Abstract
Multiple oncogenic viruses, including lymphoproliferative disease virus (LPDV) and reticuloendotheliosis virus (REV), have been detected in wild turkeys (Meleagris gallopavo). The prevalence of infection with these viruses appears to be more common than overt disease; thus, data on the manifestation of associated disease in wild turkeys are scarce. Diagnostic records from wild turkeys submitted to the Southeastern Cooperative Wildlife Disease Study from 1980 to 2017 were reviewed to identify cases of neoplasia. Neoplasia was reported in 59 of 851 (6.9%) wild turkeys submitted. Of the cases of neoplasia tested by polymerase chain reaction, LPDV was detected in 34 of 58 (59%), REV in 10 of 39 (26%), both viruses in 3 of 39 (8%), and no retroviruses detected in 5 of 39 (13%) turkeys. The most common gross lesions observed among turkeys with neoplasms were emaciation (30/40; 75%); nodules in the skin (26/59; 44%), liver (17/59; 29%), or spleen (9/59; 15%); and splenomegaly (14/59; 24%). Microscopically, nodules were composed of pleomorphic round cells with large eccentric nuclei and prominent nucleoli resembling lymphocytes or lymphoblasts (57/59; 97%) except for 2 cases, one of myeloid cell origin and the other with primarily spindloid cells. This study indicates the need to characterize the pathogenesis and potential health threat posed by REV and LPDV to wild turkeys. Experimental infection studies and the development of additional diagnostic tests to confirm the role of retroviruses in lymphoproliferative disease are warranted.
Keywords
Wild turkeys (Meleagris gallopavo) are a popular game species across much of North America and are an aesthetically and economically important natural resource. 3 Efforts by state and federal wildlife agencies and independent nonprofit organizations have successfully led to increased wild turkey population numbers over the past several decades; however, more recently, anecdotal evidence of turkey population declines reported in numerous states have caused concern. 7,8 Wild turkeys are susceptible to a variety of infectious diseases, such as avian pox, histomoniasis, and mycoplasmosis, among others. Furthermore, the role of viruses in inducing neoplastic disease in wild turkeys has become a recent topic of investigative interest. 10,18,20,23,29
Several oncogenic viruses have been associated with tumor development in domestic poultry, including domestic turkeys. 26,28 The most well characterized of these viruses include retroviruses such as avian leucosis virus (ALV) and the closely related Rous sarcoma virus and reticuloendotheliosis virus (REV; family Retroviridae; genus Gammaretrovirus), as well as the herpesvirus that causes Marek’s disease (MD). 25,28 Avian leucosis virus is the most common and economically significant virus of the leucosis/sarcoma virus group in domestic poultry. 27,33 Depending on the strain, this virus causes a variety of neoplastic and nonneoplastic conditions in chickens, including erythroblastosis, nephroblastoma, and osteopetrosis, among others. 14 REV can cause diverse disease syndromes in turkeys, chickens, ducks (Anas spp.), geese (Anser spp.), quail (Colinus spp.), and prairie chickens (Tympanuchus cupido), including lymphoproliferative lesions, acute reticular cell neoplasms, and nonneoplastic runting disease syndromes. 5,26
Lymphoproliferative disease of domestic turkeys was first described in the 1970s in the United Kingdom and was not reported again until several years later in Israel. 4,19 At this time, the disease was attributed to lymphoproliferative disease virus (LPDV; family Retroviridae; genus Alpharetrovirus) infection with multiorgan pleomorphic infiltrates of lymphocytes, plasma cells, lymphoblasts, and reticular cells in 7- to 18-week-old birds. 26 Similar lesions have historically been reported in wild turkeys; however, detection of LPDV in North America was not attempted until 2009. 9,10
Since its initial detection in wild turkeys in the United States in 2009, LPDV has garnered the attention of hunters, biologists, and wildlife veterinarians. 2 Subsequent surveillance has revealed that this virus is commonly detected and widespread in wild turkey populations in the United States and Canada. 1,22,32 Infection in wild turkeys has been associated with multiorgan lymphoid proliferation similar to that reported in domestic turkeys. 2 However, the potential impacts of LPDV on the health of individuals and wild turkey populations are poorly defined. While vertical and horizontal transmissions, both via direct and indirect contact, are presumed mechanisms of transmission of most retroviruses in domestic poultry, the mechanisms of transmission that resulted in widespread infection of wild turkeys with LPDV across North America are unknown, but experimental studies in domestic birds have shown horizontal transmission between groups in close contact. 2,28
The purpose of this study was to describe the gross and histological lesions, geographic distribution, and demographic parameters of wild turkeys with neoplastic disease submitted to the Southeastern Cooperative Wildlife Disease Study (SCWDS) over a 37-year period. In addition, we assessed for an association between neoplastic disease and evidence of infection with retroviruses, particularly LPDV and REV.
Materials and Methods
Diagnostic case records of 851 wild turkeys submitted to the SCWDS from January 1, 1980, through December 31, 2017, were reviewed for diagnoses of neoplasia. These cases were defined as individual wild turkeys with microscopic evidence of neoplastic lesions in 1 or more organs. Data compiled for each case included state and county of origin, year submitted, sex, age class, gross and microscopic lesions, and ancillary test results. Age estimates were categorized as poult (up to 1 year), subadult (1 year and up to 2 years), or adult (2 years and older). 31
Samples submitted for examination varied from whole carcasses to formalin-fixed and/or fresh tissues of select organs collected at field necropsy. Tissues representing major organ systems were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 5 μm, mounted on glass slides, and stained with hematoxylin and eosin for histologic evaluation. Immunohistochemical labeling to characterize neoplastic round cells was attempted on 4 cases (3 LPDV positive and 1 REV positive) using CD3 (1:1000, A0452; Dako, Carpinteria, CA) and CD79a (1:50, CM067C; BioCare Medical, Pacheco, CA). In addition, 1 turkey with unique cell lineage underwent Luna staining according to published methods. 30
Polymerase chain reaction for retroviral DNA was done either at SCWDS (LPDV) or the Poultry Diagnostic and Research Center (REV, ALV, and avian herpesviruses), both at the University of Georgia. For polymerase chain reaction (PCR) for all viruses, DNA was extracted from tissues in which lesions were observed or bone marrow using a QIAamp DNA mini kit (Qiagen, Valencia, CA) or from formalin-fixed, paraffin-embedded (FFPE) tissues using a QIAamp FFPE DNA kit according to the manufacturer’s guidelines. LPDV PCR was performed using primers based on the Israeli prototype strain of LPDV that covered a region spanning the p31 and CA genes (5′-ATGAGGACTTGTTAGATTGGTTAC-3′, 5′-TGATGGCGTCAGGGCTATTTG-3′). PCR products were amplified using a GoTaq Flexi DNA polymerase system (Promega, Madison, WI). REV PCR was performed using primers rev-1 and rev-2 (5′-CATACTGGAGCCAATGGTT-3′, 5′-AATGTTGTACCGAAGTACT-3′) to amplify a 291–base pair region of the env gene. Tissues were selected for testing varied based on gross and microscopic lesion severity, availability of stored tissues, and previous experience and were most commonly bone marrow, liver, and skin. 2,32 When fresh or formalin-fixed tissues were not available, DNA was extracted from scrolls of FFPE tissues and used for PCR.
Results
Details on number of cases, location of origin of each case, tissues affected, and results of virus testing are provided in Supplemental Table S1. Evidence of neoplasia was identified in 59 of 851 (6.9%) wild turkey cases submitted to the SCWDS from 1980 to 2017. Case submissions included 2 of 59 (3%) from 1980 to 1989, 5 of 59 (8%) from 1990 to 1999, 5 of 59 (8%) from 2000 to 2009, and 47 of 59 (80%) from 2010 to 2017. Thirty-two (54%) turkeys were male, 25 (42%) were female, and sex was unrecorded in 1 (2%). Forty-seven (80%) turkeys were adults, 10 (17%) were subadults, and 1 was unrecorded (2%). Turkeys with neoplasia originated from a widespread geographic region, including Alabama, Arkansas, Florida, Georgia, Kentucky, Louisiana, Michigan, Mississippi, Missouri, New Jersey, North Carolina, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia.
Fresh tissues were tested for LPDV and REV by PCR in 49 of 59 (83%) and 12 of 59 (20%) cases, respectively, and from FFPE in 9 of 59 (15%) and 27 of 59 (46%) cases, respectively. Among 58 cases tested for LPDV, DNA was detected in 34 (59%) cases, all of which were from fresh tissue. No LPDV DNA was detected from FFPE blocks in cases submitted prior to 2009 (the first year of LPDV detection in a wild turkey in the United States). Among 39 cases tested for REV, DNA was detected in 10 cases (26%), 5 of which were from FFPE blocks and 5 from fresh tissue. Both viruses were detected in 3 of 39 (8%), and no viruses were detected in 5 of 39 (13%) cases in which both viruses were tested. The details of viral testing results are in Supplemental Table S1.
Most cases (40/59; 69%) included full postmortem examination. Among these, 30 of 40 (75.0%) carcasses were in poor nutritional condition. Gross skin lesions consisting of diffuse or focally extensive subcutaneous edema associated with dermal round cell neoplasms with ulceration (case No. 49 in Suppl. Table S1; Fig. 1) or multifocal, raised, crusted, tan to yellow nodules (case No. 52; Fig. 2) on the nonfeathered skin were observed in 26 of 59 (44%) cases. However, 4 of these had concurrent microscopic lesions diagnostic for avian pox virus infection (ie, epithelial hyperplasia and ballooning degeneration of keratinocytes with intracytoplasmic, eosinophilic inclusion bodies; case No. 7, Fig. 8). Multifocal, white to yellow, pinpoint to nodular foci were detected in the spleen (9/59, 15%) and liver (17/59, 29%) (Fig. 3 [case No. 22] and Fig. 4 [case No. 19], respectively). Splenomegaly, including those containing pinpoint/nodular foci, was reported in 14 of 59 cases (24%) (Fig. 3). Other less common gross findings associated with neoplastic cells included white plaques on the mucosal surface of the esophagus, crop, and/or pharynx (5/59, 9%); mottling on the surface of the kidneys (3/59, 5%); pale streaks or nodules in the myocardium (3/59, 5%); white plaques in the mucosal lining of the small or large intestines (2/59, 3%); and 1 each of the following (1/59; 2%): pinpoint tan plaques on the air sacs, dark purple masses in the muscular layer of the esophagus, markedly thickened crop wall, and petechial hemorrhages on the pancreas.
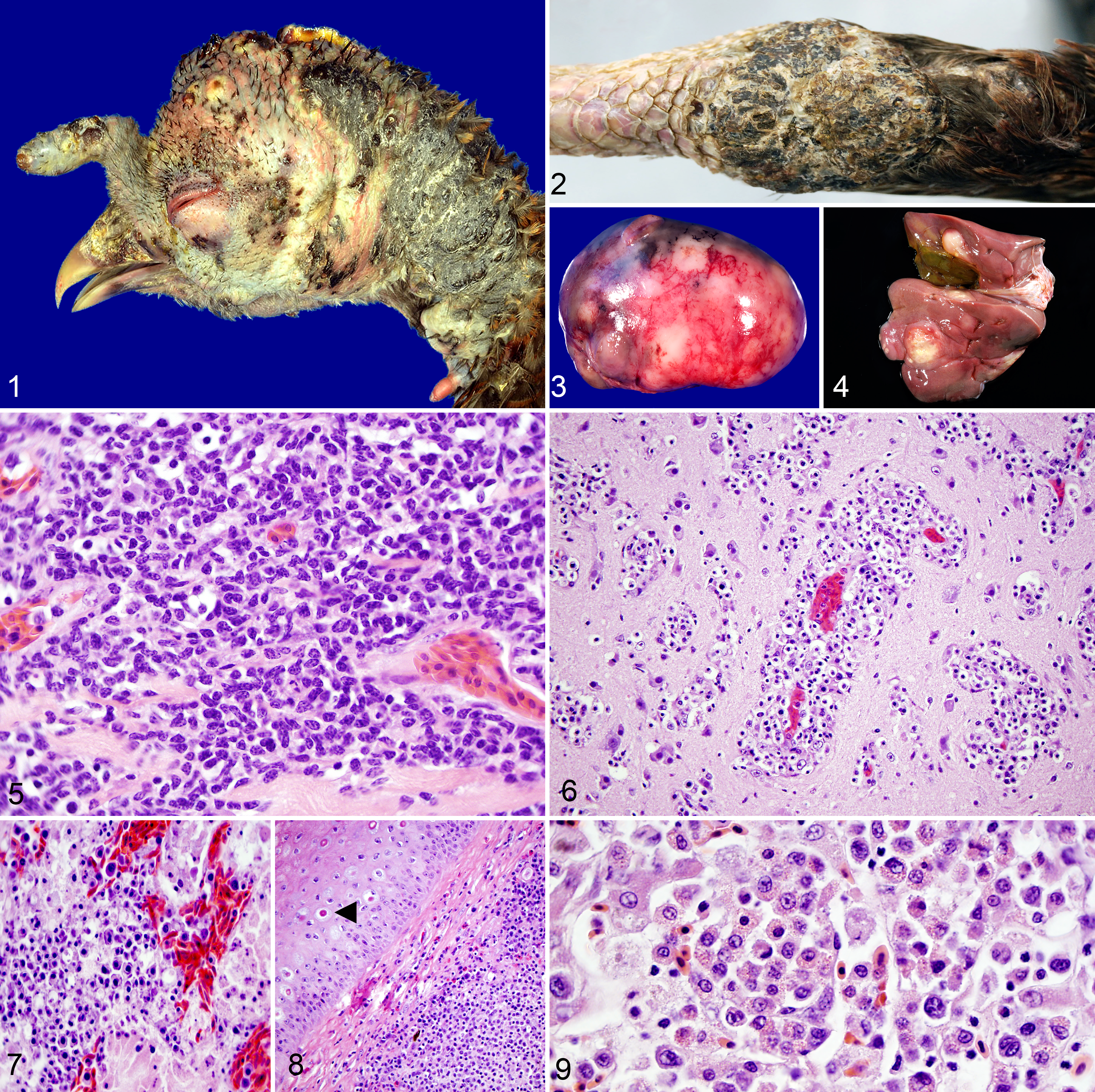
Lymphoma, wild turkey.
Microscopically, in the majority of turkeys (57/59; 97%), the pattern and predominant cell type within neoplasms consisted of sheets of pleomorphic round cells with dense, well-defined cell borders; round, basophilic nuclei with finely stippled chromatin; and variable amounts of pale, eosinophilic to basophilic cytoplasm. These cells morphologically resembled lymphoblasts with fewer plasma cells (case No. 14, Fig. 5). Mitotic figures were rare (<1 per high-power 40× field), and anisocytosis and anisokaryosis were mild to moderate. In affected skin, the dermis was often expanded by these neoplastic cells usually with overlying epidermal ulceration and hyperkeratosis. Neoplastic cells in the liver were often distributed in the parenchyma surrounding central arteries and veins with sinusoidal expansion to complete architectural effacement. Neoplastic cell infiltrates were also in the myocardium, brain (case No. 22, Fig. 6), renal interstitium (case No. 51, Fig. 7), skeletal muscle, esophagus, lung, crop, and spleen of some turkeys. One turkey had single-cell hepatocellular necrosis in the liver and salpingitis and oophoritis associated with neoplastic cell infiltration.
Two LPDV-positive turkeys had strong, granular, cytoplasmic immunohistochemical labeling for CD3 in medium to large neoplastic round cells in the kidney and heart (case No. 48) as well as skin (case No. 49). One REV-positive case (case No. 51) had rare, weak, granular, cytoplasmic CD3-immunolabeling of round cells in the liver. No immunolabeling for CD79a was evident in tissues from any of these 3 turkeys.
Coinfection with LPDV and turkey herpes virus (THV) was diagnosed in 1 turkey (case No. 25). This neoplasm consisted of multiorgan round cell invasion of the liver, spleen, bone marrow, heart, lung, brain, skeletal muscle, proventriculus, ventriculus, and adrenal gland. The neoplastic round cells were consistent with myeloid cells, with large, round to ovoid, occasionally indented nuclei with coarsely clumped chromatin and 1 to 2 prominent nucleoli, and well-defined, pale, eosinophilic cytoplasm filled with bright, eosinophilic granules (Fig. 9). Neoplastic cells did not stain with Luna stain and had no immunohistochemical labeling for CD3 or CD79, ruling out eosinophils, T lymphocytes, and B lymphocytes, respectively.
An additional case (case No. 31) had multiple, tan-white nodules in the liver and a similar, single nodule in the spleen. These masses consisted of streams of spindloid cells with variably distinct cell borders; sparse, wispy eosinophilic cytoplasm; and a centrally located, oval to oblong nucleus with finely stippled chromatin and nondistinct nucleoli. Cell pleomorphism was mild with individualized, binucleated cells with cytomegaly and karyomegaly throughout the neoplasm. Neoplastic tissues from this turkey tested negative for REV, LPDV, and ALV subtype J by PCR.
Discussion
Wild turkeys are common across the southeastern United States and a popular game species. This study evaluated a large data set of over 800 birds over a 37-year period and demonstrated that lymphoid neoplasia is not uncommon (∼7% prevalence). Lymphoproliferative disease virus was detected in over half of these cases, less commonly REV, and 3 turkeys concurrently infected with LPDV and REV had lesions that could be attributed to either virus, precluding definitive identification of a single cause. The higher percentage of detections of LPDV and REV in the past 7 years may be related to increased submission and testing.
Reticuloendotheliosis (without LPDV coinfection) was diagnosed in 6 wild turkeys submitted to the SCWDS since 1980. In most reticuloendotheliosis cases, gross lesions in the liver and spleen were similar to those observed in wild turkeys with LPDV-associated neoplasia. REV-infected wild turkeys did not have concurrent skin lesions, unlike those with LPDV. In past studies, skin lesions were not commonly reported in domestic turkeys but were seen in Attwater’s (Tympanuchus cupido attwateri) and greater (T. cupido) prairie chickens infected with REV. 12,24,25,34 In 1 study, REV rarely resulted in diarrhea and enteritis in domestic turkeys, and round cells were observed in the small intestines of 2 birds with REV infection. 24
In the present study, the first case of reticuloendotheliosis was detected in 1980, which suggests that REV and/or LPDV likely has been present for many years in wild turkeys prior to the first published reports. Two previous reports of reticuloendotheliosis in wild turkeys exist, only one of which confirmed clinical disease. 17,21 In addition, MD-like lesions were reported in 2 wild turkeys from Florida, but laboratory tests for the virus were negative, and MD has not been confirmed in wild birds. 6,11 One published report suggested ALV as a potential cause of disease in a wild turkey based on abnormal calcium deposits and diffuse periosteal proliferation, similar to what has been observed in domestic poultry. However, definitive identification of this virus in wild turkeys has not been confirmed. 15
Similar to previous studies, the present data suggest that LPDV prevalence is high in wild turkeys in the eastern United States and central Canada, and coinfections with REV and LPDV are common. 2,22 The lower prevalence of REV compared to LPDV in the present and previous studies may be falsely low due to decreased sensitivity of PCR using FFPE tissues vs fresh-frozen tissues. 2,22 The wide host range and widespread distribution of REV in the United States, concurrent with detections in wild turkeys, warrant further investigations into its epidemiology and pathogenesis. 17,20,21,29
Over half of the turkeys with neoplastic disease (including 5 of 6 with reticuloendotheliosis) for which carcasses were examined were in poor nutritional condition, which suggests that neoplasia associated with viral infection may adversely affect the overall health status of these birds. The most common gross lesions reported were multifocal nodular lesions on the nonfeathered portions of skin in the head, neck, and legs. These lesions can easily be confused with nodular lesions caused by avian poxvirus, but histologic lesions allow for differentiation of these diseases. 13 Unlike wild turkeys with LPDV and REV, skin lesions have not been associated with lymphoproliferative disease in domestic birds, for unknown reasons. 4,16 Pale proliferative foci in the liver and/or spleen were the next most commonly observed lesions in wild turkeys with LPDV-associated disease, followed by splenomegaly. Both gross and microscopic lesions observed in the present study are similar to those reported in domestic turkeys with LPDV-associated neoplastic disease and reticuloendotheliosis. 24,25 Furthermore, the cell morphology and patterns of distribution within tissues appear to be similar in both wild and domestic turkeys. 2,4 This suggests that histopathology should not be used as the sole method to distinguish these 2 diseases.
Demographic data from wild turkeys in the present study suggest that there was no sex predilection for LPDV detection or disease but that age may play a role in infection status. For example, round cell neoplasia, including those with LPDV or REV infection, was diagnosed in adult and subadult wild turkeys but not in poults. This differs from domestic turkeys, in which poults are the age class most often documented with clinical disease associated with LPDV and REV. 4,24 In prairie chickens, the ages at the time of REV-associated lymphoma development and mortality ranged from 1 to 4 years in 1 study and were consistently less than 14 months in another. 12,34 Due to their small size and limited mobility, sick or dead wild turkey poults are less likely to be found and submitted as diagnostic specimens compared to older, larger birds. As a result, the impact of these viruses on wild turkey poults, as well as the full range of associated clinical syndromes and the potential effects on wild turkey populations, is unknown. Furthermore, the virulence and pathogenesis of LPDV and REV in wild and domestic turkeys have yet to be fully elucidated.
A single turkey in the present study had a neoplasm affecting the liver, spleen, heart, lungs, brain, ventriculus, and proventriculus, comprising a round cell of different morphology from the above, suggestive of myeloid origin. This turkey was coinfected with LPDV and THV. To the authors’ knowledge, neoplasia originating from myeloid cells has not yet been reported in a wild turkey. The histologic classification of the spindle cell neoplasm in 1 turkey was not determined.
The 2 LPDV-positive (REV-negative) infected turkeys assessed by immunohistochemical labeling had strong CD3 labeling in the majority of neoplastic round cells in various tissues, indicating abundant neoplastic T lymphocytes. One REV-positive and 1 THV- and LPDV-coinfected turkey had no immunolabeling for CD3. The presence of B lymphocytes could not be adequately assessed due to lack of available B-cell markers validated for birds. CD79a labeling was attempted in tissues from these birds but did not provide reliable results, presumably due to lack of antibody cross-species reactivity. There is additional need to characterize neoplastic round cells in future research.
Bone marrow has been determined to be the most sensitive tissue for LPDV detection but is not routinely collected and submitted for histopathology. 32 In the present study, wild turkey cases with lymphoid neoplasia but from which no viruses were detected may reflect true, non-virus-associated spontaneous neoplasia. However, false virus-negative results are a possibility and may be attributable to poor sample quality (eg, long postmortem intervals, numerous freeze-thaw cycles), lack of ideal tissue selected for testing, and poor extraction recovery of viral nucleic acids from FFPE blocks. Other study limitations include those inherent to long-term retrospective data collection. These may include inconsistencies in provided case histories, variations in sample processing and necropsy techniques, and differing availability of ancillary tests at the time of diagnosis. In addition, submission biases exist across the geographic region of study, as some state wildlife agencies may be more likely to submit diagnostic samples, which may be deemed higher priority for submission in specific seasons (eg, spring or fall hunting season) or with a particular gross presentation.
This study highlights the importance of testing for multiple viruses in wild turkeys with lymphoid neoplasia, as gross and histological lesions alone are unreliable and nonspecific. While REV is much less commonly reported in wild turkeys, fewer cases were historically tested for REV compared to LPDV in the present study. Likewise, testing was seldom performed for ALV and herpesviruses, and it is important to broaden diagnostic viral testing panels to include these viruses, particularly in cases where LPDV or REV is not detected. Gaining a better understanding of the prevalence of virus infection and disease in wild turkeys is important to better assess the potential role of these viruses and associated disease in wild turkey population health.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819864306 - Multicentric Round Cell Neoplasms and Their Viral Associations in Wild Turkeys (Meleagris gallopavo) in the Southeastern United States
Supplemental Material, DS1_VET_10.1177_0300985819864306 for Multicentric Round Cell Neoplasms and Their Viral Associations in Wild Turkeys (Meleagris gallopavo) in the Southeastern United States by Kevin D. Niedringhaus, Nicole M. Nemeth, Holly S. Sellers, Justin D. Brown and Heather M. A. Fenton in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank the many SCWDS diagnosticians and staff that contributed their expertise to these cases. They also thank the state wildlife agencies that support the SCWDS and for their contribution of case material. The Athens Veterinary Diagnostic Laboratory provided technical laboratory expertise, including histology processing. Sarah Coker and Kayla Guinn at the SCWDS and Erich Linnemann at the Poultry Diagnostic Research Center assisted with molecular testing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the sponsorship of the Southeastern Cooperative Wildlife Disease Study by the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia. Support from the states to the SCWDS was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
