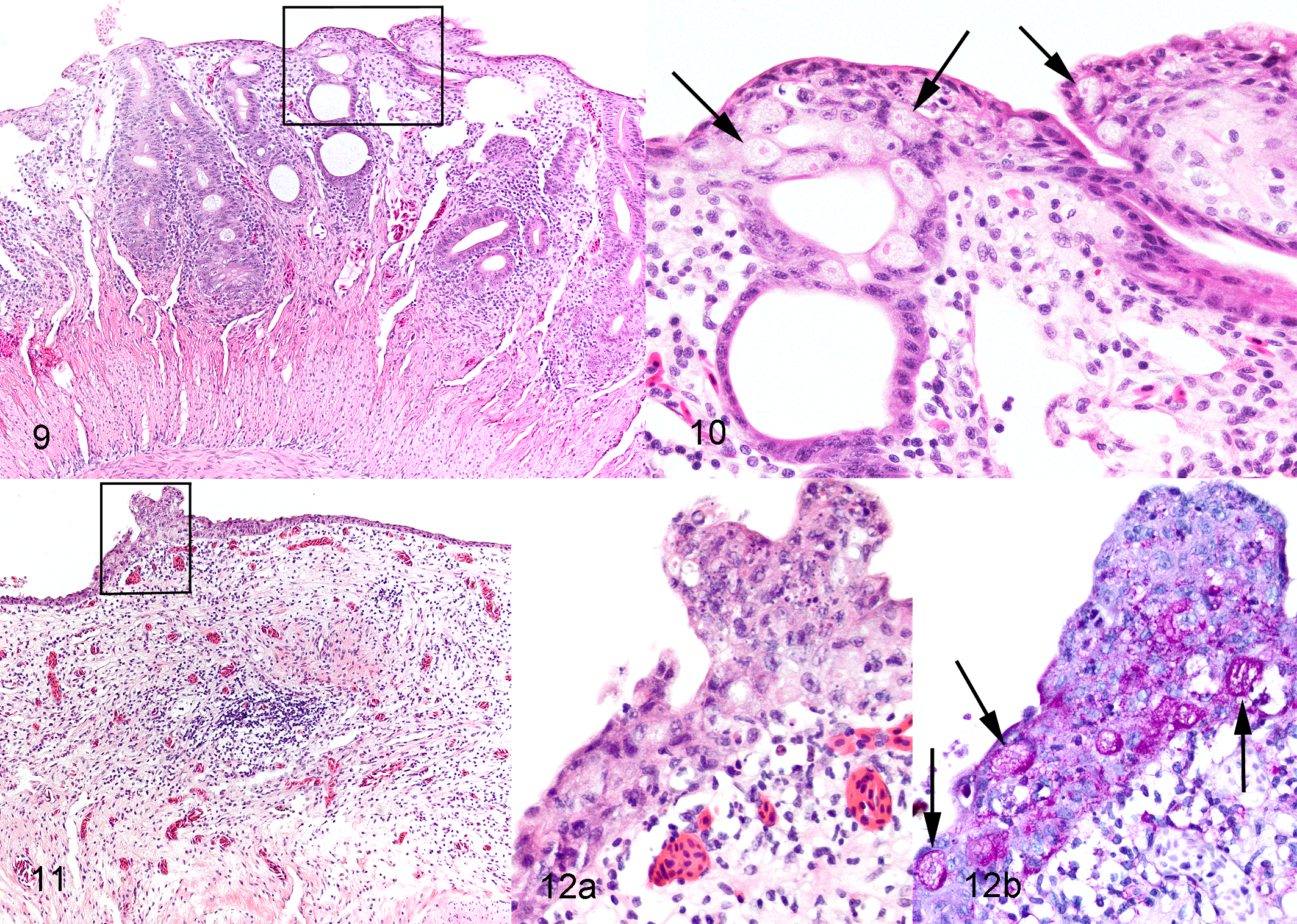
Invasive entamoebiasis, colon, cane toads.
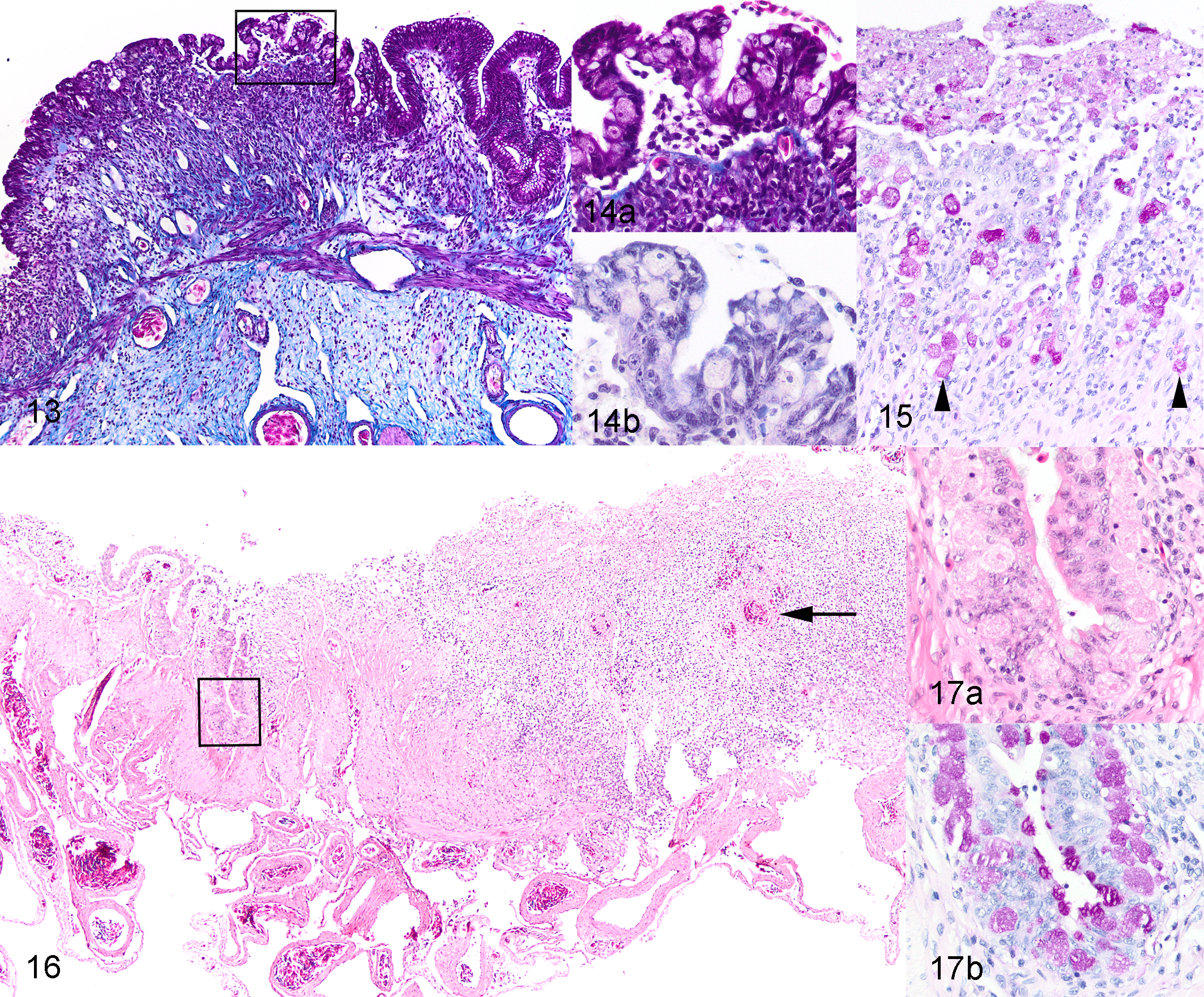
Invasive entamoebiasis, colon, cane toads.
We recorded gastric entamoebiasis in 5 toads, all from the outbreak, and all with concurrent small intestinal entamoebiasis and moderate to severe colonic entamoebiasis (Table 1, Suppl. Table S1). Lesions in the stomach were multifocal and characterized by mild to moderate disorganization of the surface and gastric pit epithelium with intraepithelial amoebas and mild to moderate associated lymphohistiocytic infiltration of the lamina propria (Figs. 18–20). There was no gross or histological evidence of spread of amoebas beyond the gastrointestinal tract in the 11 toads from early in the outbreak, in which a wider range of tissues was examined histologically. Of note is that these toads were all clinically ill (often severely), and all but 1 had severe lesions of invasive entamoebiasis involving the intestinal tract (Suppl. Table S1). Thus, if lesions of entamoebiasis were to occur in other organs, these cases were well selected to detect it.
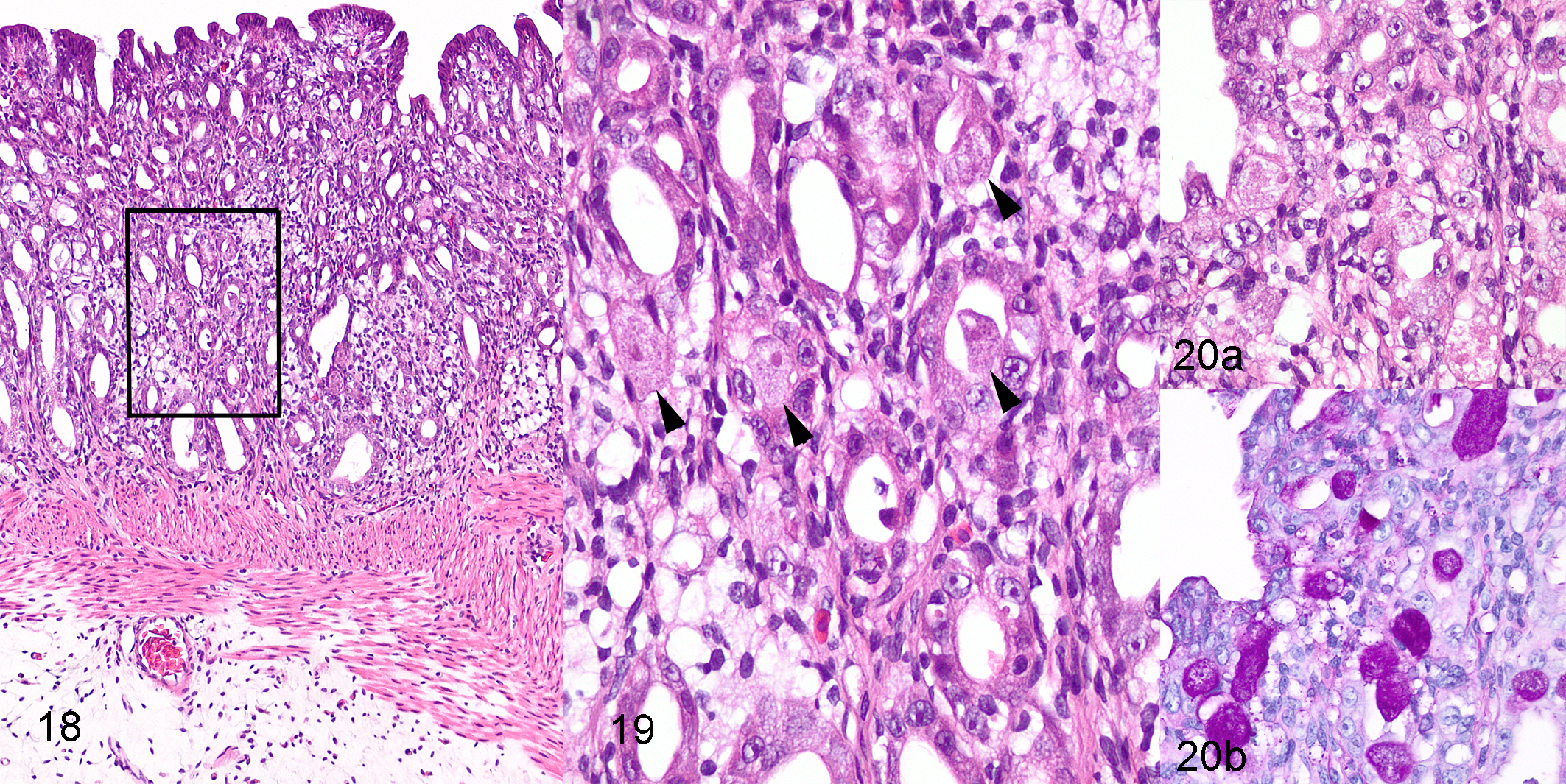
Invasive entamoebiasis, stomach, cane toad, toad 1.
Histologically, amoebas were detected in only a single form and were similar in all toads. Amoebas were ovoid, with a mean diameter of 14 μm (range, 9–23) and mean length of 18 μm (range, 11–28) (based on measurement of 30 intraepithelial amoebas in 3 toads). Amoebas had abundant finely granular cytoplasm that stained pale eosinophilic with HE and pale magenta with PAS (Figs. 12b, 15, 17b, 20b). Nuclei were small and uniform in size, with mean diameter of 3.4 μm (range, 3–4), central to eccentric, usually single but occasionally double in larger amoebas. Nuclei were vesicular, with a dense rim of peripheral chromatin and a single, small, central karyosome (Fig. 14b). These features are consistent with the histomorphology of Entamoeba spp. trophozoites. 2,22,23
Clinical Pathology
There was no significant difference in number of WBCs per 1000 RBCs between toads with and without invasive entamoebiasis (Table 2). Although this measure of absolute number of WBCs is likely less accurate than WBC counts by hemocytometry, it provides a reasonable estimate when the packed cell volumes are within normal limits or, as in our study, similar between 2 comparison groups. 10,42 Given similar absolute numbers of WBCs, toads with invasive entamoebiasis exhibited marked neutrophilia and moderate lymphopenia relative to those without invasive disease. Toads with invasive entamoebiasis also had a significant monocytosis compared to those without (Table 2). None of the plasma biochemistry analytes were significantly different between toads with and without invasive entamoebiasis (Table 2). Values of additional analytes for 8 toads with invasive entamoebiasis were (mean ± standard deviation) as follows: potassium, 3.2 ± 0.5 mmol/l; glucose, 3.1 ± 1.4 mmol/l; calcium, 1.6 ± 0.4 mmol/l; phosphorus, 0.7 ± 0.1 mmol/l; aspartate aminotransferase, 181.0 ± 97.0 U/l; and creatinine kinase, 780.9 ± 601.8 U/l. Using the VetScan method of analysis, except for total protein values in a few of the toads, other analytes for these 8 toads (plasma sodium, bile acids, uric acid, albumin, and globulin) were below the limits of detection, and therefore VetScan values for these analytes are not reported.
Hematology and Plasma Biochemistry of Cane Toads With and Without Invasive Entamoebiasis.a
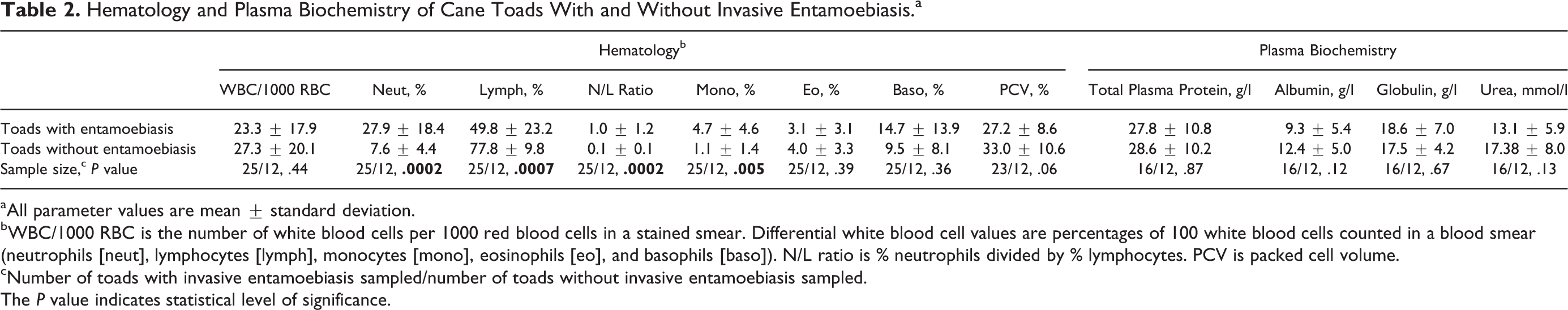
aAll parameter values are mean ± standard deviation.
bWBC/1000 RBC is the number of white blood cells per 1000 red blood cells in a stained smear. Differential white blood cell values are percentages of 100 white blood cells counted in a blood smear (neutrophils [neut], lymphocytes [lymph], monocytes [mono], eosinophils [eo], and basophils [baso]). N/L ratio is % neutrophils divided by % lymphocytes. PCV is packed cell volume.
cNumber of toads with invasive entamoebiasis sampled/number of toads without invasive entamoebiasis sampled.
The P value indicates statistical level of significance.
Other Diseases
Other diseases detected in the study toads (either by gross or histological examination) were fungal infection, presumptively caused by Mucor spp. (mucormycosis); spinal arthropathy; lungworm infection; and healed distal limb amputations (Suppl. Table S1). Presumptive mucormycosis was the most common concurrent disease and affected the skin and/or parenchymatous organs and/or gastrointestinal tract, being present in 7 toads with concurrent invasive entamoebiasis. Grossly, severe disseminated disease was evident in 2 toads and was characterized by multifocal ulcerative granulomatous lesions in the skin and discrete white granulomas in numerous parenchymatous organs. In the 5 other affected toads, presumptive mucormycosis was not appreciable grossly but was evident histologically as a few small granulomas associated with PAS-positive multiloculated fungal spherules in the liver or lamina propria of the stomach or intestine. Although we did not perform specific testing such as fungal culture or molecular diagnostics to confirm the etiologic agent, these typical features are consistent with other descriptions of mucormycosis in cane toads in Australia. 47 Spinal arthropathy was diagnosed grossly in 3 toads with concurrent invasive entamoebiasis. Grossly, affected toads had 1 or 2 enlarged or collapsed and hemorrhagic intervertebral joints, consistent with previous descriptions of the condition in cane toads. 44 Mild (up to 5 lungworms per lung) to moderate (5–10 lungworms per lung) parasitism was noted grossly in 23 of the 25 toads with invasive entamoebiasis and 8 of the 12 toads without invasive entamoebiasis. Grossly, parasite morphology was consistent with Rhabdias spp. lungworms, which are common in cane toads. These parasites are generally not associated with negative consequences in terms of survival or body condition 6 except under extreme environmental conditions. 20
Discussion
Previously, environmental 18S rDNA amplicon sequencing and species-specific PCR were used to identify the presence of a novel species of Entamoeba in colon scrapings from 17 of the 18 toads from this study that were tested. 45 The new species was closely related to but distinct from E. ranarum, which typically affects captive amphibians, 21,36,37,55 and E. invadens, which infects reptiles. 29,41
Eight of the toads in which the novel Entamoeba sp. DNA was detected had invasive entamoebiasis (8/17); however, the concentration of DNA for the novel Entamoeba sp. was directly proportional (P < .05) with lesion severity. 45 Previously published results also demonstrated a significant negative correlation between colonic amoebiasis severity and body condition and a positive correlation with clinical illness score. 45 The current study complements these previous investigations by presenting detailed gross, histological, and clinical pathological findings associated with invasive entamoebiasis in wild cane toads. Our data demonstrate a close histological link between invasive parasites and gastrointestinal lesions, lack of amoebas visible within normal tissue, and no other disease agents consistently associated with gastrointestinal pathology in the toads. Although histomorphology is not definitive in identifying amoebas to species level, the histomorphology of the amoebas in the toads is consistent with that of members of the genus Entamoeba. 2,22,23,29,35 The histology presented in this case series therefore supports the previous inference that a novel Entamoeba sp. is the main cause of the outbreak of severe clinical disease, poor body condition, and probable mortality in cane toads. 45 We did examine several toads that were found dead at the site during the outbreak, grossly and histologically, and this led to a suspicion of enteritis. However, under tropical conditions, even in toads found only a few hours after death, the intestinal tract decomposed too rapidly for accurate evaluation. Thus, we necessarily had to limit documentation of the pathology of toads during the outbreak to those found sick at the site and immediately euthanized.
Histopathologically, the lesions in toads mainly involved the mucosal epithelium and included mild to marked hyperplasia, attenuation, superficial ulceration, and loss of folds and crypts. Mild superficial epithelial changes have been described in early cases of human entamoebiasis diagnosed through rectal biopsies (rather than autopsy cases). 38 In these relatively mild human lesions, amoebas are sometimes seen directly on the basement membrane, separating it from the damaged surface epithelium. This undermining of the epithelium, causing it to slough, is also a feature of early or mild lesions of entamoebiasis in monkeys. 4 The same pathology was seen in the toad lesions. Likewise, in experimental infection in reptiles, early lesions included superficial areas of lytic necrosis, amoebas intermingled with the necrotic cells, and hyperplasia and sloughing of adjacent epithelium. 40 The limited extent and decreased severity of lesions in the small intestine in toads are similar to the situation seen in natural infections with entamoebiasis in snakes. 40,41
Although intestinal ulceration was a feature of the toad entamoebiasis lesions, ulcerations in most cases were notably more superficial than those seen in humans and reptiles. Further, in toads, amoebas were rarely observed within ulcers or necrotic intestinal wall. In humans, lesions may progress to deep, flask-shaped ulcers in the submucosa that are associated with masses of amoebas within the necrotic wall. 25,30,48 In reptiles, the typical lesion is a deeply ulcerative, necrotizing diphtheritic colitis with surface exudate so copious as to sometimes cause near occlusion of the intestine. 18,29,40 More deeply ulcerative and necrotizing lesions might have developed in the toads given time (if they had not been euthanized). However, many affected toads were very weak or moribund at the time of euthanasia, suggesting that death was imminent.
Another feature of the toad amoebiasis was lack of spread to other organs, particularly the liver, as occasionally occurs in humans 25,48 and commonly occurs in reptiles. 18,28,29,40 The amoebas spread to the liver through the portal vascular system in these species, after deep invasion of the mucosa. 40,48 Amphibians have a similar hepatic portal system as do reptiles and mammals, 14 rendering this route of potential spread of infection available to toads. The rarity of deep mural invasion by amoebas in cane toads in our study may explain why the hepatic portal system was not infiltrated, and thus amoebas did not reach the liver and potentially other organs.
Pathogenic species of the genus Entamoeba have an arsenal of virulence factors to enable tissue invasion, including lectins for attachment to intestinal epithelium, mechanisms to cause cytotoxicity of epithelial cells (including induction of apoptosis), secretion of “amoebapore” proteins to lyse cells, a vast array of cysteine proteinases to degrade extracellular matrix, oxygen detoxification mechanisms to allow deep invasion, and finally, various tools to evade the host immune response. 19,25,27,31,39 Despite being highly divergent species phylogenetically, E. histolytica and E. invadens share numerous homologous genes for survival, including many associated with virulence. 52,53 It seems likely, therefore, that the species of Entamoeba identified in the toad, being closely related to E. invadens, 45 also shares similar basic pathogenic mechanisms with E. histolytica. However, based on the lesions in toads, there may also be some important differences in the toad Entamoeba sp. infection dynamics that result in an inability for deep tissue invasion. Such differences might involve pathogen factors such as cysteine proteinases or oxygen detoxification mechanisms, or host factors that alter the pathology. 24 For example, in humans, the intestinal milieu, including presence and types of nutrients and resident bacterial flora, may influence the ability of Entamoeba to invade. 34,49 In reptiles, anorexia may predispose to development of invasive entamoebiasis due to neutral gastric pH in anorectic snakes favoring survival of ingested infective cysts. 40 In humans and mouse models of infection, the suite of cytokines displayed in the face of infection or severity/type of inflammatory response may dictate susceptibility to and severity of invasive disease. 19,26 Further study of the pathology of cane toad entamoebiasis compared to that in other species, as well as determinants of susceptibility of cane toads to invasive entamoebiasis, may more broadly inform the pathogenesis.
Because little is known about amphibian hematological and plasma biochemical responses with disease, 8,9 we performed preliminary investigations into how these parameters may be influenced by invasive entamoebiasis in toads. Neutrophils and monocytes were elevated in toads with invasive entamoebiasis, suggestive of an inflammatory response. 9 Along with elevated neutrophils, there was a decrease in lymphocytes, resulting in an increased N/L ratio. In cane toads, this ratio is elevated by injection of lipopolysaccharide (G. Brown, personal communication, 2014) and may represent a response to Gram-negative septicemia or endotoxemia resulting from intestinal ulceration. 1 Alternatively, the elevated N/L ratio could, at least in part, represent a stress leukogram. 17 Because Entamoeba sp. may be present in the intestinal lumen in the absence of invasive disease, 21,36,45 these findings suggest that white blood cell counts may be diagnostically useful in assessing presence of invasive entamoebiasis in amphibians when amoebas are detected in feces in live animals. However, published values for normal average N/L ratios in 6 other species of bufonid toads range from 0.12 to 3.4, 17 encompassing the averages for cane toads with and without invasive entamoebiasis. That wide interspecific variation underlines the value of having a suitable comparison group to aid in interpretation. Interestingly, despite the gross observation of hemorrhagic colonic content in some toads with invasive entamoebiasis, affected toads on average were not anemic, exhibiting no significant decrease in packed cell volumes.
We performed preliminary investigations into alterations of additional clinical biochemical values in 8 clinically ill toads with invasive entamoebiasis from early in the outbreak. Although these data suffer from a low sample size and lack of a suitable control group (in the form of data from toads in the same population without invasive entamoebiasis), comparison with published values in other anuran species provides some insight. For example, compared to captive bullfrogs, toads with invasive entamoebiasis have similar levels of potassium and mildly lower levels of calcium and phosphorus. 12 In a study of normal tree frogs and frogs with the skin disease chytridiomycosis, the diseased frogs suffered osmotic imbalance that included severe hypokalemia, presumed to be due to loss of electrolytes through affected skin. 51 Our toads with invasive entamoebiasis had similar potassium levels to the normal frogs in that study, a further suggestion that potassium levels in our toads may have been unremarkable. We also measured glucose in this small sample of clinically ill toads with invasive disease. Glucose in amphibians may normally be substantially lower than in other terrestrial vertebrates, with values less than 2.8 mmol/l being common. 54 Therefore, the average 3.1 mmol/l plasma glucose in diseased toads may be within normal. The values for our final 2 analytes, creatine phosphokinase and aspartate aminotransferase, appear slightly high compared to reference ranges for other species but are similar to values in normal frogs in one other study. 51 In other vertebrates, creatine phosphokinase is considered to originate solely from muscle, and concurrent elevations in aspartate aminotransferase are interpreted as also likely originating from muscle. 3 Any elevation in values for these parameters in our toads seems likely to reflect the effects of handling and intracardiac blood sampling, rather than invasive entamoebiasis.
The mechanism of severe illness and, probably in some cases, death in toads with invasive entamoebiasis may be due to one or a combination of the myriad effects that occur when extensive regions of intestinal mucosa are compromised, including profound fluid and electrolyte imbalances, and, with small intestinal involvement, malassimilation and malabsorption of nutrients. 50 Our limited clinical biochemical investigations into toads with invasive entamoebiasis did not reveal obvious alterations in this regard. However, rather than indicating a lack of usefulness for clinical biochemistry in sick amphibians, this may indicate the need for an expanded range of biochemical analytes in sick amphibians, including suitable control groups. Another possible mechanism of severe illness or death in toads with invasive entamoebiasis could be septicemia or endotoxemia arising from the damaged intestinal mucosa. 29 We did not see gross or histological evidence of systemic bacterial infection, but bacterial culture of 3 aseptically obtained organs would have been more definitive.
In conclusion, invasive entamoebiasis in free-ranging cane toads results primarily in colonic pathology, variably characterized by hyperplasia, erosion, ulceration, and fibrosis, with abundant associated intraepithelial amoebas and only rare infiltration of amoebas into the lamina propria. Lesions occasionally extend into the distal small intestine and, less commonly, the stomach, with no direct evidence of spread to other organs, such as liver. Hematology and clinical chemistry of toads with invasive entamoebiasis are unremarkable, except for affected toads exhibiting neutrophilia, monocytosis, and lymphopenia.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819868729 - Pathology Associated With an Outbreak of Entamoebiasis in Wild Cane Toads (Rhinella marina) in Tropical Australia
Supplemental Material, DS1_VET_10.1177_0300985819868729 for Pathology Associated With an Outbreak of Entamoebiasis in Wild Cane Toads (Rhinella marina) in Tropical Australia by Catherine M. Shilton, Jan Šlapeta, Richard Shine and Gregory P. Brown in Veterinary Pathology
Footnotes
Acknowledgements
We thank staff at Berrimah Veterinary Laboratory for technical assistance. We are grateful to the 3 anonymous reviewers for comments on an earlier version of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Australian Research Council (grant FL120100074 to R. Shine and FT120100095 to G. P. Brown). Financial assistance for production of histology slides was provided by Wildlife Health Australia funds to C. M. Shilton.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
