Abstract
The microsporidia Nosema apis and Nosema ceranae are major honey bee pathogens that possess different characteristics in terms of the signs they produce, as well as disease development and transmission. Although the ventricular epithelium is generally considered the target tissue, indirect observations led to speculation that N. ceranae may also target other structures, possibly explaining at least some of the differences between these 2 species. To investigate the tropism of Nosema for honey bee tissues, we performed controlled laboratory infections by orally administering doses of 50 000 or 100 000 fresh mature spores of either species. The fat body was isolated from the infected bees, as well as organs from the digestive (esophagus, ventriculus, ileum, rectum), excretory (Malpighian tubules), circulatory (aorta, heart), respiratory (thoracic tracheas), exocrine (hypopharyngeal, mandibular and labial, cephalic, thoracic salivary glands), and sensory/nervous (brain, eyes and associated nerve structures, thoracic nerve ganglia) systems. Tissues were examined by light and electron microscopy at 7, 10, and 15 days postinfection. Both Nosema species were found to infect epithelial cells and clusters of regenerative cells in the ventriculus, and while the ileum and rectum contained spores of the microsporidia in the lumen, these structures did not show overt lesions. No stages of the parasites or cellular lesions were detected in the other organs tested, confirming the high tropism of both species for the ventricular epithelium cells. Thus, these direct histopathological observations indicate that neither of these 2 Nosema species exhibit tropism for honey bee organs other than the ventriculus.
Microsporidia are obligate intracellular eukaryotic parasites included in the kingdom of Fungi. 1 However, Microsporidia were more recently considered highly derived parasitic protists, sisters of Cryptomycota/Rozella, or diverging as the next branch below fungi. 2,27 Nosema apis was the first microsporidium known to infect western honey bees (Apis mellifera), 42 and it has since been found on all continents. 15 At the beginning of the 21st century, another microsporidium—Nosema ceranae—was also seen to naturally infect A. mellifera colonies in Europe, 24 and its worldwide spread in this and other Hymenopteran hosts was also subsequently confirmed. 32
Both these microsporidia may negatively affect the longevity of the individuals in a honey bee colony, disrupting age polyethism with an intensity that reflects their prevalence in the colony. 18,32 However, the clinical signs of Nosema infection differ depending on the causative agent, both at the individual and colony level. 23,25,32 For example, the typical characteristics of N. apis infection include diarrhea, observable bee mortality around the hive, and seasonality, whereas N. ceranae infection results in colony dwindling and collapse in all seasons. All adult individuals of a colony, independent of caste and sex, are susceptible to Nosema infection, and there is evidence from both in vitro rearing experiments 10 and studies of field colonies 37,41 that Nosema spp. can even infect immature honey bee stages. Indeed, under natural conditions, either coinfection or infection with a single Nosema species is possible. 30,33,35
The transmission of Nosema infection occurs mainly by the ingestion of spores in contaminated food and water, during nest cleaning activities, or via food exchange. 38 There is also evidence that both Nosema species can be sexually transmitted to a healthy queen through contact with spore-containing semen during mating, although there is no evidence of subsequent transovarian transmission to offspring. 36 Both N. apis and N. ceranae infect epithelial cells of the ventriculus (or midgut), ultimately impairing the digestive capacity of this organ. 12,20 Both species have a similar life cycle, 18,29 summarized as (1) the initial spore penetration into the midgut lumen; (2) extrusion of the polar tubule and transfer of the sporoplasm into a host cell; (3) merogony, consisting of repeated binary fission of parasitic cells; and (4) sporogony, the production of new spores that can infect the same or adjacent cells (primary spores) or be released into the midgut lumen (environmental spores). This last stage of spore release suggests that there has been host cell rupture and damage to the ventricular epithelial layer.
There have been suggestions that N. ceranae might also infect other honey bee organs, 6,7,17 although this issue remains unclear, 20 and there is no histopathological evidence that N. ceranae produces lesions in extraventricular tissue or of the presence of different stages of its life cycle at these sites. If evidence were to suggest that N. ceranae is not tissue specific, it would require a revision of our understanding of its life cycle,[14] paving the way for new hypotheses about the differential pathogenesis and epidemiology of the 2 Nosema species. Accordingly, the objective was to assess the histologic lesions and the stages of the Nosema life cycle in extraventricular tissues after experimental infection with these parasites.
Materials and Methods
According to European legislation (EU Directive 2010/63/EU 8 ), no specific permits were required for use of honey bees for this study. The biological material studied here was collected from Apis mellifera iberiensis colonies reared at the CIAPA-IRIAF (Marchamalo, Spain). One comb containing sealed worker brood was taken from a Nosema-free colony and maintained in an incubator at 34 ± 1°C until the adult worker honey bees emerged. 32 Newly hatched bees were carefully removed, transferred to cages, maintained at 33°C in an incubator, and fed ad libitum with a sucrose solution (50% w/w in distilled water) combined with 2% Promotor L (a commercial mixture of amino acids and vitamins; Calier Lab, Les Franqueses del Valles, Barcelona, Spain). Honey and pollen were not used to feed the bees to avoid potential contamination with infective Nosema spp. spores. 31
Viable and infective N. apis and N. ceranae spores were obtained from naturally infected Spanish honey bee colonies sent to our laboratory for pathological studies as described previously. 29,31 Briefly, infected honey bees were macerated in distilled water (polymerase chain reaction [PCR] grade) and centrifuged for 6 minutes at 800 g. A pellet of mature spores was recovered, Percoll purified, and confirmed to represent a single species by PCR. 30 To homogenize and revitalize the spores, 100 laboratory-born 5-day-old bees were fed for 2 weeks with the same syrup as indicated above but containing 1 million spores of either N. ceranae or N. apis. Subsequently, these bees were anesthetized with CO2 and processed as described above to obtain Percoll-purified spores of the 2 individual Nosema species. The spores obtained were stored at room temperature for less than 24 hours before use.
Experimental honey bee infection was performed by individual oral administration when the bees were 5 days old. Water (2 μl) containing 5 or 10 × 104 fresh Percoll-purified N. ceranae or N. apis spores was offered to each bee using a micropipette, 20,31,32 discarding those bees that did not ingest the entire dose. Other bees from the same batch received 2 μl of distilled water alone and served as uninfected controls. An intentionally high dose was administered relative to the approximate infective dose indicated elsewhere (for both Nosema species: 11 ID100 = 1 × 104 spores) to promote a rapid onset of infection and to favor a wider spread to tissues for which the pathogens might show weaker tropism.
Five groups of bees were established in total, each distributed among 3 replicate cages containing 25 bees. The groups were coded as follows: A5, A10, C5, C10, and T0, where the letter indicates A = N. apis, C = N. ceranae, and T = uninfected control, and the number indicates challenge with 5 or 10 × 104 spores. Groups with different letters were reared at 33°C in separate incubators to avoid cross-infection. Sugar water with added 2% Promotor L was provided ad libitum as food, and the bees were analyzed on 7, 10, and 15 days postinfection (dpi), when a notable increase in ventricular infection was expected in the conditions of rearing used. 28 As such, 8 bees were taken at random from each group at 7 and 10 dpi, and 4 bees were taken from each A5, C5, and T0 groups at 15 dpi. It was not possible to take sufficient bees from the A10 and C10 groups at 15 dpi due to mortality at this timepoint. Of the bees collected at 7 and 10 dpi, 4 were used for histological studies by light microscopy (LM) and the other 4 were studied by transmission electron microscopy (TEM). Of the bees obtained at 15 dpi, 2 were used for LM studies and the other 2 for TEM studies.
The tissues and organs examined from each bee were brain, eyes, and associated nerve structures; hypopharyngeal, mandibular, and salivary (labial, cephalic, thoracic) glands; esophagus and abdominal digestive tract (ventriculus, ileum, rectal ampulla, Malpighian tubules); thoracic nerve ganglia; aorta and heart; thoracic trachea; and the fat body. The organs and tissues obtained were prepared for LM and TEM, as described previously. 16,21 Briefly, for LM, the organs were fixed in 10% buffered formalin for 24 hours and embedded in paraffin wax, and 4-μm sections were stained with hematoxylin-eosin (HE). For TEM, the tissue was prefixed in a 2% glutaraldehyde/2.5% paraformaldehyde solution, washed 3 times in phosphate-buffered saline (PBS), postfixed in 1% osmium tetroxide, dehydrated in an ascending acetone series, and embedded in Epon-Araldite resin. Semithin (0.5-μm) sections were then stained with 1% methylene blue in a 4% sodium borate water solution, and they were first studied under a photomicroscope (Olympus Vanox AHB53; Olympus America, Melville, NY) to select the representative areas of interest. Subsequently, the Epon block was trimmed to obtain ultrathin (60-nm) sections. The grids were then dual contrasted with 2% uranyl-acetate in water and Reynold’s lead citrate solution, for 10 minutes each, and they were visualized and photographed on a Jeol 1010 electron microscope (Jeol USA, Peabody, MA) at an accelerating voltage of 80 to 100 kV.
Results
Clinical Findings
During the first 10 days of the experiment, no relevant clinical signs were observed in the infected bees (such as diarrhea), and there was no mortality among the caged bees during the first 10 dpi. At 15 dpi, bees infected with each Nosema species at the highest doses displayed some mortality, and indeed, no bees in the C10 group and only 2 bees in the A10 group survived to 15 dpi. As no samples were obtained from the C10 group at 15 dpi, no samples were taken from the A10 group at that time.
Microscopic Pathology
The histological results were similar in the different groups regardless of the Nosema species used for infection. Microscopic lesions were not detected in any cephalic or thoracic tissue from the infected honey bees, nor were N. apis or N. ceranae organisms detected in these tissues. Moreover, no lesions or organisms were seen in the different organs of the abdominal cavity, except for mature spores, which were found only in the lumen of the ileum and rectal ampulla, but pathologic changes were not detected in these tissues (Figs. 1, 2). Only the midgut (ventricle) epithelial cells were seen to be infected in any of the bees. A comparison of the infected bees did not provide any evidence of Nosema tropism for the aforementioned structures, irrespective of the species or of the spore dose administered.
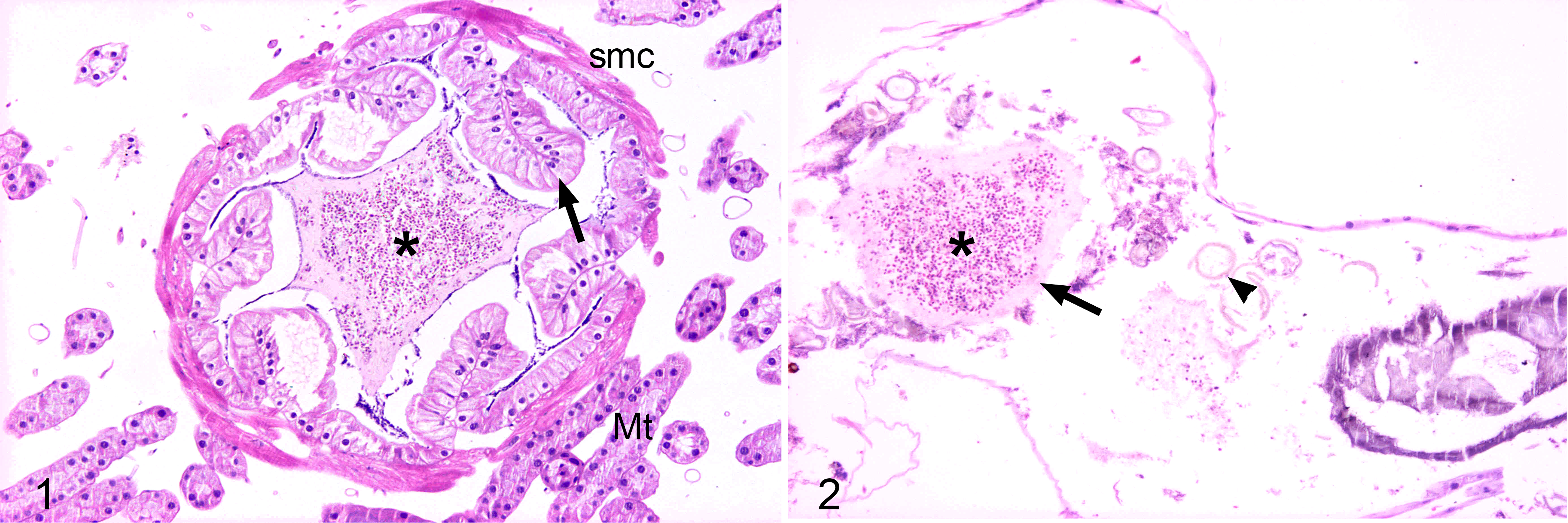
Specific tissue alterations and different stages of the Nosema life cycle were detected in the midgut of infected bees but not in the T0 control group. The microscopic lesions were similar for both Nosema species, whose different life cycle stages were distributed extensively in the ventricular epithelium.
In the bees of the C5 and C10 groups, most epithelial ventricular cells were heavily infected and had expanded cytoplasm with a brightly stained and apically displaced nucleus. There were frequent remnants of secreted globules full of mature spores as well as free mature spores in the ventricular lumen. In addition, the peritrophic membrane often appeared broken and fragmented (Fig. 3). By contrast, the circular and longitudinal musculature of the midgut was free of microsporidia and was morphologically unaltered (Fig. 3).
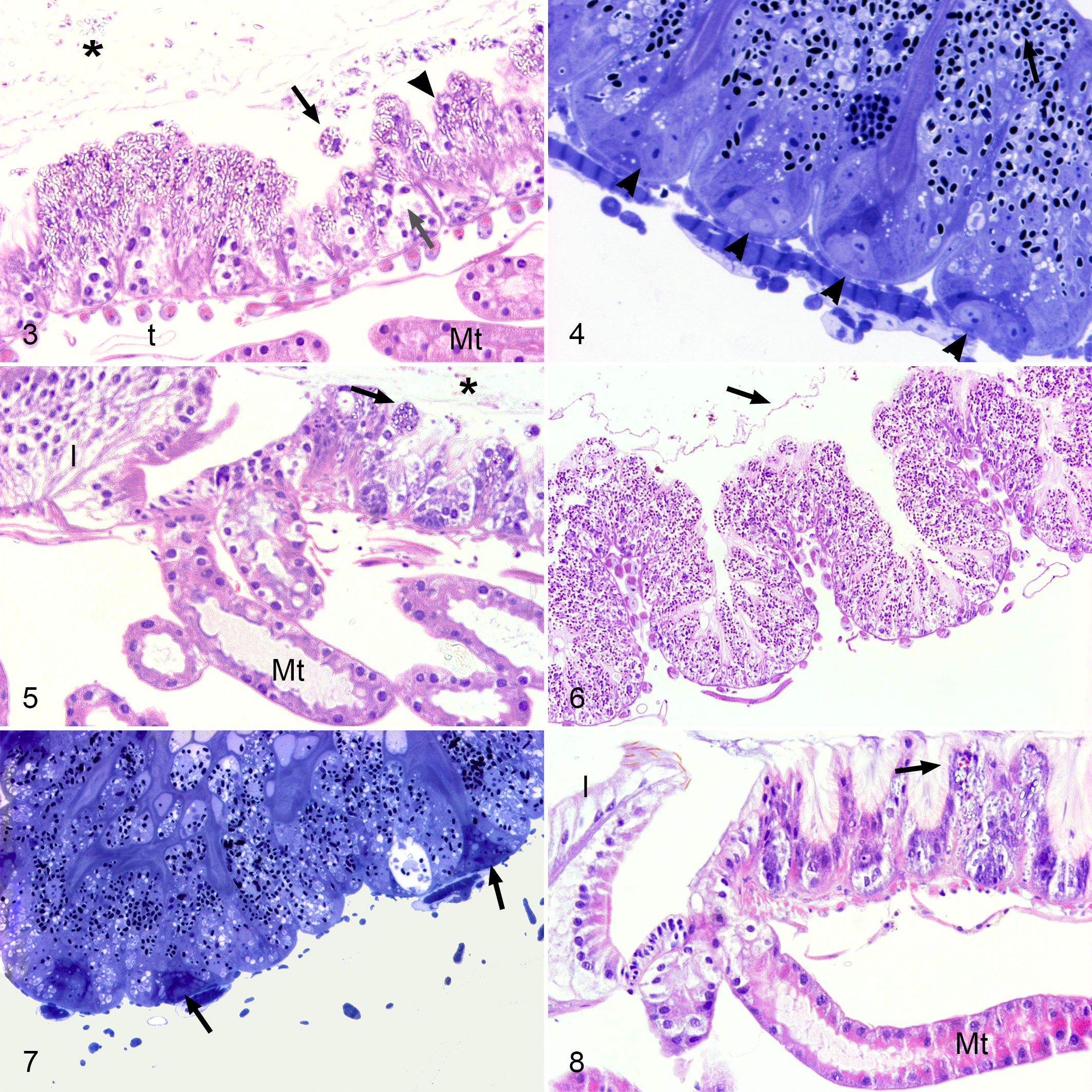
There was a predominance of basophilic mature spores in the epithelial cells at the tips of the folds. Conversely, the cells at the bottom of the folds, even those in clusters of regenerative cells, contained fewer basophilic but larger stages, indicating they contained spores in more vegetative stages (Fig. 4). A smaller number of infected ventricular epithelial cells were observed in the regions of insertion of the Malpighian tubules, although in no case were any microsporidia observed in the epithelial cells of the tubules (Fig. 5). Similar histological results were observed for groups A5 and A10 (Figs. 6–8).
Transmission Electron Microscopy
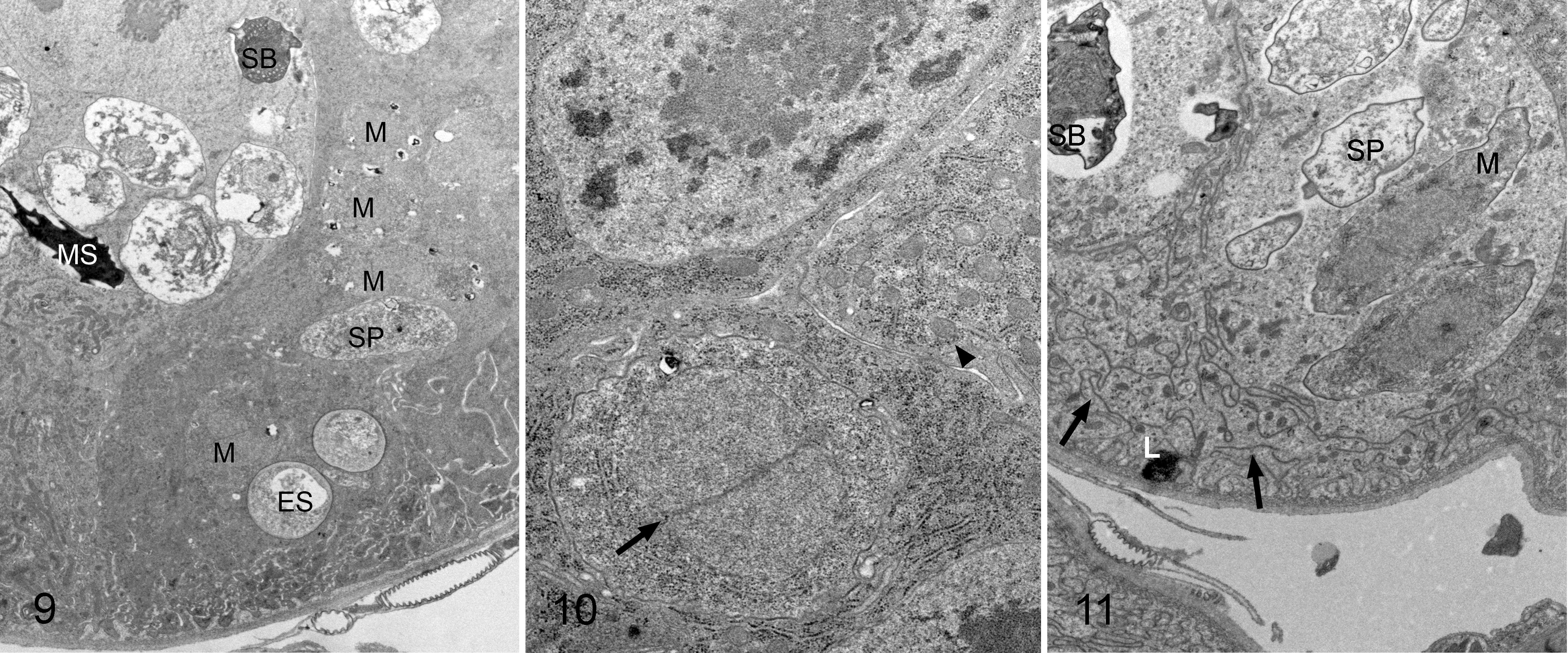
Ultrastructural examination of the ventricular parasitized cells from the bees in groups C5, C10, A5, and A10, and even of the clusters of regenerative cells, identified parasitic structures corresponding to different stages of the life cycle, such as meronts, sporonts, or mature and empty spores (Fig. 9). No differences were observed between N. apis and N. ceranae infection. All stages of the N. apis and N. ceranae life cycle had diplokaryotic nuclei, although sometimes mitochondria were grouped around it (Fig. 10). Both the epithelial ventricular cells and the clusters of regenerative cells had electrolucent cytoplasmic vacuoles, lysosomes, and a fragmented and disorganized rough endoplasmic reticulum (RER), signs of cell degeneration (Fig. 11).
Discussion
To define the tropism of Nosema spp. for extraventricular locations, we have carried out what we believe to be the first histopathological LM and TEM study of different organs and tissues from honey bees artificially infected with these microsporidia and reared under controlled conditions. It is generally accepted that the ventriculus represents the specific target organ of N. apis, 12 and although it has been proposed that spores belonging to this species might also affect honey bee glands or hemolymph, 39,40 histological confirmation of this has yet to be published.
In recent experiments on naturally infected hybrid A. mellifera worker bees from Canada, N. apis and N. ceranae were apparently detected by quantitative PCR in the intestinal tract, venom sac, and several exocrine glandular structures, either alone or in combination, 7 although no histological studies were performed on these organs. The ability of N. ceranae to infect organs outside the digestive system has also been studied in naturally infected European honey bees reared in Maryland. 6 N. ceranae proliferation was evident in epithelial cells of the ventriculus, but despite positive PCR signals, this microsporidium was not observed directly in other parts of the alimentary canal, Malpighian tubules, hypopharyngeal and salivary glands, or fat body. The authors of this latter study only showed TEM images of parasitized ventricles, similar to those published previously, 20 and did not present images of parasitized clusters of regenerative cells like the ones presented here.
On the other hand, pathways essential for controlling renewal of the ventricular epithelium have been seen to be perturbed by N. ceranae infection, 9,32,34 and the fact that clusters of regenerative cells were not parasitised 34 suggests an indirect effect of the infection at the level of these clusters and on reepithelization. 34 Therefore, our data suggest a novel mechanism of disease induced by Nosema spp., and it would be the first detection of direct infection at this level in the ventricular epithelium.
Tissue tropism of Nosema spp. was also investigated in honey bees sampled as part of a large field survey in Germany, 17 with spores detected by LM in smear preparations of the ventriculus. In those bees, it was also possible to detect N. ceranae in the hypopharyngeal glands, brain, Malpighian tubules, and fat body by PCR-RFLP (Polymerase Chain Reaction - Restriction Fragment Length Polymorphism), although again, no histological images of these organs were shown. Extraventricular infection was inferred from the detection of N. ceranae DNA, yet specific life stages of the pathogen were not directly observed in the specimens examined.
Our results are consistent with those obtained previously in honey bees that were artificially infected with N. apis or N. ceranae and reared under controlled laboratory conditions. The organs from those bees were dissected out under special conditions to prevent contamination, and small tissue samples were smeared onto slides and examined by LM. 26 In that case, spores of both microsporidia were found only in the alimentary tract and not in the brain; hypopharyngeal, salivary, and venom glands; fat body; Malpighian tubules; and hemolymph collected from the head or abdomen. Histological images were not shown in that study since the methodology used to study these tissues impeded this. Significantly, Nosema DNA was detected in several cases by PCR in that study, and indeed, surface contamination of dissected organs was reported, while stricter measures to prevent contamination in N. ceranae–infected bees resulted in a PCR signal below the threshold of reliable detection. Careful dissection technique is essential when analyzing these parasites, 7 although we argue that this cannot rule out contamination during sample preparation, which might produce false positives when very sensitive analytical methods are used (eg, PCR). Spore leakage from fragile, infected intestinal tissue and their association with external dissected organs may be problematic when contamination-free isolates are required. 26 Thus, morphologic identification of the parasites within the tissue is essential to confirm or refute tissue tropism.
The fact that 2 controlled laboratory experiments failed to detect damage or different stages of the Nosema pathogen’s life cycle, in organs outside the alimentary canal, seems to indicate these parasites are highly specific to the ventricular epithelium, reflecting a lack of tropism for other tissues. 26 The lesions observed in ventricular epithelial cells and in the clusters of regenerative cells were similar following N. apis and N. ceranae infection. Moreover, no evidence of alternative target organs for these pathogens was obtained, confirming a similar tissue tropism of the 2 species. Nevertheless, the disease produced by these 2 causative agents is quite dissimilar at the individual bee and colony levels. 23,25,29 N. apis is known to induce diarrhea in infected individuals, which is involved in the transmission of infection,[13] yet this does not occur following N. ceranae infection. 3 –5,19,21,22 Thus, the notable histopathological similarity between the 2 infections contrasts with the profound differences in clinical signs and transmission, highlighting the need for further research to elucidate the differences in the pathogenic mechanisms that are associated with these 2 honey bee microsporidia.
Footnotes
Acknowledgements
The authors thank J. Almagro, J. García, V. Albendea, C. Uceta, M. Gajero, and T. Corrales from the Laboratorio de Patología Apícola, Centro de Investigación Apícola y Agroambiental (CIAPA), IRIAF, Junta de Comunidades de Castilla-La Mancha, for their technical support. The authors also thank the National Center for Electron Microscopy (ICTS) at the UCM (Complutense University of Madrid) for their help in obtaining the electron microscopy images. The authors also wish to thank the anonymous reviewers and editors, whose comments have substantially improved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds from INIA-FEDER (National Public Research Project RTA2013-00042-06).
