Abstract
The authors recently assessed the likelihood that lifetime cancer bioassays of aspartame, methanol, and methyl tertiary butyl ether conducted with conventional (not specific pathogen free) Sprague–Dawley rats were compromised by Mycoplasma pulmonis disease. From the tumor data and other information, the authors concluded that the rats used in these bioassays likely had M pulmonis disease and that lesions of the disease were plausibly interpreted as lymphoma. Subsequently, they analyzed the nonneoplastic lesion data from these bioassays for occurrence of inflammatory lesions and found that 2,267 of 2,960 rats (76.6%) were reported to have bronchitis, the signature lesion of M pulmonis disease, and that 633 rats (21.4%) were reported to have otitis, another common lesion of the disease. Also, documentation is now available containing serologic evidence of mycoplasma infection in the rats. In contrast, the reports of 6 National Toxicology Program bioassays based on specific pathogen–free Sprague–Dawley rats listed no instances of bronchitis or otitis. These findings provide substantial additional evidence that the bioassays in question were compromised by M pulmonis disease. Therefore, the reported induction of lymphoma in these studies should not be considered in cancer risk assessments. The authors also found that inflammatory lesions were prevalent in lymph nodes, thymus, pleura, and brain. Finally, they found that of all 328 cases of lymphoimmunoblastic lymphoma affecting the lung (the primary form of lymphoma reported), 218 (66.5%) occurred within the first 104 weeks of the studies, showing that occurrence of such lesions was not due to appearance in rats surviving beyond that interval.
In our recent commentary concerning lymphomas reported to be induced in rats in the bioassays of aspartame, methanol, and methyl tertiary butyl ether (MTBE) conducted by a nonprofit cancer research organization, we assessed the neoplasm data for these studies from the organization’s Web site, published reports, and other publicly available information. 13 We concluded that the rats used in these bioassays probably had Mycoplasma pulmonis disease, that the primary form of lymphoma reported is inconsistent with recognized forms of lymphoma in rats, and that lesions of M pulmonis disease were plausibly interpreted as lymphoma. We subsequently obtained and analyzed the nonneoplastic lesion data for these studies, analyzed the age distribution of lymphoimmunoblastic lymphoma, and accessed an excerpt of a court deposition regarding serologic testing for mycoplasma infection in the organization’s rat colony. The results provide further evidence that the studies were compromised by M pulmonis disease, and they raise additional questions about the reported results. Our findings are therefore relevant to the US Environmental Protection Agency’s (EPA’s) Toxicological Review of Methanol (as based on the Integrated Risk Information System of the National Center for Environmental Assessment; see http://oaspub.epa.gov/eims/eimscomm.getfile?p_download_id=494156), namely because the draft version concludes that “methanol is likely to be carcinogenic to humans,” a conclusion based in considerable part on the organization’s methanol study.
Additional Evidence of Mycoplasma pulmonis Disease
M pulmonis disease is characterized by chronic inflammation throughout the respiratory tract—especially, the bronchi—and including the tympanic bullae. 7,8,10 –12,14 There is no other pathogen of rats that causes lesions of this type and distribution, with the single and rare exception of cilia-associated respiratory bacillus. 7,8,10 –12,14 We therefore compiled the reported occurrence of such lesions among treated and control rats in the aspartame, methanol, and MTBE bioassays from the following tables, obtained by a Freedom of Information Act request from the EPA: Nonneoplastic Lesions by Individual Animal, Chronic Lifetime, Aspartame (http://ntp.niehs.nih.gov/ntp-db/NTP_Reports/NTP_Pathology_Tbls/aspartame_tables/AspartameP09.txt), Individual Animal Pathology Data, Chronic Lifetime, Methyl Alcohol (http://www.methanol.org/pdfFrame.cfm?pdf=00117_68_p14.pdf), and Individual Animal Pathology Data, Chronic Lifetime, Methyl-Tertiary-Butyl Ether obtained by Freedom of Information Act request from the EPA (not available online).
These data include listings for inflammation in the “nose, sinus”; however, rats do not have sinuses, so it is unclear whether this represents rhinitis. Rhinitis is a common nonspecific finding in rats. 5 Therefore, we did not include rhinitis in our analysis. The tables for the aspartame and MTBE bioassays list chronic inflammation in bronchi and hyperplasia in bronchioles. We included cases with either or both as bronchitis because epithelial hyperplasia is characteristic of respiratory mucosal inflammation induced by M pulmonis. 7,8,10 –12,14 Bronchitis and otitis were the most frequently recorded findings. Table 1 shows the occurrence of these lesions. Of all 2,960 rats in the 3 bioassays, 2,267 (76.6%) were reported to have bronchitis and 633 (21.4%), otitis. Otitis was prevalent in the methanol study, being listed for 547 of 800 rats (68.4%). Respiratory tract lesions in sites other than bronchi were not recorded for the aspartame study rats.
Occurrence of Bronchitis and Otitis
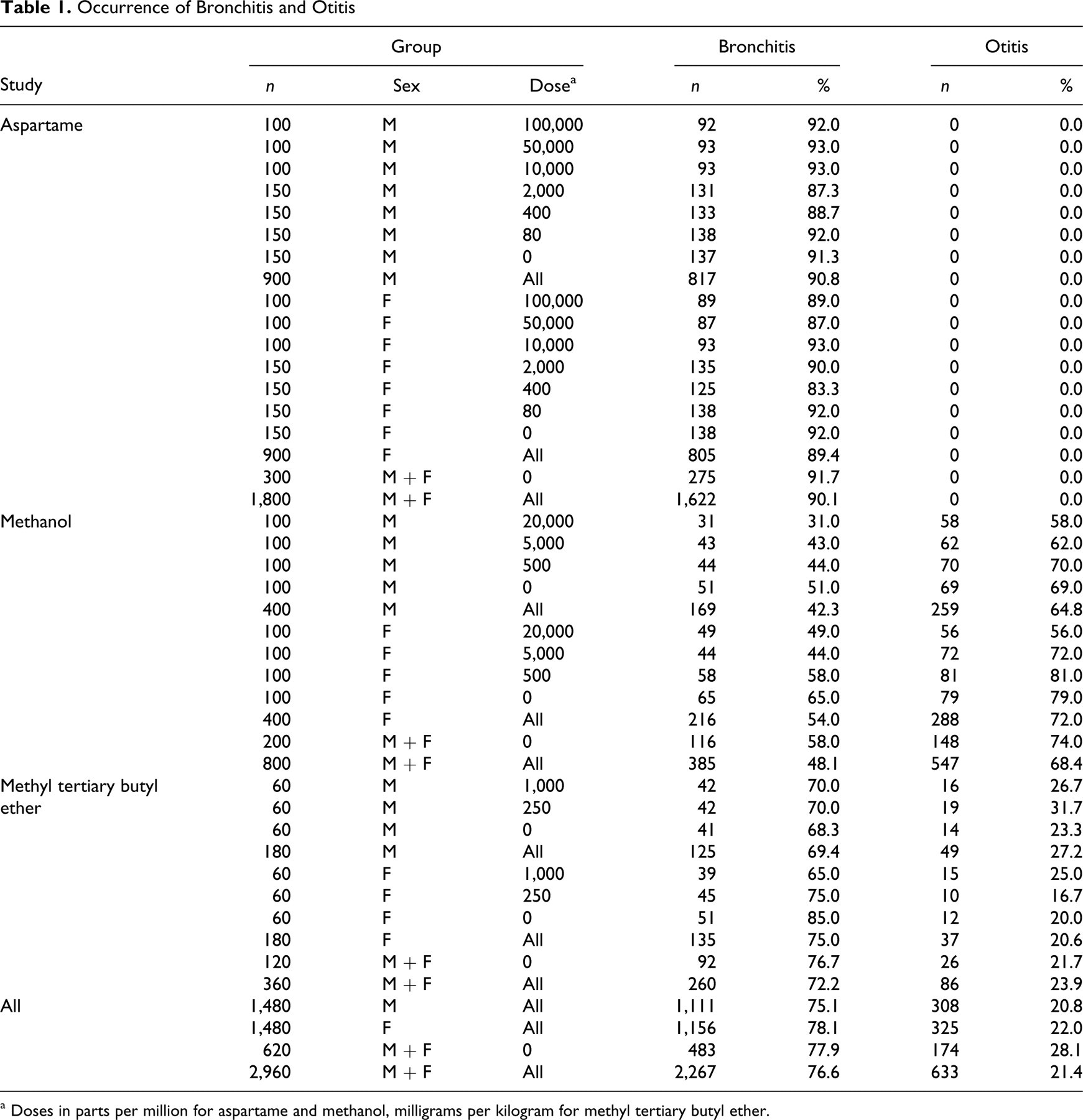
a Doses in parts per million for aspartame and methanol, milligrams per kilogram for methyl tertiary butyl ether.
The organization in question uses Sprague–Dawley (SD) rats in its bioassays.
1
–4,6
To assess occurrence of inflammatory lesions consistent with M pulmonis disease among specific pathogen–free Sprague–Dawley rats used in 2-year National Toxicology Program bioassays, we examined the nonneoplastic lesion data for control rats in 6 bioassay reports accessed via the program’s webpage (Long-Term Study Reports and Abstracts; http://ntp.niehs.nih.gov/?objectid=D16D6C59-F1F6-975E-7D23D1519B8CD7A5): Toxicology and Carcinogenesis Studies of 3,3,′4,4,′5-pentachlorobiphenyl (PCB 126) (CAS No. 57465-28-8) in Female Harlan Sprague-Dawley Rats (Gavage Studies), Toxicology and Carcinogenesis Studies of 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) (CAS No. 1746-01-6) in Female Harlan Sprague-Dawley Rats (Gavage Studies), Toxicology and Carcinogenesis Studies of 2,3,4,7,8-Pentachlorodibenzofuran (PeCDF) (CAS No. 57117-31-4) in Female Harlan Sprague-Dawley Rats (Gavage Studies), Toxicology and Carcinogenesis Studies of a Mixture of 2,3,7,8-Tetrachlorodibenzo-p-Dioxin (TCDD) (CAS No. 1746-01-6), 2,3,4,7,8-Pentachlorodibenzofuran (PeCDF) (CAS No. 57117-31-4), and 3,3,’4,4,’5-Pentachlorobiphenyl (PCB 126) (CAS No. 57465-28-8) in Female Harlan Sprague-Dawley Rats (Gavage Studies), Toxicology and Carcinogenesis Studies of 2,2,′4,4,′5,5′-Hexachlorobiphenyl (PCB 153) (CAS No. 35065-27-1) in Female Harlan Sprague-Dawley Rats (Gavage Studies), and Toxicology and Carcinogenesis Studies of a Binary Mixture of 3,3,′4,4,′5-Pentachlorobiphenyl (PCB 126) (CAS No. 57465-28-8) and 2,2,′4,4,′5,5′-Hexachlorobiphenyl (PCB 153) (CAS No. 35065-27-1) in Female Harlan Sprague-Dawley Rats (Gavage Studies).
There were no recorded instances of bronchitis or otitis, and the sentinel animal health assessment results, including those for M pulmonis serologic tests, were negative for all 6 bioassays. The predominant inflammatory lesion reported was “inflammation cellular, histiocyte” in the lung, which was recorded for 41/53 (77.4%) to 47/53 (88.7%) of the rats. Alveolar histiocytosis is a common nonspecific finding in rats and is not associated with an infectious disease.
5,10
Recently, an excerpt from a court deposition of the lead author of the reports of the organization’s MTBE bioassay 1 –4 became available as an attachment to a comment on the EPA’s draft review of methanol (Integrated Risk Information System; http://www.regulations.gov/search/Regs/contentStreamer?objectId=0900006480abe0cb&disposition=attachment&contentType=pdf). According to this testimony, the organization’s animal colony is annually screened for pathogens and parasites, and antibodies to “mycoplasma” have been found. The nature of the test and the identity of the testing laboratory were not stated, but in view of the prevalence of lesions characteristic of M pulmonis disease among the aspartame, methanol, and MTBE bioassay rats, we consider the test results sufficient to refute the argument that the presence of M pulmonis disease in the colony “has not been confirmed” 6 (http://oaspub.epa.gov/eims/eimscomm.getfile?p_download_id=494156).
Other Inflammatory Lesions
Observations discussed in our previous commentary indicate that the aspartame study rats had inflammatory lesions in organs other than those of the respiratory tract, which were unlikely due to M pulmonis, thus suggesting involvement of other pathogens. 13 We therefore evaluated occurrence of inflammatory lesions in organs other than the respiratory tract and ear and found that such lesions were unexpectedly prevalent among the rats used in these 3 studies, as shown in Table 2 . Of all 2,960 rats in the 3 bioassays, 2,462 (83.2%) were listed as having chronic inflammation of some combination of mandibular, axillary, inguinal, and/or mesenteric lymph nodes. (We excluded mediastinal lymph nodes because they would be reactive if bronchopulmonary inflammation were present.) Among the aspartame study rats, 1,269 of 1,800 (70.5%) were reported to have inflammatory changes in the thymus. Pleuritis was recorded for 1,235 (41.7%) of all 2,960 rats in the 3 bioassays and for 1,136 (63.1%) of the 1,800 aspartame study rats. Among all 2,960 rats in the 3 studies, 510 (17.2%) were reported to have meningitis, encephalitis, and/or brain abscesses. These findings are unusual and raise questions concerning histopathologic interpretation and health status of the rats. For example, it is unclear whether the lymph node changes represent true lymph node inflammation or reactive hyperplasia. Thymic inflammation is rarely diagnosed in rats, and we think that it would be difficult to find more than thymic remnants in many aged rats. Thus, the nature of the reported thymic inflammation is unclear. The reported prevalence of pleuritis and inflammatory changes in the brain and meninges seem unusual and suggest involvement of pathogens such as Streptococcus pneumoniae and Corynebacterium kutscheri, which are capable of hematogenous dissemination with localization in various organs. In contrast, few such findings were recorded for the National Toxicology Program’s bioassays cited above.
Occurrence of Other Inflammatory Lesions
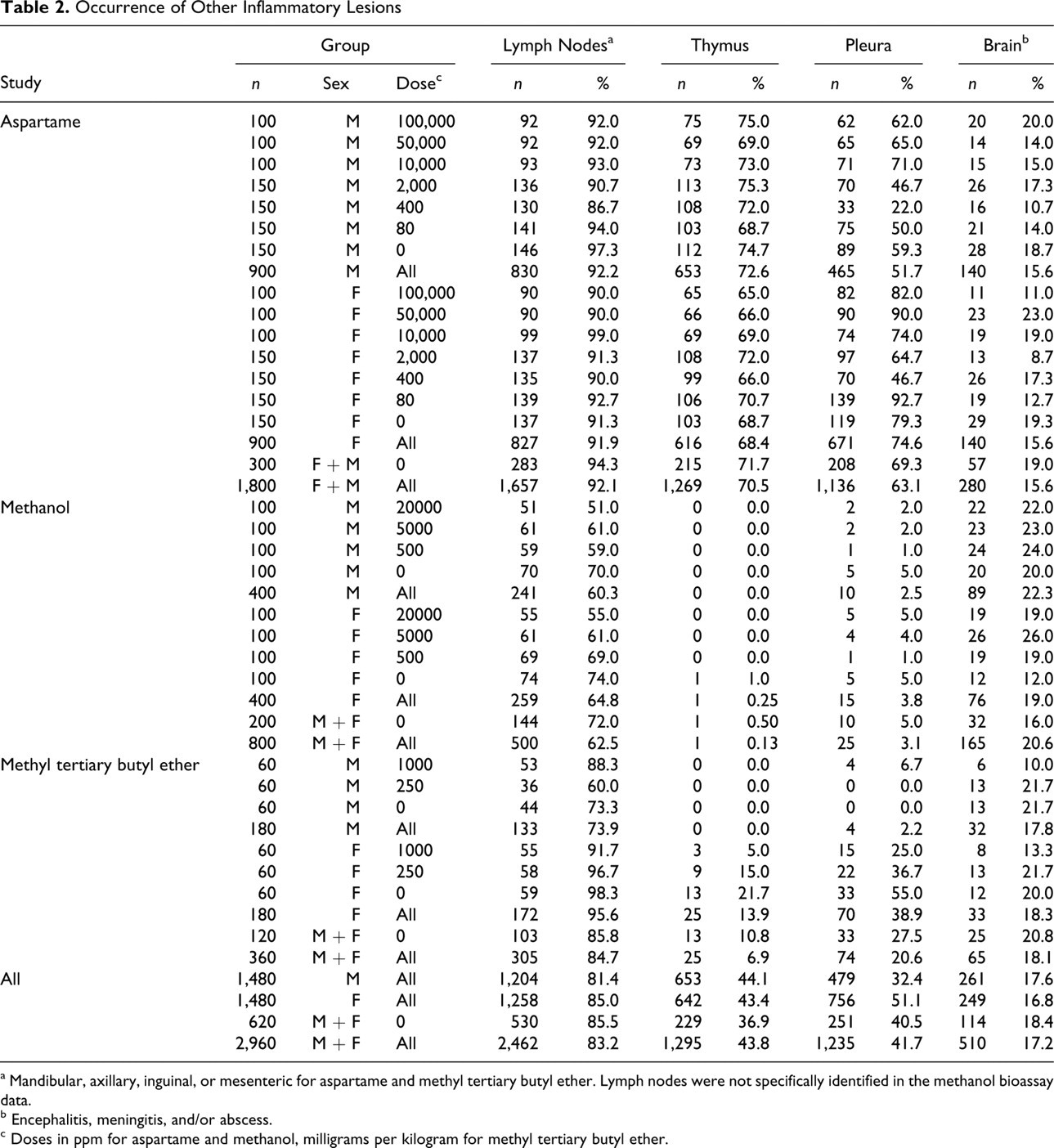
a Mandibular, axillary, inguinal, or mesenteric for aspartame and methyl tertiary butyl ether. Lymph nodes were not specifically identified in the methanol bioassay data.
b Encephalitis, meningitis, and/or abscess.
c Doses in ppm for aspartame and methanol, milligrams per kilogram for methyl tertiary butyl ether.
Organ Distribution and Age Occurrence of Lymphoma
As we discussed previously, the cellular morphology and predominantly pulmonary organ distribution of lymphoimmunoblastic lymphoma, the major form of lymphoma reported in these 3 bioassays, are atypical of lymphoma in rats and plausibly consistent with lesions of M pulmonis disease. 13 It has been argued that this interpretation is incorrect because organs other than the lung were affected and that a treatment effect remains if “lung-only” cases are excluded (http://oaspub.epa.gov/eims/eimscomm.getfile?p_download_id=494156). However, proliferation of lymphocytes and plasma cells in local and other lymph nodes and in the spleen is expected in M pulmonis disease, owing to the mitogenic properties of M pulmonis 9 and the vigorous and persistent inflammatory response the organism induces. Thus, the distribution of lesions is no more consistent with lymphoma than with M pulmonis disease, especially when one takes into account that lymphoma in rats does not typically arise in the lung and that other pathogens were likely present. 13 Therefore, if rats are excluded on the basis of lung involvement, then all rats with lymphoimmunoblastic lymphoma in the lung and any other organs must be excluded, not just rats with that diagnosis in only the lung. Table 3 shows the organs most commonly affected by lymphoimmunoblastic lymphoma in these bioassays. Among all rats in the 3 bioassays, the lung was affected in 328 (90.9%) of all 361 cases of lymphoimmunoblastic lymphoma. Thus, if all rats with lung involvement are excluded, less than 10% of cases remain. It remains to be determined whether statistical analysis of the small number of remaining cases supports existence of treatment effects.
Organ Distribution of Lymphoimmunoblastic Lymphoma a

a All rats for each bioassay (treated + control).
b Organs affected in ≥ 10% of cases.
c Mandibular, axillary, inguinal, or mesenteric for aspartame and methyl tertiary butyl ether. Lymph nodes were not identified in the methanol bioassay data.
In bioassays conducted by the organization in question, the animals are allowed to die spontaneously, and the studies are continued until all the animals have died, rather than being terminated at 104 weeks. 6 The occurrence of lymphoma in the organization’s studies, but not in bioassays of the same chemicals conducted by others, is arguably due to the longer study duration—that is, to the occurrence of lymphoma in rats surviving past 104 weeks. 6 However, as shown in Table 4 , of all rats in the 3 bioassays, 218 cases (66.5%) from 328 of lymphoimmunoblastic lymphoma affecting the lung occurred within 104 weeks. Therefore, occurrence of the lesions in question cannot be simply due to their appearance in rats surviving beyond that interval. It also has been argued that the age distribution of spontaneous deaths among rats in the organization's bioassays is inconsistent with an infectious disease and that “respiratory infection” merely represents a terminal event in aged rats. 6 However, M pulmonis disease is not an opportunistic disease of aged rats but is acquired by the young from affected mothers, and it persists for life. 7,8,10 –12,14 Therefore, lesions of the disease would be present whenever death occurred, and if the lesions were interpreted as lymphoma, then the age distribution of lymphoma diagnoses would actually be the distribution of rat deaths attributed to M pulmonis disease.
Age Distribution of Lymphoimmunoblastic Lymphoma Affecting the Lung a
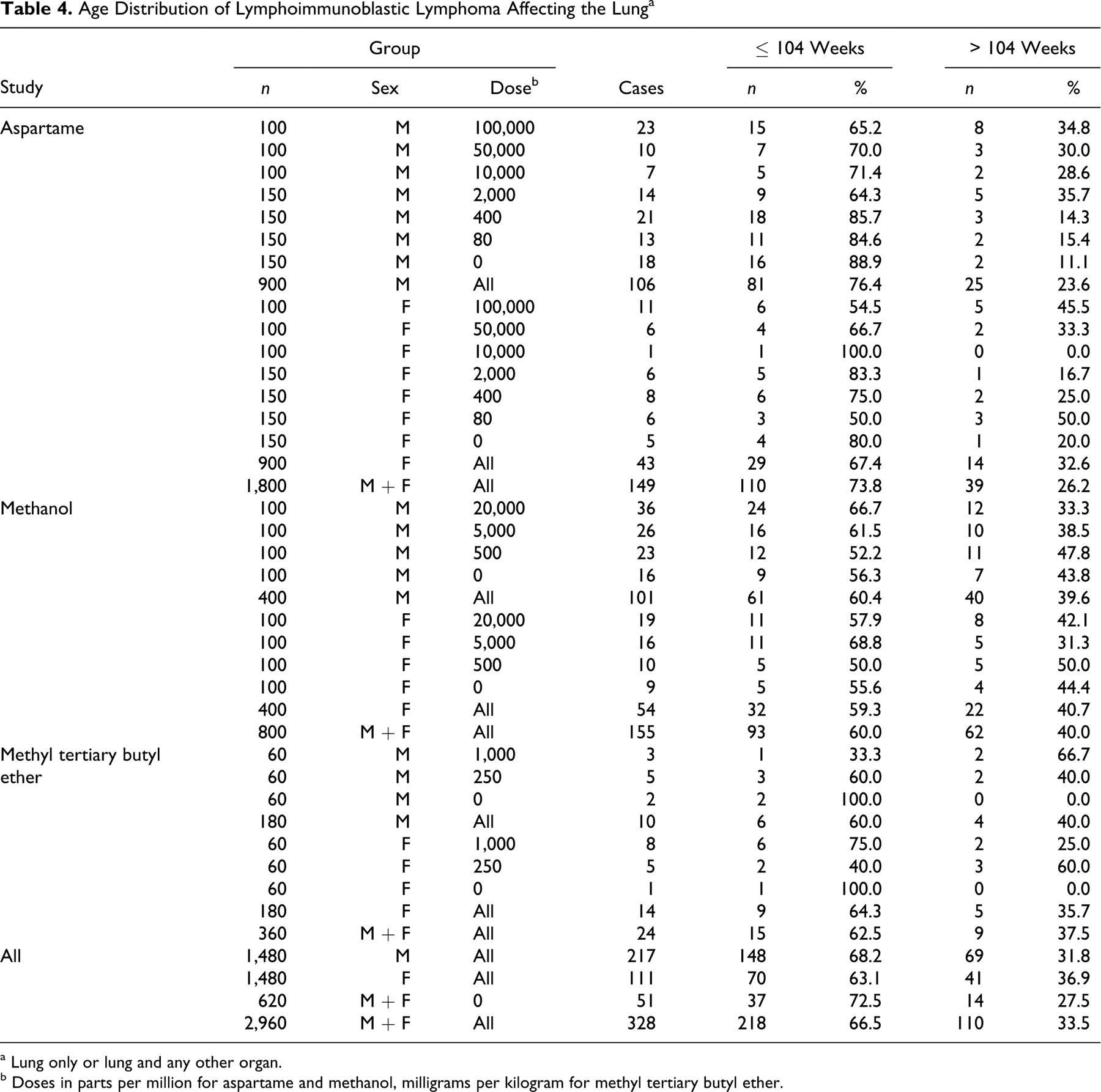
a Lung only or lung and any other organ.
b Doses in parts per million for aspartame and methanol, milligrams per kilogram for methyl tertiary butyl ether.
Conclusion
We consider the evidence discussed herein and previously 13 to be convincing that the rats in the organization’s aspartame, methanol, and MTBE bioassays had M pulmonis disease. We regard this alone as sufficient reason to question the conclusion that these chemicals induced lymphoma because it cannot be ascertained whether lesions diagnosed as pulmonary lymphoma were caused by chemical exposure, by M pulmonis, or by an interaction of the two. We also think it important to assess the accuracy of the lymphoma diagnoses because lesions of M pulmonis disease were plausibly interpreted as pulmonary lymphoma 13 and because an association between M pulmonis disease and lymphoma is not disproved, although we consider such an association unlikely. 13 For these reasons, we strongly believe that no regulatory agency should include the reported induction of lymphoma in these bioassays in cancer risk assessments unless such effects can be verified. We again emphasize that the issues of M pulmonis disease and diagnostic accuracy could be conclusively resolved by independent peer review of the slides and appropriate diagnostic testing. 13 To that end, any review of the studies in question should include individuals having expertise in pathology of M pulmonis disease, lymphomas of laboratory rats, and diagnosis and control of infectious diseases of laboratory rodents. They should have free access to the slides; they should be able to document the identity of the slides; and they should examine the slides for lesions characteristic of M pulmonis disease throughout the respiratory tract, as well as for accuracy of the lymphoma diagnoses. As we have suggested, 13 paraffin blocks of lung and other respiratory tract organs could be subjected to polymerase chain reaction–based testing for M pulmonis. Inasmuch as such testing can present technical challenges, we recommend that blood samples be obtained from the rat-breeding colony for independent serologic testing for M pulmonis and other pathogens, which is commercially available at modest cost.
Footnotes
The views expressed in this article are solely those of the authors and do not necessarily reflect the views or policies of the University of Alabama at Birmingham. The authors were paid consultants of Environ Corp, an environmental consulting firm, regarding MTBE, and E. E. McConnell is currently a study monitor of an industry supported 2-year drinking-water study of MTBE, being conducted at the Hamner Institute of Toxicology. The authors otherwise have no financial interests in—and have received no grants, contracts, or any other support, compensation, or consideration from—the manufacturers of aspartame, methanol, or MTBE and received no compensation or other incentive in regard to this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
