Abstract
Salmonella is a major foodborne pathogen and pork is one of the main sources of human salmonellosis. Understanding the pathogenesis and progression of the infection within the host is of interest to establish potential approaches to control the disease in pigs. The present study evaluates factors such as intestinal colonization, fecal shedding, and pathogen persistence by 2 studies using experimental challenge with Salmonella Typhimurium in weaned pigs and euthanasia at different time points (1, 2, and 6 and 2, 14, and 30 days postinfection [dpi], respectively). Histopathology of intestine at early time points (1 dpi and 2 dpi) showed severe damage to the epithelium together with an increase in polymorphonuclear cells and macrophages (P < .001), particularly in jejunum and ileum. Large quantities of Salmonella were detected within the contents of the ileum, cecum, and colon in early infection. Salmonella could also be observed in the medulla of tonsils and mesenteric lymph nodes. From 6 dpi onward, signs of recovery were observed, with progressive restoration of the epithelium, reduction of the inflammatory infiltrate, and elimination of Salmonella from the mucosa. Concentration of Salmonella in feces and ileum content decreased, but shedding did not cease even at 4 weeks after infection. Persistence of the bacteria in mesenteric lymph nodes was identified within the connective tissue at 14 and 30 dpi. Our results demonstrate a recovery of the disease after an initial acute phase but also show persistence within the lumen and surrounding lymphoid tissue. These findings are relevant to developing effective control strategies.
Keywords
Human salmonellosis is one of the most important foodborne zoonoses. According to the last report from the European Food Safety Agency (EFSA), Campylobacter jejuni and Salmonella spp. were the leading causes of human foodborne infections in the European Union (EU), with more than recorded 340 000 cases of these diseases. 12 After a decade of continuous decrease in the reported cases of human salmonellosis, European figures show a change in this trend, with an increase of 7000 cases from 2013 to 2015. 13 The success in control programs in poultry contrasts with the scarce results obtained in some programs established in swine. 24 Thus, as a consequence of the reduction in the incidence of salmonellosis associated with consumption of contaminated chicken and eggs, the relative number of human cases attributed to pork has increased in the past years. 21,22
Swine are a natural host for Salmonella. 6 Except in infections caused by Salmonella Choleraesuis, the host-adapted serovar to swine, 17 most Salmonella infections in pigs are mostly asymptomatic or cause a self-limiting diarrhea. 6 Among serovars affecting swine, Salmonella Typhimurium stands out. This is not only the most prevalent serovar on swine farms within the European Union (EU), 12,13,16 but multidrug resistance is also a common and frequent feature of swine S. Typhimurium isolates. 4 Last but not least, a relevant number of the human salmonellosis cases and outbreaks caused by S. Typhimurium are directly related to pork consumption. 12 These figures concern public health institutions and encourage research on this serotype.
The transmission of the bacteria occurs via the fecal-oral route. 26 After surviving extreme acidic conditions within the stomach, 1 the bacterium invades the small intestine. 10 Within the small intestine, Salmonella must also overcome challenging conditions such as low concentrations of oxygen, elevated concentrations of bile salts, osmolytes, and commensal bacterial metabolites. 1 The invasion is believed to occur mainly in the ileum. 9 After invasion of ileal epithelium, Salmonella is internalized by resident macrophages, reaches the subepithelial dome of the Peyer’s patches, and rapidly disseminates to mesenteric lymph nodes (MLNs), 34 where a combination of both inflammatory and cellular responses is able to stop the infection. 27,42 It is important to highlight the short period of time (less than 24 hours) required for Salmonella to infect pigs and be shed in high concentrations in feces. 5,20 The concentration of the pathogen remains high in feces during the first days and decreases or ceases after a few weeks. 23,25,38,44 It is hypothesized that a high proportion of infected pigs develops a carrier state, with the pathogen remaining in a latent state in certain organs such as tonsils and mesenteric lymph nodes. This persistence within the host is a key phase of the pathogen’s life cycle and also represents a challenge for disease control. 2 Indeed, little is known about how the pathogen survives for long periods of time within the host. 35,39 Moreover, the clear and precise data about fecal elimination contrast with the scarce information about the evolution of the infection in the intestine and surrounding lymphoid tissue. 5,14,24 With the aim of improving knowledge about intestinal colonization, fecal shedding, and carrier status of infected pigs, this study evaluates changes in the intestine and target lymphoid tissue (tonsils and MLNs) in a S. Typhimurium infection model in swine.
Materials and Methods
Experimental Infection and Sample Collection
Two challenge replicates (replicates A and B) were carried out. In each replicate, 16 commercial crossbred (Landrace × Large white) weaned pigs of approximately 4 weeks of age were included. Pigs were obtained from a Salmonella-free herd and moved into the isolation facilities of the University of León (Spain). All pigs were allocated in the same loose-housed box adapted to holding pigs. Box characteristics included solid concrete floor, a nipple drinker, and a feeder. A regular pelleted feed diet for weaners, free of any antimicrobial, was provided ad libitum. Temperature and room humidity were controlled and monitored on a daily basis (around 28°C and 65% humidity). Pigs were acclimated for 7 days before challenge. Two hours prior infection, 4 pigs were randomly chosen and euthanized and necropsied (control group). In both replicates, the remaining 12 pigs were orally challenged with 5 ml of broth containing 108 colony-forming units (CFU)/ml of S. Typhimurium phage type DT104, obtained from feces of naturally infected pigs (field isolate). In replicate A, 4 pigs per time point were euthanized and necropsied at 1, 2, and 6 days postinfection (dpi). In replicate B, 4 pigs per time point were euthanized and necropsied at 2, 14, and 30 dpi. During the period of study, pigs were monitored by rectal temperature and level of fecal consistency (1, normal feces; 2, soft feces; 3, watery diarrhea; 4, bloody diarrhea). Feces were also collected by rectal stimulation for Salmonella detection (once daily in replicate A; in replicate B, every second day during the first 2 weeks and every third day in weeks 3 and 4).
Information about collected samples can be accessed in Supplemental Table S1. Three sections of 10 cm each were cut from each intestinal location: distal jejunum (approximately 80 cm from ileocecal junction), distal ileum (including tissue near the ileocecal junction), tip of the cecum, and proximal colon (again taking the ileocecal junction as reference). Mesenteric lymph node (10 g) was obtained from near the ileocecal junction. Tissues collected for histological processing 10 were immediately fixed in 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) and embedded in paraffin. Feces and contents from different segments of the intestine (ileum, cecum, and colon) were stored at 4°C for microbiological analysis or frozen in liquid nitrogen for molecular quantification of Salmonella.
All procedures involving animals were performed in accordance with the European regulations regarding the protection of animals used for experimental and other scientific purposes, under the supervision of the Ethical and Animal Welfare Committee of the University of León, Spain.
Detection and Quantification of Salmonella in Feces
Salmonella detection was performed using the Annex D of the ISO-6579/2007 as previously described. 3 Fresh feces (25 g) were used for the preenrichment step of the method. One isolate per positive culture was further characterized by serotyping using the slide agglutination method to confirm the presence of the strain used in the challenge.
Samples of DNA from the frozen feces and contents were extracted using TRIsure (Bioline, Madrid, Spain) according to manufacturer instructions. TaqMan quantitative polymerase chain reaction (qPCR) assays, previously described by Martins et al, 26 were used to quantify concentrations of S. typhimurium in ileum, cecum, and colon contents as well as in feces. To prepare the standard curve, DNA from a pure broth of the Salmonella strain used in the challenge study was extracted using the DNeasy Blood & Tissue Kit (Qiagen, Valencia, CA, USA). Subsequently, known concentrations of 1.0 × 105, 5.0 × 104, 1.0 × 104, 5.0 × 103, 1.0 × 103, 5.0 × 102, 1.0 × 102, 5.0 × 101, and 0 genome equivalents (GEd) per 1 μl of DNA were used to build the reference standard curve, in which 1 genome equivalent of S. typhimurium corresponded to 5.46904 fg of DNA. A 19-mer forward primer (5′-GCGCACCTCAACATCTTTC-3′), a 22-mer reverse primer (5′-GGTCAAATAACCCACGTTCA-3′), and a fluorogenic probe (FAM ATCATCGTCGACATGC MGB/NFQ) were used in the quantification assays. Twenty-five microliters of PCR reactions contained 12.5 μl IQ Supermix 2X (Biorad, Madrid, Spain), 0.4 μM of each primer, 0.2 μM probe, 1 μM MgCl2, 200 ng DNA, and 10 μl UHQ water. PCR amplifications were performed on an iQ5 Thermo Cycler (Biorad, Madrid, Spain) under the following conditions: 95°C for 10 minutes and 50 cycles of 95°C for 15 seconds and 60°C for 1 minute.
Histopathology and Immunohistochemistry Analyses
Three samples of each collected tissue were sectioned at 5 μm and routinely processed with poly-l-lysine (Sigma-Aldrich, St. Louis, MO, USA), deparaffinized in xylene, and rehydrated through graded alcohols to distilled water. Hematoxylin and eosin staining was performed as previously described. 27 Immunohistochemistry (IHC) assays were performed with an anti-Salmonella rabbit polyclonal antibody with a specificity that was previously determined and a mouse monoclonal antibody specific for porcine macrophages (clone 4E9/11) (Biorad). The antibody reactions were visualized by a standard avidin-biotin peroxidase method (Dako, Barcelona, Spain) using diaminobenzidine (Sigma-Aldrich) as chromogen substrate. 42 To assess the specificity of each reaction, negative controls (reactions lacking the primary antibody) were performed in parallel for each section stained. After IHC labeling and image acquisition, the load of Salmonella was quantified in 10 images for each preparation using the IHC profiler plugin in ImageJ. 28 The IHC profiler algorithm, which estimates the number of positive pixels in each image, was used to enumerate Salmonella and macrophages. Neutrophils were enumerated by counting each cell type in 10 randomly selected fields (400× field) from each cut processed.
Statistical Analysis
Data were introduced into R (R-project) for further statistical analysis. 19 The assumption of normality was checked using the Shapiro-Wilk test in quantitative data (rectal temperature, macrophage counts, neutrophil counts, and Salmonella labeling). Rectal temperature values throughout the course of study followed a normal distribution, and thus differences between groups were established by repeated-measures analysis of variance, using pig as a fixed factor. The nonparametric Kruskal-Wallis test with post hoc Bonferroni correction was used to estimate differences in counts of macrophages, neutrophils, and Salmonella labeling among intestinal sections and necropsy time points. Significance of statistical analysis was set at α = 0.05.
Results
Monitoring of Pigs During the Time Course of the Study
The analysis of the presence of S. Typhimurium in feces revealed that all challenged pigs shed Salmonella in their feces from 1 dpi onward. Fecal excretion of Salmonella was constant in replicate A (all pigs and all sampling days), and most of the pigs were positive in feces during the study in replicate B. Temperature over 40°C was frequent within the first days of infection in both replicates (Figs. 1, 3). The temperature varied significantly through both studies (P < .001). Similarly, fecal scoring in the monitored pigs fluctuated in both replicates (Figs. 2, 4). All data are recorded in Supplemental Tables S2 and S3.
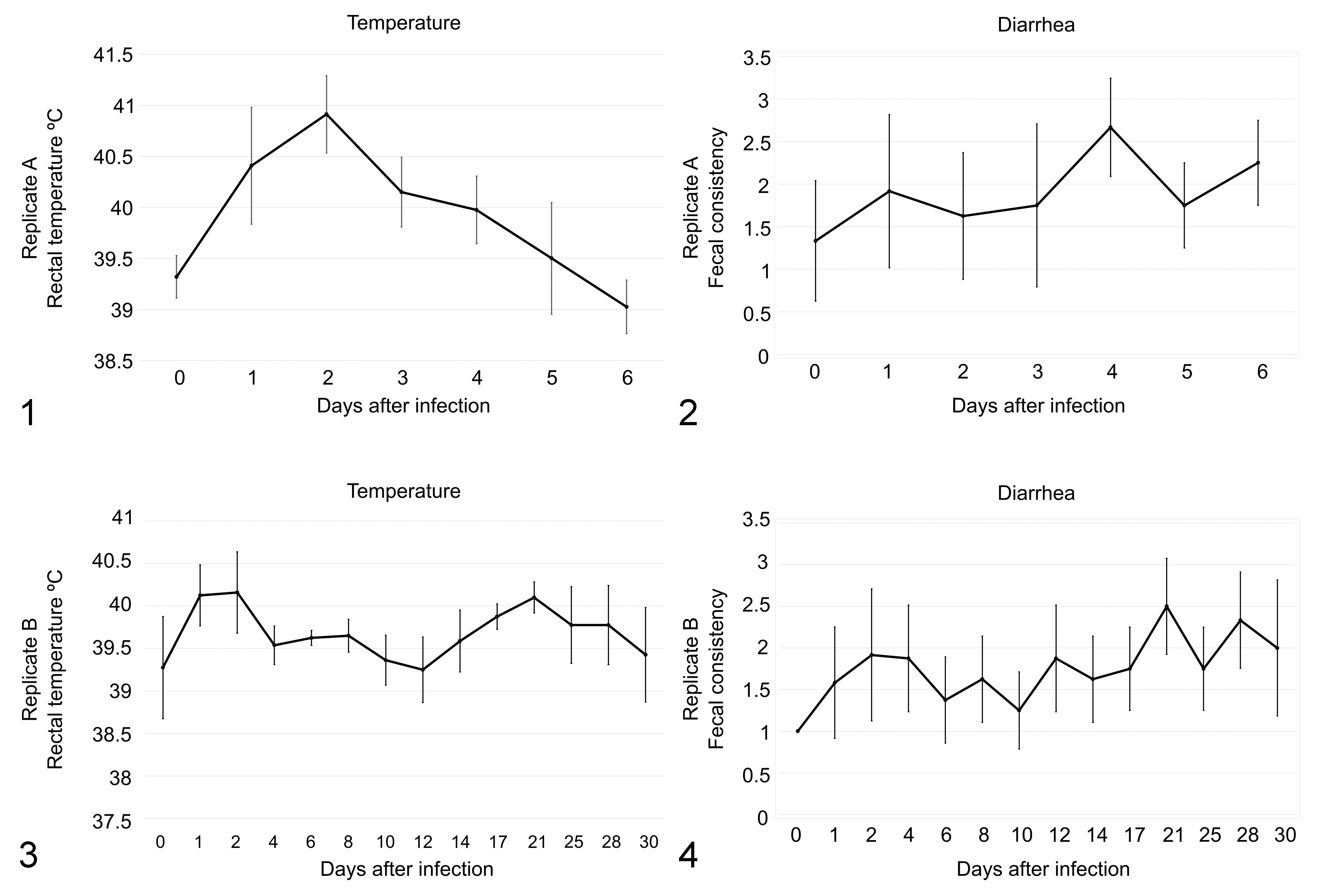
The effect of Salmonella typhimurium infection of pigs. Rectal temperature (Figs. 1, 3) was monitored at 0 to 6 days after challenge in replicate A and at 0 to 30 days after challenge in replicate B. Fecal scoring (Figs. 2, 4) was determined at the same time points for both replicates. The data show the mean and standard error of the mean. Significant changes in temperature between time points are denoted. *P < .05. **P < .01.
Location of Salmonella and Morphological Changes in the Intestine
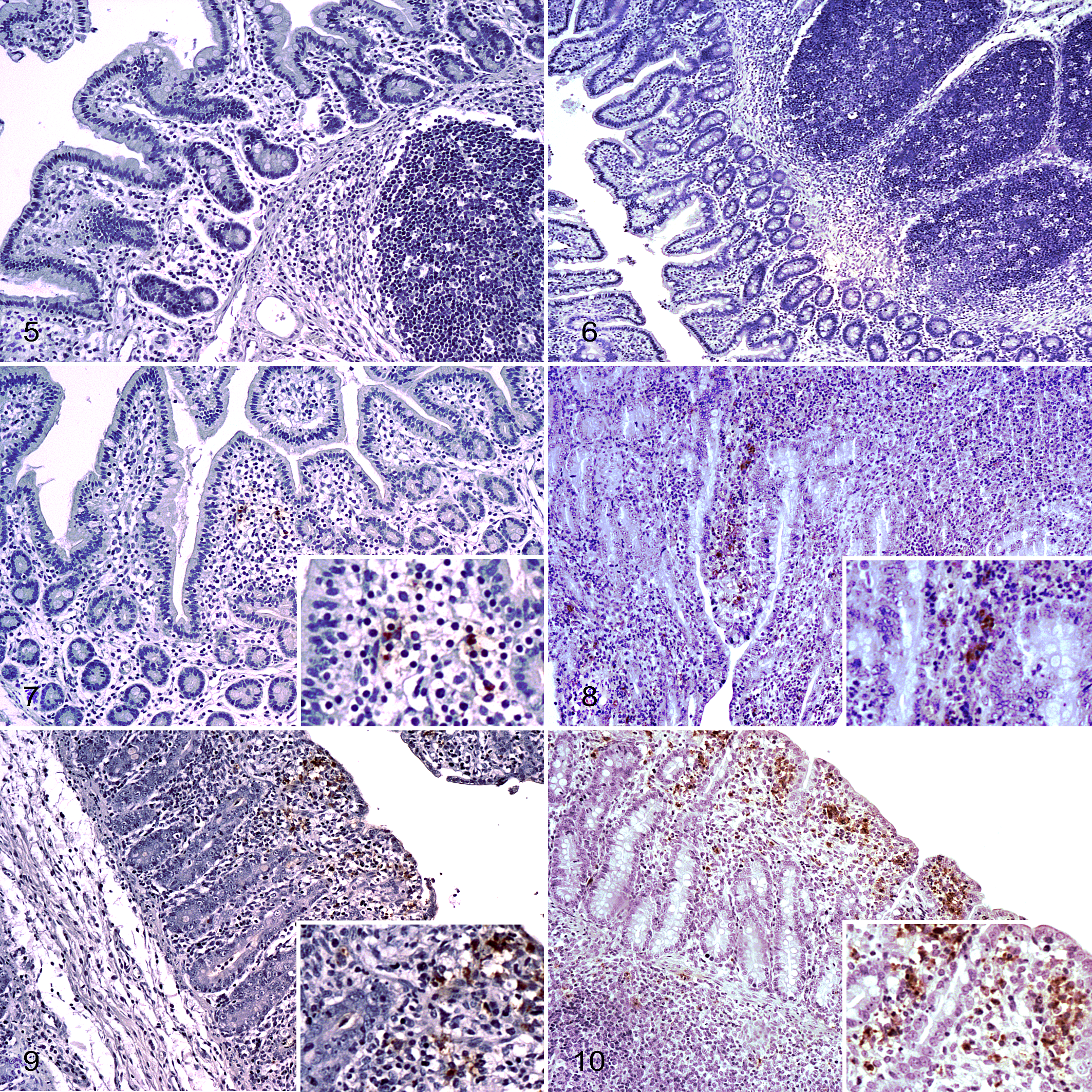
As expected, Salmonella was not observed in histological sections from control pigs (Figs. 5, 6). Reduction in the length of the epithelial villi within the small intestine (jejunum and ileum) was observed in histological sections at 1 dpi, together with the presence of Salmonella within the mucosa (Figs. 7, 8). Severe changes were observed at 2 dpi, with complete atrophy of the villi and epithelial damage in the small intestine (Figs. 9, 10). From 1 dpi to 2 dpi, we observed an increase of Salmonella labeling within the ileal mucosa (P = .025) with a similar trend in the jejunum (P = .054), but not in the colon (P = .121). Immunolabeling of Salmonella was significantly higher in the ileum compared to jejunum and colon sections at 2 dpi (P < .001). Within the ileum, Salmonella was distributed all over the epithelium, penetrating from the damaged villi through the mucosa to the submucosa, where the bacteria could be observed in the Peyer’s patches. Finally, no differences were observed in Salmonella labeling among pigs necropsied on the same day (Suppl. Table S4).
The inflammatory infiltrate was also significantly increased (P < .001) in early infected pigs (1 and 2 dpi) compared to controls (Suppl. Table S5). Indeed, the statistical analysis of neutrophil and macrophage counts among study time points showed significant differences (P < .001). No differences in neutrophil counts were observed between intestinal sections from control pigs or pigs necropsied at 1 dpi (P = .162) and 6 dpi (P = .775). In contrast, higher neutrophil counts were observed in the ileum compared to the jejunum and colon at 2 dpi (P < .001). Similarly, we observed an increase of the counts of macrophages at early infection compared to control pigs (P < .001). Counts of macrophages were higher in the ileum than in the jejunum or colon at 1 dpi and 2 dpi (P < .001).
At 2 dpi, levels of Salmonella labeling in the large intestine were lower than in the small intestine (P < .001), coinciding with lower rates of inflammatory cell infiltrate (Suppl. Tables S4 and S5). Damage in the epithelium of cecum and colon was less severe (Figs. 11, 12). In addition, Salmonella distribution was limited to the epithelium (Suppl. Table S4).
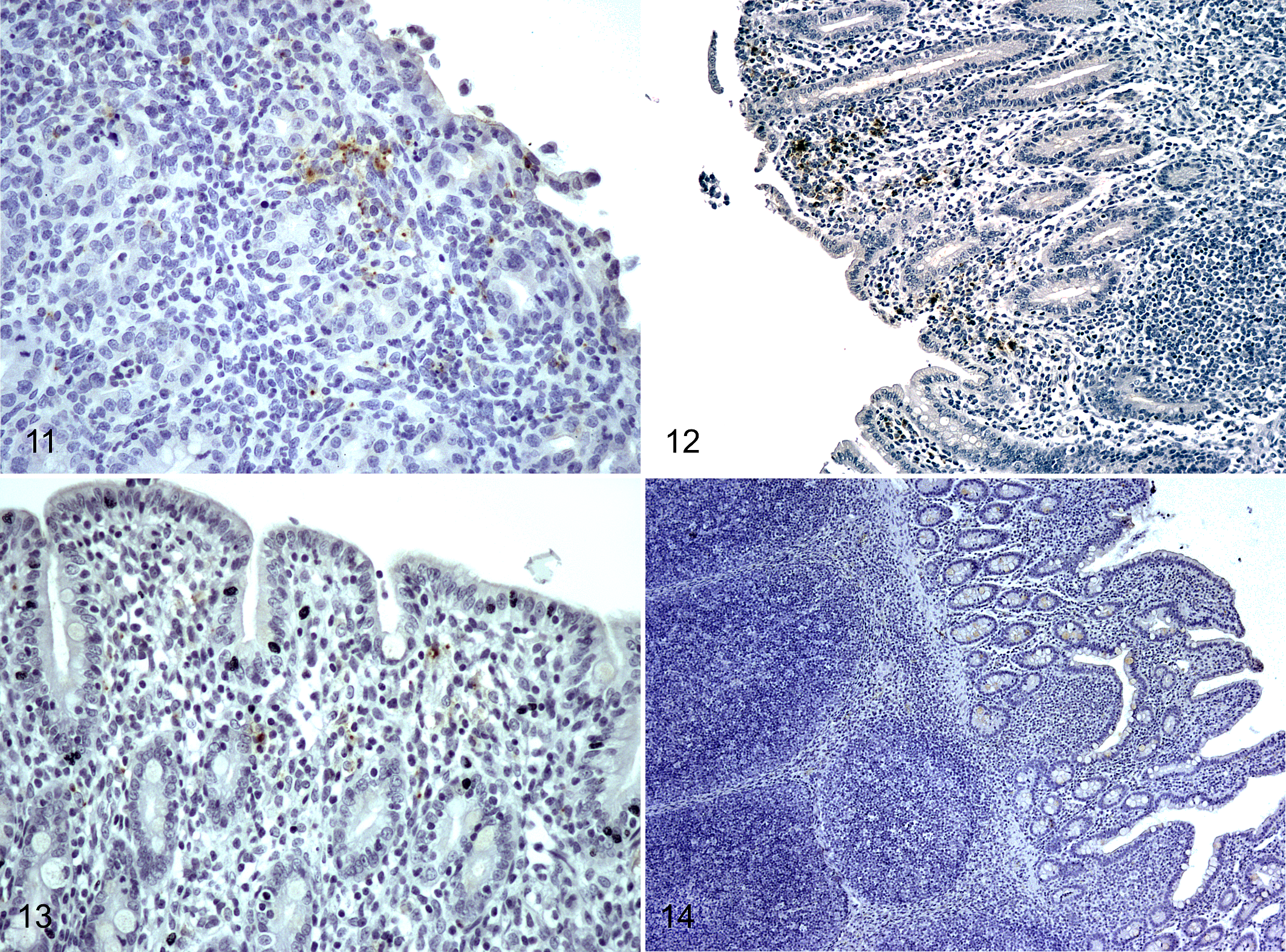
The progression of Salmonella infection was evaluated both in small and large intestine at 6, 14, and 30 dpi. Signs of initial recovery of the epithelium, such as villous development, were initially observed at 6 dpi, even though Salmonella could still be detected by IHC within the ileal mucosa (Fig. 13). It is noteworthy that the ileum was the only location where Salmonella labeling was observed at 6 dpi but substantially lower compared to early infection (P < .001; Suppl. Table S4). In fact, under the infection conditions in this study, no Salmonella labeling was detected in any intestinal section after 6 dpi. Inflammatory cell counts at 6 dpi were significantly lower than at 2 dpi (P < .01). Neutrophil counts were similar to 1 dpi pigs (P = .163), and no differences were observed in macrophage counts between 6 dpi pigs and control pigs. Lengthening of the epithelial villi and decreased inflammatory infiltrate were the main histologic changes in pigs necropsied at 14 dpi compared to earlier time points. The histologic appearance was similar at 30 dpi (Fig. 14), with complete restoration of mucosal structure.
Salmonella Infection in Secondary Lymphoid Organs
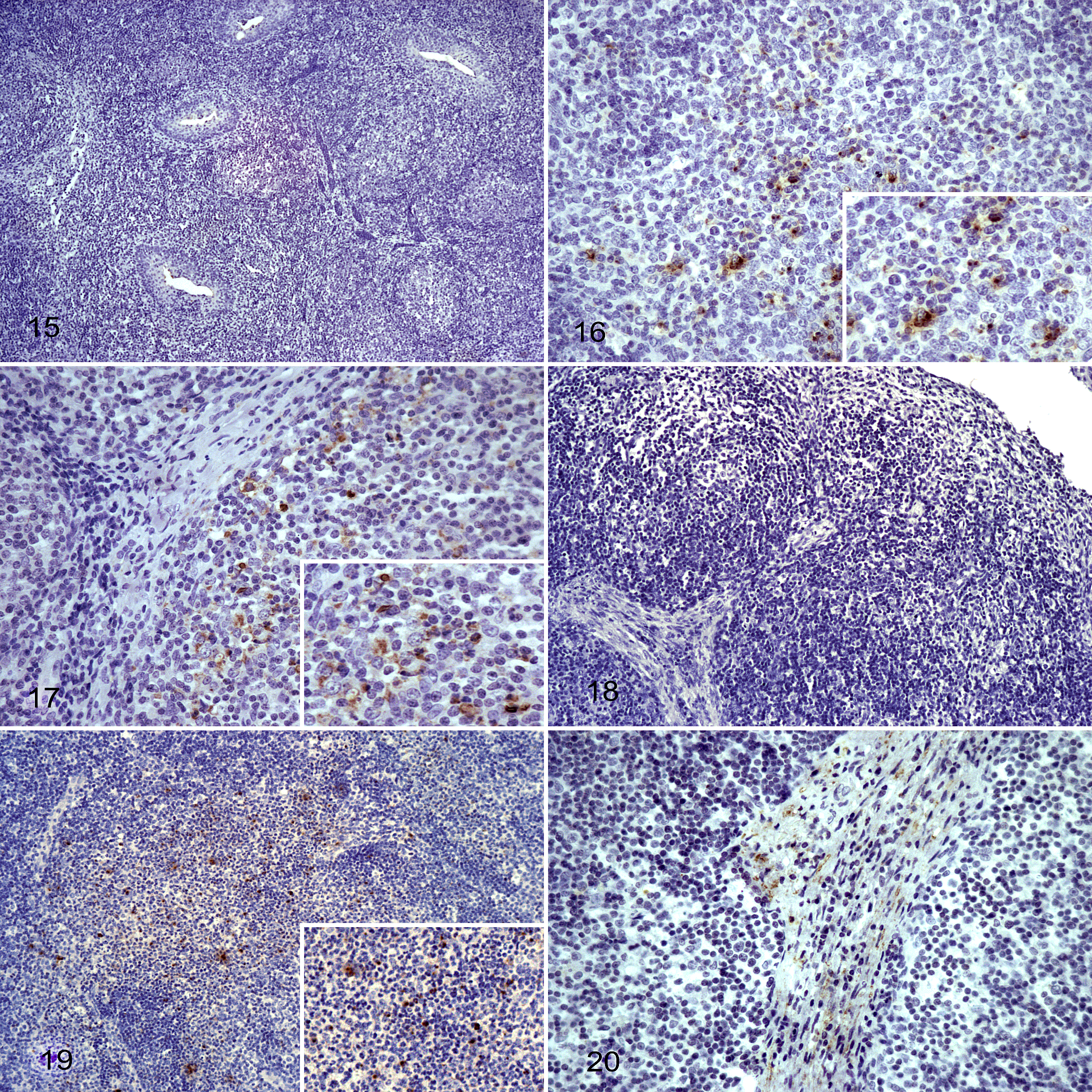
The presence of Salmonella in lymphoid tissue was evaluated by IHC in palatine tonsils and MLNs. As expected, all samples from control pigs were negative to Salmonella (Figs. 15, 18). In contrast, Salmonella was detected in tonsils and MLNs from challenged pigs at all necropsy time points. At 2 dpi, Salmonella was distributed throughout the medulla of tonsils and MLNs (Figs. 16, 19). Salmonella labeling in MLNs was significantly higher at 2 dpi (P < .001) compared to any other time point tested (Suppl. Table S4), with random distribution of the bacteria labeling in the medullary area (Figs. 16, 19). A random distribution of Salmonella was observed within the medulla of tonsils (Fig. 17) at the chronic phase of infection (14 and 30 dpi). However, in the MLNs, the bacteria were cleared from the medulla and Salmonella labeling was restricted to the connective tissue of trabeculae separating medullary sinuses (Fig. 20).
The burden of macrophages in MLNs as well as their distribution was also evaluated through the study time points (Suppl. Table S6). Interestingly, there were significant differences in immunolabeling of macrophages through the study (P < .001). Load of macrophages in MLNs from 2 dpi was significantly higher than any other necropsy time point (P < .01), except 30 dpi (P = .8). No differences were observed between control pigs and pigs necropsied at 1 dpi (P = .9), 6 dpi (P = .8), or 14 dpi (P = .6).
Detection and Quantification of Salmonella in Feces and Different Sections of the Intestine
Taqman qPCR assays were used to detect and quantify Salmonella in contents from the ileum, cecum, colon, and feces in all time points (Fig. 21). High concentrations of Salmonella, close to 105 GE per gram of digesta, were identified in all sections tested at 2 dpi. Fourteen days later, Salmonella was only quantified in relevant concentration in fecal samples (102 GE/g), while values in tissue sections were almost negligible. The concentration of Salmonella in feces remained stable until the end of study, according to the results observed in feces from pigs necropsied 30 dpi. At this time point, ileum content samples were also positive with a concentration of Salmonella of 102 GE/g. Samples from large intestine sections (cecum and colon) were negative at this time point.
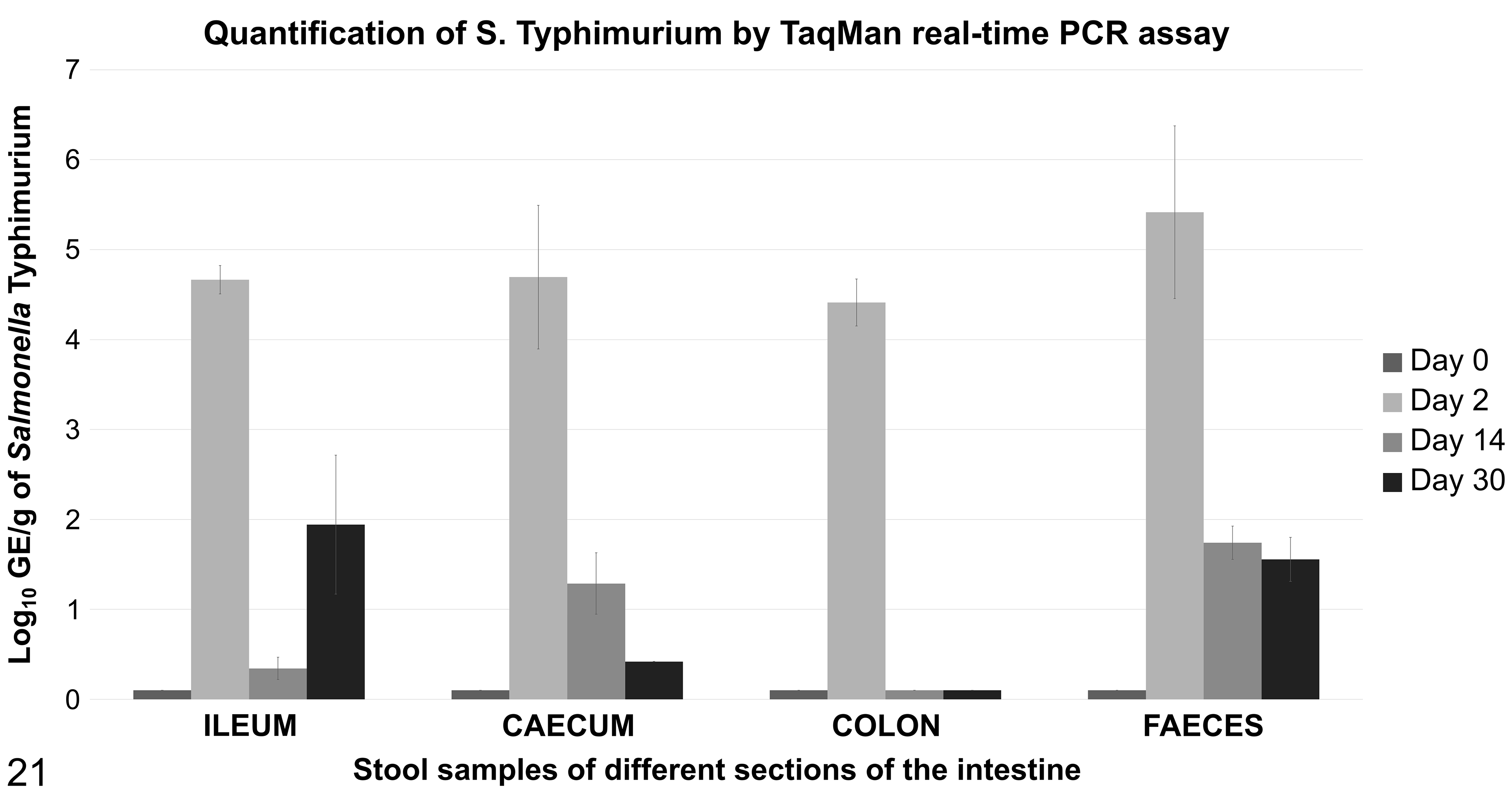
Quantification of Salmonella typhimurium by TaqMan real-time polymerase chain reaction (PCR) assay in the digesta of different sections of the intestine through the time course of the study. Log 10 genome equivalents in different locations of the gut and at different time points. Salmonella concentration decreased through time, but the pathogen could still be detected in ileum and feces at 30 days postinfection.
Discussion
Salmonella is one of the main intestinal pathogens in swine, causing not only animal disease but also a public health risk. Different research studies have investigated the epidemiology of the infection. 22,24 Their results show that, under the circumstances studied, Salmonella successfully infected the host in a few hours 14 and the pathogen was excreted in relevant concentrations on the first days after infection. 14,24 However, there are still many gaps about the pathogenesis and evolution of the infection within the intestine. By combining immunohistochemistry and microbiology techniques, we have analyzed in depth samples from 2 challenge studies with S. Typhimurium to provide new insights that help to complete this puzzle.
This study describes histological changes over time in the intestine in swine infected with S. Typhimurium. IHC results demonstrated the progressive invasion of the bacteria in the small intestinal mucosa, together with epithelial damage and atrophy of the villi within the first 48 hours of infection. The pathogen load and inflammatory cell infiltrate peaked at 2 dpi, along with severe damage of the mucosa. Presence of Salmonella, innate immune response cells (neutrophils and macrophages), and epithelial damage were particularly evident within the ileum. Our data are in line with previous works using microbiological techniques 5,14,20 or molecular biology analysis, 11 which described the distal ileum as the preferential target for Salmonella invasion. Despite this tissue tropism, 29 Salmonella could be identified in the mucosa of the cecum and colon, although in lower numbers. The lower bacterial labeling observed in the large intestine compared to the ileum could be linked to the existence of a hostile anaerobic environment for Salmonella or differences in enterocytes receptors, which may limit colonization and invasion. 10,31,32,33
The acute phase of infection, defined by clinical signs such as fever and diarrhea and shedding of high concentrations of Salmonella in feces, turns into a chronic phase, characterized by subclinical, intermittent shedding of the pathogen in feces. 20,25,45 IHC from necropsied pigs at 6 dpi (the late acute phase or early chronic phase of infection) had decreased labeling of Salmonella within the jejunum and ileum mucosa together with evidence of tissue recovery. The counts of inflammatory cells (neutrophils and macrophages) were also lower compared to early stages of the infection. This result, together with a clearance of the bacteria from the mucosa, indicates control of the infection by the host within a relatively short time frame. Complete pathogen clearance from the mucosa was observed at 14 dpi, and restoration of the villous structure to resemble the initial morphology was detected by 30 dpi.
Similar to our results of Salmonella immunolabeling within the intestinal mucosa, the concentration of Salmonella in the digesta of the different segments (as measured by qPCR) decreased throughout the period of study. Indeed, at 14 dpi and 30 dpi, the pathogen could only be detected in low concentrations in ileum and fecal samples. These results suggest the bacterium persists in the intestinal tract, even after the recovery of the intestinal wall. 7,24 The presence of Salmonella in the intestinal content without any sign of invasion of the intestinal mucosa could be associated with a flagellum expression switch, a potential strategy to remain latent and survive within the host intestine. 19,40
The function of the lymphoid tissue associated with the digestive tract is to provide a defense against ingested harmful organisms. 30,36 However, lymphoid organs, such as palatine tonsils and MLNs, have been targeted as potential locations for Salmonella carriage. 33,45 Tonsils are the first lymphoid organ exposed to ingested pathogens. Numerous studies have reported the presence of Salmonella within the tonsils after Salmonella infection, 33,45 apparently located within the connective tissue surrounding the organ. 41 Nevertheless, our histological study revealed the presence of the pathogen in the tonsillar crypts and the medullary area of the organ, regardless of the time point of study. This finding adds information to the distribution of the bacteria in the tonsil structure.
Despite the relevance of MLNs in infection persistence, little is known about how Salmonella remains alive within this organ. Within the acute phase of infection, the bacteria could be located in the medullary region of the organ. 37 Interestingly, in MLNs from pigs necropsied on days 14 and 30, we observed a lower burden of Salmonella in the organ and a redistribution of the location of the pathogen, which was restricted to the trabecular area. To our knowledge, this is the first study that reports the location of the bacteria within the MLNs at different time points. Recent studies demonstrate that Salmonella infects and survives within fibroblasts, the cell forming the connective tissue, constituting a potential reservoir for the pathogen. 15 Intestinal macrophages facilitate Salmonella survival, replication, and spread throughout the host. 18 The observed macrophages within the MLNs in acute and chronic infection are a sign of the active infection within this organ. Both macrophages and fibroblasts may be involved in the persistence of the pathogen, 18,40 acting as trojan horses where Salmonella escapes to immune surveillance and where it is hidden until conditions are more favorable to reinfect the animal. 8,39,43 Further research is needed to elucidate the target cells and potential mechanisms by which Salmonella is able to survive within the lymph nodes.
In conclusion, this study offers new insights about the time course and evolution of Salmonella infection in pigs. The results obtained show that despite the success of the bacteria to colonize and invade both the small and large intestine, the host is able to clear the infection and reestablish the intestinal barrier in a few weeks. Nevertheless, Salmonella remains in low abundance, preferentially within the small intestine lumen and in the secondary lymphoid organs, specifically in the medulla of tonsils and the within macrophages and/or fibroblasts in mesenteric lymph nodes, with the latent risk of pig reinfection and contamination of the food chain.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819843682 - Salmonella Typhimurium Infection Along the Porcine Gastrointestinal Tract and Associated Lymphoid Tissues
Supplemental Material, DS1_VET_10.1177_0300985819843682 for Salmonella Typhimurium Infection Along the Porcine Gastrointestinal Tract and Associated Lymphoid Tissues by Natividad Bellido-Carreras, Héctor Argüello, Sara Zaldívar-López, Ángeles Jiménez-Marín, Rodrigo P. Martins, Cristina Arce, Luis Morera, Ana Carvajal and Juan J. Garrido in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff from the Genetics Department at the University of Córdoba for their technical support in this study and to Edgar G. Manzanilla for his advice in the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Spanish Ministry of Economy and Competitiveness (AGL2014-54089-R). HA is a postdoctoral researcher supported by the Juan de la Cierva Postdoctoral Trainee Program of the Spanish Ministry of Economy and Competitiveness (FJCI-2016-30795). SZL was supported by a postdoctoral contract cofunded by the XXI University of Cordoba Intramural Research Program and the European Regional Development Funds (FEDER).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
