Abstract
In humans and in mouse models, precursor B-cell lymphoblastic leukemia (B-ALL)/lymphoblastic lymphoma (B-LBL) can be classified as either the pro-B or pre-B subtype. This is based on the expression of antigens associated with the pro-B and pre-B stages of B-cell development. Antigenic markers can be detected by flow cytometry or immunohistochemistry (IHC), but no comparison of results from these techniques has been reported for murine B-ALL/LBL. In our analysis of 30 cases induced by chemical or viral mutagenesis on a WT or Pax5+/– background, 18 (60%) were diagnosed as pro-B by both flow cytometry and IHC. Discordant results were found for 12 (40%); 6 were designated pro-B by IHC and pre-B by flow cytometry and the reverse for the remaining 6 cases. Discordance occurred because different markers were used to define the pro-B–to–pre-B transition by IHC vs flow cytometry. IHC expression of cytoplasmic IgM (μIgM) defined the pre-B stage, whereas the common practice of using CD25 as a surrogate marker in flow cytometry was employed here. These results show that CD25 and μIgM are not always concurrently expressed in B-ALL/LBL, in contrast to normal B-cell development. Therefore, when subtyping B-ALL/LBL in mice, an IHC panel of B220, PAX5, TdT, c-Kit/CD117, CD43, IgM, and ΚLC should be considered. For flow cytometry, cytoplasmic IgM may be an appropriate marker in conjunction with the surface markers B220, CD19, CD43, c-Kit/CD117, BP-1, and CD25.
The use of next-generation sequencing in the study of human leukemia/lymphoma is identifying an ever-increasing number of genetic mutations and chromosomal translocations suspected to be drivers of oncogenesis. 28,33,43,48 To confirm whether these suspected drivers are indeed responsible for the development of leukemia/lymphoma, they need to be validated in genetically engineered mouse models. Similarly, most of what is known regarding the etiology of leukemia/lymphoma in humans is based on studies of naturally occurring and genetically engineered mouse models of leukemia/lymphoma. 35 These models are often subsequently used to test specific targeted therapies based on these mutations; 47 therefore, it is important to ensure that they accurately reflect their human counterparts. To compare the mouse model to the human disease appropriately, precision is needed in diagnosing and classifying specific subtypes of leukemia, such as B-cell lymphoblastic leukemia (B-ALL)/lymphoblastic lymphoma (B-LBL). The diagnosis of leukemia and lymphoma is based on a combination of phenotypic, cytochemical, and molecular/cytogenetic criteria. The World Health Organization (WHO) classification of B-cell lymphoid neoplasms divides them into the categories of B-ALL/LBL and mature B-cell neoplasms, which include chronic lymphocytic leukemia (CLL) and many other lymphomas. 54 The only subclassification of B-ALL/LBL by the WHO is defined by the presence of specific genetic abnormalities, most often chromosomal translocations.
A similar diagnostic approach is used to classify leukemia/lymphoma in mice, as described in the Bethesda proposals for classification of lymphoid neoplasms in mice. 35 This murine classification does not include subtypes based on cytogenetic abnormalities, but their importance and role in leukemogenesis have been tested and verified in mouse models. 20,23,34,55 Therefore, it is important to know how the diagnostic and classification systems used for these mouse models compare to those used for humans.
B-ALL/LBL is often regarded as the malignant counterpart of normal B-cell precursors. Normal B cells originate from hematopoietic stem cells in the bone marrow, where they progress through the earliest stages of development. B-lineage precursor cells go through the pro-B and pre-B stages of development before they become immature B cells that migrate from the bone marrow to secondary lymphoid tissue. 18 Therefore, B-ALL/LBL can be subtyped as being of the pro-B or pre-B stage of development. Although this pro-B or pre-B designation is not part of the official WHO classification of lymphoid neoplasms in humans or of the Bethesda proposals for classification in mice, 35 such subtyping is often performed for research purposes. 7,15,52 The subtyping is based on the expression of antigen markers associated with the different stages of normal B-cell development (Table 1); however, there is no consensus as to the best classification system and marker combination for mouse B cells. The most frequently used systems were developed in the 1990s, known as the Hardy/Philadelphia, Basel, and Osmond classification/nomenclature systems for B-cell development. 3,17,19,29,31,32,40,49,50 These groups used different nomenclature for the stages of development and, in some cases, different markers to define these stages. Consequently, there is confusion and inconsistency in the literature, making it difficult to directly compare studies that used different systems and challenging to choose a system for a particular study. In the case of murine B-cell development, the main difference in the classification systems lies in the subdivision of the pro-B and pre-B stages, but there is general agreement that the pro-B–to–pre-B transition is defined by the completion of the productive rearrangement of the immunoglobulin heavy chain (IgH) locus and the subsequent expression of cytoplasmic immunoglobulin heavy chain μ (μIgM). 41 This transition is further characterized by the cessation of surface c-Kit/CD117 expression. 49,56 Since the initial classification systems were devised, it has been demonstrated that 90% to 95% of cells with surface expression of CD25 (IL-2 receptor α chain) concurrently express μIgM; 49 therefore, CD25 is now often used as a surrogate marker of pre-B cells as it is more amenable to detection by flow cytometry. 22,30,57 It is not possible to differentiate cytoplasmic IgM (μIgM) from surface IgM (sIgM) by immunohistochemistry (IHC). Therefore, when analyzing IgM by IHC, it is necessary to assess for light-chain differentiation; a lack of light-chain expression indicates that the cell is still in the pre-B stage and has not yet differentiated into a transitional/immature B cell. The accurate distinction between the pro-B and pre-B stages in mice is important because many of the specific antigen markers used to define stages in B-cell development differ between mice and humans and consequently cannot be directly compared. 16 What is directly comparable is the stage of maturational arrest (ie, whether the cells are at the pro-B or the pre-B stage). This is a key step in validating that the model accurately reflects the human condition and is appropriate to be used for testing specific targeted therapies.
Antigen Expression During the Stages of Mouse B-Cell Development as Defined by the 3 Main Classification Systems.a
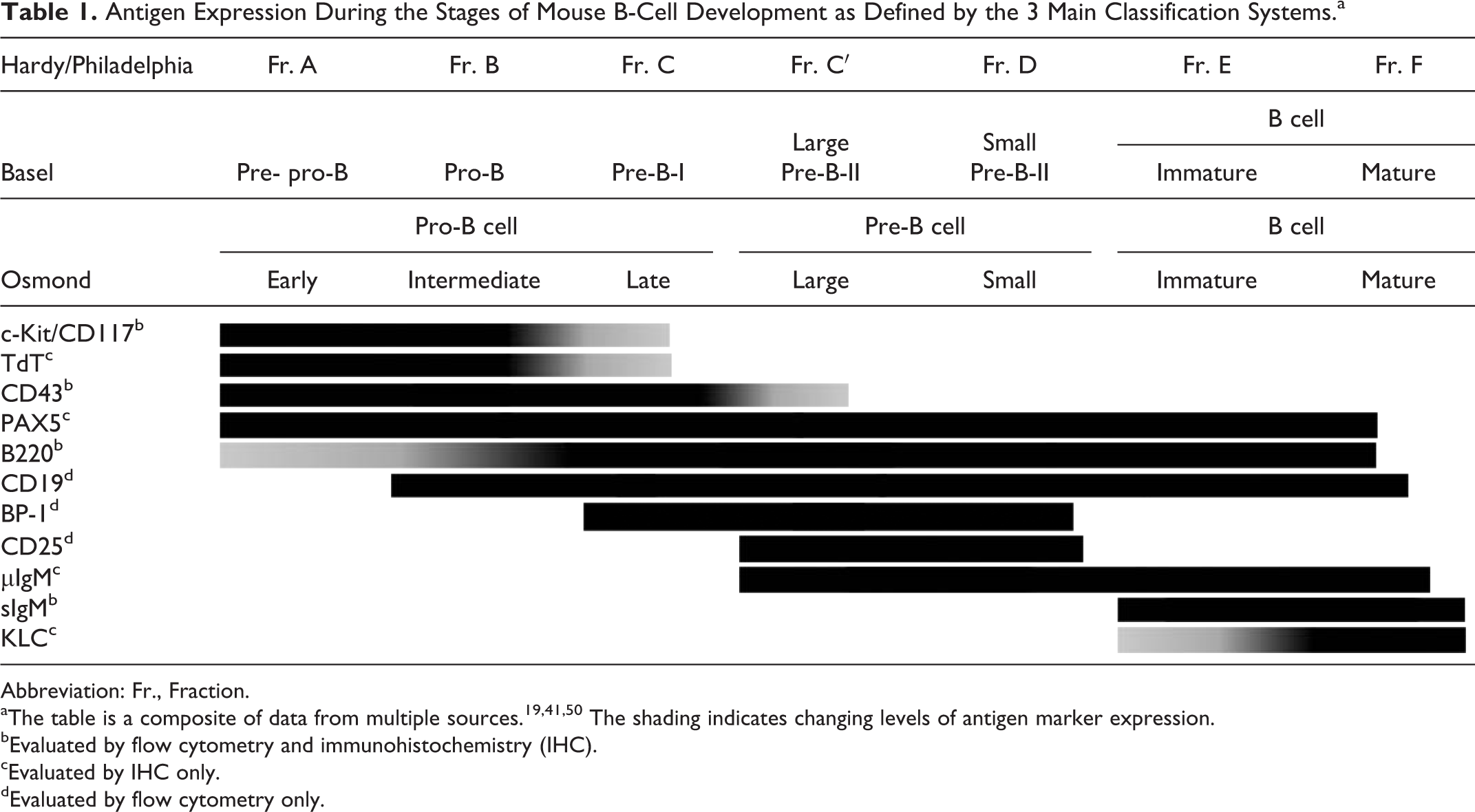
Abbreviation: Fr., Fraction.
aThe table is a composite of data from multiple sources. 19,41,50 The shading indicates changing levels of antigen marker expression.
bEvaluated by flow cytometry and immunohistochemistry (IHC).
cEvaluated by IHC only.
dEvaluated by flow cytometry only.
Although numerous modalities are employed to diagnose leukemia/lymphoma, the expression of cell surface markers, as analyzed by flow cytometry, is considered the gold standard and forms the basis of most diagnostic classifications. 10,26,60 The expression of cell surface markers can also be analyzed by IHC and can be used as an adjunct to flow cytometry or even as a replacement for it in some cases. Although flow cytometry has many advantages over IHC owing to its sensitivity and speed, sometimes only archived formalin-fixed, paraffin-embedded material is available. Therefore, whether in a diagnostic or research setting, it is important to know how well the results from IHC correlate with those from flow cytometry. In addition, IHC may provide important information regarding the expression of nuclear or cytoplasmic markers that cannot be detected by flow cytometry without prior membrane permeabilization. There have been only a few studies of the correlation between flow cytometry and IHC findings for human leukemia, although studies comparing the results obtained with these techniques have been performed for lymphoma and lymphoproliferative disease, 8,9 chronic B-cell leukemia and lymphoma, 1 and acute lymphoblastic leukemia. 2,4 Most of these studies evaluated how well IHC performed in detecting the same markers used in flow cytometry, but few evaluated and/or compared the diagnosis of the same lymphoma by IHC and flow cytometry.
To our knowledge, no previous study has compared flow cytometry and IHC for the diagnosis of murine B-cell leukemia/lymphoma in a cohort of animals with lymphoblastic leukemia/lymphoma. Therefore, our goal in the present study was to compare the subclassification of a set of mouse precursor B-ALL/LBLs from the same mice as pro-B or pre-B based on flow cytometry to the subclassification based on IHC markers and to define the IHC markers necessary for such a determination. For this purpose, we used a combination of the markers used in the previously reported Hardy/Philadelphia, 18 Basel, 50 and Osmond 31 classification/nomenclature systems for B-cell development.
Materials and Methods
Experimental Model
This study was performed by retrospectively analyzing the flow cytometric data generated in a previously published leukemia/lymphoma study 13 and by carrying out additional IHC analysis to be able to make the desired comparisons. The earlier study investigated the role of PAX5 as a tumor suppressor in ALL/LBL. Pax5 wild-type (WT) and heterozygous (Pax5+/−) mice (Pax5tm3Mbu/J 58 on a C57Bl/6 J;Sv129 background) were treated with the mutagens N-ethyl-N-nitrosourea (ENU) or Moloney murine leukemia retrovirus (MMLV). Mice given MMLV were injected with viral supernatant as newborn pups (<3 days old). All mice were thymectomized at 4 to 5 weeks of age to prevent the development of T-cell leukemia. Mice treated with ENU were injected with the mutagen 3 to 5 days after undergoing thymectomy. After the initial treatment, mice were monitored daily and were euthanized when they developed signs of illness, which included reduced activity, coat ruffling, palpable splenomegaly, or paralysis. The experimental details of each mouse in the cohort is provided in Supplemental Table S1, including sex, genotype, mutagenesis method, and the presence of an enlargement of the spleen, lymph nodes, or liver at the time of necropsy. All procedures were approved by the St. Jude Institutional Animal Care and Use Committee.
Tissue Collection and Immunophenotyping
After a mouse was euthanized, bone marrow cells were flushed from the femur with RPMI medium supplemented with 10% fetal calf serum (HyClone, Fisher Scientific, Waltham, MA), penicillin, and streptomycin. Single-cell suspensions of this bone marrow and a portion of the spleen were prepared by passing samples through a 100-μm cell strainer (BD Pharmingen, San Diego, CA). Red blood cells were lysed, and the remaining cells were stained with monoclonal antibodies conjugated to phycoerythrin (PE), fluorescein isothiocyanate (FITC), or allophycocyanin (APC). The following monoclonal antibodies were used: B220-APC, CD19-PE, CD43-FITC, BP1-PE, CD25-PE, immunoglobulin (Ig) M–FITC, Gr1-PE, Mac1-APC, c-Kit–APC, Sca1-FITC, CD3-PE, and Ter119-APC (BD Pharmingen, San Diego, CA). Data were collected using FACSCalibur or LSRII flow cytometers (BD Biosciences. San Jose, CA), and analysis was performed using FlowJo (TreeStar, Inc, Ashland, OR). The sternum and remaining portion of the spleen were fixed in 10% neutral-buffered formalin and embedded in paraffin, and 4-μm sections were stained with hematoxylin and eosin (HE) or used for IHC analysis after being heated for 1 hour at 60°C and then deparaffinized and rehydrated. IHC for c-Kit/CD117, CD43, TdT, PAX5, B220, IgM, CD3, myeloperoxidase (MPO), and κ light chain (ΚLC) was performed as previously described 45 and is detailed in Supplemental Table S2.
Diagnosis and Subtyping of B-Cell Leukemia/Lymphoma by Flow Cytometry and IHC
For each case, the evaluation for the presence of leukemia/lymphoma and the determination of the expression of each marker by IHC were performed by 2 pathologists (L.J.J. and J.E.R.). As previously reported, samples were considered positive for flow cytometry markers if more than 20% of the cells were positive. 13 Similarly, for IHC, a threshold of 20% was used, per the Proposals for the Immunological Classification of Acute Leukemias. 6 B-cell leukemias/lymphomas were subtyped according to the expression of antigen markers, as shown in Table 1. For both flow cytometry and IHC, marker expression in the spleen and bone marrow was similar. A representative flow cytometry plot and IHC panel are shown for cases subtyped as pro-B or pre-B in Figs. 1 to 4. Based on the flow cytometry data, cases were diagnosed as B-cell leukemia if they were positive for B220 and/or CD19 and negative for Gr1, CD3, and Ter119. They were subtyped as being pro-B if they expressed only a combination of B220, CD19, and/or BP1 (Fig. 1) and as pre-B (Fig. 2) if they also expressed CD25 (following the classification system in Table 1). 13 However, CD25 expression data were available for only a subset of the tumors. Based on histopathology and IHC results, cases were diagnosed as B-cell leukemia/lymphoma if they had morphologic blastic features and were positive for PAX5 and/or B220 and negative for CD3 and MPO. The blastic features observed in these cases were similar to those described in the Bethesda protocol for precursor B-cell leukemia/lymphoma; within both the pro-B and pre-B subtypes, the cells are medium to large sized with round or oval nuclei and fine stippled chromatin and variable but often single, central distinct to sometimes prominent nucleoli. 35 Although the expression of all markers was taken into account, B-cell leukemia cases were subtyped as pro-B (Fig. 3) if they were negative for IgM or as pre-B (Fig. 4) if they were positive for IgM and negative for ΚLC (following the classification in Table 1). Difficult cases and those with discordant results were reviewed jointly by using a multiheaded scope and were subsequently assigned a final interpretation.
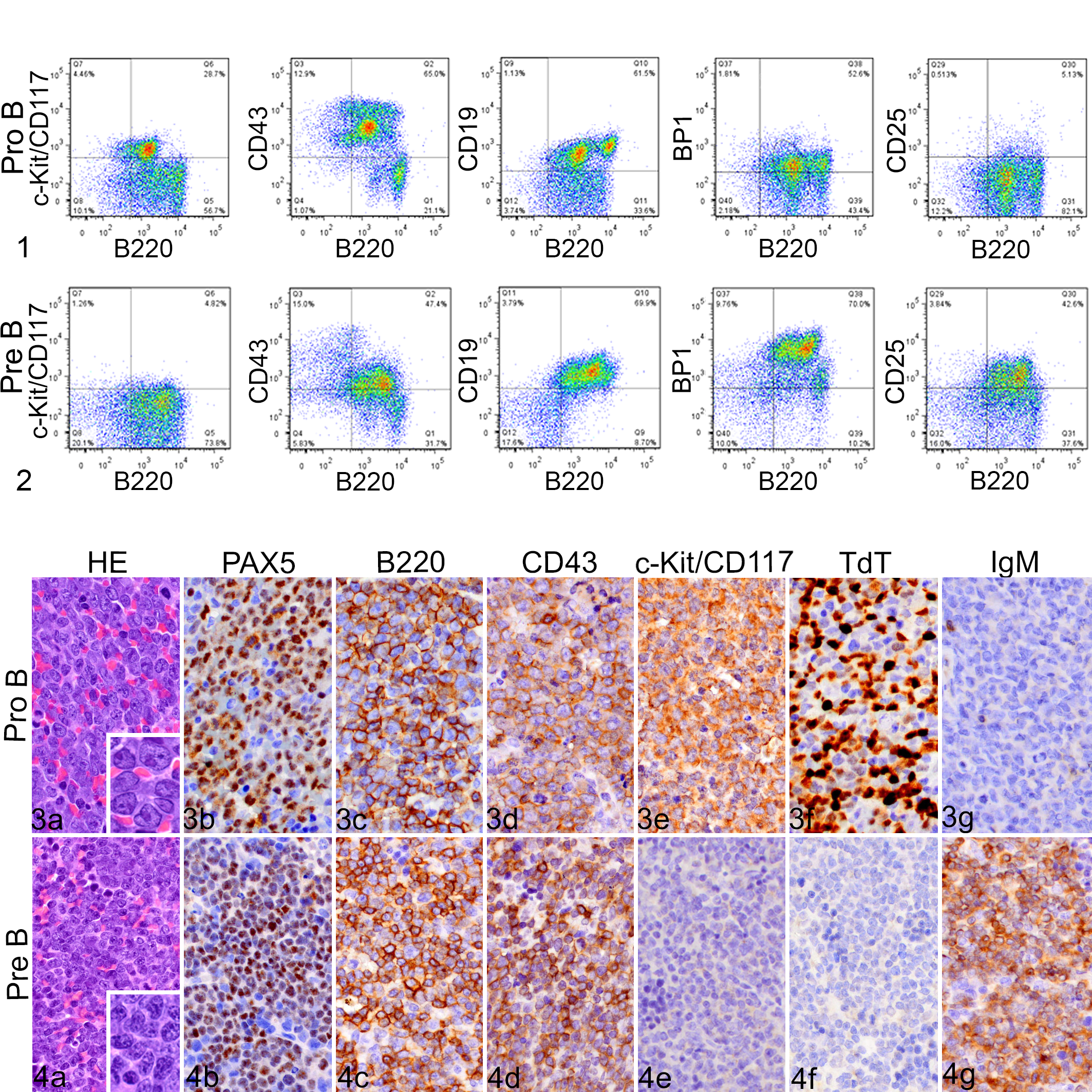
Representative flow cytometry panels for pro-B- and pre-B-cell leukemia/lymphoma. Both types are positive for B220, CD43, and CD19.
Results
All samples were negative for surface IgM by flow cytometry and for KLC by IHC; therefore, all were from cases of B-ALL/LBL (pro-B or pre-B-ALL/LBL) and none were transitional/immature B cell. Table 2 shows the expression data for all markers assessed by flow cytometry and/or IHC, along with the stage designation of pro-B or pre-B as determined independently by each modality. Of the 30 cases analyzed, the diagnosis in 18 cases (60%; case Nos. 1–18) was consistent for flow cytometry and IHC, and all 18 of these cases were diagnosed as pro-B. Of the 12 cases for which the flow cytometry and IHC results were discordant, 6 (case Nos. 19–24) were classified as pre-B by flow cytometry but as pro-B by IHC, and 6 (case Nos. 25–30) were classified as pro-B by flow cytometry but as pre-B by IHC. The main cause of discordance between results was the definition of which marker represented a transition from the pro-B to the pre-B stage. As described above, in the earlier study, 13 the pre-B stage was defined by CD25 expression in flow cytometry evaluation, whereas in the present study, the pre-B stage was defined by IHC detection of cytoplasmic μIgM expression. Another important flow cytometry and IHC marker used to differentiate the pro-B and pre-B stages is c-Kit/CD117, which is generally present in the pro-B stage but no longer expressed when cells enter the pre-B stage (Table 1). In the 6 cases (case Nos. 19–24) that were positive for CD25 (pre-B by flow cytometry) but negative for IgM (pro-B by IHC), cells were positive for c-Kit/CD117 by IHC in 4 cases and by flow cytometry in 5 cases, which was consistent with a lack of transition to the pre-B stage. Similarly, 5 of the 6 cases (case Nos. 25–30) that were negative for CD25 (pro-B by flow cytometry) and positive for IgM (pre-B by IHC) were negative for c-Kit/CD117, which was consistent with the cells having undergone a transition to the pre-B stage.
Subtyping of Murine Precursor B-Cell Lymphoblastic Leukemia by Immunophenotyping With FC and IHC.
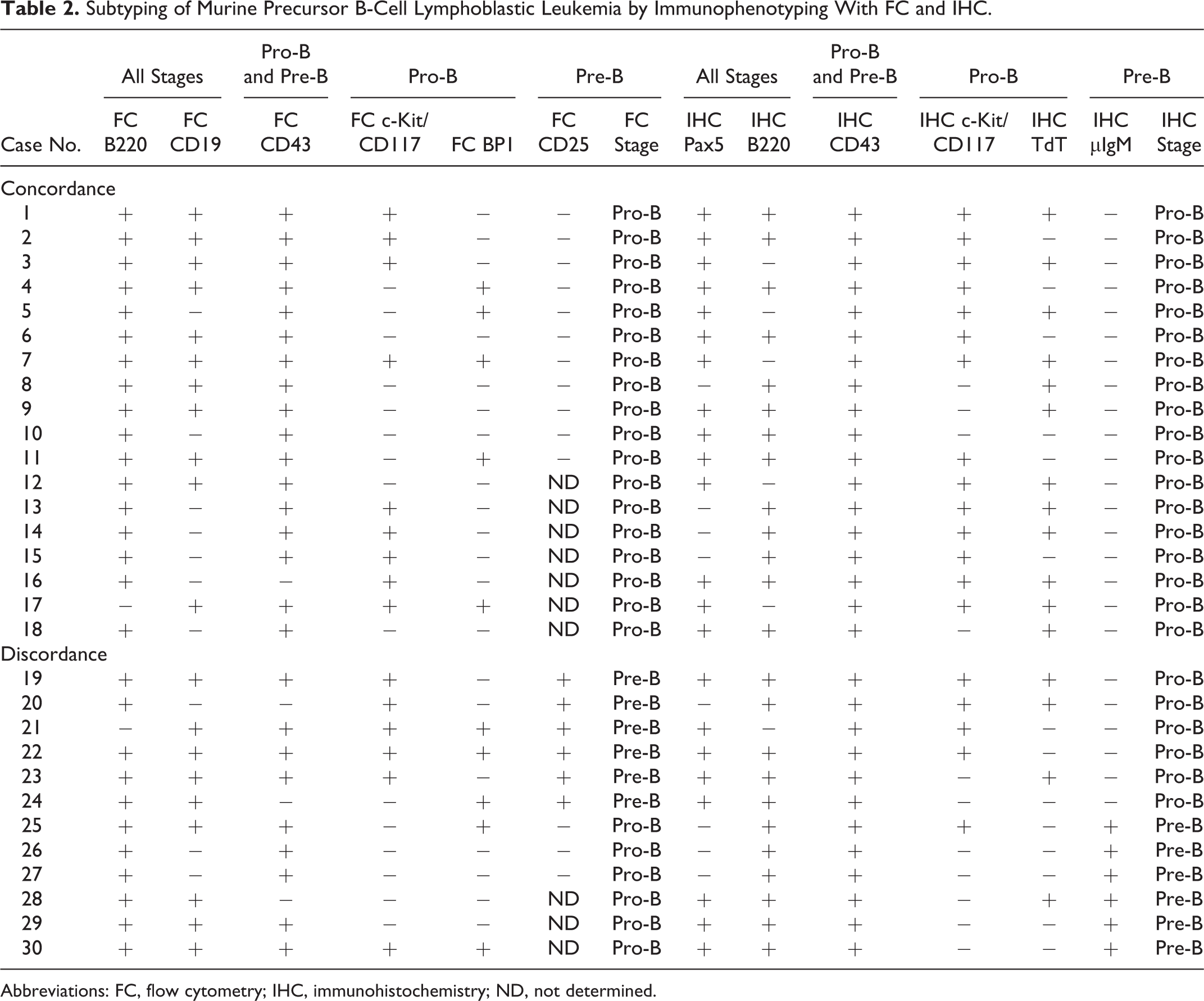
Abbreviations: FC, flow cytometry; IHC, immunohistochemistry; ND, not determined.
IHC for PAX5 was negative in 6 cases (case Nos. 8, 12, 13, 20, 25, and 27), indicating a loss of heterozygosity. All of these cases were confirmed to have a deletion of the WT Pax5 allele, as assayed by array-based comparative genomic hybridization. 13 Interestingly, 10 of the 30 cases were negative for CD19 by flow cytometry, and all of these cases either were negative for PAX5 by IHC or had a mutation in Pax5. 13 This is consistent with the fact that CD19 is entirely dependent on PAX5 for its expression. 38,39
Discussion
Profiling the stage of maturation of genetically engineered mouse models of B-cell leukemia/lymphoma is important to ensure that they faithfully reproduce the human disease, and it has potential implications for determining the cells of origin. Such profiling has demonstrated the accuracy of mouse models of common translocations that occur in B-ALL/LBL, such as ETV6-RUNX1, 59 MLL-AF4, 27 and PAX5-ELN. 24
It should be noted that in the present study, the leukemia was induced in WT and Pax5+/− mice. The Pax5 heterozygosity may have affected the phenotype of the resulting leukemias, at least in the 6 cases of the 30 examined in which loss of PAX5 was demonstrated by IHC. PAX5 influences leukemia development in 2 main ways. The less common of these is when there is a loss of heterozygosity, resulting in the absence of PAX5, as occurred in these 6 cases. This situation has been demonstrated in humans; a heterozygous germline variant of Pax5 has been reported, whereby B-ALL/LBL develops when there is a loss of the 1 WT allele. 53 Loss of PAX5 expression would result in alteration in the expression of a large number of genes. In murine WT B cells, PAX5 was shown to repress 110 genes, and when this repression is lost in Pax5–/– B cells and common lymphoid progenitors, they display promiscuous expression of lymphoid and myeloid genes. 14 Therefore, the resulting phenotypes in Pax5–/– mice may differ from those where PAX5 expression is retained. However, the more common way in which PAX5 drives leukemia is via its role as a tumor suppressor. In human B-ALL/LBL, monoallelic deletions are common, suggesting that PAX5 haploinsufficiency is a driver of leukemia, along with other cooperating mutations. 36,53 The findings from the original mouse Pax5+/− mutagenesis study from which the current cases were derived supported this role for Pax5 and identified recurrent cooperating mutations targeting tumor suppressors, Ras, and JAK-STAT signaling. 13 A similar study demonstrated that Pax5 haploinsufficiency cooperates with STAT5 activation to induce leukemia in mice. 21 Therefore, the resultant leukemias/lymphomas in the cases with retained expression of PAX5 had various underlying molecular mechanisms and thereby reflect the spectrum of phenotypes common to B-ALL/LBL.
To accurately confirm the fidelity of the mouse model to its human counterpart, it is necessary to define and correctly detect the key markers of the developmental stages. As stated earlier, many of the antigen markers used to define stages in B-cell development differ between mice and humans and therefore cannot be compared directly; 16 what is directly comparable is the stage of maturational arrest (ie, whether the cells are at the pro-B or the pre-B stage), although determining this can be challenging. As previously discussed, multiple models have been described for delineating the progressive stages of B-cell development in the mouse. The initial model, the Hardy or Philadelphia classification/nomenclature system, separates the stages into fractions of cells named alphabetically from A to F. 17 Another major model, the Basel classification/nomenclature system, identifies stages termed pre-pro B, pro-B, pre-BI, large pre-BII, and small pre-BII. 50 A third model by Osmond et al 41 uses a simpler scheme that subdivides pro-B into early, intermediate, and late and pre-B into large and small. The best approach is probably one in which these disparate systems are integrated, which is what was undertaken in the current study and outlined in Table 1. For the purposes of this discussion, the Osmond nomenclature is used. A key distinction to be made is in the transition period from the pro-B to pre-B stage. The Hardy/Philadelphia system was established based on the expression of CD43, CD24, and BP1, whereas the Basel system uses CD43, c-Kit/CD117, and CD25. A strategy based on a combination of these schemes allows for the distinction of the pro-B and pre-B stages by simultaneously evaluating the expression of BP1 and CD25, as BP1 expression begins in late pro-B and continues through large and small pre-B, whereas CD25 expression begins a step later in large pre-B and ends at the same time as BP1 (see Table 1). 5 Therefore, when flow cytometry is used, BP1 expression without CD25 expression indicates late pro-B, whereas simultaneous expression of BP1 and CD25 indicates pre-B.
Even when the expression of certain markers is agreed upon, it must be recognized that antigen markers are not completely stage restricted and that there is a progressive increase and decrease in their expression as B cells develop. Therefore, any assessment of the stage of a B-cell lymphoma/leukemia should be based on a broad panel of B-cell markers. Another consideration is that marker expression on leukemic cells does not always conform to the stages of normal maturation. In the present study, CD25 was often not expressed during the stage at which it is in normal development and vice versa. In a few cases, this also occurred with the expression of c-Kit/CD117 and TdT. In human leukemia, a common example of this is in pre-B leukemia, where TdT and CD34 are often expressed, whereas they are generally not present in normal human pre-B cells. 12 Interestingly, mice do not demonstrate this pattern for TdT; while similar to humans in their lack of TdT expression in normal pre-B cells, mice only rarely express TdT in leukemia/lymphoma pre-B cells, 46 as was seen in this study.
In this series of cases, the disparities between the classifications as pro-B or pre-B by flow cytometry and IHC were due to the mismatched expression of CD25 detected by flow cytometry and IgM detected by IHC. Cases in which CD25 but not IgM was expressed may be explained by the fact that in normal B-cell development, 5% to 10% of cells expressing CD25 do not express μIgM, and this subset is considered late pro-B. 49 Therefore, those cases (case Nos. 19–23) in which flow cytometry detected CD25 but IHC did not detect IgM were possibly pro-B, and this was further supported by the frequent expression of c-Kit/CD117, as detected by flow cytometry and/or IHC in these cases. This highlights the importance of recognizing this subset of cells in B-cell development (CD25+/μIgM– late pro-B cells). Therefore, perhaps CD25 expression should be depicted as beginning weak, with levels increasing in late pro-B. 37 The converse disparity was also present (case Nos. 25–27), whereby IgM was expressed but CD25 was not, perhaps because marker expression in leukemic cells does not always correlate exactly with that in their normal cell counterparts. 11,12,44 The expression of CD25 correlates with that of μIgM in normal B-cell development, but CD25 is not functionally required for μIgM expression; 51 thus, a leukemic cell might not express CD25 when μIgM is present. This was demonstrated in a study in which the conditional deletion of Ikaros, a gene encoding a critical transcription factor for B-cell development, led to the generation of leukemia in which a subset of cells expressed μIgM but not CD25. 25 Similarly, examination of B-cell development in mice with a targeted disruption of IL-7Rα Tyr449 revealed cells that expressed μIgM but not CD25. 42 In view of these studies and the present results, one should use caution when interpreting CD25 expression in mouse B-ALL/LBL studies, as CD25 is apparently not a reliable surrogate for μIgM in cases of abnormal B-cell development or proliferation. Therefore, our results suggest that when phenotyping B-ALL/LBL, one should evaluate the presence of pre-B cells by examining μIgM expression via IHC or by flow cytometry after performing membrane permeabilization.
There is reportedly some variability in the concordance between flow cytometry and IHC in human leukemia. One study focusing on B-ALL/LBL found a high concordance between flow cytometry and IHC results for the expression of CD10 and CD3 but not CD20, 2 whereas a study of chronic B-cell neoplasms found a high concordance for the expression of CD20 and CD5 but not CD23 or κ/λ light chains. 1 In a comparison of the detection of marker expression by IHC and flow cytometry in a cohort containing more varied diagnoses, including B-cell and T-cell lymphomas and benign lymphoproliferations, a higher concordance was found for the T-cell markers than for the B-cell markers, with the results for κ and λ light chains having the lowest concordance. 9 A study comparing the diagnoses reached using flow cytometry immunophenotyping vs histology/IHC in lymphoma and lymphoproliferative disease found 80% agreement in 9 of 11 disease groups, with the diagnosis of Hodgkin lymphoma being the most discordant; 8 however, this study did not compare the detection or use of specific markers.
To our knowledge, the present study is the first to compare the use of IHC and flow cytometry in the diagnosis of B-ALL/LBL in the same cohort of mice. The study demonstrates the importance of precisely phenotyping mouse models of leukemia to ensure that they faithfully reproduce the human disease. We suggest that when subtyping B-ALL/LBL in mice, an IHC panel of B220, PAX5, TdT, c-Kit/CD117, CD43, IgM (μIgM), and ΚLC should be considered. Our data comparisons also suggest that with flow cytometry, cytoplasmic IgM may be an appropriate marker if used in conjunction with the surface markers B220, CD19, CD43, c-Kit/CD117, BP-1, and CD25. The combined use of these IHC and flow cytometry panels will allow the subtyping of B-ALL/LBL as pro-B or pre-B in most instances. However, not all cases will have marker expression patterns that clearly conform to one or other of these designations, and molecular analyses may be necessary to determine underlying genetic mutations that would account for the disparity.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819852138 - Immunophenotyping of Murine Precursor B-Cell Leukemia/Lymphoma: A Comparison of Immunohistochemistry and Flow Cytometry
Supplemental Material, DS1_VET_10.1177_0300985819852138 for Immunophenotyping of Murine Precursor B-Cell Leukemia/Lymphoma: A Comparison of Immunohistochemistry and Flow Cytometry by Laura J. Janke, Charles G. Mullighan, Jinjun Dang and Jerold E. Rehg in Veterinary Pathology
Footnotes
Acknowledgements
We thank the St. Jude Animal Resource Center and the Veterinary Pathology Core for technical assistance. We also thank Keith A. Laycock, PhD, ELS, for scientific editing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was supported, in part, by grant P30C CA 21765 from the National Cancer Institute and by the American Lebanese Syrian Associated Charities (ALSAC).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
