Abstract
The focus of immunohistochemistry as applied to nervous system tumors is in identifying the neoplasm present and evaluating margins between normal and neoplastic tissue. Although not always utilized by specialists in neuropathology, immunohistochemistry remains useful to resolve concerns about the differentiation and rate of tumor growth. The aims of this review are to discuss the utility of immunohistochemical reagents currently used in diagnosis of canine and feline intracalvarial tumors, to indicate the applicability of some tests currently used in human nervous system tumors for domestic species, and to evaluate a few less commonly used reagents. A panel of biomarkers is usually needed to confirm a diagnosis, with groups of reagents for leptomeningeal, intraparenchymal, and ventricular neoplasms. In the future, signature genetic alterations found among feline and canine brain tumors—as correlated prospectively with diagnosis, rate of enlargement, or response to treatment—may result in new immunohistochemical reagents to simplify the task of diagnosis. Prospective studies determining the type and proportion of stem cell marker expression on patient longevity are likely to be fruitful and suggest new therapies. Due to increased frequency of biopsy or partial resection of tumors from the living patient, biomarkers are needed to serve as accurate prognostic indicators and assist in determining the efficacy of developing therapeutic options in nervous system tumors of dogs and cats.
Immunohistochemistry has been used in the diagnosis of brain tumors for the past 3 decades, 135 with the goals of improving diagnostic accuracy and predicting behavior. In companion animals as well as other species, many patients still present with large inoperable tumors for which only palliative therapy can be offered. Thus, emphasis has largely been on identifying the type of tumor and correlating that diagnosis with effects of tumor presence on brain architecture, clinical course, and signs. The earliest studies employing surgery led to disappointing results, with modest to no increase in median survival time compared to untreated patients. 40 This situation is changing, as cross-sectional imaging (magnetic resonance imaging) increasingly allows veterinary neurosurgeons to resect or biopsy neoplasms located in and around the brain. 66
Methods currently used for biopsy or excision of brain tumors include stereotactic procedures, 65,86 freehand biopsy with ultrasound guidance, 37,61 endoscopic biopsy, 60 and aspiration. 35 The overall diagnostic yield for stereotactic excisional biopsies in necropsy-confirmed cases can be as high as 95%, 65,86 while methods utilizing freehand techniques have had a yield as low as 25%. 37 For example, 65 21 of 22 brain tumor biopsies removed by stereotactic surgeries were successfully diagnosed by histopathology. Prolonged survival—which, for meningiomas, is on the order of several years 34,59 —has increased the emphasis on predicting aggressiveness and the probability of recurrence from excisional biopsies. Although surgical removal of meningioma increases the survival, there is a high incidence of recurrence, usually at the site of the original mass, which results in eventual euthanasia. 30,34
Successful characterization of brain tumors in biopsies from dogs and cats has an increasing role in diagnosis and in guiding adjunctive treatments. 88 If masses are surgically inaccessible, biopsies of unresectable lesions can be performed. The advantage of necropsy specimens of brain tumors is that the pathologist has the opportunity to examine large areas of the mass and the margins between neoplastic and normal tissue. Examination of excised tumor fragments or small biopsies places emphasis on defining tumor margins and making a diagnosis from small pieces of potentially distorted tissue.
A confounding factor in understanding nervous system tumor grading relates to the classification system used. At present, the veterinary World Health Organization classification 63 provides a relatively unembellished system of classifying brain tumors that has been followed in older texts. However, many veterinary neuropathologists now feel that an enhanced system of tumor grading is needed, and they prefer the classification used for human brain tumors. 73 There is, however, a lack of prospective evidence for extrapolating the human grading system to other species.
An important consideration regarding use of immunohistochemical diagnosis, particularly in biopsies, is a heterogeneity of maturation that may arise from neoplastic stem cells. Stains for stem cells—such as nestin, beta III tubulin, CD34, and CD133—can identify some of these cell populations but are of limited assistance in determining the differentiation of the tumor. However, stem cell profiles may be helpful in indicating effective treatments because stem cells are thought resistant to conventional therapy and, in humans, the percentage of these cells correlates with recurrence and poor prognosis. Moreover, the presence of clones of varied differentiation arising from stem cells explains why irregular staining is so common in brain tumors when differentiation biomarkers are used. A number of investigators have utilized human markers to confirm the presence of stem cells in canine brain tumors. 16,98,100,119
Before immunohistochemistry, cytochemical stains were developed with the objective of differentiating mature elements of the nervous system from one another, and they are not used extensively in tumor diagnosis at the present time. Silver and gold stains developed to mark normal cells of the neuropil have a reputation of unpredictability. 41 More frequently used in human neuropathology is the reticulin stain to identify basal lamina in meningiomas and nerve sheath tumors 14,58 and stains that detect argyrophilic nucleolar organizing regions, which, though cumbersome to quantitate, can be used to separate more from less aggressive parenchymal and meningeal tumors. 102 PTAH (phosphotungstic acid–hematoxlyn) has been used to stain the basal bodies of cilia in ependymomas. More sophisticated stains were needed to detect various immature and mature tumor cells, and immunohistochemistry provided these more specific biomarkers.
Brain tumors occur with a reported frequency of 14.5 per 100 000 dogs and 2.2 to 3.5 per 100 000 cats 17 and have been recorded in 1% to 3% of all canine necropsies. 121 It has been estimated that 80% to 90% of brain tumor diagnoses in domestic animals apply to dogs and cats. 65 In contrast, a summary of reported central nervous system (CNS) tumors in other species is fragmentary and probably inaccurate. 38,54,91
Materials and Methods
For purposes of illustrating the staining demonstrated herein, records of the University of Missouri Veterinary Medical Diagnostic Laboratory database were searched by diagnosis. Available paraffin blocks were retrieved; hematoxlyn and eosin (HE) slides were reviewed to confirm the presence of neoplastic tissue and the diagnosis; and 5-μm-thick sections were prepared from selected cases, rehydrated, and stained by immunoperoxidase methods indicated in Table 1. 3,3′-Diaminobenzidine (DAB) was used as the chromogen and Meyer’s hematoxlyn as counterstain.
Antibodies and Immunohistochemistry Procedures.a

P, rabbit polyclonal; M, mouse monoclonal; Olig2, oligodendrocytes transcription factor 2; GFAP, glial fibrillary acidic protein; CNPase, 2′,3′-cyclic nucleotide 3′phosphodiesterase.
aEnvision was the detection system for each.
Review of the Literature
Metastatic Neoplasms
Many systemic tumors readily metastasize to the brain and can confound the diagnosis of primary tumors when a systemic neoplasm is not suspected. Metastatic carcinoma cells that lack cohesiveness can be troublesome in this regard (Fig. 1). Concurrent primary and secondary brain tumors need to be distinguished from multiple primary tumors, which have been reported in a dog. 5 When of embolic origin, florid metastases are multiple, spherical, and usually located near the gray-white junction, forming expansile masses that displace tissue. Of 177 secondary canine brain tumors, 29% were hemangiosarcomas, 25% were pituitary tumors invading the brain by extension, 12% were lymphomas, and 12% were metastatic carcinomas. 116 Of 160 brain tumors in cats, metastatic brain tumors, including lymphoma, accounted for 16.9% of the total. 132 Metastatic pulmonary carcinoma was the most common embolic mass lesion. (A few of the more commonly used reagents for identifying metastatic brain neoplasms are listed in later in the article.)
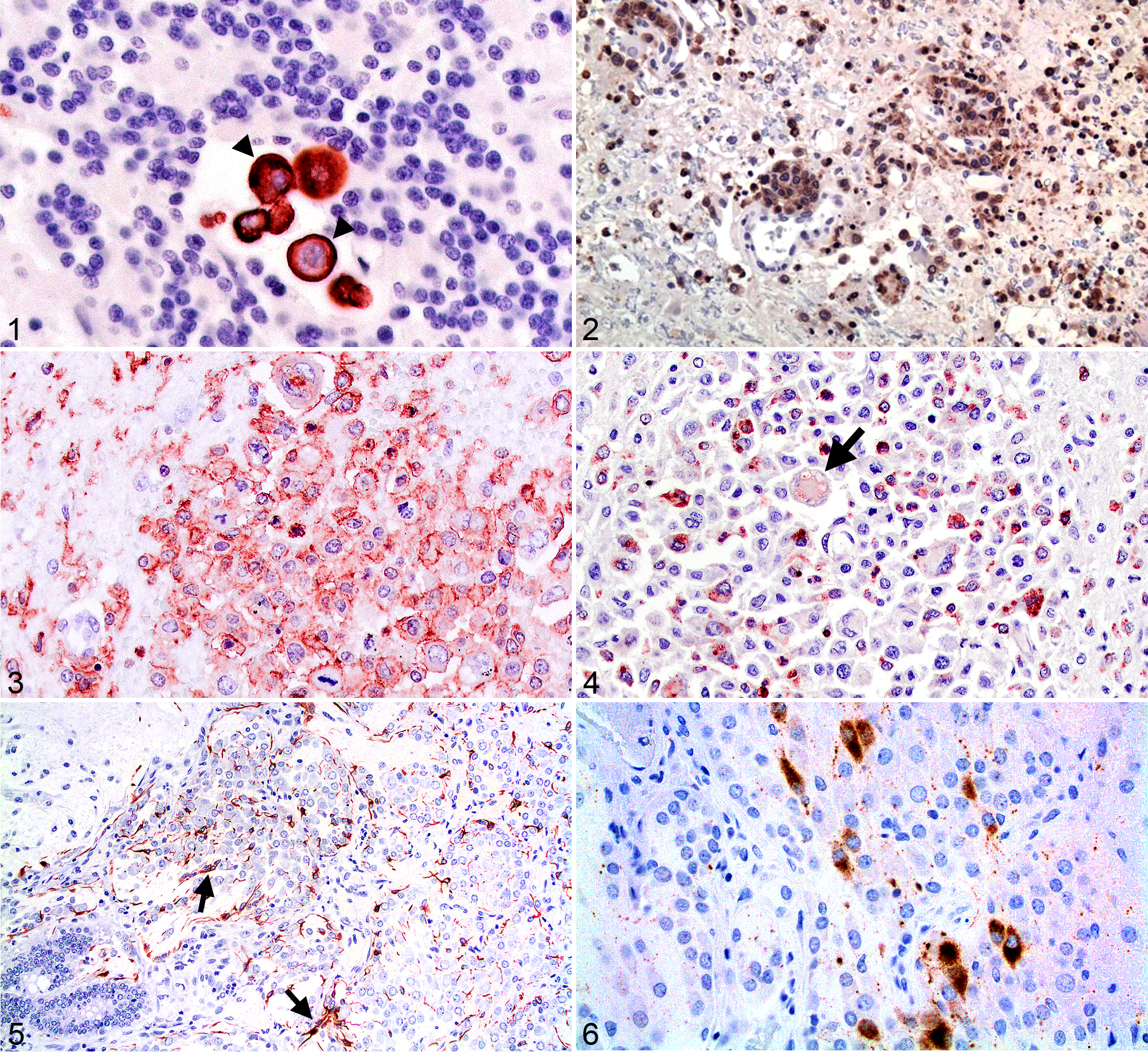
It is beyond the scope of this article to present all the possibilities for immunohistochemical techniques used to determine the origin of metastatic brain lesions. However, pathologists should keep an open mind when dealing with biopsies of potential secondary masses.
Image-guided resection of metastatic lesions increases the symptom-free interval and improves the quality of life in humans so that correct identification is crucial. 126 Eighty percent of metastases in humans affect the cerebrum, a surgically accessible site. 69 Immunohistochemistry has potential use in biopsy or excision of these lesions, determining whether the mass is metastatic or a primary neoplasm. Human neurosurgeons are occasionally faced with clinically significant metastatic neoplasms that arise from an unknown external source, and in about half of these cases, the primary tumor still has not been identified at the time of the patient’s death. 99
Metastatic tumors most often affect the canine spinal cord indirectly by infiltration of the epidural space and by instability or fracture of vertebrae than by intra-axial growth. 143 Carcinomas arising in the nasal mucosa and paranasal sinuses enter the CNS by following the olfactory nerves across the ethmoid turbinates, and meningiomas may enter via the optic foramens.
Occurrence of Intracalvarial Tumors
Of primary intracalvarial tumors, meningiomas and nerve sheath tumors can be multiple. 84,127 The results of 2 studies with a combined total of 211 cases 65,117 identified meningiomas as the most common intracalvarial neoplasm in dogs (47.4%). Astrocytomas were the most common primary intra-axial neoplasms, followed by oligodendrogliomas, choroid plexus tumors, and lymphosarcomas (17.1%, 12.8%, and 3.3%, respectively). A case report detected 2 different concurrent neoplasms in a dog. 117 In cats, combining data from 3 studies (total 262 animals), 131,132,145 meningioma was also the most prevalent intracalvarial tumor, followed by lymphoma, then other primary parenchymal tumors (61.5%, 26.7%, and 11.8% of the total, respectively). Two studies mentioned a total of 21 cats that had another primary tumor in addition to meningioima. 131,132
Metastases of brain tumors to sites outside the CNS is infrequent but may become more likely as postsurgical survival times increase. Meningioma is the neoplasm most frequently involved in extracranial metastasis. 108 A recently reported meningioma penetrated through the calvarium of a cat without surgical intervention. 56 Local implants of several intra-axial tumors may also produce metastases along ventricles or the subarachnoid space.
Round Cell Tumors
Nervous system lymphoma may be a multicentric growth or solely of brain origin. Lymphoma has been described as the third-most common metastatic brain tumor in dogs, comprising 12% of the total in one accounting. 116 During a similar period, the database contained 7 primary brain lymphomas, an indication that primary tumors constitute roughly a quarter of the total. 117 In another study, primary lymphoma accounted for 1 of 8 cases involving the brain. 18 Lymphoma is considered to be the most common secondary brain tumor in cats, accounting for 39 of 76 (51.3%) of all secondary tumors recorded. However, primary CNS lymphoma represented 35% of all lymphomas and 37% in a second study. 130,131 All feline brain lymphomas were secondary in an earlier study. 145
Both B- and T-cell lymphomas occur in the brains of dogs and cats, including angiocentric tumors. Commonly used immunohistochemical techniques are usually effective in distinguishing B- and T-cell lymphomas, similar to those at other sites in the body. Using formalin-fixed, paraffin-embedded (FFPE) tissue, most laboratories have available CD3 versus CD79a/CD20/immunoglobulin testing to characterize T- and B-cell origin tumors, respectively. 20,71,89 Staining should be negative with dedicated histiocytic markers such as Iba1. Other markers used in describing CNS lymphomas are CD45 and CD34 in dogs and cats. 46,138,140 CD45 is a common leukocyte antigen and therefore considered a general leukocytic marker. 140 CD34 is a transmembrane glycoprotein present on immature B lymphocytes that is lost later during development; it is not expressed after normal cells leave the marrow and has been used to infer the leukemic origin of brain lymphomas in animals. 138 Especially in the case of angioblastic lymphomas, leukocytes that are responding to tissue damage of vasogenic origin may make it difficult to distinguish neoplastic versus inflammatory processes until individual cell morphology is taken into account (Fig. 2).
Histiocytic sarcomas are increasingly identified in the brain, either as mass lesions or diffuse leptomeningeal infiltrates. A recent case series of canine histiocytic sarcomas by Ide et al 47 documented the pleomorphic appearance of constituent cells and found that a few normal lymphocytes are also commonly present. Tumors consisted of large, pleomorphic, vacuolated uni- and multinucleated cells, closely resembling gitter cells. Phagocytosis of lipid by these tumors can be documented by oil red O staining. Most cases have been described in dogs, but at least 1 feline case had similar morphologic characteristics and was confirmed by positive α-1-chymotrypsin labeling. 50 Like all sarcomas, histiocytic sarcomas are vimentin positive, but tumor cells should be negative for S100, glial fibrillary acidic protein (GFAP), cytokeratin, neurofilament proteins, and lymphocytic markers. 146
Histiocytic sarcomas comprised 8 of 177 neoplasms in the brains of dogs and were approximately half as common as lymphomas. 117 In an earlier work, Affolter and Moore found 1 localized histiocytic sarcoma and 1 metastatic in the brain in 20 cases, 2 about a 5% prevalence overall as a primary brain neoplasm. Based on FFPE tissues, CD45A+, CD18+, CD3–, and CD79a– constituted the most useful biomarker panel for diagnosis. 55 CD18 is relatively specific in dogs for cells of the monocyte–macrophage lineage (Fig. 3). 80 Occasionally, dim CD18-positive flow cytometric staining has been reported in feline lymphomas. 29 Lysozyme and RCA-1 lectin are additional markers successfully used in FFPE tissue. 128,133 Ultrastructurally, numerous lysosomes are present, and at least some tumor cells possess multifocal cytoplasmic staining by lysozyme immunohistochemistry (Fig. 4). 13,128
Histiocytic sarcomas can be grouped into those positive by Iba1 and canine HLA-DR reagents and those positive with CD204 and CD163. These 2 groups are thought to correspond with M1 and M2 macrophage populations, respectively, and tumors may be of either lineage. 47 M2 markers are thought consistent with microglial origin. Frozen sections, less commonly available for routine diagnosis, were additionally positive for MCHII, Cd1a, CD11c, and ICAM-1 expression. 47 Differentiation from granulomatous meningoencephalitis relies on the more abundant lymphocytes in granulomatous meningoencephalitis and more consistent perivascular orientation. 122 Variably sized tumor cells, mitoses, and extensive phagocytic activity are other helpful phenotypic characteristics of histiocytic sarcoma.
The following review of immunohistochemistry of canine and feline nervous system parenchymal tumors is based on the veterinary World Health Organization classification. 63
Neural Tumors
Neoplasms of neuronal origin are rare, and a useful description of the morphologic characteristics of this group has recently been published. 136 Included herein are primitive neuroectodermal (embryonic) tumors and those of more differentiated phenotype. 64,136 Neuroblastomas and gangliocytoma/ganglioglioblastomas may occur within or external to the CNS; peripheral tumors presumably arise from autonomic or enteric neural structures.
Less differentiated neuronal tumors have been grouped into primitive neuroepithelial tumors (PNET), among them neuroblastoma, medulloblastoma, neuresthesioblastoma, and medulloepithelioma of the eye. All consist of poorly differentiated neuroblasts that grow as cell sheets and form neuroblastic (Homer-Wright) rosettes. Tumor cells can express neural and/or glial antigens, and finding a combination of positive immunohistochemical staining, combined with characteristic HE morphology, is used in diagnosis. 118,137
Neuron-specific enolase and neurofilament expression are the standard immunohistochemical markers for the group. 64 Most human neoplasms stain with synaptophysin, 85 and canine medulloblastomas have been reported that stain with this marker as well. 79,136 In humans, medulloblastomas often have mutations found on chromosome 17 or at other sites that are not present in other PNETs, 82,96 so there is sentiment in the human literature for separating them from other tumors in the PNET group. 85 In fact, human medulloblastoma now has 4 prognostic categories based solely on the tumor’s genetic alterations, but there are no immunohistochemical tests to detect these abnormalities.
Canine PNETs may stain irregularly. 32 These tumors should contain cells with neural differentiation, but the population may have a mixed pattern, with areas of GFAP-positive glial differentiation, vimentin, or cytokeratin expression. 39 One canine tumor diagnosed as a PNET was positive for vimentin, neuron-specific enolase (NSE), and S100 but negative for synaptophysin. 16 Others suggest that diffuse cytoplasmic synaptophysin, NSE, and possibly c-Kit are the most useful markers in a subpopulation of canine medulloblastomas. 79 Still others indicate that neuron-specific nuclear protein (NeuN) and MAP 2 early-lineage protein markers are more helpful, although they are not specific for PNETs. 9 In a series of canine and feline olfactory PNETs, NeuN—a neuron-specific DNA-binding protein—labeled both nuclei and cytoplasms of cells in most tumors. 9 Additional stem cell markers include nestin, beta III tubulin, and doublecortin, in line with the primitive nature of these tumors. 49 Doublecortin is a regulator of cytoskeletal organization and is involved in neuronal and glial migration; 24 mutations in this gene may result in developmental anomalies such as polymicrogyria. Nestin is an intermediate filament of immature neuronal cells active during development, but it does not separate neuronal and glial tumors in people. 66 Neurofilament protein M, chromogranin A, and tyrosine hydroxylase are other markers used to characterize PNETs. 81,123
Neurocytomas are primitive neoplasms arising near the ventricles. They comprise small basophilic cells, morphologically similar in many respects to PNETs and oligodendrogliomas. 110 They are thought to arise in humans from expansion of residual adult neuronal progenitors in situ. Cells derived from human neurocytomas can be persuaded to form neurospheres of glial differentiation in vitro under appropriate conditions, suggesting a stem cell component. 114 Neurocytomas are extremely rare in animals: 3 canine cases have been reported, and they stained with neuronal markers synaptophysin, NeuN, and NSE but did not express neurofilaments, chromogranin A, or S100. 104 In dogs and people, some neurocytomas contain GFAP positive processes, but it is not clear whether they are processes of normal astrocytes trapped between tumor cells or those of a subpopulation of neoplastic cells. 45,110
Gangliocytomas, neoplasms of more differentiated neurons, often occur outside the CNS and consist of cells that more closely resemble differentiated neurons. 63 The presence of binucleated tumor cells of neuronal character is considered a helpful diagnostic pattern in humans. 85 They may contain reactive glial cells, but the neuronal population is considered to be neoplastic. If the neurons are intermingled with neoplastic glial cells, positive with GFAP, then the mass is termed ganglioglioma (Fig. 5). Patches of fibrillar neuropil may separate recognizable neurons. Intratumoral neurons can be positive for expression of neurotransmitter related molecules such as tyrosine hydroxylase and can express other neurotransmitters or calcium-binding molecules relevant to specific types of neuronal differentiation (Fig. 6). 96
Meningiomas
Meningiomas, as previously noted, are the most common extra-axial intracalvarial or intravertebral neoplasms. 86,95 Small tumors found at postmortem may be clinically insignificant. Solitary tumors and multiple masses can be found in cats (up to 17%) and dogs. 31,83,132 Based on the current World Health Organization veterinary histological classification system, 63 canine meningiomas are categorized into 2 major groups: (1) benign slow-growing (meningothelial, fibroblastic, transitional, psammomatous, angiomatous, papillary, granular, myxoid) and (2) faster-growing anaplastic types. Because of their external location, meningiomas are commonly biopsied or excised. 31,34,59,88 Nasal meningiomas of dogs are thought to be more aggressive and invade into the cerebrum through the cribriform plate. 94 Surgery and radiation therapy have led to success in prolonging life in dogs with frontal lobe meningiomas, which previously had a poor prognosis. 134 Meningiomas metastasize outside the cranium very rarely, but extracranial metastases or extensions through the calvarium are possible, especially of anaplastic tumors. 56,67,108,113
Vimentin is the classical immunohistochemical marker for detecting all subtypes of meningiomas, even anaplastic tumors, and most cases express S100 (Fig. 7). 64,74,117 Some but not all cases express NSE. 6,87 Laminin is deposited between cells in many cases but can be irregular or haphazard in others (Fig. 8). Laminin may not differentiate meningiomas from other spindle cell neoplasms such as nerve sheath tumors. 36 Diffuse meningeal sarcomatosis can usually be identified by the bizarre appearance of tumor cells without using immunohistochemistry. 63 Suspect sarcomatosis cases should be stained to rule out histiocytic sarcoma.
A majority of meningiomas in dogs and cats possess progesterone receptors but lack estrogen receptors. 1 Progesterone receptor expression in particular has been shown to be inversely related to proliferation labeling indices and is associated with positive response to radiotherapy. 77,127 In addition, 6 of 30 meningiomas in one study and 11 of 15 in another were pancytokeratin positive. 6,100 However, pancytokeratin stained relatively few cells, without apparent regard to benignancy or malignancy. 100 When the labeling percentage is low, pancytokeratin could be less useful in identifying small biopsies as meningiomas. A few tumors express GFAP, but only a small number of cells may be positive. 6 Epithelial membrane antigen (EMA) has been found very useful in distinguishing human meningiomas from other spindle cell tumors, 85 but no commercial reagent reacting with FFPE is apparently available. 100 In people, EMA-positive meningiomas tend to be benign, while more malignant tumors are likely to be positive for pancytokeratin. Analogous markers would be ideal in prognosticating animal meningiomas from biopsies, as an adjunct to determining mitotic or Ki-67 labeling indices, the latter being the best indicator of malignancy available at present. Neither cyclooxygenase-2 nor matrix metalloproteinase staining predict the clinical course of animal meningiomas. 76,105 A high percentage of animal meningiomas express CD34. 100 This marker has been used to distinguish human meningiomas from nerve sheath tumors, but no canine nerve sheath tumors or meningeal sarcomas have been tested.
Additional new potential markers have been described for canine and feline meningiomas. Cells of the arachnoid have desmosomes, and some authors report that the use of desmoplakin is useful in identifying human meningiomas. 3 E-cadherin and claudin-1 are markers that detect junctional complexes found in normal leptomeninges. E-cadherin identified a high percentage of canine meningiomas in one study and may prove useful in the diagnosis of canine and feline tumors. 100 E-cadherin forms cytoplasmic complexes with catenin, which binds to actin, but its presence or absence does not seem correlated with tumor grade. 23 E-cadherin does not distinguish between human schwannomas and meningiomas, being expressed in both. Choroid plexus tumors and some human metastatic carcinomas may also be positive. Ide et al 49 examined the differential immunohistochemical labeling of invasive meningiomas and discovered that invasive tumors expressed N-cadherin instead of the E-cadherin seen in benign tumors. Nuclear expression of β-catenin and doublecortin were not specific but were contributory to diagnosis. Ki-67 index was also increased in invasive tumors. Doublecortin activity was described as useful in this study and expressed at the periphery of invasive meningiomas, decorating the tumor margins. Doublecortin, not expressed in normal meninges, might with further study become a useful tumor boundary marker. 49
The expression of vascular endothelial growth factor isoforms has been associated with peritumoral edema in a variety of canine brain tumors, but expression in meningiomas is low compared to that observed in high-grade parenchymal tumors. 22 Those meningiomas possessing high levels of vascular endothelial growth factor expression had shorter survival, which may be related to increased tumor vascularity. 97
Granular cell meningioma has been considered an uncommon variant of meningioma but is probably a completely unrelated tumor. Its staining properties are variable in dogs 43,70 and a cat. 78 Granular cell tumors are also occasionally found intra-axially. 70 All investigators agree that granular cell tumors are not of leukocytic origin. 43 These neoplasms are variably positive for expression of vimentin and stain with S100 but are negative for expression of GFAP and leukocyte antigens. 109,125
Peripheral Nerve Sheath Tumors
An important differential for meningioma is intracranial peripheral nerve sheath tumor (PNST), which arises from Schwann cells, perineural fibroblasts, or a mixture. These neoplasms may be either subdivided into schwannomas, neurofibromas, and neurofibrosarcomas or as grouped PNST. PNSTs are encountered in the CNS as they extend from the periphery through nerve foramens, or they can arise intrathecally, presenting as extra-axial masses that look like meningiomas (Fig. 9). 112 These neoplasms surround ganglionic neurons in their path. Some authors indicate that malignancy is determined by mitotic index and degree of anaplasia. 64 S100, laminin, and claudin-1 staining (Figs. 10, 11) is useful in separating these tumors from metastatic sarcomas and fibrosarcoma. 14,53 Laminin staining is usually more extensive and linear in PNSTs compared to meningiomas. Application of some of the newer meningioma markers may be useful in further separating the two.
Intra-axial Masses
More deeply buried parenchymal neoplasms are increasingly being biopsied with or without thought of extirpation, before attempting other therapies. These masses are considered to be astrocytomas or oligodendrogliomas for the most part, although lack of a consistently positive diagnostic marker for oligodendrogliomas might have resulted in low diagnoses of mixed gliomas, which could behave differently than neoplasms of a pure cell lineage.
Astrocytic Tumors
Astrocytomas have historically been the most prevalent intra-axial primary tumor, at least in North America. Well differentiated astrocytomas are firm, gray-tan masses, often possessing a vague boundary with adjacent brain. Astrocytomas distort brain architecture by infiltration, and neoplastic cells tend to elaborate at least a few cytoplasmic processes. Advancing tumor borders can be microscopically vague, and neurons are found surrounded by neoplastic cells. Mitotic index and Ki-67 labeling index as methods to identify mitoses and cell proliferation, respectively, are helpful in determining the rate of tumor expansion. 136
One important question in evaluating biopsies from astrocytomas involves determining the margins of infiltrative growth. Neoplastic growth is often bordered by enlarged, reactive astrocytes having a similar HE morphology to tumor cells. In terms of pattern recognition, reactive astrocyte nuclei tend not to crowd one another, while human astrocytoma tumor cells transgress on one another’s territories. 85 The intermediate filament common to astrocytomas is GFAP, as is true of normal astrocytes, and the positive staining of neoplastic cells is a long-accepted means of diagnosis. 60 Staining may be variably intense. Vimentin is also usually positive. 19 Five of 7 morphologically diagnosed feline astrocytomas also had positive GFAP cytoplasmic staining, 4,26,106 but NSE positivity was documented only in 1 of 5 studies 26 and S100 in 2. Positive p53 staining was recorded in 1 case. 4 Prognosis most often relies on mitotic or Ki-67 labeling indices. Larger case series are needed to determine whether there are variations in staining phenotypes, particularly in cats, which less commonly develop astrocytomas than do dogs.
Several tumor markers have been applied to human astrocytomas to solve the diagnostic issues related to infiltrative margins. To date, the most widely used marker for this purpose in humans has been p53. In human astrocytomas that possess mutated p53, the molecule has a longer half-life than the wild-type molecule found in normal cells, and so it is more readily detected immunohistochemically. 10,72 However, this immunohistochemical marker is neither specific for astrocytoma nor especially sensitive. Moreover, in people, not all mutant p53 genes produce an immunohistochemically detectable increase in p53 in the tumor, 10 and reactive astrocytes may occasionally be strongly positive. 68
However, this protein has been immunohistochemically detected in 9 of 31 canine astrocytomas 120 and in 1 feline tumor. 4 Genetic studies have demonstrated that p53 mutations are rare in canine astrocytomas 119,120,144 so that accumulation of product may be insufficient for positive immunohistochemistry. In addition, epidermal growth factor receptor has been detected in higher-grade human and canine astrocytomas, while platelet-derived growth factor alpha was found to correlate with higher grade in human oligodendroglial tumors. 42,115
In humans, p53 labeling is generally used as a part of a diagnostic panel that is also designed to detect an amplified mutation of isocitrate dehydrogenase 1 (IDH1). Most IDH1 mutations in human astrocytomas occur at a single, common site, creating a unique antigenic epitope for producing antibodies that react with only the mutant protein. This biomarker has been used in differentiating astrocytomas from oligodendrogliomas 28 and, along with p53 expression, is helpful in correctly identifying the margins between neoplastic and reactive tissue. 11 Expression of the mutant IDH1 is also correlated with a better prognosis in people (86 months vs 10 months). 125 Unfortunately, a similar IDH1 mutation has not been found in canine glial tumors. 101 Further investigation may demonstrate other mutations in canine tumors that could be used to similar advantage in diagnosis.
Glioblastoma (high-grade, poorly differentiated astrocytoma) is characterized in HE sections by necrosis and perivascular palisades. This tumor has minimal to mixed differentiation along several glial lineages. Tumors are soft and hemorrhagic and contain anisomorphic, bizarre cells. Although borders with normal tissue may be more evident because of their soft nature, glioblastomas grow diffusely and aggressively along white matter tracts and rapidly kill humans. In animals, glioblastomas are rare tumors, and there is no evidence that they arise from preexisting benign astrocytomas. In humans, a mutant of the epidermal growth factor receptor protein is useful in identification. 28 Microarray analysis has demonstrated greater expression of epidermal growth factor receptor in higher-grade canine astrocytic tumors as well. 42
Glioblastoma is distinct from gliomatosis cerebri, a diffusely infiltrating neoplasm that has in various studies been described as being of oligodendroglial, astroglial, or uncertain origins. The staining variability among studies indicates that more than 1 cell of origin may be possible; thus, obtaining immunohistochemical support of the diagnosis can be frustrating. 2,33,63,98 Stem cell markers have not been utilized with these rare tumors.
Oligodendroglioma
Neoplastic oligodendrocytes do not elaborate cytoplasmic processes or produce myelin. Besides the more common solitary masses, multifocal oligodendrogliomas have been reported in 3 dogs, 62 but additional tumors were suspected to be implants from a single site of tumorigenesis.
Oligodendrogliomas are often merely identified on the basis of microscopic pattern and negative expression of GFAP, although the presence of reactive astrocytic processes throughout the mass can be confounding (Fig. 12), especially in biopsies sampled near tumor margins. No immunohistochemical reagents are consistently agreed on for identifying oligodendrogliomas. Myelin basic protein has been used without much success to identify these tumors, which is not surprising given the lack of myelin production by tumor cells. 64,134 In other studies, diagnosis was aided by the finding of factor VIII–positive and smooth muscle actin–positive characteristic glomeruloid vascular loops in the mass. 21,75 These vascular loops are commonly seen in HE sections as well. Vascular loops can be found in other types of tumors. One investigator found patches of neurofilament-positive staining tumor cells in a canine oligodendroglioma, although this is not the general case. 92
Having a better marker for neoplastic oligodendrocytes would assist diagnosis when biopsy specimens are small or distorted and could identify glial tumors of mixed differentiation. One candidate marker protein is 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase), which is expressed early in myelination and may account for up to 4% of total CNS myelin protein. This marker has been used in confirming the presence or absence of nonneoplastic oligodendrocytes in various conditions. 136 It is a marker used by the authors in a small number of cases and seems to hold promise in identifying canine oligodendrogliomas (Table 2, Fig. 13), as it has been in human oligodendroglial tumors. 111 This reagent stains normal oligodendrocyte cell bodies and processes, as well as the cytoplasms of tumor cells. In a pilot study of canine and feline gliomas, this reagent identified 6 of 6 canine oligodendrogliomas, although there was weak cytoplasmic staining in 2 of 3 astrocytomas as well (Table 2). This reagent did not react with normal or neoplastic feline tissue.
Comparison Canine and Feline Oligodendrogliomas and Astrocytomas Stained With GFAP, CNPase, and Olig2 Reagents.
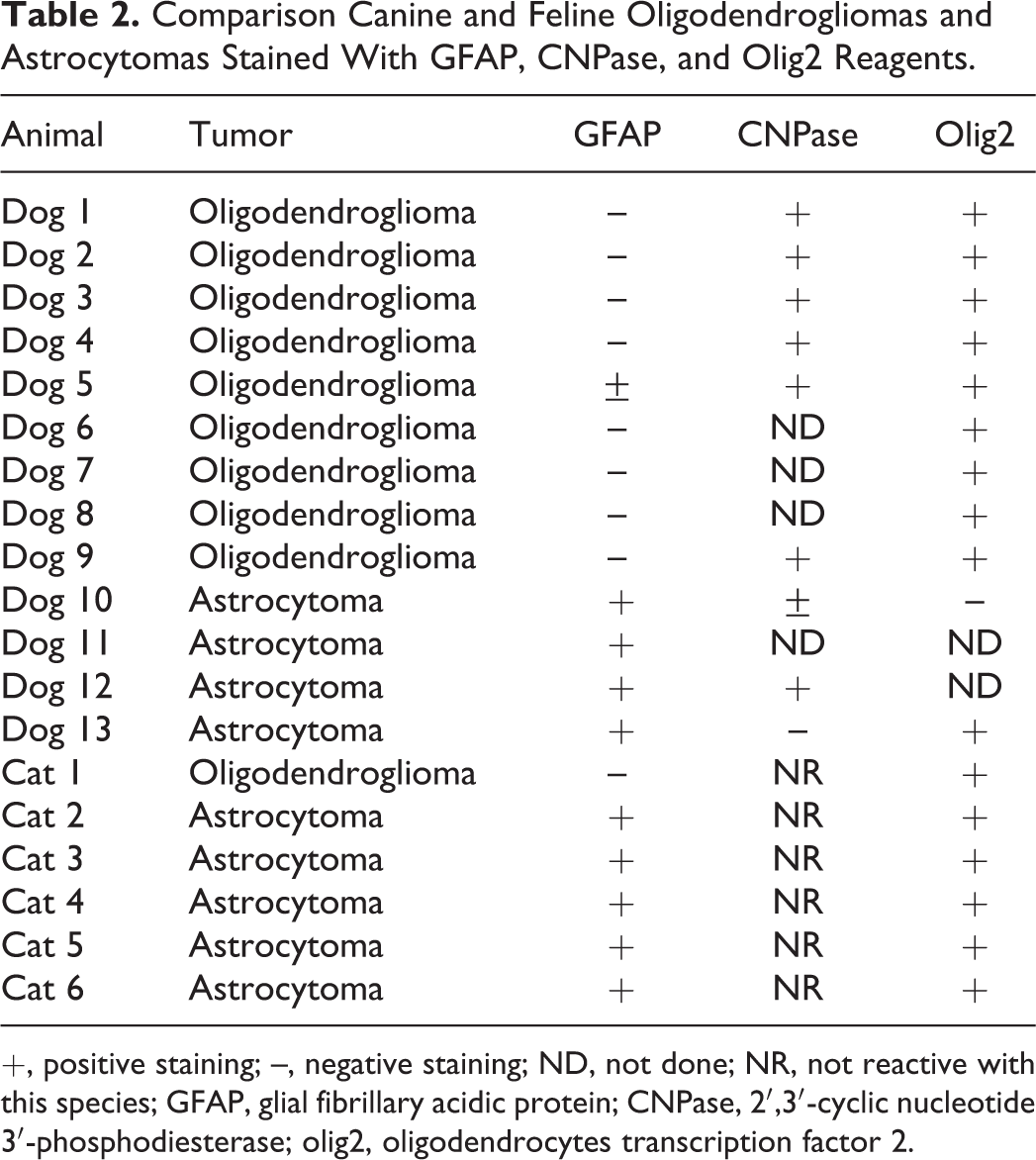
+, positive staining; –, negative staining; ND, not done; NR, not reactive with this species; GFAP, glial fibrillary acidic protein; CNPase, 2′,3′-cyclic nucleotide 3′-phosphodiesterase; olig2, oligodendrocytes transcription factor 2.
Oligodendrocyte transcription factor 2 (olig2) is another marker that has been used in human neoplasms and that successfully identified a case of gliobastoma multiforme of oligodendroglial type in a dog. 33 Olig2 transcription factor is required for oligodendrocyte differentiation and determines patterning in embryonic progenitors. The gene is not required for astrocyte development. 107 Olig2 has been used for positive nuclear immunohistochemical staining of human normal and neoplastic oligodendrocytes, although it was not fully able to separate them. 103 In test staining of a small number of cases from our archives, nuclear staining was found in all oligodendrogliomas (9 of 9 dogs and 1 of 1 cat; Fig. 14, Table 2). Unfortunately, canine and feline astrocytomas also commonly had positive nuclear staining, although nonneoplastic astrocytes were negative. Thus, the reagent may lack the desired specificity for tumor diagnosis in animals (Fig. 15). A more structured study is needed, but others have found similar results. 136 A similar problem has been encountered among human gliomas, and olig2 is now less commonly used to separate astrocytic and oligodendroglial tumors. 52,103,105 In any event, this nuclear marker may be useful in pointing out intersections between low and high cell density at margins of glioma biopsies (Fig. 14). More work is needed to identify better positive markers for neoplastic oligodendrocytes in animals, especially in cats.
Alpha-internexin has been found to be useful in separating oligodendogliomas and mixed tumors from astrocytic neoplasms in a large human case series. 25 This protein, although an intermediate filament protein of neurons, 44 is expressed in oligodendrogliomas. 27 Although not absolute, expression is associated with human IDH I mutations (discussed above). 27 This marker deserves investigation in diagnosis of gliomas in dogs and cats, although these species seem to have their own unique mutational tumor profiles. 129
Ependymoma
Ependymomas are traditionally described as topographically associated with ventricular surfaces, although a recent publication reports cases involving the pial surface of the brain without ventricular involvement. 142 Some of the more anaplastic neoplasms invade the brain. Exfoliation of ependymomas may result in implants (drop metastases) along cerebrospinal fluid pathways. Multiple subtypes of ependymomas have been described, 130,139 including some that lack the characteristic papillary pattern and rosette formation expected in classic tumors. Most have positive expression of vimentin and GFAP by immunohistochemistry 64,131,139 and are not stained with pancytokeratin antibodies. Two malignant ependymomas, with rapid spread in the subarachnoid space, have been recently reported in cats. In another patient, a solitary tumor was successfully excised. 115 Relatively few studies have examined biomarkers in animal ependymomas. In these studies, all ependymomas were identified by positive vimentin staining and GFAP reaction, 136 although ependymomas not expressing GFAP have been identified in dogs and cats. 84,142 Some feline ependymomas are pancytokeratin positive. 84,142 EMA is a useful positive marker of this tumor in humans. A single report of use of this reagent described negative staining in canine choroid plexus tumors, using lung as a positive control, 102 but the technique has not been widely used.
There is recent description of 3 feline cases of subependymomas, which have a solid histologic pattern. The cell of origin in not known, but these masses occur near the ventricle; these cases are uniformly GFAP positive but pancytokeratin negative. 142
Choroid Plexus Tumors
Choroid plexus tumors occur at sites of preexisting choroid plexus. They are a fairly common parenchymal tumor of dogs and readily biopsied. Grossly and microscopically, they appear as rather exuberant versions of normal choroid plexus (red and frond-like). They cause hydrocephalus because of their location and may implant extensively. 64
Human choroid plexus epithelium is thought to arise along with ependyma and thus expresses S100 and GFAP. Tumors may stain positively with these reagents and for vimentin. Vimentin positivity includes not just the tela choroidea but also the choroid plexus epithelium. 12,93 The neoplastic choroidal epithelium expresses cytokeratin (Fig. 16), a chief differentiative feature. Choroid plexus tumors in people stain with GFAP, S100, and EMA (like ependymoma) but uniquely express transthyretin and rarely synaptophysin. 85 These markers have not been explored in animals. Compared to metastatic papillary carcinomas metastasizing to the brain, cytokeratin staining is frequently segmental (Fig. 16). 51 Cytokeratins 7 and 20 have been used to distinguish choroid plexus carcinoma from metastatic papillary carcinomas in people, and Ide found some cytokeratin positive tumors in animals. 48,51 Biomarkers for the glial glutamate transporter (EEAT-1) have also been used in conjunction with location in humans to successfully distinguish choroid plexus neoplasms from papillary metastatic tumors, 8 without expression in normal choroid plexus. 7 The utility of this biomarker in identifying and demarcating choroid plexus tumors of domestic species is not known.
Oftentimes choroid plexus tumors do not present a great problem in diagnostic identification, but markers are lacking that distinguish benign and malignant tumors or predict implantation. In a large series, E-cadherin and β-catenin were expressed in nearly all canine tumors regardless of grade, 90 and GFAP expression appears variable and is usually minimal. According to Westworth et al, diagnoses of malignant choroid plexus tumors require at least 4 of the following 6 morphologic criteria: more than 5 mitoses per 10 high-power field, nuclear atypia, multilayered epithelium, high cell density, solid areas of cell growth, and multifocal areas of necrosis. 141 Ide, in examining 4 canine choroid plexus tumors (1 papilloma and 3 carcinomas), found them to be variably cytokeratin and minimally GFAP positive. 48
Conclusions
From the forgoing discussion, it becomes obvious that the search is incomplete for distinctive combinations of immunohistochemical markers that are capable of conclusively and specifically diagnosing different brain tumors and predicting their behavior. The more commonly used reagents for veterinary tumors are listed in Table 3. Although it is possible to separate meningeal from neuronal from glial neoplasms by their combined morphologic and immunohistochemical characteristics, there is considerable overlap and variation of positivity among tumors within each group so that immunohistochemical staining must be taken in context with the phenotype and location of the mass. Multiple immunohistochemical techniques are usually needed, which can be beyond the scope of some laboratories in the context of an everyday setting for an everyday price. Mutational hot spots are useful in diagnosis of human brain tumors, but the same mutations are not present in the analogous canine tumors. Recent studies on gene expression patterns between human and canine brain tumors reveals some similarities and some differences in tumorigenesis. 114,125,129 CNPase and Olig2 merit more investigation if only for use as reagents to separate gliomas as a group from other neoplasms.
Immunohistochemical Markers Used for Identification of Brain Tumors.
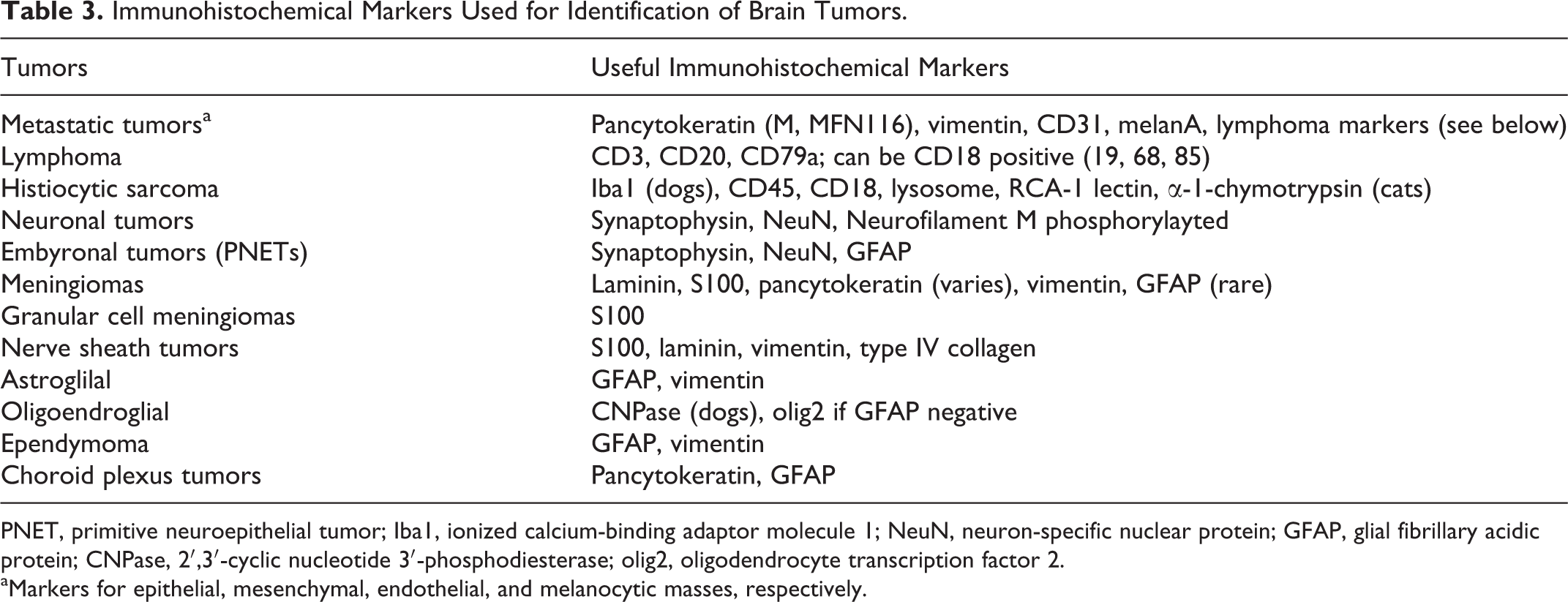
PNET, primitive neuroepithelial tumor; Iba1, ionized calcium-binding adaptor molecule 1; NeuN, neuron-specific nuclear protein; GFAP, glial fibrillary acidic protein; CNPase, 2′,3′-cyclic nucleotide 3′-phosphodiesterase; olig2, oligodendrocyte transcription factor 2.
aMarkers for epithelial, mesenchymal, endothelial, and melanocytic masses, respectively.
The profiling of human brain tumors increasingly relies on the detection and estimation of the numbers of stem cells that are multipotent, undifferentiating, and self-sustaining. 124 This is particularly true in the case of gliomas. It has been demonstrated that these cells are responsible for heterogeneous potential and, thus, immunohistochemical staining of more differentiated cells in the masses, and it appears that common stem cell biomarkers such as class III beta-tubulin, nestin, and CD133 are in fact common to several types of neoplasms 15,57,66 These stem cell reagents may ultimately have a greater predictive value of survival interval, recurrence, and response to therapy compared to those markers that detect differentiated cell types.
Footnotes
Acknowledgements
We gratefully thank Howard Wilson for his help in preparing the illustrations and the histology and immunohistochemistry technicians at the University of Missouri Veterinary Medical Diagnostic Lab.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
