Abstract
Tissues from 196 experimental and confirmed natural cases of African horse sickness (all 9 serotypes) were examined with a standardized and validated immunohistochemical assay for detection of the causative virus. The study confirmed that heart and lung are the main target tissues for African horse sickness virus (across all serotypes), followed closely by spleen. It also indicated that microvascular endothelial cells and monocyte–macrophages are the main target cells for virus replication. The importance of monocytes as target cells was emphasized, with relatively few tissue macrophages containing antigen in the lung and spleen, respectively. The results were largely in agreement with those of previous studies, but the large number of cases examined permitted more precise description of the location and distribution of antigen in different tissues. Comparison with descriptions of tissue and cell tropism of other orbiviruses indicated similarity with African horse sickness. Immunohistochemistry was shown to be a useful and consistent technique for demonstrating target cells, but the difficulty of identifying cell types—in particular, different types of monocyte–macrophages—is a limitation.
Keywords
African horse sickness (AHS) is one of the most lethal systemic illnesses of horses, with mortality rates in susceptible animals of up to 95%. 11,14,23,34 It is caused by AHS virus (AHSV), an Orbivirus (family Reoviridae), with 9 known antigenically distinct serotypes, 14,34 all of which have been implicated in outbreaks of AHS in South Africa (G. H. Gerdes, personal communication, 2008). AHSV is transmitted primarily by biting midges of the genus Culicoides—especially, C imicola, the most widely distributed of all Culicoides species, as well as the closely related C bolitinos, which was only recently revealed as a vector. 16,30 –34,42,48 It is endemic in sub-Saharan Africa, but outbreaks have also occurred in North Africa, Asia, and Europe. 14,34,42
Peracute and subacute disease in naïve horses manifests principally as respiratory and circulatory disturbances. 14,39 The peracute pulmonary (“dunkop” or “thin head”) form results in rapid death owing to fibrin-rich pulmonary edema and hydrothorax. 14,34 Signs of the subacute cardiac (“dikkop” or “thick head”) form of the disease include edema of the subcutaneous tissues and intermuscular fascia of the head, neck, and shoulders (particularly, the supraorbital fossa and fascia in the region of the nuchal ligament). 14 These animals are thought to die of heart failure, especially in light of the characteristic serofibrinous hydropericardium and severe coalescing ecchymoses and suggillations that are commonly observed on the epicardial and endocardial surfaces of the heart at postmortem. 14 However, the edema in these subacute cases is typically dorsal and localized, reaching only as far down as the scapular and humeral areas, 19 implying that heart failure is not the most likely mechanism. In our experience, many cases manifest as acute or mixed forms of the disease, with respiratory and cardiac manifestations. 14,23,45 Finally, there is a mild fever form of AHS, which is largely characterized by a transient rise in rectal temperature. 14,34
The postmortem tissues routinely submitted to laboratories for diagnosis during AHS outbreaks usually comprise spleen, heart, liver, lymph node, and lung, 22 and this is also the case in South Africa (G. H. Gerdes, personal communication, 2003). Previous studies indicate heart, lung, and spleen as major target organs of AHSV. 10,12,20,25,28,51,52 A number of studies have indicated that the principal target cells are microvascular endothelial cells in the heart and lung and, to a lesser extent, other organs, as well as monocyte–macrophages in circulation and the interstitium. 10,51,52
To standardize and validate an immunohistochemical assay as a diagnostic test for AHS, 128 positive spontaneous fatal cases of AHS were examined and compared to 119 negative cases. The procedure and results are described in detail elsewhere. 13 The present study reviewed not only those cases, which included multiple representatives of all 9 serotypes, but also archival natural and experimental cases of AHS—all of which enabled a thorough evaluation of the cell- and tissue-based tropism of natural and experimental AHSV in formalin-fixed paraffin-embedded tissues from horses. Four cases of experimental inoculation with suspected neurotropic vaccine strains were included to confirm that these strains are not viscerotropic. The results largely confirm the findings of previous studies in terms of target tissues and cells and provide a reliable guide to the location of AHSV antigen in the main target tissues.
Materials and Methods
In sum, 128 natural AHSV-positive cases less than 10 years old and 68 archival natural and experimental cases more than 10 years old (1923–1996) were examined by immunohistochemistry (IHC). The 128 cases, examined to standardize and validate an IHC assay as a diagnostic test for AHS, involved routine diagnostic cases in which the horses died naturally or were euthanized in extremis. They included a diverse selection of cardiac (12 cases), pulmonary (19 cases), and predominantly mixed (32 cases) clinical forms of AHS and multiple representatives of all 9 serotypes (Table 1 ). 13
Positively Labeled Cases of Each African Horse Sickness Virus Serotype a
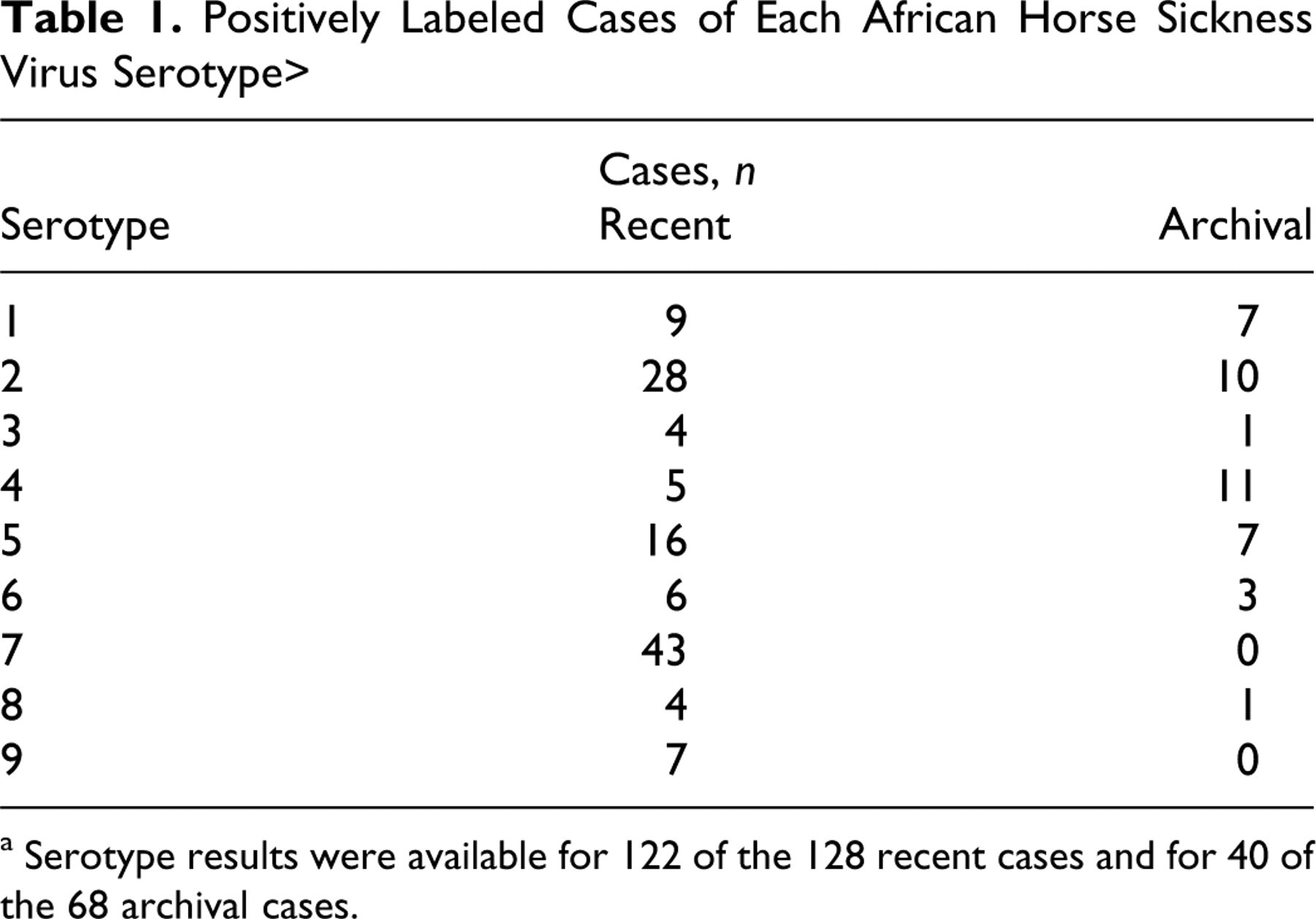
a Serotype results were available for 122 of the 128 recent cases and for 40 of the 68 archival cases.
All the cases were positive on virus isolation, and serotype results were available for 122 of the 128 cases (by means of virus neutralization or polymerase chain reaction; Table 1). The selected cases exhibited only mild to moderate autolysis on examination of sections stained with hematoxylin and eosin (HE). Not all the target tissues of lung, heart, and spleen were present in each case; 115, 76, and 110 samples of lung, heart, and spleen were available for examination, respectively. One spleen, heart, and lung sample for each case was examined, given that previous studies have indicated that AHSV does not show a preference for particular localities in these organs. 10,51,52 In some cases, additional tissues were available and thus tested—namely, 18 liver samples, 11 kidney, 8 gastrointestinal tract (GIT), 4 lymph node (bronchial, retropharyngeal, and/or mesenteric), 1 thyroid, and 1 adrenal gland.
The archival cases older than 10 years consisted of 52 spontaneous cases and 16 experimental cases of AHS. AHSV was isolated in all these cases, but in many the clinicopathological form of the disease was not recorded. However, the respiratory (4 cases), cardiac (7 cases), and mixed AHS forms (22 cases) were included, as were 7 of the 9 serotypes (Table 1); serotype results were also available for 40 of the 68 archival cases (Table 1). These cases yielded the following samples: 55 lung, 53 heart, 54 spleen, 8 liver, 5 kidney, 7 GIT, 7 lymph node, 6 brain, 5 skeletal muscle. Other tissues were present in a small number of cases and are not discussed any further.
Four archival cases had been intranasally inoculated with neurotropic vaccine strains of AHSV—namely, Karen (serotype 7; 2 cases), 7/60 (serotype 9), and A501 (serotype 1)—which were attenuated by serial intracerebral passage in mice. 17,18,29,40 In addition to the 119 AHS-negative horses examined as controls in the standardization/validation study, 13 tissues from 11 equine encephalosis virus–positive (EEV-positive) horses were examined as controls. AHSV was not isolated from any of the EEV-positive cases.
Preparation and examination of the 128 cases less than 10 years old is described in detail elsewhere. 13 The 68 archival cases, as well as the 119 negative cases and 11 EEV-positive cases, were treated in the same way. Briefly, formalin-fixed tissues were embedded in wax, sectioned, and mounted according to a standard operating procedure in the Section of Pathology, Department of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria. 4 For the 128 positive cases (with tissues less than 10 years old) and the 119 AHSV-negative cases, four 3- to 4-μm-thick sections were cut per block as follows: 2 test sections at least 10 μm apart (for comparison of labellingimmunolabelling within target tissues for validation purposes), 1 section for labeling with the negative control reagent, and one for HE labeling. 13 However, for the archival and EEV-positive cases, only 3 sections were cut per tissue block (1 test section, 1 negative reagent control section, and 1 HE section).
All test sections were immunolabeled using a polyclonal anti-AHSV serum (dilution of 1:1500 and incubated with tissue sections for 45 minutes at 37°C) and the basic avidin–biotin complex system described in a previous study. 51,52 Some modifications were made to suit conditions in the Section of Pathology laboratory, including the use of a red substrate, NovaRED (catalogue no. SK-4800, Vector Laboratories, Burlingame, California) to contrast brownish tissue pigments—especially, hemosiderin in horse spleen. 13 The anti-AHSV serum was raised in rabbits with sucrose gradient–purified, intact AHSV serotype 9 particles as antigen. 13,51,52 In addition, an enzymatic epitope retrieval method was used. 51,52 Accordingly, tissue sections were incubated with 0.05% (w/v) Protease XIV (catalogue no. P5147-5G, Sigma Chemical Co, St. Louis, Missouri) at 37°C for 20 minutes. 13 For each tissue block, a negative control section was labeled with the same avidin–biotin complex technique, but the anti-AHSV serum was substituted with an unrelated polyclonal anti-rabies virus serum that was raised in rabbits against purified rabies ribonucleoprotein (originally from the Health of Animals Laboratory, Guelph, Ontario, Canada).
For 1 of the 4 archival cases in which various suspected neurotropic strains of AHSV were intranasally inoculated into horses, the routine immunodetection method failed to elicit positive labeling in the sections of brain. For this case, labeling was repeated (on the sections of brain) using alternative antigen retrieval methods, more concentrated primary antiserum (1:500), and/or a longer incubation of the AHSV antiserum with the tissue sections (90 minutes and overnight) and more sensitive detection systems, including LSAB+ Kit, HRP (catalogue no. K0679, DakoCytomation, Glostrup, Denmark) and the NovoLink Min Polymer Detection System (catalogue no. RE7290-K, Novocastra Laboratories Ltd, Newcastle Upon Tyne, United Kingdom). The alternative antigen retrieval method involved incubating 2 mg of Protease XIV with 200 ml of 1X Dako Target Retrieval Solution (10X concentrate product; catalogue no. S1699, DakoCytomation) for 40 minutes at 96°C. Tissues were also subjected to microwave heating in different buffers (without Protease), pH 6.0–6.2 and 9 for 20, 30, and 40 minutes.
All sections were examined with a light microscope at 400× magnification, and the degree of labeling was scored on a scale of 1 to 3. 13 The interpretation of positive labeling was based on the labeling results as previously described and illustrated. 10,13,51,52 Positive labeling (using the NovaRED substrate) was indicated by the presence of fine brick-red granules and small bead- or dotlike deposits, which occurred singly or in small composite deposits, in the cytoplasm of target cells. 10,51,52 The identification of tissue macrophages depended on cell morphology, location, and, if present, pigment granules in the cytoplasm of mononuclear cells (eg, carbon or bile pigment in pulmonary macrophages and hepatic Kupffer cells, respectively).
Results
Irrespective of serotype and the clinical and macroscopic presentation of AHS, the lung and heart were confirmed as important target tissues for AHSV in horses, followed by the spleen. There was no AHSV-specific positive labeling in 119 negative-tissue control cases, in 11 EEV-positive cases, or in any of the reagent (irrelevant antiserum) control sections.
The nature and distribution of positive labeling in 128 cases less than 10 years old have been described elsewhere. 13 Briefly, positive labeling in the heart tended to be more beaded than granular, whereas in lung and spleen, positive labeling was beaded and granular—with a multifocal clustered to dustlike pattern tending to be more common in the spleen. 13
Monocyte–macrophages and microvascular endothelial cells were confirmed as the most important target cells for AHSV.
Specific positive labeling was observed in 127 of 129 (98%) AHSV-positive horses for which heart samples were available. Positive labeling occurred predominantly within the cytoplasm of normal, swollen, or, to a far lesser extent, shrunken and fragmented (apoptotic/necrotic) endothelial cells lining capillaries in the loose interstitial connective tissue between cardiac muscle cells (Fig. 1). To a far smaller degree, positive granular to beaded or dotlike intracytoplasmic positive labeling was also observed in circulating mononuclear (monocyte-like) leukocytes within blood vessels in the interstitium that were morphologically normal, swollen, or, less frequently, apoptotic/necrotic. Rarely, dotlike positive labeling appeared to be on the surface of erythrocytes in interstitial capillaries, but in many cases, it was difficult to determine if the labeling was in erythrocytes or in the cytoplasm of endothelial cells. Occasional red blood cells appeared to be labeled diffusely positive.
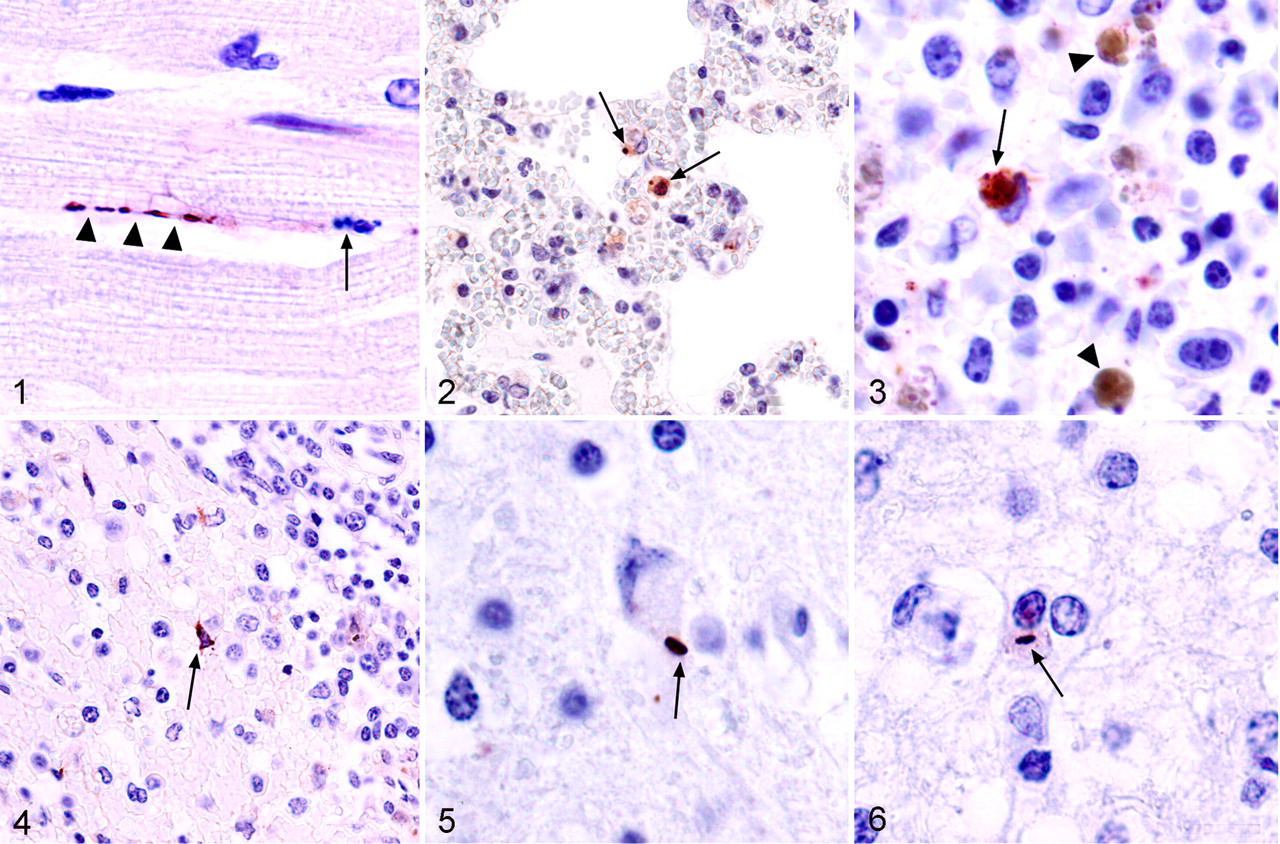
In 170 sections of lung from the 170 AHSV-positive horses where lung samples were available, positive labeling occurred in the cytoplasm of intravascular mononuclear leukocytes with monocyte–macrophage-like morphology (Fig. 2); in the tapered cytoplasm of normal, swollen, or, more rarely, shrunken and fragmented (apoptotic/necrotic) endothelial cells lining pulmonary microvessels; and rarely in the cytoplasm of macrophages in the pulmonary interstitial connective tissue. Positive labeling was usually observed in pulmonary intravascular mononuclear leukocytes and microvascular endothelial cells. Pulmonary intravascular mononuclear leukocytes were quite consistently targeted by AHSV, irrespective of the location of the lung sample. Assessed subjectively, it appeared that when positive labeling in intravascular monocyte–macrophages predominated in the lung, there would be numerous positively labeled circulating monocyte-like leukocytes in multiple organs. However, when endothelial cells were strongly positive in the lung, they tended to be prominently labeled in other organs as well. There was negligible positive labeling associated with erythrocytes in the lung.
Positive labeling was observed in 158 sections of spleen (96%) from the 164 AHSV-positive horses for which spleen samples were available. In the spleen, positive granules and beads were observed most commonly in the cytoplasm of monocyte-like cells and/or large tissue macrophages throughout the red pulp and marginal zone (Fig. 3). The macrophages often contained clumped, globular hemosiderin pigment in addition to AHSV-specific positive granules (Fig. 3). Positive labeling also occurred in endothelial cells lining precapillary arterioles, capillaries, and postcapillary venules in the red pulp and, to a lesser extent, vascular channels in the marginal zone at the periphery of the white pulp, as well as in occasional dendritic cells with stellate cytoplasm in the red pulp and marginal zone (Fig. 4). Positive labeling was rarely seen in large macrophage-like and/or dendritic cells and in occasional endothelial cells lining precapillary arterioles and capillaries within the depleted white pulp. An occasional positive dot appeared to be associated with the surface of a small number of red blood cells in the red pulp, but in general, there was only scant positive labeling of erythrocytes in the spleen.
The number of AHSV-positive cases for which other organs were available was low. The liver sections in 24 of 26 (92%) virus-positive cases with liver samples showed obvious specific positive labeling in the form of granules, beads, and dots in the cytoplasm of intravascular monocyte-like cells, swollen, shrunken, or necrotic/apoptotic sinusoidal lining cells (Kupffer cells and/or endothelial cells) and very rarely in endothelial cells lining venules and precapillary arterioles in portal tracts.
Positive labeling was observed in only 9 of 16 (56%) sections of kidney and was scant, occurring in the form of granules and small beads in the cytoplasm of intravascular monocyte-like cells in interstitial microvessels and glomerular capillaries and, rarely, in endothelial cells of glomerular capillaries and/or interstitial microvessels. In 5 of 5 sections of edematous cervical muscle that were examined, positive beads and granules were seen within the cytoplasm of endothelial cells lining interstitial capillaries and, to a lesser extent, in the cytoplasm of circulating monocyte-like cells. In these samples, targeted cells (with or without AHSV antigen) often appeared swollen or, more rarely, necrotic/apoptotic. Myofibers were often teased apart because of edema, but aside from the rare swollen fiber (still with visible cross-striations in longitudinal section), there was no evidence of significant myodegeneration or necrosis in the examined sections. Virus-specific positive labeling was observed in 14 of 15 sections of GIT from horses with AHS. In these sections of stomach and/or small intestine, positive granules were scarce but most commonly observed in the cytoplasm of microvascular endothelial cells in the muscular tunic, as well as the mucosa, submucosa, and serosa. Dotlike positive labeling was occasionally observed in the cytoplasm of circulating mononuclear leukocytes.
Of the 11 sections of lymph node examined, only 4 showed convincing positive labeling. In these cases, the positive labeling was generally very scarce, with most of the positive beads and/or granules occurring either in microvascular endothelial cells in the cortex or medulla or, more commonly, in monocyte–macrophage-like cells within cortical and/or medullary sinuses.
Cerebrum was examined from only 4 archival cases from 1962 and 1963, where various suspected neurotropic strains of AHSV were inoculated intranasally into horses. Generally, gliosis, accompanied by numerous infiltrating lymphocytes, was marked and extensive in multifocal areas of cerebral gray matter. Within these lesions and occasionally in overlying leptomeninges, there was frequently a severe diffuse vascular (predominantly adventitial) cellular infiltrate, characterized by lymphocytes, plasma cells, and fewer macrophages. Neuronal injury (including necrosis) was occasionally observed within these foci of polioencephalitis.
In 3 of 4 of these cases challenged with neurotropic AHSV, there was clear AHSV-specific positive labeling within patchy foci of lymphocytic encephalitis in the brain. Specific positive labeling took the form of variably sized beads, some quite large, through to fine dustlike granules in the cytoplasm of injured neurons (Fig. 5), microglia, and occasional astrocytes (Fig. 6). Application of the rabies virus antiserum in the place of the AHSV antiserum failed to reveal positive labeling in the same sections of brain. Despite the presence of obvious foci of nonsuppurative encephalitis and neuronal injury in the sections of brain, there was no AHSV-specific positive labeling in any of the brain sections from one case, despite repeated labeling attempts using different techniques as described above. No specific positive labeling was observed in any of the usual target organs (heart, lung, or spleen) from these 4 cases.
Discussion
The large sample size used in this study confirmed lung and heart as major target tissues for AHSV, followed closely by the spleen, across all known serotypes. However, ranking of the target tissues can vary according to the method used to detect the virus or viral antigen. For example, an enzyme-linked immunosorbent assay detected the greatest amount and longest persistence of antigen in the spleen, although significant amounts of antigen also occurred in lung, heart, and liver. 21 This is likely due to the tendency for positive labeling in the spleen to be more localized when compared to heart and lung tissue. 10 In addition, the spleen is a blood-rich organ, and although it is possible that many red blood cells carry a few viral particles on their surface (adding up to a lot of virus in the organ in general), it may well be that not enough virus is carried on any single red blood cell to be visible by IHC. The present study confirmed previous findings that specific positive labeling usually occurred throughout AHSV-positive lung and heart samples, as previously described. 10,51,52
The minimal positive labeling found in only 4 lymph nodes from 11 naturally and experimentally infected horses with lymph node samples was in stark contrast to the findings in an experimental study of AHS. 51,52 AHSV is known to occur in high concentration in lymph nodes and to replicate in them early in the course of infection. 14,19 However, as based on reverse transcription polymerase chain reaction to detect viral RNA in an AHSV-infected horse, most amplicons were obtained from RNA extracted from the heart, lung, spleen, and liver, with fewer from the kidney and cervical lymph node and least from the bronchial and mesenteric lymph nodes. 7 Widespread depletion of lymphoid elements was common in histological sections of spleen and lymph node from AHSV-positive horses in this study, which have resulted in the amount of virus left in association with these cells falling below the concentration necessary for detection by IHC. Thus, detection of AHSV antigen in lymph nodes may depend on the stage of infection, with more consistent detection of antigen occurring in lymph nodes from horses early in the course of infection.
Based on only a few samples, the results for the remaining organs examined were similar to previous findings, with antigen being quite consistently present in the liver and GIT but not the kidney. 51,52 The positive results on 5 samples of edematous neck muscle from archival cases more than 25 years old largely concur with similar findings using in situ hybridization (ISH). 10
Microvascular endothelial cells and monocyte–macrophages were demonstrated to be the most commonly targeted cells, with other cell types labeling positive less frequently. This largely confirms previous findings in experimentally infected horses using transmission electron microscopy (TEM), 12,20,25 ISH, 10 and IHC. 51,52
In horses with AHS, increased numbers of mononuclear leukocytes have been observed in and around blood vessels in histological and ultrastructural sections of lung and heart in particular, 12,25,27,28,36,43 but the types of monocyte–macrophages targeted by AHSV have not always been determined. That antigen was most commonly found in intravascular mononuclear leukocytes of the monocyte–macrophage series suggests a probable tropism for monocytes. Previous work has shown that AHSV infects and replicates readily in monocytes in vitro, 44 and the presence of AHSV in the cytoplasm of monocytes in tissues has been mentioned. 12,25,51,52 An earlier study was less specific, referring to the presence of AHSV-positive hybridization signal in “large mononuclear cells” in the marginal zone of the spleen, which were considered to be monocytes or large lymphocytes. 10
The consistently greater number of positively labeled intravascular mononuclear cells in the lungs than that in any other organ suggests that these cells, which have a monocyte-like morphology under the light microscope, may be specific to the lung. An ultrastructural study of the lungs from 5 horses experimentally infected with AHSV found that AHSV commonly infects and replicates in so-called pulmonary intravascular macrophages (PIMs). 12 PIMs are a resident population of mature phagocytes that resemble mature macrophages of the mononuclear phagocyte system in structure and function. 2 These cells have long been recognized as the site of substantial uptake of blood-borne particles and microbes in the lungs of a number of domestic animal species—particularly, ruminants and pigs but also horses and cats. 3,6,35,49,50 Because no TEM was done in the present study, the positively labeled intravascular mononuclear leukocytes in the lung could not be conclusively identified as PIMs.
Tissue macrophages were not a common or prominent target cell in most organs examined. A previous study mentioned AHSV-specific positive labeling in macrophages in spleen and lymph nodes but in no other organ. 51,52 Various ultrastructural studies on AHSV in lung and spleen samples have demonstrated structures indicative of viral infection in only a small number of tissue macrophages in these organs. 12,20 AHSV was found to readily infect equine monocyte and young macrophage cell cultures, but more mature equine macrophages were refractory to infection. 44
The occasional positive labeling of red blood cells was difficult to interpret. Although it has been clearly demonstrated that AHSV is associated with erythrocytes in the blood of infected horses, 37 no positive labeling of erythrocytes was reported in the previous studies using IHC. 51,52 The authors suggested that either there was too little erythrocyte-associated viral antigen for detection by IHC or the AHS viral antigen is held in indentations and invaginations of the red blood cell membrane, as described for other orbiviruses, 1,5,8 thus eluding detection by the primary antiserum. The latter argument seems unlikely because in histological sections erythrocytes can be sliced and are not seen as whole anyway. More important, mature red blood cells are not target cells for AHSV replication, because they lack the necessary cellular components for virus replication.
Various ultrastructural studies on AHS have demonstrated virus in occasional fibroblasts, smooth muscle cells, and pericytes. 12,20,41 In the present study, none of these cells were apparently targeted by AHSV in the tissues examined. In support of these findings, AHS viral nucleic acid and antigens have not been reported in fibroblasts, smooth muscle cells, or pericytes with ISH and IHC. 10,51,52 It is often difficult to classify cells with certainty in immunolabeled sections owing to the relative lack of cellular detail and contrast when compared to that of HE-stained sections.
Infection with AHSV of microvascular endothelial cells representing distinct anatomic sites occurred irrespective of the serotype and clinical form of AHS. Endothelial cells from different anatomical locations display antigenic heterogeneity. 38 Horses infected with a particular serotype of AHSV may have a heterogeneous mixture of highly virulent and less virulent viruses in their blood; furthermore, more-virulent viruses might have a predilection for pulmonary microvascular endothelial cells, whereas less-virulent particles might selectively multiply in lymphoid tissues or in endothelial cells lining blood and lymph vessels of the head and neck. 19 However, this hypothesis does not appear to have been investigated.
Studies on related orbiviruses have indicated similar tissue and cell tropism to AHSV. The viruses that cause bluetongue (BTV) and epizootic hemorrhagic disease (EHDV) have been shown (based on TEM, ISH, IHC, and immunofluorescence) to infect and replicate in microvascular endothelial cells in a similar selection of organs and tissues. 9,24,26,47 The association of affected cells with particular anatomic sites has also been described in EHDV and BTV infections. 15,46 Tropism for intravascular monocytes has been described for BTV and EHDV 5,9 and has been observed in infections with EEV (A. D. Pardini, unpublished data, 2007).
The discrepancy in the findings in horses inoculated intranasally with neurotropic strains of AHSV is difficult to explain because the sections from the horse with no immunolabeling had similar, although less severe, histologic lesions. Notably, the A501 strain used in that case supposedly produced less severe neurological signs in horses when compared to the strains used to inoculate the other horses. 18 It is possible that the amount of viral antigen was beneath the threshold necessary for detection by the immunolabeling method described in the present study. Alternatively, the immune system may have already cleared the virus (after the initial damage was done), leaving behind only residual inflammation. Unfortunately, no information was available on the dose administered or the virus titer in the intranasal inoculum for any of the experimentally infected horses. The lack of labeling in spleen, lung, and heart from these horses is not unexpected, given that the neurotropic mouse strains of AHSV apparently retained few of their viscerotropic properties. 18 In addition, inoculation via the intranasal route makes it less likely that significant quantities of virus would reach the viscera.
As reported in previous studies, damaged endothelial cells do not always contain virus, which suggests that the microvascular effects that are important in the pathogenesis of AHS may be multifactorial, resulting from a combination of direct endothelial damage and the release of cytokines by affected endothelial cells and monocyte–macrophages. 12,20,25 In our opinion, direct and indirect microvascular damage may play a more important role than that of heart failure in the pathogenesis of the dorsal and localized edema seen so often in horses with the subacute and mixed forms of AHS. This view is supported by the observation of scant specific labeling in the microvasculature of the edematous supraorbital fat in affected horses. 51,52 In the present study, there was also ample specific labeling of endothelial cells and monocytes in microvessels in the interstitium of edematous cervical muscles. In our experience, based on findings from the present study and ongoing work, there is evidence of swelling and, rarely, apoptosis/necrosis of microvascular endothelial cells and monocytes (a small number containing viral antigen) in sections of cervical muscle and supraorbital fat from affected animals. Future studies might usefully attempt to resolve this debate, as well as discover and characterize viral receptors on target cell membranes and enable pulmonary intravascular macrophages to be identified using light microscopy.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
