Abstract
Genetically engineered mouse lines on a C57BL/6J background are widely employed as preclinical models to study neurodegenerative human disorders and brain tumors. However, because of the lack of comprehensive data on the spontaneous background neuropathology of the C57BL/6J strain, discriminating between naturally occurring changes and lesions caused by experimental mutations can be challenging. In this context, this study aims at defining the spectrum and frequency of spontaneous brain changes in a large cohort of C57BL/6J mice and their association with specific biological variables, including age and sex. Brains from 203 experimentally naive and clinically unremarkable C57BL/6J mice were collected and analyzed by means of histopathology and immunohistochemistry. Mice ranged in age from 3 to 110 weeks with 89 females, 111 males, and 3 unknowns. Sixteen different spontaneous lesion categories were described in this cohort. Age-related neurodegenerative and/or neuroinflammatory findings represented the most common pathologic changes and included (1) Hirano-like inclusions in the thalamic neurons, (2) neuroaxonal dystrophy in the medulla oblongata, (3) periodic acid–Schiff–positive granular deposits in the neuropil of the hippocampus, and (4) progressive neuroinflammation characterized by microgliosis and astrogliosis. Neoplastic conditions, developmental abnormalities, and circulatory disorders were rarely observed incidental findings. In conclusion, this study describes spontaneous age-related brain lesions of the C57BL/6J mouse and provides a reference for evaluating and interpreting the neuropathological phenotype in genetically engineered mouse models developed and maintained on this congenic background.
Keywords
Definition of the background or incidental lesions associated with a specific mouse strain is vital to the correct interpretation of pathological endpoints in the experimental context. 10,30,60,62 C57BL/6J is arguably the most common inbred mouse strain employed in biomedical research, representing the reference congenic/consomic background to generate mouse lines harboring both spontaneous and genetically engineered mutations. 67 C57BL/6J also represents the preferred congenic background for numerous mutant models of neurological conditions, including important neurodegenerative disorders and brain tumors. As an example, genetically engineered mouse (GEM) models carrying mutations in genes responsible for familial Parkinson disease, such as LRRK2, PINK1, PRKN, and SNCA, have been preferentially developed and maintained on an incipient or full C57BL/6J congenic background. 14,27,31,35,42,72 Similarly, the C57BL/6J background is prevalent in GEM lines featuring mutations of tumor suppressor genes, including Pten, Trp53, and Ptch1, responsible for the development of both glial and primitive neuroectodermal tumors. 15,75
Exhaustive historical control data on spontaneous neuropathological changes do exist for the mouse strains or stocks that are most commonly used in toxicological pathology (eg, CD-1, B6C3F1). 61 The neuropathology of certain progeroid mouse models, such as the senescence accelerated mice P8 (SAMP8) or GEM lines with Ercc5 deficiency, is also well characterized. 2,4 However, comprehensive studies detailing the spontaneous brain findings in C57BL/6J mice are lacking.
To address the critical lack of knowledge in this area, our study aims at providing an exhaustive description of the spectrum, nature, and frequency of spontaneous brain pathology in a large cohort of C57BL/6J mice over the course of their natural life span. Our assessment also unveils pathological associations between specific lesion categories and physiological variables, including aging and sex. A correct understanding of spontaneous brain changes in such an important mouse strain is essential for distinguishing experimentally induced lesions from those inherently determined by the genetic background and/or arising as a result of normal aging.
Materials and Methods
Animals and Husbandry
The 203 animals considered in this study belonged to a large cohort of experimentally naive and clinically unremarkable (ie, unremarkable in appearance and behavior to animal care staff, neurological assessment was not routinely performed) C57BL/6J mice used as age- and sex-matched wild-type (WT) controls for the neuropathological characterization of a series of GEM lines maintained on a C57BL/6J background. The mouse population consisted of 111 males, 89 females, and 3 animals of unknown sex, and the age ranged from 3 to 110 weeks. The following age categories were considered within the study population: juvenile (3–9 weeks, 35 mice including 17 males, 15 females, and 3 unknown), young adult (10–26 weeks, 70 mice including 30 males and 40 females), adult (27–77 weeks, 51 mice including 35 males and 16 females), and aged (more than 77 weeks, 47 mice including 29 males and 18 females).
All the analyzed mice were kept under the same husbandry conditions within the SPF facility of the Catholic University of Leuven (KU Leuven). Mice were group housed (4 mice) in filter-top polysulfone cages (H-Temp; Tecniplast, Buggugiate, Italy) containing BK8/15 Lignocel wood bedding (J. Rettenmaier & Söhne GmbH, Rosenberg, Germany). Food (standard rodent diet, Ssniff R/M-H, Soest, Germany) and UV-filtered drinking water were provided ad libitum. Animal rooms were maintained at 22°C ± 2°C with a 45% to 70% relative humidity range and 12/12-hour light/dark cycle. Mice were included in a health monitoring program developed in accordance with the Federation of European Laboratory Animal Science Associations (FELASA) guidelines. Procedures involving animals were performed in accordance with the guidelines of the KU Leuven Animal Care and Use Ethical Committee, which specifically approved this study (reference number 072/2015).
Brain Collection and Pathological Examination
Most of the animals were euthanized via carbon dioxide asphyxiation and then immediately decapitated. Skin and soft tissue (including eyes and adnexa) were removed from the head. Four holes were drilled with a 26-gauge needle along the midsagittal parietal and frontal sutures of the skull to allow more rapid penetration of the fixative solution into the cranial cavity. Finally, the head was immersed in 50 ml of 10% neutral buffered formalin (NBF) and fixed for at least 2 weeks.
In a few instances, mice were anesthetized with a mixture of xylazine (25 mg/ml), ketamine (20 mg/ml), and atropine (20 ng/ml), followed by intracardial perfusion of ice-cold phosphate-buffered saline (PBS) (approximately 10 ml) and ice-cold 4% paraformaldehyde (PFA) (approximately 15 ml). The entire head was then collected as previously described and immersion fixed for at least 72 hours in 4% PFA at 4°C.
Upon complete fixation, the brains were carefully removed from the skull and trimmed, as explained hereafter, using a mouse brain matrix (BSMYS001-1; Zivic Instruments, Pittsburgh, PA). Each brain was first sliced along a midsagittal plane. Five coronal slices were then obtained from the left hemibrain using the following anatomical landmarks: paraflocculi, mammillary bodies, caudal profile of the optic chiasm, and the most ventral aspect of the olfactory tubercles. Coronal slicing of the entire brain without midsagittal separation was performed in a small subset of brain samples, especially from the younger age group. Whole body and brain weight were recorded for 78 individuals in total from the young adult, adult, and aged groups.
All samples were routinely processed for paraffin embedding, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). Both coronal and sagittal brain sections stained with HE were examined histologically. Two serial sections each of sagittal and the coronal samples were typically examined. The histopathologic criteria outlined in the INHAND diagnostic scheme were applied for the classification of brain lesions. 41
Immunohistochemistry (IHC) was performed on additional sections obtained from selected cases (the methods are described in Suppl. Table S1). Periodic acid–Schiff (PAS) and phosphotungstic acid hematoxylin (PTAH) histochemistry was also performed on a selected subset of samples following the protocols of the Armed Forces Institute of Pathology. 59
Quantitative Analysis of Gliosis
Quantitative analysis of glial fibrillary acidic protein (GFAP) and ionized calcium-binding adapter molecule 1 (Iba1) IHC was performed on 2 sets of 5 sagittal brain samples selected from the male young adult and aged group, respectively. The resulting slides were scanned using an Aperio VERSA 200 (Leica Biosystems, Buffalo Grove, IL). Quantification of the immunoreactivity for each marker was assessed in highly comparable microanatomical regions (ie, cerebellar cortex, cerebellar white matter, medulla, cerebral cortex, hippocampus, thalamus, and hypothalamus) using the Positive Pixel Count v9 algorithm in the Aperio ImageScope software.
Statistical Analysis
Statistical analyses were performed with Microsoft Excel 2016 (Microsoft Corporation, Redmond, WA). Chi-square tests were used to analyze variations in the spectrum and frequency of lesions across the different age and/or sex groups identified in the study population. Unpaired 2-tailed Student’s t test was used to assess the statistical significance of the brain weights across the different age and/or sex groups as well as the quantitative analysis of GFAP and Iba1 IHC performed in the young adult and aged groups. P < .05 was considered statistically significant.
Results
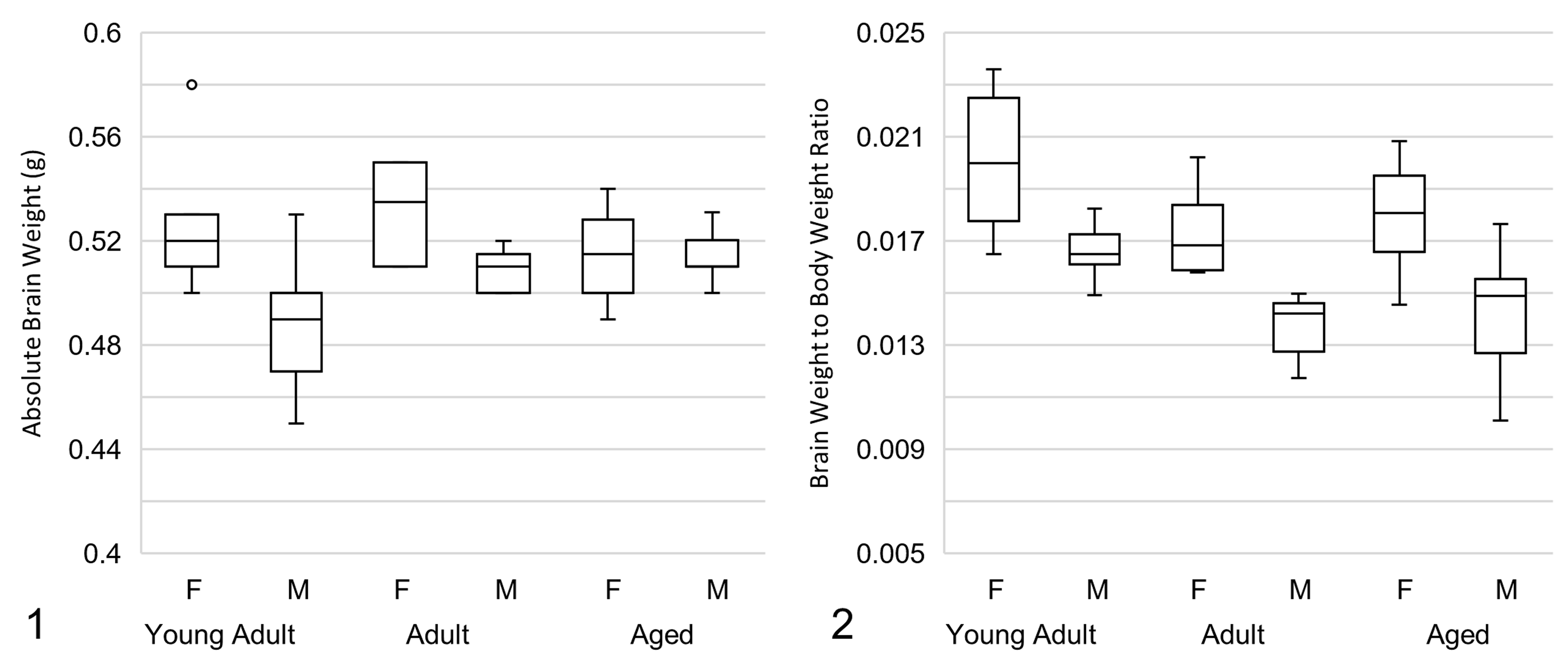
The spectrum and frequency of spontaneous brain lesions and results of the IHC analysis are summarized in Tables 1 and 2, respectively. There was a small, statistically significant increase in absolute brain weight in the adult and aged groups vs the young adult group (P = .023 and P = .013, respectively; Figures 1 and 2). The brain to body weight ratio showed a small but highly significant decrease in the adult and aged groups vs the young adult group (P < .005 for both comparisons). Absolute brain weight was slightly higher in females vs males in the young adult and adult groups (P < .005 for both comparisons). The brain to body weight ratio was slightly higher in females in all 3 groups (P < .001 for all comparisons). Brain weight of the juvenile group was not assessed.
Frequency of Spontaneous Lesions in Clinically Normal C57BL/6J Mice.
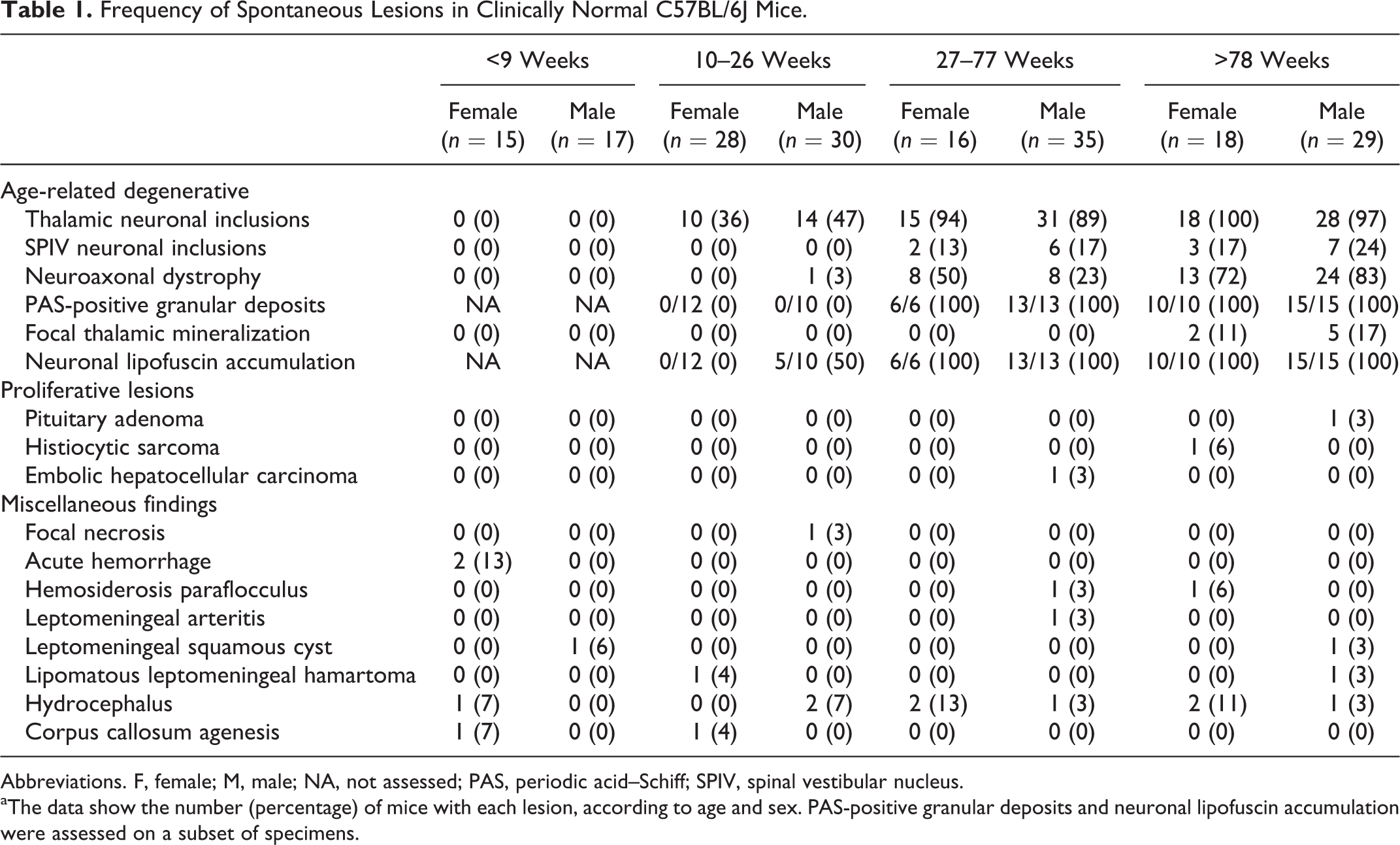
Abbreviations. F, female; M, male; NA, not assessed; PAS, periodic acid–Schiff; SPIV, spinal vestibular nucleus.
aThe data show the number (percentage) of mice with each lesion, according to age and sex. PAS-positive granular deposits and neuronal lipofuscin accumulation were assessed on a subset of specimens.
Histochemical and Immunohistochemical Findings in Selected Spontaneous Brain Lesions of C57BL/6J Mice.
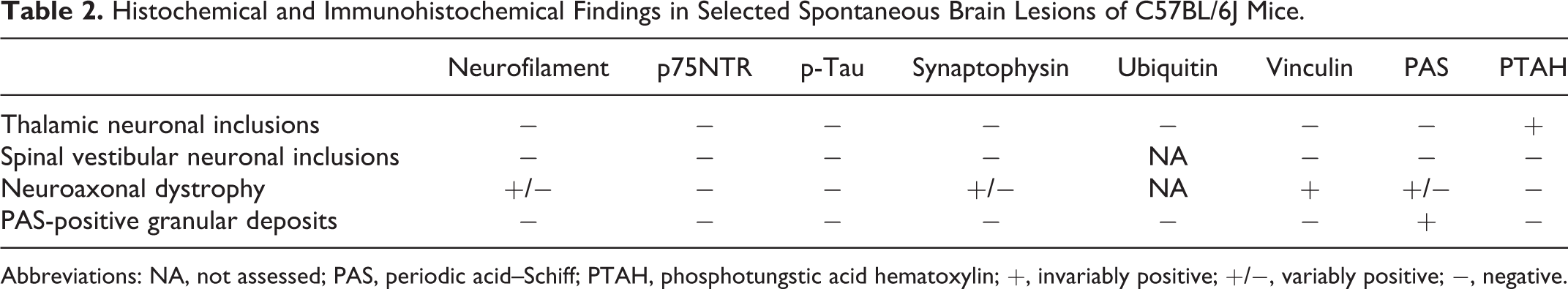
Abbreviations: NA, not assessed; PAS, periodic acid–Schiff; PTAH, phosphotungstic acid hematoxylin; +, invariably positive; +/−, variably positive; −, negative.
Age-Related Degenerative Lesions
Neurons in the thalamus of young adult, adult, and aged C57BL/6J mice frequently contained 2 × 4 to 8 μm eosinophilic, refractile cytoplasmic inclusions that varied in shape from oval to rod-shaped and angular (Fig. 3). These inclusions have been referred to as Hirano-like because of the morphological similarities to Hirano bodies observed in humans. Neurons rarely contained 2 separate Hirano-like inclusions with similar morphologic appearance. These inclusions were found most frequently in the dorsal and medial thalamus, affecting up to 100% of the neurons in these regions. In 2 very old males, the inclusions were also found in neurons of the cornu ammonis and cerebral cortex. The adult and aged groups had a greater prevalence of thalamic inclusions compared to juvenile and young adult groups. The youngest individuals with inclusions were 10 weeks old. There was no difference in prevalence between the sexes. The Hirano-like inclusions were strikingly autofluorescent upon epifluorescent illumination of HE-stained sections with a long bandpass filter cube (Leica I3, Leica Biosystems, Buffalo Grove, IL) (Fig. 4). The inclusions stained dark blue with PTAH (Fig. 5).
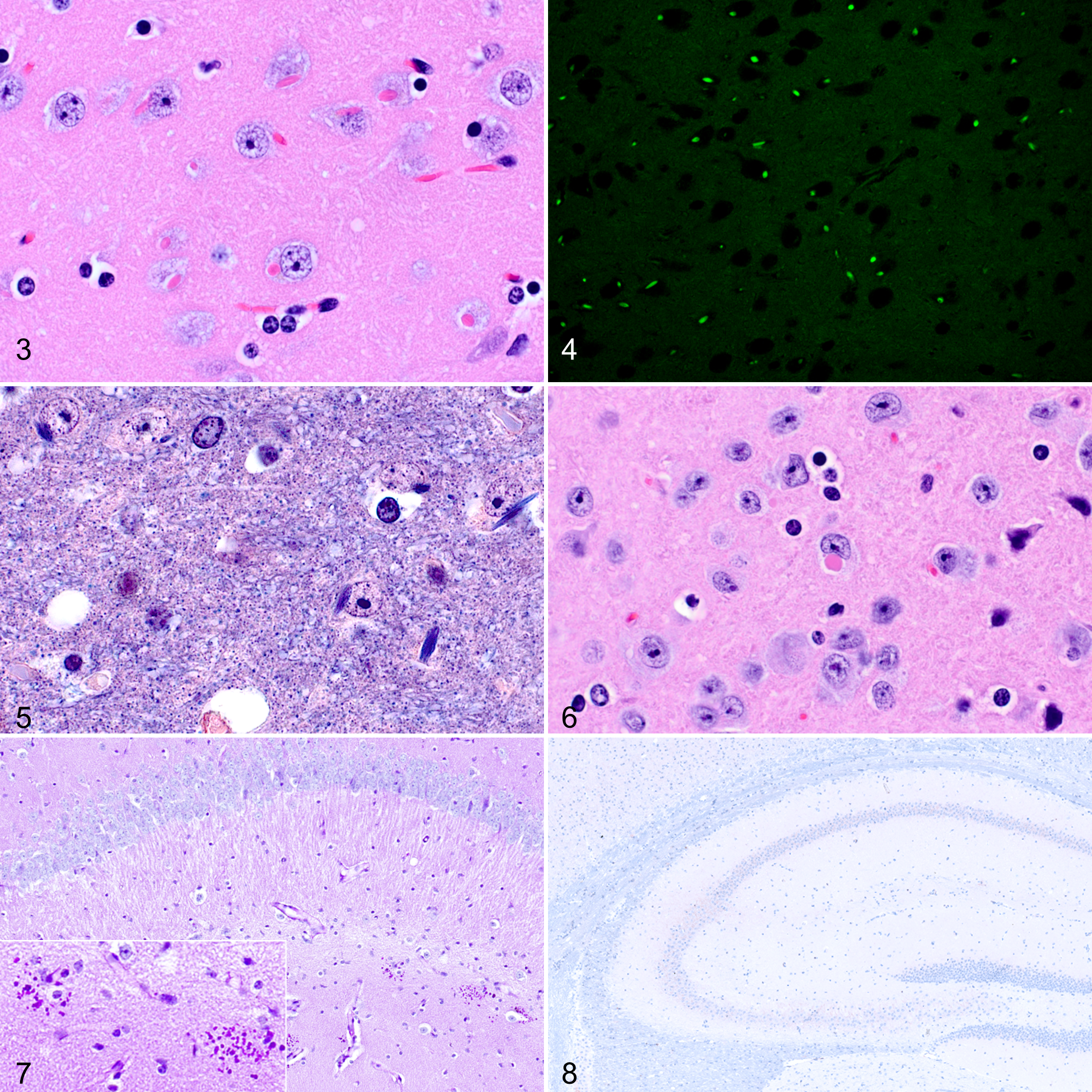
Another type of neuronal inclusion (distinct from the Hirano-like inclusions described in the thalamus) was recognized in the spinal vestibular nucleus. These latter inclusions appeared as 4- to 8-μm roundish amorphous eosinophilic cytoplasmic bodies that were typically separated from the cytoplasm by a narrow, clear halo (Fig. 6). These inclusions were identified only in mice in the adult and aged groups. Typically, only 1 or a few scattered neurons in the nucleus contained inclusions. The youngest individual with inclusions in the spinal vestibular nucleus was 65 weeks. There was no significant difference between the prevalence in adult vs aged groups. The prevalence of these inclusions was not significantly associated with sex. These inclusions were negative with PTAH staining.
Inclusion bodies in both thalamic and spinal vestibular nuclei were consistently negative for PAS reaction and negative for IHC labeling with vinculin, neurofilament, phosphorylated tau (p-Tau), and ubiquitin (Table 2).
PAS-positive granular deposits were characterized by clusters of small (approximately 2–4 μm in diameter) granular structures not associated with neurons or blood vessels that occurred typically in the stratum radiatum of CA1 but also in CA2, the dentate gyrus, and the subiculum (Fig. 7). Deposits ranged from 1 to 5 or more per coronal hemisection and tended to be more numerous in aged animals. These PAS-positive granular deposits were not evident on HE or PTAH staining. PAS-positive granular deposits were infrequently found in the cerebellar and piriform cortex. PAS-positive granular deposits were absent from each of 22 individuals (12 females aged 11 weeks and 10 males aged 17 weeks) examined from the young adult group. PAS-positive granular deposits were present in 6 of 6 females from the adult group, 13 of 13 males from the adult group, 10 of 10 females from the aged group, and 15 of 15 males from the aged group. Immunohistochemical analysis of the granular deposits was invariably negative for neurofilament, ubiquitin, p75 neurotrophin receptor (p75NTR), synaptophysin, vinculin, and p-Tau (Table 2, Fig. 8, and Suppl. Fig. S1).
Neuroaxonal dystrophy was more frequently observed in the cuneate nucleus and gracile nucleus of adult and aged mice. Neuroaxonal dystrophy was characterized by the presence of many swollen axons most conspicuously in the cuneate nucleus (Fig. 9a). The youngest individual with this change was a 16.9-week-old male in the young adult group. Comparison of the prevalence across age groups revealed a statistically significant increase in the frequency of neuroaxonal dystrophy as the mice become older (young adult group, 1/58 [2%]; adult group, 16/51 [31%]; aged group, 37/47 [79%]). The spheroids exhibited diffuse, homogeneous immunoreactivity for vinculin (Fig. 9b) and inconsistent, granular positivity for PAS, neurofilament, and synaptophysin (Fig. 9c–e). No p-Tau immunoreactivity was detected in these structures (Table 2 and Fig. 9f).
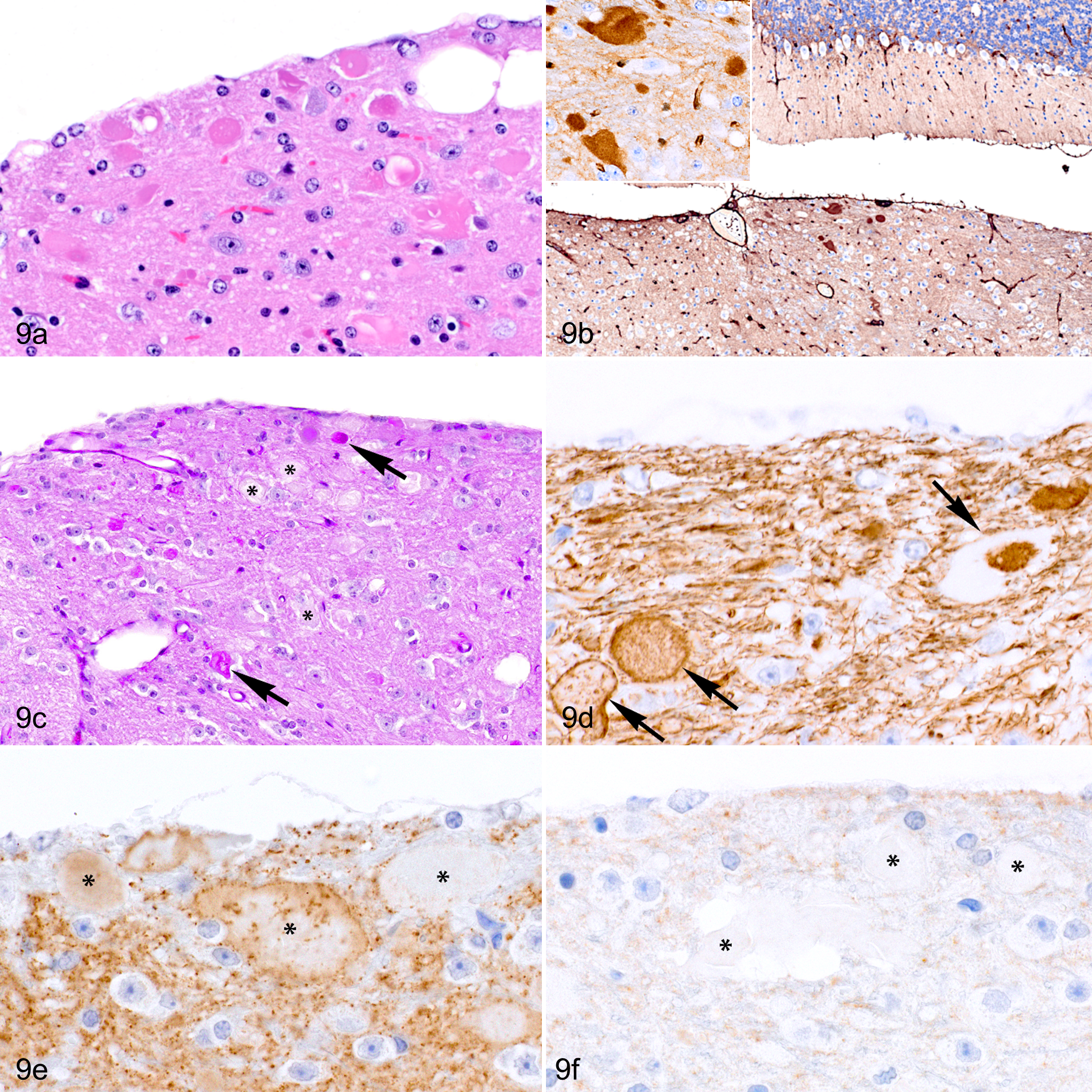
Neuroaxonal dystrophy, cuneate nucleus, 103-week-old female C57BL/6J mouse. (a) Several swollen hypereosinophilic axons (axonal spheroids) in the cuneate nucleus. Hematoxylin and eosin (HE). (b) The swollen axons exhibit strong, diffuse immunoreactivity for vinculin. (c) Some axonal spheroids contain material that is periodic acid–Schiff (PAS) positive (arrows) while others are PAS negative (asterisks). (d, e) Axonal spheroids (arrows and asterisks) have inconsistent and nonhomogeneous immunolabeling for neurofilament (d) and synaptophysin (e) but an absence of immunoreactivity for phosphorylated tau (p-Tau) (f).
Neurons of the deep cerebellar nuclei and cerebellar Purkinje neurons contained punctate light brown, PAS-positive granules in the perikarya interpreted as lipofuscin accumulation. Lipofuscin granules were absent or infrequently observed in the 22 animals examined in the young adult group. Animals in the aged group (n = 25) typically contained denser and more prominent aggregates of lipofuscin granules in the deep cerebellar nuclei and, to a lesser extent, in Purkinje cells (Suppl. Fig. S2–S5). A mild increase in PAS-positive cytoplasmic lipofuscin granules could also be appreciated in pyramidal neurons of the hippocampus and neurons in the cerebral cortex.
Focal mineralization of the thalamus (ie, globoid mineralization), typically associated with arterioles, was observed in 2 females (2/18, 11%) and 5 males (5/29, 17%) in the aged group.
Gliosis
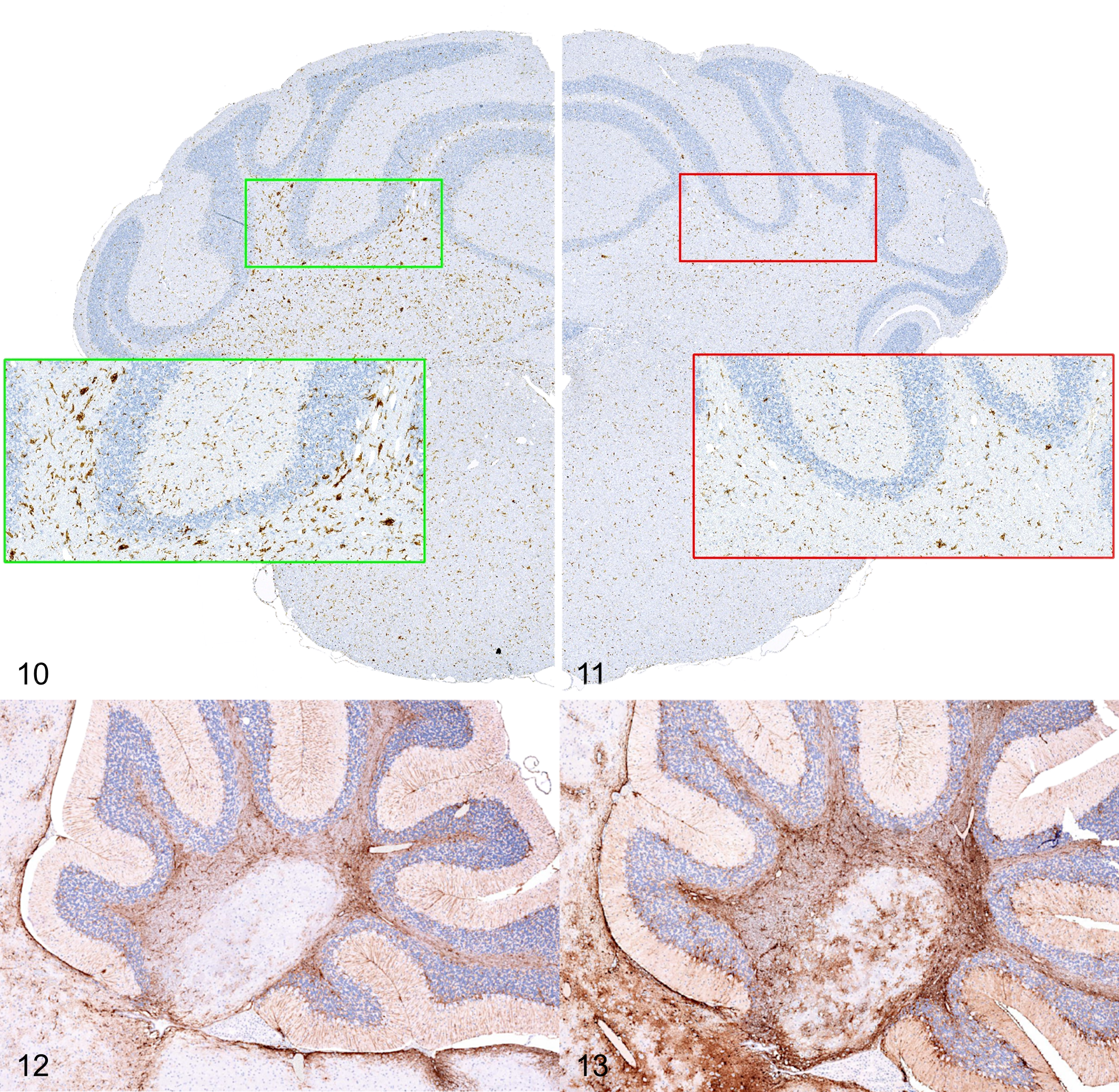
The IHC evaluation of glial markers Iba1 and GFAP was performed on a total of 31 male mice across the young adult (n = 8), adult (n = 10), and aged (n = 13) groups. This analysis revealed that older mice develop a distinct pattern of microglial activation characterized by small multifocal clusters/nodules of hypertrophic (reactive) Iba1-positive cells mainly affecting the white matter of the cerebellum (arbor vitae) and the reticular formation of the midbrain and medulla oblongata (Figs. 10, 11). Other white matter tracts/structures such as corpus callosum and cerebral peduncles were less consistently involved. An increase in GFAP immunoreactivity was also noted in the arbor vitae of older mice (Figs. 12, 13). This finding was evident as an increased density of reactive GFAP-positive astrocytes characterized by expanded cellular processes with prominent branching. To test the statistical significance of these observations, we decided to undertake a quantitative approach on comparable sets of GFAP and Iba1-labeled samples selected from the male young adult and aged groups, respectively. The aged group showed a significant increase in Iba1 staining vs the young adult group in all regions of interest that were compared—cerebellum, cerebellar white matter, medulla, cerebral cortex, hippocampus, thalamus, and hypothalamus. The fold-increase in the percentage of the region with positive pixels varied from 1.6 to 3.3 times. The aged group showed a significant increase in GFAP staining vs young adults in the cerebellar white matter and the cerebral cortex only. The fold-increase in percent positive pixels was 1.9 times in the cerebellar white matter and 1.6 times in the cerebral cortex.
Proliferative Lesions
There was a very low prevalence of proliferative lesions. A pituitary adenoma was found in 1 male, aged 78 weeks (1/203). Metastatic histiocytic sarcoma was found in 1 female, aged 82 weeks (1/203). Emboli of hepatocellular carcinoma cells were in the choroid plexus vasculature in 1 male, aged 77 weeks (1/203).
Miscellaneous Lesions
Focal necrosis in the paramedian lobe of the cerebellum was observed in one 11-week-old male. Acute hemorrhage in the temporoparietal region was observed in two 8-week-old females. Hemosiderosis within the paraflocculus was identified in 1 male aged 77 weeks and 1 female aged 105 weeks. Focal chronic lymphocytic leptomeningeal arteritis was observed in one 39-week-old male. Squamous cysts were identified in one 9-week-old male and one 103-week-old male, and both were in the same location along the ventral midline, ventral to the organum vasculosum of the lamina terminalis. Focal nodules composed of mature adipocytes (lipomatous hamartomas) were found in the interhemispheric leptomeninges of one 10-week-old male and one 84-week-old male (Suppl. Fig. S6). Mild hydrocephalus occurred at a low prevalence in this population of clinically normal mice, occurring in 5 females and 4 males. Agenesis of the corpus callosum was found in 2 females, one 7-week-old and one 10-week old; this was associated with hydrocephalus in the 7-week-old female.
Discussion
This study contributes a comprehensive assessment of brain lesions naturally occurring in experimentally naive and clinically normal C57BL/6J mice over the course of their natural life span. Age-related neurodegenerative and neuroinflammatory changes were the most prominent spontaneous brain lesions observed in our mouse population. On the contrary, neoplastic conditions, developmental abnormalities, and circulatory disorders were rarely observed.
Our analysis identified a series of poorly characterized senile lesions (ie, thalamic and spinal vestibular neuronal inclusions, neuroaxonal dystrophy, microglial nodules) that either have not been reported before or that have been sporadically described but not thoroughly investigated at a population scale. The nature and pathobiological significance of these reported pathological changes are currently unclear.
The development of inclusion bodies in the perikaryon of thalamic neurons was the most conspicuous age-related finding observed in C57BL/6J mice. Similar eosinophilic inclusions of murine thalamic neurons have been previously characterized 17,18,55 and were found to be frequent in aging B6C3F1/N 76 and SAMP8 mice 2 as well as in younger mice with thiamine deficiency or the quaking mutation. 1 Based on histochemical properties (ie, intense staining with eosin and with PTAH) and ultrastructural features, the inclusions appear to result from the progressive agglomeration of cytoskeletal filaments arranged in cross-linked parallel stacks. 1,17,18,76 In our study, these inclusions have been referred to as Hirano-like because of their striking morphological similarities to the Hirano bodies identified in a variety of dementing diseases in humans, including Guamanian parkinsonism-dementia complex, Alzheimer disease, 26,50 Pick disease, and dementia with Lewy bodies, 33 as well as in the course of normal brain aging. 20,25,26,33 Although the exact role of Hirano bodies in aging pathophysiology is unknown, they likely reflect age-related alterations, which are accelerated in various neurodegenerative diseases. 25 Like the inclusions identified in the thalamic neurons of aging C57BL/6J mice, Hirano bodies in humans are refractile eosinophilic rod-like structures that are PTAH positive. 64,65 However, Hirano bodies occur preferentially in the pyramidal neurons of the cornu ammonis (CA) fields of the hippocampus, and their number increases with age. 22,25,33 Interestingly, we observed that Hirano-like inclusions in older C57BL/6J mice are not always limited to the thalamic neurons but also affect other brain regions, including the pyramidal neurons of the CA. Hirano bodies in humans consist of abnormal accumulation of F-actin microfilaments and actin-binding proteins, such as α-actinin and vinculin, arranged in a paracrystalline structure. 22,28,33 Hirano bodies also contain epitopes of tau protein, neurofilament subunits, ubiquitin, and β amyloid precursor protein (β-APP). 23,33,45,53,63 Contrarily, the molecular nature of the Hirano-like inclusions in aging C57BL/6J mice remains unclear. Although vinculin, neurofilament, p-Tau, and ubiquitin are previously identified constituents of Hirano bodies in humans, the Hirano-like inclusions in the thalamus of C57BL/6J mice were consistently negative for these proteins as determined by our IHC investigation.
Unlike the Hirano-like inclusions, we found no previous reports of the inclusions identified in the spinal or descending vestibular nucleus, nor is there a specific analogue for this finding in other species. On a morphologic and speculative basis, we suggest that this finding represents the disorderly accumulation of cytoplasmic proteins with an unclear neuropathologic significance, similar to age-related neuronal inclusions in other species, including Hirano bodies. 26,49,57
Our study confirms that brain aging in C57BL/6J mice is associated with deposition of PAS-positive granules in the hippocampus and, to a lesser degree, cerebellum and piriform cortex. This evidence is in line with previous studies indicating a clear genetic predisposition of the C57BL/6J strain for this phenotype, which includes the identification of 2 major quantitative trait loci (QTLs) intervals on chromosomes 7 and 10, respectively. 44,47 A distinct strain-related proclivity for the development of PAS-positive granular deposits also exists in other C57BL/6 substrains and inbred lines, including AKR, SAMP8, and SJL/J mice. On the contrary, strains such as BALB/cJ, CBA/J, DBA/2J, and A/J mice do not manifest the phenotype. 38 –40,47,51 The pathobiological nature of the PAS-positive granular deposits is still largely uncharacterized. Several lines of evidence indicate that age-dependent hippocampal granules may be the result of oxidative stress. 44,47,58,71 Previous IHC studies have reported that granules react with antibodies targeting diverse molecular constituents, including hyperphosphorylated tau, amyloid β, ubiquitin, and p75NTR. 44,69 However, caution must be exercised when interpreting the signal from antibody staining as immunoglobulin M is a common contaminant of monoclonal ascites and polyclonal sera and can recognize a glycosidic neoepitope present in the granular structures, leading to false-positive results. 46,48 Further suggesting the unspecific nature of the immunoreactivity described in previous works, our study failed to demonstrate any labeling of granular deposits using highly purified monoclonal antibodies against ubiquitin, p75NTR, and p-Tau.
Neuroaxonal dystrophy mainly affecting the relay nuclei of the medulla oblongata was frequently observed in the brain of aging C57BL/6J mice. Neuroaxonal dystrophy is a neurodegenerative disorder primarily affecting the central nervous system (CNS) that is characterized by discrete swellings (spheroids) in the distal segment of axons. Impairment of the bidirectional transport machinery of axons plays a major role in the pathogenesis of neuroaxonal dystrophy. In this context, spheroids usually exhibit abnormal accumulation of molecular components that are typically involved in the maintenance of axonal integrity and transport mechanisms, including structural constituents, motor proteins, and cargoes (eg, neurofilaments, tau, ubiquitin, synaptophysin, kinesin, dynein). 8,16,54,73 Primary inherited forms of neuroaxonal dystrophy have been reported in humans as well as in mice, dogs, cats, horses, and sheep. 3,5,8,12,16,21,68,73 Senile neuroaxonal dystrophy also occurs as part of the aging process and is a common incidental finding in humans and domestic and laboratory animals.* The spheroids developed by aged C57BL/6J mice exhibit distinct characteristics. First, the lesions in aged C57BL/6J mice include accumulation of neurofilament and synaptophysin that are not typically described in spheroids from spontaneous mouse models of inherited neuroaxonal dystrophy or geriatric dogs. 7,8 Second, the spheroids in aged C57BL/6J mice are PAS positive, but this is infrequently reported in other spheroids, although it has been previously documented in familial neuroaxonal dystrophy in cats. 12 Perhaps the most striking attribute of senile spheroids in C57BL/6J mice was the intense and diffuse immunoreactivity for vinculin. As a critical component of focal adhesions, vinculin plays a pivotal role in several axonal functions, including regeneration and growth cone guidance/extension. 66,74 Interestingly, increased immunoreactivity for vinculin has been reported in the distal stump of neuromas resulting from the experimental transection of the rat sciatic nerve. 74 However, axonal accumulation of vinculin has not been previously associated with neuroaxonal dystrophy or spheroid formation.
Neuronal lipofuscin accumulation was mild, most obvious in deep cerebellar nuclei, and more clearly appreciated with PAS reaction than with HE stain. Neuronal degeneration was not observed in association with lipofuscin accumulation, and this is compatible with physiologic, age-related lipofuscin accumulation. 29,41
Quantitative increase in microglial and astrocytic activation has been consistently demonstrated as an aging finding in rodents, including C57BL/6J mice. 11,43,52,56 The senile activation and expansion of glia in the mouse brain closely recapitulates the features of progressive neuroinflammation observed in other aging animals, including humans. 6,13 Neuroinflammation is considered an important determinant for the development of several neurodegenerative conditions, including Alzheimer and Parkinson diseases. 32,77 Since the C57BL/6J mouse is often the preferred genetic background to model these human disorders, it is critical to understand how spontaneous neuroinflammation develops in this mouse strain to provide a correct interpretation of the translation potential of similar findings in the experimental context. In general, the progression of age-related gliosis in mice is more severe in females, and the white matter appears to be more affected than the gray matter. 11,43,52,56 Our study similarly demonstrated gliosis (especially microgliosis) in the brain of aged male mice compared to young adult male mice. Distinct clusters/nodules of activated microglia preferentially affecting the white matter were also observed.
In conclusion, the accurate description of these findings and their association with age and sex is critical to better determine the spectrum and frequency of strain-related background changes and to discriminate them from the effect of specific experimental interventions. These data are informative and relevant to the preclinical research settings that make use of mutant mice on a C57BL/6J background to study aging and neurodegenerative conditions.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819859878 - Spontaneous Incidental Brain Lesions in C57BL/6J Mice
Supplemental Material, DS1_VET_10.1177_0300985819859878 for Spontaneous Incidental Brain Lesions in C57BL/6J Mice by James C. Tarrant, Patrick Savickas, Lorna Omodho, Marco Spinazzi and Enrico Radaelli in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
