Abstract
We identified multiple extraintestinal cystacanths during routine postmortem examination of 3 small Indian mongooses and 2 African green monkeys from the Caribbean island of St. Kitts. In mongooses, cystacanths were encysted or free in the subcutaneous tissue, skeletal muscle, or peritoneal or pericardial cavities, whereas in the monkeys, they were in the cavity and parietal layer of the, tunica vaginalis, skeletal muscle, and peritoneal cavity. Morphological, histological, and molecular characterization identified these cystacanths as Oncicola venezuelensis (Acanthocephala: Oligacanthorhynchidae). There was minimal to mild lymphoplasmacytic inflammation associated with the parasite in the mongooses and moderate inflammation, mineralization, hemorrhage, and fibrosis in the connective tissue between the testis and epididymis in 1 monkey. We identified a mature male O. venezuelensis attached in the aboral jejunum of a feral cat, confirming it as the definitive host. Termites serve as intermediate hosts and lizards as paratenic hosts. This report emphasizes the role of the small Indian mongoose and African green monkey as paratenic hosts for O. venezuelensis.
Small Indian mongooses (Herpestes auropunctatus) and African green monkeys (Chlorocebus aethiops sabaeus) are introduced species in the Caribbean region, and their populations have grown to an invasive level on the Caribbean island of St. Kitts. 1,4 During routine postmortem examination at Ross University School of Veterinary Medicine (RUSVM), we incidentally identified cyst-like structures in 3 mongooses and in 2 African green monkeys.
The 3 mongooses were trapped for another research study with an RUSVM-approved IACUC protocol (number 17.04.13). The location of the animals is illustrated in Fig. 1. Both monkeys were feral adult males trapped on the southeast peninsula of St. Kitts and brought to a quarantine facility (on August 2015 and May 2018 respectively). These monkeys were part of another research study with an approved Institutional Animal Care and Use Committee protocol (number 17.04.18). The first monkey was dewormed twice with ivermectin, had 2 negative tuberculin tests, had normal complete blood count (CBC) and blood chemistry, had no history of any disease or trauma, weighed 5.6 kg, and had a body condition score (BCS) of 4 out of 5 at the time of euthanasia (June 2017). The second monkey also had a BCS of 4 out of 5, was dewormed and vaccinated accordingly, and had no significant clinical history at the time of euthanasia (November 2018).
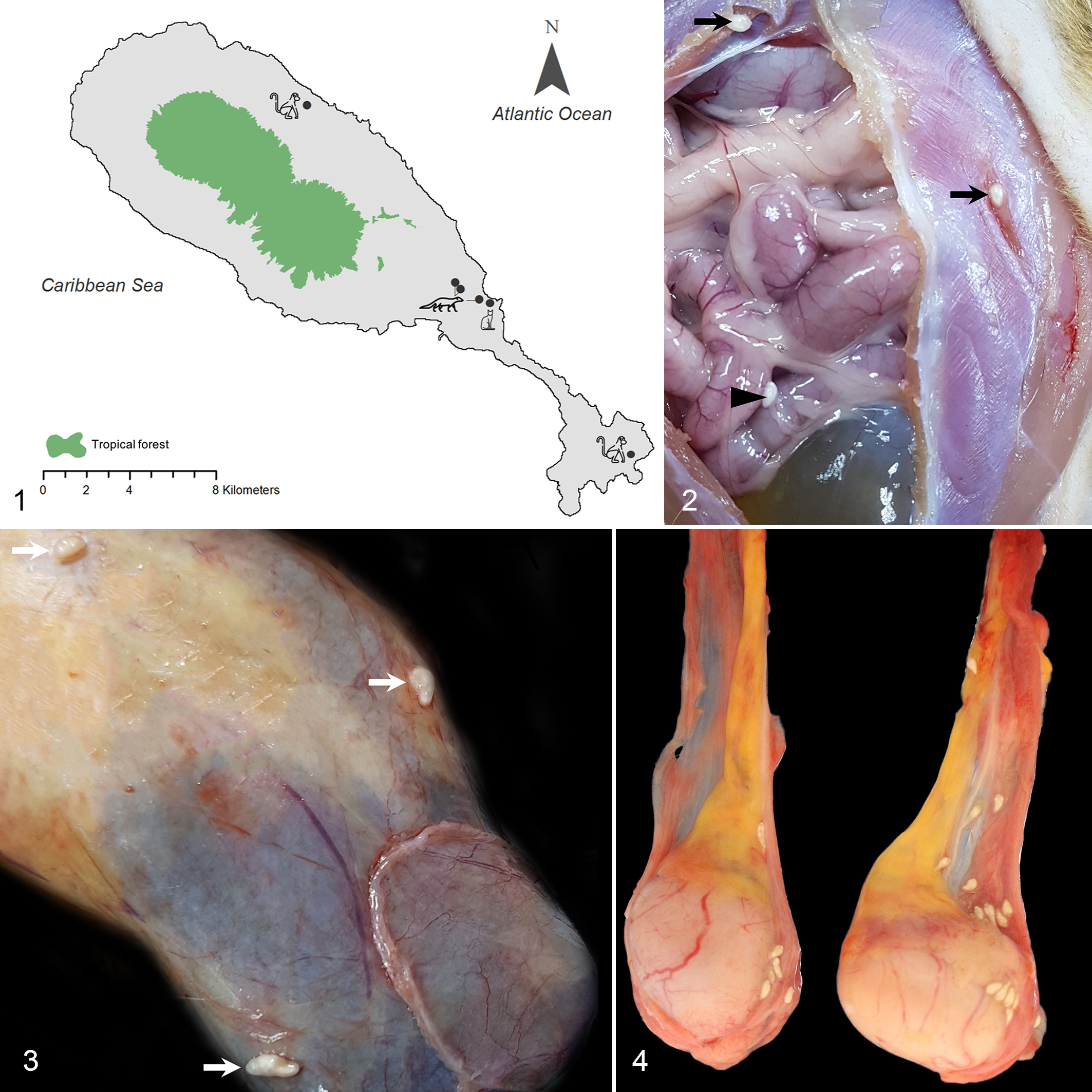
Map of St. Kitts illustrating the origin of the animals: 2 monkeys, 3 mongooses, and a feral house cat.
During postmortem examination, cyst-like structures were removed from different body locations (Table 1) and fixed in 95% ethanol. DNA extractions were performed on 3 whole cystacanth stages for each host species. For each DNA extract, polymerase chain reaction (PCR) targeting 2 regions of the ribosomal RNA (rRNA) gene were performed in triplicate following protocols described by Santos et al. 6 The partial large subunit rRNA (28 S) was amplified using the primer set C1/D2, and the internal transcribed spacer regions were amplified using the primers BD1/BD2. Amplicons of the expected sizes were gel purified and sequenced using the same primers as in the amplification PCRs. Following bidirectional sequencing, sequences were controlled, assembled, and aligned in ClustalX (http://www.clustal.org/).
Paratenic Hosts and Tissue Localization of Oncicola venezuelensis Cystacanths.

aAge was approximated.
The parasites in mongooses and monkeys were white, with a wide anterior third, measuring 4 to 5 mm long and 1 to 1.5 mm wide (Figs. 2–4). During light microscopic examination of fresh specimens, we observed the cuticle, hypodermal layer with lacunar canals, proboscis with hooks, and long ribbon-like coiled lemnisci extending close to the posterior end (Fig. 5). Some parasites were held in tap water overnight to promote evagination of proboscides (Fig. 5 inset). Histologic examination of hematoxylin and eosin (HE)–stained slides (Fig. 6) revealed the morphological features described above but also the lack of a digestive tract and a pseudocoelom, enabling the classification of the parasites as juvenile members (cystacanths) of the phylum Acanthocephala (thorny-headed worm). Encysted juvenile acanthocephalans lack reproductive organs but have all the other features of adults. 3
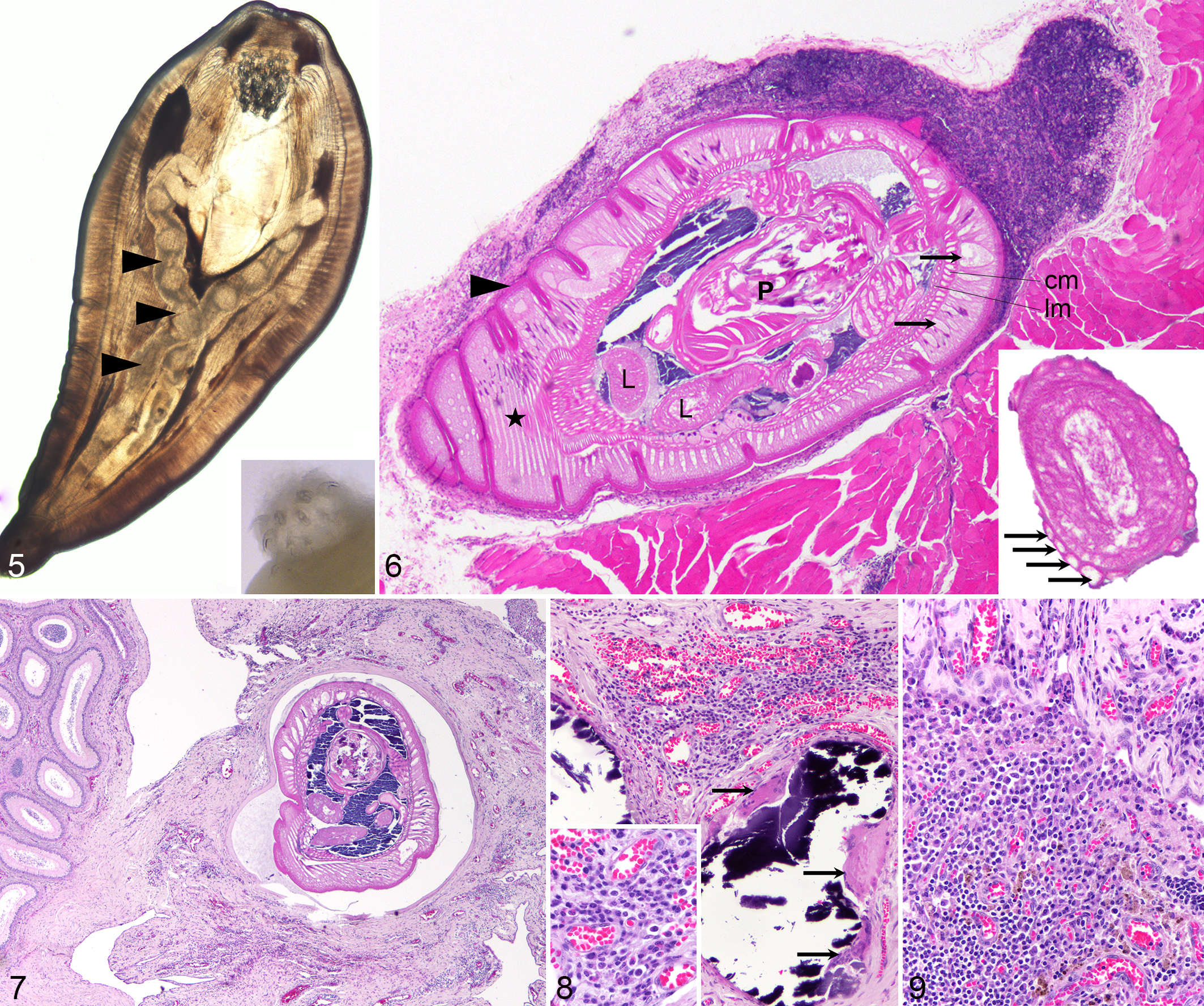
Oncicola venezuelensis cystacanths in paratenic hosts. Figure 5. Male African green monkey, case No. 4. The cystacanth has an inverted hooked proboscis and lemniscus (arrowheads). Light microscopy. Inset: everted proboscis with hooks. Stereomicroscopy. Figure 6. Mongoose, case No. 2. Cystacanth encysted in brown adipose tissue surrounding skeletal muscle. The parasite has a cuticle (arrowhead), a thick hypodermis (star) with lacunar channels (arrows), followed by a circular (cm) and a longitudinal muscle layer (lm), bordering a pseudocoelom, with lemnisci (L) and a retracted proboscis (P). A lymphoid nodule is visible in the apical part surrounding the parasite. Hematoxylin and eosin (HE). Inset: lemniscus, surrounded by compressor muscles (arrows). HE. Figures 7–9. African green monkey, case No. 4. HE. Figure 7. A viable cystacanth is in a pseudocyst, within connective tissue next to epididymis. There is mild to moderate inflammation, congestion, and fibrosis of the adjacent tissue. Figure 8. The connective tissue between testis and epididymis contains 2 foci of mineralization, close to the viable cystacanth, with multinucleated giant cells (arrows), surrounded by locally extensive accumulation of lymphocytes, plasma cells, macrophages, and eosinophils with hemorrhage and congestion. Inset: detail of inflammatory cells. Figure 9. Further away from the viable cystacanth, there is mesothelial hyperplasia of the tunica vaginalis (upper third of image) and infiltration with lymphocytes, plasma cells, and fewer macrophages. Hemosiderin is multifocally present.
The cystacanths were located intraperitoneally or within or adjacent to skeletal muscle in the 3 mongooses and in 1 monkey (Suppl. Fig. S1). They infiltrated the brown adipose tissue (mongooses) or white adipose tissue (monkeys; Suppl. Fig. S2) surrounding muscle fascia and were surrounded by minimal to mild inflammation, consisting of lymphocytes with fewer plasma cells, sometimes forming organized lymphoid aggregates (Fig. 6). In the monkey, some degenerated cystacanths were seen within skeletal muscle, suggesting chronicity, with foamy macrophages surrounding one of the cystacanths. Similar changes were seen for the cystacanths in the omentum (intraperitoneal). Most of the cystacanths that seemed grossly to be located intramuscularly were histologically observed to be within adipose tissue adjacent to the skeletal muscle, attributed to the parasite taking the path of less resistance when migrating. Mild multifocal skeletal muscle atrophy surrounding the pseudocysts was also observed. In the monkeys, however, the spectrum of changes was more severe, with cystacanths found in the testicular vaginal tunics (Figs. 3, 4), in the peritoneal cavity, and in skeletal muscle fascia. Some cystacanths of Oncicola venezuelensis were free in the cavity of the tunica vaginalis (cavum vaginale), and some were encysted between the tunica vaginalis and tunica albuginea, with minimal or no associated inflammation. However, in 1 monkey, the connective tissue between the testis and epididymis was expanded, suggestive of a linear parasitic tract, with the presence on one end of a viable larval acanthocephalan, surrounded by a pseudocyst (Fig. 7). A nearby area showed tissue necrosis and mineralization, associated with multinucleated giant cells (Fig. 8), and a more distal area had fibrosis, chronic inflammation, and hemosiderin free or within macrophages (Fig. 9). Overall, the connective tissue in this area was expanded by mild to moderate and occasionally nodular infiltrates of lymphocytes and plasma cells, with fewer macrophages and eosinophils, surrounded by multifocal congestion and hemorrhage. There was focally extensive mesothelial hyperplasia of the tunica vaginalis, in the parietal and visceral layers.
For the internal transcribed spacer (ITS) regions, 736 bp of identical sequence data was generated for cystacanths from both the mongoose and monkeys hosts. This contiguous sequence had >99% identity to O. venezuelensis Acantocephala: Oligacanthorhynchidae (KU521566) in the DNA databases. Large subunit 28 S rRNA sequences (∼747 bp) were also identical between cystacanths from both mongooses and monkeys and confirmed a very high identity (>99%) to previous reports of O. venezuelensis (KU521567). 6 All sequences generated from this study have been submitted to GenBank under the accession numbers MK377340 to MK377344.
Adult acanthocephalans parasitize the intestinal tract of different vertebrate species. A domestic cat and ocelots (Leopardus pardalis) were previously reported to be definitive hosts for Oncicola spp. (with a small intestinal location), and birds, mongooses, or lizards (Anolis spp.) serve as paratenic hosts. 2,6 The spined (hooked) proboscis is everted and used by adults for attachment to the intestinal wall. This is followed by absorption of nutrients through the body wall and lacunar channels in the hypodermis. Embryonated eggs develop from ova in the “egg balls” within the pseudocoelom and are released by adult females in the feces, subsequently ingested by invertebrate intermediate hosts where they encyst to form a cystacanth. Upon ingestion of an infected intermediate host or paratenic host by the definitive host, the cycle is completed. 3,8
Monkeys and mongooses will eat insects as part of their diet. Cockroaches 7 and termites 5 are known intermediate hosts for acanthocephalans and could easily represent part of the diet for mongooses and monkeys in St. Kitts, as previously proven for similar environments in the Caribbean. 2 Anolis lizards can also represent part of the diet for monkeys, mongooses, and domestic cats in the area. Cystacanths of O. venezuelensis were previously detected in subcutaneous nodules of Anolis lizards, which represent paratenic hosts. 6
Similar to other islands in the Caribbean, there are a high number of feral cats on St. Kitts. Intestinal acanthocephalans were not previously reported in cats in St. Kitts. Around 100 cats were autopsied over the past 3 years at RUSVM, with 38 of them being part of a study specifically investigating intestinal lesions and parasites. In one of these cats (adult, female, spayed, tabby, domestic shorthaired), we found in the aboral part of the jejunum, firmly attached to the mucosa, a 7-mm-long white parasite identified as a mature male O. venezuelensis based on morphology and histology (Suppl. Figs. S3, S4). This confirms the previous report that the feral house cat is a definitive host for O. venezuelensis in the Caribbean. 2 Even though it was previously hypothesized that the mongoose could represent a definitive host for O. venezuelensis, the current paradigm is that extraintestinal infections in mongooses are likely incidental. 2 The presence of intramuscular, intraabdominal, or pericardial cystacanths in mongooses is most likely due to the consumption of Anolis lizards or termites, which are known intermediate hosts. 5
There is a previous report of multiple encysted larval acanthocephalan parasites in the testis/tunica vaginalis in 1 African green monkey that originated from St. Kitts and was exported to a research facility in the United States. 8 The parasites were not identified but shared the location as well as many morphological features with our cases. There was no inflammation associated with the juvenile acanthocephalans in the previous report. Interestingly, the testicular location, reported in our cases and in the previous report, suggest a possible site predilection for the juvenile forms of acanthocephalans in African green monkeys.
In this study, we describe lesions associated with extraintestinal cystacanths of O. venezuelensis in small Indian mongooses and 2 African green monkeys. In the jejunum of the cat, we detected an adult male O. venezuelensis. Although generally mild in the mongoose, the spectrum of tissue changes in the monkeys could potentially result in an impairment of the reproductive function, due to the local effects of chronic inflammation and fibrosis. It remains to be proven if cystacanths of O. venezuelensis in monkey have a predilection site for the testes/tunica vaginalis.
Supplemental Material
Supplemental Material, DS_JVDI_10.1177_0300985819848502 - Extraintestinal Acanthocephalan Oncicola venezuelensis (Oligacanthorhynchidae) in Small Indian Mongooses (Herpestes auropunctatus) and African Green Monkeys (Chlorocebus aethiops sabaeus)
Supplemental Material, DS_JVDI_10.1177_0300985819848502 for Extraintestinal Acanthocephalan Oncicola venezuelensis (Oligacanthorhynchidae) in Small Indian Mongooses (Herpestes auropunctatus) and African Green Monkeys (Chlorocebus aethiops sabaeus) by Anne A. M. J. Becker, Sreekumari Rajeev, Mark A. Freeman, Amy Beierschmitt, Victoria Savinon, Judit M. Wulcan and Pompei Bolfa in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to David Hilchie, histology lab manager, for the excellent histology slides; K. C. Hill and A. Knapp for trapping and performing the autopsies of mongooses; and Katalina Zuniga-Brenes for kindly donating one of the monkeys to RUSVM Pathology services.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
