Abstract
Beta toxin (CPB) is known to be an essential virulence factor in the development of lesions of Clostridium perfringens type C enteritis in different animal species. Its target cells and exact mechanism of toxicity have not yet been clearly defined. Here, we evaluate the suitability of a neonatal piglet jejunal loop model to investigate early lesions of C. perfringens type C enteritis. Immunohistochemically, CPB was detected at microvascular endothelial cells in intestinal villi during early and advanced stages of lesions induced by C. perfringens type C. This was first associated with capillary dilatation and subsequently with widespread hemorrhage in affected intestinal segments. CPB was, however, not demonstrated on intestinal epithelial cells. This indicates a tropism of CPB toward endothelial cells and suggests that CPB-induced endothelial damage plays an important role in the early stages of C. perfringens type C enteritis in pigs.
Keywords
Clostridium perfringens is an anaerobic, Gram-positive, spore-forming bacterial pathogen of animals and humans. 6 Its pathogenesis is dependent on the production of potent exotoxins. C. perfringens type C produces 2 out of a total of 4 so-called typing toxins, alpha- (CPA) and beta toxin (CPB), but additional toxins, such as beta-2 toxin, enterotoxin, and TpeL, can be secreted. 4 It is the etiologic agent of necrotic enteritis (NE), a disease causing high morbidity and mortality in suckling piglets 7 but also affecting other mammalian hosts, such as calves, goats, horses, and humans. 1,6,8 CPB has been identified as an essential virulence factor of type C strains using 2 small laboratory animal models. 5,9 The exact mechanism of toxicity and the natural target cells of CPB are, however, not yet unequivocally defined. It was hypothesized that CPB directly causes epithelial necrosis; however, scientific proof for a direct toxic effect of the toxin on epithelial cells is lacking. We recently provided evidence that small intestinal microvascular endothelial cells could be directly targeted by CPB; however, results were based on in vitro studies and analysis of end-stage lesions of naturally affected piglets. 2,3 Experimental infections in rabbits and mice indicated that species differences in the reaction to this enteric infection exist. 5,9 The goal of this limited study was (1) to modify an experimental porcine intestinal loop infection model to reproduce early lesions of NE and (2) to determine CPB binding to potential target cells in the small intestine during early stages of NE in a naturally affected animal species.
The porcine C. perfringens type C strain JF 3721 2 , the type C reference strain NCTC 3180, and as a control, the porcine C. perfringens type A isolate JF 36932 were used. Bacteria were grown anaerobically overnight at 37°C in TGY (3% tryptic soy broth, 2% glucose, 1% yeast extract, 0.1% L-cysteine). Overnight cultures (100 μl) were added to 10 ml of TGY and grown anaerobically until early log phase (appr. 1.5 × 107 bacteria per ml determined by CFU counts from serial dilutions). Animal experiments were approved by the ethical committee of the Faculty of Veterinary Medicine, Ghent University. Three 12-hour-old and one 6-hour-old Rattlerow Seghers Pietrain crossbred piglets were obtained from AgriVet (Melle, Belgium). Sows were not vaccinated against C. perfringens type C, and no cases of NE were previously diagnosed in the herd. Intestinal loops were constructed using the procedures of Vandenbroucke et al. 10 Individual loops of approximately 2 cm length were inoculated with 500 μl of log phase cultures or 500 μl sterile TGY supplemented with 200 μg/ml trypsin inhibitor (Sigma-Aldrich). Each inoculation was performed in duplicate loops with 1 empty loop in between. Inocula and infection times are summarized in Table 1. In 1 animal (No. 4), a time course experiment for 6–10 hours was performed by reopening the abdominal suture after 2 and 4 hours and inoculation of additional loops. Piglets were maintained under anesthesia no longer than 12 hours, after which they were euthanized using T61 intravenously (0.3 ml/kg bodyweight; Intervet, Ukkel, Belgium). After euthanasia, the small intestine was immediately excised; individual loops were opened, fixed in 10% buffered formalin for 12 hours, and processed routinely for histological sectioning. Histological examinations were performed according to a standardized grading scheme. In short, capillary dilatation was scored on a severity scale of 1–5 as an average of five 400× fields, using the following guidelines: 0 = none; 1 = capillaries dilated in mucosa of necrotic villi only; 2 = capillaries dilated in mucosa underlying necrotic and nonaffected villous epithelium; 3 = capillaries dilated in mucosa and submucosa; 4 = capillaries dilated in mucosa, submucosa, and muscularis; 5 = capillaries dilated in all intestinal layers. The total necrotic area of each sample was scored as an average of all sections on each slide as 0 = no necrotic enterocytes observed; 1 = few necrotic cells focally on villous tip; 2 = multifocal villous tip necrosis, less than 10% of villi affected; 3 = multifocal villous tip necrosis, 10%–50% of villi affected; 4 = greater than 50% of villous tips are necrotic; 5 = greater than 50% of villous tips are necrotic, with deep mucosal necrosis. Immunohistochemical stainings for CPB and as control CPA (Monoclonal Antibody 10A2 [mAb-CPB], monoclonal antibody 6H7 1F3 [CPA], Center for Veterinary Biologics, Ames, Iowa) were performed as described by Miclard et al. 3
Animals, Inocula, Infection Times, and Main Macroscopic and Histologic Lesions a
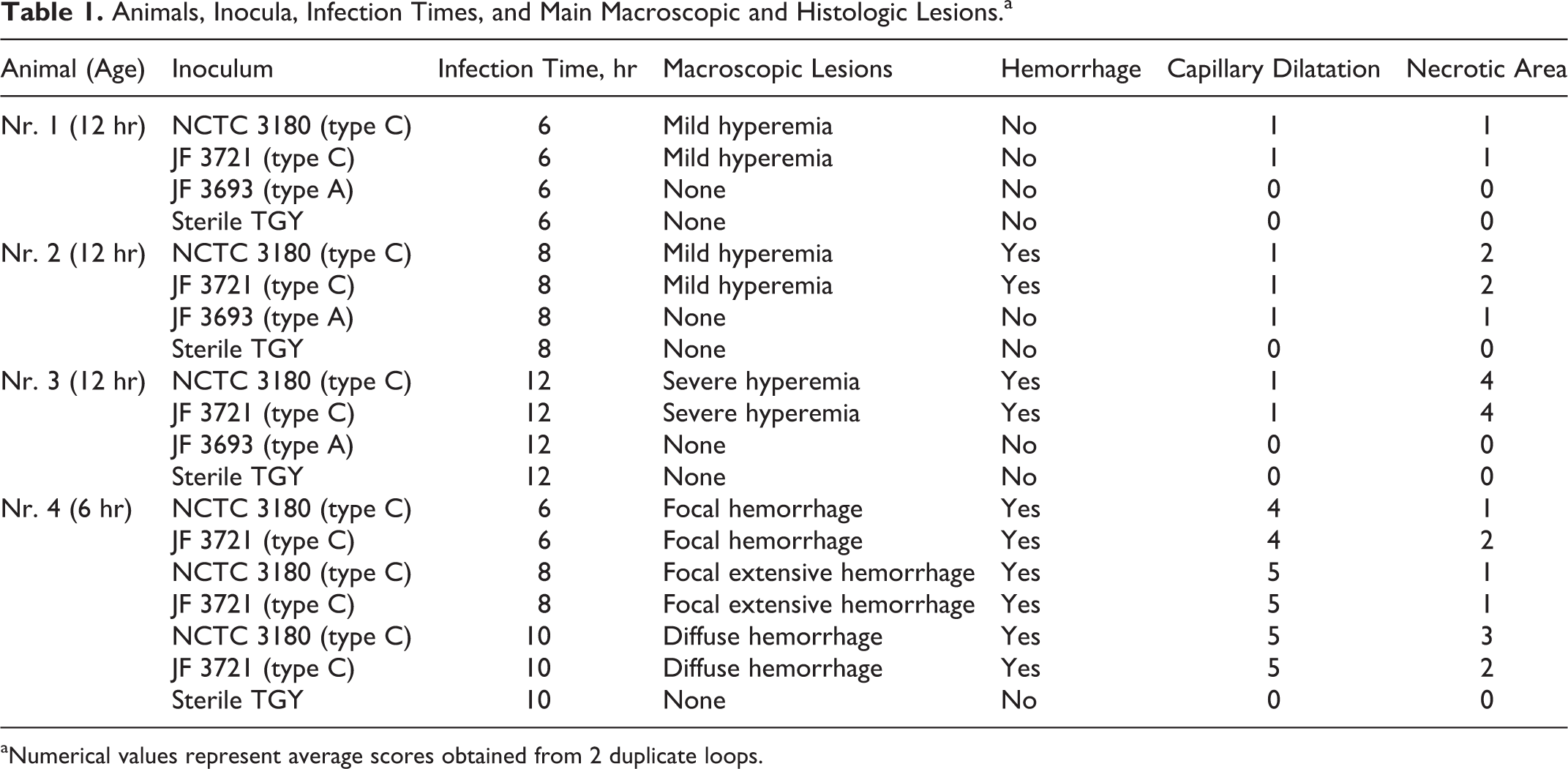
aNumerical values represent average scores obtained from 2 duplicate loops.
Pathological Lesions
In animal Nos. 1–3, macroscopic lesions, including hyperemic and roughened mucosa, only occurred in jejunal loops inoculated with the 2 C. perfringens type C strains (Table 1). Macroscopic lesions in the time course experiment, using a 6-hour-old piglet, were more pronounced, and infection with both C. perfringens type C strains induced progressive hemorrhagic lesions (Table 1). Histologically, mild lesions, evident as mild villous shortening and multifocal villous tip epithelial cell sloughing and necrosis associated with dilatation of underlying capillaries, occurred at 6 and 8 hours postinoculation (pi) with the 2 C. perfringens type C isolates in animal Nos. 1 and 2 (Table 1, Fig. 1). Twelve hours pi, type C strain–infected loops showed moderate villous shortening, multifocal necrosis of villous tip epithelial cells, and capillary dilatation in the affected villous (Fig. 2). Thrombosis was not present in samples from animal Nos. 1–3. In animal No. 4, at 6 hours pi, multifocal villous tip epithelial necrosis and marked capillary dilatation throughout the mucosa, submucosa, and muscularis were evident. This progressed to widespread capillary dilatation and hemorrhage in the lamina propria at 8 hours (Fig. 3) and marked villous shortening, with loss of epithelial cells and massive hemorrhage in the lamina propria and submucosa at 10 hours pi. Few foci of necrosis and thrombosis of small vessels in the lamina propria were visible at the 8- and 10-hour time points. Immunohistochemically, CPB was detected at the cytoplasmic membrane of endothelial cells in the lamina propria of all loops inoculated with C. perfringens type C strains. In the multifocal lesions present in animal Nos. 1–3, CPB signal was present in areas with villous tip necrosis only (Figs. 4, 5). In animal No. 4, CPB signal on endothelial cells increased progressively with the severity of histological lesions. At 6 hours pi, CPB was detected at endothelial cells of affected villi only. At 8 hours pi, CPB staining also extended to submucosal vessels (Fig. 6). At 10 hours pi, there was diffuse staining of vessels throughout the entire sections extending into the submucosa, including areas with an intact appearing epithelium. CPB signal was not detected at epithelial cells in any loop infected with type C strains. Additionally, no CPB signal was detectable in any of the control loops.
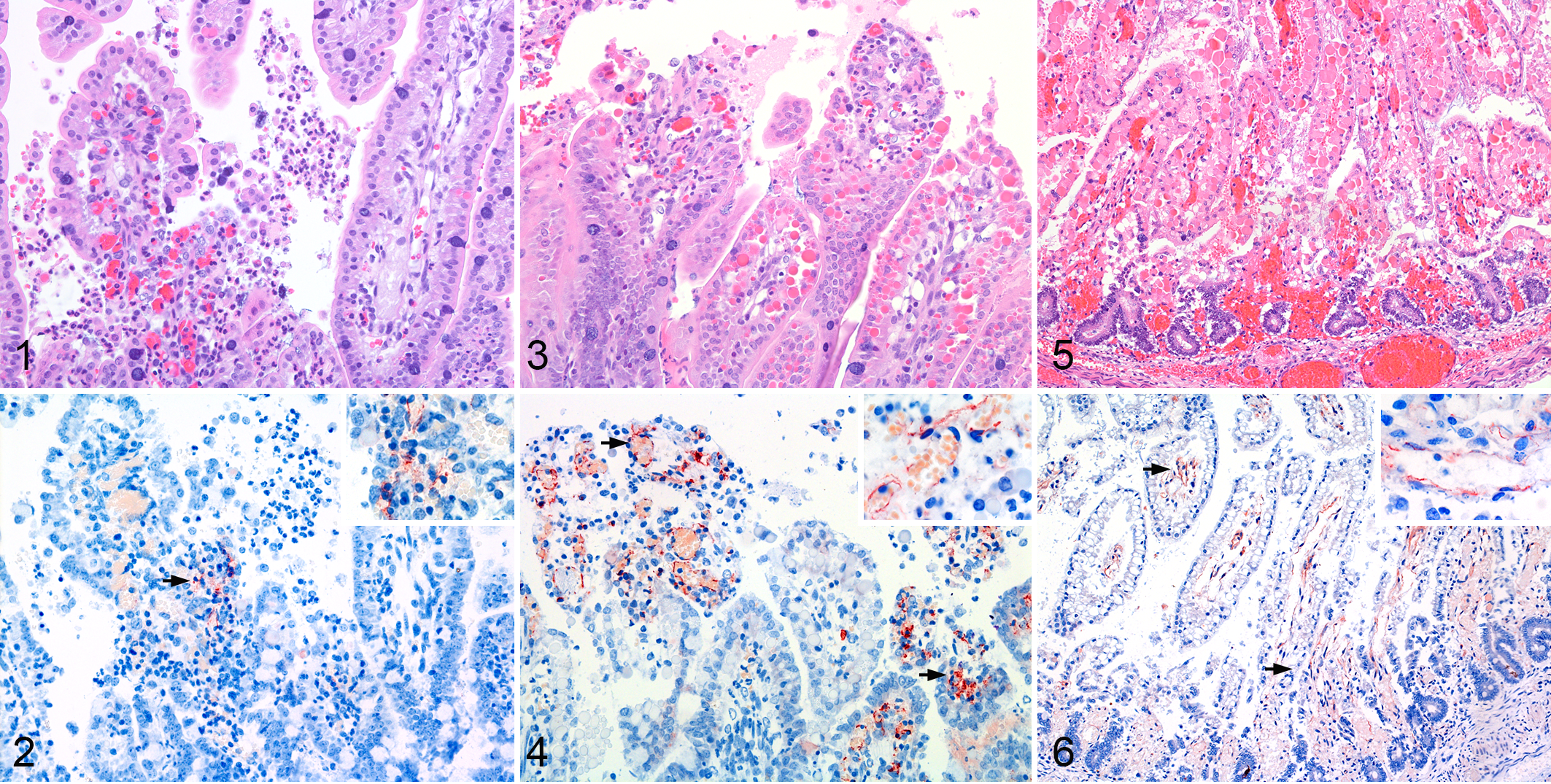
Discussion
In summary, our results demonstrate that the porcine neonatal jejunal loop model is suitable to induce early stage lesions of C. perfringens type C enteritis in a naturally affected host species and thus has potential for further research applications. The essential role of CPB in C. perfringens type C enteritis has recently been demonstrated by a combination of reverse genetics and small laboratory animal infection models. 5,9 These models are well suited to determine essential virulence factors and mechanisms of C. perfringens type C enteritis. However, susceptibility of target cells to toxins might differ among host species, and infectious models based on naturally affected species represent valuable additions to existing models. Albeit based on a limited number of animals, our results indicate that the development of early lesions of C. perfringens type C enteritis was associated with binding of CPB to microvascular endothelial cells in the small intestinal mucosa. In early-stage lesions, CPB signal at endothelial cells was only detectable in affected jejunal villi. In advanced lesions, CPB signal was localized to endothelial cells in all layers of the intestinal loops. These findings suggest increased resorption of CPB from the intestinal lumen and hematogenous distribution of CPB within the intestinal microvasculature. Our results correspond to previous studies, where we demonstrated endothelial localization of CPB in spontaneous lesions of NE in piglets. 3 In contrast to end-stage lesions in naturally affected animals, we did not observe widespread vascular necrosis in this experimental model, suggesting that vascular necrosis and thrombosis most likely occur at later time points. Due to animal welfare reasons, animals were euthanized by 12 hours pi; thus, end point lesions did not develop in our experiments. Further experiments using longer infection times would be necessary to investigate the exact time course of pathological lesions in more detail.
In contrast to an endothelial tropism of CPB, we were unable to demonstrate CPB binding to jejunal epithelial cells. However, epithelial damage was clearly demonstrated in our experiments. Because CPB was able to reach endothelial cells in the lamina propria, an initial epithelial defect leading to tissue penetration of CPB is conceivable. Whether such initial epithelial damage is due to a direct effect of C. perfringens type C on the epithelial cells or a consequence of the endothelial damage cannot be resolved by our limited experiments. Additionally, as CPB could be toxic to epithelial cells at concentrations below the detection level of our immunohistochemistry protocol, our results do not exclude a direct toxic effect on the epithelium. Further in vitro studies to determine the potential toxic effect of CPB on porcine small intestinal epithelial cells are currently being performed in our laboratory.
Footnotes
Acknowledgements
We thank Leen Timbermont, Christian Puttevils, Delphine Ameye, and Pashk Selitaj for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a Ghent University Visiting Foreign Researcher Grant and a Short International Visit Grant from the Swiss National Science Foundation (IZK0Z3_133953). Monoclonal antibodies were kindly provided by G. Shrinivas, Center for Veterinary Biologics, USDA, Ames, Iowa.
