Abstract
Neurologic manifestations other than cerebellar hypoplasia are rarely associated with feline panleukopenia virus (FPV) infection in cats. Here the authors describe lymphoplasmacytic meningoencephalitis and neuronal necrosis in 2 cats autopsied after exhibiting ataxia and nystagmus. Gross changes consisted of cerebellar herniation through the foramen magnum, with flattening of cerebrocortical gyri and narrowing of sulci. Histologically, lymphoplasmacytic meningoencephalitis, extensive neuronal necrosis, and neuroaxonal degeneration with digestion chambers were present in the telencephalon and brain stem in both cats. Frozen brain tissue of both cats was positive for parvoviral antigen via fluorescent antibody testing, and formalin-fixed, paraffin-embedded tissue sections of brain were immunoreactive for parvovirus antigen and positive for parvoviral DNA on in situ hybridization. Frozen brain tissue from 1 case was positive for parvovirus NS1 and VP2 genes using conventional polymerase chain reaction, and subsequent DNA sequencing and phylogenetic analysis revealed that the viral strain was a FPV. Reverse transcription quantitative polymerase chain reaction on formalin-fixed, paraffin-embedded brain tissue revealed high levels of parvovirus in both cases, supporting an acute and active viral infection. Although rare, FPV infection should be considered in cases of lymphoplasmacytic meningoencephalitis and neuronal necrosis in cats.
Although many parvoviruses can infect cats, infection is most commonly associated with feline panleukopenia virus (FPV), a nonenveloped, single-stranded DNA virus of the family Parvoviridae. 10,12,25,26 FPV is closely related to other parvoviruses and infects primarily domesticated cats but also wild felids worldwide. 10 Infection typically takes place in tissues with a high mitotic rate, such as intestine, bone marrow, and lymphoid tissue. 12 Transmission occurs via direct contact with contaminated feces, urine, blood, and placenta or by indirect contact through the environment. 15 The high risk for transmission and the high stability of the virus in the environment create a serious threat of infection in shelters or other environments with high population density or multiple animals from different sources. 15
Clinical disease associated with FPV or other parvoviruses in young or adult cats is usually progressive and fatal, leading to fever, vomiting, and diarrhea, associated with pancytopenia and necrotizing enteritis. 12 Fetal infection by FPV causes a wide range of clinical and pathologic manifestations. Early-term infection most commonly leads to abortion or stillbirths, whereas late-term or neonatal infection is more often associated with cerebellar hypoplasia, hydrocephalus, hydranencephaly, and retinal dysplasia. 1 –3,12,14,23,25 Parvoviral infection of rapidly replicating external germinal layer cells and Purkinje cells leads to cerebellar hypoplasia and ataxia. 1,18,20 Other rare neurologic changes associated with parvoviral infection in cats include demyelination with or without neuronal degeneration 4,8,16,27 and lymphohistiocytic cerebellar meningoencephalitis. 22 Some of these rare manifestations can affect adult cats 8,27 and can occur without the classic cerebellar involvement. 4,8,16,27
Here we characterize the neuropathologic and molecular features of 2 cases of lymphoplasmacytic meningoencephalitis and neuronal necrosis associated with parvoviral infection in cats. Both individuals were part of a group of 3 rescued stray cats that were waiting for adoption and shared the same room in a veterinary clinic. All 3 cats had acute onset of neurologic signs. Case 1 was an 8-week-old female domestic shorthaired cat with a 1-week history of ataxia and nystagmus that was euthanized because of a poor prognosis. Case 2 was a 5-month-old male neutered domestic shorthaired cat with a 24-hour history of acute ataxia that died 1 day after cat 1 was euthanized. A third cat was also ataxic and died 4–5 days before the other 2 cats exhibited clinical signs; no other information was provided about that cat. All individuals had tested negative for feline immunodeficiency virus and feline leukemia virus. Cases 1 and 2 were submitted for autopsy.
Gross anatomic changes in both cats consisted of cerebellar herniation through the foramen magnum, with mild widespread flattening of cerebrocortical gyri and narrowing of sulci (Fig. 1). Representative samples of brain, heart, trachea, lungs, liver, spleen, kidneys, urinary bladder, gastrointestinal tract, lymph nodes, thyroid and parathyroid glands, adrenal glands, and bone marrow were fixed in 10% buffered formalin, routinely processed for histology, and stained with hematoxylin and eosin. For the brain, sections of frontal, parietal, temporal, and occipital telencephalon, basal nuclei, thalamus, hippocampus, mesencephalon, cerebellum, and pons were examined histologically. 28 Neurohistologic changes were similar in both cases and consisted of multiple perivascular accumulations of small to moderate numbers of lymphocytes and plasma cells within the neuroparenchyma and leptomeninges (Fig. 2). Endothelial cells were swollen, and nodular areas of microgliosis were present throughout the neuroparenchyma (Fig. 3). Extensive neuronal necrosis was present within the frontal and parietal cerebral cortex and hippocampus (Fig. 4), as well as the vestibular and abducens nuclei in case 1. Affected neurons had hypereosinophilic and shrunken cytoplasm, with nuclear pyknosis or karyorrhexis. Dilation of the periaxonal spaces, with swollen axonal spheroids or foamy macrophages (Gitter cells, digestion chambers), were present throughout the white matter of the distal brain stem, especially within the facial nerve fibers (Fig. 5). The neuroparenchyma adjacent to the affected areas throughout the brain was vacuolated because of edema. Small intestinal crypt necrosis and mucosal collapse, typical of parvoviral infection, was present in case 2. No other pathologic changes were present in either cat.
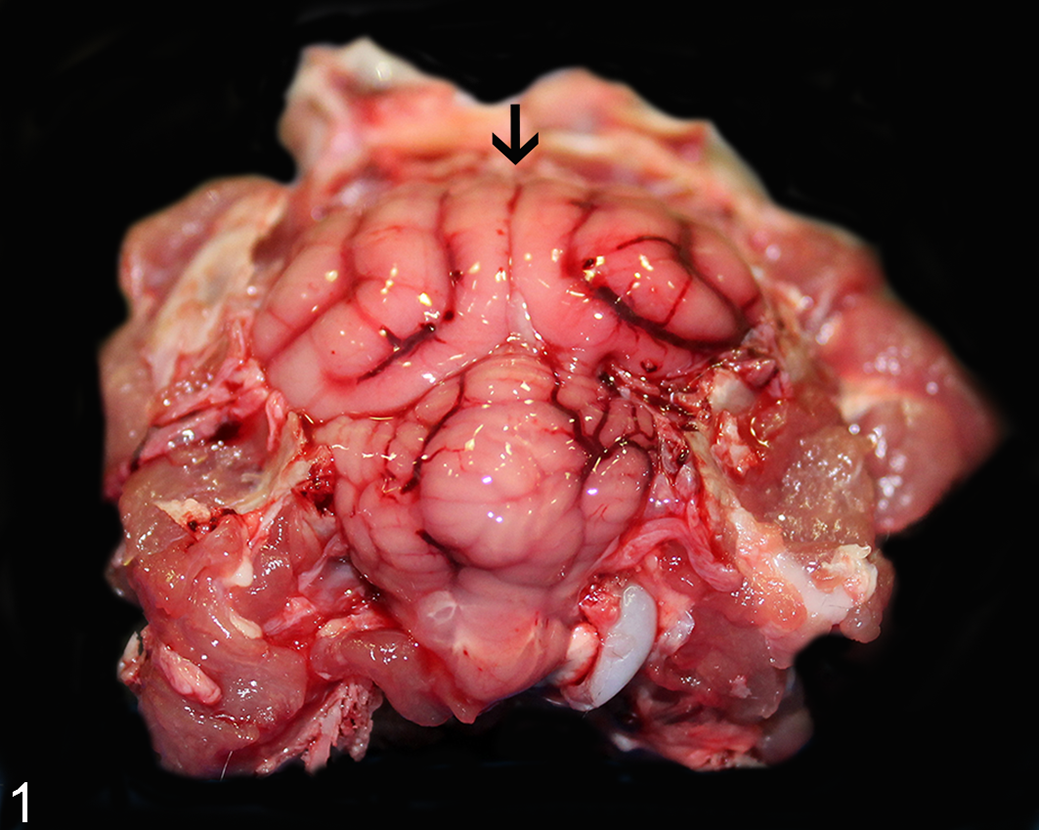
Necrotizing meningoencephalitis, brain, cat, case 1. The cerebellar vermis is herniated through the foramen magnum. There is also flattening of cerebrocortical gyri (arrows) due to cerebral edema.
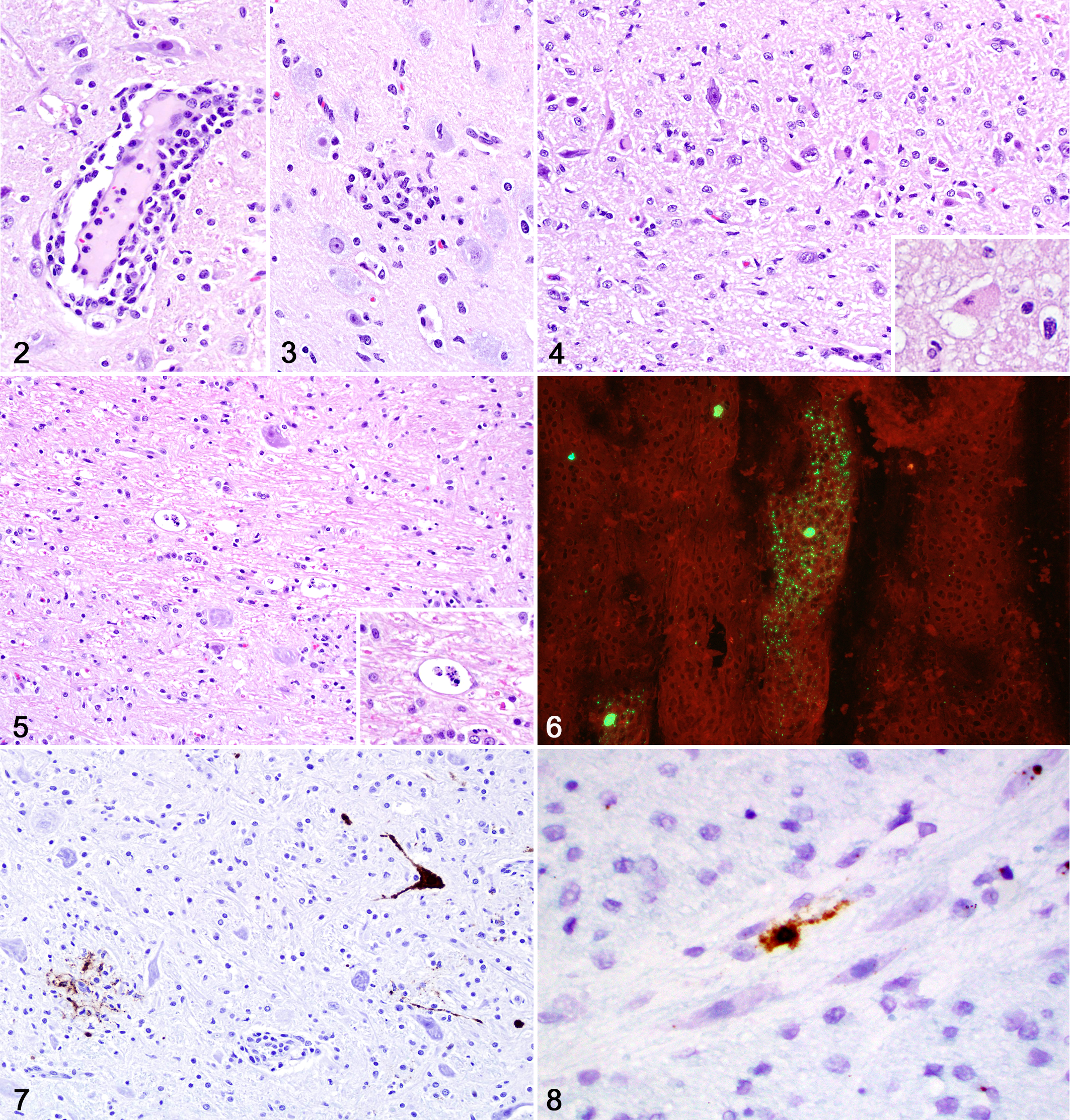
Feline parvovirus–associated meningoencephalitis, brain, cat.
Using fluorescent antibody testing (FAT), frozen brain tissue (cases 1 and 2) was positive for feline parvovirus (Fig. 6) and negative for rabies, feline infectious peritonitis virus, and Toxoplasma gondii. Immunohistochemistry (IHC) for feline parvovirus (mouse monoclonal, 1:500 dilution for 60 minutes; Bio-Rad, Hercules, CA; MCA2064) was conducted using formalin-fixed tissue sections of brain and small intestine (cases 1 and 2). Control tissues included canine small intestine with confirmed parvoviral enteritis. Widespread, robust immunolabeling was detected mainly in cerebrocortical neurons, Purkinje neurons, and Golgi neurons and less commonly in microglial nodules and endothelial cells throughout the brain in both cases (Fig. 7). Immunolabeling was also present in the cerebellar external granular layer neurons in case 1. Immunolabeling was also detected within necrotic areas in the intestine in case 2; no immunolabeling was present in the intestine in case 1. In situ hybridization (ISH) was performed on formalin-fixed sections of brain stem (cases 1 and 2) according to previously published work. 7,13 A strong parvoviral ISH signal was localized within the cytoplasm and nuclei of necrotic and viable neurons across the brain (Fig. 8).
A conventional polymerase chain reaction (PCR) was performed on frozen brain tissue from case 2. Viral DNA was extracted using a commercial kit (QIAmp cador Pathogen Kit, Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Using published primers, 5,6 a 583-bp fragment of the capsid protein 2 gene (VP2) and a 2007-bp fragment of the nonstructural protein 1 gene (NS1) were amplified. A previously sequenced field strain of canine parvovirus (CPV) was used as a positive control, as reported elsewhere. 9 PCR products from both VP2 and NS1 genes were purified and submitted for Sanger DNA sequencing, as previously described. 24 Basic Local Alignment Search Tool analysis (http://www.ncbi.nlm.nih.gov/BLAST) was used on each sequence to identify related viruses. Phylogenetic trees were built using the NS1 and VP2 sequences from this study and 46 VP2 gene nucleotide sequences and 31 NS1 gene sequences retrieved from GenBank. The retrieved sequences were representative strains from FPV, mink enteritis virus, and CPV from different parts of the world. VP2 and NS1 sequences from this study are available in the public database under accession numbers MH581488 and MH577049, respectively. Sequence alignments were performed using ClustalW software, and phylogenetic trees were inferred using the neighbor-joining method 21 available in Molecular Evolutionary Genetics Analysis version 7 (http://www.megasoftware.net). 11 Evolutionary distances were computed using the maximum composite likelihood method. The percentage of replicate trees in which the associated taxa clustered together using bootstrapping (1000 iterations) is shown next to the branches. The amplified sequences were 99% homologous to NS1 and VP2 gene sequences from FPV, CPV, and mink enteritis virus available in GenBank. Phylogenetic analysis of NS1 placed this sequence in the FPV clade (Suppl. Fig. S1). Demarcation criteria from the International Committee on Taxonomy of Viruses for the parvovirus genus are based only on the NS gene; therefore, the strain investigated in this study was classified as FPV. The VP2 gene phylogeny (Suppl. Fig. S2) had a closer association with mink enteritis virus, followed by FPV, which might be the consequence of recombination events and might have an impact on the unusual tropism and virulence of the strain.
For reverse transcription quantitative PCR (RT-qPCR), nucleic acid was extracted from two 10-μm sections of formalin-fixed, paraffin-embedded brain tissue (cases 1 and 2) sectioned into sterile Eppendorf tubes. Samples were deparafinized in lysis buffer for 60 minutes at 70°C and then incubated overnight at 60°C with proteinase K. After incubation the tubes were briefly centrifuged and allowed to cool to room temperature; 200 μL of lysate from each tube was transferred to a 96-well plate for extraction using a Biomek 4000 automated workstation (Beckman Coulter, Indianapolis, Indiana) Total Nucleic Acid preset program and Agencourt Formapure Kit (Beckman Coulter) according to the manufacturer’s instructions. RT-qPCR was performed on these samples using primers and probe to amplify the VP2 gene, according to previously published work. 7 Supporting the conventional PCR and sequencing results, high levels of parvovirus were amplified by RT-qPCR in both cases (1.35 × 105 and 1.64 × 105 copies/200 μg of nucleic acid in cases 1 and 2, respectively).
The diagnosis of parvoviral meningoencephalitis and neuronal necrosis in the present cases was achieved on the basis of the FAT, IHC, ISH, and PCR results. Subsequent DNA sequencing and phylogenetic analysis revealed that the NS1 gene sequence best matched the FPV clade, indicating that the parvovirus in these cats was closely related with FPV strains detected in cats from Italy and Japan; the estimated confidence levels of the VP2 gene sequence further support this hypothesis. An acute and active viral infection and a causal relationship were supported by the results of the RT-qPCR. 7
Gross neuropathologic changes associated with parvoviral infection in cats have been limited to cerebellar hypoplasia, hydrocephalus, and hydranencephaly. 1,12,25 Evidence of cerebral edema, such as cerebellar herniation with flattening of cerebrocortical gyri and narrowing of sulci, have not been described in cases of parvoviral infection in cats and were present in both of the present cases. 1,8,12,16,18,20,25,27 The neurohistologic changes in the present cases were also distinct from those previously attributed to parvoviral infection in cats, which include cerebellar hypoplasia, depletion of the granular layer, Purkinje cell heterotopia and vacuolation, Purkinje cell dendrite disarray, and a reduction of myelinated fibers. 4,8,16,20,22,27 No gross or histologic evidence of cerebellar hypoplasia was present in these cases.
Other central nervous system manifestations of parvoviral infection in cats are rare and consist mainly of neurodegenerative changes with minimal or no accompanying inflammation. 4,8,16,22,27 FPV infection was suspected in cases of bilateral and symmetric demyelination of the brain stem and spinal cord, as well as neuronal degeneration in the gray matter of the spinal cord in the 1970s, but infection could not be confirmed, and the role of FPV in the development of those changes remains elusive. 4 Mild cerebellar lymphohistiocytic meningoencephalitis was reported in a cat with parvoviral infection in which immunolabeling was detected in neurons, macrophages and microglia, and ependymal cells; no further viral characterization was conducted. 22 Focal neuronal satellitosis and neuronophagia were described in a subset of cats that died of necrotizing enteritis due to FPV infection; immunolabeling was detected in neurons of the interthalamic adhesion and glial cells. 8 Neuronal vacuolation with positive immunolabeling for FPV was described in the thoracic spinal cord of a cat that also had necrotizing enteritis, similar to one of the present cases. 16 The presence of tissue FPV was confirmed via PCR and sequencing of viral DNA in only 2 of these previously reported occasions. 8,16 In addition, vacuolation of the neuropil and neuronal degeneration in the lateral geniculate nuclei, cerebral cortex, hippocampus, and pons was associated with CPV-2 in a group of cats; immunolabeling was detected mainly in neurons and less often glial and endothelial cells, and the diagnosis was supported via PCR and DNA sequencing. 27
The successful replication of parvovirus is highly dependent on the host cell’s achieving the S phase of its cycle, which makes tissues with high a mitotic rate a preferred target for infection in young and adult individuals. 17 Intrauterine or neonatal central nervous system infection occurs mainly within dividing neuroblasts of the cerebellar external granular layer. 1,10,12,17,25 Purkinje neurons have been shown to be postmitotic after 40 days of gestation, 19 but parvoviral infection has been demonstrated within these cells during late pregnancy or neonatal life, 1,2,8,18 –20,27 which suggests that parvoviruses are able to replicate in terminally differentiated neurons. 25 In the present cases, viral antigen was detected within neurons, glial cells, and endothelial cells using IHC, whereas viral nucleic acid was present only within neurons. These results differ from a previous study using the same ISH technique for detection of CPV-2 in the hearts of dogs, which revealed a more widespread parvoviral signal with ISH. 7 The reasons for these discrepancies are elusive and may be related to technical differences in fixation, IHC protocols, or antibodies (mouse monoclonal vs rabbit polyclonal) 7 or to differences in the dynamics of viral replication, with active replication within neurons and latent replication within other cells. 16
In summary, we describe previously undocumented neuropathologic changes associated with parvoviral infection in 2 cats. Viral antigen was detected within neurons and endothelial cells via FAT and IHC, and parvoviral nucleic acid was detected within neurons via ISH. Phylogenetic analysis of the NS1 gene classified the strain as a FPV. Abundant viral nucleic acid (by RT-qPCR), suggesting an acute and active viral infection, was associated with these neuropathologic changes. Although uncommon, parvoviral infection should be considered in cases of lymphoplasmacytic meningoencephalitis and neuronal necrosis in cats.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819837723 - Lymphoplasmacytic Meningoencephalitis and Neuronal Necrosis Associated With Parvoviral Infection in Cats
Supplemental Material, DS1_VET_10.1177_0300985819837723 for Lymphoplasmacytic Meningoencephalitis and Neuronal Necrosis Associated With Parvoviral Infection in Cats by Anna Kokosinska, Grazieli Maboni, Kathleen M. Kelly, Alex Molesan, Susan Sanchez, Jeremiah T. Saliki and Daniel R. Rissi in Veterinary Pathology
Footnotes
Acknowledgements
We thank Jillian Fishburn (Athens Veterinary Diagnostic Laboratory, University of Georgia College of Veterinary Medicine) for support with the FAT and Dr Randall Renshaw (Department of Population Medicine and Diagnostic Services, Cornell University College of Veterinary Medicine, Ithaca, NY) for the support with RT-qPCR.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
