Abstract
Immunothrombosis is a potentially beneficial physiological process that aids innate immunity and host defense against pathogen invasion. However, this process can also be damaging when it occurs to excess or in critical blood vessels. Formation of extracellular traps by leukocytes, particularly neutrophils, is central to our understanding of immunothrombosis. In addition to degranulation and phagocytosis, extracellular traps are the third mechanism by which neutrophils combat potential pathogens. These traps consist of extracellular DNA decorated with bactericidal cellular proteins, including elastase, myeloperoxidase, and cathepsins. Neutrophils can release these structures as part of a controlled cell-death process or via a process termed vital NETosis that enables the cells to extrude DNA but remain viable. There is accumulating evidence that NETosis occurs in companion animals, including dogs, horses, and cats, and that it actively contributes to pathogenesis. Numerous studies have been published detailing various methods for identification and quantification of extracellular trap formation, including cell-free DNA, measurements of histones and proteins such as high-mobility group box–1, and techniques involving microscopy and flow cytometry. Here, we outline the present understanding of these phenomena and the mechanisms of extracellular trap formation. We critically review the data regarding measurement of NETosis in companion animals, summarize the existing literature on NETosis in veterinary species, and speculate on what therapeutic options these insights might present to clinicians in the future.
Keywords
The innate immune system actively participates in the initiation and propagation of thrombosis, through the activity of neutrophils, monocytes, and dendritic cells that can initiate and accelerate fibrin formation and activate platelets. Although thrombosis is typically thought of as pathologic, in some scenarios it may represent a beneficial physiological process that aids innate immunity and host defense against pathogen invasion. This protective process is now termed immunothrombosis 46,74 and is a proposed consequence of the conserved links between inflammation and coagulation. 119,120 Formation of extracellular traps by leukocytes, particularly neutrophils, is central to our understanding of the genesis of immunothrombosis. While immunothrombosis may be protective in some settings, dysregulated or excessive clot formation likely contributes to morbidity and mortality in trauma, sepsis, heat stroke, and hemolytic anemia. Immunothrombosis may therefore represent a novel therapeutic target in these conditions, aided by distinct pathways and molecular mediators that separate immunothrombosis from normal hemostasis. Here, we discuss the cellular mechanisms that underpin neutrophil extracellular trap (NET) formation and immunothrombosis, appraise the methods for identification of NETs, review the existing veterinary literature, and suggest potential avenues for future therapeutic intervention.
Mechanisms of NET Formation
Neutrophils are the sentinel cells of innate immunity responsible for defending the host against invading microorganisms. Once recruited from the bloodstream to sites of infection, neutrophils may phagocytose, degranulate, and generate reactive oxygen species to effectively eliminate invading pathogens. 146 In response to certain stimuli, neutrophils can also enhance their antimicrobial activities by releasing NETs, composed of extracellular chromatin. 18 Attached to these web-like scaffolds of cell-free DNA (cfDNA) are histones and neutrophil granule proteins like myeloperoxidase (MPO), lactoferrin, cathepsin G, neutrophil elastase (NE), and antimicrobial peptides such as defensins. 101,105,166 These components of NETs are crucial to their antimicrobial properties.
The process of NET formation, termed NETosis, is the outcome of a number of highly regulated cellular events induced by a variety of stimuli. The cellular orchestration of NETosis is dependent on the species and the stimulus, and it is currently the topic of intense investigation. NETs are released upon direct exposure to microorganisms or through recognition of pathogen-associated molecular patterns within the bloodstream or body compartments accessible via diapedesis. Ex vivo exposure of feline and equine neutrophils to Leishmania and Streptococcus equi or Streptococcus capitis, respectively, results in NETosis. 166,218 In vivo, in dogs with sepsis, NETs have been documented in the blood, in the lungs, and in abdominal and pleural effusion samples. 81,111 As part of the outer membrane of Gram-negative bacteria, lipopolysaccharide (LPS) or endotoxin initiates inflammation by activating neutrophils via the pattern recognition receptor, Toll-like receptor 4 (TLR4). In a model system, high-dose LPS stimulates canine neutrophils to undergo NETosis. 112 In comparison to those of dogs, equine neutrophils are extremely sensitive to LPS. Equine neutrophils undergo extensive NETosis when exposed to a fraction (1/100,000) of the LPS concentration required to induce NETosis in dogs. In addition to direct interactions with microorganisms and molecular patterns, cytokines such as interleukin (IL)–8, complement component C5a, or autoantibodies have also been shown to induce NETosis in human neutrophils. 89
Certain cellular events are essential for NETosis. After stimulation by the protein kinase C activator phorbal 12-myristate 13-acetate (PMA) or by LPS, canine neutrophils undergo sequential morphological changes including chromatin decondensation, loss of membrane integrity, and release of cfDNA (Fig. 1). 112 Human neutrophils undergo loss of the nuclear envelope and mixing of karyosomes and cytoplasmic granules prior to the release of cfDNA. 49 Similar to human and murine neutrophils, canine neutrophils undergo extensive NETosis in response to PMA. 81,112 Although the exact mechanism of PMA-induced NETosis remains unclear, PMA likely signals multiple intracellular pathways essential for mediating NETosis. First, PMA triggers the generation of reactive oxygen species by inducing calcium influx and activating NADPH oxidase. Reactive oxygen species generation must occur prior to the activation of p38 mitogen-activated protein kinase and extracellular signal-regulated kinase phosphorylation during NETosis. 80,90 In mice, the synergistic action of NE and MPO within the nucleus following translocation from the cytoplasm promotes chromatin decondensation. 156 Nuclear decondensation is also dependent on post-translational modification of histones, a group of positively-charged proteins responsible for hyper-condensing DNA into nucleosomes. Citrullination (also called deimination) of histones, catalyzed by the enzyme peptidylarginine deiminase 4 (PAD4), alters the electrostatic interactions between histones and DNA by decreasing the net positive charge of histones. This, in turn, causes chromatin decondensation facilitating the release of cfDNA. 105,167,217 In canine neutrophils, PAD4 inhibition by the chemical Cl-amidine blocks histone citrullination and prevents NETosis mediated by either LPS or PMA. 112
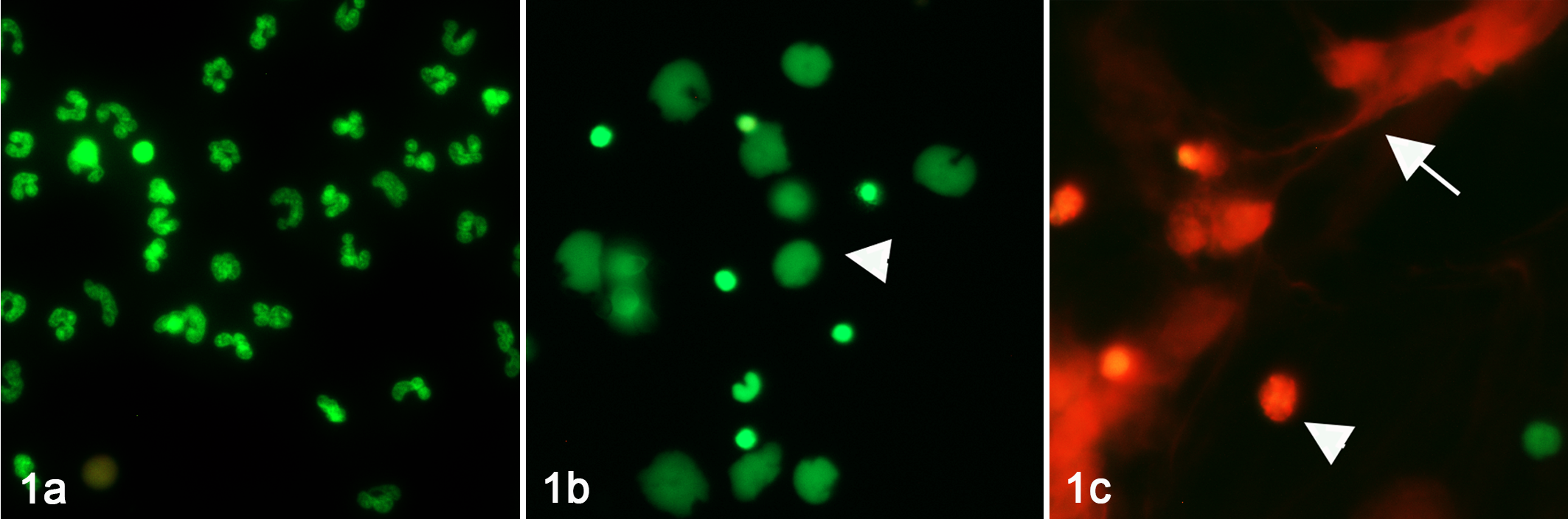
Fluorescence microscopy of live canine neutrophils demonstrating the sequential morphological changes seen during NETosis. Isolated living neutrophils, activated with PMA, were stained with either a cell-permeant nucleic acid dye (green) or cell-impermeant nucleic acid dye (red), and incubated for 30 (1a), 90 (1b), or 180 (1c) minutes prior to imaging. Only cell-free DNA or cells without an intact cell membrane are stained red. (1a) At 30 minutes, all neutrophils have an intact cell membrane with lobulated nuclei. (1b) By 90 minutes, chromatin within the cells are beginning to decondense (arrowhead). (1c) By 180 minutes, most neutrophils have lost their membrane integrity, and web-like cell-free DNA (arrows) are released.
Types of NETosis
Many of the early studies of NETosis relied heavily on electron microscopy or fluorescence microscopy to visualize neutrophils generating NETs. These techniques require fixation and immunolabeling and hence limit the potential physiologic relevance of the phenomena observed. As a result, NETosis was initially considered a type of cell death because expulsion of cfDNA and granule proteins requires disintegration of both the nuclear envelope and plasma membrane. This type of NETosis, described by some investigators as “suicidal” or “lytic,” occurs with PMA stimulation when cells can no longer maintain homeostasis following the event. 230 Developments in intravital microscopy enabled investigators to observe NETosis in vivo and demonstrate that living neutrophils can maintain their functional capacities following NETosis. In human skin infections and in mouse sepsis models, direct encounters of neutrophils with Staphylococcus aureus and E. coli lead to release of cfDNA. 141,231 The remaining anuclear neutrophils maintain an intact cell membrane, perform chemotaxis, and phagocytize nearby bacteria. 231 This phenomenon, termed vital NETosis, has yet to be demonstrated in veterinary species.
Platelet-Neutrophil Interactions in NETosis
Platelets play key roles in innate immunity. Human, murine, and canine platelets express functional TLRs, allowing them to directly respond to pathogen-associated molecular patterns. 30,113,188 Activated platelets interact with neutrophils to facilitate NETosis. 30 In ex vivo systems, LPS-induced NETosis in human, canine, and murine neutrophils is limited, 112,118,160 but the response can be amplified by the presence of LPS-activated platelets, suggesting that platelet-neutrophil interactions promote and facilitate NETosis. In mouse endotoxemia models, platelet depletion significantly attenuates NET formation within liver sinusoids. Formation of platelet-neutrophil aggregates and NETosis in this model is dependent on platelet TLR4. 30 In addition to LPS, thromboxane and thrombin-mediated activation of platelets also support robust NETosis in humans and mice. 24
For platelet-mediated NETosis to occur, both cell types must synergistically interact via adhesive and soluble interactions. Platelet activation must first take place to initiate intracellular signaling cascades that induce secretion of soluble mediators, and upregulate and activate adhesion molecules. Platelet activation induced by agonists such as adenosine diphosphate and thrombin results in degranulation of alpha-granules with subsequent membrane surface P-selectin expression. Agonist-mediated activation also leads to inside-out signaling that alters the conformation of the extracellular domains of the αIIbβ3 integrin and, thereby, increases its affinity for ligands.
In mice, platelet-neutrophil interactions mediated by adhesive interactions between platelet P-selectin and its neutrophil receptor, P-selectin glycoprotein ligand–1 (PSGL-1), are essential for NETosis during sepsis. 48,187 Such findings, however, could not be replicated in humans, suggesting that P-selectin/PSGL-1–mediated NETosis may be species dependent. 22,139 It is known that equine peripheral blood mononuclear cells express PSGL-1 as sialylated homodimers. Several post-translational modifications and receptor dimerization are required for high-affinity P-selectin binding by equine PSGL-1. 225 However, in contrast to human PSGL-1, sulfation of equine PSGL-1 is not required for P-selectin binding. Clearly, there are interspecies differences in the structure of these proteins that could influence their ability to mediate NET formation. In humans, neutrophils express the neutrophil integrin αMβ2 (MAC-1), which binds to the platelet heterodimeric glycoprotein 1bα (GP1bα) via von Willebrand factor and can induce NETosis in vitro. 169 In mice, fibrinogen, which normally binds to its platelet receptor, αIIbβ3, also binds to neutrophil integrin MAC-1 to form an adhesive interaction between the 2 cell types (Fig. 2).

A schematic diagram demonstrating the molecular mechanisms of platelet-neutrophil interaction involved in NETosis. Platelet-neutrophil interaction can be broadly divided into either adhesive or soluble interactions.
Activated platelets also secrete soluble mediators capable of stimulating nearby neutrophils to produce NETs. Upon activation, human and mouse platelets upregulate and secrete high mobility group box–1 (HMGB-1), a damage-associated molecular pattern. 140,209,210 This protein binds to its neutrophil receptor, the receptor for advanced glycation end-products to facilitate NETosis. 139,209 In mouse septic peritonitis models, platelet-specific HMGB-1 knockout mice produce significantly fewer NETs, leading to bacteremia and an amplified systemic inflammatory response. This suggests that platelet-derived HMGB-1 is critical for bacterial clearance and survival in sepsis. The pathways downstream of receptor for advanced glycation end-products activation that lead to NETosis, however, remain to be determined. 40,41,139,234 Other platelet-derived chemokines, like platelet factor 4 and RANTES (CCL5), can directly activate neutrophils to undergo adhesion. The heterodimerization of platelet-derived platelet factor 4 and CCL5 binding to neutrophil G-coupled protein receptors also enhances NETosis in mice (Fig. 2). 169
Role of NETs in Innate Immunity
NETs exert their antimicrobial activities by physically entrapping microorganisms, directly killing pathogens, or hindering their dissemination from the point of entry. 18 The negatively charged surface of extracellular chromatin facilitates the binding of organisms like bacteria Candida albicans and Leishmania. Adhered microbes are then killed by high concentrations of antimicrobial proteins such as histones and cathepsin G. It was recently demonstrated that NETs derived from dogs with sepsis can bind directly to bacteria, suggesting that the active entrapment of bacteria by NETs may occur in dogs in vivo. 111,221 Microorganisms that evolve to dismantle the structural components of NETs are considered more virulent. For instance, Staphylococcus aureus degrades NETs by converting DNA to deoxyadenosine, which causes macrophage apoptosis, allowing the bacteria to escape from NETs and evade phagocytosis. 197 The respiratory pathogen Streptococcus pneumoniae can disseminate into the bloodstream by expressing endA, a virulence factor that degrades DNA. 10
Immunothrombosis
Microvascular thrombosis induced by NETs is an important first line of defense as it prevents systemic dissemination of pathogens via the circulation. With their multifaceted ability to stimulate thrombus generation, NETs are key players in immunothrombosis. 133 The structural components of NETs can directly activate platelets, facilitate thrombus formation, and inhibit fibrinolysis and the natural anticoagulant pathways. In dogs, neutrophil-derived cfDNA decreases fibrinolysis, while NET proteins accelerate clot formation. 82 Large fragments of double-stranded cfDNA measuring at 1.5-10 kbp collected from human septic patients strengthen clot formation by binding to factor XII and high-molecular-weight kininogen, both critical for activation of the contact pathway of coagulation. 53,176 NETs’ ability to trap and accumulate both circulating cells and soluble coagulation components is likely central to their thrombogenic abilities. 152 The web-like structure of cfDNA fortifies thrombus formation by binding to circulating erythrocytes, platelets, and clotting factors, including tissue factor and fibrin. 133 Moreover, histones on NETs can activate platelets and increase thrombin generation via platelet TLR2 and TLR4, and histones induce platelets to release polyphosphates and expose phosphatidylserine and activated factor V. 152,179 These platelet changes in turn support the prothrombinase complex (FXa/FVa/Ca2+) assembly. Histones exhibit further prothrombotic effects by modulating the activation of the natural anticoagulant, protein C. 5,179 In vitro, cathepsin G and NE can further strengthen clots by degrading tissue factor pathway inhibitor bound on human endothelial cells. 189 Fibrin degradation is impeded by NETs as DNA forms a ternary complex with plasmin and fibrin, inhibiting plasmin-mediated fibrinolysis. 152 Recent studies in DNAse-deficient mice suggest that NETs may also promote vascular occlusion independently of coagulation activation and fibrin clot formation. 86,152
NET-related thrombosis is an important component of pathogen control provided by NETs; as such, NETs highlight the intersection of blood coagulation and antimicrobial defense. When NETs trap tissue factor pathway inhibitor, which NET serine proteases subsequently inactivate, NETs promote extrinsic coagulation and arterial thrombosis in mice. 135 During systemic infection, the resulting arterial thrombi immobilize bacteria in the microvasculature, preventing pathogen spread. However, the same mechanism can result in undesired large vessel occlusion and thrombosis.
Acute infections, perhaps through NET generation, predispose patients to deep vein thrombosis, a major cause of cardiovascular death in humans. 183 In a murine venous stasis model, a link between NET-driven thrombosis and deep vein thrombosis development was established. In this model, venous stasis resulted in a rapid accumulation of neutrophils, NET formation, and subsequent activation of FXII. 211 DNase1 administration reduced NETs and suppressed deep vein thrombosis growth, demonstrating the dependence of thrombosis on NETs. 211 Tissue factor expressed by hematopoietic cells, platelets, and FXII, with which NETs co-localize, was also critical for thrombosis in this model. 152,211 Similarly, murine deep vein thrombosis models show reduced thrombus formation in PAD4-deficient mice that cannot generate NETs, again suggesting that NETs are integral to deep vein thrombosis. 131 NETs have also been demonstrated in close association with von Willebrand factor in thrombi in murine and baboon deep vein thrombosis models. 51,133 NET markers are elevated in a multitude of human diseases associated with increased thrombotic risk, such as cancer and rheumatoid arthritis, and NET markers correlate with disease state in patients with thrombotic microangiopathies. 52,133 Interestingly, human patients with vascular occlusions secondary to severe bacterial infections have decreased plasma ability to degrade NETs ex vivo and form intravascular NET clots. 86
In addition to the deleterious effects of thrombus generation, NET components can be directly toxic to host tissues as well as microbes. NET histones, and to a lesser extent MPO, are cytotoxic to both endothelial and epithelial cells. 170 Many diseases and their animal models demonstrate the capacity of NETs to cause collateral host tissue damage. For example, in murine models of LPS-induced acute lung injury, NETs are generated and cause organ injury and local and systemic inflammation; NET degradation by DNase protects against lung injury. 118 Circulating NET markers are also increased in the plasma of human patients with acute respiratory distress syndrome and correlate with disease severity and mortality. 104 Thus, the antimicrobial effects of NETs can cause significant bystander damage.
Biomarkers of NETosis
Multiple putative biomarkers have been investigated for the identification and quantification of NETosis, but presently there is no gold standard. 94,137 All of the available techniques have both strengths and weaknesses and distinct specificities for NETosis versus other mechanisms of DNA extrusion or passive release. It should be recognized that biomarkers that lack specificity for NETosis can still provide other valuable insights into pathogenesis. For instance, regardless of whether cfDNA is released by NETosis or other mechanisms, its procoagulant properties likely contribute to the morbidity and mortality of pathogenic immunothrombosis. 58
Cell-Free DNA (cfDNA)
Dyes capable of fluorescence when bound to nucleic acids are the most frequently used method for quantifying extracellular DNA in body fluids 48,52,83,127,134,185 and for experiments using isolated neutrophils. 61,81 Total cfDNA provides a high-throughput, quantitative measure of DNA release applicable to a wide range of species. 29,155 However, measurement of cfDNA alone does not distinguish NETosis from double-stranded DNA derived from other sources. Extracellular DNA may derive from release of NET-like structures by non-neutrophils (eg, eosinophils, macrophages), from release during apoptotic or necrotic cell death, or from enucleation of maturing erythroid precursors. 6,60,98,128,201 Extracellular DNA from apoptosis, necrosis, and erythroid maturation is not accompanied by cytoplasmic proteins. This may have pathophysiologic and therapeutic relevance as the presence of cytoplasmic proteins affects the properties of NETs, DNA, and histones. 128 Human clinical studies initially suggested that cfDNA likely originated from NETosis if there is a concurrent increase in DNA-histone complexes and neutrophil-derived proteins. 175,205 However, such correlations do not rule out release of cfDNA from endothelial cells or parenchymal cells, as demonstrated by investigations using bone marrow chimeric mice. 204 More specialized approaches, such as determining the tissue of origin based on epigenetic signatures, have been used in human studies but have yet to be applied to companion animal samples. 98,190
Biomarker studies require careful design to ensure they reliably reflect in vivo cfDNA concentrations, including control of pre-analytical factors. 45 For example, plasma is preferred to serum, as serum can contain high concentrations of DNA released from leukocytes after sample collection. 45,103,193 The time between collection and processing, 20,227 centrifugation conditions (duration, relative centrifugal force), 45 and storage conditions 45,215 also influence DNA concentrations, although the effect of delayed processing may be minimized by use of tubes containing a stabilizing agent. 151 Investigators must also consider if DNA extraction is required. DNA has been measured directly in serum 214 and plasma, 52,134,147 but autofluorescence 185 and interfering substances may introduce inaccuracy. 83,185 The most commonly used dyes are PicoGreen 48,127,134,214 and Sytox Green. 147,185 Manufacturer’s instructions for the cell-viability dye Sytox Green do not list specific interfering substances, but an effect of growth medium is mentioned. 162 This suggests that a complex matrix such as plasma may also contain interfering substances. For PicoGreen, the manufacturer lists numerous interfering substances, some of which are routinely present in plasma (eg, albumin, IgG). 161 In addition, icteric and hemolytic interferences occur for both dyes. 83,185 To avoid interferences, DNA extraction can be performed before plasma DNA quantification. 83 However, extraction does not provide 100% recovery of DNA, and performance varies between different extraction protocols. 108,186,227
NET Components and NETosis Inducers
Increased concentrations or gene expression of inducers of NETosis (eg, IL-8, TNF-α, and HMGB1) is reported in veterinary patients with a variety of clinical conditions. 56,62,70,200,202,212 This suggests NETosis may occur in various disease states, but it should be recognized that these markers are not specific for NETosis but contribute to numerous inflammatory and cell death pathways. 28,33,163 As discussed above, NETosis is an umbrella term for a variety of molecular pathways that result in DNA release, 94 so it is possible that NETs may be produced without concurrent increases in the concentrations of a particular inducer. 75
Concentrations or activity of individual NET components is increased in plasma and other body fluids in a number of clinical conditions. 3,100,199,200 These include neutrophil granule proteins (eg, NE, 3,18 MPO 3,18,199 ), nuclear proteins (eg, histones, 18 HMGB1 56,158 ), and DNA-histone complexes termed nucleosomes. 100,199 Similar to cfDNA or NET inducers, these markers are not specific for NETosis. For instance, NE and MPO are released during neutrophil degranulation, 96 while histones and HMGB1 are present in all nucleated cells and can be released during forms of cell death other than NETosis. 4,85,174
Citrullinated Histones
Citrullinated histones are purported to be highly specific markers for NETosis. 100,118,194,195 This is based on evidence that knocking out or inhibiting PAD4 (the enzyme responsible for histone citrullination during NETosis) reduces NET formation. 14,110,217 However, involvement in NETosis does not necessarily imply specificity, since H3 citrullination has also been reported in neutrophils undergoing apoptosis, autophagy, and necrosis. 168 In addition, studies aiming to document NETosis in clinical conditions have used western blots 100 and ELISAs 195,196 to quantify citrullinated histones in body fluids, but these assays do not determine the cells from which the histones originated. A quantitative ELISA for citrullinated H3 is not currently available for use in veterinary species. The enzymes PAD2 and PAD4 responsible for histone citrullination are expressed in a variety of cell types, 216 and overexpression of PAD4 has been documented in cells other than neutrophils in cancer 25 and autoimmunity. 136 Citrullination at various residues of histone H3 has been cited as evidence of NETosis 100,195 but is also detectable in non-neutrophil cell types, including oligodendrocytes, 136 cultured fibroblast-like synoviocytes, 14 and cancer cell lines. 27 The sensitivity of citrullinated histones for NETosis detection is also debated. 94 Investigations using PMA to trigger NETosis have produced conflicting results regarding H3 citrullination. Some studies suggest PAD4 activation and histone citrullination are essential for PMA-induced NETosis, while others have documented PMA induced-NETosis in the absence of citrullination. 94,148,149 Antibody performance likely contributes to these inconsistent results. 148 There are marked differences in the signals generated for western blots of the same neutrophil lysates using distinct anti-citrulline antibodies, to the extent that using an alternative antibody can lead to a different interpretation of the experiment. 148 These observations regarding antibody performance raise important issues regarding appropriate controls for measurement of NET markers in biological fluids. A widely used rabbit polyclonal anti-histone H3 antibody (abcam ab5103) shows weak reactivity with non-stimulated neutrophil lysates. 148 Anecdotally, longer incubation times are also reported to increase H3 citrullination in unstimulated neutrophils. 94 These are not necessarily problems for ex vivo experiments as signals can be compared between stimulated and control neutrophil lysates prepared after standardized incubation times and containing consistent concentrations of histones. However, such issues are more difficult to negate when comparing body fluids from different animals, which may contain different total histone H3 concentrations and histones originating from neutrophils at different stages of maturity.
Complexes of NET Components
Demonstration of DNA or histones complexed with neutrophil granule proteins (eg, NE or MPO) is generally considered specific for NETosis. 137 Three approaches that are commonly used to quantify DNA/granule protein complexes are ELISA, immunofluorescence, and flow cytometry. It should be noted, however, that extracellular interactions between negatively charged DNA and positively charged proteins such as NE and MPO might mimic NETs 194 and create false-positive results. 84
Sandwich ELISAs have been constructed with an anti-DNA and anti-myeloperoxidase antibody pair to quantify DNA/myeloperoxidase complexes in human and murine body fluids. 24,91,134,214 The assays have yet to be fully validated, however, and presently there are no equivalent commercial kits. Creation of a robust ELISA for NET fragments is challenging, not least because NETs are highly variable in size. Large DNA fragments might physically block access of the anti-protein antibody to its substrate. This was proposed as the reason for erroneous results with a commercial anti-citrullinated histone/myeloperoxidase complex ELISA that was withdrawn from the market shortly after release (Cayman Chemical, personal communication). Exposing samples to brief DNase digestion before analysis to reduce the size of the DNA components might help, 88 but care must be taken to avoid overdigestion that may disrupt the NETs and produce false-negative results.
Immunofluorescence demonstrating spatial overlap of staining for DNA and protein components has been used to detect NETs in isolated neutrophil preparations, 18 tissue sections, 16 body fluid smears, 11,65,66,111 and in vivo. 141 Quantification can be performed by manually counting NETs, but this is subjective and susceptible to error induced by microscopic field selection. 36 Automated or semi-automated alternatives are described and could improve objectivity and reduce the labor-intensive nature of manual quantification. 17,66,145 Further work is required to fully validate microscopy-based biomarkers for use in clinical samples and to investigate the possibility that cells lysed during blood smear production may mimic NETs. 121
Several strategies are described for quantification of NETs in whole blood or isolated neutrophil preparations by flow cytometry, sometimes combined with image analysis. 55,101,138,233 These focus on identification of early morphologic changes associated with NETosis 233 or cell-associated NETs rather than circulating NET fragments. 55,101,138 Flow cytometry offers the potential to overcome the subjectivity that affects many immunostaining studies, 101,138 but information regarding the performance of flow cytometry as a biomarker of NETosis is currently limited. 125 For whole blood studies, the effects of pre-analytical factors (eg, time between collection and processing) have not been reported, and there is a lack of detailed descriptions of analytical performance criteria such as coefficients of variation. 55,101 Further work is needed to assess these various aspects of flow cytometric NETosis detection, but this area clearly holds future promise.
There is now a substantial body of literature describing measurements of NETosis biomarkers in a variety of sample types collected from various companion animal patient populations. The major limitation of this literature base, however, is the lack of specificity of many of these biomarkers for NETosis. As discussed above, there are multiple potential sources of biomarkers, such as cfDNA and histones, aside from neutrophils undergoing NETosis. In particular, apoptosis and necrosis likely contribute to the concentrations of HMGB-1, cfDNA, nucleosomes, and histones reported in the literature. Indeed, the cfDNA measured in patients with cancer may actually originate from the neoplastic cells themselves, potentially through apoptosis or cell necrosis. 8,9,102 In other scenarios, plasma cfDNA measured in companion animals may not even represent host DNA but could be present due to concurrent parasitemia due to Babesia spp., Leishmania, or filarial nematodes. 1
As such, currently the best evidence for NETosis in veterinary medicine likely comes from measurements of citrullinated histones and from imaging studies that document the presence of NETs in samples collected and handled so as to prevent ex vivo activation. Studies that have measured multiple biomarkers of NETosis are likely more robust. As previously noted, however, almost all potential NETosis biomarkers have overlapping origins that can confound interpretation of histone, nucleosome, and HMGB-1 measurements.
NETosis in Diseases of Animals
Infection
Some of the most compelling evidence for NETosis in companion animals relates to their role in antimicrobial and antiparasitic host defense. cfDNA and nucleosome concentrations have been investigated as prognostic biomarkers in canine sepsis. 107 A prospective study enrolled 45 dogs with sepsis, 10 dogs with nonseptic systemic inflammatory response syndrome, and 15 healthy controls. Biomarker concentrations were measured at admission, and patients were followed to hospital discharge. cfDNA and nucleosome concentrations were significantly higher in dogs with sepsis compared to healthy controls. cfDNA concentrations were also significantly higher in dogs with sepsis that had bacteremia compared to those with negative blood cultures, suggesting that this cfDNA may derive from intravascular NETosis. As such, the findings suggest that point-of-care cfDNA measurement might aid identification of bacteremia in dogs with signs of sepsis.
Canine neutrophils release NETs in vitro in response to E. coli lipopolysaccharide. 112 NETs were visualized by fluorescence microscopy of living neutrophils and using immunofluorescent microscopy for the NET components cfDNA, citrullinated histone H3, and myeloperoxidase. This process appears to be dependent on PAD4 because it was inhibited by the PAD4 inhibitor Cl-amidine. Compared to NETs induced by PMA, LPS stimulation resulted in smaller NETs with less extracellular citrullinated histone H3. In one case series of dogs with sepsis, NETs were identified using immunofluorescence microscopy in cytology samples of septic foci. 111 Samples of endotracheal tracheal wash, bronchoalveolar lavage fluid, and abdominal and pleural effusion collected from 3 dogs with sepsis and 2 other nonseptic disease processes were fixed, permeabilized, and stained for MPO, citrullinated H3, and cfDNA. NETs were identified based on co-localization of these components and quantified compared to the total number of neutrophils. NETs were identified in the fluid samples collected from all 3 septic dogs and in limited numbers from a dog with sterile chronic bronchitis.
A prospective study evaluated uteri removed from normal dogs during routine spay procedures and from dogs with pyometra. 164 Uterine contents were evaluated by bacterial culture and for identification of NETs by immunocytochemistry for histone, NE, or MPO; fluorescence microscopy; and scanning electron microscopy. The study identified NETs in uterine contents in all pyometra cases and could be seen entrapping bacteria. No bacteria and no NETs were identified in the control uteri.
A similar study by the same group evaluated the in vitro capacity of equine neutrophils to generate NETs by chemical activation or through ex vivo stimulation with bacteria associated with equine metritis, including Streptococcus equi zooepidemicus, E. coli, and Staphylococcus capitis. 166 Incubation of neutrophils with bacteria alone significantly increased NETosis compared to neutrophils treated only with PMA. Streptococcus equi zooepidemicus induced fewer NETs than did E. coli or Staphylococcus capitis. In addition, NETs were present in endometrial mucus samples taken from mares with bacterial endometritis. Although NETs may play protective roles in combating the bacteria that cause endometritis, certain components of NETs have the capacity to trigger endometrial fibroplasia that could lead to excessive collagen deposition and endometrial fibrosis. In an in vitro study, incubation of endometrial tissue explants with NET components including NE, cathepsin G, and MPO was associated with an increase in type I collagen protein expression. 165 It remains to be determined if this process can also occur in vivo and if it contributes to the pathogenesis of equine endometrial fibrosis. In a study evaluating the peritoneal fluid of women with endometriosis, NETs were present in 49% of patients, compared to only 18% of controls. In addition, when NETs were present, there were significantly more of them in the peritoneal fluid of women with endometriosis compared to controls. 12
Feline neutrophils stimulated with protozoan parasites release structures comprising DNA and histones consistent with NETs. 218 Neutrophils from cats with symptomatic FeLV infections have significantly greater NET release by unstimulated neutrophils compared to FeLV– and FeLV+ asymptomatic cats. This suggests that feline NET release can be modulated by viral infection and suggests the mechanism is overactivated in FeLV+ cats.
Although macrophages are the definitive host cells for Leishmania in dogs, neutrophils are the first cells to encounter the parasite following inoculation. Canine blood neutrophils isolated from healthy dogs were exposed to L. infantum promastigotes in vitro. 159 The neutrophils efficiently engulfed the parasite and generated intracellular superoxide radicals, but there was minimal release of NETs. This may suggest that NETosis is not an important host defense response against Leishmania or that the conditions necessary for efficient NETosis (eg, concurrent cell types such as platelets) were not present in the experimental system employed. Trypanosoma cruzi efficiently induced canine blood neutrophils to form NETs that were decorated with NE, as evaluated by cfDNA measurement and immunofluorescence microscopy. 35 Although T. cruzi can induce NET formation, the role of this host defense response in vivo is uncertain, because T. cruzi can circulate for years in dogs. The parasite can reside intracellularly in macrophages, which may reduce NET formation and help protect it from the host’s defenses.
Canine Immune-Mediated Hemolytic Anemia
Immune-mediated hemolytic anemia in dogs is strongly associated with thrombosis 37 ; hence, there is considerable interest in potential links between NETs and immunothrombosis in this condition. Nucleosomes (DNA-histone complexes) may also play physical roles in promoting and propagating clot formation, since they contribute to the physical clot structure and impair clot dissolution by inhibiting fibrinolysis. 178 cfDNA competes with plasmin on fibrin, which results in decreased clot lysis. 47 cfDNA can also suppress fibrinolysis by accelerating the inactivation of tissue plasminogen activator (tPA) by plasminogen-activator inhibitor-1 (PAI-1). 117 Canine NETs have been demonstrated to increase the rate of thrombus formation and reduce the potential for fibrinolysis of any resulting blood clots. 82 There is solid evidence that NETosis occurs in dogs with IMHA. Nucleosomes were measured in the serum of dogs with primary immune-mediated hemolytic anemia using a commercially available ELISA and were significantly higher in IMHA cases than controls. 81 A subsequent prospective observational study of 28 dogs with IMHA and 20 healthy controls was conducted to assess the cfDNA concentrations as measured by PicoGreen fluorescence. 83 Concentrations of cfDNA were significantly higher in dogs with IMHA compared to controls, and high cfDNA concentrations were associated with increased odds of death. The origin of this cfDNA is uncertain, but it appears to result from increased release rather than decreased degradation, because DNase activity was not different between cases and controls. In a comparable study by a separate group, concentrations of cfDNA, nucleosomes, and citrullinated histone were measured in plasma samples from 35 dogs with IMHA and from 56 healthy controls. 100 cfDNA concentrations were above the reference interval in 17% of dogs with IMHA, while nucleosome concentrations were increased in almost all dogs with IMHA. Citrullinated histone H3 was detected in 84% of samples from dogs with IMHA.
Asthma
Equine asthma, also known as recurrent airway obstruction, is a disease characterized by reversible airway obstruction, bronchoconstriction, and neutrophilic inflammation. Bronchoalveolar lavage fluid from horses with active recurrent airway obstruction contained NETs, while BAL fluid from horses with recurrent airway obstruction in remission did not. 31 Interestingly, horses express 2 distinct secretoglobin family 1A members. These proteins are secreted by bronchiolar epithelial cells and are a major constituent of airway surface fluid. Their concentrations are reduced in human asthma and in equine recurrent airway obstruction. Ex vivo, recombinant secretoglobin 1A1A increases neutrophil oxidative burst and phagocytosis, reduces neutrophil chemotaxis, and inhibits NET formation. It is undetermined if these phenomena are mechanistically and causally related or if they represent epiphenomena in equine recurrent airway obstruction.
Low-density granulocytes are a distinct subset of proinflammatory cells that can be isolated from the peripheral blood mononuclear cell fractions of patients with immune-mediated and neoplastic diseases. These cells have an increased propensity for NETosis. 23 A study compared the low-density neutrophil profiles of horses with severe equine asthma to those of healthy controls. 73 The response profile of the low-density neutrophils in these animals was most consistent with a pro-inflammatory low-density granulocyte morphology. Specifically, these cells had increased expression of N-formylmethionine-leucyl-phenylalanine receptors and an increased capacity to produce NETs. However, there were no differences between the healthy and the asthmatic horses.
NETs within the lungs have been associated with the severity of airway obstruction and inflammation in humans with asthma. Similarly, NETosis was 2-fold higher in lavage fluid from the lungs of asthmatic horses compared to controls. 206 Induced NETosis was enhanced in airway neutrophils from asthmatic horses relative to controls, but there was no difference in NETosis by blood neutrophils between healthy and asthmatic horses. Similarly, glucocorticoids did not affect NETosis by blood neutrophils but did diminish NETosis by airway neutrophils.
Toxicant Exposure
Two studies have evaluated the effect of environmental toxicants on NET formation by canine neutrophils. Exposure of canine neutrophils to sodium arsenite leads to formation of extracellular structures composed of DNA decorated with histones, NE, and MPO. 219 These structures were visualized by fluorescence confocal microscopy and appear consistent with previous descriptions of NETs. The mechanisms of NET formation in response to sodium arsenite are unknown at this time, however. Exposure of canine neutrophils to nickel nitrate hexahydrate (a common heavy metal used in batteries) also causes release of NETs based on fluorescence confocal microscopy observations. 220 In addition, nickel exposure increased cfDNA release as quantified by a PicoGreen fluorescence assay. The mechanism of nickel-induced NET formation remains obscure, and the clinical significance of these findings is uncertain, particularly because environmental exposures of companion animals to nickel or arsenic are rare.
Trauma
A preliminary study assessed several techniques for measurement of cfDNA in canine plasma obtained from dogs with cancer, with sepsis, and following trauma. 108 Concentrations were also measured in samples from healthy controls. Compared to controls, plasma cfDNA concentrations were significantly increased in dogs with sepsis, trauma, and neoplasia. The findings were extended to a prospective case-control study in which plasma cfDNA and nucleosome concentrations were evaluated as prognostic biomarkers in canine trauma. 106 The study enrolled 49 dogs with moderate-severe trauma and followed them to hospital discharge. Concentrations of cfDNA and nucleosomes were positively correlated and were significantly higher in injured dogs compared to healthy controls. Only nucleosome concentration correlated with injury severity scores. Non-survivors had significantly higher nucleosome concentrations than survivors. Although cfDNA concentrations have been documented to be increased in dogs following trauma, the kinetics of this biomarker over time are unknown. A prospective study evaluated 3 groups of dogs undergoing ovariohysterectomy, cranial cruciate ligament repair, or hemilaminectomy and measured cfDNA concentrations repeatedly in the 72 hours following surgery. 223 This study suggested that the biological half-life of plasma cfDNA is ∼6 hours. In dogs undergoing hemilaminectomy, cfDNA concentrations differed significantly between the time of admission and 6–12 hours after surgery. The authors suggested that the short half-life of cfDNA might be useful for assessment of acute severe tissue injury.
Other Conditions
A study compared the plasma cfDNA concentrations in a heterogeneous population of 97 dogs with various disease processes to those of 24 healthy dogs. 20 Plasma cfDNA was measured by fluorometry without prior DNA extraction. The diseased dog population had significantly higher cfDNA than clinically normal dogs and the concentration of cfDNA correlated with disease severity as assessed by American Society of Anesthesiologists status. Within the diseased population, non-survivors had significantly higher cfDNA concentrations than survivors. No attempt was made to identify the origin of the cfDNA measured; hence, the contribution of NETosis to these results cannot be determined. The authors concluded that measurement of canine cfDNA could be a useful nonspecific disease indicator and prognostic tool.
Strenuous exercise has been demonstrated to increase plasma cfDNA concentrations in dogs, 78 but cfDNA concentrations do not appear to be useful for identification of exertional rhabdomyolysis in racing sled dogs. 38 Dogs with gastric dilation and volvulus had significantly greater plasma cfDNA concentrations compared to controls, but cfDNA concentrations were not predictive of complications or of survival. 200
In an experimental model in dogs, autologous blood clots were injected to induce pulmonary thromboembolism, and then plasma cfDNA concentrations were measured with a PicoGreen assay. 203 Blood clot size correlated with induced pulmonary vascular resistance, pulmonary artery pressure, and plasma cfDNA concentrations. The cfDNA release was thrombus-dependent, because microsphere lung embolization did not alter plasma cfDNA concentrations. The same group also demonstrated that autologous blood clots allowed to undergo fibrinolysis release DNA in vitro.
Heat stroke in dogs is also associated with NETosis. In a prospective study, serum histones were measured at hospital admission in 16 dogs with heatstroke and compared to those of 7 healthy controls. Serum histone concentrations in dogs with heatstroke were significantly higher than in controls. 19 Serum histone concentrations were also significantly higher among non-survivors and in those dogs with severe hemostatic derangements. The correlation between serum histones and coagulation abnormalities may reflect disease severity or might suggest a mechanistic link between histone release and hemostatic derangement. Further investigation of this finding is required. The correlation between histone concentrations and coagulation abnormalities is of interest, because histones may be amenable to pharmacologic manipulation.
NETs as Therapeutic Targets
Since the first description of NET formation in 2004, 18 and the recognition of the thrombotic potential of these structures, 46,57 there has been an explosion of literature in this field. As understanding of the nature, mechanisms, and consequences of NETosis has grown, so has interest in therapeutic manipulation of this process. The complex and orchestrated nature of NETosis itself, and the multicomponent nature of the structures generated, provides numerous putative therapeutic targets. The major treatment strategies currently under investigation are summarized in Table 1, and the most important are discussed in detail below. To date, no studies of therapies targeting NETosis in companion animals have been reported.
Major Treatment Strategies Currently Under Investigation for the Management of Immunothrombosis.
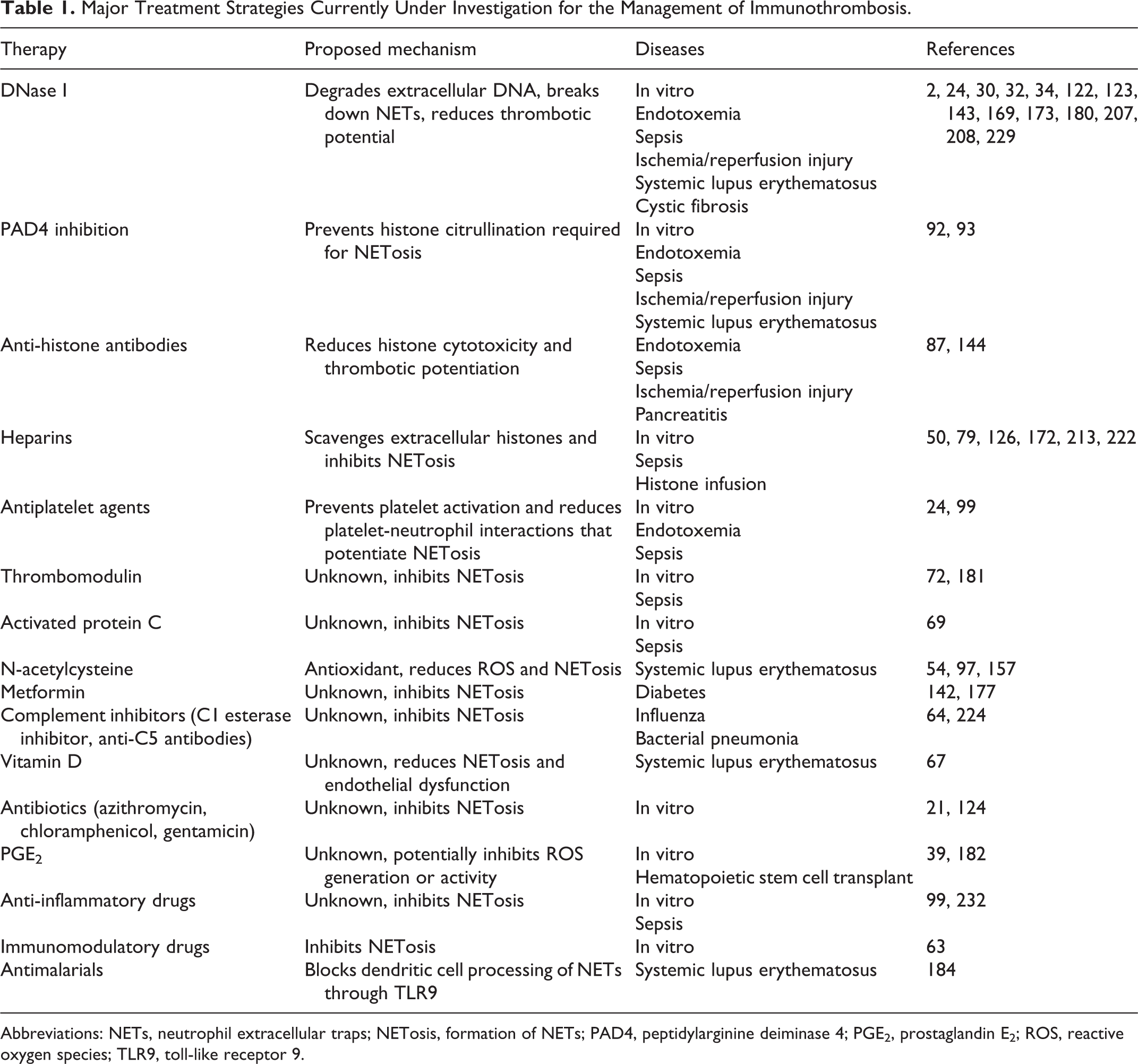
Abbreviations: NETs, neutrophil extracellular traps; NETosis, formation of NETs; PAD4, peptidylarginine deiminase 4; PGE2, prostaglandin E2; ROS, reactive oxygen species; TLR9, toll-like receptor 9.
While NETs and immunothrombosis may offer clinicians the potential to prevent and treat disease, it is clear that NETs are Janus-faced. Constrained, regulated, and controlled NETosis is likely protective, while excessive NETosis contributes to organ dysfunction and mortality through immunothrombosis. This poses substantial challenges for clinicians seeking to intervene therapeutically. 171 For instance, treatment of murine Staphylococcus aureus skin infections with DNase, an enzyme that digests DNA, leads to systemic bacterial dissemination. 230 Similarly, early treatment of septic mice with DNAse leads to deterioration in their condition, but late treatment reduces mortality. 123 A similar study also demonstrated that systemic treatment with recombinant DNase attenuated sepsis-induced organ dysfunction and improved survival in septic mice. 32 Much work remains to be done identifying the safest and most effective treatment strategies and determining which patients will benefit.
DNase
NETs are primarily composed of extracellular DNA. This cfDNA is crucial for the capture of microbes but also plays a prominent role in immunothrombosis by restraining and co-localizing high-molecular-weight kininogen in proximity to the primary factors of the contact pathway of coagulation. 153 The helical structure of DNA is also important for contact pathway activation. Digestion of DNA by DNase abolishes these procoagulant effects. 59,191 Humans with acute thrombotic microangiopathies have increased plasma concentrations of NETosis biomarkers. These patients have deficient DNase activity, such that supplementation of plasma with recombinant human DNase I may restore endogenous NET-degradation activity in these patients. 86
There is considerable interest in the use of DNase I in the management of autoimmune conditions such as systemic lupus erythematosus. 7,130 Human patients in the 1950s and 1960s were given the compound on an experimental basis with anecdotal temporary improvement in their conditions. Development of recombinant human DNase I in the 1990s enabled management of sputum viscosity in patients with cystic fibrosis. 180 Studies in mouse models of systemic lupus erythematosus suggest that recombinant DNase I may improve kidney histologic lesions and prolong survival, 122,207 but these effects are not consistent. 208 A phase Ib, randomized, blinded placebo-controlled trial of 17 patients with systemic lupus erythematosus nephritis suggested that recombinant DNase I is safe, achieves appreciable activity after IV administration, and was not associated with the development of neutralizing antibodies. No beneficial effects on markers of the disease process were discernable, however. 34
Treatment of mice subjected to ischemia-reperfusion injury with DNase I (during ischemia and reperfusion periods) significantly reduced NETosis, inflammatory cell infiltration, and thrombin-antithrombin complex generation and improved perfusion in post-ischemic muscle. This DNase treatment did not alter skeletal muscle fiber injury or concentrations of proinflammatory molecules, however. 2
Treatment of patients with suspected sepsis is an attractive possibility, but using DNase carries risk because the cause of the inflammatory response seen clinically is not always readily apparent, and it is possible to mistake the nature of the pathogen. NETs may be essential for defense against fungal pathogens, and inadvertent DNase intervention might cause a deterioration in the patient’s condition. 15 DNase treatment of sepsis is also complicated by temporal effects. Studies to date have used mouse cecal-ligation puncture models and suggest that early therapy is deleterious, while late treatment reduces organ damage and bacterial dissemination. 123 Concurrent treatment with DNase and antibiotics resulted in improved survival, reduced bacteremia, and organ dysfunction in a separate murine study, however, 32 which might indicate that combined therapies may limit any detrimental effects of the DNase therapy and potentiate the benefits.
Experimental studies have linked NETosis to acute lung injury, 173 a phenomenon that is likely dependent on platelet-neutrophil interactions. 24 DNase therapy in disorders like transfusion-related acute lung injury may be beneficial. 198 Results from murine studies of DNase therapy in lung injury are not consistent, however, with 2 studies evaluating DNase therapy in models of ventilator-induced lung injury demonstrating varying levels of benefit from this intervention. 169,229 These disparities may result from different disease severities at the time of intervention, particularly if DNase therapy alters aspects of neutrophil biology other than NETs, such as inhibiting cell recruitment. The tissue location in which NETosis is occurring (eg, blood vs lung vs liver, etc) may influence the outcome of therapy by allowing the spread of organisms in one area, while reducing thrombotic complications in another. 30,143
PAD4 Inhibition
The enzyme PAD4 is a key therapeutic target for diminishing NETosis because of its essential role in catalyzing the histone citrullination crucial for NET formation. 109 Sepsis models utilizing PAD4-knockout mice demonstrated that PAD4 deficiency improves survival and decreases the severity of organ dysfunction without exacerbating bacteremia. 13,132 In a separate murine study of lethal endotoxemia, LPS-induced NETosis was inhibited by treatment with the PAD2/PAD4 inhibitor YW3-56. 116 This therapy also reduced pulmonary vascular leakage and diminished the resulting lung injury and translated into a survival benefit. Systemic treatment of mice with inhibitors of PAD4 improves survival in sepsis models 115 and protects hepatocytes from liver ischemia-reperfusion injury. 77 PAD4 is also an important target in autoimmune disease. In mouse models of SLE, inhibition of PAD4 with the drug BB-Cl-amidine decreased autoimmune responses, protected against vascular damage, decreased proteinuria, and reduced skin and endothelial dysfunction. 92,93 The same PAD4 inhibitor also reduced mortality in a murine sepsis model. 14 Despite all these potential benefits, the long-term physiologic consequences of systemic PAD4 inhibition are unknown. As such, therapeutic strategies aimed at PAD4 may have unintended consequences, and safe therapies targeting this enzyme may be difficult to develop.
Anti-Histone Antibodies
In the extracellular environment, histones are pro-inflammatory, cytotoxic, and prothrombotic. 192 Histones facilitate thrombus formation in both platelet-dependent and platelet-independent manners 59 and impair clot dissolution by inhibiting fibrinolysis. 178 Histones are key components of NETs, making them a rational potential target for therapeutic interventions. Multiple strategies have been used to reduce the cytotoxicity, organ dysfunction, and mortality that can arise from increased concentrations of histones in tissues or in circulation, 192 but several histone subtypes exist. This complicates therapeutic intervention, particularly because histone subtype varies with the disease process. For instance, antibodies against histone H3 may reduce mortality in mouse models of pancreatitis, 87 while in mouse models of hepatic ischemia/reperfusion injury, histone neutralization using antibodies against both histones H3 and H4 significantly protects against injury. 76 In a histone-induced acute kidney injury model, antibodies against histone H2A and H4 suppressed renal inflammation, neutrophil infiltration, and tubular cell necrosis and improved excretory renal function. 4 The same antibodies also reduced mortality in lipopolysaccharide, tumor necrosis factor or cecal-ligation, and puncture models of sepsis in mice. 226 Separately, antibodies against H1 reduce the mortality of mice in LPS models of sepsis, 95 and anti-H3 antibodies also significantly improve survival in mouse cecal-ligation and puncture sepsis models. 115 As such, anti-histone antibody therapies will likely need to be targeted to individual disorders, and this may also be species-specific. At this time, anti-histone antibody therapies have been deployed only in mice; hence, it is uncertain if such therapies will generalize to humans or to companion animals.
Heparins and Antiplatelet Agents
Antithrombotic therapies may be of value in the management of excessive NETosis and immunothrombosis. 114 Potential therapeutic strategies include the use of unfractionated heparin, low-molecular-weight heparins, and antiplatelet agents to target distinct aspects of immunothrombosis pathogenesis. 126,172 Extracellular histones released during NETosis can be scavenged by heparin and may prevent histone interactions with tissues and with platelets. 50 These effects are noteworthy, because treatment of dogs with individually dose-adjusted heparin improves survival in IMHA, 71 a disease in which NETosis has been repeatedly demonstrated. 81,83,100 Heparin, through unknown mechanisms, has been shown to inhibit NETosis in human neutrophils ex vivo. 126 Unfractionated heparin improves survival in mouse cecal-ligation and puncture models of sepsis better than antibiotics alone, while the combination of antimicrobial drug therapy and heparin was superior to either. 213 In rat models of histone-induced organ dysfunction, both unfractionated heparin and low-molecular-weight heparins reduced histone-induced leukopenia and thrombocytopenia, diminished organ dysfunction, and improved survival. 79 Non-anticoagulant heparin, which binds to extracellular histones but has minimal affinity for antithrombin, has also been shown to reduce histone-mediated cytotoxicity, attenuate endotoxin-mediated lung injury, and improve survival in septic mice. 222
Pharmaceutical activated protein C is no longer available for use in humans 129 ; however, interest remains in determining how the drug affects NETosis. In human sepsis, plasma histone concentrations are inversely related to endogenous aPC activity, 44 and several studies suggest that aPC reduces the toxicity of histones 179,226 and inhibits NETosis. 69 Similarly, thrombomodulin attenuates histone-induced thrombin generation and endothelial cell death in vitro, likely through its ability to activate protein C. 154 Thrombomodulin also reduces NETosis and blunts the prothrombotic state resulting from cecal-ligation and puncture-induced peritonitis in rats. 72 Recombinant human thrombomodulin is used successfully in humans with sepsis-induced DIC in Japan, 68,228 and Phase III clinical trials are ongoing in the USA (NCT01598831, NCT03517501). If these prove successful, then some of the benefits may derive from inhibition of immunothrombosis.
Platelet activation and platelet-neutrophil interaction are crucial for NETosis in vivo. Thus, antiplatelet therapy may attenuate NETosis and reduce the potential consequences of excessive immunothrombosis. Endotoxin-mediated platelet activation is associated with increased thromboxane A2 generation. 150 This is noteworthy because pretreatment of mice with aspirin decreases NETosis and lung injury following endotoxemia. 24 Similarly, endotoxin-mediated platelet activation is dependent on ADP in canine and equine platelets, 112 which suggests that clopidogrel therapy might attenuate platelet-neutrophil interaction and NETosis. In humans with sepsis, prior antiplatelet therapy is associated with improved outcome. 26,43 This finding prompted initiation of a clinical trial investigating aspirin therapy in people with sepsis, 42 which completed the enrollment of 17,000 patients in June 2018. Publication of this trial is still pending. Platelet inhibition in sepsis may have unexpected effects, however. In a murine peritonitis model, pretreatment of mice with aspirin resulted in suppression of NETosis coupled with increased bacteremia. 99 As with other therapies to regulate NETosis, timing of therapy, as well as type and location of infection, may alter the impact of antiplatelet therapy on outcomes in humans and animals with sepsis.
Conclusions
The processes of NETosis and immunothrombosis are complex, orchestrated, and integral components of the innate immune response, but when misdirected or in excess, these processes can be harmful to the host. Detecting NET components such as cfDNA or histones is straightforward, but accurately and definitively determining the origin of these components is much more difficult. Developing ways to enhance differentiation of NETosis from apoptosis and necrosis will be crucial to the continued development of this field and will underpin the use of therapeutic interventions. The study of NETs and immunothrombosis holds much future promise both diagnostically and therapeutically, but a considerable amount of work remains to be done in this arena.
Footnotes
Author Contribution
Robert Goggs, Unity Jeffery, Dana N. LeVine, Ronald H. L. Li All authors contributed equally to this manuscript
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
