Abstract
There is limited published information about disease in wild sea snakes and no standardized guideline for postmortem examination of sea snakes. Identifying causes of morbidity and mortality of marine vertebrate species has been pivotal to understanding disease factors implicated in stranding events and assisting with the formulation of conservation plans. Additionally, postmortem findings can provide valuable information on life history traits and the ecology of these reclusive species. Sick, moribund, or dead sea snakes are intermittently washed ashore along Australian and international beaches and provide an opportunity to examine a subset of the population and identify causes of population decline. We present an illustrated description of sea snake anatomy and describe a systematic approach to postmortem examination of sea snakes. We describe common pathologic conditions identified from clinical and postmortem examinations of stranded Australian sea snakes from southeast Queensland. Notable pathologic conditions include traumatic injury, inflammatory conditions, parasitic infections, and neoplasia.
Introduction
Identifying causes of morbidity and mortality in many marine vertebrate species including marine turtles, dugongs, whales, and dolphins has been pivotal to developing an understanding of disease factors implicated in stranding events and assisting with the formulation of conservation plans for some species.14,44
A standardized set of guidelines for examining dead sea snakes is essential to determine causes of morbidity and mortality. Documentation of stranding location and postmortem examination also provides valuable information on life history traits and the ecology of these reclusive species (e.g., through identification of stomach contents and reproductive status). We present here an illustrated guide to assist veterinarians and biologists with postmortem examination of dead sea snakes and include descriptions of common pathologic conditions, as well as photographic illustration of healthy and diseased sea snakes. Unless otherwise indicated, the pathologic conditions described below (and shown in images) are from detailed examinations of 167 sea snakes stranded along the southeast Queensland (SEQ) coast from north Fraser Island (24.6450°S), Queensland to the Queensland–New South Wales border (28.1650°S) between 2005 and 2015. Detailed gross postmortem information was available for 92 snakes and histologic examination was undertaken in 82 snakes. The remaining 75 snakes underwent antemortem examination 20 only.
Almost all sea snakes are venomous. It is strongly recommended that anyone who encounters a stranded sea snake contact authorities or personnel familiar with handling venomous snakes. Sea snakes in Australia are protected under the Commonwealth’s Environment Protection and Biodiversity (EPBC) Act 1999, the Great Barrier Reef Marine Park Act 1975, and Queensland Nature Conservation Act 1992, and appropriate permits and authorization should be sought before commencing any postmortem examination or collection of samples.
Sea snake species
Current status of Hydrophiinae populations
True sea snakes (family Elapidae, subfamily Hydrophiinae: Hydrophiini) are viviparous and entirely aquatic, unlike their partially terrestrial and oviparous counterparts the sea kraits (subfamily Hydrophiinae, genus Laticauda).53,55 Sea snakes comprise >60 species distributed throughout tropical and subtropical Indo-Pacific waters and inhabit shallow coastal waters, coral reefs, and river mouths, as well as deep ocean water. 52 Despite their status as a protected species, some sea snake populations around Australia are declining (Australian Fisheries Management Authority. 2010. Threatened, endangered, and protected (TEP) species management fact sheet no. 6: Seasnakes. Available at: https://goo.gl/lAh0mc),8,26,61 whereas population trends elsewhere are largely unknown (Livingstone S, et al. 2009. Status of the world’s sea snakes: IUCN red list assessment. Brisbane, Australia. Available at: https://goo.gl/KhxmGN).
Species-specific differences
Although belonging to the largely terrestrial family Elapidae, sea snakes possess characteristic features vital for a marine existence. These most notably include a paddle shaped tail, valve-like nostrils, circular pupils, a sublingual salt gland, viviparous reproduction, a laterally compressed body, greatly reduced ventral scales, and skin capable of transcutaneous exchange of gas (oxygen and carbon dioxide) and water as well as photoreception.7,67 The heart in sea snakes is located more caudally than their terrestrial counterparts, with the head–heart distance estimated to be 23–42% of total body length (compared with 15–23% in terrestrial snakes). 24 Among the sea snakes, species-specific differences include variation in venom apparatus development (fish-egg eating Emydocephalus annulatus have atrophied fangs and venom glands 22 ), head size (some genera are microcephalic), 55 scale rugosity, 4 and skin permeability to water. 13
Postmortem examination
External examination
Safety
Almost all species of sea snakes are highly venomous, and the venom is regarded as neurotoxic and myotoxic. 49 The handling of dead or moribund venomous snakes poses a risk to the handler 28 because venom glands continue to hold venom after the snake has died. Many zoos and venomous snake facilities have in-house policies regarding the handling of dead snakes and in most instances the head is wrapped in tape or tubing to keep the jaws together prior to postmortem examination (R Jackson, pers. comm., 2016).
Signalment and data collection
Collecting information surrounding sea snake strandings (e.g., location, history, observation of recent environmental or climatic events) is an important part of any assessment and can greatly assist with determining a cause of death. If, for example, a snake is found in a net off-shore or if there is evidence of increased boating activity in the area, it may prompt the investigator to look more closely at the lungs for evidence of drowning or injury that may be associated with human interaction. Recording an exact stranding location (including GPS latitude and longitude coordinates if possible) may reveal new information about range distribution or ecologic patterns of a species not previously identified in that area, which is important when compiling data to predict common stranding locations. Photographic records of the rescue site, the snake in situ, external injuries, and body condition should be obtained where possible, and include an identification number, reference scale, and date of collection. 15 Recording the date and time of rescue or death informs the final examiner of the postmortem interval, which will in turn assist with interpretation of changes and guide options for further testing. Records and images can be utilized by government for annual reports of stranding and mortality.
Correct species identification (using published keys11,53 or online resources 16 ) informs the examiner of a snake’s habitat niche, distribution range, and its conservation status (i.e., rare, endangered, etc.). Key features for identification include scalation of the head, ventral and mid-body scale count, snout-to-vent length (see Morphometric measurement below) and, in some cases, skin color and pattern, although skin color and pattern should never be the sole criteria for identification, as variations can occur within species. When a species cannot be identified at the time of rescue, the examiner can submit the entire specimen or photographs of the stranded snake (dorsal and ventral surfaces of full body as well as the top, side, and bottom of the head) to the relevant state government museum.
Morphometric measurement
Measurements are important in species determination and in assessment of maturity and body condition. Morphometric data should include at least snout-to-vent length (from the tip of the snout to the caudal edge of the anal scale), tail length (from caudal edge of anal scale to the tip of the tail; Fig. 1A), and body weight. Other measurements less commonly used include head length, 59 head width (Fig. 1B), and mid-body circumference (Table 1). Head width is measured across the broadest part of the head (usually just behind the eyes) using calipers. Mid-body circumference is measured by encircling the snake’s body with a non-stretching measuring ribbon, at approximately two-thirds the distance from the snout to the tail tip, avoiding the stomach. Body condition should be assessed as emaciated, poor, fair, good, or excellent. 20
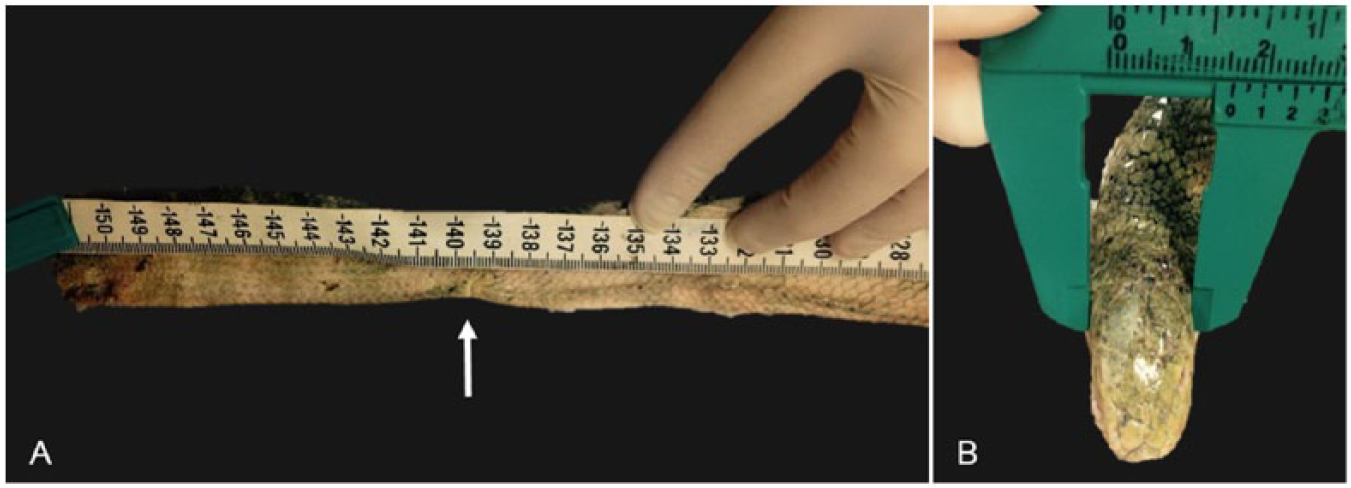
Morphometric measurements in sea snakes.
Morphometric measurements of 3 species of healthy Australian sea snakes (Hydrophis spp.) collected from Weipa, Queensland, Australia in October–November 2011.*

Range is provided in parentheses after mean values.
Sex determination
The sex of sea snakes is almost impossible to determine without exteriorizing the male genitalia or hemipenes, probing the vent for hemipenes, or visualizing the genitalia at postmortem examination. Skin color, pattern, or scalation are generally not sex-specific, except for the presence of a prolonged rostral scale in adult males of E. annulatus. 25 Sexual dimorphism is exhibited in some sea snakes with females often larger than males, 32 a possible consequence of vivipary. 60 Sexual dimorphism has been investigated in snout-to-vent length, tail length, total body length, weight, scale counts, scale keeling, body pattern, and head length, 24 but there appears to be little consistency across species and no single feature is determinate in all species of sea snake. Based on data for 96 healthy wild-caught sea snakes (collected by the authors from Weipa 12.7458°S, 141.8980°E in October–November 2011) comprising Hydrophis elegans, Hydrophis peronii, and Hydrophis curtus, males were on average heavier than females (Table 1). H. elegans and H. peronii males on average had a greater snout-to-vent length and head width, and H. elegans and H. curtus males also had a slightly greater mid-body circumference.
Maturity/age
As in many other reptilian species, maturity in sea snakes can be difficult to determine because growth rate is likely to fluctuate with water temperature, food availability, and attainment of sexual maturity. 9 Broad age categories often used are immature/juvenile and mature. 42 The onset of sexual maturity is heralded by the presence of oviductal eggs in females and mature spermatozoa in males (Burns GW. Aspects of population movements and reproductive biology of Aipysurus laevis, the olive sea snake. [PhD dissertation]. Armidale, Australia: University of New England, 1984). Newborn juvenile sea snakes can be identified by the presence of a remnant umbilicus or navel scar. 42
Carcass condition
Standard codes (Meager JJ, Limpus CJ. Marine wildlife stranding and mortality database annual report 2011. III. Marine turtle. Available at: https://goo.gl/SVJ8Ck) may assist with classifying the level of decomposition. A standard carcass condition code is available for sea turtles 15 but not for sea snakes. Postmortem autolysis is accelerated by submergence, warm water (>26.5°C), low water current (Jones H. Aging sea turtle remains: a decay rate experiment. [Honor’s thesis]. Brisbane, Australia: The University of Queensland, 2015), sepsis, and active infection prior to death. Increased autolysis may result in overestimation of the postmortem interval. 63 The time elapsed between collection and examination should also be considered when assessing decomposition rate. Storage and/or transport at ambient temperature and freezing can impact carcass condition and limit options for subsequent testing. Histologic identification of the cause of death is more likely if the carcass is in good condition.
Examination of the head, vent, and tail
All sea snakes possess highly contractile circular pupils and have large sinuses in the choroid that probably function to equalize ocular pressure during diving. 36 Similar to terrestrial snakes, they possess spectacles over their eyes to protect the delicate cornea from damage. Although the eyes are not the sole sensory method for detection of prey, they are important for identifying movement. Damage to the eye or spectacle can have significant implications for survival and could be a cause of poor body condition in sea snakes. The eyes should be examined thoroughly, 20 looking for damage or irregularities in the shape of the eye, iris, and spectacle. A normal healthy globe should have a clear cornea, and the eyes should be of equal size. Lesions within the mouth may impact vision as the flow of lacrimal secretions may become obstructed leading to distension of the sub-spectacular space (bullous spectaculopathy). 20
Great care must be taken when examining the oral cavity of a sea snake, using instruments so that the examiner’s hands do not enter the mouth. The mucosa should be thoroughly examined for signs of ulceration, hemorrhage, necrosis, foreign bodies, or inflammation (stomatitis or glossitis). The mandible and maxilla should be examined for evidence of fractures or deformities.
The vent of a healthy snake should be clean, free of any discharge or accumulated fecal matter, and the mucosa should be a uniform pale pink. The vent should also be examined for evidence of prolapse, laceration, hemipene eversion, deformity, and foreign bodies.
The tail should be examined on both sides for evidence of injury and epibiota (see below). Photoreceptors have been identified on the tip and dorsal surface of the tail of sea snakes and are thought to assist with avoidance of predators and concealment.32,67 Damage to these areas or coverage by these epibiota could result in increased risk of injury or predation. Injury, partial amputation, and heavy epibiosis of the tail have been observed in many stranded sea snakes and may interfere with swimming.
Epibiota
Epibiota, including barnacles, algae, and decapods, are encountered in large numbers in the marine environment and can colonize the skin of many marine animals, including sea snakes. To combat this accumulation, many species of sea snakes frequently rub against firm substrates to reduce their epibiota load. In addition, sea snakes are thought to utilize their natural shedding process and shed skin more frequently than their terrestrial counterparts. 50 The presence of large numbers of epibionts on a stranded sea snake should alert the examiner to potential illness or injury. Epibionts reported to colonize sea snakes include >40 species from the taxa Foraminifera, Hydrozoa, Polychaeta, Cirripedia, Decapoda, Gastropoda, Bivalvia, Bryozoa, and Chordata, and >8 species of diatoms and algae. 48 The sessile barnacle Platylepas ophiophilus (Fig. 2A), the pedunculate barnacle Lepas anserifera (Fig. 2B), and algae (Fig. 2C) were the most common findings on stranded sea snakes in SEQ. The pedunculate barnacle Conchoderma virgatum (Fig. 2D) was also seen in low numbers. The significance of epibionts remains largely unknown although some reports suggest their presence may be detrimental.50,66 It is plausible that the inability to remove barnacles and other epibionts may result from functional constraints, such as spinal or soft tissue injury, or underlying disease resulting in altered nervous system function or debility. Anecdotal evidence suggests there is a correlation between high epibiotic load and disease, although heavy epibiotic burdens do not seem to be correlated with traumatic injury, presumably because such injury is often acute. Retained shed (dysecdysis) may also build up around persistent epibionts predisposing the snake to further shedding problems as well as localized skin infections at the site of attachment. Epibiont load should be recorded as an estimated percentage of body covered, with separate estimates done for each epibiont species if more than one is present. Organisms can be manually removed and placed in 80% ethanol or 10% buffered formalin for subsequent identification, or photographs of organisms can be sent to an experienced marine parasitologist.

Epibiota found on stranded sea snakes in southeast Queensland, Australia.
Internal examination
Before commencing a postmortem examination, the mouth should be carefully taped closed. Where histologic examination of the brain is intended, the whole head can be removed and placed in formalin, 27 which will also deactivate most venoms. 45 If a laboratory will not accept the intact head of a venomous snake, the brain can be carefully dissected from the skull prior to submission (see Nervous system). Alternatively, the lower jaw can be left attached to the remainder of the body and the head removed by cutting through each temporomandibular joint and then severing the head from the spine at the atlanto-occipital joint and placing it in formalin (Fig. 3A, 3B).
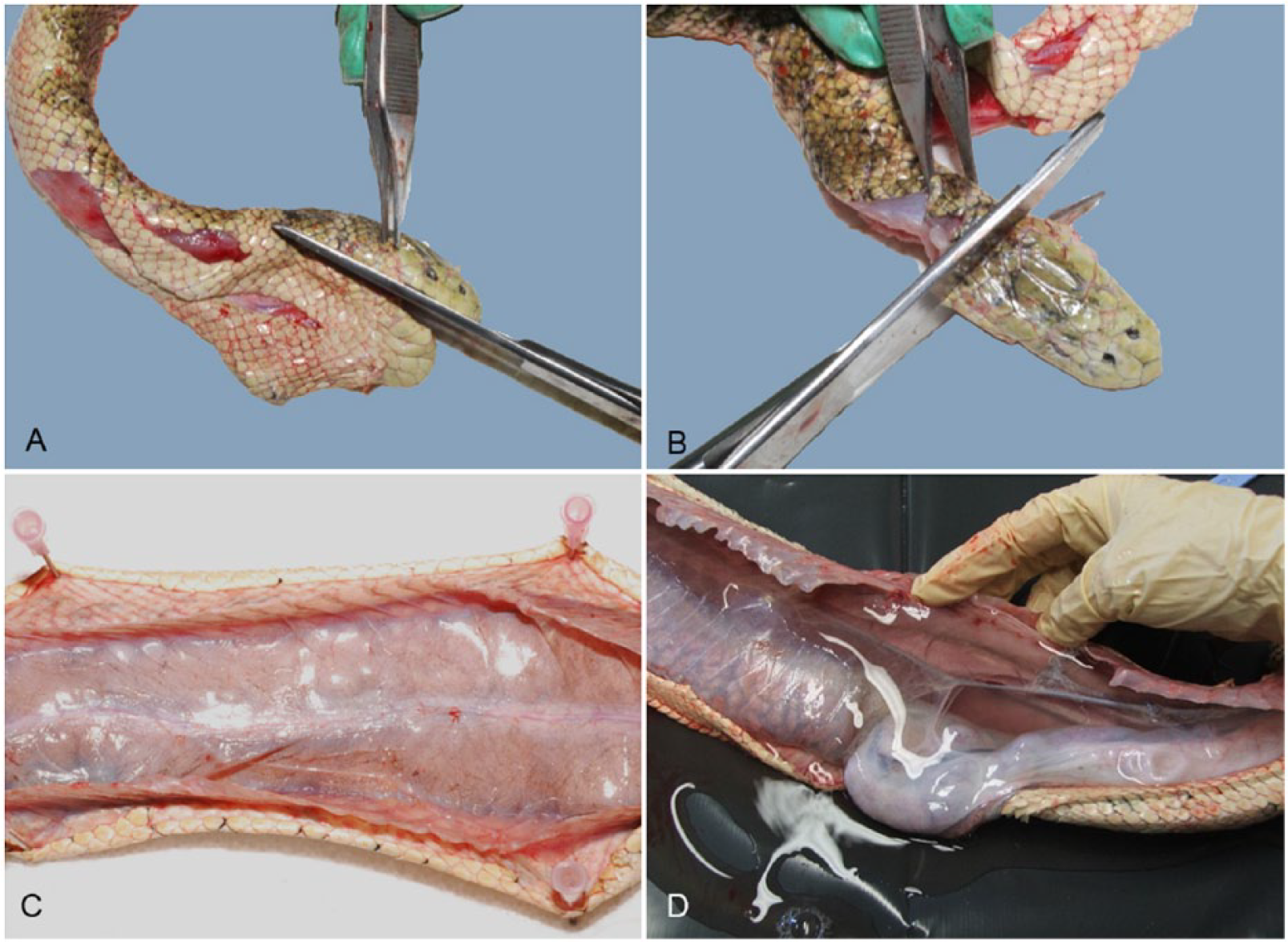
Methods for removing the head and opening the celomic cavity of a sea snake.
The celomic cavity can be opened by making an incision through the skin and body wall on one side of the ventral scales (about midway along the body) and extending the incision cranially and caudally using sharp scissors. To overcome the tendency of the carcass to roll to one side (given the laterally compressed body), the skin on both sides of the midline can be reflected and pinned to a Styrofoam or wax board placed beneath the snake (Fig. 3C). Alternatively, the snake can be placed in (left or right) lateral recumbency and the body wall can be reflected dorsally to examine the celomic cavity in situ (Fig. 3D).
Examination of the celomic cavity
The celomic membrane should be inspected for damage and then incised similarly to the skin. The organs should be examined in situ (Fig. 4A) for any obvious abnormalities or developing embryos (Fig. 4B) that could rupture during handling, before the entire viscera is removed. In healthy sea snakes in good or excellent body condition, abundant fat bodies can be seen in the caudal half of the celomic cavity, often obscuring much of the intestines and colon, splenopancreas, reproductive organs, and kidneys. Minimal celomic fluid is normally present and it is clear and slightly viscous as in other species. Vascular or hepatic conditions can lead to accumulation of a large volume of celomic fluid (Fig. 3D).
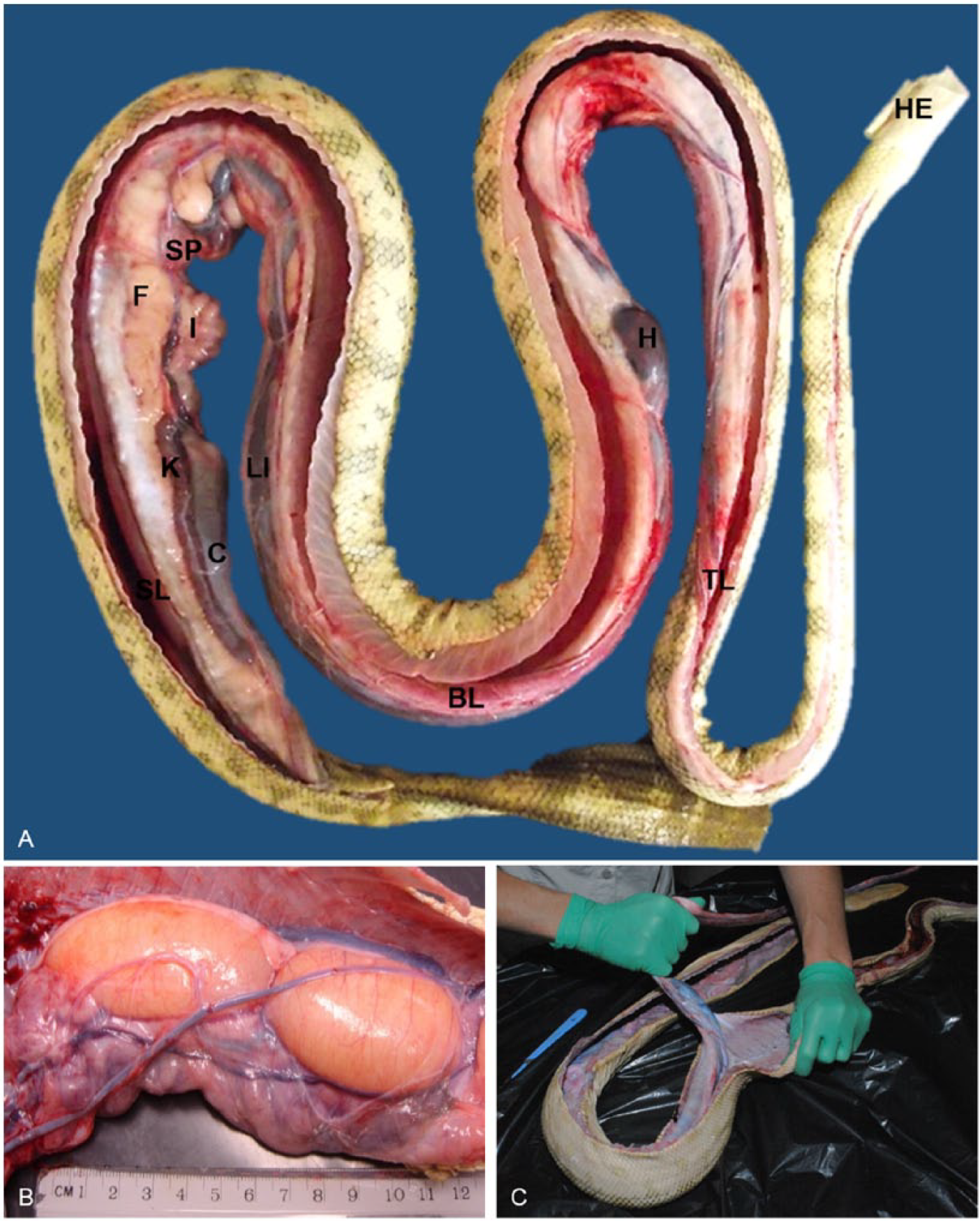
In situ anatomy of sea snakes.
Removal of visceral organs and examination of the body wall
Removal of the organs is best achieved by transecting the esophagus and trachea immediately caudal to the pharynx and pulling the viscera caudally to the cloaca, severing or tearing connective tissue attachments as required (Fig. 4C). After the visceral organs have been removed, the body wall should be examined for signs of penetrating injury, fractures to the ribs or spine, or granulomatous lesions in the hypaxial or epaxial muscles (as may be seen with microsporidial infection 21 ).
Detailed examination of body systems, including common pathologic findings
Integumentary system
Sea snake skin is semipermeable to oxygen and water, which is thought to assist with diving ability in the aquatic environment.23,30 Compared to their terrestrial counterparts, the scales are generally smaller (particularly the ventral scales) and the degree of imbrication (overlap) varies with species. 24 Sea snake skin is usually soft and smooth, but in some species (such as H. peronii and H. curtus) the scales are highly keeled, and scale rugosity may be more developed in males in the breeding season. 3
The presence of dysecdysis (often associated with epibiota) or hyperemia of the skin is abnormal and can be an indirect indicator of systemic illness or injury. Skin wounds (including puncture wounds), scars, partial evisceration, and spinal abnormalities indicate trauma, which may be inflicted during capture of prey, by predator attack (e.g., by sea eagles or marine species; Fig. 5A–D), or by trawling. Superficial skin ulceration and or inflammation may be seen where barnacles have been forcibly removed (during capture or examination; Fig. 5E). Linear lacerations are unlikely to be caused by predators and are more likely to be the result of propeller injury or interaction with trawlers including net and fishing line entanglement 20 (Fig. 5F) or inappropriate use of snake handling equipment. A breach in the integument may predispose to bacterial or microsporidial infection, 21 or to scarring that could impede locomotion and predispose to colonization by epibiota.
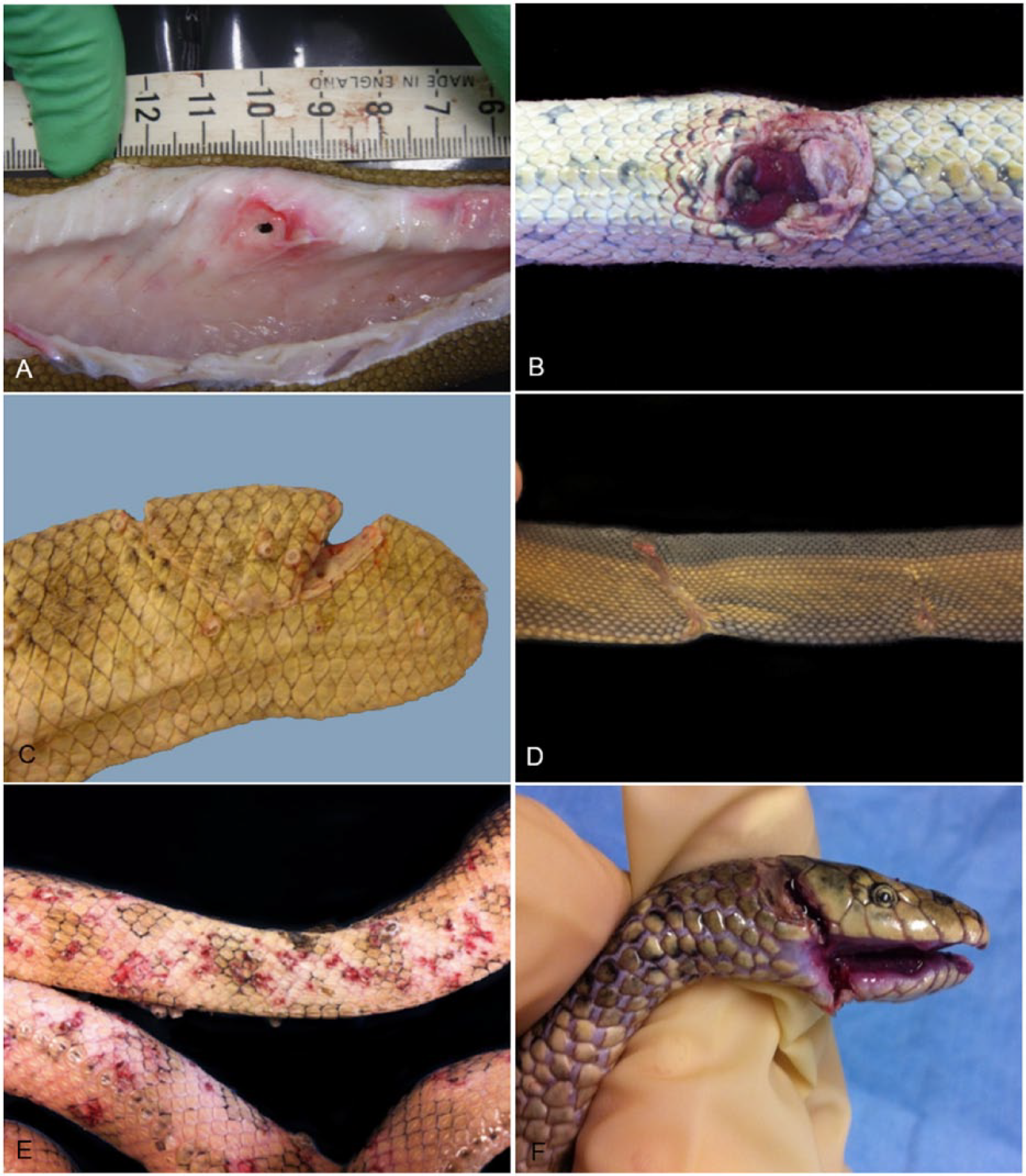
Wounds in stranded sea snakes from southeast Queensland.
Musculoskeletal system
Muscle
The somatic musculature is cylindrical, with the hypaxial and epaxial muscles used predominantly for lateral undulatory locomotion. Cachexia with skeletal muscle atrophy is often observed in sea snakes suffering from chronic debilitation resulting in reduced body condition scores 20 (Fig. 6A, 6B). Microsporidiosis affecting the hypaxial and epaxial musculature has been reported in 3 species of hydrophiid sea snakes, 21 although the origin of infection is unknown.
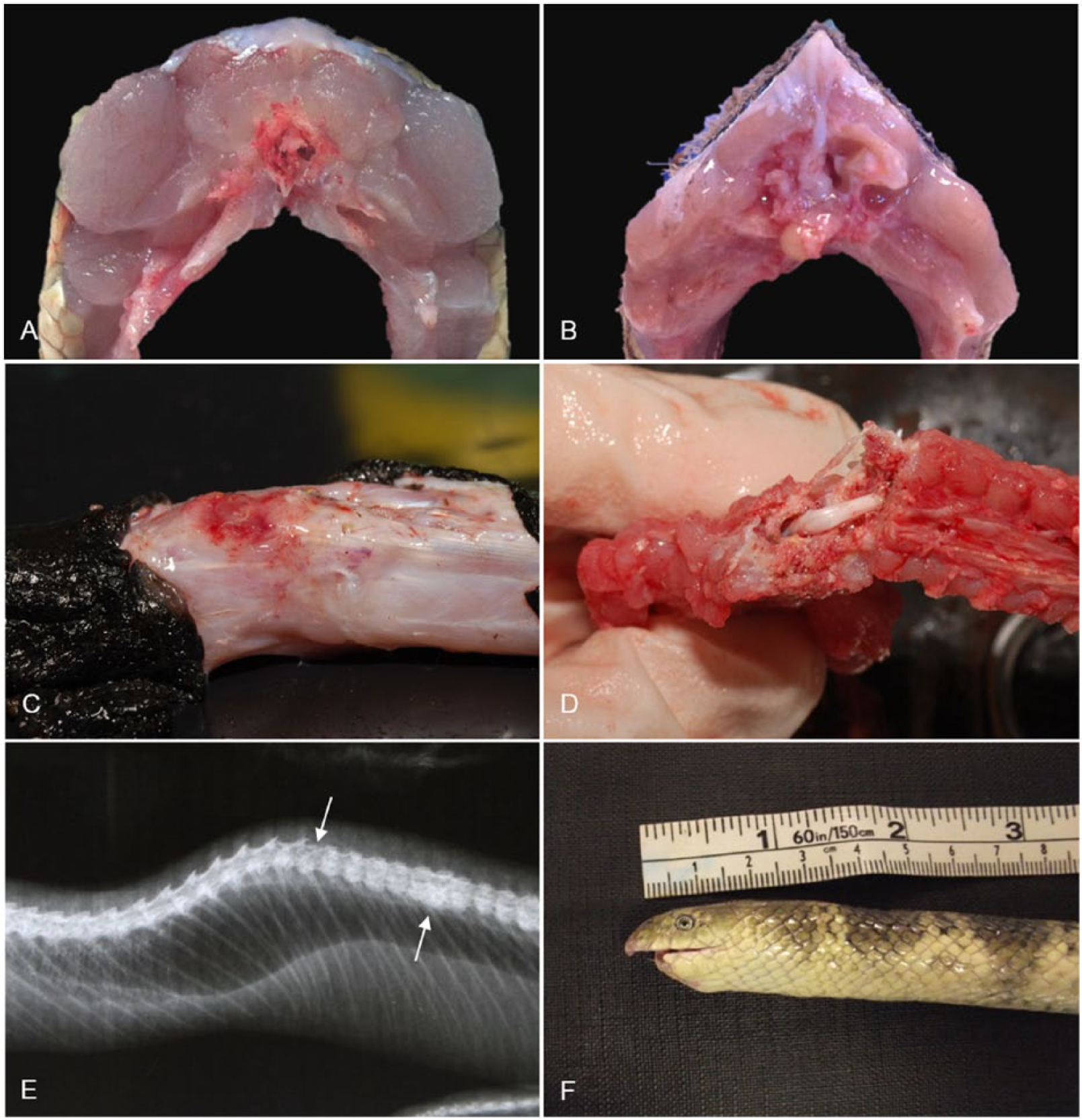
Musculoskeletal lesions in stranded sea snakes from southeast Queensland.
Bone
One of the most common conditions in sea snakes washed ashore is traumatic injury to the spine or jaw and/or head, including vertebral fractures and mandibular amputations (Fig. 6C–F). These may be accompanied by heterophilic-to-pyogranulomatous osteomyelitis. The cause is usually difficult to identify, although trawling is known to result in traumatic injuries to sea snakes with factors such as the weight of the catch and how the snakes are treated on deck likely to affect survival, 64 and is a potential cause of sea snake strandings.
Cardiovascular system
Sea snakes possess a 3-chambered heart that lies more caudally than in their terrestrial counterparts. The benefit of this is not fully understood although a more centrally located heart may allow for more efficient shunting of blood to different parts of the body or may assist in equalizing blood pressure between the caudal and cranial part of the body during diving. 31 Two aortae (left and right) and a single pulmonary artery carry blood from the heart. The left aorta crosses over the lung and esophagus from the anatomic right side of the heart to the left side of the body (Fig. 7A). As in other reptiles, there is an incomplete ventricular septum. 39 The muscular ventricle can be visualized by incising and reflecting the thin pericardium (Fig. 7B), and thereafter the entire heart can be exteriorized for examination.
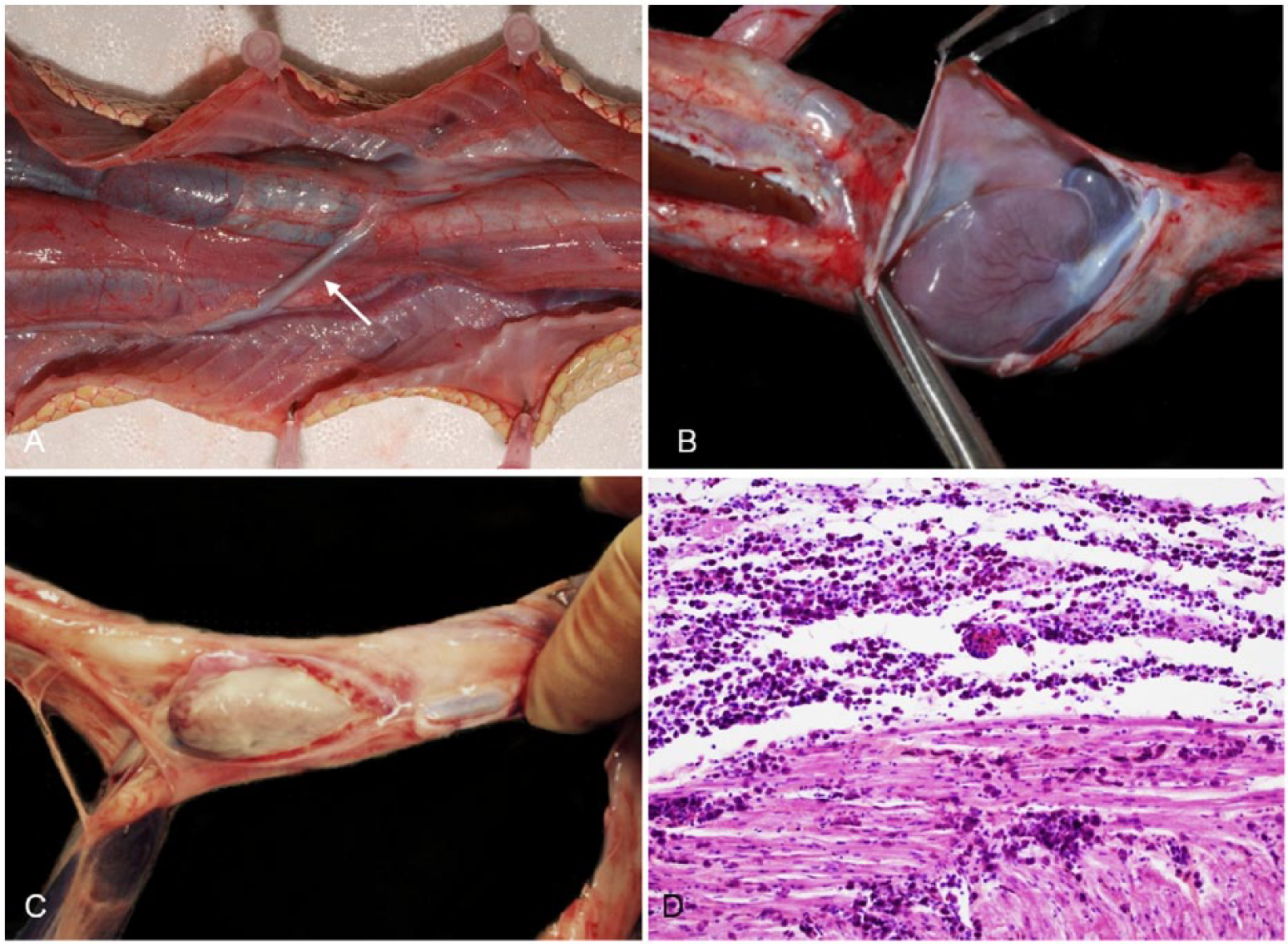
Cardiovascular anatomy and pathology in sea snakes.
Cardiovascular disease is rarely observed in sea snakes. Pericarditis and myocarditis were identified in one stranded sea snake in SEQ that was lethargic and emaciated. The pericardium was grossly thickened, opaque, and partially adhered to the epicardium (Fig. 7C). The heart was pale and mottled, and histologic examination revealed a dense infiltrate of macrophages and heterophils expanding the pericardium and throughout the myocardium (Fig. 7D).
Given their diving capabilities, sea snakes could potentially suffer from decompression sickness, particularly if caught in trawler nets, which are rapidly brought to the ocean surface. It is postulated that a sea snake’s ability regarding cutaneous expulsion of nitrogen and shunting of blood away from the lung may reduce the risk of decompression sickness.29,58 This syndrome requires further investigation in sea snakes.
Respiratory system
Sea snakes possess a single lung divided into 3 main anatomic sections: the tracheal lung; the bronchial lung; and the saccular lung. The tracheal and bronchial lungs are collectively termed the vascular lung 35 and are responsible for gas exchange. The tracheal lung is defined grossly by cartilaginous rings along its length, which are complete rings cranially, becoming incomplete caudally, and disappear as the tracheal lung merges with the bronchial lung. The internal surface of the bronchial lung (and sometimes the distal tracheal lung) comprises a network of muscle fibers giving it a mesh-like appearance (Fig. 8A inset). The length and surface area of the bronchial lung varies dramatically with species, spanning almost the entire length of the respiratory tract in some species (Fig. 8A); in others, it forms only a very small section before abruptly ending at the junction with the saccular lung (Fig. 8B). The saccular lung is relatively avascular and is thought to function mostly for air storage. 6 The anatomic variations in the lungs of sea snake species may reflect different diving abilities and foraging behaviors.
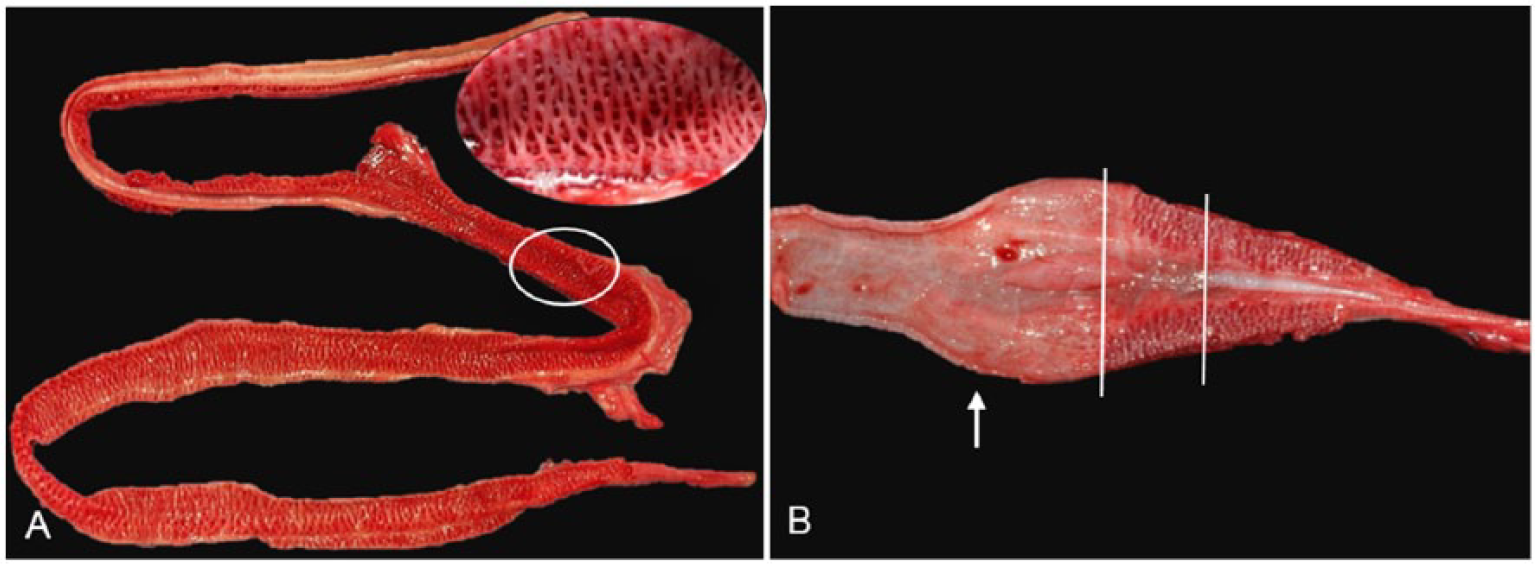
Lung anatomy varies with species of sea snake.
Hydrophitrema gigantica (syn. Pulmovermis cyanovitellosus; https://goo.gl/zaKIjj), a large hemiurid trematode, is relatively common in the lungs of sea snakes. 54 The number and location of H. gigantica in the lung vary widely, with >45 sometimes present in the lungs of stranded sea snakes in SEQ. On postmortem examinations, 29 of 92 (32%) sea snakes had H. gigantica in their lungs, and 22 of the 29 (76%) snakes with H. gigantica were in suboptimal body condition (i.e., fair, poor, or emaciated). Although the presence of H. gigantica is generally regarded as benign, it may be associated with mucosal trauma and hemorrhage in the bronchial lung (Fig. 9A, 9B) and saccular lung. Histologic changes included congestion, lymphangiectasia, shallow faveolae, parenchymal fibrosis, epithelial hypertrophy, cuboidal metaplasia, and focal heterophilic infiltration.
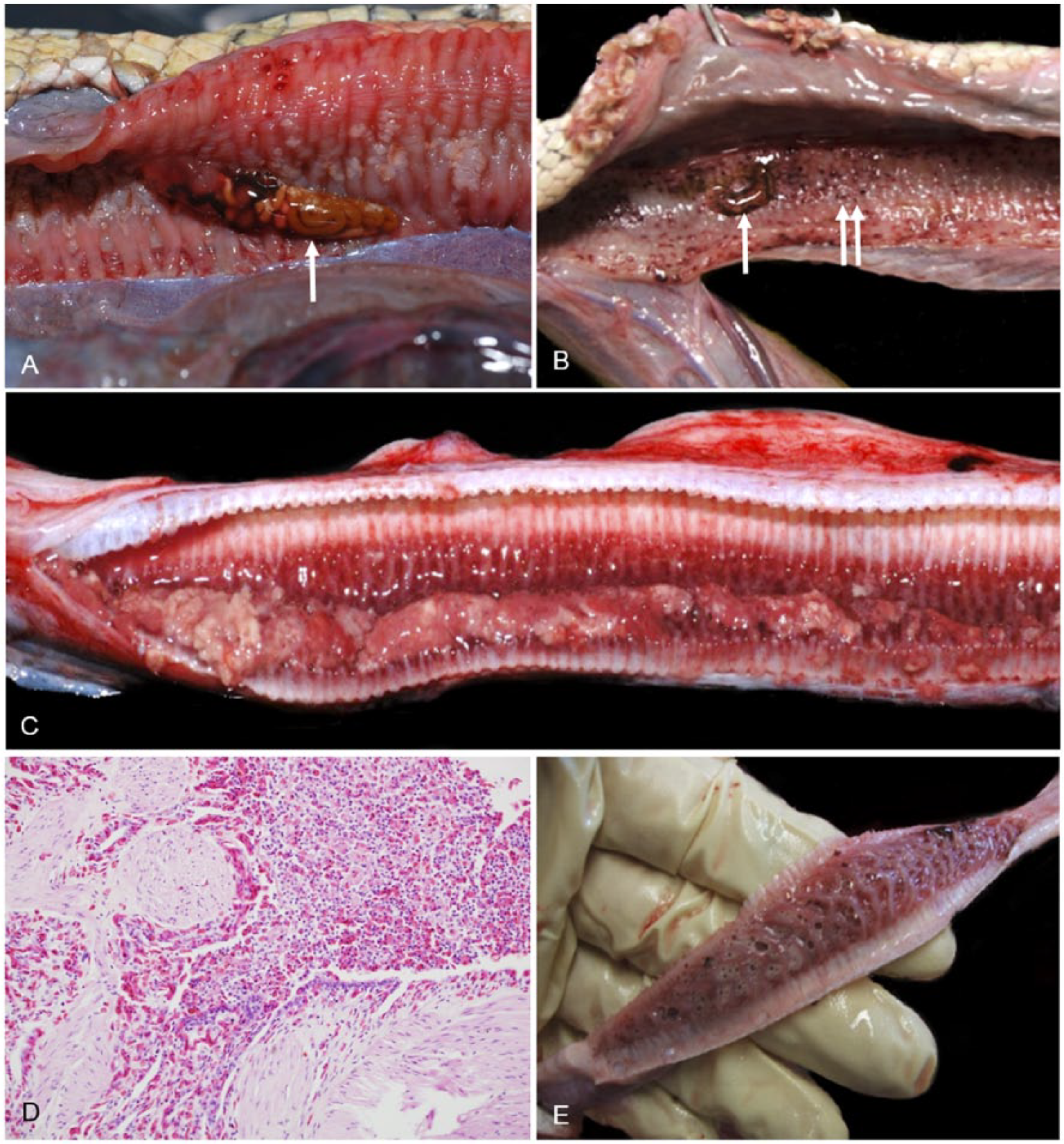
Lung lesions and parasites in stranded sea snakes from southeast Queensland.
Other pathologic conditions identified in the lungs of stranded sea snakes from SEQ include pulmonary fibrosis, interstitial pneumonia, and suppurative bronchopneumonia (Fig. 9C, 9D). Foamy exudate and water was also seen in the lungs of one sea snake presumed to have drowned (Fig. 9E). Although a cause for these pulmonary conditions was not always evident, in some cases large numbers of H. gigantica or aspiration of sea water were noted and, in one case, bacteria (Chryseobacterium spp., formerly Flavobacterium spp.) were isolated from lung tissue.
Gastrointestinal system (including liver and pancreas)
The gastrointestinal tract of sea snakes is very similar to terrestrial snakes with a long esophagus, glandular and nonglandular regions of the stomach, and a long convoluted intestine. Prey selection varies between sea snake species but often includes bony fish, marine eels, and eel-like fish. Some specialist feeders, such as E. annulatus and Aipysurus eydouxii, feed solely on fish eggs. Digestion is rapid in sea snakes compared to terrestrial snakes, 24 but where ingested prey is found it should be collected and identified where possible.
Oral cavity
The most common oral conditions detected in stranded sea snakes in SEQ were stomatitis, glossitis, and jaw amputation (see Musculoskeletal system). Stomatitis was identified in 18 of 167 (11%) snakes and varied in appearance from reddened oral mucosa with petechial hemorrhage to caseous exudation. Glossitis, often associated with trauma to the tongue, was identified in 3 of 167 (2%) snakes. Vibrio alginolyticus was cultured from a swab of the oral cavity of one snake with severe stomatitis and glossitis.
Stomach, small intestine, and large intestine
Pathologic conditions of the gastrointestinal tract observed in sea snakes from SEQ included foreign body penetration (e.g., fish spines in the oral mucosa; Fig. 10A), intestinal intussusception (Fig. 10B), gaseous intestinal dilation (Fig. 10C), gastric ulceration (Fig. 10D), granulomatous gastritis and enteritis (Fig. 1E), lymphocytic gastritis, colitis, gastric nematodiasis, intestinal cestodiasis, and intestinal trematodiasis. Also observed was multifocal serosal granulomatous serositis often containing cestode or nematode larvae, or trematode eggs (Fig. 10E, 10F). Nematode parasites identified in the lumen of the stomach of stranded sea snakes included Paraheterotyphlum australe, 40 which were often seen as an entangled mass of adults (Fig. 10G). Encysted plerocercoids of Callitetrarhynchus gracilis were occasionally observed in the intestinal serosa (Fig. 10H).
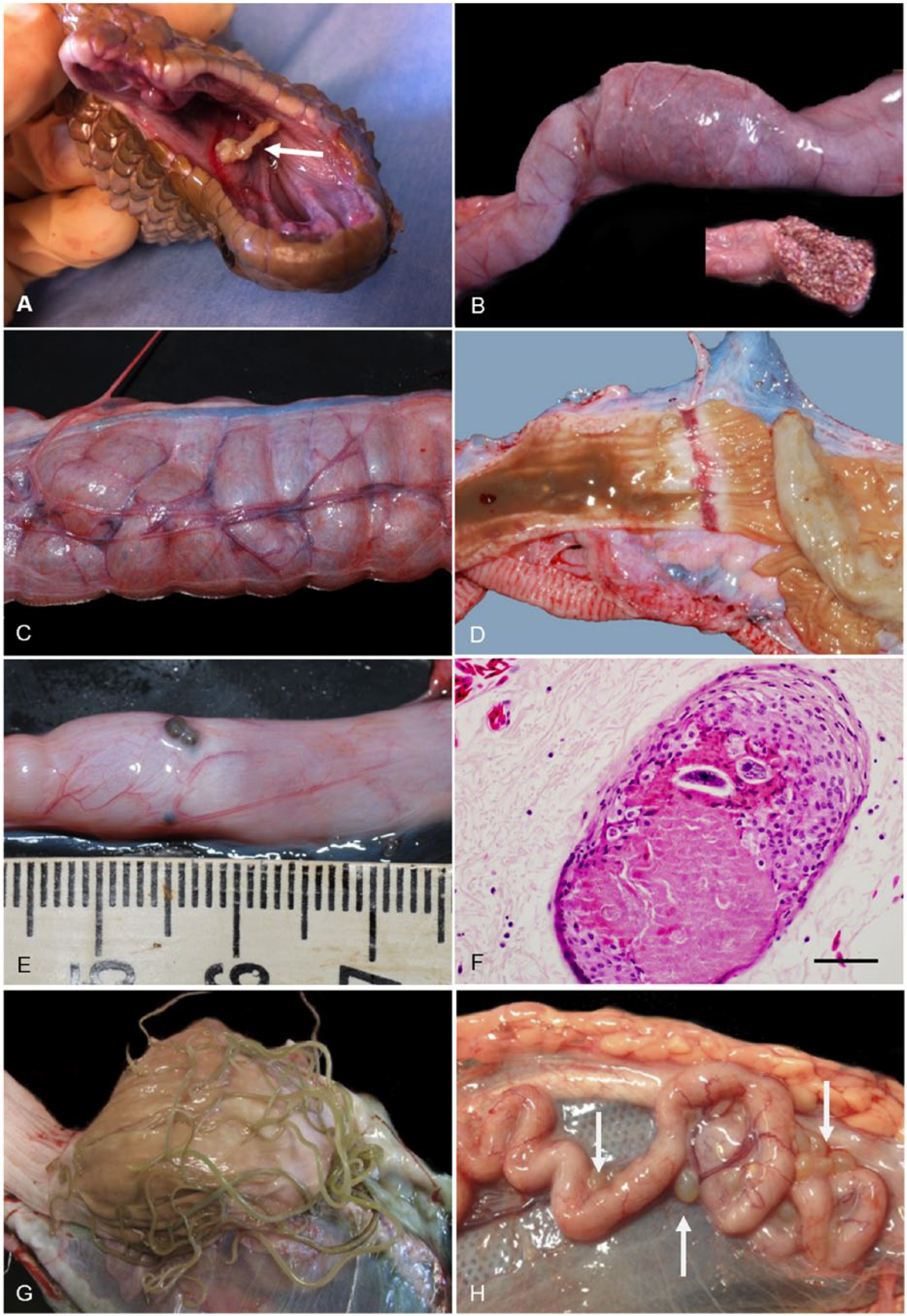
Gastrointestinal lesions in stranded sea snakes from southeast Queensland.
Liver
Focal or diffuse discoloration of the liver may be the result of melanomacrophage hypertrophy and hyperplasia (Fig. 11A) or hepatic lipidosis and vacuolar degeneration of hepatocytes (Fig. 11B). These changes are likely to be a result of chronic debilitation, emaciation, stress, or chronic inflammation1,37 and were identified relatively frequently in stranded sea snakes in SEQ, often in snakes in emaciated-to-poor body condition suffering from various traumatic and inflammatory conditions. Inflammatory lesions were also occasionally noted in the liver of sea snakes, including necrotizing pyogranulomatous or granulomatous hepatitis and hepatic fibrosis. Disseminated bacterial infection (Acinetobacter spp.), affecting the liver, kidney, and mesentery was the most likely cause of one case of hepatitis. A case of hepatic fibrosis was attributed to cardiac insufficiency with portal hypertension. In one snake, the liver had ruptured in a region of chronic heterophilic inflammation and fibrosis (Fig. 11C). A cause for the inflammation was not determined, but the rupture seemed to have been the result of trauma, indicated by the presence of several other injuries. Traumatic injury, including partial and complete bisection of the liver, was evident in some individuals, and was suspected to be caused by inappropriate use (excessive compression) of snake handling tongs commonly used on trawling vessels. Multifocal hyperplasia of bile ducts was a relatively common finding often accompanied by a similar proliferative change in exocrine pancreatic ducts. Adenocarcinoma of possible ductal origin was present in 8 sea snakes and, in each of these, a carcinoma, apparently of ductal origin, was also present in the pancreas. It is not known whether these tumors represent concurrent primary hepatic and pancreatic neoplasms or (more likely) hepatic metastases of a primary pancreatic neoplasm. The latter is supported by the fact that all 8 sea snakes with liver neoplasms were a subset of 13 with pancreatic neoplasms (see below), 2 of which also had renal metastasis, and that hepatic tumors varied from multiple, small disseminated nodules to large, solitary masses (Fig. 11D, 11E). The hepatic tumors consisted of dense aggregates of irregular ducts lined by large cuboidal, columnar, or polygonal cells (Fig. 11F) with 3 mitoses per 10 high-power fields (hpf).
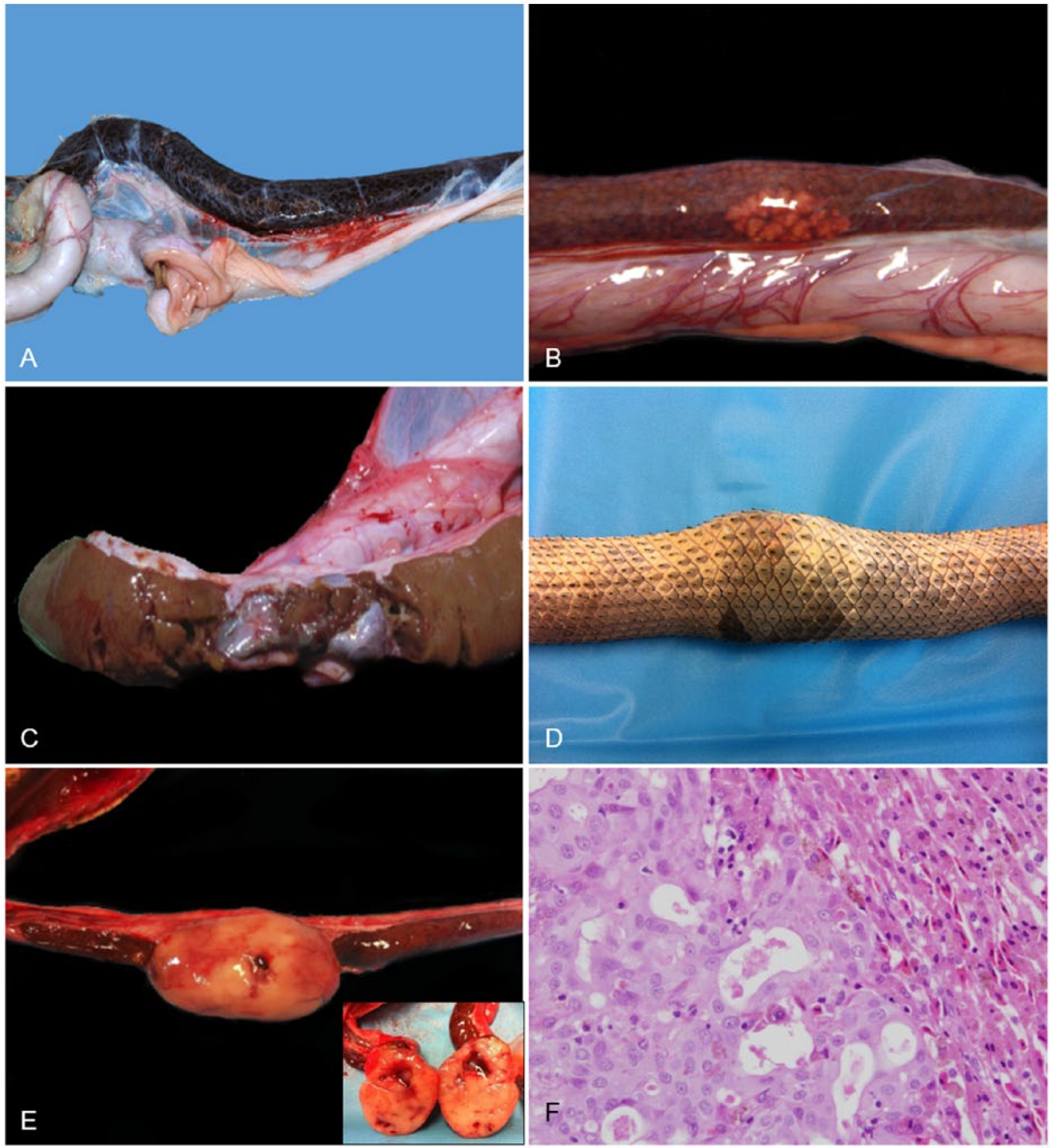
Liver lesions in stranded sea snakes from southeast Queensland.
Pancreas
The exocrine pancreas is a fleshy, smooth organ clearly identified adjoining the spleen (splenopancreas) and small intestine, and is closely associated with the gall bladder (Fig. 12A–C). Gross abnormalities in stranded sea snakes in SEQ included enlargement, discoloration, and a nodular appearance. Nonproliferative histologic lesions included diffuse atrophy of the exocrine pancreas, pancreatitis, necrotizing pancreatitis, granulomatous pancreatitis, larval cestodiasis (plerocercoids), and larval nematodiasis. A spectrum of proliferative lesions was noted in the exocrine pancreatic ducts. Neoplasms consistent with ductal adenocarcinoma were evident in the pancreas of 13 snakes, 9 of which also had multiple foci of ductular hyperplasia in adjacent pancreatic tissue (Fig. 12D). This ductular hyperplasia was not appreciated in the pancreata of animals unaffected by neoplasms. The neoplasms were typically poorly circumscribed and infiltrative, often obliterating the pancreatic tissue and extending into the adjacent spleen. They were composed of large cuboidal, columnar, or polygonal cells forming variably sized ducts in a loose collagenous stroma (Fig. 12E). The neoplastic cells had ample amphophilic cytoplasm and, in 6 of 13 tumors, up to 15% of these cells contained an eosinophilic inclusion of 3–8 µm diameter that was typically located within a well-defined, cytoplasmic vacuole. These inclusions tinctorially resembled zymogen granules but were single and much larger than the zymogen granules in the adjacent pancreatic acinar cells. Transmission electron microscopic evaluation did not clarify the nature of these inclusions, although it remains possible they contain mucus or lipid. The nuclei of neoplastic cells were typically round-to-ovoid with stippled or vesicular chromatin; karyomegaly was prominent in some tumors. Mitotic rate varied from <1 to 5 per 10 hpf, and intra-tumoral necrosis was also often evident. Of the 13 sea snakes with pancreatic adenocarcinomas, morphologically similar neoplasms were present in the liver of 8 snakes, the kidney of 2 snakes, and the spleen of 4 snakes. Splenic involvement likely reflected the direct extension of the pancreatic neoplasm into the adjacent splenic tissue, rather than metastasis. A cause for these tumors has not been identified although a toxin and/or xenobiotic or an infectious agent would seem a plausible cause for proliferative changes within both the biliary and pancreatic ducts. Transmission electron microscopic evaluation of tumor tissues from 2 sea snakes failed to reveal evidence of viral particles.
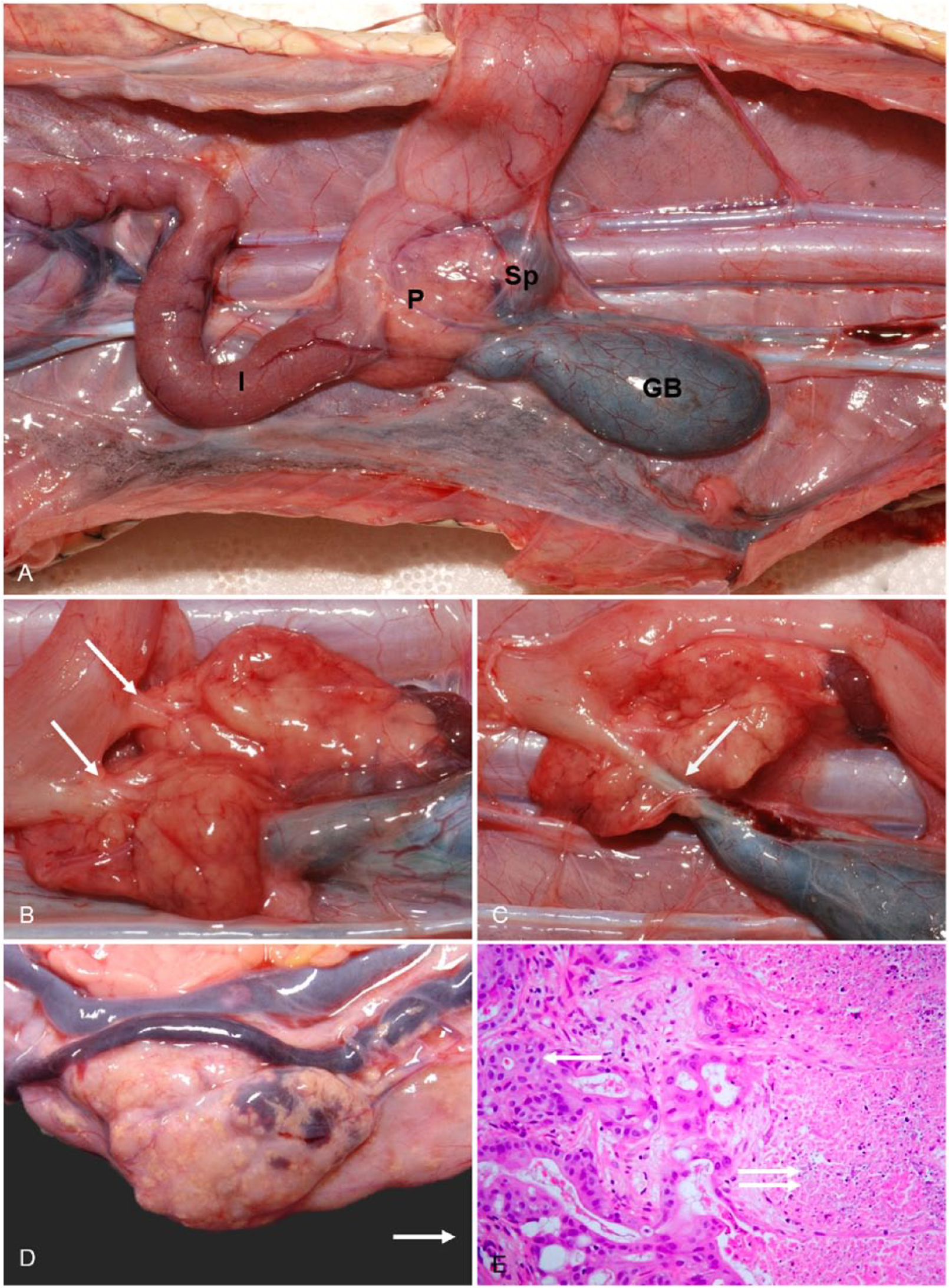
Pancreatic anatomy and lesions.
Urinary system
The mechanisms of electrolyte homeostasis in sea snakes are more complex than in terrestrial snakes given the requirement to maintain lower plasma electrolyte concentrations than their hyperosmotic environment. Their paired kidneys are similar to terrestrial snakes and lie alongside the colon (Fig. 13A) and excrete potassium, magnesium,5,65 and nitrogenous waste (as uric acid) as well as small amounts of sodium (5%). 5 Sodium excretion through the kidneys is insufficient to maintain homeostasis and thus sea snakes employ extra-renal electrolyte excretion via a posterior sublingual salt gland. 12 Similar to terrestrial snakes, sea snakes possess a sexual segment of the kidney 57 primarily in males. Renal lesions were identified histologically in 11 of 82 (13%) stranded sea snakes in SEQ and included granulomatous and lymphocytic nephritis, glomerulonephritis, gout, and (in 2 of 82 snakes) neoplasia (presumed metastatic pancreatic adenocarcinoma).
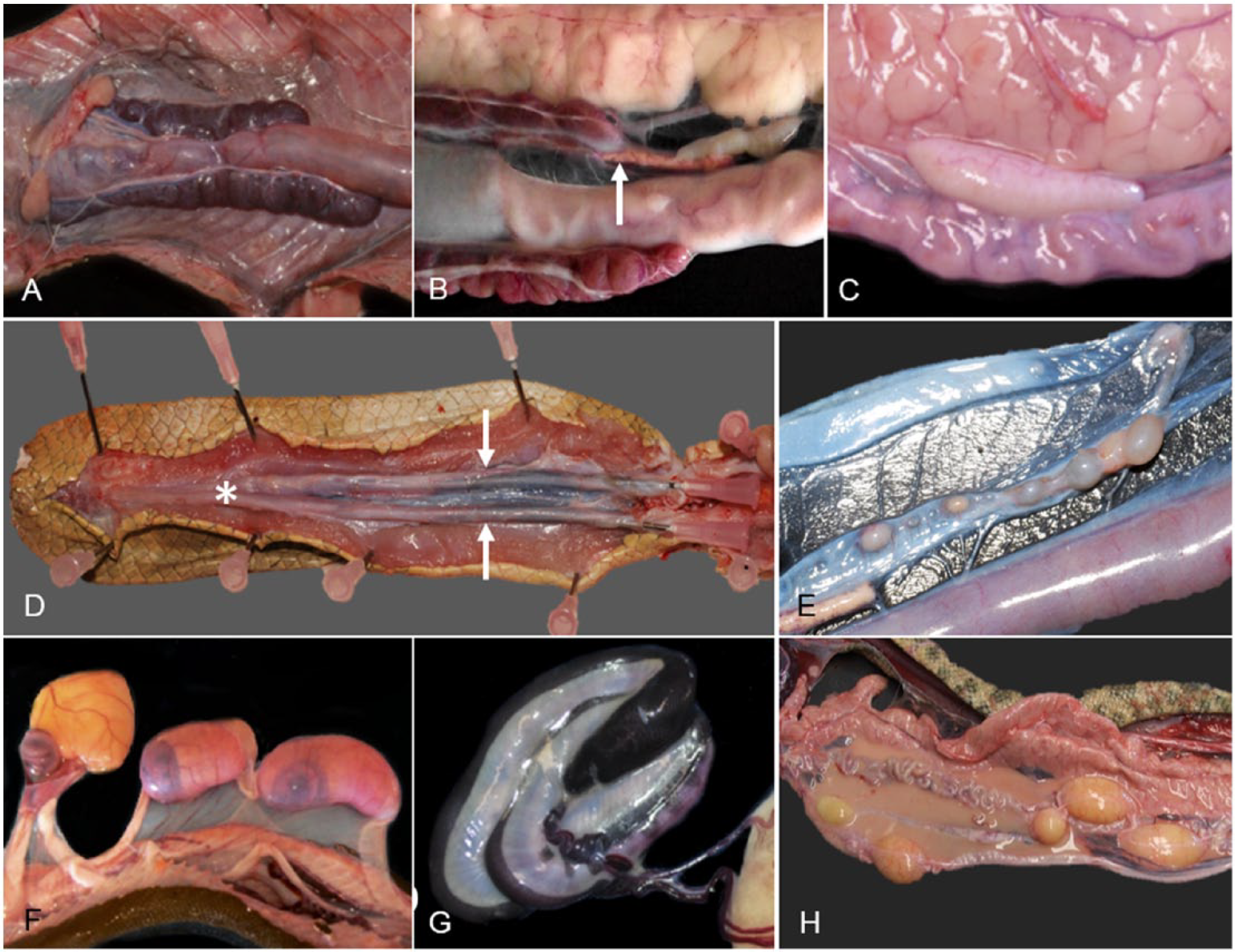
Urogenital system and adrenal glands of sea snakes.
Genital system
Testicles and ovaries in sea snakes are grossly similar to those in terrestrial snakes. The testes are paired, elongate, cream-colored structures situated cranial to the kidneys (Fig. 13C). The testes enlarge in some mature males prior to the appearance of gravid females within a population.24,32 Spermatogenesis in sexually active males is characterized by a prominent lining of seminiferous tubules with spermatozoa within the lumen as well as within efferent ducts and epididymal ducts. There was histologic evidence of active spermatogenesis in 15 of 52 (29%) autopsied stranded adult male sea snakes in SEQ. A seasonal relationship was evident, with peak spermatogenesis during the spring and summer months (September–January; unpublished data). Genital pathology was identified histologically in 4 of 52 (8%) male snakes and included testicular degeneration and atrophy. The hemipenes are bag-like structures, covered in spines and hooks that lie in individual sheaths extending almost the entire length of the tail (Fig. 13D) and are held in place by the retractor penis muscles. During mating, the male will evert one or both hemipenes. Trauma to, or amputation of, the tail may result in concurrent injury or amputation of the hemipenes or retractor muscles, although not observed in sea snakes from the current SEQ study.
The female reproductive system comprises paired elongate ovaries containing follicles at various stages of development, and paired oviducts located in the distal third of the celomic cavity. Hydrophiinae: Hydrophiini sea snakes are viviparous, with gestation lasting up to 9 mo in some species. 32 Reproductive effort is considerably greater in sea snakes than terrestrial snakes; however, clutch size is often much smaller. 33 During nonbreeding periods, the ovaries contain previtellogenic follicles that can be difficult to distinguish macroscopically within celomic fat and connective tissue but appear as small pale-to-white nodules near the intestine (Fig. 13E). Autopsy of 25 stranded female sea snakes in SEQ identified developing oviductal embryos in 4 snakes (1 H. elegans and 3 Hydrophis major) examined in October and November 2009 and near–full-term fetuses in 2 snakes (Hydrophis platurus; Fig. 13F, 13G) examined in March 2010 and April 2012. A number of sea snake species have been observed to give birth to young in autumn (March–May).18,41 Genital lesions were identified histologically in 4 of 25 (16%) stranded female sea snakes in SEQ and included ovarian atrophy, yolk rupture with associated celomitis (Fig. 13H), and heterophilic cloacitis and metritis. Sex was not identified in the remaining (15) autopsied snakes.
Hematopoietic system
The hematopoietic system in reptiles is relatively poorly studied, and there is little published information regarding sea snakes.19,34 The spleen is located in close proximity to the pancreas and gall bladder (Fig. 12A) and distinguishable from the pancreas by its dark-red to purple color. There is one case report of disseminated round cell neoplasia in a captive Laticauda colubrina (sea krait) with infiltration of the heart, lung, liver, intestines, skin, and kidneys by neoplastic cells, and the presence of high numbers of large immature cells in a peripheral blood film consistent with lymphoid leukemia. 10 A stranded male H. major in SEQ had marked leukocytosis (estimated at 40 × 109/L) on peripheral blood film, with high numbers of circulating atypical cells (82% of circulating leukocytes) indicative of a acute myeloid leukemia of probable monocytic lineage (Fig. 14A). Postmortem histopathology revealed infiltration of the splenic parenchyma by large round-to-ovoid cells with often convoluted nuclei, clumped chromatin, and ample amphophilic cytoplasm (Fig. 14B). Multinucleate cells were common, and there were up to 12 mitoses per hpf. Similar cells were also present in hepatic sinusoids, as well as in blood vessels in the lung, kidney, and intestine. Extravascular aggregates of these cells were also evident throughout the wall of the intestine (including in the region of the intussusception in Fig. 10B), in the oral mucosa and submucosa, and within the medullary cavity of bones of the skull. Other splenic changes identified histologically in 6 of 82 (7%) stranded sea snakes in SEQ included lymphoid atrophy, multifocal granulomatous and necrotizing splenitis, melanomacrophage hypertrophy and hyperplasia, and (in 4 of 82 snakes) neoplasia (presumed extension of pancreatic adenocarcinoma).
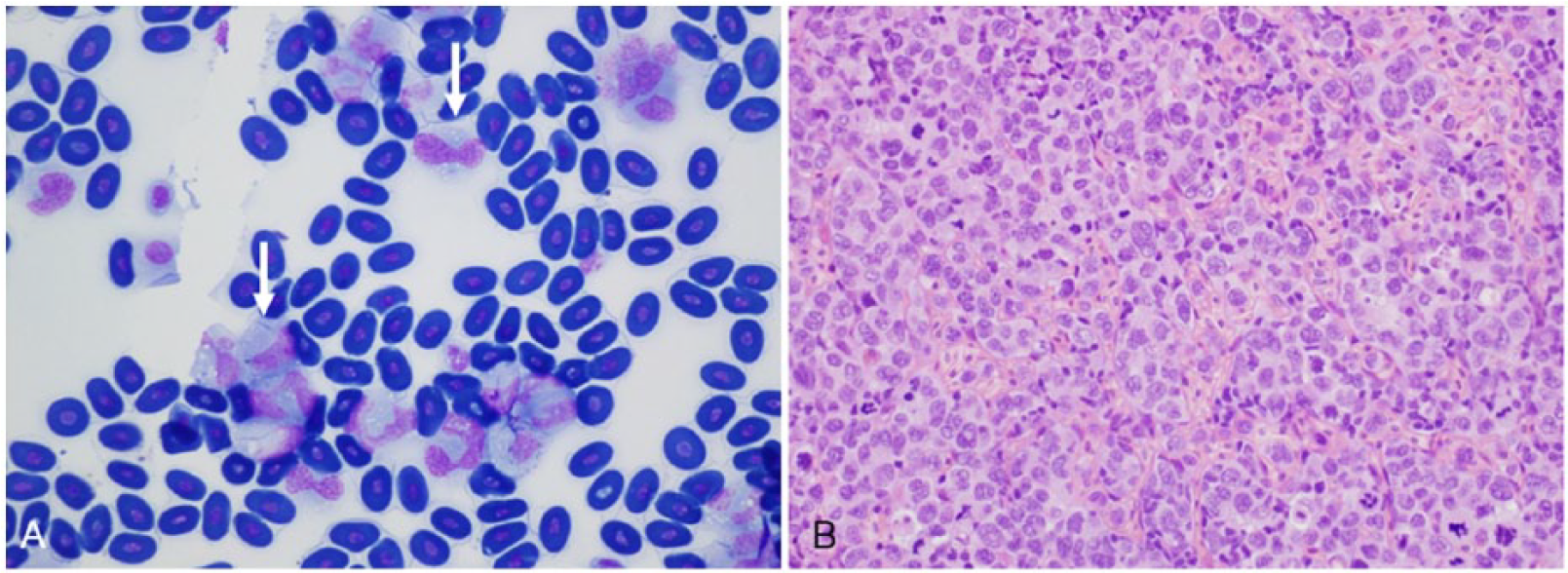
Leukemia in a stranded Hydrophis major from southeast Queensland.
Endocrine system
The thyroid gland is a single organ located proximal to the base of the heart. The adrenal glands are elongate pale structures in the connective tissue cranial to the poles of the kidneys and caudal to the reproductive organs (Fig. 13B).
Nervous system
The central nervous system (CNS) including the brain and spinal cord is considerably more difficult to extract from venomous snakes than from nonvenomous snakes given the inherent risk of envenomation, but it should be included in postmortem examination. The brain can be removed using small bone cutters or rongeurs, either before or after fixation in formalin, by entering the cranial cavity through the foramen magnum, after first removing the skin and muscles overlying the skull. This approach can be used even if the jaws are taped together. The spinal cord can be removed from large snakes by sequentially removing the dorsal portion of the vertebral bodies using fine bone cutters. 43 Alternatively, in small snakes, the vertebral column can be removed and fixed whole in formalin, after first removing the muscles around the column. CNS changes observed histologically in 4 of 82 (5%) sea snakes in SEQ included hemorrhage due to traumatic injury, meningitis, and regional myelopathy (often associated with traumatic injury).
Sensory organs
Eyes
The eyes of sea snakes are essentially spherical, with circular pupils and spherical lenses. 36 The eyes are covered by a thick spectacle, and damage to this structure can result in catastrophic impairment of vision. Ocular injury is one of the most common conditions identified in stranded sea snakes and varies from spectacle damage to traumatic proptosis or complete enucleation. Damage to the nasolacrimal duct, often due to cranial or oral trauma or stomatitis, can result in bullous spectulopathy as a result of disruption of the drainage of the sub-spectacular space. 20 One stranded sea snake from SEQ had a collapsed globe and concurrent histiocytic conjunctivitis of unknown cause.
Olfactory organs
The olfactory system is highly developed in sea snakes and consists of the olfactory epithelium, vomeronasal organ, and Bowman glands, 36 working in conjunction with the tongue for chemoreception. Damage to the head, oral cavity, or tongue can result in significant reduction in chemosensory ability and may lead to starvation, cachexia, and death.
Sampling protocols
Specimen collection and storage for histologic examination
In order to investigate morbidity and mortality in sea snakes, a minimum tissue sample set should include the following: skin, skeletal muscle, heart, lung (tracheal, bronchial, and saccular sections), stomach, small and large intestine, liver, pancreas, kidney, gonad, spleen, endocrine organs (including thyroid and adrenal), brain and spinal cord, bone including bone marrow, eye, and olfactory organs. Tissue samples for formalin fixation should not exceed 5 mm in thickness, except for the brain and spinal cord, which should be fixed whole (and may be fixed within the cranium or vertebral column). Tissues should be fixed in 10% neutral-buffered formalin at a ratio of ~1 part tissue to 10 parts formalin. If buffered formalin is not available, alternative fixatives can be made using sodium phosphate, fresh water, and 37% formaldehyde; sea water and 37% formaldehyde (https://goo.gl/7X5exw); or aqueous neutral-buffered zinc formalin solution (Z-Fix, Anatech, Battle Creek, MI). 51 Tissues require fixation in formalin for at least 24 h before further processing.
Specialized sampling techniques
Fresh tissue or swabs should be collected for culture from lesions where bacterial or viral etiologies are suspected. Fresh tissue samples can also be stored in RNAlater (Thermo Fisher Scientific, Waltham, MA) solution for subsequent molecular investigation including by PCR with appropriate primers, which is increasingly being used for microorganism detection and identification. When parasites are grossly identifiable, they should be collected and fixed appropriately with either hot or cold fixation methods 17 before storing in 10% formalin or 80% ethanol. Tissue samples can also be placed in 10% formalin and/or 3% glutaraldehyde in 0.1 M cacodylate buffer for histologic and ultrastructural characterization of parasites. Fresh frozen tissue samples can be kept for PCR identification of parasites.
Toxicologic analysis
As apex reptilian predators, sea snakes are potential bioaccumulators of toxic compounds, and the potential carcinogenicity, mutagenicity, and teratogenicity of certain pollutants warrants significant concern. Persistent organic pollutants, polybrominated diphenyl ethers, and perfluorinated compounds have been widely reported to accumulate in the fat, blood, liver, and kidneys of fish,2,47 turtles, 38 and other marine species. 62 Polycyclic aromatic hydrocarbons have recently been detected in high concentration in the gut, liver, and kidney of one sea snake species (Hydrophis schistosus; formerly Enhydrina schistosa), 46 and heavy metals (lead, cadmium, copper, vanadium, nickel, and zinc) have been detected in the liver, kidney, skin, and muscle tissues of H. curtus. 56 Specimens of skin, liver, kidney, celomic fat, blood, muscle, and ingested food should be placed in separate plastic bags or containers and stored frozen at −20°C.
Footnotes
Acknowledgements
We thank staff at the Australia Zoo Wildlife Hospital, Sea World (Gold Coast), and Underwater World (Mooloolaba) for assisting with assessment and triage of stranded sea snakes, and the Australia Zoo Rescue Team, Fraser Island Queensland Parks and Wildlife Service rangers Ivan Thrash and Linda Berhendorff, and Dr. Col Limpus for assistance with rescuing and transporting stranded animals for assessment. We also extend our appreciation to Professor Janet Paterson-Kane for histologic evaluation of some cases, IDEXX Laboratories for facilitating processing and assessment of samples, and Lyndal Hulse (The University of Queensland) and Dr. John Mackie (QML Vetnostics) for assisting with images of blood films.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received funding from the Wildlife Disease Association Australasian Section for some histopathological processing, and The University of Queensland, The School of Veterinary Science Donor/Bequest Research Fund BEQPMI2014.
